S-4/A: Registration of securities issued in business combination transactions
Published on August 11, 2021
Table of Contents
As filed with the Securities and Exchange Commission on August 11, 2021
Registration No. 333-257535
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
AMENDMENT NO. 1
TO
FORM S-4
REGISTRATION STATEMENT
UNDER
THE SECURITIES ACT OF 1933
GS ACQUISITION HOLDINGS CORP II
(Exact name of registrant as specified in its charter)
| Delaware (State or other jurisdiction of incorporation or organization) |
6770 (Primary Standard Industrial Classification Code Number) |
83-0974996 (I.R.S. Employer Identification Number) |
200 West Street
New York, NY, 10282
Telephone: (212) 902-1000
(Address, including zip code, and telephone number, including area code, of registrants principal executive offices)
Thomas R. Knott
Chief Executive Officer
GS Acquisition Holdings Corp II
200 West Street
New York, NY, 10282
Telephone: (212) 902-1000
(Name, address, including zip code, and telephone number, including area code, of agent for service)
Copies to:
| Michael J. Aiello Brian Parness Weil, Gotshal & Manges LLP 767 Fifth Avenue New York, NY 10153 (212) 310-8000 |
Thomas D. Logan, CEO Mirion Technologies, Inc. 1218 Menlo Drive Atlanta, GA 30318 (470) 870-2700 |
Alan F. Denenberg Stephen Salmon Bryan M. Quinn Davis Polk & Wardwell LLP 1600 El Camino Real Menlo Park, CA 94025 (650) 752-2000 |
Valerie Ford Jacob Freshfields Bruckhaus Deringer LLP 601 Lexington Avenue New York, NY 10022 (212) 277 4000 |
Approximate date of commencement of proposed sale of the securities to the public: As soon as practicable after this registration statement becomes effective and upon completion of the transactions.
If the securities being registered on this Form are being offered in connection with the formation of a holding company and there is compliance with General Instruction G, check the following box: ☐
If this Form is filed to register additional securities for an offering pursuant to Rule 462(b) under the Securities Act, check the following box and list the Securities Act registration statement number of the earlier effective registration statement for the same offering. ☐
If this Form is a post-effective amendment filed pursuant to Rule 462(d) under the Securities Act, check the following box and list the Securities Act registration statement number of the earlier effective registration statement for the same offering. ☐
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, a smaller reporting company, or emerging growth company. See the definitions of large accelerated filer, accelerated filer, smaller reporting company, and emerging growth company in Rule 12b-2 of the Exchange Act. (Check one):
| Large accelerated filer | ☐ | Accelerated filer | ☐ | |||
| Non-accelerated filer | ☒ | Smaller reporting company | ☐ | |||
| Emerging growth company | ☒ | |||||
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 7(a)(2)(B) of the Securities Act. ☐
If applicable, place an ☒ in the box to designate the appropriate rule provision relied upon in conducting this transaction:
Exchange Act Rule 13e-4(i) (Cross-Border Issuer Tender Offer) ☐
Exchange Act Rule 14d-1(d) (Cross-Border Third-Party Tender Offer) ☐
CALCULATION OF REGISTRATION FEE
|
|
||||||||
| Title of each class of securities to be registered |
Amount to be registered(1)(2) |
Proposed maximum offering price per unit |
Proposed maximum aggregate offering price(3) |
Amount of registration fee(4) |
||||
| GSAH Class A Common Stock, $0.0001 par value per share |
24,878,039(2) | $10.00 | $248,780,390.00 | $27,141.94(5) | ||||
|
|
||||||||
|
|
||||||||
| (1) | Based on the maximum number of shares of Class A common stock, par value $0.0001 per share, of the registrant estimated to be issuable in connection with the Business Combination described herein, excluding (1) the portion of such shares issuable to certain funds affiliated with Charterhouse Capital Partners LLP and (2) any shares of Class B common stock, par value $0.0001 per share, issuable to certain members of management of Mirion Technologies (TopCo), Ltd. |
| (2) | Pursuant to Rule 416(a) of the Securities Act, there are also being registered an indeterminable number of additional securities as may be issued to prevent dilution resulting from stock splits, stock dividends or similar transactions. |
| (3) | Computed in accordance with Rule 457 of the Securities Act. |
| (4) | Calculated pursuant to Rule 457 of the Securities Act by calculating the product of (i) the proposed maximum aggregate offering price and (ii) 0.0001091. |
| (5) | The registrant previously paid the registration fee in connection with a prior filing of this Registration Statement. |
The registrant hereby amends this registration statement on such date or dates as may be necessary to delay its effective date until the registrant shall file a further amendment which specifically states that this registration statement shall thereafter become effective in accordance with Section 8(a) of the Securities Act of 1933 or until the registration statement shall become effective on such date as the Securities and Exchange Commission, acting pursuant to said Section 8(a), may determine.
Table of Contents
EXPLANATORY NOTE
This proxy statement/prospectus relates to a Business Combination Agreement, dated June 17, 2021 (as it may be amended from time to time, the Business Combination Agreement), by and among GS Acquisition Holdings Corp II, a Delaware corporation (the Company or GSAH), Mirion Technologies (TopCo), Ltd., a Jersey private company limited by shares (Mirion), for the limited purpose set forth therein, CCP IX LP No. 1, CCP IX LP No. 2, CCP IX Co-Investment LP and CCP IX Co-Investment No. 2 LP (collectively, the Charterhouse Parties), for the limited purpose set forth therein, each of the other persons set forth on Annex I thereto (together with the Charterhouse Parties, the Supporting Mirion Holders), and, for the limited purpose set forth therein, the other holders of A Ordinary Shares and B Ordinary Shares from time to time becoming a party thereto by executing a Joinder Agreement, a copy of which is attached to this proxy statement/prospectus as Annex A (each, a Joining Seller and collectively, the Joining Sellers and, together with each Supporting Mirion Holder, each, a Seller and, collectively, the Sellers).
Pursuant to the Business Combination Agreement, among other things and subject to the terms and conditions contained therein, the parties thereto will enter into a business combination transaction, pursuant to which Mirion will combine with a subsidiary of the Company as described below (the Business Combination, and the transactions contemplated by the Business Combination Agreement, the Transactions).
The proposed Business Combination will establish the Company as the corporate parent of Mirion. At the Closing, in order to implement a structure similar to that of an Up-C, the Company will establish a Delaware corporation (IntermediateCo) as a subsidiary of the Company. Sellers will be permitted to elect to receive equity consideration either in the form of (i) shares of GSAH Class A common stock, par value $0.0001 per share (GSAH Class A common stock) or (ii) paired interests consisting of shares of GSAH Class B common stock, par value $0.0001 per share (GSAH Class B common stock) paired together with shares of IntermediateCo Class B common stock, par value $0.0001 per share (IntermediateCo Class B common stock). Holders of shares of IntermediateCo Class B common stock will have a redemption right for such shares to be settled, at the option of the Company, with (i) shares of GSAH Class A common stock (on a one-for-one basis) or (ii) a cash amount per share based on an average trailing stock price of GSAH Class A common stock. In the event of a redemption request by a holder of shares of IntermediateCo Class B common stock, the Company may, at its option, effect a direct exchange of cash or Company Class A common stock for shares of IntermediateCo Class B common stock in lieu of such a redemption. Upon redemption or exchange of shares of IntermediateCo Class B common stock, the corresponding shares of GSAH Class B common stock will be cancelled. The Company will hold 100% of the voting shares of IntermediateCo Class A common stock, par value $0.0001 per share (IntermediateCo Class A common stock). As further described in the Second Amended and Restated Certificate of Incorporation to be in effect after the Closing, each holder of the Companys Class B common stock will be entitled to one vote for each share of GSAH Class B common stock held of record by such holder on all matters on which stockholders generally are entitled to vote and the holders of GSAH Class B common stock shall not be entitled to dividends of cash or property on such shares of GSAH Class B common stock.
Subject to the terms of the Business Combination Agreement, the consideration to be paid in connection with the Business Combination is $1,700,000,000 and will be paid in a combination of equity and cash consideration. The cash consideration will be an amount equal to $1,310,000,000; provided, that if the Minimum Cash Condition is not met, and Mirion and the Charterhouse Parties elect to waive the Minimum Cash Condition, then the Cash Consideration will be equal to $1,310,000,000 less the amount by which $1,310,000,000 exceeds the Available Closing Cash. In exchange for the A Ordinary Shares of $0.01 each in the capital of Mirion, the B Ordinary Shares of $0.01 each in the capital of Mirion and certain loan notes due 2026 issued by Mirion Technologies (HoldingSub1), Ltd, each Seller may elect to receive cash or equity consideration or a combination thereof, which equity consideration shall be in the form of either shares of GSAH Class A common stock or shares of GSAH Class B common stock combined with shares of IntermediateCo Class B common stock that will be majority owned by the Company.
i
Table of Contents
The Available Closing Cash will be an amount equal to (i) the amount of funds contained in the Companys trust account (after reduction for the aggregate amount of payments required to be made in connection with any valid stockholder redemptions), plus (ii) the aggregate amount of cash that has been funded to and remains with the Company pursuant to the Subscription Agreements as of immediately prior to the closing of the Business Combination (the Closing), plus (iii) the amounts delivered pursuant to the Mirion Debt Refinancing (as defined in the Business Combination Agreement), plus (iv) the cash and cash equivalents of Mirion and its subsidiaries on a consolidated basis as of the date of the Closing (the Closing Date), plus (v) the proceeds, if any, from the sale by the Company to GSAM Holdings LLC (GSAM Holdings) of shares of GSAH Class A common stock, pursuant to the Backstop Agreement, less (vi) the total amount required to be paid to fully satisfy all obligations related to Mirions credit agreement as of the Closing Date, less (vii) certain transaction expenses, less (viii) $50,000,000 (collectively, the Available Closing Cash).
Concurrently with the execution of the Business Combination Agreement, the Company entered into subscription agreements (the Subscription Agreements) with certain investors (collectively, the PIPE Investors), pursuant to, and on the terms and subject to the conditions of which, the PIPE Investors have collectively subscribed for 90,000,000 shares of GSAH Class A common stock for an aggregate purchase price equal to $900,000,000 (the PIPE Investment and, such shares, the PIPE Shares). The PIPE Investment will be consummated substantially concurrently with the Closing.
This proxy statement/prospectus serves as:
| | a proxy statement/prospectus for the special meeting of the Company, where the GSAH stockholders will vote on, among other things, proposals to (i) approve the Business Combination Agreement and the transactions contemplated thereby, including the Business Combination, (ii) approve the issuance of GSAH Common Stock in connection with (x) the Transactions (including as may be required under the NYSE) and (y) the PIPE Investment; (iii) adopt the Companys second amended and restated certificate of incorporation, (iv) approve, on a non-binding basis, certain governance provisions in the Companys second amended and restated certificate of incorporation; (v) approve the Mirion Technologies, Inc. Omnibus Incentive Plan; (vi) elect the directors constituting the board of directors of the post-combination company; and (vii) approve the adjournment of the Special Meeting, if necessary, to permit further solicitation of proxies because there are not sufficient votes to approve and adopt any of the foregoing; and |
| | a prospectus for the shares of GSAH Class A common stock (other than to certain funds affiliated with Charterhouse Capital Partners LLP and GSAH Class B common stock that certain of Mirions shareholders will receive as consideration in the Transactions). |
ii
Table of Contents
PRELIMINARY SUBJECT TO COMPLETION, DATED AUGUST 11, 2021
GS ACQUISITION HOLDINGS CORP II
200 West Street
New York, NY 10282
Dear GS Acquisition Holdings Corp II Stockholder:
We cordially invite you to attend a special meeting (the Special Meeting) of the stockholders of GS Acquisition Holdings Corp II, a Delaware corporation (we, us, our, GSAH or the Company), which will be held via live webcast on [●] at [●] a.m. [Eastern Time] or at such other time, on such other date and at such other place to which the meeting may be adjourned or postponed. The special meeting can be accessed by visiting [●], where you will be able to listen to the meeting live and vote during the meeting.
On June 17, 2021, the Company entered into a business combination agreement (the Business Combination Agreement) with Mirion Technologies (TopCo), Ltd., a Jersey private company limited by shares (Mirion), for the limited purpose set forth therein, CCP IX LP No. 1, CCP IX LP No. 2, CCP IX Co-Investment LP and CCP IX Co-Investment No. 2 LP (collectively, the Charterhouse Parties), for the limited purpose set forth therein, each of the other persons set forth on Annex I thereto (together with the Charterhouse Parties, the Supporting Mirion Holders), and, for the limited purpose set forth therein, the other holders of A Ordinary Shares and B Ordinary Shares from time to time becoming a party thereto by executing a Joinder Agreement (each, a Seller and, collectively, the Sellers). A copy of the Business Combination Agreement is attached to this proxy statement/prospectus as Annex A (each, a Joining Seller and collectively, the Joining Sellers and, together with each Supporting Mirion Holder, each a Seller and, collectively, the Sellers).
Pursuant to the Business Combination Agreement, among other things and subject to the terms and conditions contained therein, the parties thereto will enter into a business combination transaction, pursuant to which Mirion will combine with a subsidiary of the Company as described below (the Business Combination, and the transactions contemplated by the Business Combination Agreement, the Transactions).
The proposed Business Combination will establish the Company as the corporate parent of Mirion. At the Closing, in order to implement a structure similar to that of an Up-C, the Company will establish a Delaware corporation (IntermediateCo) as a subsidiary of the Company. Sellers will be permitted to elect to receive equity consideration either in the form of (i) shares of GSAH Class A common stock, par value $0.0001 per share (GSAH Class A common stock) or (ii) paired interests consisting of shares of GSAH Class B common stock paired together with shares of IntermediateCo Class B common stock, par value $0.0001 per share (IntermediateCo Class B common stock). Holders of shares of IntermediateCo Class B common stock will have a redemption right for such shares to be settled, at the option of the Company, with (i) shares of GSAH Class A common stock (on a one-for-one basis) or (ii) a cash amount per share based on an average trailing stock price of GSAH Class A common stock. In the event of a redemption request by a holder of shares of IntermediateCo Class B common stock, the Company may, at its option, effect a direct exchange of cash or Company Class A common stock for shares of IntermediateCo Class B common stock in lieu of such a redemption. Upon redemption or exchange of shares of IntermediateCo Class B common stock, the corresponding shares of GSAH Class B common stock will be cancelled. The Company will hold 100% of the voting shares of IntermediateCo Class A common stock, par value $0.0001 per share (IntermediateCo Class A common stock). As further described in the Companys Amended and Restated Certificate of Incorporation to be in effect after the Closing, each holder of the GSAH Class B common stock will be entitled to one vote for each share of GSAH Class B common stock held of record by such holder on all matters on which stockholders generally are entitled to vote and the holders of GSAH Class B common stock shall not be entitled to dividends of cash or property on such shares of GSAH Class B common stock.
In accordance with the terms and subject to the conditions of the Business Combination Agreement, the consideration to be paid in connection with the Business Combination is $1,700,000,000 and will be paid in a
Table of Contents
combination of equity and cash consideration. The cash consideration will be an amount equal to $1,310,000,000; provided, that if the Minimum Cash Condition is not met, and Mirion and the Charterhouse Parties elect to waive the Minimum Cash Condition, then the Cash Consideration will be equal to $1,310,000,000 less the amount by which $1,310,000,000 exceeds the Available Closing Cash. In exchange for the A Ordinary Shares of $0.01 each in the capital of Mirion, the B Ordinary Shares of $0.01 each in the capital of Mirion and certain loan notes due 2026 issued by Mirion Technologies (HoldingSub1), Ltd, each Seller may elect to receive cash or equity consideration or a combination thereof, which equity consideration shall be in the form of either shares of GSAH Class A common stock or shares of GSAH Class B common stock combined with shares of IntermediateCo Class B common stock that will be majority owned by the Company.
The Available Closing Cash will be an amount equal to (i) the amount of funds contained in the Companys trust account (after reduction for the aggregate amount of payments required to be made in connection with any valid stockholder redemptions), plus (ii) the aggregate amount of cash that has been funded to and remains with the Company pursuant to the Subscription Agreements as of immediately prior to the closing of the Business Combination (the Closing), plus (iii) the amounts delivered pursuant to the Mirion Debt Refinancing (as defined in the Agreement), plus (iv) the cash and cash equivalents of Mirion and its subsidiaries on a consolidated basis as of the date of the Closing (the Closing Date), plus (v) the proceeds, if any, from the sale by the Company to GSAM Holdings LLC (GSAM Holdings) of shares of GSAH Class A common stock, pursuant to the Backstop Agreement, less (vi) the total amount required to be paid to fully satisfy all obligations related to Mirions credit agreement as of the Closing Date, less (vii) certain transaction expenses, less (viii) $50,000,000 (collectively, the Available Closing Cash).
Concurrently with the execution of the Business Combination Agreement, the Company entered into subscription agreements (the Subscription Agreements) with certain investors (collectively, the PIPE Investors), pursuant to, and on the terms and subject to the conditions of which, the PIPE Investors have collectively subscribed for 90,000,000 shares of GSAH Class A common stock for an aggregate purchase price equal to $900,000,000 (the PIPE Investment and, such shares, the PIPE Shares). The PIPE Investment will be consummated substantially concurrently with the Closing.
In connection with the Business Combination, certain other related agreements have been, or will be entered into on or prior to the date the Business Combination is consummated including the Amended and Restated Registration Rights Agreement, the Subscription Agreements, the Backstop Agreement, the Option Agreements and the Amended and Restated Sponsor Agreement, in each case, as defined in the accompanying proxy statement/prospectus. For additional information, see Proposal No. 1Approval of the Business CombinationRelated Agreements in the accompanying proxy statement/prospectus.
At the Special Meeting, our stockholders will be asked to consider and vote upon: among other things, proposals to (i) approve the Business Combination Agreement and the transactions contemplated thereby, including the Business Combination (the Business Combination Proposal), (ii) approve the issuance of GSAH Common Stock in connection with (x) the Transactions (including as may be required under the NYSE) and (y) the PIPE Investment; (iii) adopt the Companys second amended and restated certificate of incorporation (the Charter Proposal), (iii) approve, on a non-binding basis, certain governance provisions in the Companys second amended and restated certificate of incorporation (the Governance Proposal), (vi) approve the Mirion Technologies, Inc. Omnibus Incentive Plan; (vi) elect nine directors constituting the board of directors of the post-combination company; and (vii) adjourn the Special Meeting, if necessary, to permit further solicitation of proxies because there are not sufficient votes to approve and adopt any of the foregoing (the Adjournment Proposal). The Transactions contemplated by this Business Combination Agreement will be consummated only if the Business Combination Proposal, the NYSE Proposal, the Charter Proposal and the Director Election Proposal (collectively, the Condition Precedent Proposals) are approved at the Special Meeting. Each of the Condition Precedent Proposals is cross-conditioned on the approval of each other. Each of these proposals is more fully described in the accompanying proxy statement/prospectus, which each stockholder is encouraged to read carefully and in its entirety.
ii
Table of Contents
Our publicly-traded GSAH Class A common stock, units and warrants are currently listed on the NYSE under the symbols GSAH, GSAH.U and GSAH WS, respectively. We intend to apply to continue the listing of GSAH Class A common stock and warrants on the NYSE under the symbols MIR and MIR.WS, respectively, upon the closing of the Business Combination, though such securities may not be listed, for instance if there is not a sufficient number of round lot holders. At the closing of the Business Combination, each unit will separate into its components consisting of one share of GSAH Class A common stock and one-fourth of one warrant. GSAH is currently awaiting preliminary listing approval from NYSE in order to submit its listing application and believes the combined entity will satisfy all criteria for listing upon completion of the Business Combination. As such, GSAH expects to obtain NYSE listing approval before the Closing; notwithstanding, GSAH can provide no assurances that NYSE will approve the listing application. NYSEs determination may not be known at the time stockholders are asked to vote on the Business Combination and the closing is not conditioned on NYSEs approval of the continued listing, but the closing of the PIPE Investment is conditioned on the PIPE Shares being listed on the NYSE.
Pursuant to our Amended and Restated Certificate of Incorporation (the GSAH Certificate of Incorporation), a holder of our public shares (a public stockholder) may request that we redeem all or a portion of such stockholders public shares for cash if the Business Combination is consummated. Holders of units must elect to separate the units into the underlying public shares and warrants prior to exercising redemption rights with respect to the public shares. If holders hold their units through a broker, bank or other nominee, holders must notify their broker, bank or other nominee that they elect to separate the units into the underlying public shares and warrants, or if a holder holds units registered in its own name, the holder must contact Continental Stock Transfer & Trust Company, N.A., our transfer agent, directly and instruct it to do so. Public stockholders may elect to redeem their public shares even if they vote for the Business Combination Proposal or any other proposal. If the Business Combination is not consummated, the public shares will be returned to the respective holder, broker, bank or other nominee. If the Business Combination is consummated, and if a public stockholder properly exercises its right to redeem all or a portion of the public shares that it holds, including by timely delivering its shares to our transfer agent, we will redeem such public shares for a per-share price, payable in cash, equal to the pro rata portion of the trust account established at the consummation of our initial public offering (the trust account), calculated as of two business days prior to the consummation of the Business Combination, including interest (net of taxes payable). For illustrative purposes, as of [●], 2021, this would have amounted to approximately $[●] per outstanding public share. If a public stockholder properly exercises its redemption rights in full, then it will be electing to exchange all of its public shares for cash and will not own any public shares of the post-business combination company. See Special Meeting of GSAH StockholdersRedemption Rights in the accompanying proxy statement/prospectus for a detailed description of the procedures to be followed if you wish to redeem your public shares for cash. Holders of our outstanding warrants do not have redemption rights in connection with the Business Combination. Unless otherwise specified, the information in the accompanying proxy statement/prospectus assumes that none of our public stockholders exercise their redemption rights with respect to their shares of GSAH Class A common stock.
Notwithstanding the foregoing, a public stockholder, together with any affiliate of such public stockholder or any other person with whom such public stockholder is acting in concert or as a group (as defined in Section 13(d)(3) of the Securities Exchange Act of 1934, as amended (the Exchange Act)), will be restricted from redeeming its public shares with respect to more than an aggregate of 15% of the public shares. Accordingly, if a public stockholder, alone or acting in concert or as a group, seeks to redeem more than 15% of the public shares, then any such shares in excess of that 15% limit would not be redeemed for cash. In addition, pursuant to the GSAH Certificate of Incorporation, in no event will we redeem public shares in an amount that would cause our net tangible assets (as determined in accordance with Rule 3a51-1(g)(1) of the Exchange Act) to be less than $5,000,001. In such case, we would not proceed with the redemption of our public shares and the Business Combination, and instead may search for an alternate initial business combination.
The Business Combination Agreement is also subject to the satisfaction or waiver of certain closing conditions as described in the accompanying proxy statement/prospectus. These conditions to closing in the Business Combination Agreement are for the sole benefit of the parties thereto and may be waived by such parties. There
iii
Table of Contents
can be no assurance that the parties to the Business Combination Agreement would waive any such provision of the Business Combination Agreement.
GS Sponsor II LLC (the Sponsor) and GS Acquisition Holdings II Employee Participation LLC have each agreed to, among other things, vote in favor of the Business Combination Proposal and the other proposals to be voted upon at the Special Meeting, and to waive their redemption rights in connection with the consummation of the Business Combination with respect to any shares of our common stock held by them. Our shares of GSAH Class B common stock will be excluded from the pro rata calculation used to determine the per-share redemption price. As of the date of the accompanying proxy statement/prospectus, the Sponsor and the other holders of GSAH Class B common stock own an aggregate of 20% of our outstanding shares of common stock.
We are providing the accompanying proxy statement/prospectus and accompanying proxy card to our stockholders in connection with the solicitation of proxies to be voted at the Special Meeting and at any adjournments or postponements of the Special Meeting. Information about the Special Meeting, the Business Combination and other related business to be considered by the Companys stockholders at the Special Meeting is included in the accompanying proxy statement/prospectus. Whether or not you plan to attend the Special Meeting, all of our stockholders are urged to read the accompanying proxy statement/prospectus, including the Annexes and the accompanying financial statements of the Company and Mirion, carefully and in its entirety. In particular, you should also carefully consider the risk factors described in Risk Factors beginning on page [●] of the accompanying proxy statement/prospectus.
After careful consideration, our Board of Directors has unanimously approved the Business Combination Agreement and the Business Combination, and unanimously recommends that our stockholders vote FOR adoption of the Business Combination Agreement and approval of the Business Combination, and FOR all other proposals presented to our stockholders in the accompanying proxy statement/prospectus. When you consider our Board of Directors recommendation of these proposals, you should keep in mind that our directors have interests in the Business Combination that may conflict with your interests as a stockholder. See the section entitled Certain Relationships and Related Persons Transactions for additional information.
The approval of each of the Business Combination Proposal, the Governance Proposal, the NYSE Proposal, the Incentive Plan Proposal and the Adjournment Proposal require the affirmative vote of a majority of the votes cast by holders of our outstanding shares of common stock represented at the special meeting by attendance via the virtual meeting website or by proxy and entitled to vote thereon at the Special Meeting. The approval of the Charter Proposal requires the affirmative vote of holders of a majority of our outstanding shares of common stock entitled to vote thereon at the Special Meeting. Directors are elected by a plurality of the votes cast in the Director Election Proposal; this means that the nine individuals nominated for election to our Board of Directors who receive the most FOR votes will be elected to the Board of Directors following the Closing.
Your vote is very important. Whether or not you plan to attend the Special Meeting, please vote as soon as possible by following the instructions in the accompanying proxy statement/prospectus to make sure that your shares are represented at the Special Meeting. If you hold your shares in street name through a broker, bank or other nominee, you will need to follow the instructions provided to you by your broker, bank or other nominee to ensure that your shares are represented and voted at the Special Meeting. The Business Combination is conditioned on the approval of each of the Condition Precedent Proposals at the Special Meeting. Each of the Condition Precedent Proposals is cross-conditioned on the approval of each other. The Adjournment Proposal is not conditioned upon the approval of any other proposal. Each of these proposals is more fully described in the accompanying proxy statement/prospectus, which each stockholder is encouraged to read carefully and in its entirety.
If you sign and return your proxy card without indicating how you wish to vote, your proxy will be voted FOR each of the proposals presented at the Special Meeting. If you fail to return your proxy card or fail to instruct
iv
Table of Contents
your broker, bank or other nominee how to vote, and do not attend the Special Meeting via the virtual meeting website, the effect will be, among other things, that your shares will not be counted for purposes of determining whether a quorum is present at the Special Meeting and will not be voted.
TO EXERCISE YOUR REDEMPTION RIGHTS, YOU MUST DEMAND IN WRITING THAT YOUR PUBLIC SHARES ARE REDEEMED FOR A PRO RATA PORTION OF THE FUNDS HELD IN THE TRUST ACCOUNT AND TENDER YOUR SHARES TO OUR TRANSFER AGENT AT LEAST TWO BUSINESS DAYS BEFORE THE SCHEDULED DATE OF THE SPECIAL MEETING. YOU MAY TENDER YOUR SHARES BY EITHER DELIVERING YOUR STOCK CERTIFICATE TO THE TRANSFER AGENT OR BY DELIVERING YOUR SHARES ELECTRONICALLY USING THE DEPOSITORY TRUST COMPANYS DWAC (DEPOSIT WITHDRAWAL AT CUSTODIAN) SYSTEM. IF THE BUSINESS COMBINATION IS NOT COMPLETED, THEN THESE SHARES WILL BE RETURNED TO YOU OR YOUR ACCOUNT. IF YOU HOLD THE SHARES IN STREET NAME, YOU WILL NEED TO INSTRUCT THE ACCOUNT EXECUTIVE AT YOUR BROKER, BANK OR OTHER NOMINEE TO WITHDRAW THE SHARES FROM YOUR ACCOUNT IN ORDER TO EXERCISE YOUR REDEMPTION RIGHTS.
On behalf of our Board of Directors, I would like to thank you for your support and look forward to the successful completion of the Business Combination.
| Sincerely, |
| /s/ Thomas R. Knott |
| Thomas R. Knott |
| Chief Executive Officer, Chief Financial Officer and Secretary |
NEITHER THE SECURITIES AND EXCHANGE COMMISSION NOR ANY STATE SECURITIES REGULATORY AGENCY HAS APPROVED OR DISAPPROVED THE TRANSACTIONS DESCRIBED IN THE ACCOMPANYING PROXY STATEMENT/PROSPECTUS, PASSED UPON THE MERITS OR FAIRNESS OF THE BUSINESS COMBINATION OR RELATED TRANSACTIONS OR PASSED UPON THE ADEQUACY OR ACCURACY OF THE DISCLOSURE IN THE ACCOMPANYING PROXY STATEMENT/PROSPECTUS. ANY REPRESENTATION TO THE CONTRARY CONSTITUTES A CRIMINAL OFFENSE.
The information contained in this document is subject to completion or amendment. A registration statement relating to these securities has been filed with the United States Securities and Exchange Commission. These securities may not be sold nor may offers to buy be accepted prior to the time the registration statement becomes effective. This document is not an offer to sell these securities and it is not soliciting an offer to buy these securities, nor shall there be any sale of these securities, in any jurisdiction in which such offer, solicitation or sale is not permitted or would be unlawful prior to registration or qualification under the securities laws of any such jurisdiction.
The accompanying proxy statement/prospectus is dated [●], 2021 and is expected to be first mailed to our stockholders on or about [●], 2021.
v
Table of Contents
NOTICE OF SPECIAL MEETING OF
STOCKHOLDERS OF GS ACQUISITION HOLDINGS CORP II
TO BE HELD [●], 2021
To the Stockholders of GS Acquisition Holdings Corp II:
NOTICE IS HEREBY GIVEN that a special meeting (the Special Meeting) of the stockholders of GS Acquisition Holdings Corp II, a Delaware corporation (we, us, our or the Company), will be held via live webcast on [●] at [●] a.m. [Eastern Time]. The special meeting can be accessed by visiting [●], where you will be able to listen to the meeting live and vote during the meeting.
You are cordially invited to attend the Special Meeting to conduct the following items of business:
| | Proposal No. 1Approval of the Business CombinationTo consider and vote upon a proposal to approve and adopt the Business Combination Agreement, dated as of June 17, 2021 (as it may be further amended from time to time, the Business Combination Agreement), by and among the Company, Mirion Technologies (TopCo), Ltd., a Jersey private company limited by shares Mirion, for the limited purpose set forth therein, CCP IX LP No. 1, CCP IX LP No. 2, CCP IX Co-Investment LP and CCP IX Co-Investment No. 2 LP (collectively, the Charterhouse Parties), for the limited purpose set forth therein, each of the other persons set forth on Annex I thereto and, for the limited purpose set forth therein, the other holders of A Ordinary Shares and B Ordinary Shares from time to time becoming a party thereto by executing a Joinder Agreement, a copy of which is attached to this proxy statement/prospectus as Annex A; |
| | Proposal No. 2The NYSE ProposalTo consider and vote upon a proposal to approve, for purposes of complying with applicable listing rules of the New York Stock Exchange (the NYSE), (a) the issuance of more than 20% of the Companys outstanding Class A common stock in connection with the Business Combination, including the PIPE Investment, and (b) the issuance of shares of the GSAH Class A common stock and the GSAH Class B common stock to a Related Party (as defined in Section 312.03 of the NYSEs Listed Company Manual) in connection with the Business Combination (we refer to this proposal as the NYSE Proposal); |
| | Proposal No. 3The Charter ProposalTo consider and vote upon a proposal to adopt the Second Amended and Restated Certificate of Incorporation in the form attached to the accompanying proxy statement/prospectus as Annex B (the New Mirion Charter), which, if approved, would take effect upon the closing of the Business Combination (we refer to this proposal as the Charter Proposal); |
| | Proposal No. 4The Governance Proposals To consider and vote upon a proposal to approve, on a non-binding advisory basis, certain governance provisions in the New Mirion Charter, presented separately in accordance with the United States Securities and Exchange Commission (SEC) requirements (we refer to this proposal as the Governance Proposal); |
| | Proposal No. 5The Director Election ProposalTo consider and vote upon a proposal to elect nine directors to serve, effective upon the closing of the Business Combination, on our Board of Directors until the 2022 annual meeting of stockholders, and until their respective successors are duly elected and qualified, or until their earlier resignation, removal or death (we refer to this proposal as the Director Election Proposal); |
| | Proposal No. 6The Incentive Plan ProposalTo consider and vote upon a proposal to approve the Mirion Technologies, Inc. Omnibus Incentive Plan (the Incentive Plan), including the authorization of the initial share reserve under the Incentive Plan (we refer to this proposal as the Incentive Plan Proposal); and |
| | Proposal No. 7The Adjournment ProposalTo consider and vote upon a proposal to approve the adjournment of the Special Meeting to a later date or dates, if necessary, to permit further solicitation and vote of proxies in the event that there are insufficient votes for, or for any other reason in connection with, the approval of one or more of the other proposals at the Special Meeting (we refer to this proposal as the Adjournment Proposal). |
Table of Contents
The Business Combination is conditioned on the approval of each of the Condition Precedent Proposals at the Special Meeting. Each of the Condition Precedent Proposals is cross-conditioned on the approval of each other. The Adjournment Proposal is not conditioned upon the approval of any other proposal. Each of these proposals is more fully described in this proxy statement/prospectus, which each stockholder is encouraged to read carefully and in its entirety.
The record date for the Special Meeting is [●], 2021. Only stockholders of record at the close of business on that date may vote at the Special Meeting or any adjournment thereof. A complete list of our stockholders of record entitled to vote at the Special Meeting will be available for ten days before the Special Meeting at our principal executive offices for inspection by stockholders during ordinary business hours for any purpose germane to the Special Meeting.
Pursuant to our Amended and Restated Certificate of Incorporation (the GSAH Certificate of Incorporation), a holder of our public shares (a public stockholder) may request that we redeem all or a portion of such stockholders public shares for cash if the Business Combination is consummated. Holders of units must elect to separate the units into the underlying public shares and warrants prior to exercising redemption rights with respect to the public shares. If holders hold their units through a broker, bank or other nominee, holders must notify their broker, bank or other nominee that they elect to separate the units into the underlying public shares and warrants, or if a holder holds units registered in its own name, the holder must contact Continental Stock Transfer & Trust Company, N.A., our transfer agent, directly and instruct it to do so. Public stockholders may elect to redeem their public shares even if they vote for the Business Combination Proposal or any other proposal. If the Business Combination is not consummated, the public shares will be returned to the respective holder, broker, bank or other nominee. If the Business Combination is consummated, and if a public stockholder properly exercises its right to redeem all or a portion of the public shares that it holds, including timely delivering its shares to our transfer agent, we will redeem such public shares for a per-share price, payable in cash, equal to the pro rata portion of the trust account established at the consummation of our initial public offering (the trust account), calculated as of two business days prior to the consummation of the Business Combination, including interest (net of taxes payable). For illustrative purposes, as of [●], 2021, this would have amounted to approximately $[●] per outstanding public share. If a public stockholder properly exercises its redemption rights in full, then it will be electing to exchange all of its public shares for cash and will not own any public shares of the post-business combination company. See Special Meeting of GSAH StockholdersRedemption Rights in this proxy statement/prospectus for a detailed description of the procedures to be followed if you wish to redeem your public shares for cash. Holders of our outstanding warrants do not have redemption rights in connection with the Business Combination. Unless otherwise specified, the information in this proxy statement/prospectus assumes that none of our public stockholders exercise their redemption rights with respect to their shares of GSAH Class A common stock.
Notwithstanding the foregoing, a public stockholder, together with any affiliate of such public stockholder or any other person with whom such public stockholder is acting in concert or as a group (as defined in Section 13(d)(3) of the Securities Exchange Act of 1934, as amended (the Exchange Act)), will be restricted from redeeming its public shares with respect to more than an aggregate of 15% of the public shares. Accordingly, if a public stockholder, alone or acting in concert or as a group, seeks to redeem more than 15% of the public shares, then any such shares in excess of that 15% limit would not be redeemed for cash. In addition, pursuant to the GSAH Certificate of Incorporation, in no event will we redeem public shares in an amount that would cause our net tangible assets (as determined in accordance with Rule 3a51-1(g)(1) of the Exchange Act) to be less than $5,000,001. In such case, we would not proceed with the redemption of our public shares and the Business Combination, and instead may search for an alternate initial business combination.
GS Sponsor II LLC (the Sponsor) and GS Acquisition Holdings II Employee Participation LLC have each agreed to, among other things, vote in favor of the Business Combination Proposal and the other proposals described herein to be presented at the Special Meeting, and to waive their redemption rights in connection with the consummation of the Business Combination with respect to any shares held by them. Our shares of GSAH Class B common stock will be excluded from the pro rata calculation used to determine the per-share redemption price. As of the date of this proxy statement/prospectus, the Sponsor and our officer and directors own an aggregate of 20% of our outstanding shares of Company common stock.
ii
Table of Contents
Concurrently with the execution of the Business Combination Agreement, the Company entered into subscription agreements (the Subscription Agreements) with certain investors (collectively, the PIPE Investors), pursuant to, and on the terms and subject to the conditions of which, the PIPE Investors have collectively subscribed for 90,000,000 shares of GSAH Class A common stock for an aggregate purchase price equal to $900,000,000 (the PIPE Investment and, such shares, the PIPE Shares). The PIPE Investment will be consummated substantially concurrently with the Closing.
Your vote is very important. Whether or not you plan to attend the Special Meeting, please vote as soon as possible by following the instructions in this proxy statement/prospectus to make sure that your shares are represented at the Special Meeting. If you hold your shares in street name through a broker, bank or other nominee, you will need to follow the instructions provided to you by your broker, bank or other nominee to ensure that your shares are represented and voted at the Special Meeting. The Business Combination is conditioned on the approval of each of the Condition Precedent Proposals at the Special Meeting. Each of the Condition Precedent Proposals is cross-conditioned on the approval of each other. The Adjournment Proposal is not conditioned upon the approval of any other proposal. Each of these proposals is more fully described in this proxy statement/prospectus, which each stockholder is encouraged to read carefully and in its entirety.
Your attention is directed to the remainder of this proxy statement/prospectus following this notice (including the Annexes and other documents referred to herein) for a more complete description of the proposed Business Combination and related transactions and each of the proposals. You are encouraged to read this proxy statement/prospectus carefully and in its entirety, including the Annexes and other documents referred to herein. If you have any questions or need assistance voting your shares, please contact Innisfree M&A Incorporated, our proxy solicitor, by calling: (877) 456-3463 (toll free) or Banks and Brokers may call collect: (212) 750-5833.
| By Order of the Board of Directors |
| /s/ Thomas R. Knott |
| Thomas R. Knott Chief Executive Officer, Chief Financial Officer and Secretary |
New York, NY
[●], 2021
iii
Table of Contents
| Page | ||||
| 1 | ||||
| 7 | ||||
| 10 | ||||
| QUESTIONS AND ANSWERS ABOUT THE PROPOSALS FOR OUR STOCKHOLDERS |
15 | |||
| 32 | ||||
| SELECTED UNAUDITED PRO FORMA CONDENSED COMBINED FINANCIAL INFORMATION |
66 | |||
| 68 | ||||
| UNAUDITED PRO FORMA CONDENSED COMBINED FINANCIAL INFORMATION |
128 | |||
| 151 | ||||
| 159 | ||||
| 205 | ||||
| 207 | ||||
| 209 | ||||
| 211 | ||||
| 212 | ||||
| 219 | ||||
| 220 | ||||
| GSAHS MANAGEMENTS DISCUSSION AND ANALYSIS OF FINANCIAL CONDITION AND RESULTS OF OPERATIONS |
231 | |||
| 234 | ||||
| 258 | ||||
| MIRIONS MANAGEMENTS DISCUSSION AND ANALYSIS OF FINANCIAL CONDITION AND RESULTS OF OPERATIONS |
263 | |||
| 293 | ||||
| 295 | ||||
| 303 | ||||
| 310 | ||||
| 321 | ||||
| 323 | ||||
| 329 | ||||
| 340 | ||||
| 341 | ||||
| 341 | ||||
| 342 | ||||
| 342 | ||||
| 342 | ||||
| 343 | ||||
| F-1 | ||||
ANNEXES
| Annex A |
Business Combination Agreement | A-1 | ||
| Annex B |
Form of New Mirion Charter | B-1 | ||
| Annex C |
Form of New Mirion Bylaws | C-1 | ||
| Annex D |
Amended and Restated Sponsor Agreement | D-1 | ||
| Annex E |
Form of Subscription Agreement | E-1 | ||
| Annex F |
Form of Amended and Restated Registration Rights Agreement | F-1 | ||
| Annex G |
Backstop Agreement | G-1 | ||
| Annex H |
Option Agreement | H-1 | ||
| Annex I |
Form of Equity Incentive Plan | I-1 |
i
Table of Contents
Unless otherwise stated in this proxy statement/prospectus or the context otherwise requires, references to:
| | A Ordinary Shares are to the A Ordinary Shares of $0.01 each in the capital of Mirion, having the rights set out in the Existing Company Articles; |
| | Amended and Restated Registration Rights Agreement are to that certain Amended and Restated Registration Rights Agreement, dated as of the Closing, by and between the GS Holders and the Sellers a copy of which is attached to this proxy statement/prospectus as Annex F as it may be further amended from time to time; |
| | Amended and Restated Sponsor Agreement are to that certain Amended and Restated Sponsor Agreement, the Company, dated June 29, 2020, by and among the Insiders; |
| | ASC 480 are to the Accounting Standards Codification 480-10-S99-3A |
| | ASC 815 are to the Accounting Standards Codification 815; |
| | B Ordinary Shares are to the B Ordinary Shares of $0.01 each in the capital of Mirion, having the rights set out in the Existing Company Articles; |
| | Available Closing Cash are to an amount equal to (i) the amount of funds contained in the Companys trust account (after reduction for the aggregate amount of payments required to be made in connection with any valid stockholder redemptions), plus (ii) the aggregate amount of cash that has been funded to and remains with the Company pursuant to the Subscription Agreements as of immediately prior to the closing of the Business Combination, plus (iii) the amounts delivered pursuant to the Mirion Debt Refinancing (as defined in the Business Combination Agreement), plus (iv) the cash and cash equivalents of Mirion and its subsidiaries on a consolidated basis as of the date of the Closing, plus (v) the proceeds, if any, from the sale by the Company to GSAM Holdings of shares of GSAH Class A common stock, pursuant to the Backstop Agreement, less (vi) the total amount required to be paid to fully satisfy all obligations related to Mirions credit agreement as of the Closing Date, less (vii) certain transaction expenses, less (viii) $50,000,000; |
| | Backstop Agreement are to that certain Backstop Agreement, dated as of June 17, 2021, by and between the Company and GSAM Holdings; |
| | Board, Board of Directors, or GSAH Board are to the board of directors of the Company; |
| | Business Combination are to the transactions contemplated by the Business Combination Agreement; |
| | Business Combination Agreement are to that certain Business Combination Agreement, dated June 17, 2021 (as it may be amended from time to time), by and among GSAH, Mirion, the Charterhouse Parties (each acting by its general partner, Charterhouse General Partners (IX) Limited), the other Supporting Mirion Holders, and, Joining Sellers. |
| | Cash Consideration are to $1,310,000,000; provided, however, if the Minimum Cash Condition is not satisfied, and the Company and the Charterhouse Parties elect to waive the Minimum Cash Condition within fifteen (15) Business Days of receipt by the Company and the Charterhouse Parties of a written notice from the SPAC that the Minimum Cash Condition has not been satisfied and is incapable of being satisfied, Cash Consideration shall mean $1,310,000,000 less the Cash Shortfall; |
| | Cash Shortfall are to the amount by which $1,310,000,000 exceeds the Available Closing Cash; |
| | Charterhouse Demand Period are to the exclusive right for Charterhouse Holders to for a 90-day period beginning on the 181st day after the Closing to exercise a single demand right; |
| | Charterhouse Director Nomination Agreement are to that certain Director Nomination Agreement, dated as of June 17, 2021 and as may be amended, restated or otherwise modified from time to time, by and among the Company and the Charterhouse Parties. |
1
Table of Contents
| | Charterhouse Holders or the Charterhouse Parties are to CCP IX LP No. 1, CCP IX LP No. 2, CCP IX Co-Investment LP and CCP IX Co-Investment No. 2 LP (each acting by its general partner, Charterhouse General Partners (IX) Limited); |
| | Closing are to the consummation of the Transactions; |
| | Closing Date are to the date on which the Transactions are consummated; |
| | Code are to the Internal Revenue Code of 1986, as amended; |
| | Common Stock are to the GSAH Class A common stock and the GSAH Class B common stock, together; |
| | Company, GSAH, we, us, and our are to GS Acquisition Holdings Corp II, including, as applicable, following the consummation of the Business Combination (at which time, it will have changed its name to Mirion Technologies, Inc.); |
| | Condition Precedent Proposal are to the NYSE Proposal, the Charter Proposal and the Director Election Proposal; |
| | COVID-19 are to SARS-CoV-2 or COVID-19, and any evolutions thereof or any other epidemics, pandemics or disease outbreaks; |
| | Demanding Holders are to each of (i) the Charterhouse Parties, (ii) the GS Holders or (iii) the holders of at least thirty percent (30%) in interest of the then outstanding registrable securities; |
| | DGCL are to the General Corporation Law of the State of Delaware; |
| | DWAC are to the Deposit Withdrawal at Custodian; |
| | DTC are to The Depository Trust Company; |
| | Exchange Act are to the Securities Exchange Act of 1934, as amended; |
| | Existing Mirion Articles are to Mirions Memorandum and Articles of Association, as in effect on the date of the Business Combination, and following the effectiveness of the Mirion Articles Amendment, as so amended and restated; |
| | Existing Mirion Shares are to, collectively, the A Ordinary Shares and the B Ordinary Shares; |
| | Exit are to the transfer of shares (whether through a single transaction or a series of transaction) as a result of which any person, or persons connected (as defined in Section 252 of the U.K. Companies Act) or acting in concert (as defined in the City Code on Takeovers and Mergers) with such person, holds more than 50% of the Class A and Class B ordinary shares of Mirion; |
| | Exit Bonuses are to the cash bonuses that certain executive officers of Mirion are entitled to subject to the applicable executive officer remaining actively employed with Mirion in good standing through the date of such Exit; |
| | FATCA are to the Foreign Account Tax Compliance Act; |
| | FDI are to foreign direct investment; |
| | founder shares are to the shares of GSAH Class B common stock, of which 18,750,000 shares are outstanding as of the date of this proxy statement/prospectus (17,425,000 shares of which are held by the Sponsor and 1,325,000 shares of which are held by GS Employee Participation); |
| | FCPA are to the United States Foreign Corrupt Practices Act; |
| | Freed Employment Agreement are to that certain Employment Agreement, dated as of July 16, 2016, by and between Mirion Technologies, Inc. and Mike Freed; |
| | FTC are to the U.S. Federal Trade Commission; |
| | GAAP are to the Generally Accepted Accounting Principles in the United States of America; |
2
Table of Contents
| | Goldman Sachs are to The Goldman Sachs Group, Inc., a Delaware corporation (NYSE: GS) and its affiliates (other than the Company); |
| | GS Director Nomination Agreement are to that certain Director Nomination Agreement, dated as of June 17, 2021, and as may be amended, restated or otherwise modified from time to time, by and among the Company and the Sponsor; |
| | GS Employee Participation are to GS Acquisition Holdings II Employee Participation LLC; |
| | GS Holders are to, collectively, the Sponsor, GS Employee Participation, GSAM Holdings; |
| | GSAH are to GS Acquisition Holdings Corp II, prior to the consummation of the Business Combination; |
| | GSAH Certificate of Incorporation are to GSAHs amended and restated certificate of incorporation, dated June 29, 2020; |
| | GSAH Class A common stock are to the GSAH Class A common stock, par value $0.0001 per share; |
| | GSAH Class B common stock are to the GSAH Class B common stock, par value $0.0001 per share; |
| | GSAH common stock are to, collectively, the GSAH Class A common stock and GSAH Class B common stock; |
| | GSAM are to Goldman Sachs Asset Management, a division of The Goldman Sachs Group, Inc.; |
| | GSAM Holdings are to GSAM Holdings LLC; |
| | HSR Act are to the Hart-Scott-Rodino Antitrust Improvements Act of 1976, as amended; |
| | Incentive Plan are to the Mirion Technologies, Inc. Ombnibus Equity Incentive Plan attached to this proxy statement/prospectus as Annex I; |
| | Insiders are to the Company, the Sponsor, GSAM Holdings and GS Employee Participation; |
| | Initial Stockholders are to the Sponsor and GS Employee Participation who collectively hold all of our founder shares; |
| | IntermediateCo are to Mirion IntermediateCo, Inc., a Delaware corporation; |
| | IntermediateCo Charter are to the proposed Certificate of Incorporation of IntermediateCo, in the form set forth as Exhibit C of the Business Combination Agreement; |
| | IntermediateCo Class A common stock are to the IntermediateCo Class A common stock, par value $0.0001 per share; |
| | IntermediateCo Class B common stock are to the IntermediateCo Class B common stock, par value $0.0001 per share; |
| | Investment Company Act are to the Investment Company Act of 1940, as amended; |
| | IPO or initial public offering are to GSAHs initial public offering, consummated on July 2, 2020; |
| | JOBS Act are to the Jumpstart Our Business Startups Act of 2012; |
| | Joining Sellers are to holders of A Ordinary Shares and B Ordinary Shares from time to time becoming party to the Business Combination Agreement by executing a Joinder Agreement, a copy of which is attached to this proxy statement/prospectus as Annex A; |
| | Logan Employment Agreement are to that certain Employment Agreement, dated as August 15, 2006, by and between Mirion and Thomas D. Logan, as amended; |
| | management or management team of an entity are to the officers and directors of such entity; |
| | Management Notes are to payment-in-kind loan notes issued by UKTopco to certain members of Mirion management; |
| | maximum redemption scenario are to a scenario in which, in connection with the Business Combination, the maximum number of shares of GSAH Class A common stock are redeemed such that the remaining |
3
Table of Contents
| funds held in the trust account after the payment of the redeeming shares pro-rata allocation along with the proceeds from the PIPE Investment are sufficient to satisfy the Minimum Cash Condition. Based on the amount of $750.1 million in our trust account as of March 31, 2021, including accrued dividends, and taking into account the anticipated gross proceeds of approximately $900.0 million from the PIPE Investment, approximately 36.6 million shares of GSAH Class A common stock may be redeemed and still enable GSAH to have sufficient cash to satisfy the cash closing conditions in the Business Combination Agreement; |
| | Ministry are to the Finnish Ministry of Economic Affairs and Employment; |
| | Monitoring Act are to the Finnish Act on the Monitoring of Foreign Corporate Acquisitions; |
| | Minimum Cash Condition are to the $1,310 million available for Cash Consideration; |
| | Mirion or Mirion TopCo are to Mirion Technologies (TopCo), Ltd; |
| | Mirion Articles Amendment are to no later than ten (10) Business Days following the date of the Business Combination Agreement, Mirion and the Supporting Mirion Holders took, or caused to be taken, such actions as were required to amend and restate the Existing Mirion Articles in the form set forth as Exhibit P of the Business Combination Agreement; |
| | Mirion Credit Agreement are to Mirions credit agreement, together with all related loan documents, as entered into in March 2019 and as amended thereafter. |
| | Mirion Debt Refinancing are to the repayment, refinancing or payment of consent fees related to the existing indebtedness of Mirion and its subsidiaries under the Mirion Credit Agreement; |
| | NPP are to nuclear power plant; |
| | New Mirion are to the name of the Company following the consummation of the Transactions; |
| | New Mirion Board are to the Board of Directors of New Mirion; |
| | New Mirion Bylaws are to the proposed Amended and Restated Bylaws of the Company, a form of which is attached hereto as Annex C, which will become the Companys bylaws upon the consummation of the Business Combination; |
| | New Mirion Charter are to the proposed Second Amended and Restated Certificate of Incorporation of the Company, a form of which is attached hereto as Annex B, which will become the Companys certificate of incorporation upon the consummation of the Business Combination; |
| | New Mirion Class A common stock are to GSAH Class A common stock upon the consummation of the Business Combination; |
| | New Mirion Class B common stock are to GSAH Class B common stock upon the consummation of the Business Combination; |
| | New Mirion common stock are to New Mirion Class A common stock together with New Mirion Class B common stock; |
| | New Mirion Organizational Documents are to the New Mirion Bylaws and the New Mirion Charter; |
| | NYSE are to the New York Stock Exchange; |
| | no redemption scenario are to a scenario in which, in connection with the Business Combination, no shares of GSAH Class A common stock are redeemed; |
| | Non-Reliance Periods are to, collectively, the Companys audited balance sheet as of July 2, 2020, unaudited interim financial statements for the quarterly period ended September 30, 2020 and audited financial statements as of and for the period ended December 31, 2020, as reported in the Companys Current Report on Form 8-K filed July 9, 2020, Quarterly Report on Form 10-Q filed November 13, 2020 and Annual Report on Form 10-K filed on March 31, 2021, respectively. |
4
Table of Contents
| | NYSE are to the New York Stock Exchange; |
| | Option Agreement are to that certain Option Agreement, dated as of the Closing, by and among GSAM Holdings and certain Sellers; |
| | PIPE Investment are to the private placement pursuant to which the PIPE Investors have collectively subscribed for 90,000,000 shares of GSAH Class A common stock for an aggregate purchase price equal to $900,000,000 (the PIPE Investment); |
| | PIPE Investors are to GSAM Holdings and certain other accredited investors (as defined in Rule 501 under the Securities Act) that will invest in the PIPE Investment; |
| | PIPE Shares are to the GSAH Class A common stock issued in connection with the PIPE Investment; |
| | PIK Notes are to the Shareholder Notes and the Management Notes; |
| | post-business combination company are to the Company following the consummation of the Business Combination; |
| | primary capital are to the cash available at Closing to the combined company to operate the post-closing business and for general corporate purposes; |
| | private placement warrants are to the 8.5 million private placement warrants outstanding as of the date of this proxy statement/prospectus; |
| | public shares are to the shares of GSAH Class A common stock (including those that underlie the units) that were initially offered and sold by GSAH in its IPO; |
| | public stockholders are to the holders of our public shares (including the Sponsor and the other holders of GSAH Class B common stock, provided that each of their status as a public stockholder shall only exist with respect to such public shares); |
| | public warrants are to the redeemable warrants (including those that underlie the units) that were initially offered and sold by GSAH in its IPO; |
| | redemption are to each redemption of public shares for cash pursuant to the GSAH Certificate of Incorporation; |
| | RRA Parties are to, collectively, the GS Holders, the Sellers and each other person who executes a joinder to the Amended and Restated Registration Rights Agreements; |
| | Sarbanes-Oxley Act are to the Sarbanes-Oxley Act of 2002; |
| | SEC are to the U.S. Securities and Exchange Commission; |
| | Securities Act are to the Securities Act of 1933, as amended; |
| | Sellers are to, collectively, the Joining Sellers and the other Supporting Mirion Holders; |
| | Schopfer Employment Agreement are to that certain Third Amended and Restated Employment Agreement, dated as of May 1, 2021, by and between Brian Schopfer and Mirion; |
| | Schopfer Severance Period are to that certain twelve (12)-month period after the date of Brian Schopfers termination during which he will be entitled, in addition to any accrued amount, to (i) a continuation of his annual base salary, (ii) a pro rata portion of Mr. Schopfers annual incentive bonus for the fiscal year in which the termination of his employment with Mirion occurs, payable at the same time as such payment would otherwise have been made to Mr. Schopfer had his employment with Mirion not been terminated, and (iii) continued payment by Mirion, for the Schopfer Severance Period or, if earlier, until the date on which Mr. Schopfer commences employment with and becomes eligible for health care benefits from a new employer, of the premiums associated with group health continuation coverage premiums for Mr. Schopfer and his dependents under COBRA; |
5
Table of Contents
| | Shareholder Notes are to payment-in-kind loan notes issued by UKTopco to certain Mirion TopCo shareholders; |
| | Special Meeting are to the special meeting of the stockholders of the Company that is the subject of this proxy statement/prospectus; |
| | Sponsor or GS Sponsor are to GS Sponsor II LLC, a Delaware limited liability company; |
| | Subscription Agreements are to, collectively, those certain subscription agreements entered into between the Company and the PIPE Investors; |
| | Supporting Mirion Holders are to the parties set forth on Annex I of the Business Combination Agreement, together with the Charterhouse Parties; |
| | Total Consideration are to $1,700,000,000; |
| | Transaction Payments are to are to any compensatory payments or benefits to which Thomas D. Logan becomes entitled in connection with a change in ownership or effective control (under Section 280G(b)(2) of the Code) of Mirion; |
| | Transactions are to the Business Combination and the other transactions as contemplated by the Business Combination Agreement; |
| | transfer agent are to Continental Stock Transfer & Trust Company, N.A. (Continental); |
| | trust account are to the trust account of GSAH that holds proceeds from its IPO and the sale of the private placement warrants; |
| | trustee are to Wilmington Trust, N.A.; |
| | units are to the units of the Company, each unit representing one share of GSAH Class A common stock and one-third of one redeemable warrant to acquire one share of GSAH Class A common stock, that were initially offered and sold by GSAH in its IPO (less the number of units that have been separated into the underlying public shares and underlying warrants upon the request of the holder thereof); |
| | UKBA are to the UK Anti-Bribery Act; |
| | UKTopco are to Mirion Technologies (HoldingSub1), Ltd. |
| | warrants are to public warrants and private placement warrants. |
Unless otherwise stated in this proxy statement/prospectus or as the context otherwise requires, (1) all references in this proxy statement/prospectus to Class A common stock or warrants include such securities underlying the units and (2) all references in this proxy statement/prospectus to public stockholders who properly exercise their redemption rights or similar references mean public stockholders who properly exercise, and do not properly withdraw, such election.
6
Table of Contents
CAUTIONARY STATEMENT REGARDING FORWARD-LOOKING STATEMENTS
This proxy statement/prospectus contains statements that are forward-looking and as such are not historical facts. This includes, without limitation, statements regarding the financial position, business strategy and the plans and objectives of management for future operations, including as they relate to the potential Business Combination, of the Company. These statements constitute projections, forecasts and forward-looking statements, and are not guarantees of performance. Such statements can be identified by the fact that they do not relate strictly to historical or current facts. When used in this proxy statement/prospectus, words such as anticipate, believe, continue, could, estimate, expect, intend, may, might, plan, possible, potential, predict, project, should, strive, seeks, plans, scheduled, would and similar expressions may identify forward-looking statements, but the absence of these words does not mean that a statement is not forward-looking. When the Company discusses its strategies or plans, including as they relate to the potential Business Combination, it is making projections, forecasts or forward-looking statements. Such statements are based on the beliefs of, as well as assumptions made by and information currently available to, the Companys management.
Forward-looking statements in this proxy statement/prospectus may include, for example, statements about:
| | GSAHs ability to complete the Business Combination or, if GSAH does not complete the Business Combination, any other initial business combination; |
| | satisfaction or waiver (if applicable) of the conditions to the Business Combination, including, among other things: the satisfaction or waiver of certain customary closing conditions, including, among others, the Minimum Cash Condition, receipt of approvals from governmental authorities in certain foreign jurisdictions (or expiration of applicable waiting periods in those jurisdictions), the existence of no material adverse effect at the Company or Mirion and receipt of certain stockholder approvals contemplated by this proxy statement/prospectus; |
| | the occurrence of any other event, change or other circumstances that could give rise to the termination of the Business Combination Agreement; |
| | the projected financial information, anticipated growth rate, and market opportunity of Mirion; |
| | the ability to obtain or maintain the listing of the post-business combination companys Class A common stock, warrants and units on the NYSE following the Business Combination; |
| | our public securities potential liquidity and trading; |
| | our ability to consummate the PIPE Investment or raise financing in the future; |
| | our success in retaining or recruiting, or changes required in, our officers, key employees or directors following the completion of the Business Combination; |
| | members of GSAHs management team allocating their time to other businesses and potentially having conflicts of interest with GSAHs business or in approving the Business Combination; |
| | the use of proceeds not held in the trust account or available to us from interest income on the trust account balance; |
| | factors relating to the business, operations and financial performance of Mirion and its subsidiaries, including: |
| | global economic weakness and uncertainty; |
| | risks relating to the continued growth of Mirions customers markets; |
| | failure to meet or anticipate technology changes; |
| | the unpredictability of Mirions future operational results; |
| | disruption of Mirions customers orders or Mirions customers markets; |
7
Table of Contents
| | less favorable contractual terms with large customers; |
| | risks associated with governmental contracts; |
| | failure to mitigate risks associated with long-term fixed price contracts; |
| | risks associated with information technology disruption or security; |
| | risks associated with the implementation and enhancement of information systems; |
| | failure to properly manage Mirions supply chain or difficulties with third-party manufacturers; |
| | competition in the infrastructure technologies industry; |
| | failure to realize the expected benefit from any rationalization and improvement efforts; |
| | disruption of, or changes in, Mirions independent sales representatives, distributors and original equipment manufacturers; |
| | failure to obtain performance and other guarantees from financial institutions; |
| | failure to realize sales expected from Mirions backlog of orders and contracts; |
| | changes to law, including tax law; |
| | ongoing tax audits; |
| | risks associated with future legislation and regulation of Mirions customers markets both in the United States and abroad; |
| | costs or liabilities associated with product liability; |
| | Mirions ability to attract, train and retain key members of its leadership team and other qualified personnel; |
| | the adequacy of Mirions insurance coverage; |
| | a failure to benefit from future acquisitions; |
| | failure to realize the value of goodwill and intangible assets; |
| | the global scope of Mirions operations; |
| | risks associated with Mirions sales and operations in emerging markets; |
| | exposure to fluctuations in foreign currency exchange rates; |
| | Mirions ability to comply with various laws and regulations and the costs associated with legal compliance; |
| | adverse outcomes to any legal claims and proceedings filed by or against us; |
| | Mirions ability to protect or enforce its proprietary rights on which its business depends; |
| | third party intellectual property infringement claims; |
| | liabilities associated with environmental, health and safety matters; |
| | risks associated with Mirions limited history of operating as an independent company; |
| | potential net losses in future periods; and |
| | other factors detailed under the section entitled Risk Factors. |
The forward-looking statements contained in this proxy statement/prospectus are based on current expectations and beliefs concerning future developments and their potential effects on us or Mirion. There can be no assurance
8
Table of Contents
that future developments affecting us or Mirion will be those that we or Mirion have anticipated. These forward-looking statements involve a number of risks, uncertainties (some of which are beyond GSAHs control or the control of Mirion) or other assumptions that may cause actual results or performance to be materially different from those expressed or implied by these forward-looking statements. These risks and uncertainties include, but are not limited to, those factors described under the heading Risk Factors. Should one or more of these risks or uncertainties materialize, or should any of our assumptions prove incorrect, actual results may vary in material respects from those projected in these forward-looking statements. Neither we nor Mirion undertake any obligation to update or revise any forward-looking statements, whether as a result of new information, future events or otherwise, except as may be required under applicable securities laws.
Before any GSAH stockholder grants its proxy or instructs how its vote should be cast or votes on the proposals to be put to the Special Meeting, such stockholder should be aware that the occurrence of the events described in the Risk Factors section and elsewhere in this proxy statement/prospectus may adversely affect us.
9
Table of Contents
This summary term sheet, together with the sections entitled Questions and Answers About the Proposals for Our Stockholders and Summary of the Proxy statement/prospectus, summarizes certain information contained in this proxy statement/prospectus, but does not contain all of the information that is important to you. You should read this proxy statement/prospectus, including the attached Annexes and the accompanying financial statements of the Company and Mirion, carefully and in its entirety for a more complete understanding of the matters to be considered at the Special Meeting.
| | GS Acquisition Holdings Corp II, a Delaware corporation, (the Company or GSAH) is a special purpose acquisition company formed for the purpose of effecting a merger, capital stock exchange, asset acquisition, stock purchase, reorganization or similar business combination with one or more businesses. |
| | On July 2, 2020, we completed our IPO of 75,000,000 units, including 5,000,000 units issued pursuant to the partial exercise by the underwriters of their option to purchase additional units in full, at a price of $10.00 per unit, generating proceeds to us of $750,000,000 before underwriting discounts and expenses. Each unit consisted of one share of GSAH Class A common stock and one-quarter of one redeemable warrant, with each whole warrant exercisable for one share of GSAH Class A common stock at a price of $11.50 per share. Simultaneously with the closing of the IPO, we closed the private placement of an aggregate of 8,500,000 warrants, each exercisable to purchase one share of GSAH Class A common stock at an exercise price of $11.50 per share, to the Sponsor, at a price of $2.00 per private placement warrant, generating proceeds of $17,000,000. Each warrant sold in the IPO and the private placement will become exercisable 30 days after the completion of our initial business combination, and will expire at 5:00 p.m., New York City time, five years after the completion of our initial business combination or earlier upon redemption or liquidation. Subject to the terms and conditions contained in the warrant agreement governing the warrants (the warrant agreement), we may redeem the warrants either for cash once the warrants become exercisable or for shares of GSAH Class A common stock commencing 90 days after the warrants become exercisable. For more information regarding the warrants, please see the section entitled Description of New Mirion Securities. |
| | Mirion provides products, services and software that allow its customers to safely leverage the power of ionizing radiation for the greater good of humanity. Mirions solutions have critical applications in the medical, nuclear energy and defense markets, as well as in laboratories and scientific research, analysis and space exploration. Many of Mirions markets are characterized by the need to meet rigorous regulatory standards, design qualifications and operating requirements. Throughout Mirions history, Mirion has successfully leveraged the strength of its expertise in ionizing radiation to continually drive innovation and expand the commercial applications of its core technology competencies. Through its facilities in 12 countries, Mirion supplies its solutions in the Americas, Europe, Africa, the Middle East and Asia Pacific regions. |
| | On June 17, 2021, the Company, Mirion, for the limited purpose set forth therein, the Charterhouse Parties, for the limited purpose set forth therein, the other Supporting Mirion Holders and, for the limited purpose set forth therein, the other holders of Existing Mirion Shares from time to time becoming a party thereto by executing a Joinder Agreement, entered into the Business Combination Agreement, a copy of which is attached to this proxy statement/prospectus as Annex A. |
| | Pursuant to the terms of the Business Combination Agreement, the parties thereto will enter into a business combination transaction pursuant to which Mirion will combine with a subsidiary of the Company. |
| | The proposed Business Combination will establish the Company as the corporate parent of Mirion. At the Closing, in order to implement a structure similar to that of an Up-C, the Company will establish a Delaware corporation (IntermediateCo) as a subsidiary of the Company. Sellers will be permitted to elect to receive equity consideration either in the form of (i) shares of GSAH Class A common stock or (ii) paired interests consisting of shares of GSAH Class B common stock paired together with shares of IntermediateCos Class B common stock. Holders of shares of IntermediateCos Class B common stock will |
10
Table of Contents
| have a redemption right for such shares to be settled, at the option of the Company, with (i) shares of GSAH Class A common stock (on a one-for-one basis) or (ii) a cash amount per share based on an average trailing stock price of GSAH Class A common stock. In the event of a redemption request by a holder of shares of IntermediateCo Class B common stock, the Company may, at its option, effect a direct exchange of cash or Company Class A common stock for shares of IntermediateCo Class B common stock in lieu of such a redemption. Upon redemption or exchange of shares of IntermediateCo Class B common stock, the corresponding shares of GSAH Class B common stock will be cancelled. The Company will hold 100% of the voting shares of IntermediateCo Class A common stock. As further described in the New Mirion Charter to be in effect after the Closing, each holder of GSAH Class B common stock will be entitled to one vote for each share of GSAH Class B common stock held of record by such holder on all matters on which stockholders generally are entitled to vote and the holders of GSAH Class B common stock shall not be entitled to dividends of cash or property on such shares of GSAH Class B common stock. |
| | In accordance with the terms of the Business Combination Agreement, and subject to the adjustments set forth therein, the consideration to be paid in connection with the Business Combination is $1,700,000,000 and will be paid in a combination of equity and cash consideration. The cash consideration will be an amount equal to $1,310,000,000; provided, that if the Minimum Cash Condition is not met, and Mirion and the Charterhouse Parties elect to waive the Minimum Cash Condition, then the Cash Consideration will be equal to $1,310,000,000 less the amount by which $1,310,000,000 exceeds the Available Closing Cash. In exchange for the A Ordinary Shares, the B Ordinary Shares and certain loan notes due 2026 issued by Mirion Technologies (HoldingSub1), Ltd, each Seller may elect to receive cash or equity consideration or a combination thereof, which equity consideration shall be in the form of either shares of GSAH Class A common stock or shares of GSAH Class B common stock combined with shares of IntermediateCo Class B common stock that will be majority owned by the Company. The Available Closing Cash will be an amount equal to (i) the amount of funds contained in the Companys trust account (after reduction for the aggregate amount of payments required to be made in connection with any valid stockholder redemptions), plus (ii) the aggregate amount of cash that has been funded to and remains with the Company pursuant to the Subscription Agreements as of immediately prior to the Closing, plus (iii) the amounts delivered pursuant to the Mirion Debt Refinancing, plus (iv) the cash and cash equivalents of Mirion and its subsidiaries on a consolidated basis as of the Closing Date, plus (v) the proceeds, if any, from the sale by the Company to GSAM Holdings of shares of GSAH Class A common stock, pursuant to the Backstop Agreement, less (vi) the total amount required to be paid to fully satisfy all obligations related to Mirions credit agreement as of the Closing Date, less (vii) certain transaction expenses, less (viii) $50,000,000. |
| | Concurrently with the execution of the Business Combination Agreement, the Company entered into the Subscription Agreements with the PIPE Investors, pursuant to, and on the terms and subject to the conditions of which, the PIPE Investors have collectively subscribed for 90,000,000 shares of GSAH Class A common stock for an aggregate purchase price equal to $900,000,000. The PIPE Investment will be consummated substantially concurrently with the Closing. |
It is anticipated that, upon completion of the Business Combination:
| | the PIPE Investors (including GSAM Holdings, assuming no syndication of its subscription) will own approximately 44% of the outstanding GSAH common stock; |
| | our public stockholders will own approximately 37% of the outstanding GSAH common stock; |
| | the Sellers, other than current members of Mirion management, will own approximately 15% of outstanding GSAH common stock; |
| | Sellers who are current members of Mirion management will own approximately 4% of the outstanding GSAH common stock; and |
| | the GS Sponsor will own 0% of the outstanding GSAH common stock (assuming, for this purpose, that none of the founder shares performance vesting conditions have been satisfied at the time of completion of the Business Combination). |
11
Table of Contents
These levels of ownership interest: (i) assume that no public shares are elected to be redeemed in connection with the Business Combination, (ii) exclude the 18,750,000 outstanding founder shares held by the GS Sponsor (all of which are subject to the performance vesting conditions and forfeiture as described herein), (iii) assume no exercise of any warrants to purchase Class A common stock that will remain outstanding immediately following the Business Combination and (iv) exclude the issuance of any shares under the Incentive Plan following the Business Combination. If the actual facts are different from these assumptions, the above levels of ownership interest will be different.
For more information, see Summary of the Proxy Statement/ProspectusOwnership of the Company Following the Business Combination and Unaudited Pro Forma Condensed Combined Financial Information.
| | In evaluating the Business Combination, our Board considered a number of factors, including Mirions highly attractive business model, Mirions deep relationships with a diverse customer base, Mirions strong recurring revenue, Mirions experienced and proven management team, Mirions strong balance sheet, other alternatives, terms of the Business Combination Agreement, continued ownership by sellers and the role of the independent directors. For more information about our decision-making process, as well as other factors, uncertainties and risks considered, see the section entitled Proposal No. 1Approval of the Business Combination GSAHs Board of Directors Reasons for the Approval of the Business Combination. |
| | Pursuant to the GSAH Certificate of Incorporation, a public stockholder may request that we redeem all or a portion of such stockholders public shares for cash if the Business Combination is consummated. Holders of units must elect to separate the units into the underlying public shares and warrants prior to exercising redemption rights with respect to the public shares. If holders hold their units through a broker, bank or other nominee, holders must notify their broker, bank or other nominee that they elect to separate the units into the underlying public shares and warrants, or if a holder holds units registered in its own name, the holder must contact Continental Stock Transfer & Trust Company, N.A., our transfer agent, directly and instruct it to do so. Public stockholders may elect to redeem their public shares even if they vote FOR the Business Combination Proposal or any other proposal. If the Business Combination is not consummated, the public shares will be returned to the respective holder, broker, bank or other nominee. If the Business Combination is consummated, and if a public stockholder properly exercises its right to redeem all or a portion of the public shares that it holds, including by timely delivering its shares to our transfer agent, we will redeem such public shares for a per-share price, payable in cash, equal to the pro rata portion of the trust account, calculated as of two business days prior to the consummation of the Business Combination, including interest (net of taxes payable). For illustrative purposes, as of [●], 2021, this would have amounted to approximately $10.25 per outstanding public share. If a public stockholder properly exercises its redemption rights in full, then it will be electing to exchange all of its public shares for cash and will not own any public shares of the post-business combination company. Holders of our outstanding warrants do not have redemption rights in connection with the Business Combination. Please see the section entitled Special Meeting of GSAH StockholdersRedemption Rights. |
| | In addition to voting on the proposal to approve and adopt the Business Combination Agreement and approve the Business Combination (we refer to this proposal as the Business Combination Proposal), at the Special Meeting, our stockholders will be asked to vote upon: |
| | a proposal to approve, for purposes of complying with applicable listing rules of NYSE, (a) the issuance of more than 20% of the Companys outstanding Class A common stock in connection with the Business Combination, including the PIPE Investment, and (b) the issuance of shares of the Companys Class A common stock and the Companys Class B common stock to a Related Party (as defined in Section 312.03 of the NYSEs Listed Company Manual) in connection with the Business Combination (we refer to this proposal as the NYSE Proposal); |
| | a proposal relating to adopting the New Mirion Charter, which, if approved, would take effect upon the closing of the Business Combination (we refer to this proposal as the Charter Proposal); |
12
Table of Contents
| | a proposal to approve, on a non-binding basis, certain governance provisions in the New Mirion Charter, in accordance with SEC requirements (we refer to this proposal as the Governance Proposal) |
| | a proposal to elect nine directors to serve, effective upon the closing of the Business Combination, with each director on our Board having a term that expires at the post-business combination companys annual meeting of stockholders in 2022, and until their respective successors are duly elected and qualified, or until their earlier resignation, removal or death (we refer to this proposal as the Director Election Proposal); |
| | a proposal to approve the Incentive Plan, including the authorization of the initial share reserve under the Incentive Plan (we refer to this proposal as the Incentive Plan Proposal); and |
| | a proposal to adjourn the Special Meeting to a later date or dates, if necessary, to permit further solicitation and vote of proxies in the event there are insufficient votes for, or for any other reason in connection with, the approval of one or more of the other proposals at the Special Meeting (we refer to this proposal as the Adjournment Proposal). |
See Proposal No. 1Approval of the Business Combination, Proposal No. 2The NYSE Proposal, Proposal No. 3The Charter Proposal, Proposal No. 4The Governance Proposal, Proposal No. 5The Director Election Proposal, Proposal No. 6The Incentive Plan Proposal and Proposal No. 7The Adjournment Proposal. The Business Combination is conditioned on the approval of each of the Condition Precedent Proposals at the Special Meeting. Each of the Condition Precedent Proposals is cross-conditioned on the approval of each other. The Adjournment Proposal is not conditioned upon the approval of any other proposal. Each of these proposals is more fully described in this proxy statement/prospectus, which each stockholder is encouraged to read carefully and in its entirety.
| | Upon consummation of the Business Combination, our Board anticipates increasing its initial size from five directors to nine directors, with each director on our Board having a term that expires at the post-business combination companys annual meeting of stockholders in 2022, and until their respective successors are duly elected and qualified, or until their earlier resignation, removal or death. Please see the sections entitled Proposal No. 5The Director Election Proposal and Management of New Mirion Following the Business Combination for additional information. |
| | Unless waived by the parties to the Business Combination Agreement, and subject to applicable law, the closing of the Business Combination is subject to a number of conditions set forth in the Business Combination Agreement including, among others, (i) the Company having at least an aggregate of $1.310 billion in cash available at Closing; (ii) the registration statement becoming effective in accordance with the Securities Act; (iii) customary bringdown conditions; (iv) no material adverse effect of either the Company or Mirion having occurred; and (v) to the extent requested by the Company, Mirion having issued a notice of suspension or termination of business with certain partners. There can be no assurance that the parties to the Business Combination Agreement would waive any such provision of the Business Combination Agreement. For more information about the closing conditions to the Business Combination, please see the section entitled Proposal No. 1Approval of the Business CombinationThe Business Combination AgreementConditions to Closing of the Business Combination. |
| | The proposed Business Combination, including our business following the Business Combination, involves numerous risks. For more information about these risks, please see the section entitled Risk Factors. |
| | When you consider the recommendation of our Board in favor of approval of the Business Combination Proposal and the other proposals included herein, you should keep in mind that the Sponsor and our directors have interests in such proposal that are different from, or in addition to, those of our stockholders and warrant holders generally. Our Board was aware of and considered these interests, among other matters, in evaluating and negotiating the Business Combination and transaction agreements and in recommending to our stockholders that they vote in favor of the proposals presented at the Special Meeting, including the |
13
Table of Contents
| Business Combination Proposal. GSAH stockholders should take these interests into account in deciding whether to approve the proposals presented at the Special Meeting, including the Business Combination Proposal. See Proposal No. 1Approval of the Business CombinationInterests of Goldman Sachs Parties and Certain Other Persons in the Business Combination. |
14
Table of Contents
QUESTIONS AND ANSWERS ABOUT THE PROPOSALS FOR OUR STOCKHOLDERS
The questions and answers below highlight only selected information from this document and only briefly address some commonly asked questions about the proposals to be presented at the Special Meeting, including with respect to the proposed Business Combination. The following questions and answers do not include all the information that is important to our stockholders. We urge our stockholders to read this proxy statement/prospectus, including the Annexes and the accompanying financial statements of the Company and Mirion carefully and in their entirety to fully understand the proposed Business Combination and the voting procedures for the Special Meeting, which will be held via live webcast on [●], 2021 at [●] a.m. [Eastern Time], or at such other time, on such other date and at such other place to which the meeting may be adjourned or postponed.
| Q: | Why am I receiving this proxy statement/prospectus? |
| A: | Our stockholders are being asked to consider and vote upon, among other proposals, a proposal to approve and adopt the Business Combination Agreement and approve the Business Combination. The Business Combination Agreement provides for, among other things, the parties thereto entering into a business combination transaction pursuant to which Mirion will combine with a subsidiary of the Company. |
The proposed Business Combination will establish the Company as the corporate parent of Mirion. At the Closing, in order to implement a structure similar to that of an Up-C, the Company will establish a Delaware corporation, IntermediateCo, as a subsidiary of the Company. Sellers will be permitted to elect to receive equity consideration either in the form of (i) shares of GSAH Class A common stock or (ii) paired interests consisting of shares of GSAH Class B common stock paired together with shares of IntermediateCo Class B common stock. Holders of shares of IntermediateCo Class B common stock will have a redemption right for such shares to be settled, at the option of the Company, with (i) shares of GSAH Class A common stock (on a one-for-one basis) or (ii) a cash amount per share based on an average trailing stock price of GSAH Class A common stock. In the event of a redemption request by a holder of shares of IntermediateCo Class B common stock, the Company may, at its option, effect a direct exchange of cash or Company Class A common stock for shares of IntermediateCo Class B common stock in lieu of such a redemption. Upon redemption or exchange of shares of IntermediateCo Class B common stock, the corresponding shares of GSAH Class B common stock will be cancelled. The Company will hold 100% of the voting shares of IntermediateCo Class A common stock. As further described in the New Mirion Charter to be in effect after the Closing, each holder of GSAH Class B common stock will be entitled to one vote for each share of GSAH Class B common stock held of record by such holder on all matters on which stockholders generally are entitled to vote and the holders of GSAH Class B common stock shall not be entitled to dividends of cash or property on such shares of GSAH Class B common stock
See the section entitled Proposal No. 1Approval of the Business Combination for more detail.
A copy of the Business Combination Agreement is attached to this proxy statement/prospectus as Annex A and you are encouraged to read it carefully and in its entirety.
THE VOTE OF STOCKHOLDERS IS IMPORTANT. STOCKHOLDERS ARE ENCOURAGED TO VOTE AS SOON AS POSSIBLE AFTER READING THIS PROXY STATEMENT/PROSPECTUS, INCLUDING THE ANNEXES AND THE ACCOMPANYING FINANCIAL STATEMENTS OF THE COMPANY AND MIRION, CAREFULLY AND IN ITS ENTIRETY.
| Q: | When and where is the Special Meeting? |
| A: | The Special Meeting will be held via live webcast on [●], 2021 at [●]:00 a.m. [Eastern Time], or at such other time, on such other date and at such other place to which the meeting may be adjourned or postponed. The special meeting can be accessed by visiting [●], where you will be able to listen to the meeting live and vote during the meeting. |
15
Table of Contents
| Q: | What are the specific proposals on which I am being asked to vote at the Special Meeting? |
| A: | In addition to voting on a proposal to approve and adopt the Business Combination Agreement and approve the Business Combination, at the Special Meeting, GSAH is asking holders of its common stock to consider and vote upon: |
| | a proposal to approve, for purposes of complying with applicable listing rules of the NYSE, (a) the issuance of more than 20% of the Companys outstanding Class A common stock in connection with the Business Combination, including the PIPE Investment, and (b) the issuance of shares of the Companys Class A common stock and the Companys Class B common stock to a Related Party (as defined in Section 312.03 of the NYSEs Listed Company Manual) in connection with the Business Combination; |
| | a proposal relating to adopting the New Mirion Charter, which, if approved, would take effect upon the closing of the Business Combination; |
| | a proposal to approve, on a non-binding basis, certain governance provisions in the New Mirion Charter; |
| | a proposal to elect nine directors to serve, effective upon the closing of the Business Combination, with each director on our Board having a term that expires at the post-business combination companys annual meeting of stockholders in 2022, and until their respective successors are duly elected and qualified, or until their earlier resignation, removal or death; |
| | a proposal to approve the Incentive Plan, including the authorization of the initial share reserve under the Incentive Plan; and |
| | a proposal to adjourn the Special Meeting to a later date or dates, if necessary, to permit further solicitation and vote of proxies in the event there are insufficient votes for, or for any other reason in connection with, the approval of one or more of the other proposals at the Special Meeting. |
If our stockholders do not approve each of the Condition Precedent Proposals, then unless certain conditions in the Business Combination Agreement are waived by the applicable parties to the Business Combination Agreement, the Business Combination Agreement could terminate and the Business Combination may not be consummated. See Proposal No. 1Approval of the Business Combination, Proposal No. 2The NYSE Proposal, Proposal No. 3The Charter Proposal, Proposal No. 4The Governance Proposal, Proposal No. 5The Director Election Proposal, Proposal No. 6The Incentive Plan Proposal and Proposal No. 7The Adjournment Proposal. We will hold the Special Meeting to consider and vote upon these proposals. This proxy statement/prospectus contains important information about the Business Combination and the other matters to be acted upon at the Special Meeting. Our stockholders should read it carefully and in its entirety.
After careful consideration, GSAHs Board of Directors has determined that the Business Combination Proposal, the NYSE Proposal, the Charter Proposal, the Governance Proposal, the Director Election Proposal, the Incentive Plan Proposal and the Adjournment Proposal are in the best interests of GSAH and its stockholders and unanimously recommends that you vote or give instruction to vote FOR each of those proposals.
The existence of financial and personal interests of one or more of GSAHs directors may result in a conflict of interest on the part of such director(s) between what he or they may believe is in the best interests of GSAH and its stockholders and what he or they may believe is best for himself or themselves in determining to recommend that stockholders vote for the proposals. See Proposal No. 1Approval of the Business CombinationInterest of Goldman Sachs Parties and Certain Other Persons in the Business Combination for a further discussion of these considerations.
| Q: | Are the proposals conditioned on one another? |
| A: | Yes. The Business Combination is conditioned on the approval of each of the Condition Precedent Proposals at the Special Meeting. Each of the Condition Precedent Proposals is cross-conditioned on the approval of each other. The Adjournment Proposal is not conditioned upon the approval of any other proposal. |
16
Table of Contents
| Q: | Why is GSAH proposing the Business Combination? |
| A: | GSAH was organized to effect a merger, capital stock exchange, asset acquisition, stock purchase, reorganization or similar business combination with one or more businesses. |
Mirion provides products, services and software that allow its customers to safely leverage the power of ionizing radiation for the greater good of humanity. Mirions solutions have critical applications in the medical, nuclear energy and defense markets, as well as in laboratories and scientific research, analysis and space exploration. Many of Mirions markets are characterized by the need to meet rigorous regulatory standards, design qualifications and operating requirements. Throughout Mirions history, Mirion has successfully leveraged the strength of its expertise in ionizing radiation to continually drive innovation and expand the commercial applications of its core technology competencies. Through its facilities in 12 countries, Mirion supplies its solutions in the Americas, Europe, Africa, the Middle East and Asia Pacific regions. For more information about Mirion see Information about Mirion.
Based on its due diligence investigations of Mirion and the industry in which it operates, including the financial and other information provided by Mirion in the course of our due diligence investigations, our Board believes that the Business Combination with Mirion is in the best interests of us and our stockholders and presents an opportunity to increase stockholder value. However, there is no assurance of this. See Proposal No. 1Approval of the Business Combination GSAHs Board of Directors Reasons for the Approval of the Business Combination for additional information.
Although our Board believes that the Business Combination with Mirion presents a unique business combination opportunity and is in the best interests of us and our stockholders, our Board did consider certain potentially material negative factors in arriving at that conclusion. These factors are discussed in greater detail in the section entitled Proposal No. 1Approval of the Business Combination GSAHs Board of Directors Reasons for the Approval of the Business Combination, as well as in the sections entitled Risk Factors Risks Related to Mirions Business.
| Q: | What will Mirion stockholders receive in return for GSAHs acquisition of all of the outstanding equity interests of Mirion? |
| A: | As a result of and upon the consummation of the Business Combination, among other things, all outstanding equity interests of Mirion will be cancelled in exchange for the right to receive the Transaction Consideration. |
Subject to the terms of the Business Combination Agreement, the consideration to be paid in connection with the Business Combination is $1,700,000,000 and will be paid in a combination of equity and cash consideration. The cash consideration will be an amount equal to $1,310,000,000; provided, that if the Minimum Cash Condition is not met, and Mirion and the Charterhouse Parties elect to waive the Minimum Cash Condition, then the Cash Consideration will be equal to $1,310,000,000 less the amount by which $1,310,000,000 exceeds the Available Closing Cash. In exchange for the A Ordinary Shares of $0.01, the B Ordinary Shares of $0.01 and certain loan notes due 2026 issued by Mirion Technologies (HoldingSub1), Ltd, each Seller may elect to receive cash or equity consideration or a combination thereof, which equity consideration shall be in the form of either shares of the Companys Class A common stock or shares of the Companys Class B common stock combined with shares of IntermediateCo Class B common stock that will be majority owned by the Company.
The Available Closing Cash will be an amount equal to (i) the amount of funds contained in the Companys trust account (after reduction for the aggregate amount of payments required to be made in connection with any valid stockholder redemptions), plus (ii) the aggregate amount of cash that has been funded to and remains with the Company pursuant to the Subscription Agreements as of immediately prior to the Closing, plus (iii) the amounts delivered pursuant to the Mirion Debt Refinancing, plus (iv) the cash and cash equivalents of Mirion and its subsidiaries on a consolidated basis as of the Closing Date, plus (v) the proceeds, if any, from the sale by the Company to GSAM Holdings of shares of the Companys Class A
17
Table of Contents
common stock, pursuant to the Backstop Agreement, less (vi) the total amount required to be paid to fully satisfy all obligations related to the Mirion Credit Agreement as of the Closing Date, less (vii) certain transaction expenses, less (viii) $50,000,000.
For further details, see Proposal No. 1Approval of the Business CombinationThe Business Combination Agreement.
| Q: | What equity stake will current stockholders of GSAH and the PIPE Investors, including GSAM Holdings, hold in us after the closing? |
| A: | As of March 31, 2021, there were 93,750,000 shares of GSAH common stock outstanding, which includes the 18,750,000 founder shares held by the GS Sponsor and the 75,000,000 public shares. As of March 31, 2021, there are outstanding warrants to purchase an aggregate of 27,250,000 shares of GSAH common stock, which includes 8,500,000 private placement warrants held by the Sponsor and approximately 18,750,000 public warrants. Therefore, as of March 31, 2021 (without giving effect to the Business Combination), our fully diluted share capital would be approximately 121,000,000 shares of GSAH common stock. |
It is anticipated that, upon completion of the Business Combination:
| | the PIPE Investors (including GSAM Holdings, assuming no syndication of its subscription) will own approximately 44% of the outstanding GSAH common stock; |
| | our public stockholders will own approximately 37% of the outstanding GSAH common stock; |
| | the Sellers, other than current members of Mirion management, will own approximately 15% of outstanding GSAH common stock; |
| | Sellers who are current members of Mirion management will own approximately 4% of the outstanding GSAH common stock; and |
| | the GS Sponsor will own 0% of the outstanding GSAH common stock (assuming, for this purpose, that none of the founder shares performance vesting conditions have been satisfied at the time of completion of the Business Combination). |
These levels of ownership interest (i) assume that no public shares are elected to be redeemed in connection with the Business Combination, (ii) exclude the 18,750,000 outstanding founder shares held by the GS Sponsor (all of which are subject to the performance vesting conditions and forfeiture as described herein), (iii) assume no exercise of any warrants to purchase Class A common stock that will remain outstanding immediately following the Business Combination and (iv) exclude the issuance of any shares under the Incentive Plan following the Business Combination. In addition, the consideration to be paid to the Mirion Sellers in connection with the Business Combination is allocated first in respect of the PIK Notes and second in respect of the Existing Mirion Shares. The PIK Notes accrue payment-in-kind (PIK) interest daily at a rate of 11.5% annually and are contemplated to be sold at the Closing for a price equal to the full outstanding principal amount together with all accrued but unpaid interest up to but excluding the Closing Date. See Certain Relationships and Related Persons TransactionsMirions Related Person TransactionsShareholder Notes. Accordingly, as the amount of accrued and unpaid interest increases, the holders of the PIK Notes will receive more consideration as compared to the holders of the Existing Mirion Shares. This has the effect over time of increasing the amount of shares of GSAH Class A Common Stock to be issued to the Mirion Sellers (excluding members of Mirion management) and reducing the number of shares of GSAH Class B common stock that will be issued to Mirion management (there is no incremental dilution to Public Stockholders). For purposes of the ownership levels described herein, we have assumed an amount of principal and interest of the PIK Notes as if the Closing Date will be October 31, 2021, but for all other purposes have assumed the Closing Date will be March 31, 2021.
If the actual facts are different from these assumptions, the above levels of ownership interest will be different.
18
Table of Contents
The founder shares are subject to vesting in three equal tranches, based on the volume-weighted average price of the Companys GSAH Class A common stock being greater than or equal to $12.00, $14.00 and $16.00 per share (each, a Founder Share Vesting Event) for any 20 trading days in any 30 consecutive trading day period. Vesting of the founder shares will be accelerated upon certain sale events based on the per share price of the GSAH Class A common stock in such sale event. Holders of the founder shares are entitled to vote such founder shares and receive dividends and other distributions with respect to such founder shares prior to vesting, but such dividends and other distributions with respect to unvested founder shares will be set aside by the Company and shall only be paid to the holders of the founder shares upon the vesting of such founder shares. The founder shares will be forfeited to the Company for no consideration if they fail to vest within five years of the closing of the Business Combination.
In conjunction with the Business Combination Agreement, the Sponsor issued 3,200,000 membership interests to Thomas Logan, the Chief Executive Officer of Mirion, 700,000 membership interests to Brian Schopfer, the Chief Financial Officer of Mirion, and 4,200,000 membership interests to Lawrence Kingsley, who is expected to be Chairman of the Board of New Mirion (collectively, the Profits Interests). The Profits Interests are intended to be treated as profits interests for U.S. income tax purposes, pursuant to which Messrs. Logan, Schopfer and Kingsley will have an indirect interest in the founder shares held by the Sponsor. The Profits Interests are subject to service and performance vesting conditions, including the occurrence of the Closing, and do not fully vest until all of the applicable conditions are satisfied. In addition, the Profits Interests are subject to certain forfeiture conditions. See Certain Relationships and Related Persons TransactionsMirions Related Person TransactionsProfits Interests.
In addition, we expect that the Sellers who are current members of Mirions management team will elect to receive shares of GSAH Class B common stock of the Company that will have voting rights but no economic interest in the Company, paired with shares of IntermediateCo Class B common stock (non-voting) of a newly formed subsidiary (IntermediateCo) of GSAH (the Paired Interests). The share calculations assume that only members of Mirions management team will elect to receive shares of GSAH Class B common stock and that the other Sellers receiving shares of GSAH common stock will elect to receive shares of GSAH Class A common stock.
The following table illustrates varying ownership levels in New Mirion (as a percentage of outstanding common stock) immediately following the consummation of the Business Combination based on the assumptions above plus a maximum redemption scenario which assumes the maximum number of shares of GSAH Class A common stock are redeemed such that the remaining funds held in the trust account after the payment of the redeeming shares pro-rata allocation along with the proceeds from the PIPE Investment are sufficient to fund the Minimum Cash Condition.
| Pro Forma Class A Share Ownership in the Company | ||||||||||||||||
| No Redemptions | Maximum Redemptions (1) | |||||||||||||||
| Number of Shares (millions) |
Percentage of Outstanding Shares |
Number of Shares (millions) |
Percentage of Outstanding Shares |
|||||||||||||
| PIPE Investors (2) |
90.0 | 44 | % | 90.0 | 50 | % | ||||||||||
| Public Stockholders |
75.0 | 37 | % | 38.4 | 21 | % | ||||||||||
| Mirion Sellers (excluding Mirion Management (3) |
30.0 | 15 | % | 30.0 | 17 | % | ||||||||||
| GS Sponsor (4) |
| 0 | % | | 0 | % | ||||||||||
| GS Backstop(5) |
| 0 | % | 12.5 | 7 | % | ||||||||||
| Pro Forma Class B Share Ownership in the Company | ||||||||||||||||
| No Redemptions | Maximum Redemptions | |||||||||||||||
| Number of Shares (millions) |
Percentage of Outstanding Shares |
Number of Shares (millions) |
Percentage of Outstanding Shares |
|||||||||||||
| Mirion Management (3)(4) |
9.0 | 4 | % | 9.0 | 5 | % | ||||||||||
19
Table of Contents
| (1) | Assumes that 36.6 million public shares (being our estimate of the maximum number of public shares that could be redeemed in connection with the Business Combination in order to satisfy the related Minimum Cash Condition contained in the Business Combination Agreement) are redeemed in connection with the Business Combination. |
| (2) | Includes 20 million GSAH Class A shares subscribed for by GSAM Holdings, assuming no syndication of its subscription. |
| (3) | Mirion Sellers have the option of receiving either shares of GSAH Class A common stock or Paired Interests at closing. We have assumed that all Mirion Sellers with the exception of members of Mirion management will elect to receive GSAH Class A common stock. |
| (4) | Excludes 18,750,000 founder shares that convert from shares of GSAH Class B common stock to shares of GSAH Class A common stock upon the closing of the Business Combination and are subject to certain vesting and forfeiture conditions described above. |
| (5) | The maximum redemptions scenario assumes there is a Cash Shortfall such that GSAM Holdings will purchase 12,500,000 shares of GSAH Class A common stock under the Backstop Agreement. |
For more information, see Summary of the Proxy Statement/ProspectusOwnership of the Company Following the Business Combination and Unaudited Pro Forma Condensed Combined Financial Information.
| Q: | What will happen in the Business Combination? |
| A: | Pursuant to the Business Combination Agreement, among other things and subject to the terms and conditions contained therein, the parties thereto will enter into a business combination transaction, pursuant to which Mirion will combine with a subsidiary of the Company as described below. |
The proposed Business Combination will establish the Company as the corporate parent of Mirion. At the Closing, in order to implement a structure similar to that of an Up-C, the Company will establish a Delaware corporation, IntermediateCo, as a subsidiary of the Company. Sellers will be permitted to elect to receive equity consideration either in the form of (i) shares of the Companys Class A common stock or (ii) paired interests consisting of shares of GSAH Class B common stock paired together with shares of IntermediateCo Class B common stock. Holders of shares of IntermediateCo Class B common stock will have a redemption right for such shares to be settled, at the option of the Company, with (i) shares of GSAH Class A common stock (on a one-for-one basis) or (ii) a cash amount per share based on an average trailing stock price of the Companys Class A common stock. In the event of a redemption request by a holder of shares of IntermediateCo Class B common stock, the Company may, at its option, effect a direct exchange of cash or GSAH Class A common stock for shares of IntermediateCo Class B common stock in lieu of such a redemption. Upon redemption or exchange of shares of IntermediateCos Class B common stock, the corresponding shares of GSAH Class B common stock will be cancelled. The Company will hold 100% of the voting shares of IntermediateCo Class A common stock. As further described in the New Mirion Charter to be in effect after the Closing, each holder of GSAH Class B common stock will be entitled to one vote for each share of GSAH Class B common stock held of record by such holder on all matters on which stockholders generally are entitled to vote and the holders of GSAH Class B common stock shall not be entitled to dividends of cash or property on such shares of GSAH Class B common stock.
See the section entitled Proposal No. 1Approval of the Business Combination for more detail.
| Q: | Following the Business Combination, will the Companys securities continue to trade on a stock exchange? |
| A: | Yes. We intend to apply to continue the listing of GSAH Class A common stock and warrants on the NYSE under the symbols MIR and MIR.WS, respectively, upon the closing of the Business Combination, though such securities may not be listed, for instance if there is not a sufficient number of round lot holders. At the closing of the Business Combination, each unit will separate into its components consisting of one share of GSAH Class A common stock and one-fourth of one warrant. GSAH is currently awaiting |
20
Table of Contents
| preliminary listing approval from NYSE in order to submit its listing application and believes the combined entity will satisfy all criteria for listing upon completion of the Business Combination. As such, GSAH expects to obtain NYSE listing approval prior to the Closing notwithstanding, GSAH can provide no assurances that NYSE will approve the listing application. NYSEs determination may not be known at the time stockholders are asked to vote on the Business Combination and the closing is not conditioned on NYSEs approval of the continued listing, but the closing of the PIPE Investment is conditioned on the PIPE Shares being listed on the NYSE. |
| Q: | How has the announcement of the Business Combination affected the trading price of GSAH Class A common stock? |
| A: | On June 16, 2021, the trading date before the public announcement of the Business Combination, GSAHs public units, GSAH Class A common stock and warrants closed at $10.43, $10.00 and $1.81, respectively. On August 6, 2021, the most recent practicable date prior to the date of this proxy statement/prospectus, the Companys public units, Class A common stock and warrants closed at $10.45, $9.96 and $1.88, respectively. |
| Q: | Is the Business Combination the first step in a going private transaction? |
| A: | No. The Company does not intend for the Business Combination to be the first step in a going private transaction. One of the primary purposes of the Business Combination is to provide a platform for Mirion to access the U.S. public markets. |
| Q: | Will the management of Mirion change in the Business Combination? |
| A: | We anticipate that all of the executive officers of Mirion will remain with the post-business combination company. |
Additionally, Thomas D. Logan, Lawrence D. Kingsley, Jyothsna (Jo) Natauri, Christopher Warren, Steven Etzel, [●], [●], [●] and [●] have each been nominated to serve as directors of the post-business combination company upon completion of the Business Combination. Please see the sections entitled Proposal No. 5The Director Election Proposal and Management of New Mirion Following the Business Combination for additional information.
| Q: | Will the Company obtain new financing in connection with the Business Combination? |
| A: | Yes. The PIPE Investors have agreed to purchase in the aggregate approximately 90,000,000 shares of GSAH Class A common stock, for approximately $900,000,000 of gross proceeds, in the PIPE Investment. The PIPE Investment is contingent upon, among other things, stockholder approval of the Business Combination Proposal and the closing of the Business Combination. See Proposal No. 1Approval of the Business CombinationRelated AgreementsSubscription Agreements. In addition, a subsidiary of Mirion entered into a debt commitment letter in connection with the Business Combination Agreement. The funds under the debt commitment letter are available to, among other things, (i) fund the Business Combination, (ii) repay and terminate the existing indebtedness of certain subsidiaries of Mirion and (iii) pay all fees, premiums, expenses and other transaction costs incurred in connection with the foregoing. The aggregate commitment of the senior secured credit facilities consists of a $830 million dollar-denominated first lien term facility and a $90 million revolving facility. See Description of New Mirion Indebtedness. |
| Q: | What conditions must be satisfied to complete the Business Combination? |
| A: | The Business Combination Agreement is subject to the satisfaction or waiver of certain customary closing conditions, including, among others, (i) the Minimum Cash Condition; (ii) the registration statement becoming effective in accordance with the Securities Act; (iii) customary bringdown conditions; (iv) no material adverse effect having occurred; and (v) to the extent requested by the Company, Mirion having issued a notice of suspension or termination of business with certain partners. Any of the conditions to the |
21
Table of Contents
| obligations of GSAH, Mirion and the Sellers may be waived in writing by mutual agreement of GSAH, Mirion and the Charterhouse Parties. Any of the conditions to the obligations of GSAH may only be waived in writing by GSAH. In addition, any of the conditions to the obligations of Mirion and the Sellers may only be waived in writing by mutual agreement of Mirion and the Charterhouse Parties. For more information about conditions to the consummation of the Business Combination, see Proposal No. 1Approval of the Business CombinationThe Business Combination Agreement. |
| Q: | When do you expect the Business Combination to be completed? |
| A: | It is currently expected that the Business Combination will be consummated in the second half of 2021. This date depends, among other things, on the approval of the proposals to be put to GSAHs stockholders at the Special Meeting and certain regulatory approvals. However, such meeting could be adjourned if the Adjournment Proposal is adopted by GSAHs Stockholders at the Special Meeting and GSAH elects to adjourn the Special Meeting to a later date or dates, if necessary, to permit further solicitation and vote of proxies in the event there are insufficient votes for, or for any other reason in connection with, the approval of one or more of the other proposals at the Special Meeting. For a description of the conditions for the completion of the Business Combination, see Proposal No. 1Approval of the Business CombinationThe Business Combination Agreement. |
| Q: | What happens if the Business Combination is not consummated? |
| A: | If GSAH is not able to complete the Business Combination with Mirion by July 2, 2022 (or if such date is extended at a duly called meeting of stockholders, such later date) and is not able to complete another business combination by such date, GSAH will (1) cease all operations except for the purpose of winding up, (ii) as promptly as reasonably possible but no more than ten business days thereafter redeem the public shares, at a per-share price, payable in cash, equal to the aggregate amount then on deposit in the trust account including interest but less taxes payable (less up to $100,000 of interest to pay dissolution expenses), divided by the number of then outstanding public shares, which redemption will completely extinguish public stockholders rights as stockholders (including the right to receive further liquidating distributions, if any), and (iii) as promptly as reasonably possible following such redemption, subject to the approval of GSAHs remaining stockholders and GSAHs Board, dissolve and liquidate, subject in each case to GSAHs obligations under Delaware law to provide for claims of creditors and the requirements of other applicable law. |
| Q: | How many votes do I have at the Special Meeting? |
| A: | Our stockholders are entitled to one vote on each proposal presented at the Special Meeting for each share of our common stock held of record as of [●], 2021 the record date for the Special Meeting. As of the close of business on the record date, there were [●] outstanding shares of our common stock. |
| Q: | Did GSAHs Board obtain a third-party valuation or fairness opinion in determining whether or not to proceed with the Business Combination? |
| A: | No. Neither the GSAH Board nor any committee thereof is required to obtain an opinion from an independent investment banking or accounting firm that the price that we are paying for Mirion is fair to us from a financial point of view. Neither the GSAH Board nor any committee thereof obtained a third party valuation in connection with the Business Combination. In analyzing the Business Combination, the GSAH Board conducted due diligence on Mirion and reviewed comparisons of selected financial data of Mirion with certain of its peers in the industry and the financial terms set forth in the Business Combination Agreement. Based on the foregoing, the GSAH Board concluded that the Business Combination was in the best interest of GSAHs stockholders. |
22
Table of Contents
| Q: | Do I have redemption rights? |
| A: | If you are a holder of public shares, you have the right to request that we redeem all or a portion of your public shares for cash provided that you follow the procedures and deadlines described elsewhere in this proxy statement/prospectus. Public stockholders may elect to redeem all or a portion of the public shares held by them regardless of if or how they vote in respect of the Business Combination Proposal or any other proposal set forth herein. If you wish to exercise your redemption rights, see the answer to the next question: How do I exercise my redemption rights? |
Notwithstanding the foregoing, a public stockholder, together with any affiliate of such public stockholder or any other person with whom such public stockholder is acting in concert or as a group (as defined in Section 13(d)(3) of the Exchange Act), will be restricted from redeeming its public shares with respect to more than an aggregate of 15% of the public shares. Accordingly, if a public stockholder, alone or acting in concert or as a group, seeks to redeem more than 15% of the public shares, then any such shares in excess of that 15% limit would not be redeemed for cash. In addition, pursuant to the GSAH Certificate of Incorporation, in no event will we redeem public shares in an amount that would cause our net tangible assets (as determined in accordance with Rule 3a51-1(g)(1) of the Exchange Act) to be less than $5,000,001. In such case, we would not proceed with the redemption of our public shares and the Business Combination, and instead may search for an alternate initial business combination.
The Sponsor and GS Employee Participation have each agreed to waive their redemption rights with respect to all of the shares of our common stock held by them in connection with the consummation of the Business Combination. The founder shares will be excluded from the pro rata calculation used to determine the per-share redemption price.
| Q: | How do I exercise my redemption rights? |
| A: | If you are a public stockholder and wish to exercise your right to redeem the public shares, you must: |
| (1) | (a) hold public shares, or (b) if you hold public shares through units, elect to separate your units into the underlying public shares and warrants prior to exercising your redemption rights with respect to the public shares; |
| (2) | prior to 5:00 p.m. Eastern Time on [●], 2021 (two business days before the scheduled date of the Special Meeting) submit a written request to Continental Trust & Transfer Company N.A., our transfer agent, that we redeem all or a portion of your public shares for cash, affirmatively certifying in your request if you ARE or ARE NOT acting in concert or as a group (as defined in Section 13d-3 of the Exchange Act) with any other stockholder with respect to shares of our common stock at the following address: |
Continental Stock Transfer & Trust Company
1 State Street 30th Floor
New York, New York 10004
Attention: Mark Zimkind
E-Mail: mzimkind@continentalstock.com
; and
| (3) | deliver your public shares either physically or electronically through the Deposit Withdrawal at Custodian (DWAC) system of The Depository Trust Company (DTC) to our transfer agent. |
Notwithstanding the foregoing, a public stockholder, together with any affiliate of such public stockholder or any other person with whom such public stockholder is acting in concert or as a group (as defined in Section 13(d)(3) of the Exchange Act), will be restricted from redeeming its public shares with respect to more than an aggregate of 15% of the public shares. Accordingly, if a public stockholder, alone or acting in concert or as a group, seeks to redeem more than 15% of the public shares, then any such shares in excess of that 15% limit would not be redeemed for cash.
23
Table of Contents
Holders must complete the procedures for electing to redeem their public shares in the manner described above prior to 5:00 p.m., Eastern Time, on [●], 2021 (two business days before the scheduled date of the Special Meeting) in order for their shares to be redeemed.
The address of our transfer agent is listed under the question Who can help answer my questions? below.
Holders of units must elect to separate the units into the underlying public shares and warrants prior to exercising redemption rights with respect to the public shares. If holders hold their units through a broker, bank or other nominee, holders must notify their broker, bank or other nominee that they elect to separate the units into the underlying public shares and warrants, or if a holder holds units registered in its own name, the holder must contact the transfer agent directly and instruct it to do so.
Public stockholders will be entitled to request that their public shares be redeemed for a pro rata portion of the amount then on deposit in the trust account as of two business days prior to the consummation of the Business Combination including interest earned on the funds held in the trust account and not previously released to us (net of taxes payable). For illustrative purposes, as of [●], 2021, this would have amounted to approximately $[●] per outstanding public share. However, the proceeds deposited in the trust account could become subject to the claims of GSAHs creditors, if any, which could have priority over the claims of the public stockholders. Therefore, the per share distribution from the trust account in such a situation may be less than originally expected due to such claims. Whether you vote, and if you do vote irrespective of how you vote, on any proposal, including the Business Combination Proposal, will have no impact on the amount you will receive upon exercise of your redemption rights. It is expected that the funds to be distributed to public stockholders electing to redeem their public shares will be distributed promptly after the consummation of the Business Combination.
Any request for redemption, once made by a holder of public shares, may be withdrawn at any time up to the deadline for exercising redemption requests (being two business days before the scheduled date of the Special Meeting) and thereafter, with our consent, until the consummation of the Business Combination. If you deliver your shares for redemption to our transfer agent, and later decide prior to such deadline not to elect redemption, you may request that our transfer agent return the shares (physically or electronically) to you. You may make such request by contacting our transfer agent at the phone number or address listed at the end of this section.
Any corrected or changed written exercise of redemption rights must be received by our transfer agent prior to the vote taken with respect to the Business Combination Proposal at the Special Meeting (or if we so elect, prior to the consummation of the Business Combination). No request for redemption will be honored unless the holders public shares have been delivered (either physically or electronically) to our transfer agent, at least two business days before the scheduled date of the Special Meeting.
If a holder of public shares properly makes a request for redemption and the public shares are delivered as described above, then, if the Business Combination is consummated, we will redeem the public shares for a pro rata portion of funds deposited in the trust account, calculated as of two business days prior to the consummation of the Business Combination, including interest (net of taxes payable).
If you are a holder of public shares and you exercise your redemption rights, such exercise will not result in the loss of any warrants that you may hold.
| Q: | If I am a holder of units, can I exercise redemption rights with respect to my units? |
| A: | No. Holders of outstanding units must elect to separate the units into the underlying public shares and warrants prior to exercising redemption rights with respect to the public shares. If you hold your units through a broker, bank or other nominee, you must notify your broker, bank or other nominee that you elect to separate the units into the underlying public shares and warrants, or if you hold units registered in your own name, you must contact our transfer agent directly and instruct them to do so. You are requested to cause your public shares to be separated and delivered to our transfer agent, by 5:00 p.m., Eastern Time, on [●], 2021 (two business days before the scheduled date of the Special Meeting) in order to exercise your redemption rights with respect to your public shares. |
24
Table of Contents
| Q: | What are the U.S. federal income tax consequences of exercising my redemption rights? |
| A: | The U.S. federal income tax consequences to a stockholder of exercising its redemption rights will depend on the particular facts and circumstances. Please see the section entitled Proposal No. 1Approval of the Business CombinationUnited States Federal Income Tax Considerations to Stockholders Exercising Redemption Rights. We urge you to consult your tax advisors regarding the tax consequences of exercising your redemption rights. |
| Q: | Can the Sponsor or our officer and directors redeem their founder shares in connection with consummation of the Business Combination? |
| A: | No. The Sponsor and GS Employee Participation have each agreed to waive their redemption rights with respect to all of the founder shares in connection with the consummation of the Business Combination. The founder shares will be excluded from the pro rata calculation used to determine the per-share redemption price. |
| Q: | Is there a limit on the number of shares I may redeem? |
| A: | Yes. A public stockholder, together with any affiliate of such public stockholder or any other person with whom such public stockholder is acting in concert or as a group (as defined in Section 13(d)(3) of the Exchange Act), will be restricted from redeeming its public shares with respect to more than an aggregate of 15% of the public shares. Accordingly, if a public stockholder, alone or acting in concert or as a group, seeks to redeem more than 15% of the public shares, then any such shares in excess of that 15% limit would not be redeemed for cash. However, in no event is your ability to vote all of your shares (including those shares held by you or by a group in excess of 15% of the shares sold in our IPO) for or against our Business Combination restricted. |
| Q: | Is there a limit on the total number of shares that may be redeemed? |
| A: | Yes. Pursuant to the GSAH Certificate of Incorporation, in no event will we redeem public shares in an amount that would cause our net tangible assets (as determined in accordance with Rule 3a51-1(g)(1) of the Exchange Act) to be less than $5,000,001. In such case, we would not proceed with the redemption of our public shares and the Business Combination, and instead may search for an alternate initial business combination. |
| Q: | Will how I vote affect my ability to exercise redemption rights? |
| A: | No. Stockholders may elect to redeem all or a portion of the public shares held by them regardless of if or how they vote in respect of the Business Combination Proposal or any other proposal set forth herein. As a result, the Business Combination Agreement can be approved by stockholders who will redeem their shares and no longer remain stockholders, leaving stockholders who choose not to redeem their shares holding shares in a company with a potentially less-liquid trading market, fewer stockholders, potentially less cash and the potential inability to meet the listing standards of the NYSE. |
| Q: | If I am a Company warrant holder, can I exercise redemption rights with respect to my warrants? |
| A: | No. The holders of our warrants have no redemption rights with respect to our warrants. |
| Q: | Do I have appraisal rights in connection with the proposed Business Combination? |
| A: | No. Neither our stockholders nor our warrant holders have appraisal rights in connection with the Business Combination under the DGCL. |
25
Table of Contents
| Q: | What happens to the funds deposited in the trust account after consummation of the Business Combination? |
| A: | Following the closing of the IPO, an amount equal to $750 million ($10.00 per unit) of the net proceeds from the IPO and the sale of the private placement warrants was placed in the trust account. As of [●], 2021, funds in the trust account totaled $[●], including $[●] of accrued dividends. These funds will remain in the trust account, except for the withdrawal of interest to pay taxes, if any, until the earliest of (1) the completion of a business combination (including the closing of the Business Combination), (2) the redemption of any public shares properly tendered in connection with a stockholder vote to amend the GSAH Certificate of Incorporation to modify the substance or timing of GSAHs obligation to redeem 100% of its public shares if it does not complete its initial business combination by July 2, 2022; and (3) the redemption of all of the public shares if GSAH is unable to complete its initial business combination by July 2, 2022 (or if such date is extended at a duly called meeting of stockholders, such later date), subject to applicable law. |
Upon consummation of the Business Combination, the funds deposited in the trust account will be released to pay holders of our public shares who properly exercise their redemption rights; to pay a portion of the Cash Consideration and transaction fees and expenses associated with the Business Combination; and for working capital and general corporate purposes of the post-business combination company. See Summary of the Proxy Statement/ProspectusSources and Uses of Funds for the Business Combination.
| Q: | What do I need to do now? |
| A: | You are urged to read this proxy statement/prospectus, including the Annexes and the accompanying financial statements of the Company and Mirion, carefully and in its entirety and to consider how the Business Combination will affect you as a stockholder or warrant holder. Our stockholders should then vote as soon as possible in accordance with the instructions provided in this proxy statement/prospectus and on the enclosed proxy card. |
| Q: | How do I vote? |
| A: | If you are a holder of record of shares of our common stock on the record date for the Special Meeting, you may vote during the Special Meeting via the meeting website or by submitting a proxy for the Special Meeting. You may submit your proxy by completing, signing, dating and returning the enclosed proxy card in the accompanying pre-addressed postage-paid envelope. If you hold your shares in street name, which means your shares are held of record by a broker, bank or other nominee, you should contact your broker, bank or other nominee to ensure that votes related to the shares you beneficially own are properly counted. In this regard, you must provide the broker, bank or other nominee with instructions on how to vote your shares or, if you wish to attend the Special Meeting and vote via the special meeting website, obtain a valid proxy from your broker, bank or other nominee. |
| Q: | If my shares are held in street name, will my broker, bank or other nominee automatically vote my shares for me? |
| A: | No. If your shares are held in a stock brokerage account or by a bank or other nominee, you are considered the beneficial holder of the shares held for you in what is known as street name. If this is the case, this proxy statement/prospectus may have been forwarded to you by your broker, bank or other nominee, or its agent, and you may need to obtain a proxy form from the institution that holds your shares and follow the instructions included on that form regarding how to instruct your broker, bank or other nominee as to how to vote your shares. Under the rules of various national and regional securities exchanges, your broker, bank or other nominee cannot vote your shares with respect to non-discretionary matters unless you provide instructions on how to vote in accordance with the information and procedures provided to you by your |
26
Table of Contents
| broker, bank or other nominee. We believe all of the proposals presented to our stockholders will be considered non-discretionary and therefore your broker, bank or other nominee will not vote your shares without your instruction. Your broker, bank or other nominee can vote your shares only if you provide instructions on how to vote. As the beneficial holder, you have the right to direct your broker, bank or other nominee as to how to vote your shares and you should instruct your broker, bank or other nominee to vote your shares in accordance with directions you provide. If you do not provide voting instructions to your broker, bank or other nominee on a particular proposal on which your broker, bank or other nominee does not have discretionary authority to vote, your shares will not be voted on that proposal. This is called a broker non-vote. An abstention will be counted towards the quorum requirement for each of the proposals presented at the Special Meeting but a broker non-vote will not. In connection with (i) the Business Combination Proposal and the Adjournment Proposal, abstentions and broker non-votes will have no effect, (ii) the NYSE Proposal and the Incentive Plan Proposal, abstentions will be counted as a vote cast at the Special Meeting and will have the same effect as a vote AGAINST the proposal but broker non-votes will have no effect and (iii) the Charter Proposal, abstentions and broker non-votes will have the same effect as voting AGAINST the proposal. |
| Q: | Who is entitled to vote at the Special Meeting? |
| A: | GSAH has fixed [●], 2021 as the record date for the Special Meeting. If you were a stockholder of GSAH at the close of business on the record date, you are entitled to vote on matters that come before the Special Meeting. However, a stockholder may only vote his, her or its shares if he, she or it is present at the Special Meeting by attendance via the virtual meeting website or is represented by proxy at the Special Meeting. |
| Q: | What happens if I sell my shares of common stock before the Special Meeting? |
| A: | The record date for the Special Meeting is earlier than the date of the Special Meeting and earlier than the date that the Business Combination is expected to be completed. If you transfer your shares of common stock after the applicable record date, but before the Special Meeting, unless you grant a proxy to the transferee, you will retain your right to vote at the Special Meeting with respect to such shares but the transferee, and not you, will have the ability to redeem such shares (if time permits). |
| Q: | What constitutes a quorum at the Special Meeting? |
| A: | A majority of the outstanding shares of our common stock entitled to vote as of the record date at the Special Meeting must be present by attendance via the virtual meeting website or represented by proxy, at the Special Meeting to constitute a quorum and in order to conduct business at the Special Meeting. Abstentions will be counted as present for purposes of determining a quorum but broker non-votes will not. Our Initial Stockholders, who currently own approximately 20% of our outstanding shares of common stock, will count towards this quorum. In the absence of a quorum, the chairman of the Special Meeting has power to adjourn the Special Meeting. Based on the number of outstanding shares of our common stock as of the record date for the Special Meeting, [●] shares of our common stock will be required to achieve a quorum at the Special Meeting. |
| Q: | What vote is required to approve each proposal at the Special Meeting? |
| A: | The following votes are required for each proposal at the Special Meeting: |
| | The approval of each of the Business Combination Proposal, the Governance Proposal, the Incentive Plan Proposal and the Adjournment Proposal requires the affirmative vote of a majority of the votes cast by holders of our outstanding shares of common stock represented by attendance via the virtual meeting website or by proxy and entitled to vote thereon at the Special Meeting. Generally, only votes FOR or AGAINST are considered to be votes cast. Accordingly, a Company stockholders failure to vote by proxy or to vote during |
27
Table of Contents
| the Special Meeting, as well as an abstention from voting and a broker non-vote with regard to the such proposals, will have no effect on such proposals. Abstentions will be counted in connection with the determination of whether a valid quorum is established but will have no effect on such proposals. |
| | NYSE Proposal: The approval of the NYSE Proposal requires the affirmative vote of holders of a majority of the votes cast by holders of our outstanding shares of common stock represented at the Special Meeting via the virtual website or by proxy and entitled to vote thereon at the Special Meeting. A Company stockholders failure to vote by proxy or to vote during the Special Meeting and a broker non-vote with regard to the NYSE Proposal will have no effect on the NYSE Proposal. The NYSE considers abstentions to be votes cast and included in the number of shares of which a majority is required to vote in favor. Accordingly, abstentions will have the same effect as a vote AGAINST the NYSE Proposal. |
| | Charter Proposal: The approval of the Charter Proposal requires the affirmative vote of holders of a majority of our outstanding shares of common stock entitled to vote thereon at the Special Meeting. Accordingly, a Company stockholders failure to vote by proxy or to vote during the Special Meeting, as well as an abstention from voting and a broker non-vote with regard to the Charter Proposal will have the same effect as a vote AGAINST the Charter Proposal. |
| | Director Election Proposal: Directors are elected by a plurality of all of the votes cast by holders of shares of common stock represented at the Special Meeting via the virtual meeting website or by proxy and entitled to vote thereon at the Special Meeting. This means that the nine director nominees who receive the most affirmative votes will be elected to serve as directors following the closing of the Business Combination. Stockholders may not cumulate their votes with respect to the election of directors. Assuming a valid quorum is established, abstentions and broker non-votes will have no effect on the election of directors. |
See How does the Initial Stockholders intend to vote their shares? for information on how the Initial Stockholders intend to vote their shares.
| Q: | Why is GSAH proposing the governance proposal? |
| A: | As required by applicable SEC guidance, GSAH is requesting that its stockholders vote upon, on a non-binding advisory basis, a proposal to approve certain governance provisions contained in the New Mirion Charter that materially affect stockholder rights. This separate vote is not otherwise required by Delaware law separate and apart from the Charter Proposal, but pursuant to SEC guidance, GSAH is required to submit these provisions to its stockholders separately for approval. However, the stockholder vote regarding this proposal is an advisory vote, and is not binding on GSAH and the Board (separate and apart from the approval of the Charter Proposal). Furthermore, the Business Combination is not conditioned on the separate approval of the governance proposal (separate and apart from approval of the Charter Proposal). Please see the section entitled Proposal No. 4The Governance Proposal. |
| Q: | What are the recommendations of GSAHs Board? |
| A: | GSAHs Board believes that the Business Combination Proposal and the other proposals to be presented at the Special Meeting are in the best interest of GSAHs stockholders and unanimously recommends that its stockholders vote FOR the Business Combination Proposal, FOR the NYSE Proposal, FOR the Charter Proposal, FOR the Governance Proposal, FOR each of the director nominees set forth in the Director Election Proposal, FOR the Incentive Plan Proposal and FOR the Adjournment Proposal, in each case, if presented to the Special Meeting. |
The existence of financial and personal interests of one or more of GSAHs directors may result in a conflict of interest on the part of such director(s) between what he or they may believe is in the best interests of GSAH and its stockholders and what he or they may believe is best for himself or themselves in determining to recommend that stockholders vote for the proposals. See the section entitled Proposal No. 1Approval of the Business CombinationGSAHs Board of Directors Reasons for the Approval of the Business Combination for a further discussion of these considerations.
28
Table of Contents
| Q: | How do the Initial Stockholders intend to vote their shares? |
| A: | The Sponsor and GS Employee Participation have each agreed to, among other things, vote in favor of the Business Combination Proposal and the other proposals described herein to be presented at the Special Meeting. As of the date of this proxy statement/prospectus, the Initial Stockholders own approximately 20% of the outstanding shares of our common stock. |
At any time at or prior to the Business Combination, subject to applicable securities laws (including with respect to material nonpublic information), the Sponsor, Mirion Stockholder or our or their respective directors, officers, advisors or respective affiliates may (1) purchase public shares from institutional and other investors who vote, or indicate an intention to vote, against any of the Condition Precedent Proposals, or elect to redeem, or indicate an intention to redeem, public shares, (2) execute agreements to purchase such shares from such investors in the future, or (3) enter into transactions with such investors and others to provide them with incentives to acquire public shares, vote their public shares in favor of the Condition Precedent Proposals or not redeem their public shares. Such a purchase may include a contractual acknowledgement that such stockholder, although still the record holder of GSAHs shares, is no longer the beneficial owner thereof and therefore agrees not to exercise its redemption rights. In the event that the Sponsor, Mirion stockholder or our or their respective directors, officers, advisors or respective affiliates purchase shares in privately negotiated transactions from public stockholders who have already elected to exercise their redemption rights, such selling stockholder would be required to revoke their prior elections to redeem their shares. The purpose of such share purchases and other transactions would be to (1) increase the likelihood of approving the Condition Precedent Proposals and (2) limit the number of public shares electing to redeem, including to satisfy any redemption threshold.
Entering into any such arrangements may have an adverse effect on the market price of our common stock (e.g., by giving an investor or holder the ability to effectively purchase shares at a price lower than market, such investor or holder may therefore become more likely to sell the shares he or she owns, either at or prior to the Business Combination). If such transactions are effected, the consequence could be to cause the Business Combination to be consummated in circumstances where such consummation could not otherwise occur. Purchases of shares by the persons described above would allow them to exert more influence over the approval of the proposals to be presented at the Special Meeting and would likely increase the chances that such proposals would be approved. We will file or submit a Current Report on Form 8-K to disclose any material arrangements entered into or significant purchases made by any of the aforementioned persons that would affect the vote on the proposals to be put to the Special Meeting or the redemption threshold. Any such report will include descriptions of any arrangements entered into or significant purchases by any of the aforementioned persons.
| Q: | May I change my vote after I have mailed my signed proxy card? |
| A: | Yes. GSAH stockholders may send a later-dated, signed proxy card to GSAHs Secretary at GSAHs address set forth below so that it is received by GSAHs Secretary prior to the vote at the Special Meeting (which is scheduled to take place on [●], 2021) or attend the Special Meeting via the virtual Special Meeting website and vote. GSAH stockholders also may revoke their proxy by sending a notice of revocation to GSAHs Secretary, which must be received by GSAHs Secretary prior to the vote at the Special Meeting. However, if your shares are held in street name by your broker, bank or other nominee, you must contact your broker, bank or other nominee to change your vote. |
| Q: | If I am not going to attend the Special Meeting via the virtual Special Meeting website, should I return my proxy card instead? |
| A: | Yes. Whether you plan to attend the Special Meeting or not, please read the enclosed proxy statement/prospectus carefully and in its entirety, and vote your shares by completing, signing, dating and returning the enclosed proxy card in the postage-paid envelope provided. |
29
Table of Contents
| Q: | What will happen if I sign and return my proxy card without indicating how I wish to vote? |
| A: | If you sign and return your proxy card without indicating how you wish to vote, your proxy will be voted FOR each of the proposals presented at the Special Meeting. |
| Q: | What happens if I fail to take any action with respect to the Special Meeting? |
| A: | If you fail to take any action with respect to the Special Meeting and the Business Combination is approved by stockholders and the Business Combination is consummated, you will become a stockholder or warrant holder of the post-business combination company, as applicable. If you fail to take any action with respect to the Special Meeting and the Business Combination is not approved, you will remain a stockholder or warrant holder of GSAH, as applicable. However, if you fail to vote with respect to the Special Meeting, you will nonetheless be able to elect to redeem your public shares in connection with the Business Combination (if time permits). |
| Q: | What should I do with my stock certificates, warrant certificates or unit certificates? |
| A: | Our stockholders who exercise their redemption rights must deliver (either physically or electronically) their stock certificates to our transfer agent prior to the Special Meeting. |
Holders must complete the procedures for electing to redeem their public shares in the manner described above prior to 5:00 p.m., Eastern Time, on [●], 2021 (two business days before the scheduled date of the Special Meeting) in order for their shares to be redeemed.
Our warrant holders should not submit the certificates relating to their warrants. Public stockholders who do not elect to have their public shares redeemed for the pro rata share of the trust account should not submit the stock certificates relating to their public shares.
| Q: | What should I do if I receive more than one set of voting materials? |
| A: | GSAH stockholders may receive more than one set of voting materials, including multiple copies of this proxy statement/prospectus and multiple proxy cards or voting instruction cards. For example, if you hold your shares in more than one brokerage account, you will receive a separate voting instruction card for each brokerage account in which you hold shares. If you are a holder of record and your shares are registered in more than one name, you will receive more than one proxy card. Please complete, sign, date and return each proxy card and voting instruction card that you receive in order to cast a vote with respect to all of your shares of our common stock. |
| Q: | Who will solicit and pay the cost of soliciting proxies for the Special Meeting? |
| A: | We will pay the cost of soliciting proxies for the Special Meeting. GSAH has engaged Innisfree M&A Incorporated (Innisfree) to assist in the solicitation of proxies for the Special Meeting. GSAH has agreed to pay Innisfree a fee of $40,000, plus disbursements. GSAH will reimburse Innisfree for reasonable out-of-pocket expenses and will indemnify Innisfree and its affiliates against certain claims, liabilities, losses, damages and expenses. GSAH will also reimburse banks, brokers and other custodians, nominees and fiduciaries representing beneficial owners of our common stock for their expenses in forwarding soliciting materials to beneficial owners of our common stock and in obtaining voting instructions from those owners. GSAHs management team may also solicit proxies by telephone, by facsimile, by mail, on the Internet or during the Special Meeting via the Special Meeting website. They will not be paid any additional amounts for soliciting proxies. |
| Q: | Who can help answer my questions? |
| A: | If you have questions about the proposals or the Business Combination or if you need additional copies of the proxy statement/prospectus or the enclosed proxy card, you should contact: |
30
Table of Contents

Innisfree M&A Incorporated (Innisfree)
501 Madison Avenue, 20th Floor
New York, NY 10022
Shareholders may call toll free: (877) 456-3463
Banks and Brokers may call collect: (212) 750-5833
You also may obtain additional information about GSAH from documents filed with the SEC by following the instructions in the section entitled Where You Can Find More Information. If you are a holder of public shares and you intend to seek redemption of your public shares, you will need to send a letter demanding redemption and deliver your public shares (either physically or electronically through DTCs DWAC System) to Continental Stock Transfer & Trust Company, N.A., our transfer agent, at the address below prior to the Special Meeting. Holders must complete the procedures for electing to redeem their public shares in the manner described above prior to 5:00 p.m., Eastern Time, on [●], 2021 (two business days before the scheduled date of the Special Meeting) in order for their shares to be redeemed. If you have questions regarding the certification of your position or delivery of your stock, please contact Continental Stock Transfer & Trust Company, N.A.:
Continental Stock Transfer & Trust Company
1 State Street 30th Floor
New York, New York 10004
Attention: Mark Zimkind
E-Mail: mzimkind@continentalstock.com
31
Table of Contents
SUMMARY OF THE PROXY STATEMENT/PROSPECTUS
This summary highlights selected information from this proxy statement/prospectus and does not contain all of the information that is important to you. To better understand the proposals to be submitted for a vote at the Special Meeting, including the Business Combination, you should read this proxy statement/prospectus, including the Annexes and the accompanying financial statements of the Company and Mirion, carefully and in its entirety. The Business Combination Agreement, a copy of which is attached to this proxy statement/prospectus as Annex A, is the primary legal document that governs the Business Combination and the other transactions that will be undertaken in connection with the Business Combination. The Business Combination Agreement is also described in detail in this proxy statement/prospectus in the section entitled Proposal No. 1Approval of the Business Combination.
Unless otherwise specified, all share calculations (i) assume that no public shares are elected to be redeemed in connection with the Business Combination, (ii) exclude the 18,750,000 outstanding founder shares held by the GS Sponsor (all of which are subject to the performance vesting conditions and forfeiture as described herein), (iii) assume no exercise of any warrants to purchase GSAH Class A common stock that will remain outstanding immediately following the Business Combination, (iv) exclude the issuance of any shares under the Incentive Plan following the Business Combination and (v) assume that only members of Mirions management team will elect to receive shares of GSAH Class B common stock and that the other Sellers receiving shares of GSAH common stock will elect to receive shares of GSAH Class A common stock. If the actual facts are different from these assumptions, the share calculations, including ownership interest of the post-business combination company, will be different.
Parties to the Business Combination
The Company
GS Acquisition Holdings Corp II is a blank check company incorporated as a Delaware corporation on May 31, 2018, and formed for the purpose of effecting a merger, capital stock exchange, asset acquisition, stock purchase, reorganization or similar business combination with one or more businesses. GSAH has neither engaged in any operations nor generated any operating revenue to date. Based on GSAHs business activities, it is a shell company as defined under the Exchange Act because it has no operations and nominal assets consisting almost entirely of cash.
On July 2, 2020, GSAH completed its IPO of 75,000,000 units, including 5,000,000 units issued pursuant to the partial exercise by the underwriters of their option to purchase additional units in full, at a price of $10.00 per unit, generating proceeds to us of $750,000,000 before underwriting discounts and expenses. Each unit consisted of one share of GSAH Class A common stock and one-quarter of one redeemable warrant, with each whole warrant exercisable for one share of GSAH Class A common stock at a price of $11.50 per share. Simultaneously with the closing of the IPO, GSAH closed the private placement of an aggregate of 8,500,000 warrants, each exercisable to purchase one share of GSAH Class A common stock at an exercise price of $11.50 per share, to the Sponsor, at a price of $2.00 per private placement warrant, generating proceeds of $17,000,000. Each warrant sold in the IPO and the private placement will become exercisable 30 days after the completion of our initial business combination, and will expire at 5:00 p.m., New York City time, five years after the completion of our initial business combination or earlier upon redemption or liquidation. Subject to the terms and conditions contained in the warrant agreement, the warrants may be redeemed either for cash once they become exercisable or for shares of GSAH Class A common stock commencing 90 days after they become exercisable.
On the closing date of the IPO, GSAH placed $750,000,000 of proceeds (including $26,250,000 of deferred underwriting discount) from the IPO and the sale of the private placement warrants into the trust account. The proceeds held in the trust account may only be invested in United States government securities within the meaning of Section 2(a)(16) of the Investment Company Act having a maturity of 180 days or less or in money
32
Table of Contents
market funds meeting certain conditions under Rule 2a-7 promulgated under the Investment Company Act which invest only in direct U.S. government treasury obligations. These funds will remain in the trust account, except for the withdrawal of interest to pay taxes, if any, until the earliest of (1) the completion of a business combination (including the closing of the Business Combination), (2) the redemption of any public shares properly tendered in connection with a stockholder vote to amend the GSAH Certificate of Incorporation to modify the substance or timing of GSAHs obligation to redeem 100% of its public shares if it does not complete its initial business combination by July 2, 2022; and (3) the redemption of all of the public shares if GSAH is unable to complete its initial business combination by July 2, 2022 (or if such date is extended at a duly called meeting of stockholders, such later date), subject to applicable law.
GSAHs publicly-traded Class A common stock, units and warrants are currently listed on the NYSE under the symbols GSAH, GSAH.U and GSAH WS, respectively. We intend to apply to continue the listing of GSAH Class A common stock and warrants on the NYSE under the symbols MIR, and MIR.WS, respectively, upon the closing of the Business Combination. At the closing of the Business Combination, each unit will separate into its components consisting of one share of GSAH Class A common stock and one-fourth of one warrant.
GSAHs principal executive office is located at 200 West Street, New York, New York 10282 and its telephone number is (212) 902-1000. GSAHs corporate website address is www.GSacquisition.com. GSAHs website and the information contained on, or that can be accessed through, the website is not deemed to be incorporated by reference in, and is not considered part of, this proxy statement/prospectus.
Mirion
Mirion provides products, services and software that allow its customers to safely leverage the power of ionizing radiation for the greater good of humanity. Mirions solutions have critical applications in the medical, nuclear energy and defense markets, as well as in laboratories and scientific research, analysis and space exploration. Many of Mirions markets are characterized by the need to meet rigorous regulatory standards, design qualifications and operating requirements. Throughout Mirions history, Mirion has successfully leveraged the strength of its expertise in ionizing radiation to continually drive innovation and expand the commercial applications of its core technology competencies. Through its facilities in 12 countries, Mirion supplies its solutions in the Americas, Europe, Africa, the Middle East and Asia Pacific regions. As of March 31, 2021 and June 30, 2020, Mirion had an accumulated deficit of $860.6 million and $729.7 million, respectively. For the nine months ended March 31, 2021 Mirion experienced a net loss of $131.0 million.
Mirions principal executive office is located at 1218 Menlo Drive, Atlanta, GA 30318
For more information about Mirion see Information about Mirion.
The following is a summary of the proposals to be put to GSAH stockholders at the Special Meeting and certain transactions contemplated by the Business Combination Agreement. Each of the proposals below, except the Adjournment Proposal, is cross-conditioned on the approval of each other. The Adjournment Proposal is not conditioned upon the approval of any other proposal set forth in this proxy statement/prospectus. The transactions contemplated by the Business Combination Agreement will be consummated only if the Condition Precedent Proposals are approved at the Special Meeting.
Business Combination Proposal
As discussed in this proxy statement/prospectus, we are asking our stockholders to approve and adopt the Business Combination Agreement, dated as of June 17, 2021, by and among GSAH, Mirion, and the Sellers, a copy of which is attached to this proxy statement/prospectus as Annex A. The Business Combination Agreement provides that, among other things, the parties thereto will enter into the Business Combination pursuant to which Mirion will combine with a subsidiary of the Company. Proposal No. 1Approval of the Business Combination.
33
Table of Contents
Structure of the Transactions
On June 17, 2021, the Company, Mirion, for the limited purpose set forth therein, the Charterhouse Parties, for the limited purpose set forth therein, the other Supporting Mirion Holders and, for the limited purpose set forth therein, the other holders of Existing Mirion Shares from time to time becoming a party thereto by executing a Joinder Agreement, entered into the Business Combination Agreement, a copy of which is attached to this proxy statement/prospectus as Annex A.
Pursuant to the terms of the Business Combination Agreement, the parties thereto will enter into a business combination transaction pursuant to which Mirion will combine with a subsidiary of the Company.
The proposed Business Combination will establish the Company as the corporate parent of Mirion. At the Closing, in order to implement a structure similar to that of an Up-C, the Company will establish a Delaware corporation (IntermediateCo) as a subsidiary of the Company that will merge with and into Mirion TopCo with Mirion TopCo surviving. Sellers will be permitted to elect to receive equity consideration either in the form of (i) shares of GSAH Class A common stock or (ii) paired interests consisting of shares of Company Class B common stock paired together with shares of IntermediateCo Class B common stock.
We expect the Mirion Sellers (excluding members of Mirion management) to elect to receive shares of GSAH Class A Common Stock and members of Mirion management to elect to receive shares of GSAH Class B Common Stock.
Holders of shares of IntermediateCo Class B common stock will have a redemption right for such shares to be settled, at the option of the Company, with (i) shares of Company Class A common stock (on a one-for-one basis) or (ii) a cash amount per share based on an average trailing stock price of Company Class A common stock. In the event of a redemption request by a holder of shares of IntermediateCo Class B common stock, the Company may, at its option, effect a direct exchange of cash or Company Class A common stock for shares of IntermediateCo Class B common stock in lieu of such a redemption. Upon redemption or exchange of shares of IntermediateCo Class B common stock, the corresponding shares of Company Class B common stock will be cancelled. The Company will hold 100% of the voting shares of IntermediateCo Class A common stock and greater than 80% of the shares of IntermediateCo Class B common stock. As further described in the New Mirion Charter to be in effect after the Closing, each holder of Company Class B common stock will be entitled to one vote for each share of Company Class B common stock held of record by such holder on all matters on which stockholders generally are entitled to vote and the holders of Company Class B common stock shall not be entitled to dividends of cash or property on such shares of Company Class B common stock. See Description of New Mirion Securities.
The structure of the Business Combination and the resulting combined enterprise was selected because it facilitated multiple goals for the Business Combination in a tax-efficient manner i.e., it permits GSAH stockholders, GSAH warrant holders and shareholders of Mirion TopCo, including members of Mirion management, in each case, who are U.S. taxpayers to participate in the Business Combination while deferring U.S. taxation until such time as the applicable investor exits its investment in the combined enterprise. For example, the structure enables the public company to exercise full control over the Mirion operating entities and to file consolidated U.S. federal income tax returns with Mirions U.S. operating subsidiaries. It also affords the shareholders of GSAH Class A common stock and GSAH Class B common stock voting power commensurate with their economic interests in combined enterprise. As it pertains to members of Mirion management who are U.S. taxpayers, the ability of Mirion TopCo shareholders to elect to receive shares of GSAH Class B Common Stock is expected to facilitate their tax-efficient participation in the Business Combination. The structure does not provide other advantages particular to members of Mirion management and does not advantage or disadvantage other Mirion TopCo shareholders, who are not U.S. taxpayers. Moreover, the structure does not advantage or disadvantage any GSAH stockholders, GSAH warrant holders or the PIPE Investors, beyond permitting those who are U.S. taxpayers to participate in the Business Combination in a tax-efficient manner.
34
Table of Contents
The following diagram illustrates the ownership structure of GSAH immediately following the Business Combination (percentages shown as basic ownership of common stock) and based on the assumptions described under Ownership of the Company Following the Business Combination and assuming that no GSAH public stockholder exercises redemption rights with respect to its shares for a pro rata portion of the funds in GSAHs trust account:
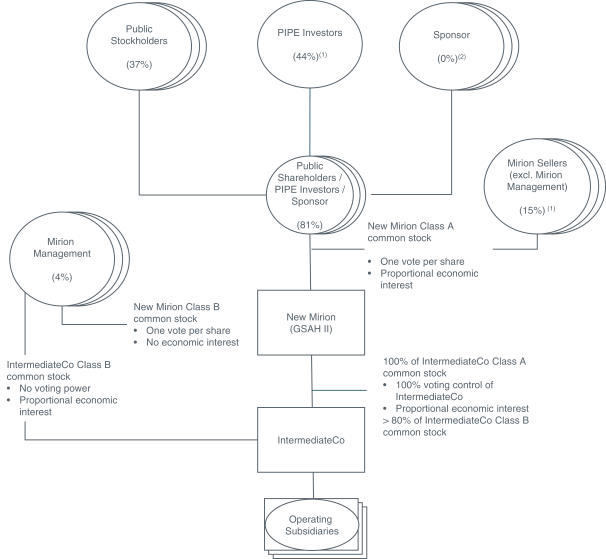
| (1) | Includes 20,000,000 GSAH Class A shares subscribed for by GSAM Holdings, assuming no syndication of its subscription. Neither the Backstop Agreement nor the Option Agreement is exercisable in the no redemptions scenario as shown above because there will be not be a Cash Shortfall. See Related AgreementsBackstop Agreement and Related AgreementsOption Agreement. |
| (2) | Excludes 18,750,000 founder shares that convert from shares of GSAH Class B common stock to shares of GSAH Class A common stock upon the closing of the Business Combination and are subject to certain vesting and forfeiture conditions described elsewhere in this prospectus. |
35
Table of Contents
Consideration to the Sellers in the Business Combination
Subject to the terms of the Business Combination Agreement and adjustments set forth therein, the consideration to be paid in connection with the Business Combination is $1,700,000,000 (the Total Consideration ) and will be paid in a combination of equity and cash consideration. The cash consideration will be an amount equal to $1,310,000,000; provided, that if the Minimum Cash Condition is not met, and Mirion and the Charterhouse Parties elect to waive the Minimum Cash Condition, then the cash consideration will be equal to $1,310,000,000 less the amount by which $1,310,000,000 exceeds the Available Closing Cash. In exchange for the A Ordinary Shares of $0.01 each in the capital of Mirion, the B Ordinary Shares of $0.01 each in the capital of Mirion and certain loan notes due 2026 issued by Mirion Technologies (HoldingSub1), Ltd, each Seller may elect to receive cash or equity consideration or a combination thereof, which equity consideration shall be in the form of either shares of GSAHs Class A common stock or shares of GSAHs Class B common stock combined with shares of Class B common stock of a to be formed Delaware corporation that will be majority owned and controlled by GSAH. The Available Closing Cash will be an amount equal to (i) the amount of funds contained in GSAHs trust account (after reduction for the aggregate amount of payments required to be made in connection with any valid stockholder redemptions), plus (ii) the aggregate amount of cash that has been funded to and remains with GSAH pursuant to the Subscription Agreements as of immediately prior to the closing of the Business Combination (the Closing), plus (iii) the amounts delivered pursuant to the Debt Financing (as defined in the Agreement), plus (iv) the cash and cash equivalents of Mirion and its subsidiaries on a consolidated basis as of the date of the Closing (the Closing Date), plus (v) the proceeds, if any, from the sale by GSAH to GSAM Holdings LLC (GSAM Holdings) of shares of the GSAH Class A common stock, pursuant to the Backstop Agreement, less (vi) the total amount required to be paid to fully satisfy all obligations related to Mirions credit agreement as of the Closing Date, less (vii) certain transaction expenses, less (viii) $50,000,000 (collectively, the Available Closing Cash).
Mirion Material Adverse Effect
Under the Business Combination Agreement, certain representations and warranties of Mirion are qualified in whole or in part by a material adverse effect standard for purposes of determining whether a breach of such representations and warranties has occurred. In addition, it is a condition to the performance of GSAHs obligations to close the transactions contemplated by the Business Combination Agreement that no Material Adverse Effect occurs between signing and Closing. Pursuant to the Business Combination Agreement, a Material Adverse Effect means any state of facts, development, effect, change, circumstance, event or occurrence, that, individually or in the aggregate, has had, or would reasonably be expected to have, a material adverse effect on the business, assets, results of operations or financial condition of Mirion and its subsidiaries, taken as a whole; provided, however, that in no event would any of the following (or the effect of any of the following), alone or in combination, be deemed to constitute a Material Adverse Effect or be taken into account in determining whether a Material Adverse Effect has occurred or would reasonably be expected to occur: (i) any change in law or GAAP or any interpretation or enforcement thereof, (ii) any change in interest rates or economic, political, business, financial, commodity, currency or market conditions generally, (iii) any national or international political or social conditions in countries in which, or in the proximate geographic region in which, Mirion or any of its subsidiaries operates, including the engagement by the United States or such other countries in hostilities or the escalation thereof, whether or not pursuant to the declaration of a national emergency or war, or the occurrence or the escalation of any military or terrorist attack upon the United States or such other country, or any territories, possessions, or diplomatic or consular offices of the United States or such other countries or upon any military installation, equipment or personnel of the United States or such other country, (iv) any earthquake, hurricane, tsunami, tornado, flood, mudslide, wild fire or other natural disaster, act of God or other force majeure event, (v) any change generally affecting any of the industries or markets in which Mirion or any of its subsidiaries operates, (vi) the announcement or execution of the Business Combination Agreement, or the pendency, performance or consummation of the transactions contemplated thereby, including the impact thereof on relationships, contractual or otherwise, with customers, suppliers, distributors, partners, licensors, providers or employees, (vii) the taking of any action required by the terms of Business Combination
36
Table of Contents
Agreement or with the prior written consent of GSAH, (viii) any failure of Mirion and its subsidiaries, taken as a whole, to meet any projections, forecasts or budgets (provided that clause (viii) shall not prevent or otherwise affect a determination that any change or effect underlying such failure to meet projections or forecasts has resulted in, or contributed to, or would reasonably be expected to result in, a Material Adverse Effect, to the extent that such change or effect is not otherwise excluded from this definition of Material Adverse Effect) and (ix) COVID-19 or any other epidemic, pandemic or disease outbreak, or any law, directive, pronouncement or guideline issued by a governmental authority, the Centers for Disease Control and Prevention (or similar national or international organization), the World Health Organization or industry group providing for business closures, changes to business operations, sheltering-in-place or other restrictions that relate to, or arise out of, an epidemic, pandemic or disease outbreak (including the COVID-19 pandemic) or any change in such law, directive, pronouncement or guideline or interpretation thereof following the date of the Business Combination Agreement or Mirions or any of its subsidiaries compliance therewith; provided that, in the case of clauses (i)-(v) and (ix), such changes may be taken into account to the extent that such changes have had a disproportionate impact on Mirion and its subsidiaries, taken as a whole, as compared to other competitors or similarly situated companies operating in the industries or markets in which the Mirion and its subsidiaries operate.
Seller Material Adverse Effect
Under the Business Combination Agreement, certain representations and warranties of the Sellers are qualified in whole or in part by a material adverse effect standard for purposes of determining whether a breach of such representations and warranties has occurred. Pursuant to the Business Combination Agreement, a Seller Material Adverse Effect means, with respect to the Sellers, any state of facts, development, effect, change, circumstance, event or occurrence that, individually or in the aggregate, has prevented, materially delayed or materially impaired, or would reasonably be expected to prevent, materially delay or materially impair, the ability of the Sellers, considered as a group, to consummate the transactions contemplated by the Business Combination Agreement.
SPAC Material Adverse Effect
Under the Business Combination Agreement, certain representations and warranties of GSAH are qualified in whole or in part by a material adverse effect standard for purposes of determining whether a breach of such representations and warranties has occurred. In addition, it is a condition to the performance of Mirion and the Sellers obligations to close the transactions contemplated by the Business Combination Agreement that no SPAC Material Adverse Effect occurs between signing and Closing. Pursuant to the Business Combination Agreement, a SPAC Material Adverse Effect means, with respect to the SPAC, any state of facts, development, effect, change, circumstance, event or occurrence that, individually or in the aggregate, has prevented, materially delayed or materially impaired, or would reasonably be expected to prevent, materially delay or materially impair, the ability of the SPAC to consummate the transactions contemplated by the Business Combination Agreement.
Closing of the Business Combination
The Closing of the Business Combination is expected to occur by electronic exchange of documents at 10:00 a.m. Eastern Time, which will not be later than the third business day after the satisfaction or waiver of the conditions described below under the subsection Conditions to Closing of the Business Combination. Notwithstanding the foregoing, the Closing of the Business Combination may occur at such other time, date and location as may be mutually agreed upon in writing by GSAH, Mirion and the Charterhouse Parties.
Conditions to Closing of the Business CombinationConditions to Each Partys Obligations
The respective obligations of the parties to the Business Combination Agreement to effect the Closing and the other transactions contemplated by the Business Combination Agreement are subject to the satisfaction, at or prior to the Closing of the Business Combination, of each of the following conditions:
| | No provision of law, and no judgment, injunction, order or decree of any applicable governmental authority, will prohibit the consummation of the Closing. |
37
Table of Contents
| | Any applicable waiting period under the HSR Act (and any extensions thereof or any timing agreements, understandings or commitments obtained by request or other action of the United States Federal Trade Commission or the Antitrust Division of the United States Department of Justice, as applicable) will have expired or been terminated. |
| | The parties to the Business Combination Agreement will have received specified pre-Closing authorizations, consents, clearances, waivers and approvals of certain governmental authorities in connection with the execution, delivery and performance of the Business Combination Agreement and the transactions contemplated thereunder. |
| | The Registration Statement will have become effective in accordance with the Securities Act, no stop order shall have been issued by the SEC with respect to the Registration Statement and no action seeking such stop order shall have been threatened or initiated. |
| | The required vote of GSAHs stockholders to approve the Business Combination Proposal, the NYSE Proposal, the Charter Proposal, the Director Election Proposal, the Incentive Plan Proposal and the Adjournment Proposal shall have been duly obtained in accordance with the DGCL, the GSAH Certificate of Incorporation and bylaws, and the rules and regulations of the NYSE; |
| | GSAH will have at least $5,000,001 of net tangible assets following the exercise of any redemption rights by the Companys holders of Class A common stock in accordance with the GSAH Certificate of Incorporation and bylaws. |
Conditions to Closing of the Business CombinationConditions to GSAHs Obligations
The obligation of GSAH to consummate the Closing is subject to the satisfaction, at or prior to the Closing of the Business Combination, of each of the following conditions, any of which may be waived, in writing, by GSAH:
| | Mirion and the Sellers will have performed in all material respects all of their respective obligations under the Business Combination Agreement required to be performed thereby on or prior to the Closing Date. |
| | (i) The representations and warranties of Mirion contained in Section 4.09(b) of the Business Combination Agreement must be true and correct in all respects as of the date of the Business Combination Agreement and as of the Closing Date, as if made at and as of the Closing Date; (ii) the fundamental representations and warranties of Mirion (i.e., representations related to organization and qualification, capitalization, authority and brokers) must be true and correct in all but de minimis respects (without giving effect to any limitations as to materiality or Material Adverse Effect or any similar limitation set forth therein) as of the date of the Business Combination Agreement and as of the Closing Date, as if made at and as of the Closing Date (except to the extent such representations and warranties expressly relate to an earlier date, and in such case, shall be so true and correct in all but de minimis respects at and as of such earlier date); and (iii) each other representation and warranty of Mirion contained in Article 4 of the Business Combination Agreement must be true and correct (without giving effect to any limitations as to materiality or Material Adverse Effect or any similar limitation set forth therein) as of the date of the Business Combination Agreement and as of the Closing Date, as if made at and as of the Closing Date (except to the extent such representations and warranties expressly relate to an earlier date, and in such case, shall be true and correct at and as of such earlier date), except, in the case of this clause (iii), where the failure to be so true and correct, individually or in the aggregate, has not had, and would not reasonably be expected to have, a Material Adverse Effect. |
| | The representations and warranties of the Sellers contained in Article 3 of the Business Combination Agreement must be true and correct (without giving effect to any limitations as to materiality or |
38
Table of Contents
| Material Adverse Effect or any similar limitation set forth therein) as of the date of the Business Combination Agreement and as of the Closing Date, as if made at and as of such date (except to the extent such representations and warranties expressly relate to an earlier date, and in such case, shall be true and correct at and as of such earlier date), except where the failure to be so true and correct, individually or in the aggregate, has not had, and would not reasonably be expected to have, a Seller Material Adverse Effect. |
| | GSAH will have received a certificate signed by an officer of Mirion, dated the Closing Date, certifying that the conditions specified in the three immediately preceding bullet points have been fulfilled. |
| | A majority of the holders of the A Ordinary Shares and the B Ordinary Shares of Mirion will have delivered the drag along notice, which they have agreed to do within five business days of the effectiveness of this registration statement. |
| | No Material Adverse Effect will have occurred since the date of the Business Combination Agreement. |
| | Mirion and its subsidiaries will have issued notice of suspension or termination of any contracts to certain sales channel partners and otherwise ceased doing business with such sales channel partners as requested in writing by GSAH. |
Conditions to Closing of the Business CombinationConditions to Mirions and the Sellers Obligations
The obligation of Mirion and the Sellers to consummate the Closing are subject to the satisfaction, at or prior to the Closing of the Business Combination, of each of the following conditions, any of which may be waived, in writing, by Mirion and the Charterhouse Parties:
| | GSAH will have performed in all material respects all of its obligations under the Business Combination Agreement required to be performed by it on or prior to the Closing Date. |
| | (i) The representations and warranties of GSAH contained in Section 5.09(b) of the Business Combination Agreement must be true and correct in all respects as of the Closing Date, as if made at and as of such date; (ii) the fundamental representations and warranties of GSAH (i.e., representations related to organization and qualification, capitalization, authority and brokers) must be true and correct (without giving effect to any limitations as to materiality or material adverse effect or any similar limitation set forth therein) in all but de minimis respects as of the Closing Date, as if made at and as of such date (except to the extent such representations and warranties expressly relate to an earlier date, and in such case, shall be so true and correct in all but de minimis respects at and as of such earlier date); and (iii) each other representation and warranty of GSAH contained in Article 5 of the Business Combination Agreement shall be true and correct (without giving effect to any limitations as to materiality or material adverse effect or any similar limitation set forth therein) as of the Closing Date, as if made at and as of such date (except to the extent such representations and warranties expressly relate to an earlier date, and in such case, shall be true and correct at and as of such earlier date), except, in the case of this clause (iii), where the failure to be so true and correct has not had, and would not reasonably be expected to have, a SPAC Material Adverse Effect. |
| | Mirion will have received a certificate signed by an officer of GSAH, dated the Closing Date, certifying that the conditions specified in the two immediately preceding bullets have been fulfilled. |
| | The Available Closing Cash will not be less than $1,310,000,000. |
| | Since the date of the Business Combination Agreement, there will have been no development, effect, change, circumstance, event or occurrence, that, individually or in the aggregate, has had, or would reasonably be expected to have a SPAC Material Adverse Effect. |
39
Table of Contents
Representations and Warranties
Under the Business Combination Agreement, the Sellers made customary representations and warranties about such Seller relating to: existence and power; authorization; governmental authorization; noncontravention; ownership; and brokers fees.
Under the Business Combination Agreement, Mirion made customary representations and warranties about it relating to: existence and power; authorization; governmental authorization; noncontravention; capitalization; subsidiaries; financial statements; absence of certain changes; undisclosed material liabilities; material contracts; litigation; compliance with laws and court orders; real property; intellectual property; insurance coverage; employees; employee benefit plans; taxes; environmental matters; affiliate transactions; brokers fees; anti-bribery; anti-corruption; international trade; sanctions; data privacy; customers and suppliers; product liabilities and recalls; PPP loans; and debt financing.
Under the Business Combination Agreement, GSAH made customary representations and warranties about it relating to: corporate existence and power; authorization; governmental authorization; noncontravention; capital structure; SEC documents; controls; listing; registration statement and proxy statement; absence of certain changes; litigation; compliance with applicable laws; taxes; employees and employee benefit plans; affiliate transactions; properties; brokers fees; financial ability; PIPE financing; and trust account.
Covenants of the Parties
Covenants of Mirion
Mirion made certain covenants under the Business Combination Agreement, including, among other things, the covenants set forth below.
| | Mirion has agreed to, and to cause its subsidiaries to, from the date of the Business Combination Agreement until the earlier of the Closing or the termination of the Business Combination Agreement in accordance with its terms, operate in the ordinary course of business consistent with past practice. |
| | Subject to certain exceptions, from the date of the Business Combination Agreement until the earlier of the Closing or the termination of the Business Combination Agreement in accordance with its terms, Mirion will not, and will cause its subsidiaries not to: |
| | change or amend its organizational documents; |
| | make, declare, set aside, establish a record date for or pay any dividend or distribution; |
| | enter into, assume, assign, partially or completely amend any material term of, modify any material term of or terminate any collective bargaining or similar agreement (including agreements with works councils and trade unions and side letters) to which Mirion or its subsidiaries is a party or by which it is bound, other than in the ordinary course of business consistent with past practice; |
| | (i) issue, deliver, sell, transfer, pledge, dispose of or place any lien (other than a permitted lien) on any shares or any other equity or voting securities of Mirion or any of its subsidiaries or (ii) issue any securities (including any shares, voting securities or loan capital) or grant any options, appreciation rights, share units, profits interests, warrants or other rights to purchase or obtain any shares or any other equity or voting securities or loan capital of Mirion and/or any of its subsidiaries; |
| | sell, assign, transfer, convey, abandon, subject to a lien, or otherwise dispose of any owned real property; |
40
Table of Contents
| | sell, assign, transfer, convey, lease, license, abandon, allow to lapse or expire, subject to or grant any lien (other than permitted liens) on, or otherwise dispose of, any material assets, rights or properties (including leased real property) of Mirion and its subsidiaries, taken as a whole, other than in the ordinary course of business; |
| | waive, release, compromise, settle or satisfy any pending or threatened claim or compromise or settle any liability, (i) if such settlement would require payment by Mirion in an amount greater than $1,000,000 individually or in the aggregate, (ii) to the extent such settlement includes an agreement to accept or concede material injunctive relief or (iii) to the extent such settlement involves a governmental authority or alleged criminal wrongdoing; |
| | agree to modify in any respect materially adverse to Mirion and its subsidiaries any confidentiality or similar contract or agreement to which Mirion or any of its subsidiaries are a party; |
| | directly or indirectly acquire by merging or consolidating with, or by purchasing a substantial portion of the assets of, or by purchasing all of or a substantial equity interest in, or by any other manner, any business or any corporation, partnership, limited liability company, joint venture, association or other entity or Person or division thereof; |
| | make any loans or advance any money or other property; |
| | redeem, purchase or otherwise acquire any shares (or other equity interests) of Mirion or any of its subsidiaries or any securities or obligations convertible (whether currently convertible or convertible only after the passage of time or the occurrence of certain events) into or exchangeable for any shares of capital stock (or other equity interests) of Mirion or any of its subsidiaries; |
| | adjust, split, combine, subdivide, recapitalize, reclassify or otherwise effect any change in respect of any shares or other equity interests or securities of Mirion; |
| | make any change in its customary accounting principles or methods of accounting materially affecting the reported consolidated assets, liabilities or results of operations of Mirion and its subsidiaries; |
| | adopt or enter into a plan of complete or partial liquidation, dissolution, merger, consolidation or recapitalization; |
| | make, change or revoke any material tax election, adopt or change any material accounting method with respect to taxes, file any amended material tax return, settle or compromise any material tax liability, surrender any right to claim a material refund of taxes, consent to any extension or waiver of the limitations period applicable to any material tax claim or assessment (other than any extension pursuant to an extension to file any tax return), change its tax residence or start to trade to a material extent through a permanent establishment or other taxable presence, or enter into any material closing agreement with respect to any tax; |
| | (i) increase or grant any increase in the compensation, bonus, or other benefits (other than de minimis fringe or other benefits) of, or pay, grant or promise any bonus to, certain key employees; (ii) grant or pay any severance or change in control pay or benefits to, or otherwise increase the severance or change in control pay or benefits of, any current or former service provider; (iii) enter into, amend (other than immaterial amendments) or terminate any employee benefit plan, policy, program, agreement, trust or arrangement; (iv) take any action to accelerate the vesting or payment of, or otherwise fund or secure the payment of, any compensation or non-de minimis benefits under any employee plan; (v) grant any equity or equity based compensation awards; or (vi) hire or terminate (other than for cause) certain key employees; |
41
Table of Contents
| | voluntarily fail to maintain in full force and effect material insurance policies covering the Mirion and its subsidiaries and their respective properties, assets and businesses in a form and amount consistent with past practices; |
| | enter into any transaction or amend in any material respect any existing agreement with any related party; |
| | enter into any agreement that materially restricts the ability of Mirion or its subsidiaries to engage or compete in any line of business or enter into a new line of business; |
| | make any capital expenditure that in the aggregate exceeds $2,500,000; |
| | receive, collect, compile, use, store, process, share, safeguard, secure (technically, physically or administratively), dispose of, destroy, disclose, or transfer (including cross-border) any personal information (or fail to do any of the foregoing, as applicable) in violation of any (i) applicable privacy laws, (ii) external-facing privacy policies or notices of, or (iii) contractual obligations with respect to any personal information; |
| | incur, guarantee or otherwise become liable for (whether directly, contingently or otherwise) any indebtedness; and |
| | enter into, assume, assign, partially or completely amend any material term of, modify any material term of or terminate certain material contracts for which payments to or from Mirion or any of its subsidiaries would be expected to exceed $5,000,000 annually or $20,000,000 in the aggregate. |
| | Subject to confidentiality obligations and certain other restrictions, Mirion will, and will cause its subsidiaries to, afford to GSAH reasonable access to Mirions properties, books, contracts, commitments, records and appropriate officers and employees, and will use its commercially reasonable efforts to furnish GSAH with financial and operating data and other information concerning the affairs of Mirion and its subsidiaries as GSAH may reasonably request solely for purposes of consummating the transactions contemplated by the Business Combination Agreement. |
| | Mirion and each Seller (on behalf of itself and its respective controlled affiliates) waives any past, present or future claim of any kind against, and any right to access, the Trust Account, the trustee and GSAH, or to collect from the Trust Account any monies that may be owed to them by GSAH or any of its affiliates for any reason whatsoever, and will not seek recourse against the Trust Account at any time for any reason whatsoever. |
| | The Charterhouse Parties have agreed to exercise their drag-along right and to send a drag-along notice to the Mirion shareholders within five business days following the date this registration statement becomes effective. If any holders of Existing Mirion Shares, having been served such drag-along notice, have not delivered the relevant joinder agreement and election agreement by the fifth day prior to the Closing Date, such documents will be delivered on their behalf by an authorized individual pursuant to the Existing Mirion Articles. In the event any holders of Existing Mirion Shares fail to deliver their individual election notice, they will be deemed to have elected to receive GSAH Class A common stock as their consideration under the Business Combination Agreement. |
| | Mirion, the Charterhouse Parties and GSAH will use their reasonable best efforts to agree on a final steps plan regarding certain pre-Closing transactions. Prior to the Closing, Mirion will, and will cause its subsidiaries to, to take, or cause to be taken, all actions and to do, or cause to be done, all things necessary to effectuate the agreed-upon pre-Closing step plan, with such amendments, modifications, re-orderings and the like as Mirion may determine to be reasonably necessary and desirable to effect the transactions contemplated by the Business Combination Agreement, subject to certain consent rights of GSAH and the Charterhouse Parties. |
42
Table of Contents
| | Mirion will take certain actions to supplement existing policies, procedures and practices concerning sales channel partners, including ceasing to do business with channel partners in certain jurisdictions, if requested by GSAH. |
| | Mirion and the Sellers will settle and terminate certain related party agreements. |
| | Mirion will reasonably cooperate with GSAH and GSAHs lender to obtain, owners and lenders title insurance policies with respect to owned real property, dated as of the Closing Date, issued from a title insurance company and in amounts reasonably satisfactory to GSAH, including to deliver such customary affidavits from officers of Mirion and its subsidiaries as reasonably requested by the title insurance company, including any affidavit required by the title company in order to issue a non-imputation endorsement. |
| | Neither Mirion, any Seller nor any of their respective controlled affiliates, directly or indirectly, will engage in any transactions involving the securities of GSAH prior to the time of the making of a public announcement regarding all of the material terms of the transactions contemplated by the Business Combination Agreement. |
| | No later than ten (10) Business Days prior to the Closing, Mirion will take such action as may be necessary such that, as of the Closing, (i) certain loan agreements between Mirion and certain of its officers will be terminated and of no further continued force or effect without any obligations or liabilities surviving the Closing, and (ii) all accounts payable to either party to such agreements will be settled and fully discharged with no further obligation or liability to either party. |
| | Mirion will cause Mirion Technologies (HoldingSub2) Ltd., a limited liability company incorporated in England and Wales with a company number 09299632 (the DCL Beneficiary) to use its reasonable best efforts to, and will cause each of its subsidiaries to use its reasonable best efforts to, arrange and obtain debt financing on terms and conditions not less favorable than (taken as a whole) those set forth in the debt commitment letter delivered by Mirion in connection with the execution of the Business Combination Agreement (or such other terms as are reasonably acceptable to the DCL Beneficiary and GSAH). |
| | Prior to the Closing, Mirion will, or shall cause its applicable subsidiaries to, repay in full any loans applied for or received by Mirion or any of its subsidiaries pursuant to the Paycheck Protection Program (PPP Loans) outstanding as of the date of the Business Combination Agreement (including any PPP Loans that as of the date of the Business Combination Agreement are subject to (i) an application for forgiveness, (ii) an audit by the U.S. Small Business Administration or the lender of such PPP Loan, or (iii) an escrow agreement). |
| | Neither Mirion nor any Seller will take, nor will Mirion or any Seller permit any of their respective affiliates or representatives to take, whether directly or indirectly, any action to solicit, initiate or engage in discussions or negotiations with, or enter into any agreement with, or encourage, or provide information to, any person or entity concerning any purchase of any of Mirions equity securities or the issuance and sale of any securities of, or membership interests in, Mirion or its subsidiaries or any merger or sale of substantial assets involving Mirion or its subsidiaries, other than immaterial assets or assets sold in the ordinary course of business. |
Covenants of GSAH
GSAH made certain covenants under the Business Combination Agreement, including, among other things, the covenants set forth below.
| | Subject to certain exceptions, from the date of the Business Combination Agreement until the earlier of the Closing or the termination of the Business Combination Agreement in accordance with its terms, GSAH will not: |
| | change, modify or amend the Trust Agreement, or organizational documents; |
43
Table of Contents
| | declare, set aside or pay any dividends on, or make any other distribution in respect of any outstanding capital stock of, or other equity interests in, GSAH; split, combine or reclassify any capital stock of, or other equity interests in, GSAH; or repurchase, redeem or otherwise acquire, or offer to repurchase, redeem or otherwise acquire, any capital stock of, or other equity interests in, GSAH; |
| | make, change or revoke any material tax election, adopt or change any material accounting method with respect to taxes, file any amended material tax return, settle or compromise any material tax liability, surrender any right to claim a material refund of taxes, consent to any extension or waiver of the limitations period applicable to any material tax claim or assessment (other than any extension pursuant to an extension to file any tax return), change its tax residence or start to trade to a material extent through a permanent establishment other taxable presence, or enter into any material closing agreement with respect to any tax; |
| | enter into, renew or amend in any material respect any transaction or contract with a related party of GSAH; |
| | waive, release, compromise, settle or satisfy any pending or threatened material claim or compromise or settle any liability; |
| | incur, guarantee or otherwise become liable for (whether directly, contingently or otherwise) any indebtedness; |
| | offer, issue, deliver, grant or sell, or authorize or propose to offer, issue, deliver, grant or sell, any capital stock of, other equity interests, equity equivalents, stock appreciation rights, phantom stock ownership interests or similar rights in, GSAH or any securities convertible into, or any rights, warrants or options to acquire, any such capital stock or equity interests; |
| | amend, modify or waive any of the terms or rights set forth in any private placement warrant; or |
| | merge or consolidate, restructure, reorganize or completely or partially liquidate or dissolve, or adopt or enter into a plan of complete or partial liquidation, dissolution, restructuring, recapitalization or other reorganization of GSAH. |
| | GSAH will take all necessary action to cause the Board of Directors GSAH as of immediately following the Closing to consist of nine (9) directors, of whom one (1) shall be the Chief Executive Officer of GSAH upon the Closing (i.e., the Chief Executive Officer of Mirion immediately prior to the Closing), two (2) shall be named by the Sponsor, one (1) shall be named by the Charterhouse Parties and the remainder shall be mutually agreed by the Charterhouse Parties, Mirion and GSAH prior to the Closing; |
| | Prior to the Closing, GSAH will file an amendment to its certificate of incorporation with the Secretary of State of the State of Delaware in the form agreed by the parties to the Business Combination Agreement; |
| | Concurrently with the Closing, GSAH shall cause the existing Registration Rights Agreement, dated June 29, 2020, to be amended and restated in the form of the Amended and Restated Registration Rights Agreement agreed by the parties to the Business Combination Agreement. |
| | GSAH will use reasonable best efforts to ensure that it remains listed as a public company, and that shares of Class A common stock of GSAH remain listed, on the NYSE. |
| | GSAH shall use reasonable best efforts to keep current and timely file all reports required to be filed or furnished with the SEC and otherwise comply in all material respects with its reporting obligations under applicable law. |
44
Table of Contents
| | Unless otherwise approved in writing by Mirion and the Charterhouse Parties, GSAH will not permit any material amendment or modification to be made to, or any waiver of any provision or remedy under, or any replacements or terminations of, the Subscription Agreements in any manner adverse to Mirion or the Charterhouse Parties. GSAH will use its reasonable best efforts to take, or cause to be taken, all actions and do, or cause to be done, all things necessary, proper or advisable to consummate the transactions contemplated by the PIPE Subscription Agreements on the terms and conditions described therein, including using its reasonable best efforts to enforce its rights under the PIPE Subscription Agreements to cause the PIPE Investors to pay the applicable purchase price under each PIPE Investors applicable Subscription Agreement in accordance with its terms. |
| | Unless otherwise approved in writing by Mirion and the Charterhouse Parties, GSAH will not permit any material amendment or modification to be made to, or any waiver of any provision or remedy under, or any replacement or termination of, the Backstop Agreement in any manner adverse to Mirion or the Charterhouse Parties. |
| | Upon the satisfaction (or waiver by GSAH) of the conditions set forth in Article 11 of the Business Combination Agreement, and in accordance with and pursuant to the Trust Agreement at the Closing, (A) GSAH will cause the documents, opinions and notices required to be delivered to the trustee pursuant to the Trust Agreement to be so delivered and (B) GSAH will make arrangements to cause the trustee to (1) pay as and when due all amounts payable to all holders, as of the date of the Business Combination Agreement, of Class A common stock and Class B common stock of GSAH who shall have previously validly elected to redeem their shares pursuant to a redemption of such stockholders shares and (2) promptly thereafter, pay all remaining amounts then available in the Trust Account in accordance with the Business Combination Agreement and the Trust Agreement. |
| | GSAH will use its reasonable best efforts to, and will cause its respective representatives to use their reasonable best efforts to, provide all cooperation in connection with the arrangement of the debt financing as may be reasonably requested by Mirion that is necessary or customary for financings of the type contemplated by the debt commitment letter. |
| | Subject to certain limitations, GSAH agrees to take all steps necessary or advisable to eliminate impediments under any antitrust, competition, or other applicable law (including mitigation measures imposed by CFIUS, ITAR or MINEFI) that are asserted by any governmental authority or any other party having jurisdiction over the transactions contemplated by the Business Combination Agreement. |
| | GSAH will not take, nor will it permit any of its affiliates or representatives to take, whether directly or indirectly, any action to solicit, initiate, continue or engage in discussions or negotiations with, or enter into any agreement with, or encourage, respond, provide information to or commence due diligence with respect to, any person or entity, concerning, relating to or which is intended or is reasonably likely to give rise to or result in, any offer, inquiry, proposal or indication of interest, written or oral relating to any business combination. |
Joint Covenants
| | GSAH, the Sellers and Mirion will use their respective reasonable best efforts to take, or cause to be taken, all actions and to do, or cause to be done, all things necessary or advisable under applicable law to consummate the transactions contemplated by the Business Combination Agreement. |
| | The Sellers, GSAH and Mirion will cooperate with one another (i) in determining whether any action by or in respect of, or filing with, any governmental authority is required, or any actions, consents, approvals or waivers are required to be obtained from parties to any material contracts, in connection with the consummation of the transactions contemplated by the Business Combination Agreement and (ii) in using their respective reasonable best efforts to take such actions or make any such filings, furnishing information required in connection therewith and seeking timely to obtain any such actions, consents, approvals or waivers. |
45
Table of Contents
| | The parties to the Business Combination Agreement will negotiate in good faith to establish a directors and officers liability insurance policy, to be in place as of the Closing, that includes full prior acts coverage and continuity. |
| | The parties to the Business Combination Agreement will consult with each other before issuing any press release or making any public statement with respect to the Business Combination Agreement or the transactions contemplated thereby and will not issue any such press release or make any such public statement prior to such consultation. |
| | Each party to the Business Combination Agreement shall give prompt notice to the other parties of (a) certain actions or investigations; (b) the occurrence or non-occurrence of any event whose occurrence or non-occurrence, as the case may be, could reasonably be expected to cause any condition set forth in Section 11.02 or Section 11.03 of the Business Combination Agreement not to be satisfied at any time from the date the Business Combination Agreement to the Closing; (c) any notice or other communication from any third-party alleging that the consent of such third-party is or may be required in connection with the transactions contemplated by the Business Combination Agreement; and (d) any regulatory notice, report or results of inspection from a governmental authority in respect of the transactions contemplated by the Business Combination Agreement. |
Survival of Representations, Warranties and Covenants; No Indemnification
None of the representations, warranties, covenants and agreements in the Business Combination Agreement or in any instrument, document or certificate delivered pursuant to the Business Combination Agreement survive the Closing, except for those covenants and agreements contained therein which by their terms expressly apply in whole or in part after the Closing and then only to such extent until such covenants and agreements have been fully performed. Neither Mirion nor the Sellers will have any indemnification obligations pursuant to the Business Combination Agreement. None of the foregoing will limit the rights of GSAH to pursue recoveries under any buyer-side representations and warranties insurance policy.
Termination
The Business Combination Agreement may be terminated at any time prior to the Closing as follows:
| | by mutual written agreement of GSAH, Mirion and the Charterhouse Parties; |
| | by GSAH, on the one hand, and Mirion and the Charterhouse Parties, on the other hand, if the Closing has not been consummated on or before November 30, 2021 (as may be extended under the Business Combination Agreement, or by mutual agreement of the parties, the End Date); provided, however, that if the conditions specified in Sections 11.01(b) and 11.01(c) of the Business Combination Agreement have not been satisfied by November 30, 2021, either GSAH, on the one hand, or Mirion and the Charterhouse Parties, on the other hand, may by written notice to the other party, extend the End Date to January 31, 2022; provided, further however, that if, following such extension of the End Date, the conditions specified in Sections 11.01(b) and 11.01(c) of the Business Combination Agreement have not been satisfied by January 31, 2022 because of a failure to receive the specified approval or approvals noted on Section 11.01(c) of the Company Disclosure Schedule, either GSAH, on the one hand, or Mirion and the Charterhouse Parties, on the other hand, may by written notice to the other party, extend the End Date to March 31, 2022; |
| | by written notice from either Mirion and the Charterhouse Parties, on the one hand, or GSAH, on the other hand, to the other(s) if consummation of the transactions contemplated by the Business Combination Agreement would violate any nonappealable final order, decree or judgment of any court or governmental authority having competent jurisdiction; |
46
Table of Contents
| | by written notice to Mirion and the Charterhouse Parties from GSAH if there is any breach of any representation, warranty or covenant on the part of Mirion or the Sellers set forth in the Business Combination Agreement, such that the conditions specified in Sections 11.02(a), 11.02(b) and 11.02(c) of the Business Combination Agreement would not be satisfied at the Closing, subject to applicable cure periods as specified in the Business Combination Agreement; |
| | by written notice to GSAH from Mirion and the Charterhouse Parties if there is any breach of any representation, warranty or covenant on the part of GSAH set forth the Business Combination Agreement, such that the conditions specified in Sections 11.03(a) and 11.03(b) of the Business Combination Agreement would not be satisfied at the Closing, subject to applicable cure periods as specified in the Business Combination Agreement; |
| | by written notice from either Mirion and the Charterhouse Parties, on the one hand, GSAH, on the other hand, to the other(s) if the approval of the Business Combination Proposal, the NYSE Proposal, the Charter Proposal, the Director Election Proposal, the Incentive Plan Proposal and the Adjournment Proposal by the required vote of GSAHs stockholders is not obtained; or |
| | by written notice from either Mirion and the Charterhouse Parties, on the one hand, or GSAH, on the other hand, to the other(s) if the Minimum Cash Condition is incapable of being satisfied and Mirion and the Charterhouse Parties have not waived the Minimum Cash Condition within fifteen (15) Business Days of receipt by Mirion and the Charterhouse Parties of a written notice from GSAH that the Minimum Cash Condition has not been satisfied and is incapable of being satisfied. |
If the Business Combination Agreement is terminated, the Business Combination Agreement will become void and have no effect, without any liability on the part of any party thereto or its respective affiliates, shareholders, stockholders, directors, officers, employees, agents, consultants or representatives to the other parties to the Business Combination Agreement; provided that if such termination shall result from (i) fraud, (ii) the knowing and willful failure of a party to fulfill a condition to the performance of the obligations of the other party, (iii) the knowing and willful failure of a party to perform a covenant of the Business Combination Agreement or (iv) the knowing and willful breach by a party thereto of any representation or warranty or agreement contained herein, such party will, subject to the terms of the Business Combination Agreement, be fully liable for any and all losses, damages, claims, costs or expenses incurred or suffered by the other parties as a result of such failure or breach.
Amendments
Any provision of the Business Combination Agreement may be amended if such amendment is in writing and is signed by each of Mirion, GSAH and the Charterhouse Parties, on behalf of the Sellers.
Related Agreements
This section describes certain additional agreements entered into or to be entered into pursuant to the Business Combination Agreement. For additional information, see Proposal No. 1Approval of the Business CombinationRelated Agreements.
Amended and Restated Sponsor Agreement
In connection with the execution of the Agreement, the Company amended and restated that certain letter, dated June 29, 2020, by and among the Company, the Sponsor, GSAM Holdings and GS Employee Participation (collectively, the Insiders), pursuant to which, among other things, the Company and the Insiders agreed (i) to vote any shares of the Companys securities in favor of the Business Combination and other Business Combination proposals, (ii) not to redeem any shares of the GSAH Class A common stock or the founder shares, in connection with the optional stockholder redemption, (iii) not to transfer any founder shares until the earlier of
47
Table of Contents
(x) the one year anniversary of the Closing Date and (y) the day following the trading date when the last reported sale price of the GSAH Class A common stock first equals or exceeds $12.00 per share (as adjusted for stock splits, stock dividends, reorganizations, recapitalizations and the like) for any 20 trading days within any 30 consecutive trading day period commencing at least 150 days after the Closing Date, subject to the clear market provisions in the Amended and Restated Registration Rights Agreement, (iv) not to transfer any shares issued to GSAM Holdings as part of the PIPE Investment if such shares are retained by GSAM Holdings or its affiliates (but, for the avoidance of doubt, not if distributed to GSAM Holdings or its permitted transferees employees, investment partners or clients) for a period of 180 days after the Closing, subject to the clear market provisions in the Amended and Restated Registration Rights Agreement, and (v) to be bound to certain other obligations as described in the Amended and Restated Sponsor Agreement.
Additionally, the founder shares will be subject to vesting in three equal tranches, based on the volume weighted average price of the GSAH Class A common stock being greater than or equal to $12.00, $14.00 and $16.00, respectively, per share for any 20 trading days in any 30 consecutive trading day period. Such founder shares will be forfeited to the Company for no consideration if they fail to vest within five years of the Closing. The Sponsor has issued membership interests intended to be treated as profits interests for U.S. income tax purposes to each of Lawrence D. Kingsley, who will serve as Chairman of the Board of the Company when the Business Combination closes, Thomas Logan, Chief Executive Officer of Mirion, and Brian Schopfer, Chief Financial Officer of Mirion, whereby such individuals will have an indirect interest in the founder shares held by the Sponsor, subject to vesting upon the satisfaction of service, performance and other conditions, including the Closing.
Each of the holders of the founder shares have agreed to waive the anti-dilution adjustments provided for in the Companys Amended and Restated Certificate of Incorporation applicable to the founder shares in connection with the Business Combination, including the PIPE Investment. As a result of such waiver, the 18,750,000 founder shares will automatically convert into shares of the GSAH Class A common stock on a one-for-one basis upon the consummation of the Business Combination.
The foregoing description of the Amended and Restated Sponsor Agreement does not purport to be complete and is qualified in its entirety by the terms and conditions of the Amended and Restated Sponsor Agreement, a copy of which is attached hereto as Annex D and is incorporated herein by reference.
Subscription Agreements
Concurrently with the execution of the Agreement, the Company entered into Subscription Agreements with the PIPE Investors, pursuant to, and on the terms and subject to the conditions of which, the PIPE Investors have collectively subscribed for 90,000,000 shares of the GSAH Class A common stock for an aggregate purchase price equal to $900,000,000. The PIPE Investment will be consummated substantially concurrently with the Closing.
The Subscription Agreements for the PIPE Investors (other than GSAM Holdings, whose registration rights are governed by the Amended and Restated Registration Rights Agreement) provide for certain registration rights. In particular, the Company is required to, as soon as practicable but no later than (i) 30 calendar days following the Closing Date, file with the SEC (at the Companys sole cost and expense) a registration statement registering the resale of such PIPE Shares, and use its commercially reasonable efforts to have such registration statement declared effective as soon as practicable after the filing thereof, but no later than the earlier of (x) the 60th calendar day (or 90th calendar day if the SEC notifies the Company that it will review such registration statement) following the Closing and (y) the 10th business day after the date the Company is notified (orally or in writing, whichever is earlier) by the SEC that such registration statement will not be reviewed or will not be subject to further review. Such registration statement is required to be kept effective until the earliest of (i) the date the PIPE Shares thereunder have been sold by the non-GSAM PIPE Investors, (ii) the date the PIPE Shares
48
Table of Contents
may be sold without restrictions under Rule 144 under the Securities Act, including without limitation, any volume and manner of sale restrictions that may be applicable to affiliates under Rule 144 and without the requirement for the Company to be in compliance with the current public information required under Rule 144(c)(1) (or Rule 144(i)(2), if applicable) and (iii) three years after effectiveness of such registration statement.
The Subscription Agreements will terminate with no further force and effect upon the earliest to occur of: (i) such date and time as the Business Combination Agreement is terminated in accordance with its terms; (ii) upon the mutual written agreement of the parties to such Subscription Agreement; (iii) the conditions contained in the Subscription Agreement not being satisfied or waived prior to the Closing; and (iv) the first anniversary of the date of the Subscription Agreement if the closing pursuant to the Subscription Agreement has not yet occurred.
The foregoing description of the Subscription Agreements does not purport to be complete and is qualified in its entirety by the terms and conditions of the form of Subscription Agreement, a copy of which is attached hereto as Annex E and is incorporated herein by reference.
Amended and Restated Registration Rights Agreement
At the Closing, the Company will enter into the Amended and Restated Registration Rights Agreement with the GS Holders and the Sellers (collectively, with each other person who has executed and delivered a joinder thereto, the RRA Parties), pursuant to which the RRA Parties will be entitled to registration rights in respect of certain shares of the GSAH Class A common stock and certain other equity securities of the Company that are held by the RRA Parties from time to time.
The Amended and Restated Registration Rights Agreement provides that the Company will use commercially reasonable efforts to file with the SEC a shelf registration statement pursuant to Rule 415 under the Securities Act registering the resale of certain shares of the GSAH Class A common stock and certain other equity securities of the Company held by the RRA Parties as soon as reasonably practicable but no later than 30 calendar days following the consummation of the Business Combination and use its commercially reasonably efforts to have such shelf registration statement declared effective as soon as reasonably practicable after the filing thereof and no later than the earlier of (x) the 90th calendar day following the filing date if the SEC notifies the Company that it will review such shelf registration statement and (y) the 10th business day after the date the Company is notified in writing by the SEC that such shelf registration statement will not be reviewed or will not be subject to further review.
Each of (i) the Charterhouse Parties, (ii) the GS Holders or (iii) the holders of at least thirty percent (30%) in interest of the then outstanding registrable securities (each of (i), (ii) or (iii), the Demanding Holders) will be entitled to demand registration rights in connection with an underwritten offering. The Charterhouse Parties have an exclusive right for a 90-day period beginning on the 181st day after the Closing (the Charterhouse Demand Period) to exercise a single demand right. The Demanding Holders will be, at any time and from time to time on or after the date the Charterhouse Demand Period ends, entitled to demand registrations of all or part of their registrable securities. Such demand registrations are subject to certain offering thresholds, applicable lock-up restrictions and certain other conditions.
In addition, the RRA Parties have certain piggy-back registration rights. The Amended and Restated Registration Rights Agreement includes customary indemnification and confidentiality provisions. The Company will bear the expenses incurred in connection with the filing of any registration statements filed pursuant to the terms of the Amended and Restated Registration Rights Agreement.
The foregoing description of the Amended and Restated Registration Rights Agreement does not purport to be complete and is qualified in its entirety by the terms and conditions of the form of Amended and Restated Registration Rights Agreement, a copy of which is attached hereto as Annex F and is incorporated herein by reference.
49
Table of Contents
Backstop Agreement
In connection with the execution of the Agreement, GSAM Holdings and the Company have entered into the Backstop Agreement pursuant to which GSAM Holdings has committed to purchase from the Company up to 12,500,000 shares of the GSAH Class A common stock at a price per share equal to $10.00 immediately prior to (and contingent upon) the Closing, solely to the extent necessary to fund any valid redemptions by the Companys stockholders that results in the Cash Shortfall being greater than zero dollars, contingent upon the terms and subject to the conditions set forth in the Backstop Agreement.
The foregoing description of the Backstop Agreement does not purport to be complete and is qualified in its entirety by the terms and conditions of the Backstop Agreement, a copy of which is attached hereto as Annex G and is incorporated herein by reference.
Option Agreement
In connection with the execution of the Agreement, at the Closing, GSAM Holdings and certain of the Sellers will enter into the Option Agreement pursuant to which such Sellers will agree to, at the option of GSAM Holdings and subject to there being a partial exercise of the Backstop Agreement as described above, sell to GSAM Holdings up to 12,500,000 shares of the GSAH Class A common stock to be received by such Sellers pursuant to the Business Combination Agreement at the Closing at a price per share equal to $10.00 in cash, on the terms and subject to the conditions set forth in the Option Agreement. In the event there are sufficient redemptions of public shares such that there is a Cash Shortfall, the Company may exercise its rights under the Backstop Agreement to require GSAM Holdings to purchase up to 12,500,000 shares of GSAH Class A common stock to cover such Cash Shortfall as described above. If the Company exercises its rights under the Backstop Agreement for less than 12,500,000 shares of GSAH Class A common stock, GSAM Holdings has the right, but not the obligation, under the Option Agreement to purchase up to the difference of 12,500,000 shares of GSAH Class A common stock and the amount of shares purchased under the Backstop Agreement from the Sellers party to the Option Agreement.
The foregoing description of the Option Agreement does not purport to be complete and is qualified in its entirety by the terms and conditions of the form of Option Agreement, a copy of which is attached hereto as Annex H and is incorporated herein by reference.
Ownership of the Company Following the Business Combination
As of March 31, 2021, there were 93,750,000 shares of GSAH common stock outstanding, which includes the 18,750,000 founder shares held by the GS Sponsor and the 75,000,000 public shares. As of March 31, 2021, there are outstanding warrants to purchase an aggregate of 27,250,000 shares of GSAH common stock, which includes 8,500,000 private placement warrants held by the Sponsor and approximately 18,750,000 public warrants. Therefore, as of March 31, 2021 (without giving effect to the Business Combination), our fully diluted share capital would be approximately 121,000,000 shares of GSAH common stock.
It is anticipated that, upon completion of the Business Combination:
| | the PIPE Investors (including GSAM Holdings, assuming no syndication of its subscription) will own approximately 44% of the outstanding GSAH common stock; |
| | our public stockholders will own approximately 37% of the outstanding GSAH common stock; |
| | the Sellers, other than current members of Mirion management, will own approximately 15% of outstanding GSAH common stock; |
| | Sellers who are current members of Mirion management will own approximately 4% of the outstanding GSAH common stock; and |
50
Table of Contents
| | the GS Sponsor will own 0% of the outstanding GSAH common stock (assuming, for this purpose, that none of the founder shares performance vesting conditions have been satisfied at the time of completion of the Business Combination). |
These levels of ownership interest (i) assume that no public shares are elected to be redeemed in connection with the Business Combination, (ii) exclude the 18,750,000 outstanding founder shares held by the GS Sponsor (all of which are subject to the performance vesting conditions and forfeiture as described herein), (iii) assume no exercise of any warrants to purchase Class A common stock that will remain outstanding immediately following the Business Combination, (iv) exclude the issuance of any shares under the Incentive Plan following the Business Combination and (v) assume that only members of Mirions management team will elect to receive shares of GSAH Class B common stock and that the other Sellers receiving shares of GSAH common stock will elect to receive shares of GSAH Class A common stock. In addition, the consideration to be paid to the Mirion Sellers in connection with the Business Combination is allocated first in respect of the PIK Notes and second in respect of the Existing Mirion Shares. The PIK Notes accrue payment-in-kind (PIK) interest daily at a rate of 11.5% annually and are contemplated to be sold at the Closing for a price equal to the full outstanding principal amount together with all accrued but unpaid interest up to but excluding the Closing Date. See Certain Relationships and Related Persons TransactionsMirions Related Person TransactionsShareholder Notes. Accordingly, as the amount of accrued and unpaid interest increases, the holders of the PIK Notes will receive more consideration as compared to the holders of the Existing Mirion Shares. This has the effect over time of increasing the amount of shares of GSAH Class A Common Stock to be issued to the Mirion Sellers (excluding members of Mirion management) and reducing the number of shares of GSAH Class B common stock that will be issued to Mirion management (there is no incremental dilution to Public Stockholders). For purposes of the ownership levels described herein, we have assumed an amount of principal and interest of the PIK Notes as if the Closing Date will be October 31, 2021, but for all other purposes have assumed the Closing Date will be March 31, 2021.
If the actual facts are different from these assumptions, the above levels of ownership interest will be different.
The founder shares are subject to vesting in three equal tranches, based on the volume-weighted average price of the Companys GSAH Class A common stock being greater than or equal to $12.00, $14.00 and $16.00 per share for any 20 trading days in any 30 consecutive trading day period. Vesting of the founder shares will be accelerated upon certain sale events based on the per share price of the GSAH Class A common stock in such sale event. Holders of the founder shares are entitled to vote such founder shares and receive dividends and other distributions with respect to such founder shares prior to vesting, but such dividends and other distributions with respect to unvested founder shares will be set aside by the Company and shall only be paid to the holders of the founder shares upon the vesting of such founder shares. The founder shares will be forfeited to the Company for no consideration if they fail to vest within five years of the closing of the Business Combination.
In conjunction with the Business Combination Agreement, the Sponsor issued 3,200,000 membership interests to Thomas Logan, the Chief Executive Officer of Mirion, 700,000 membership interests to Brian Schopfer, the Chief Financial Officer of Mirion, and 4,200,000 membership interests to Lawrence Kingsley, who is expected to be Chairman of the Board of New Mirion. These Profits Interests are intended to be treated as profits interests for U.S. income tax purposes, pursuant to which Messrs. Logan, Schopfer and Kingsley will have an indirect interest in the founder shares held by the Sponsor. The Profits Interests are subject to service and performance vesting conditions, including the occurrence of the Closing, and do not fully vest until all of the applicable conditions are satisfied. In addition, the Profits Interests are subject to certain forfeiture conditions. See Certain Relationships and Related Persons TransactionsMirions Related Person TransactionsProfits Interests.
In addition, we expect that the Sellers who are current members of Mirions management team will elect to receive shares of GSAH Class B common stock of the Company that will have voting rights but no economic interest in the Company, paired with shares of IntermediateCo Class B common stock (non-voting) of a newly
51
Table of Contents
formed subsidiary (IntermediateCo) of GSAH. The share calculations assume that only members of Mirions management team will elect to receive shares of GSAH Class B common stock and that the other Sellers receiving shares of GSAH common stock will elect to receive shares of GSAH Class A common stock.
The following table illustrates varying ownership levels in New Mirion (as a percentage of outstanding common stock) immediately following the consummation of the Business Combination based on the assumptions above plus a maximum redemption scenario which assumes the maximum number of shares of GSAH Class A common stock are redeemed such that the remaining funds held in the trust account after the payment of the redeeming shares pro-rata allocation along with the proceeds from the PIPE Investment are sufficient to fund the Minimum Cash Condition.
| Pro Forma Class A Share Ownership in the Company | ||||||||||||||||
| No Redemptions | Maximum Redemptions (1) | |||||||||||||||
| Number of Shares (millions) |
Percentage of Outstanding Shares |
Number of Shares (millions) |
Percentage of Outstanding Shares |
|||||||||||||
| PIPE Investors (2) |
90.0 | 44 | % | 90.0 | 50 | % | ||||||||||
| Public Stockholders |
75.0 | 37 | % | 38.4 | 21 | % | ||||||||||
| Mirion Sellers (excluding Mirion Management) (3) |
30.0 | 15 | % | 30.0 | 17 | % | ||||||||||
| GS Sponsor (4) |
| 0 | % | | 0 | % | ||||||||||
| GS Backstop(5) |
| 0 | % | 12.5 | 7 | % | ||||||||||
| Pro Forma Class B Share Ownership in the Company | ||||||||||||||||
| No Redemptions | Maximum Redemptions | |||||||||||||||
| Number of Shares (millions) |
Percentage of Outstanding Shares |
Number of Shares (millions) |
Percentage of Outstanding Shares |
|||||||||||||
| Mirion Management (3) |
9.0 | 4 | % | 9.0 | 5 | % | ||||||||||
| (1) | Assumes that 36.6 million public shares (being our estimate of the maximum number of public shares that could be redeemed in connection with the Business Combination in order to satisfy the related Minimum Cash Condition contained in the Business Combination Agreement) are redeemed in connection with the Business Combination. |
| (2) | Includes 20 million GSAH Class A shares subscribed for by GSAM Holdings, assuming no syndication of its subscription. |
| (3) | Mirion Sellers have the option of receiving either shares of GSAH Class A common stock or Paired Interests at closing. We have assumed that all Mirion Sellers with the exception of members of Mirion management will elect to receive GSAH Class A common stock. |
| (4) | Excludes 18,750,000 founder shares that convert from shares of GSAH Class B common stock to shares of GSAH Class A common stock upon the closing of the Business Combination and are subject to certain vesting and forfeiture conditions described above. |
| (5) | The maximum redemptions scenario assumes there is a Cash Shortfall such that GSAM Holdings will purchase 12,500,000 shares of GSAH Class A common stock under the Backstop Agreement. |
In the event that more than 36.6 million public shares are redeemed and Mirion and the Charterhouse Parties waive the Minimum Cash Condition, Cash Consideration will be reduced by $10.00 for each additional redemption and additional shares of GSAH common stock will be issued to the Sellers at an assumed value of $10.00 per share in lieu thereof.
52
Table of Contents
The following summarizes the pro forma GSAH Class A and GSAH Class B common stock ownership (as a percentage of outstanding common stock) under a scenario where 100% of the outstanding public shares were redeemed:
| Pro Forma Class A Share Ownership in the Company |
||||||||
| 100% Redemptions | ||||||||
| Number of Shares (millions) |
Percentage of Outstanding Shares |
|||||||
| PIPE Investors (1) |
90.0 | 50 | % | |||||
| Public Stockholders |
| 0 | % | |||||
| Mirion Sellers (excluding Mirion Management) (2) |
68.3 | 38 | % | |||||
| GS Sponsor (3) |
| 0 | % | |||||
| GS Backstop(4) |
12.5 | 7 | % | |||||
| Pro Forma Class B Share Ownership in the Company |
||||||||
| 100% Redemptions | ||||||||
| Number of Shares (millions) |
Percentage of Outstanding Shares |
|||||||
| Mirion Management (2) |
9.0 | 5 | % | |||||
| (1) | Includes 20 million GSAH Class A shares subscribed for by GSAM Holdings, assuming no syndication of its subscription. |
| (2) | Mirion Sellers have the option of receiving either shares of GSAH Class A common stock or Paired Interests at closing. We have assumed that all Mirion Sellers with the exception of members of management will elect to receive GSAH Class A shares. |
| (3) | Excludes 18,750,000 founder shares that convert from shares of GSAH Class B common stock to shares of GSAH Class A common stock upon the closing of the Business Combination and are subject to certain vesting and forfeiture conditions described below. |
| (4) | The 100% redemptions scenario assumes there is a Cash Shortfall such that GSAM Holdings will purchase 12,500,000 shares of GSAH Class A common stock under the Backstop Agreement. |
For more information, see Unaudited Pro Forma Condensed Combined Financial Information.
Date, Time and Place of the Special Meeting of GSAHs Stockholders
The Special Meeting will be held via live webcast on [●] at [●]:00 a.m. [Eastern Time], or at such other time, on such other date and at such other place to which the meeting may be adjourned or postponed. The special meeting can be accessed by visiting [●], where you will be able to listen to the meeting live and vote during the meeting.
Voting Power; Record Date
GSAH stockholders will be entitled to vote or direct votes to be cast at the Special Meeting if they owned shares of Common Stock at the close of business on [●], 2021 which is the record date for the Special Meeting. Stockholders are entitled to one vote on each proposal presented at the Special Meeting for each share of our common stock held of record as of [●], 2021 the record date for the Special Meeting. If your shares are held in street name or are in a margin or similar account, you should contact your broker, bank or other nominee to ensure that votes related to the shares you beneficially own are properly counted. GSAHs warrants do not have voting rights with respect to the proposals to be presented at the Special Meeting. As of the close of business on the record date, there were [●] outstanding shares of our common stock.
53
Table of Contents
Proposals to be Put to the Stockholders of GSAH at the Special Meeting
In addition to the Business Combination Proposal, our stockholders will be asked to consider and vote upon the following proposals at the Special Meeting:
NYSE Proposal
Assuming the Business Combination Proposal is approved, our stockholders will also be asked to consider and approve, for purposes of complying with applicable listing rules of the NYSE, (a) the issuance of more than 20% of the Companys outstanding Class A common stock in connection with the Business Combination, including the PIPE Investment, and (b) the issuance of shares of the GSAH Class A common stock and the GSAH Class B common stock to a Related Party (as defined in Section 312.03 of the NYSEs Listed Company Manual) in connection with the Business Combination. For further details, see Proposal No. 2The NYSE Proposal.
Charter Proposal
Assuming the Business Combination Proposal and the NYSE Proposal are approved, our stockholders will also be asked to consider and act upon a proposal relating to adopting the New Mirion Charter in the form attached hereto as Annex B, which, if approved, would take effect upon the closing of the Business Combination. For further details, see Proposal No. 3The Charter Proposal.
Governance Proposal
Assuming the Business Combination Proposal, the Charter Proposal and the NYSE Proposal are approved, our stockholders will also be asked to consider and approve, on a non-binding advisory basis, certain governance provisions in the second amended and restated certificate of incorporation, presented separately in accordance with SEC requirements, which, if approved, would take effect upon the closing of the Business Combination. For further details, see Proposal No. 4The Governance Proposal.
Director Election Proposal
Assuming the Business Combination Proposal, the NYSE Proposal and the Charter Proposal are approved, our stockholders will also be asked to consider and vote upon a proposal to elect nine directors to serve, effective upon the closing of the Business Combination, with each director on our Board having a term that expires at the post-business combination companys annual meeting of stockholders in 2022, and until their respective successors are duly elected and qualified, or until their earlier resignation, removal or death. For further details, see Proposal No. 5The Director Election Proposal.
Incentive Plan Proposal
Assuming the Business Combination Proposal, the NYSE Proposal, the Charter Proposal and the Director Election Proposal are approved, our stockholders will also be asked to consider and vote upon a proposal to approve the Incentive Plan, including the authorization of the initial share reserve under the Incentive Plan. For further details, see Proposal No. 6The Incentive Plan Proposal.
Adjournment Proposal
Our Board may ask our stockholders to consider and vote upon a proposal to approve the adjournment of the Special Meeting to a later date or dates, if necessary, to permit further solicitation and vote of proxies in the event that there are insufficient votes for, or for any other reason in connection with, the approval of one or more of the other proposals at the Special Meeting. For further details, see Proposal No. 7The Adjournment Proposal.
54
Table of Contents
Quorum and Required Vote for proposals at the Special Meeting
A majority of the outstanding shares of our common stock entitled to vote as of the record date at the Special Meeting must be present, by attending the Special Meeting via the virtual meeting website or represented by proxy, at the Special Meeting to constitute a quorum and in order to conduct business at the Special Meeting. Abstentions will be counted as present for the purpose of determining a quorum but a broker non-vote will not. Our Initial Stockholders, who currently own approximately 20% of our outstanding shares of Common Stock, will count towards this quorum. In the absence of a quorum, the chairman of the Special Meeting has power to adjourn the Special Meeting. Based on the number of outstanding shares of our common stock as of the record date for the Special Meeting, [●] shares of our common stock will be required to achieve a quorum at the Special Meeting.
The Sponsor and GS Employee Participation have each agreed to, among other things, vote in favor of the Business Combination Proposal and the other proposals described herein to be presented at the Special Meeting. As of the date of this proxy statement/prospectus, the Initial Stockholders own approximately 20% of the outstanding shares of our common stock.
The proposals presented at the Special Meeting require the following votes:
| | Business Combination Proposal: The approval of the Business Combination Proposal requires the affirmative vote of a majority of the votes cast by holders of our outstanding shares of common stock represented at the special meeting by attendance via the virtual meeting website or by proxy and entitled to vote thereon at the Special Meeting. Generally, only votes FOR or AGAINST are considered to be votes cast. Accordingly, a Company stockholders failure to vote by proxy or to vote during the Special Meeting, as well as an abstention from voting and a broker non-vote with regard to the Business Combination Proposal will have no effect on the Business Combination Proposal. Abstentions will be counted in connection with the determination of whether a valid quorum is established but will have no effect on the Business Combination Proposal. |
| | NYSE Proposal: The approval of the NYSE Proposal requires the affirmative vote of holders of a majority of the votes cast by holders of our outstanding shares of common stock represented at the special meeting by attendance via the virtual meeting website or by proxy and entitled to vote thereon at the Special Meeting. A Company stockholders failure to vote by proxy or to vote during the Special Meeting and a broker non-vote with regard to the NYSE Proposal will have no effect on the NYSE Proposal. The NYSE considers abstentions to be votes cast and included in the number of shares of which a majority is required to vote in favor. Accordingly, abstentions will have the same effect as a vote AGAINST the NYSE Proposal. |
| | Charter Proposal: The approval of the Charter Proposal requires the affirmative vote of holders of a majority of our outstanding shares of common stock entitled to vote thereon at the Special Meeting. Accordingly, a Company stockholders failure to vote by proxy or to vote during the Special Meeting, as well as an abstention from voting and a broker non-vote with regard to the Charter Proposal will have the same effect as a vote AGAINST the Charter Proposal. |
| | Governance Proposal: The approval of the Governance Proposal will require the affirmative vote of a majority of the votes cast by holders of our outstanding shares of common stock represented at the special meeting by attendance via the virtual meeting website or by proxy and entitled to vote at the special meeting. Accordingly, a Company stockholders failure to vote by proxy or to vote during the Special Meeting, as well as an abstention from voting and a broker non-vote with regard to the Governance Proposal, will have no effect on the Governance Proposal. Abstentions will be counted in connection with the determination of whether a valid quorum is established but will have no effect on the Governance Proposal. |
55
Table of Contents
| | Director Election Proposal: Directors are elected by a plurality of all of the votes cast by holders of shares of common stock represented at the special meeting by attendance via the virtual meeting website or by proxy and entitled to vote thereon at the Special Meeting. This means that the nine director nominees who receive the most affirmative votes will be elected. Stockholders may not cumulate their votes with respect to the election of directors. Assuming a valid quorum is established, abstentions and broker non-votes will have no effect on the election of directors. |
| | Incentive Plan Proposal: The approval of the Incentive Plan Proposal requires the affirmative vote of a majority of the votes cast by holders of our outstanding shares of common stock represented at the special meeting by attendance via the virtual meeting website or by proxy and entitled to vote thereon at the Special Meeting. A Company stockholders failure to vote by proxy or to vote during the Special Meeting and a broker non-vote with regard to the Incentive Plan Proposal will have no effect on the Incentive Plan Proposal. The NYSE considers abstentions to be votes cast and included in the number of shares of which a majority is required to vote in favor. Accordingly, abstentions will have the same effect as a vote AGAINST the Incentive Plan Proposal. |
| | Adjournment Proposal: The approval of the Adjournment Proposal requires the affirmative vote of a majority of the votes cast by holders of our outstanding shares of common stock represented at the special meeting by attendance via the virtual meeting website or by proxy and entitled to vote thereon at the Special Meeting. Generally, only votes FOR or AGAINST are considered to be votes cast. Accordingly, a Company stockholders failure to vote by proxy or to vote during the Special Meeting, as well as an abstention from voting and a broker non-vote will have no effect on the Adjournment Proposal. Abstentions will be counted in connection with the determination of whether a valid quorum is established but will have no effect on the Adjournment Proposal. |
Redemption Rights
Pursuant to the GSAH Certificate of Incorporation, a public stockholder may request that we redeem all or a portion of its public shares for cash if the Business Combination is consummated. As a holder of public shares, you will be entitled to receive cash for any public shares to be redeemed only if you:
| (1) | (a) hold public shares, or (b) if you hold public shares through units, elect to separate your units into the underlying public shares and warrants prior to exercising your redemption rights with respect to the public shares; |
| (2) | prior to 5:00 p.m. Eastern Time on [●], 2021 (two business days before the scheduled date of the Special Meeting) submit a written request to Continental Stock Transfer & Trust Company, N.A., our transfer agent, that we redeem all or a portion of your public shares for cash, affirmatively certifying in your request if you ARE or ARE NOT acting in concert or as a group (as defined in Section 13d-3 of the Exchange Act) with any other stockholder with respect to shares of our common stock; and |
| (3) | deliver your public shares either physically or electronically through DTCs DWAC system to our transfer agent. |
Holders must complete the procedures for electing to redeem their public shares in the manner described above prior to 5:00 p.m., Eastern Time, on [●], 2021 (two business days before the scheduled date of the Special Meeting) in order for their shares to be redeemed.
Holders of units must elect to separate the units into the underlying public shares and warrants prior to exercising redemption rights with respect to the public shares. If holders hold their units through a broker, bank or other nominee, holders must notify their broker, bank or other nominee that they elect to separate the units into the
56
Table of Contents
underlying public shares and warrants, or if a holder holds units registered in its own name, the holder must contact the transfer agent directly and instruct it to do so. Public stockholders may elect to redeem their public shares even if they vote for the Business Combination Proposal or any other proposal. If the Business Combination is not consummated, the public shares will be returned to the respective holder, broker, bank or other nominee. If the Business Combination is consummated, and if a public stockholder properly exercises its right to redeem all or a portion of the public shares that it holds, including timely delivering its shares to our transfer agent, we will redeem such public shares for a per-share price, payable in cash, equal to the pro rata portion of the trust account, calculated as of two business days prior to the consummation of the Business Combination, including interest (net of taxes payable). For illustrative purposes, as of [●], this would have amounted to approximately $[●] per outstanding public share. If a public stockholder properly exercises its redemption rights in full, then it will be electing to exchange all of its public shares for cash and will not own any public shares of the post-business combination company. See Special Meeting of GSAH StockholdersRedemption Rights in this proxy statement/prospectus for a detailed description of the procedures to be followed if you wish to redeem your public shares for cash.
Notwithstanding the foregoing, a public stockholder, together with any affiliate of such public stockholder or any other person with whom such public stockholder is acting in concert or as a group (as defined in Section 13(d)(3) of the Exchange Act), will be restricted from redeeming its public shares with respect to more than an aggregate of 15% of the public shares. Accordingly, if a public stockholder, alone or acting in concert or as a group, seeks to redeem more than 15% of the public shares, then any such shares in excess of that 15% limit would not be redeemed for cash. In addition, pursuant to the GSAH Certificate of Incorporation, in no event will we redeem public shares in an amount that would cause our net tangible assets (as determined in accordance with Rule 3a51-1(g)(1) of the Exchange Act) to be less than $5,000,001. In such case, we would not proceed with the redemption of our public shares and the Business Combination, and instead may search for an alternate initial business combination.
The Sponsor and GS Employee Participation have agreed to, among other things, vote in favor of the Business Combination Proposal and the other proposals described herein to be presented at the Special Meeting. As of the date of this proxy statement/prospectus, the Initial Stockholders own approximately 20% of the outstanding shares of our common stock.
Holders of our outstanding warrants do not have redemption rights in connection with the Business Combination.
Appraisal Rights
Neither our stockholders nor our warrant holders have appraisal rights in connection with the Business Combination under the DGCL.
Proxy Solicitation
Proxies may be solicited by mail, telephone or in person. GSAH has engaged Innisfree M&A Incorporated to assist in the solicitation of proxies.
If a stockholder grants a proxy, it may still vote its shares at the special meeting by attendance via the virtual meeting website if it revokes its proxy before the Special Meeting. A stockholder also may change its vote by submitting a later-dated proxy as described in the section entitled Questions and Answers About the Proposals for our StockholdersMay I change my vote after I have mailed my signed proxy card?
57
Table of Contents
Interests of Goldman Sachs Parties and Certain Other Persons in the Business Combination
When you consider the recommendation of our Board in favor of approval of the Business Combination Proposal and the other proposals included herein, you should keep in mind that the Sponsor and our directors have interests in such proposal that are different from, or in addition to, those of our stockholders and warrant holders generally. Our Board was aware of and considered these interests, among other matters, in evaluating and negotiating the Business Combination and transaction agreements and in recommending to our stockholders that they vote in favor of the proposals presented at the Special Meeting, including the Business Combination Proposal. GSAH stockholders should take these interests into account in deciding whether to approve the proposals presented at the Special Meeting, including the Business Combination Proposal. These interests include, among other things:
| | If we do not consummate a business combination by July 2, 2022 (or if such date is extended at a duly called meeting of our stockholders, such later date), we would (1) cease all operations except for the purpose of winding up; (2) as promptly as reasonably possible but not more than ten business days thereafter, redeem the public shares, at a per share price, payable in cash, equal to the aggregate amount then on deposit in the trust account, including interest (which interest shall be net of taxes payable, and less up to $100,000 of interest to pay dissolution expenses), divided by the number of then outstanding public shares, which redemption will completely extinguish public stockholders rights as stockholders (including the right to receive further liquidating distributions, if any); and (3) as promptly as reasonably possible following such redemption, subject to the approval of our remaining stockholders and our Board, dissolve and liquidate, subject in each case to our obligations under Delaware law to provide for claims of creditors and the requirements of other applicable law. In such event, the 18,750,000 shares of Class B common stock owned by our Initial Stockholders, including the Sponsor, would be worthless because following the redemption of the public shares, we would likely have few, if any, net assets and because the Sponsor and each of our officers and directors have agreed to waive their rights to liquidating distributions from the trust account with respect to such shares if we fail to complete a business combination within the required period. Additionally, in such event, the 8,500,000 private placement warrants that the Sponsor paid $17 million for will expire worthless. The [-] shares of Class A common stock that the Initial Stockholders will hold following the Business Combination, if unrestricted and freely tradable, would have had aggregate market value of approximately $[-] million based upon the closing price of $[-] per share of Class A common stock on the NYSE on [-], 2021, the most recent practicable date prior to the date of this proxy statement. Given such shares of our common stock will be subject to certain restrictions, we believe such shares have less value. The [-] private placement warrants that the Sponsor will hold following the Business Combination, if unrestricted and freely tradable, would have had an aggregate market value of approximately $[-] million based upon the closing price of $[-] per warrant on the NYSE on [-], 2021, the most recent practicable date prior to the date of this proxy statement. See the section titled Proposal No. 1Approval of the Business CombinationInterests of Goldman Sachs Parties and Certain Other Persons in the Business Combination for further information on implied ownership levels by and returns to the GSAH stockholders (including the Sponsor) at various prices. |
| | Our existing management team members and directors will be eligible for continued indemnification and continued coverage under a directors and officers liability insurance policy after the Business Combination and pursuant to the Business Combination Agreement. |
| | In order to protect the amounts held in the trust account, the Sponsor has agreed that it will be liable to us if and to the extent any claims by a third party (other than our independent registered public accounting firm) for services rendered or products sold to us, or a prospective target business with which we have discussed entering into a transaction agreement, reduce the amount of funds in the trust account to below: (1) $10.00 per public share; or (2) such lesser amount per public share held in the trust account as of the date of the liquidation of the trust account due to reductions in the value of the trust assets, in each case, net of the interest which may be withdrawn to pay taxes, except as to any claims by a third party who executed a waiver of any and all rights to seek access to the trust account |
58
Table of Contents
| and except as to any claims under our indemnity of the underwriters of our IPO against certain liabilities, including liabilities under the Securities Act. |
| | Upon completion of the Business Combination, an aggregate amount of approximately $33,000,000 in deferred underwriting discount, advisory fees and placement agent fees, will be payable to Goldman Sachs & Co. LLC, an affiliate of us and the Sponsor. In addition, Goldman Sachs will receive a committed financing fee of $18,400,000 in connection with the Debt Financing. |
| | Tom Knott, our Chief Executive Officer, Chief Financial Officer and Secretary, is a Managing Director of Goldman Sachs and Raanan A. Agus, one of our directors, is a Participating Managing Director of Goldman Sachs. |
| | Goldman Sachs Private Credit Funds, affiliates of GSAH, are a current lender to Mirion, holding $137.6 million of the USD Term Loan and 122.8 million of the EUR Term Loan under the Mirion Credit Agreement. GSAH intends to use a portion of the proceeds from the Business Combination, including the PIPE Investment, to repay the outstanding Mirion Credit Agreement and, as a result, such affiliates would receive their pro rata portion of such proceeds. |
| | GSAM Holdings has subscribed for $200 million of the PIPE Investment, for which it will receive up to 20 million shares of our Class A common stock, unless it chooses to syndicate such subscription prior to Closing. See Proposal No. 1Approval of the Business CombinationRelated AgreementsSubscription Agreements. |
| | GSAM Holdings and GSAH have entered into the Backstop Agreement pursuant to which GSAM Holdings has committed to purchase from GSAH up to 12,500,000 shares of GSAHs Class A common stock at a price per share equal to $10.00 immediately prior to (and contingent upon) the Closing, contingent upon the terms and subject to the conditions set forth in the Backstop Agreement. For additional information, see Proposal No. 1Approval of the Business CombinationRelated AgreementsBackstop Agreement. |
| | GSAM Holdings and Sellers that elect to receive cash for their Existing Mirion Shares at Closing will enter into the Option Agreement, pursuant to which such Sellers will agree to, at the option of GSAM Holdings and subject to there being a partial exercise of the Backstop Agreement, sell to GSAM Holdings up to 12,500,000 shares of the GSAH Class A common stock to be received by such Sellers pursuant to the Business Combination Agreement at the Closing at a price per share equal to $10.00 in cash, on the terms and subject to the conditions set forth in the Option Agreement, see Proposal No. 1Approval of the Business CombinationRelated AgreementsOption Agreement. |
| | Pursuant to the Amended and Restated Registration Rights Agreement, the Sponsor, GS Employee Participation and GSAM Holdings will have customary registration rights, including demand and piggy-back rights, subject to cooperation and cut-back provisions with respect to the shares of Class A common stock and warrants held by such parties. See Proposal No. 1Approval of the Business CombinationRelated AgreementsAmended and Restated Registration Rights Agreement. |
| | The GS Director Nomination Agreement will grant the Sponsor the ongoing right (but not the obligation) to appoint or nominate to the Board of Directors two (2) individuals, or the GS Sponsor Directors, to serve as director of New Mirion. |
| | The Sponsor and GS Employee Participation have agreed to, among other things, vote in favor of the Business Combination Proposal and the other proposals described herein to be presented at the Special Meeting. See Proposal No. 1Approval of the Business CombinationRelated AgreementsAmended and Restated Sponsor Agreement. As of the date of this proxy statement, the Initial Stockholders own approximately 20% of the outstanding shares of our common stock. |
59
Table of Contents
The existence of financial and personal interests of one or more of GSAHs directors may result in a conflict of interest on the part of such director(s) between what he or they may believe is in the best interests of GSAH and its stockholders and what he or they may believe is best for himself or themselves in determining to recommend that stockholders vote for the proposals. See Proposal No. 1Approval of the Business Combination Interests of Goldman Sachs Parties and Certain Other Persons in the Business Combination for a further discussion of these considerations.
Recommendation to Stockholders of GSAH
GSAHs Board of Directors believes that the Business Combination Proposal and the other proposals to be presented at the Special Meeting are in the best interest of GSAHs stockholders and unanimously recommends that its stockholders vote FOR the Business Combination Proposal, FOR the NYSE Proposal, FOR the separate Charter Proposal, FOR the Governance Proposal, FOR each nominee in the Director Election Proposal, FOR the Incentive Plan Proposal and FOR the Adjournment Proposal, in each case, if presented to the Special Meeting.
The existence of financial and personal interests of one or more of GSAHs directors may result in a conflict of interest on the part of such director(s) between what he or they may believe is in the best interests of GSAH and its stockholders and what he or they may believe is best for himself or themselves in determining to recommend that stockholders vote for the proposals. See the section entitled Proposal No. 1Approval of the Business CombinationInterests of Goldman Sachs Parties and Certain Other Persons in the Business Combination for a further discussion of these considerations.
Sources and Uses of Funds for the Business Combination
The following tables summarize the sources and uses for funding the Business Combination assuming a March 31, 2021 Closing Date, and assume (i) for the no redemptions scenario that no public stockholders exercise their redemption rights in connection with the Business Combination and (ii) for the maximum redemptions scenario that 36.6 million public shares (being our estimate of the maximum number of public shares that could be redeemed in connection with the Business Combination in order to satisfy the Minimum Cash Condition) are redeemed in connection with the Business Combination.
| Assuming No Redemptions |
Assuming Maximum Redemptions |
|||||||
| Sources: |
||||||||
| Cash inflow from PIPE Investment (1) |
$ | 900.0 | $ | 900.0 | ||||
| Cash inflow from the Companys trust account |
750.1 | 750.1 | ||||||
| Cash inflow from new debt (2) |
830.0 | 830.0 | ||||||
| Cash inflow from balance sheet |
89.5 | 89.5 | ||||||
| Cash inflow from GS backstop (3) |
| 125.0 | ||||||
|
|
|
|
|
|||||
| Total sources |
$ | 2,569.6 | $ | 2,694.6 | ||||
|
|
|
|
|
|||||
| Uses: |
||||||||
| Paydown of Mirion third-party debt (4) |
908.5 | 908.5 | ||||||
| Payment to Sellers (5) |
1,310.0 | 1,310.0 | ||||||
| Payment to redeeming public stockholders (6) |
| 366.1 | ||||||
| Cash to balance sheet |
291.1 | 50.0 | ||||||
| Payment of seller transaction expenses (7) |
11.8 | 11.8 | ||||||
| Payment of other transaction expenses (8) |
48.2 | 48.2 | ||||||
|
|
|
|
|
|||||
| Total uses |
$ | 2,569.6 | $ | 2,694.6 | ||||
|
|
|
|
|
|||||
60
Table of Contents
| (1) | Represents the issuance of 90 million shares of GSAH Class A common stock through the PIPE Investment. |
| (2) | Represents the assumed issuance of $830.0 million of new debt for the debt refinancing of the Mirion Existing Credit Facility. The interest rate and other terms will vary depending on a variety of factors, including the timing of the debt financing marketing and market conditions existing at such time. |
| (3) | Represents the cash inflow from the exercise of the option under the Backstop Agreement to require the Backstop Party to purchase up to 12.5 million shares of Class A common stock in the maximum redemption scenario and no exercise such option in the no redemption scenario. |
| (4) | Reflects the cash used to effect the debt refinancing under Mirions Existing Credit Facility. |
| (5) | Reflects the net cash consideration paid to or on behalf of the Mirion Sellers under the terms of the Business Combination Agreement. This includes the repayment of outstanding notes payable to the Mirion Sellers. |
| (6) | Reflects the maximum payment that could be made to redeeming public stockholders which would leave sufficient cash to satisfy the Minimum Cash Condition. The maximum amount of redemptions assumed is 36.6 million shares at a price of $10.00 per share. |
| (7) | Represents the payment of estimated seller transaction and transaction advisor fees and expenses. |
| (8) | Represents the payment of deferred underwriter discounts and commissions of $26.3 million and an estimated $21.9 million of other acquisition-related transaction and transaction advisor fees and expenses. |
For more information, please see the sections entitled Unaudited Pro Forma Condensed Combined Financial Information.
U.S. Federal Income Tax Considerations
The U.S. federal income tax consequences to a stockholder of exercising its redemption rights will depend on the particular facts and circumstances. Please see the section entitled Proposal No. 1Approval of the Business CombinationUnited States Federal Income Tax Considerations to Stockholders Exercising Redemption Rights. We urge you to consult your tax advisors regarding the tax consequences of exercising your redemption rights.
Expected Accounting Treatment
The Business Combination is accounted for under the scope of Financial Accounting Standards Boards Accounting Standards Codification (ASC) Topic 805, Business Combinations (ASC 805). Pursuant to ASC 805, GSAH has been determined to be the accounting acquirer. Mirion constitutes a business in accordance with ASC 805 and the business combination constitutes a change in control. Accordingly, the business combination will be accounted for using the acquisition method. Under this method of accounting, Mirion will be treated as the acquired company for financial reporting purposes and the net assets of the post-business combination company will be stated at fair value, with goodwill or other intangible assets recorded.
Regulatory Matters
Under the HSR Act and the rules that have been promulgated thereunder by the U.S. Federal Trade Commission (the FTC), certain transactions may not be consummated unless information has been furnished to the Antitrust Division of the Department of Justice (Antitrust Division) and the FTC and certain waiting period requirements have been satisfied. The Business Combination is subject to these requirements and may not be completed until the expiration of a 30-day waiting period and any extensions thereto following the filing of the required Notification and Report Forms with the Antitrust Division and the FTC or until early termination is granted.
At any time before or after consummation of the Business Combination, notwithstanding any future expiration or termination of the waiting period under the HSR Act, the applicable competition authorities could take such action under applicable antitrust laws as each deems necessary or desirable in the public interest, including
61
Table of Contents
seeking to enjoin the consummation of the Business Combination. Private parties may also seek to take legal action under the antitrust laws under certain circumstances. Even following early termination or expiration of the waiting period under the HSR Act, we cannot assure you that the Antitrust Division, the FTC, any state attorney general, or any other government authority will not attempt to challenge the Business Combination on antitrust grounds, and, if such a challenge is made, we cannot assure you as to its result.
Additionally, under applicable laws and regulatory regimes in certain foreign jurisdictions, certain transactions may not be consummated until approval is granted or applicable waiting periods expire or terminate. The Business Combination is subject to applicable foreign direct investment (FDI) laws in certain jurisdictions, and may not be consummated until approval or expiration of the applicable waiting periods under these FDI laws, including in Finland, France, Germany and possibly the United Kingdom, and may not be completed until such approvals are obtained or the applicable waiting periods have expired.
Additional approvals relating to federal and state regulations pertaining to nuclear and radioactive materials are also required, and those approvals or actions will be sought. It is presently contemplated that if any such additional regulatory approvals or actions are required, those approvals or actions will be sought. There can be no assurance, however, that any additional approvals or actions will be obtained.
Emerging Growth Company
GSAH is an emerging growth company, as defined in Section 2(a) of the Securities Act, as modified by the JOBS Act, and it may take advantage of certain exemptions from various reporting requirements that are applicable to other public companies that are not emerging growth companies, including, but not limited to, not being required to comply with the auditor attestation requirements of Section 404 of the Sarbanes-Oxley Act, reduced disclosure obligations regarding executive compensation in GSAHs periodic reports and proxy statement/prospectus, and exemptions from the requirements of holding a nonbinding advisory vote on executive compensation and stockholder approval of any golden parachute payments not previously approved.
Further, section 102(b)(1) of the JOBS Act exempts emerging growth companies from being required to comply with new or revised financial accounting standards until private companies (that is, those that have not had a Securities Act registration statement declared effective or do not have a class of securities registered under the Exchange Act) are required to comply with the new or revised financial accounting standards. The JOBS Act provides that a company can elect to opt out of the extended transition period and comply with the requirements that apply to non-emerging growth companies but any such election to opt out is irrevocable. GSAH has elected not to opt out of such extended transition period, which means that when a standard is issued or revised and it has different application dates for public or private companies, GSAH, as an emerging growth company, can adopt the new or revised standard at the time private companies adopt the new or revised standard. This may make comparison of GSAHs financial statements with certain other public companies difficult or impossible because of the potential differences in accounting standards used.
We will remain an emerging growth company until the earlier of: (1) the last day of the fiscal year (a) following the fifth anniversary of the closing of the IPO, (b) in which we have total annual gross revenue of at least $1.07 billion or (c) in which we are deemed to be a large accelerated filer, which means the market value of our common equity that is held by non-affiliates exceeds $700 million as of the last business day of our most recently completed second fiscal quarter; and (2) the date on which we have issued more than $1.00 billion in non-convertible debt securities during the prior three-year period. If the Business Combination is consummated, we currently anticipate losing our emerging growth company status at 2022 year end. References herein to emerging growth company shall have the meaning associated with it in the JOBS Act.
62
Table of Contents
Comparison of Stockholder Rights
Following the consummation of the Business Combination, the rights of our stockholders who become New Mirion stockholders in the Business Combination will no longer be governed by the GSAH Certificate of Incorporation and instead will be governed by the New Mirion Charter and the New Mirion Bylaws. See Comparison of New Mirion Stockholder Rights.
Summary of Risk Factors
In evaluating the proposals to be presented at the Special Meeting, a stockholder should carefully consider all the information contained in this proxy statement/prospectus in deciding how to vote for the proposals presented herein. The occurrence of one or more of the events or circumstances described in the section titled Risk Factors, alone or in combination with other events or circumstances, may materially adversely affect our business, financial condition and operating results. Such risks include, but are not limited to:
Risks Related to Mirions Business
| | Mirions global operations have exposed Mirion to risks associated with public health crises and epidemics/pandemics, such as COVID-19. |
| | Mirion has incurred operating losses in the past and expects to incur operating losses in the future. |
| | If Mirion is unable to develop new products or enhance existing products to meet its customers needs and compete favorably in the market, Mirion may be unable to attract or retain customers. |
| | Mirions sales cycles in certain end markets can be long and unpredictable. |
| | Mirions growth plans depend in part on growth through acquisitions, and these plans involve numerous risks. If Mirion is unable to make acquisitions, or if Mirion is not successful in integrating the technologies, operations and personnel of acquired businesses or fail to realize the anticipated benefits of an acquisition, Mirions operations may be materially and adversely affected. |
| | Mirion operates as an entrepreneurial, decentralized company, which presents both benefits and certain risks. In particular, significant growth in a decentralized operating model may put strain on certain business group resources and Mirions corporate functions, which could materially and adversely affect Mirions business, financial condition and results of operations. |
| | A failure to expand Mirions manufacturing capacity and scale Mirions capabilities to manufacture new products could constrain Mirions ability to grow its business. |
| | Mirion derives a significant portion of its revenue from international sales and Mirions operations in foreign countries are subject to political, economic, legal and other risks, which could materially and adversely affect Mirions business. |
| | Mirion relies on third-party sales representatives and distributors to assist in selling Mirions products and services, and the failure of these representatives and distributors to perform as expected or to secure regulatory approvals in jurisdictions where they are required to do so could reduce our future sales. |
| | Mirion is subject to, or may otherwise be impacted by, a variety of federal, state, local and foreign laws and regulatory regimes. Failure to comply with such laws and regulations could subject Mirion to, among other things, penalties and legal expenses which could have a material and adverse effect on Mirions business, or such laws and regulations could otherwise impact Mirion, directly or indirectly, in a manner that has a material and adverse effect on Mirions business. |
63
Table of Contents
Risks Related to the Business Combination and GSAH
| | The Sponsor and GS Employee Participation have each agreed to vote in favor of the Business Combination and the other proposals described herein to be presented at the Special Meeting, regardless of how our public stockholders vote. |
| | Neither the GSAH Board nor any committee thereof obtained a third party valuation in determining whether or not to pursue the Business Combination. |
| | Since the Sponsor and the members of GSAHs management team have interests that are different, or in addition to (and which may conflict with), the interests of our stockholders, a conflict of interest may have existed in determining whether the Business Combination is appropriate as our initial business combination. Such interests include that the Sponsor will lose its entire investment in us if our business combination is not completed. |
| | The exercise of the GSAH management teams discretion in agreeing to changes or waivers in the terms of the Business Combination Agreement, including closing conditions, may result in a conflict of interest when determining whether such changes to the terms or waivers of conditions are appropriate and in GSAHs stockholders best interest. |
| | GSAH and Mirion will incur significant transaction and transition costs in connection with the Business Combination. |
| | The announcement of the proposed Business Combination could disrupt Mirions relationships with its customers, suppliers, joint venture partners and others, as well as its operating results and business generally. |
| | The historical financial results of Mirion and unaudited pro forma financial information included elsewhere in this proxy statement may not be indicative of what our actual financial position or results of operations would have been. |
| | We have a minimum cash requirement. This requirement may make it more difficult for us to complete the Business Combination as contemplated. |
| | We have identified a material weakness in our internal control over financial reporting, and we may experience additional material weaknesses or otherwise fail to design and maintain effective internal control over financial reporting, our ability to timely and accurately report our financial condition and operating results in compliance with reporting requirements applicable for public companies in the United States could be impaired, which may adversely affect investor confidence in us and, as a result, the value of our Class A common stock. |
Risks Related to the Redemption
| | Public stockholders who wish to redeem their public shares for a pro rata portion of the trust account must comply with specific requirements for redemption that may make it more difficult for them to exercise their redemption rights prior to the deadline. If stockholders fail to comply with the redemption requirements specified in this proxy statement, they will not be entitled to redeem their public shares for a pro rata portion of the funds held in the trust account. |
| | There is some uncertainty regarding the U.S. federal income tax consequences to holders of our Class A common stock of exercising their redemption rights. |
64
Table of Contents
Risks if the Business Combination Is Not Consummated
| | If we are not able to complete the Business Combination with Mirion by July 2, 2022 (or if such date is extended at a duly called meeting of stockholders, such later date) nor able to complete another initial business combination by such date, we would cease all operations except for the purpose of winding up and we would redeem our public shares and liquidate, in which case our public stockholders may receive only $10.00 per share, or less than such amount in certain circumstances, and our warrants will expire worthless. |
| | You will not have any rights or interests in funds from the trust account, except under certain limited circumstances. To liquidate your investment, therefore, you may be forced to sell your public shares or public warrants, potentially at a loss. |
| | If the funds not being held in the trust account are insufficient to allow us to operate until at least July 2, 2022, we may be unable to complete our initial business combination. |
65
Table of Contents
SELECTED UNAUDITED PRO FORMA CONDENSED COMBINED FINANCIAL INFORMATION
The following unaudited pro forma condensed combined balance sheet as of March 31, 2021 and the unaudited pro forma condensed combined statements of operations for the year ended December 31, 2020 and the three months ended March 31, 2021 present the historical financial statements of GSAH, adjusted to reflect the Business Combination. The Company and Mirion shall collectively be referred to herein as the Companies. The Companies, subsequent to the Business Combination, shall be referred to herein as the Combined Company.
The unaudited pro forma condensed combined balance sheet as of March 31, 2021 assumes that the Business Combination was completed on March 31, 2021. The unaudited pro forma condensed combined statements of operations for the year ended December 31, 2020 and for the three months ended March 31, 2021 give pro forma effect to the Business Combination as if it had occurred on January 1, 2020.
The Companys fiscal year ends on December 31, whereas Mirions fiscal year ends June 30. Due to this difference, the unaudited pro forma condensed combined statement of operations, which we refer to as the pro forma condensed combined statement of operations, for the year ended December 31, 2020, is derived from the Companys audited consolidated statement of operations for the year ended December 31, 2020, and Mirions unaudited financial results for the twelve-month period from January 1, 2020 through December 31, 2020. The pro forma condensed combined statement of operations for the three months ended March 31, 2021, combines the unaudited consolidated statement of operations for both GSAH and Mirion during the same period. Mirions balances have been classified consistently with the Companys presentation.
On June 17, 2021, the Company entered into the Business Combination Agreement. The pro forma condensed combined information contained herein assumes the Companys stockholders approve the proposed Business Combination. The Companys public stockholders may elect to redeem their shares of Class A common stock even if they approve the proposed Business Combination. The Company cannot predict how many of its public stockholders will elect to redeem their shares of GSAH Class A common stock for cash. As a result, the Company has provided pro forma condensed combined financial statements under two different redemption scenarios:
| | Assuming no redemptions: This presentation assumes that no shares of GSAH Class A common stock are redeemed. |
| | Assuming maximum redemptions: This presentation assumes that the maximum number of shares of GSAH Class A common stock are redeemed such that the remaining funds held in the trust account after the payment of the redeeming shares pro-rata allocation along with the proceeds from the PIPE Investment are sufficient to fund the Minimum Cash Condition (not less than $1,310 million available for use as cash consideration to the Sellers and to be retained on the balance sheet of the Combined Company). Based on the amount of $750.1 million in the trust account as of March 31, 2021, inclusive of accrued dividends, and considering the anticipated gross proceeds of approximately $900.0 million from the PIPE Investment, the aggregate commitment of $830 million from a first lien term facility pursuant to the Debt Commitment Letter and approximately $125.0 million from the Backstop Party, approximately 36,610,000 shares of GSAH Class A common stock may be redeemed and still enable us to have sufficient cash to satisfy the cash closing conditions in the Business Combination Agreement, including the debt refinancing of the Mirion Existing Credit Facility. |
The actual redemptions will likely be within the scenarios described above; however, there can be no assurance regarding which scenario will be closest to the actual results. Under both scenarios, the Company is considered the accounting acquirer. See Unaudited Pro Forma Condensed Combined Financial Information for more details.
66
Table of Contents
Summary Unaudited Pro Forma Financial Information
(Dollars in millions except share and per share data)
| Pro Forma Combined (Assuming No Redemptions) |
Pro Forma Combined (Assuming Maximum Redemptions) |
|||||||
| Statement of Operations DataThree Months Ended March 31, 2021 |
||||||||
| Total Revenues |
$ | 166.2 | $ | 166.2 | ||||
|
|
|
|
|
|||||
| Net income (loss) |
$ | (44.3 | ) | $ | (44.7 | ) | ||
|
|
|
|
|
|||||
| Pro Forma weighted average common shares of Class A common stock outstandingbasic and diluted |
195,050,000 | 170,940,000 | ||||||
| Pro Forma net income (loss) per share basic and diluted available to common stockholders, Class A |
$ | (0.22 | ) | $ | (0.25 | ) | ||
| Statement of Operations DataYear Ended December 31, 2020 |
||||||||
| Total Revenues |
$ | 584.7 | $ | 584.7 | ||||
|
|
|
|
|
|||||
| Net income (loss) |
$ | (206.1 | ) | $ | (207.7 | ) | ||
|
|
|
|
|
|||||
| Pro forma weighted average common shares of Class A common stock outstandingbasic and diluted |
195,050,000 | 170,940,000 | ||||||
| Pro forma net income (loss) per share basic and diluted available to common stockholders, Class A |
$ | (1.01 | ) | $ | (1.15 | ) | ||
| Balance Sheet DataAs of March 31, 2021 |
||||||||
| Total current assets |
$ | 662.7 | $ | 421.6 | ||||
| Total assets |
$ | 3,183.7 | $ | 2,942.6 | ||||
| Total current liabilities |
$ | 261.3 | $ | 261.3 | ||||
| Total liabilities |
$ | 1,242.9 | $ | 1,242.9 | ||||
| Total stockholders equity (deficit) |
$ | 1,940.8 | $ | 1,699.7 | ||||
67
Table of Contents
You should carefully review and consider the following risk factors and the other information contained in this proxy statement/prospectus, including the Annexes and the accompanying financial statements of GSAH and Mirion, in evaluating the Business Combination and the proposals to be voted on at the Special Meeting. You should carefully consider the following risk factors in addition to the other information included in this proxy statement/prospectus, including matters addressed in the section entitled Cautionary Statement Regarding Forward-Looking Statements. GSAH or Mirion may face additional risks and uncertainties that are not presently known to GSAH or Mirion, or that GSAH or Mirion currently deems immaterial, which may also impair GSAHs or Mirions business or financial condition.
Risks Related to Mirions Business
The following risk factors apply to the business and operations of Mirion and will also apply to the business and operations of the post-business combination company. The occurrence of one or more of the events or circumstances described in these risk factors, alone or in combination with other events or circumstances, may adversely affect the ability to complete or realize the anticipated benefits of the Business Combination, and may have a material adverse effect on the business, cash flows, financial condition and results of operations of the post-business combination company. Unless the context otherwise requires, all references in this subsection to the Company, we, us or our refer to the business of Mirion prior to the consummation of the Business Combination, which will be the business of the post-business combination company following the consummation of the Business Combination.
Risks Related to Our Business and Industry
Our global operations expose us to risks associated with public health crises and epidemics/pandemics, such as COVID-19. The global spread of COVID-19 has created significant volatility, uncertainty and worldwide economic disruption, resulting in an economic slowdown of potentially extended duration.
COVID-19 has had an adverse impact on our operations and supply chains, including as a result of impacts associated with preventive and precautionary measures that we, other businesses and governments are taking. Due to these impacts and measures, we have experienced unpredictable reductions in demand for certain of our products and services. Many employers in the United States and Europe, including us, are continuing to require some of their employees to work from home or not go into their offices or customers facilities. In addition to existing travel restrictions, countries may continue to close or decline to reopen borders, impose prolonged quarantines, and further restrict travel, which could significantly impact our ability to support our sites and customers in those locations and the ability of our employees to get to their places of work to produce products, or significantly hamper our products from moving through the supply chain. As a result, COVID-19 may materially adversely affect revenue growth in certain of our businesses, primarily those serving our medical end markets, and it is uncertain how materially COVID-19 will affect our global operations generally if these impacts were to persist or worsen over an extended period of time. The extent and duration of the impacts are uncertain and dependent in part on customers returning to work and economic activity ramping up.
The impact of COVID-19 on our customers has affected our sales operations in certain ways. For example, we have experienced increased customer disputes regarding orders, delayed customer notices to proceed with production, delayed payment from customers and, on rare occasions, customers have refused to pay for their orders entirely.
Our ability to continue to manufacture products is highly dependent on our ability to maintain the safety and health of our factory employees. The ability of our employees to work may be impacted by individuals contracting or being exposed to COVID-19. While we are following the requirements of governmental authorities and taking preventative and protective measures to prioritize the safety of our employees, these measures may not be successful, and we may be required to temporarily close facilities or take other measures.
68
Table of Contents
For example, two of our facilities have undergone brief closures due to the COVID-19 pandemic. While we are staying in close communication with our sites, employees, customers and suppliers and acting to mitigate the impact of this dynamic and evolving situation, the duration and extent of the effect of COVID-19 on us is not determinable.
The duration and extent of the impact from the COVID-19 pandemic depends on future developments that cannot be accurately predicted at this time, such as the severity and transmission rate of the virus, the existence of any additional waves of the pandemic, the extent and effectiveness of containment actions, treatment and prevention measures, including vaccines, and the impact of these and other factors on our customers, employees, suppliers and other business partners. Moreover, to the extent the COVID-19 pandemic or any worsening of the global business and economic environment as a result thereof, continues to adversely affect our business and financial results, it may also have the effect of heightening or exacerbating many of the other risks described under Risks Related to Our Business Operations.
We have incurred operating losses in the past and expect to incur operating losses in the future.
As of March 31, 2021 and June 30, 2020 we had an accumulated deficit of $860.6 million and $729.7 million, respectively. For the nine months ended March 31, 2021 we experienced a net loss of $131.0 million. We cannot assure you that we will achieve positive net income in any future period. We expect our operating expenses to increase in the future as we expand our operations. Furthermore, as a public company, we will incur additional legal, accounting and other expenses that we did not incur as a private company. If our revenue and gross profit do not grow at a greater rate than our operating expenses, we will not be able to achieve and maintain profitability. We expect to incur significant losses in the future for a number of reasons, including without limitation the other risks and uncertainties described herein. Additionally, we may encounter unforeseen operating or legal expenses, difficulties, complications, delays and other factors that may result in losses in future periods. If our expenses exceed our revenue, we may never achieve or maintain profitability and our business may be harmed.
Our financial performance may be variable.
Our business depends on the demand for our radiation detection, measurement, analysis and monitoring products, our nuclear medicine and related quality management products, and services in the nuclear, defense, medical and other end markets. In the past, the demand for our products in these markets has fluctuated due to a variety of factors, many of which are beyond our control. This has caused our financial performance to fluctuate. Among the factors affecting our performance are:
| | general economic conditions, both domestically and internationally, including inflation, recession and interest rate fluctuations; |
| | the timing, number and size of orders from, and shipments to, our customers, as well as the relative mix of those orders; |
| | the timing of revenue recognition, which often requires customer acceptance of the delivered products; |
| | delays, postponements or cancellations of construction or decommissioning of NPPs caused by, for example, financing difficulties or regulatory delays; |
| | adverse economic, financial and/or political conditions, as well as manmade or natural disasters, such as pandemics, in one or more of our target end markets; |
| | variations in the volume of orders for a particular product or product line in a particular quarter; |
| | the size and timing of new contract awards; |
| | the timing of the release of government funds for procurement of our products; |
| | the degree to which new end markets emerge for our products; |
69
Table of Contents
| | the budget cycles of U.S. and foreign governments and commercial enterprises that affect timing of order placement for or delivery of our products; |
| | the tendency of commercial enterprises to fully utilize annual capital budgets prior to expiration |
| | international trade conditions, such as the tariffs imposed by both the United States and China on the import of certain goods; and |
| | changes in laws or regulations affecting our target end markets, in particular the medical market. |
If we are unable to develop new products or enhance existing products to meet our customers needs and compete favorably in the market, we may be unable to attract or retain customers.
The markets in which we compete are subject to technological changes, product obsolescence and evolving industry standards. Our ability to successfully compete in these markets and to continue to grow our business depends in significant part upon our ability to develop, introduce and sell new and enhanced products in a timely and cost-effective manner, and to anticipate and respond to changing customer requirements. We have experienced, and may in the future experience, delays in the development and introduction of new products. These delays could provide a competitor a first-to-market advantage or greater market share. Defects or errors found in our products after commencement of commercial shipment could result in delays in market acceptance of these products. Our nuclear medicine and imaging products may become obsolete or unmarketable if new technologies are introduced to the market, or if new industry standards emerge. We may not be able to leverage our assets to diversify our products and services fast enough to generate revenue beyond our current markets in a timely manner. If we are unable to diversify our product and service offerings quickly enough to respond to market changes, our financial viability may worsen.
Our ability to successfully develop and introduce new products and product enhancements, and the revenues and costs associated with these efforts, will be affected by our ability to:
| | properly identify and address customer needs; |
| | in the case of our medical end market, educate medical providers about the use of new products and services; |
| | comply with internal quality assurance systems and processes in a timely and efficient manner; |
| | manage regulatory approvals and clearances including their timing and costs; |
| | accurately predict and control costs associated with inventory overruns caused by phase-in of new products and phase-out of old products; |
| | manufacture and deliver our products in sufficient volumes on time and accurately predict and control costs associated with manufacturing, installation, warranty and maintenance of the products; |
| | meet our product development plan and launch timelines; |
| | improve manufacturing yields of components; and |
| | manage customer demands for retrofits of both old and new products. |
Lack of market acceptance for our new products will jeopardize our ability to recoup research and development expenditures, hurt our reputation and harm our business, financial condition and results of operations. Accordingly, we cannot assure you that our future product development efforts will be successful.
We operate in highly competitive markets and in some cases compete against larger companies with greater financial resources.
The market for our products and services is fragmented, with a variety of small and large competitors, where the degree of fragmentation and the identities of our competitors vary among our target end markets. Some of our
70
Table of Contents
competitors have greater financial resources than do we, and they may be able to focus those resources on developing products or services that are more attractive to potential customers than those that we offer, or on lobbying efforts to enhance their prospects of obtaining government contracts. Some of our competitors, for example, are substantially larger and better capitalized than we are and have the ability to combine solutions into an integrated offering at attractive prices. Our competitors may offer these solutions at prices below cost in order to improve their competitive positions. Any of these competitive factors could make it more difficult for us to attract and retain customers, cause us to lower our prices to compete, and reduce our market share and revenue, any of which could materially and adversely affect our business, financial condition and results of operations.
Our customers may reduce or halt their spending on our products and services.
A variety of factors may cause our existing or future customers to reduce or halt their spending on radiation detection, measurement, analysis and monitoring products and services. These factors include:
| | unfavorable financial conditions and strategies of our customers; |
| | for the nuclear end market, civic opposition to or changes in government policies regarding nuclear operations or a reduction in demand for nuclear generating capacity; |
| | accidents, terrorism, natural disasters or other incidents occurring at our facilities, the facilities of our customers or at any other place; and |
| | the decision by one or more of our customers to acquire one of our competitors or otherwise insource the services we provide. |
Our sales cycles in certain end markets can be long and unpredictable.
Our sales efforts for many of our products involve substantial discussion with customers regarding product configuration and deployment. This process can be extremely lengthy and time consuming and typically involves a significant product evaluation process. For example, the typical sales cycle for products whose procurement relates to the construction of new, or the refurbishment of existing, nuclear power plants, or NPPs, ranges from 12 to 36 months and has, in some cases, extended up to 60 months or more. In the medical end market, the typical sales cycle depends upon the type of product and whether the sales are international or within the United States, and can range from 1 to 18 months. In addition, these customers generally make a significant commitment of resources to test and evaluate our products prior to purchase. As a result, our sales process is often subject to delays associated with the lengthy approval processes that typically accompany the design, testing and adoption of new, technologically complex products. This results in us investing significant resources prior to orders being placed for our products, with no assurances that we will secure a sale.
In addition, a significant amount of time can pass before we recognize the revenue associated with an order once it has been placed. We may need a notice to proceed with an order from the customer before starting to execute the customers order, which may delay revenue recognition. We may also not recognize revenue for sales of certain of our products until the customer certifies the successful installation and operation of the product, which can be many months or, particularly with regard to our Sensing Systems and Radiation Monitoring Systems products, years following the receipt of a customer order. The installation of our equipment may also be subject to construction or scheduled outage delays unrelated to our products, which can further defer the recognition of revenue.
We exercise judgment in determining the timing of revenue by analyzing the point in time or the period over which the customer has the ability to direct the use of and obtain substantially all of the remaining benefits of the performance obligation. Revenue recognized on an over-time basis for the year ended June 30, 2020 accounted for approximately 29% of total net sales. Typically, overtime revenue recognition is based on the utilization of an input measure used to measure progress, such as costs incurred to date relative to total estimated costs. Changes in total estimated costs are recognized using the cumulative catch-up method of accounting which recognizes the
71
Table of Contents
cumulative effect of the changes on current and prior periods in the current period. Accordingly, the effect of the changes on future periods of contract performance is recognized as if the revised estimate had been the original estimate. A significant change in an estimate on one or more contracts could have a material effect on our consolidated financial position, results from operations, or cash flows.
Our long and uncertain sales cycle and the unpredictable period of time between the placement of an order and our ability to recognize the revenue associated with the order makes revenue predictions difficult, particularly on a quarterly basis, and can cause our operating results to fluctuate significantly.
Our growth plans depend in part on growth through acquisitions, and these plans involve numerous risks. If we are unable to make acquisitions, or if we are not successful in integrating the technologies, operations and personnel of acquired businesses or fail to realize the anticipated benefits of an acquisition, our operations may be materially and adversely affected.
As part of our business and growth strategy, we have made and plan to continue to make acquisitions of, or significant investments, in businesses, products or technologies that allow us to complement our existing product offerings, expand our market coverage, increase our engineering workforce, reinforce our supply chain or enhance our technological capabilities.
For example, in fiscal 2020, we acquired Premium Analyse, a key player in the radioactive gas detection market and measurement of tritium, Selmic, an electronic component manufacturer of sensors, modules, and devices serving in automotive, transportation, medical, security, defense, and telecom industries, and Capintec, a leading supplier of calibration and measurement technologies for nuclear medicine applications. We also acquired the Personal Radiation Dosimeter facility from the Helmholtz Zentrum of Munich. In fiscal 2021, we acquired Biodex, a provider of nuclear instruments, imaging equipment and rehabilitation systems, DOSImetrics, a provider of personnel dosimetry systems, and Sun Nuclear, a provider of radiation oncology quality assurance. We plan to continue exploring additional acquisition opportunities in the future. If our expected returns on these transactions are not achieved, it could adversely impact our business, results of operations and financial condition.
We plan to continue exploring additional acquisition opportunities in the future but we are unable to predict whether or when any prospective acquisition candidate will become available or the likelihood that any acquisition will be completed.
Even if we do find suitable acquisition opportunities, we may not be able to consummate the acquisitions on commercially acceptable terms or realize the anticipated benefits of any acquisitions we do undertake. Our ability to grow our business through acquisitions is subject to numerous risks, including competition for the acquisition of attractive or promising businesses or assets, the need to finance such acquisitions through cash on hand or financing, and the need to secure required governmental approvals under antitrust and competition laws in the United States and worldwide.
Where we succeed in acquiring a business or assets, we are exposed to many risks, including:
| | problems integrating the new personnel or the purchased operations, technologies or products; |
| | difficulty securing adequate working capital; |
| | unanticipated costs associated with the acquisition; |
| | negative effects on our ability to generate excess free cash flow; |
| | negative effects on profitability; |
| | adverse effects on existing business relationships with suppliers and customers; |
| | risks associated with entering markets in which we have no or limited prior experience; |
72
Table of Contents
| | loss of key employees of the acquired business; |
| | our assumption of legal or regulatory risks, particularly with respect to smaller businesses that have immature business processes and compliance programs; |
| | litigation arising from the operations before they were acquired by us; and |
| | difficulty completing financial statements and audits. |
Our inability to overcome problems encountered in connection with any acquisition could divert the attention of management, consume scarce corporate resources and otherwise harm our business. If our expected returns on these transactions are not achieved, it could adversely impact our business, results of operations and financial condition.
Many of our products and services involve the detection, identification, measurement or monitoring of radiation and the failure of our products or services to perform to specification could materially and adversely affect our business, financial condition or results of operations.
Our products and services involve the detection and monitoring of radiation and are crucial components of the safety measures employed with respect to ionizing radiation. In the medical end market, our products and services are often used, for example, to ensure that radiation oncology patients receive accurate doses of radiation. In order to ensure the safety of such patients, we are committed to upholding high standards of precision and accuracy for our products. The failure of our products to perform to specification could result in personal injury or death and property damage (including environmental contamination), or the incorrect treatment being administered to patients. Legal and regulatory actions taken in response to product failure could result in significant costs to us. Additionally, the failure of our products to perform to specification could adversely affect market perception of the quality and effectiveness of our products and services, which would harm our ability to attract new customers and could cause our existing customers to cease doing business with us.
While we have attempted to secure appropriate insurance coverage at a reasonable cost, we do not insure against all risks and a claim can exceed the limits of our policies. We cannot assure you that our insurers will pay a particular claim, or that we will be able to maintain coverage at reasonable rates in the future, or at all. We may also be subject to significant deductibles.
Our contracts with customers generally seek to limit our liability in connection with product failure, but we cannot assure you that these contractual limitations on liability will be effective or sufficient in scope in all cases or that our insurance will cover the liabilities we have assumed under these contracts. The costs of defending against a claim arising out of such failure, and any damages awarded as a result of such a claim, could adversely affect our business, financial condition and results of operations.
Certain of our products require the use of radioactive sources or incorporate radioactive materials, which subjects us and our customers to regulations, related costs and delays and potential liabilities for injuries or violation of environmental, health and safety laws.
The majority of our products designed to detect, quantify and analyze ionizing radiation require the use of radioactive sources for testing and calibration. The required radioactive sources, or other sources of ionizing radiation, e.g., X-ray machines, are held by our facilities performing these tests and calibrations. Our customers hold equivalent sources for ongoing testing and re-calibration. Customers often acquire the radioactive sources directly from third party providers but may also purchase the sources from us as accessory to the product.
Certain of our reactor instrumentation and control equipment and systems in our Industrial segment incorporate radioactive materials. In all such cases, licenses for radioactive sources and materials or other sources of ionizing radiation are provided by the appropriate regulatory authority in the relevant jurisdiction and such authorities
73
Table of Contents
may be at the state or national level. Our failure or any customers failure to obtain the necessary license for radioactive sources or materials required by or incorporated into our products could result in the cancellation or delay of purchases by our customers, or remedial action by the relevant regulators.
While the specific process and criteria for receiving a license differ from jurisdiction to jurisdiction, it generally involves an application process in which we: identify a person or persons who have appropriate training and experience to be a health physics/radiation safety officer; specify the radioactive sources or materials sought to be licensed, their physical form (i.e., sealed or unsealed) and maximum possession limits on the amount of each type of radioactive element or compound sought under the license; specifies their intended use (e.g., calibration, testing, quality assurance, manufacturing); and, set forth written policies and procedures to ensure that we have adequate measures in place to ensure health and safety. These policies and procedures typically must be designed to ensure worker, workplace and public safety, including emergency plans; set forth the proper handling, control and security of radioactive sources or materials on site; detail any disposal or decommissioning considerations; and adequately train personnel at the site in proper access to, and handling of, radioactive sources or materials. Our noncompliance with or failure to properly implement such policies and procedures could delay or otherwise preclude us from obtaining the necessary license for radioactive sources or materials required by or incorporated into our products, which could result in the cancellation or delay of purchases by our customers.
The particular license requirements in a given jurisdiction are normally tailored to the specific radioactive elements or compounds involved, their physical form, and possession limits. Once authorities complete their application review and any required follow-up, the authority issues the site a license which imposes specific on-going compliance obligations that typically include requirements for us to pay periodic licensing fees, submit periodic written compliance reports, and agree to periodic site inspections by regulators, which may be announced or unannounced. Our failure to comply with any of these on-going obligations could result in the revocation of the necessary license for radioactive sources or materials required by or incorporated into our products, which could result in the cancellation or delay of purchases by our customers.
We are subject to federal, state and local regulations governing storage, handling and disposal of these radioactive materials and waste products. Outside of the United States, we are also subject to radiation regulations that vary from country to country. The improper storage, use and disposal of such materials by us and/or our customers could result in direct or secondary liability, including penalties and fines, to us in the event of environmental contamination or physical injury. Although we believe that our safety procedures for storing, handling and disposing of these radioactive materials comply in all material respects with the standards prescribed by law and regulation, we cannot eliminate the risk of accidental contamination or injury from those radioactive materials nor can we control the practices of our customers. The sale and use of our products with radioactive sources or materials could also lead to the filing of claims if someone were to allege injury from the use of one of our products or allege that one of our products was defective. Such a claim could result in substantial damages, be costly and time-consuming to defend and adversely affect the marketability of our products and our reputation.
We and many of our customers operate in a politically sensitive environment, and the public perception of nuclear energy or nuclear medicine can affect our customers and us.
We and our customers operate in a politically sensitive environment. The risks associated with radioactive materials and the public perception of those risks can affect our business. Opposition by third parties can delay or prevent the construction of new nuclear power plants and can limit the operation of nuclear reactors. Adverse public reaction to developments in the use of nuclear power could directly affect our customers and indirectly affect our business. In the past, adverse public reaction, increased regulatory scrutiny and litigation have contributed to extended construction periods for new nuclear reactors, sometimes delaying construction schedules by decades or more or even shutting down operations. In addition, anti-nuclear groups in Germany successfully lobbied for the adoption of the Nuclear Exit Law in 2002, which requires the shutdown of all German NPPs by 2022. Adverse public reaction could also lead to increased regulation or limitations on the
74
Table of Contents
activities of our customers, more onerous operating requirements or other conditions that could have a material adverse impact on our customers and our business.
Accidents involving nuclear power facilities, including but not limited to events similar to Fukushima, or terrorist acts or other high profile events involving radioactive materials could materially and adversely affect our customers and the markets in which we operate and increase regulatory requirements and costs that could in turn materially and adversely affect our business.
Successful execution of our business model in the nuclear power end market is dependent upon a certain level of public support for nuclear power. Nuclear power faces strong opposition from certain competitive energy sources, individuals, and organizations. The accident that occurred at the Fukushima nuclear power plant in Japan beginning on March 11, 2011 increased public opposition to nuclear power in some countries, resulting in a slowdown in, or, in some cases, a complete halt to new construction of nuclear power plants, an early shut down of existing power plants, or a dampening of the favorable regulatory climate needed to introduce new nuclear technologies. As a result of the Fukushima accident, some countries that were considering launching new domestic nuclear power programs have delayed or cancelled the preparatory activities they were planning to undertake as part of such programs. If accidents similar to the Fukushima disaster or other events, such as terrorist attacks involving nuclear facilities, occur, public opposition to nuclear power may increase, regulatory requirements and costs could become more onerous and customer demand for our products in the nuclear end market could suffer, which could materially and adversely affect our business and operations.
We have, and we intend to continue pursuing, fixed-price contracts. Our failure to mitigate certain risks associated with such contracts may result in reduced margins.
We estimate that approximately a quarter of our revenue was associated with contracts with a duration of 12 months or longer and approximately 60% of such revenue was associated with contracts with fixed-price arrangements which do not provide for price escalation in the event of unanticipated cost overruns, in each case for the fiscal year ended June 30, 2021. Under these contracts, we perform our services and provide our products at a fixed price. Fixed-price contracts carry inherent risks, including risks of losses from underestimating costs, operational difficulties and other changes that may occur over the contract period. We have in the past experienced unanticipated cost overruns on some of our fixed-price contracts. If our cost estimates for a contract are inaccurate or if we do not execute the contract within our cost estimates, we may incur losses or the contract may not be as profitable as we expected. In addition, even though some of our longer-term contracts contain price escalation provisions, such provisions may not fully provide for cost increases, whether from inflation, the cost of goods and services to be delivered under such contracts or otherwise. In addition, we are sometimes required to incur costs in connection with modifications to a contract that may not be approved by the customer as to scope or price, or to incur unanticipated costs, including costs for customer-caused delays, errors in specifications or designs or contract termination, that we may not be able to recover. These, in turn, could adversely affect our business, financial condition and results of operations. The revenue, cost and gross profit realized on such contracts can vary, sometimes substantially, from the original projections due to changes in a variety of factors, such as:
| | failure to properly estimate, or changes in, the costs of material, components or labor; |
| | inflation and currency exchange rate fluctuations; |
| | unanticipated technical problems with the products or services being supplied by us, which may require that we spend our own money to remedy the problem; |
| | our suppliers or subcontractors failure to perform; |
| | difficulties of our customers in obtaining required governmental permits or approvals; |
| | changes in local laws and regulations; |
| | unanticipated delays in construction of new NPPs and decommissioning of existing NPPs; and |
| | limited history with new products and new customers. |
Furthermore, we intend to continue pursuing longer-term contracts which may continue to contain fixed-price arrangements, and the amount of revenue associated with such contracts may change in future periods. As a
75
Table of Contents
result of one or more of these factors, we may incur losses or contracts may not be as profitable as we expect, and this could materially and adversely affect our business, financial condition and results of operations.
We may not realize all of the sales expected from our backlog of orders and contracts, and amounts included in our order backlog may not result in actual revenue or translate into profits.
Although the amount of our backlog is based on signed purchase orders or other written contractual commitments, we cannot guarantee that our order backlog will result in actual revenue in the originally anticipated period or at all. For fiscal 2020, our estimated combined order backlog was $601.4 million. The majority of our combined backlog is considered firm and expected to be delivered within one year. In addition, the mix of contracts included in our order backlog can greatly affect our margins in future periods, which may not be comparable to our historical product mix and operating results. Our customers may experience project delays or cancel orders due to factors beyond our control. If customers terminate, reduce or defer firm orders, whether due to fluctuations in their business needs or purchasing budgets or other reasons, our sales will be adversely affected and we may not realize the revenue we expect to generate from our backlog or, if realized, the revenue may not translate into profit. If our order backlog fails to result in revenue in a timely manner or at all, we could experience an overall reduction in revenue and liquidity.
Risks Related to Our Business Operations
We operate as an entrepreneurial, decentralized company, which presents both benefits and certain risks. In particular, significant growth in a decentralized operating model may put strain on certain business group resources and our corporate functions, which could materially and adversely affect our business, financial condition and results of operations.
The business is organized in two reportable business segments: Medical and Industrial. Our Medical segment is based around our sales, products and services to customers in the medical market. The Industrial segment is primarily based around the nuclear energy, defense, laboratories and scientific research markets as well as other industrial markets.
The decentralization of our organization structure necessarily places significant control and decision-making powers in the hands of local management, which presents certain risks, including the risk that we may be slower to detect or react to compliance-related matters, that company-wide business initiatives may be more challenging or costly to implement, and the risk of noncompliance or failures is higher than they may be in a more centralized operating environment. In addition, key business group resources and our corporate functions, which are leanly staffed but responsible for supporting our decentralized operations, may also not be able to detect or resolve financial, operational, and compliance matters on a timely basis. Our failure to adapt our financial, operational and compliance controls and systems to effectively manage our decentralized business and comply with our obligations as a public company could materially and adversely affect our business, financial condition or results of operations.
A failure to expand our manufacturing capacity and scale our capabilities to manufacture new products could constrain our ability to grow our business.
The future growth of our business depends on our ability to successfully expand our manufacturing capacity. For example, we experienced manufacturing delays with one of our suppliers, Selmic, in connection with ramping up production of our Mirion Battlefield Dosimeter. To ensure on-time deliveries going forward, we acquired Selmic and invested resources in resolving the manufacturing issues that caused delays. Expansion of our manufacturing capacity may also require us to obtain regulatory approvals or additional financing. Delay in the expansion of our manufacturing capacity could constrain our ability to grow our business, which would adversely affect our business, financial condition and results of operations.
Similarly, we could have substantial difficulty in dealing with rapid growth in markets for new products that we may introduce. If demand for our new products increases rapidly, we will need to expand internal production capacity or implement additional outsourcing. Success in developing, manufacturing and supporting products manufactured in small volumes does not guarantee comparable success in operations conducted on a larger scale.
76
Table of Contents
Manufacturing yields and product quality may decline as production volumes increase. If we are unable to deliver products quickly and cost effectively and in the requisite volumes, our customers may decline to purchase our new products or may purchase substitute products offered by our competitors. The costs associated with implementing new manufacturing technologies, methods, and processes, including the purchase of new equipment, and any resulting delays, inefficiencies and loss of sales, could harm our results of operations.
We rely on third-party manufacturers to produce sub-components for certain of our products and services. If our manufacturers are unable to meet our requirements, or are subject to unanticipated disruptions, our business could be harmed.
We use third-party manufacturers to produce sub-components for certain of our products. From time to time demand for our products has grown faster than the supply capabilities of these vendors. For example, significant growth in our Instadose product line required additional inventory purchasing to meet demand. In many cases, these manufacturers have no obligation to supply products to us for any specific period, in any specific quantity or at any specific price, except as set forth in a particular purchase order. Our requirements represent a small portion of the total production capacities for many of our manufacturers, and our manufacturers may reallocate capacity to other customers, even during periods of high demand for our products or services. We have in the past experienced, and may in the future experience, quality control issues and delivery delays with our manufacturers due to factors such as materials shortages, outages of specialized manufacturing equipment, high industry demand, inability of our manufacturers to consistently meet our quality or delivery requirements, or long lead times for components that could delay deliveries. Component manufacturers that sell to our suppliers may decide to stop producing certain components, declaring end-of-life for critical components and limiting supply of these components. In such cases, we would need to identify component alternatives, redesign electronic components or requalify electronic designs, which would require time and resources. In addition, third-party manufacturers may have financial difficulties and face the risk of bankruptcy, especially in light of the current worldwide economic downturn. If one of our suppliers was to cancel or materially change a commitment with us or fail to meet the quality or delivery requirements needed to satisfy customer orders for our products, we could lose time-sensitive customer orders, be unable to develop or sell our products or services cost effectively or on a timely basis, if at all, and have significantly decreased revenue, which would harm our business, financial condition and results of operations. We may qualify additional suppliers in the future which would require time and resources. If we do not qualify additional suppliers, we may be exposed to increased risk of capacity shortages due to our dependence on our current suppliers.
In addition, our suppliers (and those they depend upon for materials and services) are subject to risks, including COVID-19-related supplier plant shutdowns or slowdowns, labor disputes or constraints, union organizing activities, intellectual property claims, financial liquidity, information technology failures, inclement weather, natural disasters, significant public health and safety events, supply constraints, and general economic and political conditions that could limit their ability to provide us with materials. Insurance for certain disruptions may not be available, affordable or adequate. The effects of climate change, including extreme epidemics and pandemics, weather events, long-term changes in temperature levels, sea level rise and water availability may exacerbate these risks. Such disruption has in the past and could in the future interrupt our ability to manufacture certain products.
We derive a significant portion of our revenue from international sales and our operations in foreign countries are subject to political, economic, legal and other risks, which could materially and adversely affect us.
Revenue generated from outside of North America accounted for approximately 48% of our net sales in both fiscal 2020 and 2019, and approximately 47% of our net sales for the nine months ended March 31, 2021 and 2020. We anticipate that international sales will continue to constitute a material percentage of our total net sales in future periods. As a result, our operations are subject to risks associated with global operations and sales, including:
| | foreign currency exchange fluctuations; |
77
Table of Contents
| | changes in regulatory requirements; |
| | tariffs and other barriers; |
| | timing and availability of export licenses; |
| | difficulties in accounts receivable collections; |
| | difficulties in protecting and enforcing our intellectual property; |
| | difficulties in staffing and managing international operations; |
| | difficulties in managing sales agents, distributors and other third parties; |
| | coordination regarding, and difficulties in obtaining, governmental approvals for products that may require certification; |
| | rescission or termination of contracts by governmental parties without penalty and regardless of the terms of the contract; |
| | restrictions on transfers of funds and other assets of our subsidiaries between jurisdictions; |
| | the burden of complying with a wide variety of complex foreign laws and treaties; |
| | potentially adverse tax consequences; and |
| | uncertainties relative to regional political and economic circumstances. |
We are also subject to risks associated with the imposition of legislation and regulations relating to the import or export of our products. Furthermore, the failure to comply with export control regulations and to obtain required approvals could result in loss of the ability to continue to export products, fines and penalties.
We cannot predict whether quotas, duties, taxes or other charges or restrictions upon the importation or exportation of our products will be implemented by the United States or other countries. Some of our customers purchase orders and agreements are governed by foreign laws, which often differ significantly from the laws of the United States. Therefore, we may be limited in our ability to enforce our rights under such agreements and to collect damages, if awarded. These factors may materially and adversely affect our business, financial condition and results of operations.
We rely on third-party sales representatives to assist in selling our products and services, and the failure of these representatives to perform as expected or to secure regulatory approvals in jurisdictions where they are required to do so could reduce our future sales.
We derive a significant portion of our revenue from sales through third-party sales representatives. We have established relationships with some of our third-party sales representatives recently, and we are unable to predict the extent to which our third-party sales representatives will be successful in marketing and selling our products and services. Moreover, many of our third-party sales representatives also market and sell competing products and services, which may affect the extent to which our third-party sales representatives promote our products and services. If our third party sales representatives advertise or promote or characterize our products in a manner inconsistent with our (or their) messaging, as approved by our regulatory affairs professionals, such acts could be imputed to us and we could become subject to risk or liability from government regulatory bodies or agencies for criminal or civil claims, including false claims, and we could become susceptible to individual consumer actions or class actions based on false or improper advertising and promotion, off-label promotion, failure to warn defects in our products and unfair competition or unfair trade practices claims, all of which could lead to adverse publicity, fines, penalties, judgments, money damages and other significant losses. Our future performance will also depend, in part, on our ability to attract additional third-party sales representatives who will be able to market and support our products and services effectively and accurately, especially in markets in which we have not previously sold our products and services. If we cannot retain our current third-party sales representatives or recruit additional or replacement third-party sales representatives, our business, financial condition and results of operations could be harmed.
78
Table of Contents
If our suppliers experience supply shortages and prices of commodities or components that we use in our operations increase, our results of operations could be materially and adversely affected.
We are dependent upon certain sole or limited source suppliers for critical raw materials or components of some of our products. For example, we rely on limited source suppliers for certain precious metals used in some of our radiation oncology and reactor instrumentation, scintillator materials used in our detection and identification equipment, analog sensor tubes used in certain of our imaging products, and detectors used in our dosimetry line of products.
Most of our suppliers are not required to supply us with any minimum quantities, and we cannot assure you that we will receive adequate quantities of components on a timely basis in the future. For example, a single source supplier informed us that its supplier was discontinuing the manufacture of an on-board computer module component of one of our multi-channel analyzers used by our Industrial segment to interpret signals from our detectors allowing our customers to understand levels of detectable radiation. The notification prompted us to secure a final end-of-life order in an amount sufficient to meet our anticipated production requirements at least through April 2022, the exact duration depending on sales of this particular device. Qualification and redesign efforts are underway to meet this timeline.
Our suppliers could have financial or other issues that could cause a disruption in the supply or increase the cost of components to us. Such disruptions or delays could impact our obligations to other parties. In addition, were we to change suppliers of components in some of our products, we may be required to seek new qualifications for such products, which can be a time-consuming and costly process. As a result of interruption of supply or increased component costs, we may not be able to obtain the raw materials or components that we need to fill customer orders. The inability to fill these orders could cause delays, disruptions or reductions in product shipments, require us to negotiate alternate supply arrangements with replacement suppliers where available or require product redesigns which could, in turn, damage relationships with current or prospective customers, increase costs or prices and materially and adversely affect our business, financial condition and results of operations, including through litigation.
Our reliance upon sole or limited sources of supply for certain materials or components could cause production interruptions, delays and inefficiencies.
We purchase materials, components, and equipment from third parties for use in our manufacturing operations. For example, we purchase cryogenic cooling equipment to support our spectroscopy line of products. There is a limited supply market for this type of equipment, and these products are designed specifically for use in our products. Qualification and design of new equipment will require time and resources to complete. Our income could be adversely impacted if we are unable to adjust our purchases to reflect changes in customer demand and market fluctuations, including those caused by seasonality or cyclicality. During a market upturn, suppliers may extend lead times, limit supplies, or increase prices. If we cannot purchase sufficient products at competitive prices and quality and on a timely enough basis to meet increasing demand, we may not be able to satisfy market demand, product shipments may be delayed, our costs may increase, or we may breach our contractual commitments and incur liabilities. Conversely, in order to secure supplies for the production of products, we sometimes enter into non-cancelable purchase commitments with vendors, which could impact our ability to adjust our inventory to reflect declining market demands. If demand for our products is less than we expect, we may experience additional excess and obsolete inventories and be forced to incur additional charges and our profitability may suffer.
In addition, some of our businesses purchase certain requirements from sole or limited source suppliers for reasons of quality assurance, cost effectiveness, availability, contractual obligations or uniqueness of design or technology. If these or other suppliers encounter financial, operating, quality, or other issues or if our relationship with them changes, including as a result of contractual disputes, we might not be able to quickly establish or qualify replacement sources of supply. The supply chains for our businesses could also be disrupted by supplier capacity constraints, operational or quality issues, bankruptcy or exiting of the business for other reasons,
79
Table of Contents
decreased availability of key raw materials or commodities, and external events such as natural disasters, pandemic health issues, war, terrorist actions, governmental actions, and legislative or regulatory changes. For example, some of our products incorporate microchips and other semiconductor components for which there is a global supply shortage. Any of these factors could result in production interruptions, delays, extended lead times, and inefficiencies. As discussed above, such disruptions could also result in liability from litigation.
Because we cannot always immediately adapt our production capacity and related cost structures to changing market conditions, our manufacturing capacity may at times exceed or fall short of our production requirements. Any or all of these issues could result in the loss of customers, provide an opportunity for competing products to gain market acceptance, and otherwise adversely affect our profitability. If we are not able to mitigate the impact of any disruptions in our supply chain, then our business, financial condition and results of operations may be materially and adversely impacted.
Because we compete directly with certain of our customers and suppliers, our results of operations could be materially and adversely affected in the short term if these customers or suppliers abruptly discontinue or significantly modify their relationship with us.
Some of our competitors are also our suppliers and customers. For example, we had an arrangement with a supplier of components used to manufacture our Cryo-Cycle product. That supplier was acquired by one of our competitors, after which time the supplier ceased supplying us with the components used to manufacture the Cryo-Cycle. As with our other suppliers, our competitor suppliers are not required to supply us with any minimum quantities, and we cannot assure you that we will receive adequate quantities of components on a timely basis in the future. The loss of orders stemming from the actions of our supplier or customer competitors could cause delays, disruptions or reductions in product shipments or require product redesigns that could, in turn, damage relationships with current or prospective customers, increase costs or prices, result in litigation or otherwise materially and adversely affect our business, financial condition and results of operations.
We derive a material portion of our revenue from contracts with governmental customers or their contractors. Such customers are subject to increased pressures to reduce expenses. Government-funded contracts may also contain unusual or more onerous terms and conditions that are not common among commercial customers or risk subjecting us to audits, investigations, sanctions and penalties.
U.S. government contractors and subcontractors must comply with specific procurement regulations and other requirements, including without limitation those related to ethics and business conduct, cost accounting, pricing, intellectual property, employment, cybersecurity, and supply chain issues. Accordingly, we are subject to routine audits and investigations by U.S. government agencies and held to strict compliance standards. If we fail to comply with these rules and regulations, we could be subject to reductions in the value of our government contracts, contract modification or termination, loss of valuable intellectual property rights, the assessment of criminal and civil penalties and fines, and/or suspension or debarment from government contracting and subcontracting for a period of time or permanently.
Furthermore, we have bid, and may in the future submit bids, for U.S. government contracts that require our employees to maintain various levels of security clearances and require us or our subsidiaries to maintain certain facility security clearances in compliance with Department of Defense requirements. Obtaining and maintaining security clearances for employees involves a lengthy process, and it can be difficult to identify, recruit and retain employees who already hold security clearances. If our employees are unable to obtain or retain security clearances, or if our employees who hold security clearances stop working for us, we may face delays in fulfilling contracts, or be unable to fulfill or secure new contracts, with any customer involved in classified work. Any breach of security for which we are responsible could seriously harm our business, damage our reputation and make us ineligible to work on any classified programs.
The classified work that we currently perform at one of our facilities subjects us to the industrial security regulations of the Department of Defense that are designed to safeguard against unauthorized access by foreigners and others to classified and other sensitive information. We may be subject to penalties for violations of these regulations and the U.S. government could terminate our contracts with it or decide not to renew them
80
Table of Contents
and such a situation could also impair our ability to obtain new contracts and subcontracts. The government may also change its procurement practices or adopt new contracting rules and regulations that could be costly to satisfy or that could impair our ability to obtain new contracts. See Risk Factors We must comply with the U.S. Foreign Corrupt Practices Act, or FCPA, and analogous non-U.S. anti-bribery statutes including the UK Anti-Bribery Act. Our third-party sales representatives or distributors failure to comply with such laws could subject us to, among other things, penalties and legal expenses that could harm our reputation and materially and adversely affect our business, financial condition and results of operations.
Any reduction in the capital resources or government funding of our customers could reduce our sales and impede our ability to generate revenue.
A significant portion of our sales are capital purchases by our customers. The spending policies of our customers could have a significant effect on the demand for our products. These policies are based on a wide variety of factors, including the resources available to make purchases, the spending priorities among various types of equipment, policies regarding spending during recessionary periods and changes in the political climate. In particular, certain customers can come under significant budgetary pressure and resort to cost-cutting measures.
Any changes in capital spending or changes in the capital budgets of our customers could significantly reduce demand for our products. The capital resources of our customers may be limited by the availability of equity or debt financing. In addition, a portion of our sales are to governmental and non-profit entities such as universities and hospitals, which are subject to unique budgetary pressures. Any reduction in spending or budget austerity measures could inhibit the ability of these customers to purchase our products.
Many of our large contracts have penalties for late deliveries.
In some cases, including through many of our fixed-price contracts, we have agreed to deliver a project by a scheduled date. If we fail to deliver the project as scheduled, we may be held responsible for costs associated with the delay, generally in the form of liquidated damages, in some cases up to the full value of the contract. We have in the past incurred penalties associated with late delivery on some of our contracts. In the event that a project is delayed, the total costs of the project could exceed our original estimates, and we could experience reduced profits or a loss for that project.
A failure or breach of our or our vendors information technology, or IT, data security infrastructure, or the security infrastructure of our products, or the discovery or exploitation of defects or vulnerabilities in the same, may subject us and our products to increased vulnerability to unauthorized access and cyberattacks and could materially and adversely impact our or our customers business, financial condition, reputation and operations.
We rely upon the capacity, reliability and security of our and our vendors IT and data security infrastructure and our ability to expand and continually update this infrastructure in response to the changing needs of our business. As we implement new systems or integrate existing systems, they may not perform as expected, which may result in liability or incurred costs, including litigation. We also face the challenge of supporting our older systems and implementing necessary upgrades. If we experience an issue with the functioning of an important IT system or a security breach of our IT systems, including during system upgrades and/or new system implementations, the resulting disruptions, including because of investigations or litigation, could have an adverse effect on our business, financial condition and operations. Furthermore, we collect and maintain information in digital form that is necessary to conduct our business, and we are increasingly dependent on our IT and data security infrastructure to operate our business. In the ordinary course of our business, we collect, store and transmit large amounts of confidential information, including intellectual property, proprietary business information and personal information. It is critical that we do so in a secure manner to maintain the confidentiality and integrity of such confidential information. We have established physical, electronic and organizational measures to safeguard and secure our systems to prevent data compromise and rely on
81
Table of Contents
commercially available systems, software, tools, and monitoring to provide security for our IT systems and the processing, transmission and storage of digital information. We have also outsourced elements of our IT systems and, as a result, a number of third-party vendors may or could have access to our confidential information.
Despite our implementation of security measures, our IT systems, like those of other companies, are vulnerable to damage or interruption from a variety of sources, including physical damage, telecommunications or network failures or interruptions, system malfunction, natural disasters, malicious human acts, terrorism and war. Such IT systems, including our servers, are additionally vulnerable to physical or electronic break-ins, security breaches from inadvertent or intentional actions by our employees, third-party service providers, contractors, consultants, business partners, and/or other third parties, or from cyber-attacks by malicious third parties (including the deployment of harmful malware, ransomware, denial-of-service attacks, social engineering, and other means to affect service reliability and threaten the confidentiality, integrity, and availability of information). For example, in February 2021, we experienced a ransomware attack that involved the unauthorized access to certain of our servers. While we were able to detect and stop the unauthorized access before any substantial amount of information was accessed and before the attacker was able to encrypt our systems, the attacker misappropriated certain personal and proprietary information and publicly published certain of such information. We reported the incident to the applicable government authorities in France, Germany and the United States. Additionally, one of our acquired subsidiaries experienced a ransomware attack in February 2020, prior to our acquisition of such subsidiary. The acquired subsidiary did not make any ransom payments and was able to restore its systems from backups. Although we have implemented additional security measures to prevent future ransomware attacks, we can provide no assurance that our IT systems, or those of the third parties upon which we rely, will not experience cybersecurity incidents in the future. The risk of a security breach or disruption, particularly through cyber-attacks or cyber intrusion, including by computer hackers, foreign governments, and cyber terrorists, has generally increased as the number, intensity, and sophistication of attempted attacks and intrusions from around the world have increased. We may not be able to anticipate all types of security threats, and we may not be able to implement preventive measures effective against all such security threats. The techniques used by cyber criminals change frequently, may not be recognized until launched, and can originate from a wide variety of sources, including outside groups such as external service providers, organized crime affiliates, terrorist organizations, or hostile foreign governments or agencies. It is possible that we or our third-party vendors may experience cybersecurity and other breach incidents that remain undetected for an extended period. Even when a security breach is detected, the full extent of the breach may not be determined immediately. The costs to us to mitigate network security issues, bugs, viruses, worms, malicious software programs and security vulnerabilities could be significant and, while we have implemented security measures to protect our IT and data security infrastructure, our efforts to address these issues may not be successful. There is also the potential for class action or other litigation as the result of such issues and the dissemination of personal information.
Any system failure, accident or security breach could result in disruptions to our operations or those of our customers. A material network breach in the security of our IT systems could include the theft of our intellectual property (including our trade secrets), customer information, human resources information or other confidential matter or the theft of the confidential information of our customers. To the extent that any disruption or security breach results in a loss or damage to our or our customers data, or an inappropriate disclosure of confidential, proprietary or customer information, it could cause significant damage to our reputation, affect our relationships with our customers, lead to claims against us, including civil litigation, and ultimately harm our business. In addition, we may be required to incur significant costs to protect against damage caused by these disruptions or security breaches in the future. If our IT systems fail and our redundant systems or disaster recovery plans are not adequate to address such failures, or if our business interruption insurance does not sufficiently compensate us for any losses that we may incur, our revenues and profits could be reduced and the reputation of our brand and our business could be materially and adversely affected.
We are also reliant on the security practices of our third-party service providers, which may be outside of our direct control. The services provided by these third parties are subject to the same risk of outages, other failures and security breaches described above. If these third parties fail to adhere to adequate security practices, or
82
Table of Contents
experience a breach of their systems, the data of our employees, customers and business associates may be improperly accessed, used or disclosed. In addition, our providers have broad discretion to change and interpret the terms of service and other policies with respect to us, and those actions may be unfavorable to our business operations. Our providers may also take actions beyond our control that could harm our business, including discontinuing or limiting our access to one or more services, increasing pricing terms, terminating or seeking to terminate our contractual relationship altogether, or altering how we are able to process data in a way that is unfavorable or costly to us. Although we expect that we could obtain similar services from other third parties, if our arrangements with our current providers were terminated, we could experience interruptions in our business, as well as delays and additional expenses in arranging for alternative cloud infrastructure services. Any loss or interruption to our systems or the services provided by third parties would adversely affect our business, financial condition and results of operations.
Failure to secure and protect our trade secrets or other confidential or proprietary information from disclosure or misappropriation could materially and adversely affect our business, competitiveness and financial condition.
We rely on trade secrets and confidentiality agreements to protect our unpatented know-how, technology, and other proprietary information, including unpatented proprietary radiation detection expertise, continuing technological innovation and other trade secrets some of which is licensed from third parties, and to develop and maintain our competitive position. With respect to our products, we consider trade secrets and know-how to be one of our primary sources of intellectual property rights. However, trade secrets and know-how can be difficult to protect. We seek to protect these trade secrets and other proprietary technology, in part, by entering into non-disclosure and confidentiality agreements with parties who have access to them, such as our employees, corporate collaborators, outside contractors, consultants, advisors, and other third parties. We also enter into confidentiality and invention or patent assignment agreements with our employees and consultants. We cannot guarantee that we have entered into such agreements with each party that may have or have had access to our trade secrets or proprietary information, including our technology and processes. Despite these efforts, any of these parties may breach the agreements and disclose our proprietary information, including our trade secrets, and we may not be able to obtain adequate remedies for such breaches. Enforcing a claim that a party illegally disclosed or misappropriated a trade secret is difficult, expensive, and time-consuming, and the outcome is unpredictable. In addition, some courts inside and outside the U.S. are less willing or unwilling to protect trade secrets. If any of our trade secrets were to be lawfully obtained or independently developed by a competitor or other third party, we would have no right to prevent them from using that technology or information to compete with us. If any of our trade secrets were to be disclosed to or independently developed by a competitor or other third party, it could have a material adverse effect on our competitive position, business, financial condition, and results of operations.
Our future success is dependent on our ability to retain key personnel, including our executive officers, and attract qualified personnel. If we lose the services of these individuals or are unable to attract new talent, our business will be materially and adversely affected.
Our future operating results depend in significant part upon the continued contributions of our key technical and senior management personnel, many of whom would be difficult to replace. We are particularly dependent on the continued service of Thomas D. Logan, our Chief Executive Officer and current Chairman of the Board (and expected Vice Chairman of the Board upon consummation of the business combination), Brian Schopfer, our Chief Financial Officer, and Mike Freed, our Chief Operating Officer.
Our future operating results also depend in significant part upon our ability to attract, train and retain qualified management, manufacturing and quality assurance, engineering, marketing, sales and support personnel. In particular, engineers skilled in the analog technologies used in certain of our products are in high demand and competition to attract such personnel is intense. In addition, the expected increase in construction of new NPPs may exacerbate the shortage of radiation engineers and other qualified personnel. We are continually recruiting
83
Table of Contents
such personnel; however, we cannot assure you that we will be successful in attracting, training or retaining such personnel now or in the future. There may be only a limited number of persons with the requisite skills to serve in these positions, and it may be increasingly difficult for us to hire such persons over time. The high demand for such personnel may increase the costs to us to recruit and retain employees.
The loss of any key employee, the failure of any key employee to perform in his or her current position, our inability to attract, train and retain skilled employees as needed or the inability of our officers and key employees to expand, train and manage our employee base could materially and adversely affect our business, financial condition and results of operations.
If we encounter manufacturing problems, or if our manufacturing facilities do not continue to meet federal, state or foreign manufacturing standards, we may be required to temporarily cease all or part of our manufacturing operations, which would result in delays and lost revenue.
Many of our products are complex and require the integration of a number of components from several sources of supply. We must manufacture and assemble these complex systems in commercial quantities in compliance with regulatory requirements and at an acceptable cost. In addition, the COVID-19 pandemic may impact the supply of key components such that we may not receive them in a timely manner, in sufficient quantities, or at reasonable cost. We may also experience limitations in the availability of qualified personnel as a result of shelter-in-place rules, quarantine requirements, or illness. If our manufacturing capacity does not keep pace with product demand, we will not be able to fulfill orders in a timely manner, which in turn may breach our obligations to our business partners or otherwise have a negative effect on our financial results and overall business, including as a result of litigation. Conversely, if demand for our products decreases, the fixed costs associated with excess manufacturing capacity may adversely affect our financial results.
Our manufacturing processes and the manufacturing processes of our third-party suppliers are required to comply with the FDAs Quality System Regulations, or QSR, which are medical device good manufacturing practices for any products imported into, or sold within, the United States. The QSR is a complex regulatory scheme that covers all aspects of medical device manufacture, from pre-production design validation and servicing, as such aspects bear upon the safe and effective use of the device and whether the device otherwise meets the U.S. Federal Food, Drug and Cosmetic Act, or FDCA. Other jurisdictions where our medical device products are distributed and sold have their own regulatory requirements that include quality and manufacturing requirements and controls. Furthermore, we are required to verify that our suppliers maintain facilities, procedures and operations that comply with our quality requirements. We are also subject to state licensing and other requirements and licenses applicable to manufacturers of medical devices, and we are required to comply with International Organization for Standardization, or ISO, quality system standards in order to produce products for sale in Europe and Canada, as well as various other foreign laws and regulations. Because our manufacturing processes include the production of diagnostic and therapeutic X-ray equipment and laser equipment, we are subject to the electronic product radiation control provisions of the FDCA, which requires that we file reports with the FDA, applicable states and our customers regarding the distribution, manufacturing and installation of these types of equipment. The FDA enforces the QSR and the electronic product radiation control provisions through inspections, both periodic and for cause. We have been, and will continue being subject to such inspections. FDA inspections usually occur every two to three years. During such inspections, the FDA may issue Inspectional Observations on Form FDA 483, listing instances where a manufacturer has failed to comply with the FDCA, applicable regulations and procedures, or previous warning letters.
Sometimes inspections result in warning letters which are publicly available and can result in adverse publicity. Our failure to take prompt and satisfactory corrective action in response to an adverse inspection or our failure to comply with applicable regulatory requirements and standards could result in enforcement actions, including a shutdown of our manufacturing operations, a recall of our products, civil or criminal penalties, or other sanctions, which would cause our sales and business to suffer. Any inspection or government action based on alleged violations of law could require us to expend significant time and resources in response and could generate
84
Table of Contents
negative publicity. The occurrence of any event or penalty described above may inhibit our ability to keep our products on the market and generate revenue. In addition, because some foreign regulatory approvals require approvals or clearances from the FDA, any failure to comply with FDA requirements may also disrupt our sales of products in other countries. We cannot assure you that the FDA or other governmental authorities would agree with our interpretation of applicable regulatory requirements, or that we, or our third-party suppliers have in all instances fully complied with all applicable requirements. If any of these events occur, our reputation could be harmed, we could lose customers and there could be a material adverse effect on our business, financial condition and operations, including as the result of litigation.
If we cannot achieve the required level and quality of production, we may need to outsource production or rely on licensing and other arrangements with third parties who possess sufficient manufacturing facilities and capabilities in compliance with regulatory requirements. Even if we could outsource needed production or enter into licensing or other third-party arrangements, this could reduce our gross margin and expose us to the risks inherent in relying on others. We also cannot assure you that our suppliers will deliver an adequate supply of required components on a timely basis, or that they will adequately comply with the QSR. Failure to obtain these components on a timely basis would disrupt our manufacturing processes and increase our costs, which would harm our operating results.
Our customers localization requirements, in particular in China, India and South Korea, could materially and adversely affect our business.
Many emerging markets, including China, India and South Korea, impose localization requirements which favor locally based component manufacturers and which require some degree of technology transfer to local manufacturers. Over time, such localization requirements could limit our ability to sell into such markets and could affect our ability to maintain our trade secrets. In the past, government customers have, as a condition of funding, imposed localization requirements that require the transfer of certain technology (e.g., manufacturing technology) to local manufacturers, and this requirement has affected our ability to monitor and maintain control over our intellectual property. We may be subject to similar requirements as a condition of funding in the future.
Our operations, and the operations of our suppliers, distributors or customers, could be subject to natural and manmade disasters and other business disruptions, which could materially and adversely affect our business and increase our expenses.
Our operations could be subject to natural disasters and other business disruptions, which could harm our future revenue and financial condition and increase our costs and expenses. For example, some of our facilities are located in areas with earthquake fault lines or in hurricane zones. In the event of a major earthquake or other natural or manmade disaster, we could experience business interruptions, destruction of or damage to facilities and/or loss of life, any of which could materially and adversely affect our business and increase our expenses.
Legal and Regulatory Risks
We are subject to, or may otherwise be impacted by, a variety of federal, state, local and foreign laws and regulatory regimes. Failure to comply with such laws and regulations could subject us to, among other things, penalties and legal expenses which could have a material and adverse effect on our business, or such laws and regulations could otherwise impact us, directly or indirectly, in a manner that has a material and adverse effect on our business.
Our business is subject to regulation by various federal, state, local and foreign governmental agencies. In the United States, such regulation includes the radioactive material exposure and nuclear facilities regulatory activities of the NRC, the anti-trust regulatory activities of the Federal Trade Commission and Department of Justice, the import/export regulatory activities of the Department of Commerce, the Department of State and the Department of Treasury, the regulatory activities of the Occupational Safety and Health Administration, the
85
Table of Contents
regulations of the FDA, the environmental regulatory activities of the Environmental Protection Agency, the labor regulatory activities of the Equal Employment Opportunity Commission and tax and other regulations by a variety of regulatory authorities in each of the areas in which we conduct business. We are also subject to regulation in other countries where we conduct business. In certain jurisdictions, such regulatory requirements may be more stringent than in the United States. We are also subject to a variety of U.S. federal and state employment and labor laws and regulations, including the Americans with Disabilities Act, the Federal Fair Labor Standards Act, the Worker Adjustment and Restructuring Notification Act, which requires employers to give affected employees at least 60 days notice of a plant closing or mass layoff, and other regulations related to working conditions, wage-hour pay, overtime pay, employee benefits, anti-discrimination and termination of employment. We are also subject to the employment and labor laws and regulations of the foreign jurisdictions, including France and Germany, where many of our employees are located.
Noncompliance with applicable regulations or requirements could subject us to investigations, sanctions, enforcement actions, disgorgement of profits, fines, damages, civil and criminal penalties, injunctions or debarment from government contracting or subcontracting. In addition, from time to time we have received, and may in the future receive, correspondence from former employees terminated by us who threaten to bring claims against us alleging that we have violated one or more labor or employment regulations. An adverse outcome in any such litigation could require us to pay damages.
Governmental enforcement actions could harm our business, financial condition and results of operations. If any governmental sanctions are imposed, or if we do not prevail in any civil or criminal litigation, our business, financial condition and results of operations could be materially adversely affected. In addition, responding to any action could be costly and result in a significant diversion of managements attention and resources.
We and our customers operate in highly regulated industries that require us and them to obtain, and comply with, federal, state, local and foreign government permits and approvals.
We and our customers operate in a highly regulated environment. Many of our products and services must comply with various domestic and international standards that are used by regulatory and accreditation bodies for approving such services and products. Many of our products, particularly those offered by our Industrial segment, are subject to an array of product testing under extreme temperature, pressure, radiation and seismic conditions, known collectively as a qualification, for any given nuclear reactor design. The qualification is typically owned by the party who pays for the testing and so, in certain cases, we license such qualifications from a third party. In addition, many of our products and services, particularly those offered by our Medical segment, must be certified by the National Voluntary Laboratory Accreditation Program in the United States and by other governmental agencies in international markets. The termination of any such accreditation or our failure to obtain and maintain required qualification or accreditation for our products and services may adversely affect our revenue and results of operations.
Changes in these standards and accreditation requirements may also result in our having to incur substantial costs to adapt our products. Such adaptations may introduce quality assurance issues during transition as new features and products may not perform as expected. Additionally, changes affecting radiation protection practices, including new understandings of the hazards of radiation exposure and corresponding changes in regulations, may impact how our services are used by our customers and may, in some circumstances, cause us to alter our products and services.
Our subsidiary Sun Nuclear offers oncology quality assurance products for diagnostic imaging and radiation therapy. These products may be relied upon by customers as part of their quality assurance programs for regulatory compliance, and thus could subject the company to potential risk of regulatory noncompliance or enforcement action by state or federal regulatory agencies, including but not limited to the NRC, Agreement State radiation safety agencies, the Food and Drug Administration, or FDA, the Center for Disease Control and Prevention, or CDC, and other agencies.
86
Table of Contents
In addition, our customers are required to obtain, and to comply with, federal, state, local and foreign government licenses, permits and approvals with respect to either their facilities or possession and use of radioactive sources or other radioactive materials. For example, federal agencies such as the NRC and FDA, Agreement State agencies, and others have certain regulatory responsibilities regarding medical devices, radiopharmaceuticals, and other medical products that utilize radioactive material. Any of these licenses, permits or approvals may be subject to denial, revocation or modification under various circumstances. Failure to obtain or comply with the conditions of licenses, permits or approvals may adversely affect our customers operations by suspending their activities or delaying or preventing the receipt of radioactive sources or other radioactive materials, and may subject them to penalties and other sanctions. Although existing licenses, permits or approvals are routinely renewed by various regulators, renewal could be denied or jeopardized by various factors, including but not limited to:
| | failure to comply with environmental and safety laws and regulations; |
| | failure to comply with permit conditions or violations found during inspections or otherwise; |
| | local community, political or other opposition; |
| | executive action; and |
| | legislative action. |
Furthermore, if new environmental legislation or regulations are enacted or existing laws or regulations are amended or are interpreted or enforced differently, our customers may be required to obtain additional operating licenses, permits or approvals. Regulatory issues experienced by our customers may lead to delay or cancellation of their orders for our products and services or the discontinuance of future orders. We cannot assure you that we or our customers will be able to meet all potential regulatory challenges.
Changes in industry standards and governmental regulations may increase our expenses or reduce demand for our products or services.
We compete in markets in which we and our customers must comply with supranational, federal, state, local, and other jurisdictional regulations, such as regulations governing health and safety, the environment, and electronic communications, and market standardizations. We develop, configure, and market our products and services to meet customer needs created by these regulations and standards. These regulations and standards are complex, change frequently, have tended to become more stringent over time, and may be inconsistent or conflicting across jurisdictions. Any significant change or delay in implementation in any of these regulations or standards (or in the interpretation, application, or enforcement thereof) could reduce or delay demand for our products and services, increase our costs of producing or delay the introduction of new or modified products and services, or could restrict our existing activities, products, and services. In addition, in certain of our markets our growth depends in part upon the introduction of new regulations or implementation of industry standards on the timeline we expect. In these markets, the delay or failure of governmental and other entities to adopt or enforce new regulations or industry standards, or the adoption of new regulations or industry standards which our products and services are not positioned to address, could adversely affect demand. In addition, regulatory deadlines or industry standard implementation timelines may result in substantially different levels of demand for our products and services from period to period.
We operate in a highly litigious industry and are, thus, subject to risks related to legal claims and proceedings filed by or against us, and adverse outcomes in these matters may materially harm our business.
We are subject to various claims, disputes, investigations, demands, arbitration, litigation, or other legal proceedings. Legal claims and proceedings may relate to labor and employment, commercial arrangements, intellectual property, disputes with customers or business partners, breach of contract, environmental, health and safety, property damage, theft, consumer protection, class action, mass tort and product liability, personal injury, false advertising, unfair competition or unfair trade practices, public or private nuisance, whistleblower
87
Table of Contents
litigation, fiduciary duties of our directors and officers, securities, Medicare and Medicaid reimbursement claims, false claims, radioactive contamination, indemnity, insurance and various other matters. Legal matters are inherently uncertain and we cannot predict the duration, scope, cost, outcome or consequences of such matters. Legal matters are expensive and time-consuming to defend, settle, and/or resolve, even if successfully, and may require us to implement certain remedial measures that could prove costly or disruptive to our business and operations and could result in civil or criminal fines, penalties, consent decrees, changes in business practices and exclusion from participation in various government healthcare-related programs. The unfavorable resolution of one or more of these matters could have an adverse impaact on our business, results of operations and financial condition.
We may incur material losses and expenses as a result of products liability claims brought against us.
We face an inherent business risk of exposure to products liability claims, with or without merit. This includes where our products are found to be defective in design or manufacture, a misstatement is found on product labels or marketing materials, including (but not limited to) in product warnings and instructions, or where our or our agents conduct is found to fall below the standard of care for a similarly situated medical device company. Accordingly, we should expect, in the ordinary course of business, to encounter class actions, mass tort actions, claims that allege our marketed products or products in development are mislabeled, mischaracterized or defective and violate applicable consumer protection statutes or FDA regulations or have caused, or could cause, serious adverse events or injury, including latent injury, and claims that our products have been, or should be recalled due to safety or warning defects. As discussed above, if our insurance coverage is inadequate to cover such claims or actions, we must pay the amount of any settlement or judgment in excess of the policy limits. Our failure to maintain adequate insurance coverage or failure to successfully defend against such claims, lawsuits and issues could have a material adverse effect on our business, financial condition, results of operations or cash flows.
Legal, political and economic uncertainty surrounding the exit of the United Kingdom from the European Union, or Brexit, and the implementation of the trade and cooperation agreement between the United Kingdom and the European Union could materially and adversely affect our business.
In June 2016, voters in the United Kingdom approved a referendum to withdraw the United Kingdoms membership from the European Union, which is commonly referred to as Brexit. The United Kingdoms withdrawal from the European Union occurred on January 31, 2020, but the United Kingdom remained in the European Unions customs union and single market for a transition period that expired on December 31, 2020. On December 30, 2020, the United Kingdom and the European Union entered into the Trade and Cooperation Agreement, which was applied on a provisional basis from January 1, 2021. While the economic integration does not reach the level that existed during the time the United Kingdom was a member state of the European Union, the Trade and Cooperation Agreement sets out preferential arrangements in areas such as trade in goods and in services, digital trade and intellectual property. Negotiations between the United Kingdom and the European Union are expected to continue in certain other areas which are not covered by the Trade and Cooperation Agreement. The long-term effects of Brexit will depend on the effects of the implementation and application of the Trade and Cooperation Agreement and any other relevant agreements between the United Kingdom and the European Union.
We have operations in the United Kingdom and the European Union and, as a result, we face risks associated with the potential uncertainty and disruptions that may follow Brexit and the implementation and application of the Trade and Cooperation Agreement, including with respect to volatility in exchange rates and interest rates, disruptions to the free movement of data, goods, services, people and capital between the United Kingdom and the European Union and potential material changes to the regulatory regime applicable to our operations in the United Kingdom. The uncertainty concerning the United Kingdoms future legal, political and economic relationship with the European Union could adversely affect political, regulatory, economic or market conditions in the European Union, the United Kingdom and worldwide and could contribute to instability in global political institutions, regulatory agencies and financial markets. These developments, or the perception that any of them
88
Table of Contents
could occur, have had and may continue to have a material adverse effect on global economic conditions and the stability of global financial markets and could significantly reduce global market liquidity and limit the ability of key market participants to operate in certain financial markets. In particular, it could also lead to a period of considerable uncertainty in relation to the United Kingdom financial and banking markets, as well as to the regulatory process in Europe. Asset valuations, currency exchange rates and credit ratings may also be subject to increased market volatility.
We may also face new regulatory costs and challenges as a result of Brexit that could have a material adverse effect on our operations. For example, as of January 1, 2021, the United Kingdom lost the benefits of global trade agreements negotiated by the European Union on behalf of its members, which may result in increased trade barriers that could make our business activities in areas that are subject to such global trade agreements more difficult. In addition, Brexit could lead to legal uncertainty and potentially divergent national laws and regulations as the United Kingdom determines which laws of the European Union to replace or replicate. There may continue to be economic uncertainty surrounding the consequences of Brexit that adversely impact customer confidence resulting in customers reducing their spending budgets on our services, which could materially adversely affect our business, financial condition and results of operations.
The ongoing instability and uncertainty surrounding Brexit and the implementation and application of the Trade and Cooperation Agreement, could require us to restructure our business operations in the United Kingdom and the European Union and could have an adverse impact on our business and employees in the United Kingdom and European Union.
Enhanced international tariffs, including tariffs that affect our products or components within our products, other trade barriers or global trade wars or domestic preferences could increase our costs and materially and adversely affect our business operations and financial condition.
Our global business could be negatively affected by trade barriers and other governmental protectionist measures, any of which can be imposed suddenly and unpredictably. There is currently significant uncertainty about the future trade relationships between the United States and various other countries, most significantly China, with respect to trade policies, treaties, government regulations and tariffs.
Since the beginning of 2018, there has been increasing public threats and, in some cases, legislative or executive action, from United States and foreign leaders regarding instituting tariffs against foreign imports of certain materials. During the last half of calendar year 2018, the federal government imposed a series of tariffs ranging from 7.5% to 25% on a variety of imports from China. These tariffs affect certain components that we import into the United States from our suppliers. China has responded to these tariffs with retaliatory tariffs ranging from 5% to 25% on a wide range of products from the United States, which include certain of our products. Higher duties on existing tariffs and further rounds of tariffs have been announced or threatened by the United States and Chinese leaders. Although the United States and China signed an initial trade deal in January 2020 and China announced a one year tariff exemption for medical linear accelerators in September 2019, there is no assurance that the trade deal will be signed or that the exemption on medical linear accelerators will continue beyond one year or that we will continue to qualify for such exemption. Additionally, the United States has threatened to impose tariffs on goods imported from other countries, which could also impact our or our customers operations. If these tariffs continue, if additional tariffs are placed on certain of our components or products, or if any related counter-measures are taken by China, the United States or other countries, our business, financial condition and results of operations may be materially harmed. The imposition of tariffs could also increase our costs and require us to raise prices on our products, which may negatively impact the demand for our products in the affected market. If we are not successful in offsetting the impact of any such tariffs, our revenue, gross margins and operating results may be adversely affected.
These tariffs are subject to a number of uncertainties as they are implemented, including future adjustments and changes. The ultimate reaction of other countries and the impact of these tariffs or other actions on the United
89
Table of Contents
States, China, the global economy and our business, financial condition and results of operations, cannot be predicted at this time, nor can we predict the impact of any other developments with respect to global trade. Further, the imposition of additional tariffs by the United States could result in the adoption of additional tariffs by China and other countries, as well as further retaliatory actions by any affected country. Any resulting trade war could negatively impact the global market for medical devices, including radiation therapy devices, and could have a significant adverse effect on our business. These developments may have a material adverse effect on global economic conditions and the stability of global financial markets, and they may significantly reduce global trade and, in particular, trade between China and the United States. Any of these factors could depress economic activity, restrict our access to customers and have a material adverse effect on our business, financial condition and results of operations.
We must comply with the U.S. Foreign Corrupt Practices Act, or FCPA, and analogous non-U.S. anti-bribery statutes including the UK Bribery Act. Our third-party sales representatives or distributors failure to comply with such laws could subject us to, among other things, penalties and legal expenses that could harm our reputation and materially and adversely affect our business, financial condition and results of operations.
We are required to comply with the United States Foreign Corrupt Practices Act (FCPA) which makes it unlawful to engage in bribery or to make any payments or provide any other benefits, directly or indirectly, to foreign officials for the purpose of obtaining or retaining business or to secure any other improper advantage. The FCPA also requires us, as a publicly traded company, to keep accurate books, records and accounts, and to maintain an effective system of internal accounting controls.
We operate, directly or indirectly, in more than one hundred countries around the world, many of which pose a high risk of corruption. In many countries, we also have government customers, and we utilize a network of third-party sales representatives and distributors. Based on these factors and others, our business involves a significant risk of potential FCPA violations.
All Mirion employees are informed of our responsibilities under the FCPA in the Mirion Code of Ethics and Conduct, and compliance with the FCPA is specifically mandated in detailed provisions of our agreements with third-party sales representatives and distributors. In addition, we provide live training on FCPA compliance on a regular basis for our employees who are involved in functions that necessitate such knowledge and training.
Before we became a public company, we were not subject to the accounting provisions of the FCPA. Nevertheless, as a matter of course, we continuously review and, when warranted, update and enhance our systems, procedures, contracting processes, third-party due diligence, auditing and recordkeeping to address our FCPA compliance obligations and mitigate FCPA compliance risk. In spite of this, based on the jurisdictions where we operate, the fact that we have government customers, and our use of a network of third-party sales representatives and distributors, there remains a risk that one or more employees or third parties, acting on behalf of Mirion, might engage in conduct for which we might be held responsible under the FCPA. On occasion, we may terminate distribution or other agreements with sales channel partners operating in certain non-U.S. jurisdictions based on our ongoing compliance program. This could materially impact our ability to do business in jurisdictions where we are unable to enter into agreements with alternative partners that meet our compliance standards, which could materially and adversely impact our competitive position in such jurisdictions, as well as our business, financial condition, results of operations or cash flows.
If our employees, third-party sales representatives and distributors or other agents are found to have engaged in such practices, we could suffer (i) severe penalties, including criminal and civil penalties, disgorgement, temporary or permanent debarment from public contracts, and (ii) other remedial measures, including compliance policy and procedural enhancements, improved internal controls, audits, improved compliance training and potentially employee discipline, any of which could have an adverse impact on our business, financial condition, results of operations and liquidity. Any investigation of any potential violations of the FCPA or other anti-corruption laws by U.S. or foreign authorities also could have an adverse impact on our business, financial condition and results of operations.
90
Table of Contents
Certain foreign companies, including some of our competitors, are not subject to prohibitions as strict as those under the FCPA or, even if subjected to strict prohibitions, such prohibitions may be laxly enforced in practice. If our competitors engage in corruption, extortion, bribery, pay-offs, theft or other fraudulent practices, they may receive preferential treatment from personnel of some companies, giving our competitors an advantage in securing business, or from government officials, who might give them priority in obtaining new licenses, which would put us at a disadvantage.
Legal compliance with import and export controls, as well as with sanctions, in the United States and other countries, is complex, and compliance restrictions and expenses could materially and adversely impact our revenue and supply chain.
We are subject to applicable import laws, export controls and economic sanctions laws and regulations, including rule changes, evolving enforcement practices, and other actions resulting from Executive Orders issued by the Trump and Biden administrations. Changes in import and export control or trade sanctions laws may restrict our business practices, including cessation of business activities in sanctioned countries or with sanctioned entities, and may result in claims for breach of existing contracts and modifications to existing compliance programs and training schedules. Violations of the applicable export or import control, or economic sanctions laws and regulations, such as an export to an embargoed country, or to a denied party, or the export of a product without the appropriate governmental license, may result in penalties, including fines, debarments from export privileges, and loss of authorizations needed to conduct aspects of our international business, and may harm our ability to enter into contracts with our customers who have contracts with the U.S. government. A violation of the laws and regulations enumerated above could have an adverse effect on our business, results of operations and financial condition. Additionally, we require our sales channel partners in certain non-U.S. jurisdictions to comply with certain standards as part of our trade compliance program and regularly review our partners performance of their compliance obligations. As part of these reviews, it is possible we may discover that certain partners do not meet our standards, and we may be required to terminate agreements with any non-compliant partners. Any such actions could materially and adversely impact our ability to do business in jurisdictions where we are unable to enter into agreements with alternative partners that meet our compliance standards. This in turn could materially and adversely impact our competitive position in such jurisdictions, as well as on our business, financial condition, results of operations or cash flows.
Any actual or perceived failure to comply with evolving data privacy and data security laws and regulations in the jurisdictions where we operate, both inside and outside of the United States, could lead to government enforcement actions (which could include civil or criminal penalties), private litigation or adverse publicity and could materially and adversely affect our business.
Privacy and data security have become significant issues in the United States, Europe and in many other jurisdictions where we conduct our operations. Our collection, processing, distribution, and storage of personal information is subject to a variety of laws and regulations both in the United States and abroad, which could limit the way we market and provide our products and services. Compliance with these privacy and data security requirements is rigorous and time-intensive and may increase our cost of doing business and, despite these efforts, there is a risk that we fail to comply and may become subject to government enforcement actions, fines and penalties, litigation and reputational harm, which could materially and adversely affect our business, financial condition and results of operations. In addition, the regulatory framework for the handling of personal and confidential information is rapidly evolving and is likely to remain uncertain for the foreseeable future as new privacy laws are being enacted globally and existing laws are being updated and strengthened.
For example, in May 2018, the General Data Protection Regulation, or the GDPR, superseded prior European Union data protection legislation, and it imposes more stringent European Union data protection requirements, and provides for greater penalties for noncompliance. Under the GDPR, fines of up to 20 million euro or up to 4% of the annual global turnover of the infringer, whichever is greater, could be imposed. The GDPR is wide-ranging in scope and imposes numerous additional requirements on companies that process personal data,
91
Table of Contents
including imposing special requirements in respect of the processing of personal data, requiring that consent of individuals to whom the personal data relates is obtained in certain circumstances, requiring additional disclosures to individuals regarding data processing activities, requiring that safeguards are implemented to protect the security and confidentiality of personal data, creating mandatory data breach notification requirements in certain circumstances, and requiring that certain measures (including contractual requirements) are put in place when engaging third-party processors. The GDPR also provides individuals with various rights in respect of their personal data, including rights of access, erasure, portability, rectification, restriction and objection.
Further, the United Kingdoms vote in favor of exiting the European Union, often referred to as Brexit, and ongoing developments in the United Kingdom have created uncertainty with regard to data protection regulation in the United Kingdom. As of January 1, 2021, and the expiry of transitional arrangements agreed to between the United Kingdom and the European Union, data processing in the United Kingdom is governed by a United Kingdom version of the GDPR (combining the GDPR and the Data Protection Act 2018), exposing us to two parallel regimes, each of which potentially authorizes similar fines and other potentially divergent enforcement actions for certain violations. Pursuant to the Trade and Cooperation Agreement, which went into effect on January 1, 2021, the United Kingdom and the European Union agreed to a specified period during which the
United Kingdom will be treated like a European Union member state in relation to transfers of personal data to the United Kingdom for four months from January 1, 2021. This period may be extended by two further months. Unless the European Commission makes an adequacy finding in respect of the United Kingdom before the expiration of such specified period, the United Kingdom will become an inadequate third country under the GDPR and transfers of data from the European Economic Area to the United Kingdom will require a transfer mechanism, such as the standard contractual clauses. Furthermore, following the expiration of the specified period, there will be increasing scope for divergence in application, interpretation and enforcement of the data protection law as between the United Kingdom and the European Union.
Other jurisdictions outside the European Union are similarly introducing or enhancing privacy and data security laws, rules and regulations, which could increase our compliance costs and the risks associated with noncompliance. For example, California recently enacted the California Consumer Privacy Act, or the CCPA, which creates new individual privacy rights for California consumers (as defined in the law) and places increased privacy and security obligations on companies handling personal information of consumers or households. The CCPA, which went into effect on January 1, 2020, requires covered companies to provide new disclosure to consumers about such companies data collection, use and sharing practices, provide methods for such consumers to access and delete their personal information, with exceptions, as well as allowing consumers to opt-out of certain sales or transfers of their personal information. The CCPA provides for civil penalties for violations and further provides consumers with a new private right of action in the event of a data breach involving certain sensitive information as a result of the business failure to implement reasonable security measures. This private right of action may increase the likelihood of, and risks associated with, data breach litigation. The California Attorney Generals enforcement authority under the CCPA became effective July 1, 2020, and it remains unclear how various provisions of the CCPA will be interpreted and enforced. As currently written, the CCPA impacts certain of our business activities and exemplifies the vulnerability of our business to the evolving regulatory environment related to personal information. A ballot initiative from privacy rights advocates intended to augment and expand the CCPA called the California Privacy Rights Act, or the CPRA, was passed in November 2020 and will take effect in January 2023 (with a look back to January 2022). The CPRA significantly modifies the CCPA, including by imposing additional obligations on covered companies and expanding consumers rights with respect to certain sensitive personal information, potentially resulting in further uncertainty and requiring us to incur additional costs and expenses in an effort to comply. The CPRA also creates a new state agency that will be vested with authority to implement and enforce the CCPA and the CPRA. In addition, all 50 states have laws including obligations to provide notification of security breaches of computer databases that contain personal information to affected individuals, state officers and others. Aspects of the CCPA, the CPRA, and other laws and regulations relating to data protection, privacy, and information security, as well as their enforcement, remain unclear, and we may be required to modify our practices in an effort to comply with them.
92
Table of Contents
We cannot yet fully determine the impact these or future laws, rules, and regulations concerning data privacy and security may have on our business or operations. These laws, rules and regulations may be inconsistent from one jurisdiction to another, subject to differing interpretations and may be interpreted to conflict with our practices. Additionally, we may be bound by contractual requirements applicable to our collection, use, processing and disclosure of various types of data, including personal information, and may be bound by, or voluntarily comply with, self-regulatory or other industry standards relating to these matters. Compliance with U.S. and international privacy and data security laws and regulations could require us to take on more onerous obligations in our contracts and restrict our ability to collect, use and disclose data. Because the interpretation and application of data protection laws, regulations, standards and other obligations are still uncertain, and often contradictory and in flux, it is possible that the scope and requirements of these laws may be interpreted and applied in a manner that is inconsistent with our practices and our efforts to comply with the evolving data protection rules may be unsuccessful. Failure to comply with U.S. and international privacy and data security laws and regulations could result in government enforcement actions (which could include civil or criminal penalties), private litigation or adverse publicity and could negatively affect our results of operations and business. Claims that we have violated individuals privacy rights, failed to comply with privacy and data security laws, or breached our contractual obligations, even if we are not found liable, could be expensive and time consuming to defend and could result in adverse publicity that could increase our operation costs, impact our financial performance and adversely affect enrollments.
Our ability to compete successfully and achieve future growth will depend on our ability to obtain, maintain, protect, defend and enforce our intellectual property and to operate without infringing, misappropriating or otherwise violating the intellectual property of others.
Our intellectual property, including our design, engineering, manufacturing and testing know-how, is an essential asset of our business. Failure to adequately protect our intellectual property rights could result in our competitors or other third parties offering similar products and services, potentially resulting in the loss of our competitive advantage and a decrease in our revenue, which would adversely affect our business, financial condition and results of operations. We attempt to protect our intellectual property rights through patents, trademarks, copyrights, trade secret laws, non-disclosure agreements, confidentiality procedures, employee disclosure and invention assignment agreements and other contractual provisions. We cannot guarantee that any of our pending patent applications or other applications for intellectual property registrations will be issued or granted or that our existing and future intellectual property rights will be sufficiently broad to protect our proprietary technology. While a presumption of validity exists with respect to United States patents issued to us, there can be no assurance that any of our patents, patent applications, or other intellectual property rights will not be, in whole or in part, opposed, contested, challenged, invalidated, circumvented, designed around, or rendered unenforceable. If we fail to obtain issuance of patents or registration of other intellectual property, or our patent claims or other intellectual property rights are rendered invalid or unenforceable, or narrowed in scope, pursuant to, for example, judicial or administrative proceedings including re-examination, post-grant review, inter partes, interference, opposition, or derivation proceedings, the coverage of patents and other intellectual property rights afforded our products could be impaired. Even if we are to obtain issuance of further patents or registration of other intellectual property, such intellectual property could be subjected to attacks on ownership, validity, enforceability, or other legal attacks. Any such impairment or other failure to obtain sufficient intellectual property protection could materially and adversely affect our business, financial condition and results of operations, including forcing us to, among other things, rebrand or re-design our affected products. Moreover, our patents and patent applications may only cover particular aspects of our products, and competitors and other third parties may be able to circumvent or design around our patents. Competitors may develop and obtain patent protection for more effective technologies, designs or methods. There can be no assurance that third parties will not create new products or methods that achieve similar or better results without infringing upon patents we own. If these developments were to occur, it could have an adverse effect on our business, financial condition and results of operations.
While we generally seek or apply for patent protection as and if we deem appropriate, based on the then-current facts and circumstances, we also rely upon unpatented proprietary radiation detection expertise, continuing
93
Table of Contents
technological innovation and other trade secrets some of which is licensed from third parties, to develop and maintain our competitive position. We seek to enter into confidentiality agreements with our employees and third parties who have access to our confidential or proprietary information; however, we may fail to enter into such agreements with all parties who have access to our confidential information, such agreements are often limited in duration and such agreements could be breached, and therefore they may not provide meaningful protection for our trade secrets, including our proprietary radiation detection and measurement expertise. Similarly, while we seek to enter into agreements with all of our employees and contractors who develop intellectual property during their engagement with us to assign the rights in such intellectual property to us, we may fail to enter into such agreements with all relevant employees and contractors, such agreements may be breached or may not be self-executing, and we may be subject to claims that such employees or contractors misappropriated relevant rights from their previous employers.
We cannot guarantee that the steps we have taken to protect our intellectual property will be adequate to prevent infringement of our intellectual property rights or misappropriation of our technology, trade secrets or know-how. It is possible that our efforts to protect our intellectual property rights may not:
| | prevent others from obtaining knowledge of our trade secrets through independent development or other access by legal means; |
| | prevent our competitors or other third parties from independently developing similar products, duplicating our products or designing around the patents owned by us; |
| | prevent third-party patents from having an adverse effect on our ability to do business; |
| | provide adequate protection for our intellectual property rights; |
| | prevent disputes with third parties regarding ownership of, or exclusive rights to, our intellectual property; |
| | prevent disclosure of our trade secrets and know-how to third parties or into the public domain; |
| | prevent the challenge, invalidation or circumvention of our existing patents; |
| | result in patents that lead to commercially viable products or provide competitive advantages for our products; and |
| | result in issued patents and registered trademarks from any of our pending applications. |
The laws of foreign countries also may not adequately protect our intellectual property rights. Many U.S. companies have encountered substantial infringement, misappropriation or other violations of their intellectual property rights in foreign countries. Furthermore, because filing, prosecuting, maintaining, and defending our intellectual property in all countries throughout the world would be prohibitively expensive we have not applied for patent protection or trademark or other intellectual property registrations in all jurisdictions in which we currently, or may in the future, operate. Because we conduct a substantial portion of our operations and a majority of our sales have been outside of the United States, we have significant exposure to foreign intellectual property risks.
Others have in the past attempted, and may in the future attempt, to copy or otherwise obtain and use our intellectual property without our consent. For example our customers or their end users customers may attempt to copy or otherwise obtain and use our intellectual property without our consent. Monitoring the unauthorized use of our intellectual property is difficult and we may fail to identify instances where a third party is infringing, misappropriating or otherwise violating our intellectual property. If we fail to protect our intellectual property rights adequately, we may lose an important advantage in the markets in which we compete.
We are currently party to, and may in the future initiate, litigation against one or more third parties to preserve or enforce our intellectual property rights or to challenge the validity and scope of proprietary rights asserted by others, and we could face counterclaims. Such efforts may be insufficient or ineffective, and any of our
94
Table of Contents
intellectual property rights may be challenged, which could result in them being narrowed in scope or declared invalid or unenforceable. Furthermore any such legal disputes we may initiate with our customers or companies with whom we have manufacturing relationships could substantially harm our relationships and sales. An adverse outcome in any such proceeding could subject us to significant liability for damages or invalidate our proprietary rights. Such litigation could result in significant expense to us and divert the efforts of our technical and management personnel, whether or not such litigation is ultimately determined in our favor. Further, adequate remedies may not be available in the event of an unauthorized use or disclosure of our trade secrets and manufacturing expertise. Any of the foregoing could materially and adversely affect our business, financial condition and results of operations.
We may need to defend ourselves against third-party claims that we are infringing, misappropriating or otherwise violating others intellectual property rights, which could divert managements attention, cause us to incur significant costs and prevent us from selling or using the technology to which such rights relate.
Our commercial success depends in part on avoiding infringement, misappropriation or other violations of the intellectual property and proprietary rights of third parties and other intellectual property-related disputes. There may be intellectual property rights held by others, including issued or pending patents and registered trademarks, that cover significant aspects of our technologies, products or services, and we cannot be sure that we are not infringing or violating, and have not infringed or violated, any third-party intellectual property rights. From time to time, third parties have claimed and may claim in the future that we have infringed upon, misappropriated or misused their proprietary rights, and we may be unaware of existing third-party intellectual property rights that we may be infringing.
Any of these events or claims could result in litigation. Such litigation could result in significant expense to us and divert the efforts of our technical and management personnel, whether or not such litigation is ultimately determined in our favor. In the event of an adverse result in such litigation, we could be required to pay substantial damages, cease the manufacture, use and sale of certain products, expend significant resources to develop or acquire non-infringing technology, discontinue the use of certain processes, obtain licenses to use the infringed technology or indemnify our customers. Product development or obtaining a license would likely result in significant expense to us and divert the efforts of our technical and management personnel. We cannot assure you that we would be successful in such development or acquisition or that such licenses would be available on reasonable terms, or at all. If we cannot license or develop a non-violating alternative, we would be forced to limit or stop sales of our offerings and may be unable to effectively compete. Moreover, there could be public announcements of the results of hearings, motions or other interim proceedings or developments and if securities analysts or investors perceive these results to be negative, it could have a substantial adverse effect on the price of our stock. Any of these results would materially and adversely affect our business, financial condition and results of operations.
Our use of open source software could negatively affect our ability to sell our products and subject us to possible litigation.
A portion of our products incorporate so-called open source software, and we may incorporate additional open source software in the future. Open source software is generally licensed by its authors or other third parties under open source licenses. If we fail to comply with these licenses, we may be subject to certain conditions, including requirements that we offer our products that incorporate the open source software for no cost, that we make available source code for modifications or derivative works we create based upon, incorporating or using the open source software and/or that we license such modifications or derivative works under the terms of the particular open source license. If an author or other third party that distributes such open source software were to allege that we had not complied with the conditions of one or more of these licenses, we could be required to incur significant legal expenses defending against such allegations and could be subject to significant damages, enjoined from the sale of our products that contained the open source software and required to comply with the foregoing conditions, which could disrupt the distribution and sale of some of our products and adversely affect our business, financial condition and results of operations.
95
Table of Contents
Our obligations to indemnify our customers for the infringement, misappropriation or other violation by our products of the intellectual property rights of others could require us to pay substantial damages and impose other costs and fees.
We currently have in effect, and may in the future enter into, agreements in which we agree to defend, indemnify and hold harmless our customers or suppliers from damages and costs that may arise from the infringement, misappropriation or other violation by our products of third-party patents, trademarks or other proprietary rights. We may periodically have to respond to claims and initiate or participate in litigation in connection with these indemnification obligations, which may result in our paying substantial damages. Such litigation could result in significant expense to us and divert the efforts of our technical and management personnel, whether or not such litigation is ultimately determined in our favor. Our insurance does not cover intellectual property infringement. Any of the foregoing could materially and adversely affect our business, financial condition and results of operations.
We could incur substantial costs as a result of violations of, or liabilities under, environmental laws.
Our operations and properties are subject to a variety of federal, state, local and foreign environmental, health and safety laws and regulations governing, among other things, air emissions, wastewater discharges, management and disposal of hazardous, non-hazardous and radioactive materials and waste and remediation of releases of hazardous materials. Compliance with environmental requirements could require us to incur significant operating or capital expenditures or result in significant restrictions on our operations. Our failure to comply with these environmental, health and safety laws and regulations, including failing to obtain any necessary permits, could cause us to incur substantial civil or criminal fines or penalties or enforcement actions, including regulatory or judicial orders enjoining or curtailing our operations or requiring us to conduct or fund remedial or corrective measures, install pollution control equipment or perform other actions. Under certain of these laws and regulations, we may be subject to joint and several liability for environmental investigations and cleanups, including at properties that we currently or previously owned or operated, or at sites at which waste we generated was disposed, even if the contamination was not caused by us or was legal at the time it occurred. The future identification of presently unidentified environmental conditions, more vigorous enforcement by regulatory agencies, enactment of more stringent laws, regulations or permit requirements, including relating to climate change, or other unanticipated events may arise in the future and give rise to material environmental liabilities and related costs or adversely impact the market for our products, which could materially and adversely affect our business, financial condition and results of operations.
A European Union, or EU, directive relating to the restriction of hazardous substances in electrical and electronic equipment, or RoHS directive, and an EU directive relating to waste electrical and electronic equipment, or WEEE directive, have been and are being implemented in EU member states. Among other things, the RoHS directive restricts the use of certain hazardous substances in the manufacture of electrical and electronic equipment and the WEEE directive requires producers of electrical goods to be responsible for the collection, recycling, treatment and disposal of these goods. In addition, laws similar to the RoHS and WEEE directives were passed in China in 2006 and South Korea in 2007. Governments in other countries and states, including the United States, are considering implementing similar laws or regulations.
In addition, a regulation regarding the registration, authorization and restriction of chemical substances in industrial products, or REACH, became effective in the EU in 2007. REACH and other regulations require us or our suppliers to substitute certain chemicals contained in our products with substances the EU considers less dangerous. We have assessed the impact that REACH is expected to have on us and have determined the impact at this time to be immaterial to our business and operations, but we cannot assure you that it or similar regulations will not materially affect us in the future.
The costs associated with complying with future laws and regulations could include costs associated with modifying, requalifying or reformulating our products, recycling and other waste processing costs, or legal and
96
Table of Contents
regulatory costs and insurance costs. We have recorded in the past and may be required to record in the future additional expenses for costs associated with compliance with regulations. The costs of complying with future environmental and worker health and safety laws and regulations could materially and adversely affect our business, financial condition and results of operations.
We do not control our suppliers, customers or business partners, and facts or circumstances that may occur as a result of their actions or omissions could harm our reputation and sales.
We do not control our suppliers, customers or partners, or their environmental or other practices. A violation of environmental or other laws by our suppliers, other customers or partners, or an environmental or public health incident at customer locations, including, for example, a nuclear incident at a facility to which we supplied equipment or that we serviced, or any failure of these third parties to follow generally accepted ethical business practices, could create negative publicity and harm our reputation. In addition, we may be required to seek alternative suppliers or partners if these violations or failures were to occur. We do not inspect or audit compliance of our suppliers, customers or partners with these laws or practices, and we do not require our suppliers, customers or partners to comply with a formal code of conduct. Any conduct or actions that our suppliers could take could reduce demand for our products, harm our ability to meet demand or harm our reputation, brand image, business, financial condition or results of operations.
Some of our workforce is represented by labor unions in the United States and by works councils and trade unions in the EU, and are covered by collective bargaining agreements in connection with such representations. Labor group representation may lead to work stoppages that could materially and adversely affect our business, including as a result of a failure to renegotiate a collective bargaining agreement.
As of March 31, 2021, approximately 39, or 1.5%, of our U.S. employees were unionized, and the majority of our EU employees are members of, or are represented by, works councils or trade unions and are covered by collective bargaining agreements. In addition, employees who are not currently members of, or otherwise represented by, labor organizations may seek such membership or representation, as applicable, in the future. Since 1988, we have experienced only two work stoppages, each time at our facility in Lamanon, France that lasted less than half a day. We may experience work stoppages or other labor disturbances in the future, including in connection with the renegotiation of collective bargaining agreements as they expire, which could adversely affect our business. We cannot predict how stable our relationships will be or whether we will be able to satisfy union or works council requirements without impacting our operating results and financial condition. Union and works council rules may limit our flexibility to respond to changing market conditions and the application of these rules could harm our business. The unions and works councils may also limit our flexibility in dealing with our workforce. Work stoppages and instability in our relationships could negatively impact the timely production of our products, which could strain relationships with customers and cause a loss of revenue that would adversely affect our results of operations. Additionally, any renegotiation of current collective bargaining agreements may result in terms that are less favorable to us.
The elimination or any modification of the Price-Anderson Acts indemnification authority could have adverse consequences for our business.
In the United States, the Atomic Energy Act of 1954, as amended, or the AEA, comprehensively regulates the manufacture, use and storage of radioactive materials. Section 170 of the AEA, which is known as the Price-Anderson Act, supports the nuclear services industry by offering broad indemnification for third-party public liability claims arising from a nuclear accident occurring at any commercial NPP in the United States. The Act channels the nuclear liability to the licensee plant operator and provides omnibus coverage for all firms that contribute in any way to the design, construction or operation of a licensed reactor, including vendors, contractors, suppliers, engineers, consulting firms, and transporters. The indemnification authority of the Nuclear Regulatory Commission, or NRC, and Department of Energy, or DOE, under the Price-Anderson Act has been extended by Congress numerous times since enactment in 1957, including most recently through 2025 by the
97
Table of Contents
Energy Policy Act of 2005. Extension is often largely uncontroversial, although it has met opposition at times due primarily to the view that the Act is a subsidy for the nuclear energy industry. Some of our customers are covered by the DOE indemnification provisions of the Price-Anderson Act for contractors. In addition, other jurisdictions have similar nuclear liability laws with indemnification authority to protect suppliers. If the nuclear liability and indemnification authority in the United States or other countries is eliminated or adversely modified in the future, our business could be adversely affected if the owners and operators of NPPs cancel or delay plans to build new plants or curtail the operations of existing plants. Although it is unlikely that the nuclear liability financial protection authority under the Price-Anderson Act would be completely abolished, some aspects of the Act could be changed during future reauthorizations.
Certain of our products and software are subject to ongoing regulatory oversight by the FDA or equivalent regulatory agencies in international markets and if we are not able to obtain or maintain the necessary regulatory approvals we may not be able to continue to market and sell such products which may materially and adversely affect our business.
The FDA regulates virtually all aspects of a medical device design, development, testing, manufacturing, labeling, storage, record keeping, adverse event reporting, sale, promotion, distribution and shipping. Before a new medical device, including a new intended use, indication, or claim for an existing product, can be marketed in the United States, it must first receive either premarket approval or 510(k) clearance from the FDA, unless an exemption exists. Either process can be expensive, lengthy and unpredictable. The FDAs 510(k) clearance process generally takes from three to twelve months, but it can last longer. The process of obtaining premarket approval is much more costly and uncertain than the 510(k) clearance process and it generally takes from one to three years, or even longer, from the time the application is filed with the FDA. Additionally, outside of the United States, our products are subject to clearances and approvals by foreign FDA counterparts. In order to market our products internationally, we must obtain licenses or approvals from these governmental agencies, which could include local requirements, safety standards, testing or certifications, and can be time consuming, burdensome and uncertain. Despite the time, effort and cost, there can be no assurance that a particular device or a modification of a device will be approved or cleared by the FDA, or any foreign governmental agency in a timely fashion, if at all. Even if we are granted regulatory clearances or approvals, they may include significant limitations on the indicated uses of the product, which may limit the market for those products, and how those products can be promoted.
Medical devices may only be marketed for the indications for which they are approved or cleared. The FDA and other foreign governments also may change their policies, adopt additional regulations, or revise existing regulations, each of which could prevent or delay approval or clearance of our device, or could impact our ability to market our currently approved or cleared devices. We are also subject to medical device reporting regulations, which require us to report to the FDA and other international governmental agencies if our products cause or contribute to a death or a serious injury, or malfunction in a way that would likely cause or contribute to a death or a serious injury. Further, we are subject to the QSR in the United States and ISO 13485 certification in many international markets, compliance with which is necessary to receive FDA and other international clearances or approvals to market new products, and is necessary for us to be able to continue to market a cleared or approved product in the United States or globally. After a product is placed in the market, we are also subject to oversight by the FDA and Federal Trade Commission related to the advertising and promotion of our products to ensure our claims are consistent with our regulatory clearances, that there is scientific data to substantiate our claims, and that our advertising is not false or misleading. Our products are also subject to state regulations and various international laws and regulations.
A component of our strategy is to continue to upgrade products such as HDR Vue and SunCHECK. Our previous upgrades required 510(k) clearance and international registration before we were able to offer them for sale. We expect our future upgrades will similarly require 510(k) clearance or approval; however, future upgrades may be subject to substantially more time consuming data generation requirements and uncertain premarket approval or clearance processes. If we were required to use the premarket approval process for future products or product modifications, it could delay or prevent release of the proposed products or modifications, which could harm our business.
98
Table of Contents
The FDA requires device manufacturers to make their own determination of whether or not a modification requires an approval or clearance; however, the FDA can review a manufacturers decision not to submit for additional approvals or clearances. Any modification to an FDA approved or cleared device that would significantly affect its safety or efficacy or that would constitute a major change in its intended use would require a new premarket approval or 510(k) clearance. We cannot ensure that the FDA will agree with our decisions not to seek approvals or clearances for particular device modifications or that we will be successful in obtaining premarket approvals or 510(k) clearances for modifications in a timely fashion, if at all.
We have obtained 510(k) clearance for SunCHECK to be used as an integrated patient quality assurance, machine quality assurance and data management workflow management application for radiation therapy professionals. We have made modifications to SunCHECK in the past and may make additional modifications in the future that we believe do not or will not require additional approvals or clearances. If the FDA disagrees, based on new finalized guidance and requires us to obtain additional premarket approvals or 510(k) clearances for any modifications to SunCHECK and we fail to obtain such approvals or clearances or fail to secure approvals or clearances in a timely manner, we may be required to cease manufacturing and marketing the modified device or to recall such modified device until we obtain FDA approval or clearance and we may be subject to significant regulatory fines or penalties.
The FDA and similar governmental authorities in other countries in which we market and sell our products have the authority to require the recall of our products in the event of material deficiencies or defects in design, manufacture or labeling. A government mandated recall, or a voluntary recall by us, could occur as a result of component failures, manufacturing errors or design defects, including defects in labeling and user manuals. Any recall could divert managements attention, cause us to incur significant expenses, generate negative publicity, harm our reputation with customers, negatively affect our future sales and business, require redesign of our products, and harm our operating results. In these circumstances, we may also be subject to significant enforcement action. If any of these events were to occur, our ability to introduce new or enhanced products in a timely manner would be adversely affected, which in turn would harm our future growth.
We are subject to federal, state, local and international laws and regulations related to healthcare, the violation of which could result in substantial penalties and harm our business in the medical end market.
Our operations are subject to several laws and regulations governing interactions with healthcare providers. The Medicare and Medicaid anti-kickback laws, and similar state laws, prohibit soliciting, offering, paying or accepting any payments or other remuneration that is intended to induce any individual or entity to either refer patients to or purchase, lease or order, or arrange for or recommend the purchase, lease or order of, healthcare products or services for which payment may be made under federal and state healthcare programs, such as Medicare and Medicaid. Such laws impact our sales, marketing and other promotional activities by reducing the types of financial arrangements we may have with our customers, potential customers, marketing consultants and other service providers. They particularly impact how we structure our sales offerings, including discount practices, customer support, product loans, education and training programs, physician consulting, research grants and other service arrangements. Many of these laws are broadly drafted and are open to a variety of interpretations, making it difficult to determine with any certainty whether certain arrangements violate such laws, even if statutory safe harbors are available.
In addition to such anti-kickback laws, federal and state false claims laws generally prohibit the knowing filing or causing the filing of a false claim, or the knowing use of false statements to obtain payment from government payors. Although we do not submit claims directly to payors, manufacturers can be held liable under these laws if they are deemed to cause the submission of false or fraudulent claims by providing inaccurate billing or coding information to customers, or through certain other activities, including promoting products for uses or indications that are not approved by the FDA.
99
Table of Contents
We are also subject to federal and state physician self-referral laws. The federal Ethics in Patient Referrals Act of 1989, commonly known as the Stark Law, prohibits, subject to certain exceptions, physician referrals of Medicare and Medicaid patients to an entity providing certain designated health services if the physician or an immediate family member has any financial relationship with the entity. The Stark Law also prohibits the entity receiving the referral from billing any good or service furnished pursuant to an unlawful referral. Various states have corollary laws to the Stark Law, including laws that require physicians to disclose any financial interest they may have with a healthcare provider to their patients when referring patients to that provider. Both the scope and exceptions for such laws vary from state to state.
If our past or present operations are found to be in violation of any of these anti-kickback, false claims, self-referral or other similar laws in foreign jurisdictions, we may be subject to the applicable penalty associated with the violation, which may include significant civil and criminal penalties, damages, fines, imprisonment and exclusion from healthcare programs. The impact of any such violations may lead to curtailment or restructuring of our operations, which could adversely affect our ability to operate our business and our financial results.
There are a number of federal and state laws protecting the confidentiality of certain patient health information, including patient records, and restricting the use and disclosure of that protected information. In particular, the U.S. Department of Health and Human Services, or HHS, has promulgated patient privacy rules under the Health Insurance Portability and Accountability Act, or HIPAA. These privacy rules protect medical records and other personal health information of patients by limiting their use and disclosure, giving patients the right to access, amend and seek accounting of their own health information and limiting most uses and disclosures of health information to the minimum amount reasonably necessary to accomplish the intended purpose. The HIPAA privacy standard was amended by the Health Information Technology for Economic and Clinical Health Act, enacted as part of the American Recovery and Reinvestment Act of 2009. Although we are not a covered entity under HIPAA, we are considered a business associate of certain covered entities and, as such, we are directly subject to HIPAA, including its enforcement scheme and inspection requirements, and are required to implement policies, procedures as well as reasonable and appropriate physical, technical and administrative security measures to protect individually identifiable health information we receive from covered entities. Our failure to protect health information received from customers in compliance with HIPAA or other laws could subject us to civil and criminal liability to the government and civil liability to the covered entity, could result in adverse publicity, and could harm our business and impair our ability to attract new customers.
The Sunshine Act, which was enacted by Congress as part of the Patient Protection and Affordable Care Act on December 14, 2011, requires each applicable manufacturer, which includes medical device companies, to track and report to the federal government on an annual basis all payments and other transfers of value from such applicable manufacturer to U.S. licensed physicians and teaching hospitals as well as physician ownership of such applicable manufacturers equity, in each case subject to certain statutory exceptions. Furthermore, on October 25, 2018, President Trump signed into law the Substance Use-Disorder Prevention that Promoted Opioid Recovery and Treatment for Patients and Communities Act which in part (under a provision entitled Fighting the Opioid Epidemic with Sunshine) extends the reporting and transparency requirements for physicians in the Physician Payments Sunshine Act to physician assistants, nurse practitioners, clinical nurse specialists, certified registered nurse anesthetists, and certified nurse midwives (with reporting requirements going into effect in 2022 for payments made in 2021). Such data will be made available by the government on a publicly searchable website. Failure to comply with the data collection and reporting obligations imposed by the Sunshine Act can result in civil monetary penalties ranging from $1,000 to $10,000 for each payment or other transfer of value that is not reported (up to a maximum of $150,000 per reporting period) and from $10,000 to $100,000 for each knowing failure to report (up to a maximum of $1 million per reporting period). In addition, we are subject to similar state and foreign laws related to the tracking and reporting of payments and other transfers of value to healthcare professionals, the violation of which could, among other things, result in civil monetary penalties and adversely impact our reputation and business.
100
Table of Contents
Healthcare reform legislation could materially and adversely affect demand for our products, our revenue and our financial condition.
In March 2010, the Patient Protection and Affordable Care Act, as amended by Health Care and Education Reconciliation Act, collectively referred to as the ACA were signed into law. The ACA includes a large number of health related provisions, including expanding Medicaid eligibility, requiring most individuals to have health insurance, establishing new regulations on health plans, establishing health insurance exchanges, requiring manufacturers to report payments or other transfers of value made to physicians and teaching hospitals, modifying certain payment systems to encourage more cost-effective care and a reduction of inefficiencies and waste and including new tools to address fraud and abuse. The laws also include a decrease in the annual rate of inflation for Medicare payments to hospitals and the establishment of an independent payment advisory board to suggest methods of reducing the rate of growth in Medicare spending. The expansion in the governments role in the U.S. healthcare industry may result in decreased profits to us, lower reimbursement by third-party payors for our products, or reduced volume of medical procedures conducted with our products, all of which could have a material adverse effect on our business, financial condition and results of operations. The federal government may take further action regarding the ACA, including, but not limited to, repeal or replacement action. Most recently, the Tax Cuts and Jobs Act was signed into law in December 2017, which, among other things, removed penalties for not complying with the individual mandate to carry health insurance. Additionally, all or a portion of the ACA and related subsequent legislation may be modified, repealed or otherwise invalidated through judicial challenge, which could result in lower numbers of insured individuals, reduced coverage for insured individuals and adversely affect our business. We continue to monitor the impact that the ACA may have on our business.
In addition, since the adoption of the Affordable Care Act, other legislation designed to keep federal healthcare costs down has been proposed or passed. For example, under the sequestration required by the Budget Control Act of 2011, as amended by the American Taxpayer Relief Act of 2012, Medicare payments for all items and services under Parts A and B incurred on or after April 1, 2013 have been reduced by up to 2%. Future federal legislation may impose further limitations on the coverage or amounts of reimbursement available for our products from governmental agencies or third-party payors. These limitations could have a negative impact on the demand for our products and services, and therefore on our financial position and results of operations.
Since the enactment of the ACA, the Centers for Medicare and Medicaid Services, or CMS, continues its efforts to move away from fee-for-service payments for furnishing items and services in Medicare. In the past several rulemaking cycles, CMS has increased packaging policies and created larger payment bundles across the Medicare Hospital Outpatient Prospective Payment System, or OPPS. One example is CMSs expansion of Comprehensive Ambulatory Payment Classifications, under which payment for adjunctive and secondary items, services and procedures are packaged into the most costly primary procedure at the claim level. Beyond the OPPS, CMSs Innovation Center has launched a number of alternative payment model, or APM, demonstrations that involve episode-based (i.e. bundled) payment. Since 2011, for example, Center for Medicare and Medicaid Innovation, or CMMI, has created and is in the process of creating major federal initiatives to test episode-based payments, such as the Bundled Payments for Care Improvement, Oncology Care Model, Specialty Practitioners Payment Model Opportunities. More recently, CMMI proposed a Radiation Oncology Model, which would mandate selected radiotherapy providers to participate in a prospective, episode-based payments model where payment is based on a patients diagnosis as opposed to the traditional volume-based fee-for service payment model. It is unclear what impact, if any, such initiatives will have on our business and operating results, but uncertainties surrounding the implementation of these payment models could pause or otherwise delay the purchase of our products by our customers and any resulting decrease in reimbursement to our customers may result in reduced demand for our services.
Furthermore, the Patient Access and Medicare Protection Act of 2015 froze payment for some radiation therapy delivery and related services, and requires CMS to provide a report to the U.S. Congress on the development of an APM for radiation therapy services provided in non-facility settings. While these types of payment packaging policies and episode-based payments may impact reimbursement for overall patient care, including items and
101
Table of Contents
services furnished to patients, they also create incentives for providers to carefully assess the value proposition of technology purchases and uses. The impacts of these payment and delivery system changes are in their infancy and their overall effects remain under review.
Future legislative or policy initiatives directed at reducing costs could be introduced at either the federal or state level. We cannot predict what healthcare reform legislation or regulations, if any, including any potential repeal or amendment of the ACA, will be enacted in the United States or elsewhere, what impact any legislation or regulations related to the healthcare system that may be enacted or adopted in the future might have on our business, or the effect of ongoing uncertainty or public perception about these matters will have on the purchasing decisions of our customers. However, the implementation of new legislation and regulation may materially lower reimbursements for our products, materially reduce medical procedure volumes and significantly and adversely affect our business.
If third-party payors do not provide sufficient coverage and reimbursement to healthcare providers or if there is a reduction in the number of patients with health insurance, demand for our products and our revenue could be materially and adversely affected.
Our customers rely significantly on reimbursement from public and private third-party payors procedures utilizing our radiation oncology and other medical products. Our ability to commercialize our products successfully and increase market acceptance of our products will depend in significant part on the extent to which public and private third-party payors provide adequate coverage and reimbursement for procedures that are performed with our products and the extent to which patients that are treated by our products continue to be covered by health insurance. Third-party payors may establish or change the reimbursement for medical products and services that could significantly influence the purchase of medical products and services. In addition, actions by the government, downturns in the economy and other factors outside of our control could negatively affect the number of individuals covered by health insurance. For example, in connection with COVID-19-related layoffs, many individuals have lost their employer-covered health insurance and there is uncertainty as to when or if such coverage will be re-established. If reimbursement policies or other cost containment measures are instituted in a manner that significantly reduces the coverage or payment for the procedures that are performed with our products or if there is a prolonged reduction in the number of patients eligible to be treated by our products that are covered by health insurance, our revenue may decline, our existing customers may not continue using our products or may decrease their use of our products, and we may have difficulty obtaining new customers. Such actions would likely have a material adverse effect on our operating results.
In addition, the CMS reviews reimbursement rates annually and may implement significant changes in future years, which could discourage existing and potential customers from purchasing or using our products. Further, outside of the United States, reimbursement practices vary significantly by country. Market acceptance of our products may depend on the availability and level of coverage and reimbursement in any country within a particular time.
Some of our products depend on our ability to source data from third parties who could take steps to block our access to such data. Such blocking could limit the effectiveness of these products, increase our expenses or materially and adversely impact our business.
Our SunCHECK software requires access to data such as electronic health information, or EHI, from other third-party vendors of our customers, typically original equipment manufacturers, in order to perform quality assessments. The functioning of our analytics applications and our ability to perform analytics services is predicated on our ability to establish interfaces that download the relevant data from these third party source systems on a repeated basis and in a reliable manner. The 21st Century Cures Act, often referred to simply as the Cures Act, which was enacted in 2016, contains, among other things, incentives and penalties to promote the use and efficient exchange of EHI and prevent information blocking (that is, activity that is likely to interfere with, prevent, or materially discourage access, exchange, or use of EHI, where a health information technology developer, health
102
Table of Contents
information network or health information exchange knows or should know that a practice is likely to interfere with access to, exchange or use of EHI). While the information sharing incentives created by the Cures Act are generally beneficial to our business, the implementing regulations also contain certain exceptions which would allow a market actor to block access to EHI without liability. Consequently, we may encounter vendors that engage in information blocking practices that may inhibit our ability to access the relevant data on behalf of customers and any steps we take to enforce the anti-information blocking provisions of the 21st Century Cures Act could be costly, could distract management attention from the business, and could have uncertain results.
The impact of the 21 Century Cures Act on our business is unclear at this time, due to, among other things, uncertainty regarding the interpretation of safe harbors and exceptions to the 21 Century Cures Act by industry participants and regulators.
It is unclear whether the 21 Century Cures Act may benefit us in that certain electronic health records vendors will no longer be permitted to interfere with our attempts at integration, but the rules may also make it easier for other similar companies to enter the market, creating increased competition, and reducing our market share.
Regulations related to conflict minerals may force us to incur additional expenses, may result in damage to our business reputation and may materially and adversely impact our ability to conduct our business.
As a public company, we will be subject to the requirements under the Dodd-Frank Wall Street Reform and Consumer Protection Act of 2010, or the Dodd-Frank Act, that requires us to diligence, disclose and report whether or not our devices contain conflict minerals. The implementation of these requirements could adversely affect the sourcing, availability and pricing of the materials used in the manufacture of components used in our devices. In addition, we will incur additional costs to comply with the disclosure requirements, including costs related to conducting diligence procedures to determine the sources of conflict minerals that may be used or necessary to the production of our devices and, if applicable, potential changes to devices, processes or sources of supply as a consequence of such verification activities. It is also possible that we may face reputational harm if we determine that certain of our devices contain minerals not determined to be conflict-free or if we are unable to alter our devices, processes or sources of supply to avoid such materials.
Risks Related to Our Liquidity and Capital Resources
If we cannot generate sufficient operating cash flow and obtain external financing, we may be unable to make all of our planned capital expenditures and other expenses.
Our ability to fund anticipated capital expenditures and other expenses depends on generating sufficient cash flow from operations and the availability of external financing. Since our acquisition by Charterhouse in 2015, Charterhouse has provided us with the capital and debt financing that we have used to fund our growth and operations. Charterhouses ownership in us upon the consummation of the Business Combination will reduce significantly and Charterhouse is under no obligation to continue making capital investments in us or to provide debt financing to us, and is unlikely to do so.
Our debt service obligations and our capital expenditures, together with on-going operating expenses, will be a substantial drain on our cash flow and may decrease our cash balances. The timing and amount of our capital requirements cannot be precisely determined at this moment and will depend on a number of factors, including demand for our products, product mix, changes in industry conditions and market competition. We intend to regularly assess markets for external financing opportunities, including debt and equity. Such financing may not be available when needed or, if available, may not be available on satisfactory terms, particularly in light of the limited financing available as a result of the recent global financial crisis. Any equity financing would cause further dilution to our stockholders. Our inability to obtain needed financing or to generate sufficient cash from operations may require us to abandon projects or curtail capital expenditures, and we could be materially adversely affected. If we are not able to independently generate excess free cash flow and obtain third party debt or equity financing, our ability to grow our business may be materially adversely affected.
103
Table of Contents
Our indebtedness could impair our financial condition and harm our ability to operate our business.
Certain of our subsidiaries have incurred indebtedness under a credit agreement providing for multi-currency senior secured term loan facilities and a revolving credit facility. We refer to the credit agreement, together with all related loan documents, as entered into in March 2019 and as amended thereafter, as the Mirion Credit Agreement. The obligations under the Mirion Credit Agreement are jointly and severally guaranteed on a senior basis by certain of our subsidiaries organized in the United States, Germany, the U.K., Canada, France, Belgium and Luxembourg, and secured by substantially all of the assets of those subsidiaries (subject to certain customary limitations). As of March 31, 2021, $889.5 million, net of $17.5 million of deferred financing costs, was outstanding under the Mirion Credit Agreement term loan facilities and no loans were outstanding under the Mirion Credit Agreement revolving credit facility, other than $8.9 million of letters of credit issued thereunder. An additional $1.5 million of indebtedness was outstanding under certain other loan agreements with third parties. We have also entered into various line of credit arrangements with local banks in France and Germany. All of our debt under the Mirion Credit Agreement bears interest at variable rates. If the rates were to increase significantly, our cost of borrowing would increase and the risks related to our indebtedness would be exacerbated.
On June 17, 2021, Mirion Technologies (HoldingSub2) Ltd. entered into a commitment letter (the Commitment Letter) with Goldman Sachs Lending Partners LLC and Citigroup Global Markets Inc. to provide new debt financing, subject to the closing of the Business Combination and certain other conditions, which debt financing will replace the facilities provided under the Mirion Credit Agreement. The aggregate commitment of the senior secured credit facilities pursuant to the new debt financing will consist of an $830 million dollar-denominated first lien term facility (the New Term Facility) and a $90 million revolving credit facility (the New Revolving Facility and, together with the New Term Facility, the New Credit Facilities). The actual terms of the New Credit Facilities may differ from those described in the Commitment Letter. Additionally, all of the debt under the New Credit Facilities will bear interest at a variable rate. If the rates were to increase significantly, our cost of borrowing would increase and the risks related would be exacerbated.
One of our subsidiaries has also issued notes to shareholders, or the Shareholder Notes of approximately $1.2 billion as of March 31, 2021, and $3.6 million in notes to certain members of management, or the Management Notes as of March 31, 2021. The Shareholder Notes and Management Notes rank pari passu between each other and our other unsecured obligations. The Shareholder and Management Notes can be prepaid without penalty at our option and are subordinate in right of payment to any of our indebtedness to banks or to other financial institutions (either currently existing or incurred in the future). Most of the Shareholder and Management Notes have been admitted to trading and are on the official listing of The International Stock Exchange, or TISE. The Shareholder and Management Notes bear simple annual interest at 11.5%. For the Shareholder Notes, the interest is added to the principal outstanding on December 31 of each year until 2025. Accrued interest outstanding not added to the Shareholder Notes principal was $32.1 million as of March 31, 2021. For the Management Notes, half of the interest is added to the principal outstanding on December 31 of each year until 2025, while the remaining half is payable in cash annually. Accrued interest outstanding on the Management Notes is not material. The Shareholder Notes and Management Notes will be repaid in connection with the Business Combination.
Our indebtedness may have important consequences, including, but not limited to, the following:
| | increasing our vulnerability to general economic downturns and adverse industry conditions; |
| | requiring us to dedicate a significant portion of our cash flows from operations to the payment of interest and principal on our debt, which would reduce the funds available to us for our working capital, capital expenditures or other general corporate requirements; |
| | limiting our flexibility in planning for, or reacting to, changes in our business and industry; |
| | placing us at a competitive disadvantage compared to our competitors with less indebtedness or more liquidity; and |
104
Table of Contents
| | limiting our ability to obtain additional financing in the future for working capital, capital expenditures, acquisitions, general corporate purposes or other purposes. |
Despite our levels of indebtedness, we have the ability to incur more indebtedness. Incurring additional debt could further intensify the risks described above.
We may be able to incur additional debt in the future and the terms of the Mirion Credit Agreement will not fully prohibit us from doing so. We have the ability to draw upon the undrawn portion of our $90 million Existing Revolving Facility. We also have the ability to draw upon the uncommitted accordion provided under the Mirion Credit Agreement (subject to the receipt of commitments and satisfaction of certain other conditions), which permits incremental term loans and revolving loans thereunder or certain equivalent debt outside of the Mirion Credit Agreement of up to (i) the greater of $118.0 million and 100% of consolidated EBITDA (as defined in the Mirion Credit Agreement), plus (ii) the aggregate amount of all voluntary prepayments of loans and certain other permitted indebtedness that is secured on a pari passu basis with the loans, in each case, to the extent not financed with the incurrence of certain additional long-term indebtedness, plus (iii) an unlimited amount so long as, on a pro forma basis (as defined in the Mirion Credit Agreement) (x) with respect to indebtedness secured on a pari pasu basis with the first lien obligations under the Mirion Credit Agreement, the consolidated first lien secured debt to consolidated EBITDA ratio (as defined therein) would not exceed 5.00:1.00 or, in the case of incremental facilities incurred to finance a permitted acquisition or other permitted investment, such ratio immediately prior to the incurrence; (y) with respect to indebtedness secured on a junior basis with the first lien obligations under the Mirion Credit Agreement, the consolidated secured debt to consolidated EBITDA ratio (as defined in the Mirion Credit Agreement) would not exceed 5.50:1.00 or, in the case of incremental facilities incurred to finance a permitted acquisition or other permitted investment, such ratio immediately prior to the incurrence; and (z) in the case of unsecured indebtedness either the consolidated total debt to consolidated EBITDA ratio (as defined therein) would not exceed 6.00:1.00 or the interest coverage ratio (as defined therein) would be at least 2:00:1.00 (or, in the case of incremental facilities incurred to finance a permitted acquisition or other permitted investment, the consolidated total debt to consolidated EBITDA ratio would not exceed such ratio immediately prior to the incurrence, or the interest coverage ratio would not be less than such ratio, immediately prior to the incurrence). In addition, the Mirion Credit Agreement contains other provisions allowing us to incur significant amounts of additional new debt. If new debt is added to our current debt levels, the related risks that we now face could intensify and we may not be able to meet all our respective debt obligations. In addition, the Mirion Credit Agreement does not prevent us from incurring obligations that do not constitute indebtedness under those agreements.
Additionally, the terms of the New Credit Facilities to be provided pursuant to the Commitment Letter will not fully prohibit us from incurring additional debt in the future. We will have the ability to draw upon our $90 million New Revolving Facility. Similar to the Mirion Credit Agreement, we will have the ability to draw upon the uncommitted accordion to be provided under the New Credit Facilities (subject to the receipt of commitments and satisfaction of certain other conditions), which will incremental term loans and revolving loans thereunder or certain equivalent debt outside of the New Credit Facilities documentation of up to (i) the greater of $172.0 million and 100% of consolidated EBITDA plus any unused portion of the general debt basket that is instead applied to increase the amount of the accordion, plus (ii) the aggregate amount of all voluntary prepayments of loans and certain other permitted indebtedness secured on a pari passu basis with the loans under the New Credit Facilities, in each case, to the extent not financed with the incurrence of certain additional long-term indebtedness, plus (iii) an unlimited amount, so long as (x) in the case of indebtedness secured on a pari passu basis with the first lien obligations under the New Credit Facilities, the first lien net leverage ratio (to be defined in the New Credit Facilities documentation) is equal to or less than the greater of the first lien net leverage ratio on the closing date of the New Credit Facilities or, in the case of incremental facilities incurred to finance a permitted acquisition or other permitted investment, such ratio immediately prior to the incurrence; (y) in the case of junior lien loans or loans secured by non-collateral, the secured net leverage ratio (to be defined in the New Credit Facilities documentation) is not more than the greater of 0.75x above the secured net leverage ratio on the closing date of the New Credit Facilities or, in the case of incremental facilities incurred to finance a permitted
105
Table of Contents
acquisition or other permitted investment, such ratio immediately prior to the incurrence; and (z) in the case of unsecured loans, either (A) the total net leverage ratio (to be defined in the New Credit Facilities Documentation) is not more than the greater of 1.25x above the total net leverage ratio on the closing date of the New Credit Facilities or, in the case of incremental facilities incurred to finance a permitted acquisition or other permitted investment, such ratio immediately prior to the incurrence, or (B) the interest coverage ratio (to be defined in the New Credit Facilities documentation) is greater than or equal to the lesser of 1.75:1.00 or, in the case of incremental facilities incurred to finance a permitted acquisition or other permitted investment, the interest coverage ratio immediately prior to the incurrence. In addition, the New Credit Facilities documentation will contain other provisions allowing us to incur significant amounts of additional new debt. If new debt is added to the debt that will be originally incurred under the New Credit Facilities, the related risks could intensify and we may not be able to meet all our respective debt obligations. In addition, the credit agreement governing the New Credit Facilities will not prevent us from incurring obligations that do not constitute indebtedness under those agreements. The definitive documentation for the New Credit Facilities has not been finalized and, accordingly, the actual terms of the New Credit Facilities may differ from those described in this proxy statement/prospectus. Market conditions at the time of debt financing marketing could result in more restrictive terms. Additionally, the actual terms of the New Credit Facilities may differ from those described in the Commitment Letter.
Restrictive covenants in the Mirion Credit Agreement and any future debt agreements, could restrict our operating flexibility.
The Mirion Credit Agreement contains restrictive covenants that limit our ability to engage in specified transactions and prohibit us from voluntarily prepaying certain of our other indebtedness. These covenants limit our ability to, among other things:
| | incur additional indebtedness; |
| | pay dividends on, or repurchase or make distributions in respect of, our capital stock or make other restricted payments; |
| | make certain investments, including acquisitions of other companies; |
| | sell or transfer assets; |
| | prepay, redeem, repurchase, defease or amend the terms of certain junior indebtedness; |
| | create or incur liens on our assets or enter into contractual obligations that restrict our ability to grant liens on assets or capital stock or pay dividends; and |
| | consolidate, merge, sell or otherwise dispose of all or substantially all of our assets. |
Under the Mirion Credit Agreement, in certain circumstances we also are required to satisfy and maintain a certain total level of first lien net debt to income ratio. Our ability to meet this financial ratio could be affected by events beyond our control, and there can be no assurance that we will meet that ratio.
The failure to comply with any of these covenants or any other term of the Mirion Credit Agreement would cause a default under the Mirion Credit Agreement. A default, if not waived, could result in acceleration of the outstanding indebtedness under the Mirion Credit Agreement, in which case such indebtedness would become immediately due and payable, and could also cause the acceleration of other indebtedness outstanding at such time. If any default occurs, we may not be able to pay our debt or borrow sufficient funds to refinance it. Even if new financing is available, it may not be available on terms that are acceptable to us. Complying with these covenants may cause us to take actions that we otherwise would not take or not take actions that we otherwise would take.
Unfavorable currency exchange rate fluctuations could materially and adversely affect our financial results.
Our international sales and our operations in countries other than the United States expose us to risks associated with fluctuating currency values and exchange rates. A significant amount of our international sales, costs, assets
106
Table of Contents
and liabilities are denominated in currencies other than the U.S. dollar. For fiscal 2020, approximately 39% of our sales were denominated in euros, 3% in pounds sterling, 3% in Japanese yen and 3% in Canadian dollars. For the nine months ended March 31, 2021, approximately 36% of our sales were denominated in euros, 4% in pounds sterling, 3% in Japanese yen and 2% in Canadian dollars. Gains and losses on the conversion of accounts receivable, accounts payable and other monetary assets and liabilities to U.S. dollars may contribute to fluctuations in our results of operations. In addition, increases in the value of the U.S. dollar relative to the euro could have an adverse effect on our results of operations. We do not currently purchase forward contracts to hedge against the risks associated with fluctuations in exchange rates.
Changes in our effective tax rate or adverse outcomes resulting from examination of our income tax returns could materially and adversely affect our results.
Our effective tax rate could be adversely affected by several factors, many of which are outside of our control, including:
| | earnings being lower than anticipated in countries where we are taxed at lower rates or other shifts in the mix of pre-tax profits and losses from one jurisdiction to another; |
| | our inability to use tax credits; |
| | changing tax laws or related interpretations, accounting standards and regulations and interpretations in multiple tax jurisdictions in which we operate; |
| | an increase in expenses not deductible for tax purposes, including certain stock-based compensation expense and impairment of goodwill; |
| | the tax effects of purchase accounting for acquisitions and restructuring charges and other discrete recognition of taxable events and exposures that may cause fluctuations between reporting periods; |
| | changes related to our ability to ultimately realize future benefits attributed to net operating loss and other carryforwards included in our deferred tax assets; |
| | tax assessments resulting from income tax audits or any related tax interest or penalties that would affect our income tax expense for the period in which the settlements take place; and |
| | a change in our decision to indefinitely reinvest foreign earnings. |
In addition, we may be subject to examination of our income tax returns by the Internal Revenue Service or other tax authorities. If any tax authority challenges the relative mix of our U.S. and international income, our future effective income tax rates could be adversely affected. While we regularly assess the likelihood of adverse outcomes from such examinations and the adequacy of our provision for income taxes, we cannot assure you that such provision is sufficient and that a determination by a tax authority will not have an adverse effect on our business, financial condition and results of operations.
Our effective tax rates may increase.
On April 7, 2021, the Biden administration proposed changes to the U.S. tax system. The proposals under discussion include changes to the U.S. corporate tax system that would increase U.S. corporate tax rates, impose a corporate minimum book tax and double the tax rate on and make other tax changes to GILTI earned by foreign subsidiaries. While it is expected that a tax reform bill will be introduced in the House of Representatives in the near term, many aspects of the current proposals are unclear or undeveloped. We are unable to predict which, if any, U.S. tax reform proposals will be enacted into law, and what effects any enacted legislation might have on our liability for U.S. federal income taxes. However, it is possible that the enactment of changes in the U.S. corporate tax system could have a material adverse effect on our liability for U.S. corporate tax and our consolidated effective tax rate.
107
Table of Contents
Risks Related to the Business Combination and GSAH
The Sponsor and GS Employee Participation have each agreed to vote in favor of the Business Combination and the other proposals described herein to be presented at the Special Meeting, regardless of how our public stockholders vote.
The Sponsor and GS Employee Participation have each agreed to, among other things, vote in favor of the Business Combination Proposal and the other proposals described herein to be presented at the Special Meeting. As of the date of this proxy statement, the Initial Stockholders own approximately 20% of the outstanding shares of our common stock. Accordingly, it is more likely that the necessary stockholder approval will be received than would be the case if our Initial Stockholders agreed to vote their founder shares in accordance with the majority of the votes cast by our public stockholders.
Neither the GSAH Board nor any committee thereof obtained a third party valuation in determining whether or not to pursue the Business Combination.
Neither the GSAH Board nor any committee thereof is required to obtain an opinion from an independent investment banking or accounting firm that the price that we are paying for Mirion is fair to us from a financial point of view. Neither the GSAH Board nor any committee thereof obtained a third party valuation in connection with the Business Combination. In analyzing the Business Combination, the GSAH Board conducted due diligence on Mirion. The GSAH Board also consulted with GSAHs management and its legal counsel, financial advisor and other advisors and considered a number of factors, uncertainty and risks, including, but not limited to, those discussed under Proposal No. 1Approval of the Business CombinationGSAHs Board of Directors Reasons for the Approval of the Business Combination, and concluded that the Business Combination was in the best interest of GSAHs stockholders. Accordingly, investors will be relying solely on the judgment of the GSAH Board in valuing Mirion, and the GSAH Board may not have properly valued such businesses. The lack of a third party valuation may also lead an increased number of stockholders to vote against the Business Combination or demand redemption of their shares, which could potentially impact our ability to consummate the Business Combination.
Since the Sponsor and the members of GSAHs management team have interests that are different, or in addition to (and which may conflict with), the interests of our stockholders, a conflict of interest may have existed in determining whether the Business Combination is appropriate as our initial business combination. Such interests include that the Sponsor will lose its entire investment in us if our business combination is not completed.
When you consider the recommendation of our Board in favor of approval of the Business Combination Proposal and the other proposals included herein, you should keep in mind that the Sponsor and our directors have interests in such proposal that are different from, or in addition to, those of our stockholders and warrant holders generally. Our Board was aware of and considered these interests, among other matters, in evaluating and negotiating the Business Combination and transaction agreements and in recommending to our stockholders that they vote in favor of the proposals presented at the Special Meeting, including the Business Combination Proposal. GSAH stockholders should take these interests into account in deciding whether to approve the proposals presented at the Special Meeting, including the Business Combination Proposal. See Proposal No. 1Approval of the Business CombinationInterests of Goldman Sachs Parties and Certain Other Persons in the Business Combination for a further discussion of these considerations.
The exercise of the GSAH management teams discretion in agreeing to changes or waivers in the terms of the Business Combination Agreement, including closing conditions, may result in a conflict of interest when determining whether such changes to the terms or waivers of conditions are appropriate and in GSAHs stockholders best interest.
In the period leading up to the closing of the Business Combination, events may occur that, pursuant to the Business Combination Agreement, would require GSAH to agree to amend the Business Combination
108
Table of Contents
Agreement, to consent to certain actions taken by Mirion or to waive rights that GSAH is entitled to under the Business Combination Agreement, including those related to closing conditions. Such events could arise because of changes in the course of Mirion businesses or a request by Mirion to undertake actions that would otherwise be prohibited by the terms of the Business Combination Agreement or the occurrence of other events that would have a material adverse effect on Mirion businesses and would entitle GSAH to terminate the Business Combination Agreement. In any of such circumstances, it would be at GSAHs discretion, acting through its Board, to grant its consent or waive those rights. The existence of financial and personal interests of one or more of the directors described in the preceding risk factors (and described elsewhere in this proxy statement) may result in a conflict of interest on the part of such director(s) between what he or they may believe is best for GSAH and its stockholders and what he or they may believe is best for himself or themselves in determining whether or not to take the requested action. As of the date of this proxy statement, GSAH does not believe there will be any changes or waivers that GSAHs management team would be likely to make after stockholder approval of the Business Combination Proposal has been obtained. While certain changes could be made without further stockholder approval, GSAH will circulate a new or amended proxy statement and resolicit GSAHs stockholders if changes to the terms of the transaction that would have a material impact on its stockholders are required prior to the vote on the Business Combination Proposal.
GSAH and Mirion will incur significant transaction and transition costs in connection with the Business Combination.
GSAH and Mirion have both incurred and expect to incur significant, non-recurring costs in connection with consummating the Business Combination and operating as a public company following the consummation of the Business Combination. We and Mirion may also incur additional costs to retain key employees. Certain transaction expenses incurred in connection with the Business Combination Agreement (including the Business Combination), including all legal, accounting, consulting, investment banking and other fees, expenses and costs, will be for the account of, or paid by, the party incurring such fees, expenses and costs, or otherwise paid by GSAH following the closing of the Business Combination.
GSAHs transaction expenses as a result of the Business Combination are currently estimated at approximately $33 million in deferred underwriting discount, advisory fees and placement agent fees payable to Goldman Sachs & Co. LLC, an affiliate of us and the Sponsor. The amount of the deferred underwriting discount will not be adjusted for any shares that are redeemed in connection with the Business Combination. The per-share amount we will distribute to stockholders who properly exercise their redemption rights will not be reduced by the deferred underwriting discount and after such redemptions, the per-share value of shares held by non-redeeming stockholders will reflect our obligation to pay the deferred underwriting discount.
The announcement of the proposed Business Combination could disrupt Mirions relationships with its customers, suppliers, joint venture partners and others, as well as its operating results and business generally.
Whether or not the Business Combination and related transactions are ultimately consummated, as a result of uncertainty related to the proposed transactions, risks relating to the impact of the announcement of the Business Combination on Mirions business include the following:
| | its employees may experience uncertainty about their future roles, which might adversely affect Mirions ability to retain and hire key personnel and other employees; |
| | customers, suppliers, joint venture partners and other parties with which Mirion maintains business relationships may experience uncertainty about its future and rescind their deposits, seek alternative relationships with third parties, seek to alter their business relationships with Mirion. or fail to extend an existing relationship with Mirion; and |
| | Mirion has expended and will continue to expend significant costs, fees and expenses for professional services and transaction costs in connection with the proposed Business Combination. |
109
Table of Contents
If any of the aforementioned risks were to materialize, they could lead to significant costs which may impact Mirions results of operations and cash available to fund its businesses.
Subsequent to consummation of the Business Combination, we may be exposed to unknown or contingent liabilities and may be required to subsequently take write-downs or write-offs, restructuring and impairment or other charges that could have a significant negative effect on our financial condition, results of operations and the price of our securities, which could cause you to lose some or all of your investment.
We cannot assure you that the due diligence conducted in relation to Mirion has identified all material issues or risks associated with Mirion, its business or the industry in which it competes. Furthermore, we cannot assure you that factors outside of Mirions and our control will not later arise. As a result of these factors, we may be exposed to liabilities and incur additional costs and expenses and we may be forced to later write-down or write-off assets, restructure our operations, or incur impairment or other charges that could result in our reporting losses. Even if our due diligence has identified certain risks, unexpected risks may arise and previously known risks may materialize in a manner not consistent with our preliminary risk analysis. If any of these risks materialize, this could have a material adverse effect on our financial condition and results of operations and could contribute to negative market perceptions about our securities or the post-business combination company.
Accordingly, any stockholders or warrant holders of GSAH who choose to remain our stockholders or warrant holders following the Business Combination could suffer a reduction in the value of their shares, warrants and units. Such stockholders or warrant holders are unlikely to have a remedy for such reduction in value.
The historical financial results of Mirion and unaudited pro forma financial information included elsewhere in this proxy statement may not be indicative of what our actual financial position or results of operations would have been.
The historical financial results of Mirion included in this proxy statement do not reflect the financial condition, results of operations or cash flows they would have achieved as a public company during the periods presented or those we will achieve in the future. The post-business combination companys financial condition and future results of operations could be materially different from amounts reflected in its historical financial statements included elsewhere in this proxy statement, so it may be difficult for investors to compare the post-business combination companys future results to historical results or to evaluate its relative performance or trends in its business.
Similarly, the unaudited pro forma financial information in this proxy statement is presented for illustrative purposes only and has been prepared based on a number of assumptions including, but not limited to, Mirion being treated as the acquired company for financial reporting purposes in the Business Combination, the total debt obligations and the cash and cash equivalents of Mirion on the date the Business Combination closes and the number of our public shares that are redeemed in connection with the Business Combination. Accordingly, such pro forma financial information may not be indicative of the post-business combination companys future operating or financial performance and the post-business combination companys actual financial condition and results of operations may vary materially from the pro forma results of operations and balance sheet contained elsewhere in this proxy statement, including as a result of such assumptions not being accurate. See Selected Unaudited Pro Forma Condensed Combined Financial Information.
We currently intend to only complete one Business Combination with the proceeds of our IPO and the sale of the private placement warrants, which will cause us to be solely dependent on Mirions business. This lack of diversification may negatively impact our operations and profitability.
We currently intend to only complete one Business Combination with the proceeds of our IPO and the sale of the private placement warrants. By completing our Business Combination with only a single entity our lack of diversification may subject us to numerous economic, competitive and regulatory risks. Further, we would not be
110
Table of Contents
able to diversify our operations or benefit from the possible spreading of risks or offsetting of losses, unlike other entities which may have the resources to complete several business combinations in different industries or different areas of a single industry. Accordingly, the prospects for our success will be solely dependent upon the business and financial performance of Mirion.
This lack of diversification may subject us to numerous economic, competitive and regulatory risks, any or all of which may have a substantial adverse impact upon the particular industry in which we may operate subsequent to the Business Combination. See Risks Related to Mirions Business for risks we may face as a result of consummating the Business Combination with Mirion.
We have a minimum cash requirement. This requirement may make it more difficult for us to complete the Business Combination as contemplated.
The Business Combination Agreement provides that Mirions and the Sellers obligation to consummate the Business Combination is conditioned on, among other things, the Minimum Cash Condition, which provides that the cash consideration in the Transactions will be an amount equal to $1,310,000,000; provided, that if the Minimum Cash Condition is not met, and Mirion and the Charterhouse Parties elect to waive the Minimum Cash Condition, then the Cash Consideration will be equal to $1,310,000,000 less the amount by which $1,310,000,000 exceeds the Available Closing Cash.
In addition, pursuant to the GSAH Certificate of Incorporation, in no event will we redeem public shares in an amount that would cause our net tangible assets (as determined in accordance with Rule 3a51-1(g)(1) of the Exchange Act) to be less than $5,000,001. If such conditions are not met, and such conditions are not or cannot be waived under the terms of the Business Combination Agreement, then the Business Combination Agreement could terminate and the proposed Business Combination may not be consummated.
If such conditions are waived and the Business Combination is consummated, the cash held by us and our subsidiaries (including Mirion) in the aggregate, after the closing of the Business Combination may not be sufficient to allow us to operate and pay our bills as they become due. Furthermore, our affiliates are not obligated to make loans to us or invest in us in the future after the Business Combination. The additional exercise of redemption rights with respect to a large number of our public stockholders may make us unable to take such actions as may be desirable in order to optimize our capital structure after consummation of the Business Combination and we may not be able to raise additional financing from unaffiliated parties necessary to fund our expenses and liabilities after the closing of the Business Combination. Any such event in the future may negatively impact the analysis regarding our ability to continue as a going concern at such time.
Certain regulatory approvals are closing conditions to the Business Combination. Such regulatory approvals may not be received, may take longer to receive than expected, or may be subject to conditions that are not presently anticipated, any of which could delay or prevent the Closing.
Before the Business Combination may be completed, we must obtain approvals under certain foreign direct investment regimes in France, Finland, Germany and possibly the United Kingdom. Specifically, in France, we are required to make a filing so as to obtain either (i) a written confirmation from the French Minister in charge of Economy that the Business Combination does not fall within the scope of the French Monetary and Financial Code or (ii) the approval of the Business Combination by a decision of the Minister in charge of Economy pursuant to that Code. In Finland, we are required to file an application or notification under the Act on the Screening of Foreign Corporate Acquisitions. In Germany, we are required to file a notification under the Foreign Trade and Payments Act and the accompanying Foreign Trade and Payments Regulation. In the United Kingdom, we could be required to make a mandatory filing under the National Security and Investment Act 2021 if it comes into force prior to the Closing. In addition to these foreign direct investment approvals, Mirion and its subsidiaries must obtain approvals from the U.S. Nuclear Regulatory Commission and certain U.S. state regulators in relation to the transfer of certain radioactive material licenses that they hold. Each of the foregoing regulatory approvals is a condition to Closing.
111
Table of Contents
The amount of time required to receive each of these regulatory approvals from the relevant governmental authorities could be considerable and is difficult to predict, and there can be no assurances as to whether such regulatory approvals will be received when expected, if at all. There can also be no assurances whether any conditions, limitations, obligations or restrictions will be imposed in connection with such approvals and, if imposed, whether such conditions, limitations, obligations or restrictions will have the effect of preventing or materially delaying the consummation of the Business Combination, imposing additional material costs on or materially limiting New Mirions revenue following the Business Combination, or otherwise materially reducing the anticipated benefits of the Business Combination. Further, the effects of the COVID-19 pandemic may delay receipt of the regulatory approvals necessary to complete the Business Combination.
You should also consider that regulatory approvals from governmental authorities, if received, reflect only the relevant authoritys view that the Business Combination does not contravene applicable foreign direct investment laws, related regulations or policy. Further, any particular regulatory approval is not an endorsement, recommendation or opinion of the applicable governmental authority that the proposed Business Combination is favorable to the shareholders of either party to the Business Combination from a financial point of view or that the authority has considered the adequacy or appropriateness of the commercial or legal terms of the Business Combination.
If we and Mirion and its subsidiaries do not obtain all regulatory approvals by the End Date of November 30, 2021, then, subject to certain conditions, we or Mirion and the Charterhouse Parties may extend the End Date to January 31, 2022, but if the parties do not extend the End Date then either party could terminate the Business Combination Agreement. In addition, if we and Mirion and its subsidiaries do not obtain all regulatory approvals by such extended End Date, then, subject to certain conditions, we or Mirion and the Charterhouse Parties may further extend the End Date to March 31, 2022, but if the parties do not further extend the End Date then either party could terminate the Business Combination Agreement. Accordingly, if any regulatory approvals are subject to significant conditions, or not obtained at all, our ability to consummate the Business Combination may be impaired and any such conditions could have a material and adverse impact on New Mirions future business, results of operations and financial condition.
The Sponsor, Mirion or our or their respective directors, officers, advisors or respective affiliates may elect to purchase shares from public stockholders prior to the consummation of the Business Combination, which may influence the vote on the Business Combination and reduce the public float of our Class A common stock.
At any time at or prior to the Business Combination, subject to applicable securities laws (including with respect to material nonpublic information), the Sponsor, Mirion stockholders or our or their respective directors, officers, advisors or respective affiliates may (1) purchase public shares from institutional and other investors who vote, or indicate an intention to vote, against any of the Condition Precedent Proposals, or elect to redeem, or indicate an intention to redeem, public shares, (2) execute agreements to purchase such shares from such investors in the future, or (3) enter into transactions with such investors and others to provide them with incentives to acquire public shares, vote their public shares in favor of the Condition Precedent Proposals or not redeem their public shares. Such a purchase may include a contractual acknowledgement that such stockholder, although still the record holder of GSAHs shares, is no longer the beneficial owner thereof and therefore agrees not to exercise its redemption rights. In the event that the Sponsor, Mirion stockholders or our or their respective directors, officers, advisors or respective affiliates purchase shares in privately negotiated transactions from public stockholders who have already elected to exercise their redemption rights, such selling stockholder would be required to revoke their prior elections to redeem their shares. The purpose of such share purchases and other transactions would be to (1) increase the likelihood of approving the Condition Precedent Proposals and (2) limit the number of public shares electing to redeem, including to satisfy any redemption threshold.
Entering into any such arrangements may have a depressive effect on our common stock (e.g., by giving an investor or holder the ability to effectively purchase shares at a price lower than market, such investor or holder may therefore become more likely to sell the shares he or she owns, either at or prior to the Business
112
Table of Contents
Combination). If such transactions are effected, the consequence could be to cause the Business Combination to be consummated in circumstances where such consummation could not otherwise occur. Purchases of shares by the persons described above would allow them to exert more influence over the approval of the proposals to be presented at the Special Meeting and would likely increase the chances that such proposals would be approved. In addition, if such purchases are made, the public float of our public shares and the number of beneficial holders of our securities may be reduced, possibly making it difficult to maintain or obtain the quotation, listing or trading of our securities on a national securities exchange.
Future resales of common stock after the consummation of the Business Combination may cause the market price of our securities to drop significantly, even if our business is doing well.
After the consummation of the Business Combination and subject to certain exceptions, certain Sellers will be contractually restricted from selling or transferring any shares of our common stock it received in connection with the Business Combination for a period of six months, and the Sponsor will be contractually restricted from selling or transferring the founder shares until the end of the Sponsor Lock-up Period. However, following the expiration of such lockup, neither the Sponsor nor such stockholders will be restricted from selling their shares of our common stock, other than by applicable securities laws. Additionally, the PIPE Investors will not be restricted from selling any of their shares of our common stock following the closing of the Business Combination, other than by applicable securities laws. As such, sales of a substantial number of shares of our common stock in the public market could occur at any time. These sales, or the perception in the market that the holders of a large number of shares intend to sell shares, could reduce the market price of our common stock.
As restrictions on resale end and registration statements are available for use, the sale or possibility of sale of shares by the Sponsor or its members, the PIPE Investors or certain other Sellers could have the effect of increasing the volatility in our share price or the market price of our Class A common stock could decline if the holders of currently restricted shares sell them or are perceived by the market as intending to sell them. Pursuant to the Subscription Agreements and the Amended and Restated Registration Rights Agreement, we have agreed to register the resale of the shares of Class A common stock certain Sellers receives in connection with the Business Combination, the founder shares and the shares of our common stock issued in the PIPE Investment, in accordance with the terms and subject to the conditions of the Subscription Agreements and Amended and Restated Registration Rights Agreement, as applicable.
We are not registering the shares of Class A common stock issuable upon exercise of the warrants under the Securities Act or any state securities laws at this time, and such registration may not be in place when an investor desires to exercise warrants, thus precluding such investor from being able to exercise its warrants except on a cashless basis and potentially causing such warrants to expire worthless.
We are not registering the shares of our Class A common stock issuable upon exercise of the warrants under the Securities Act or any state securities laws at this time. However, under the terms of the warrant agreement, we have agreed that as soon as practicable, but in no event later than 15 business days after the closing of our initial business combination, we will use our reasonable best efforts to file with the SEC, and within 60 business days following our initial business combination to have declared effective, a registration statement covering the issuance of the shares of our Class A common stock issuable upon exercise of the warrants and to maintain a current prospectus relating to those shares of Class A common stock until the warrants expire or are redeemed. We cannot assure you that we will be able to do so if, for example, any facts or events arise which represent a fundamental change in the information set forth in the registration statement or prospectus, the financial statements contained or incorporated by reference therein are not current, complete or correct or the SEC issues a stop order. If the shares issuable upon exercise of the warrants are not registered under the Securities Act, we will be required to permit holders to exercise their warrants on a cashless basis. However, no warrant will be exercisable for cash or on a cashless basis, and we will not be obligated to issue any shares to holders seeking to exercise their warrants, unless the issuance of the shares upon such exercise is registered or qualified under the securities laws of the state of the exercising holder or an exemption from registration is available.
113
Table of Contents
Notwithstanding the above, if our Class A common stock is at the time of any exercise of a warrant not listed on a national securities exchange such that it satisfies the definition of a covered security under Section 18(b)(1) of the Securities Act, we may, at our option, require holders of public warrants who exercise their warrants to do so on a cashless basis in accordance with Section 3(a)(9) of the Securities Act and, in the event we so elect, we will not be required to file or maintain in effect a registration statement, but we will use our best efforts to register or qualify the shares under applicable blue sky laws to the extent an exemption is not available. In no event will we be required to net cash settle any warrant, or issue securities or other compensation in exchange for the warrants in the event that we are unable to register or qualify the shares underlying the warrants under applicable state securities laws and no exemption is available. If the issuance of the shares upon exercise of the warrants is not so registered or qualified or exempt from registration or qualification, the holder of such warrant shall not be entitled to exercise such warrant and such warrant may have no value and expire worthless. In such event, holders who acquired their warrants as part of a purchase of units will have paid the full unit purchase price solely for the shares of Class A common stock included in the units. If and when the warrants become redeemable by us, we may exercise our redemption right even if we are unable to register or qualify the underlying shares of Class A common stock for sale under all applicable state securities laws.
If third parties bring claims against us, the proceeds held in the trust account could be reduced and the per share redemption amount received by stockholders may be less than $10.00 per share (which was the offering price per unit in our IPO).
Our placing of funds in the trust account may not protect those funds from third-party claims against us. Although we will seek to have all vendors, service providers (other than our independent registered public accounting firm), prospective target businesses or other entities with which we do business execute agreements with us waiving any right, title, interest or claim of any kind in or to any monies held in the trust account, there is no guarantee that they will execute such agreements or even if they execute such agreements that they would be prevented from bringing claims against the trust account, including, but not limited to, fraudulent inducement, breach of fiduciary responsibility or other similar claims, as well as claims challenging the enforceability of the waiver, in each case in order to gain advantage with respect to a claim against our assets, including the funds held in the trust account. If any third party refuses to execute an agreement waiving such claims to the monies held in the trust account, our management will perform an analysis of the alternatives available to it and will enter into an agreement with a third party that has not executed a waiver only if management believes that such third partys engagement would be significantly more beneficial to us than any alternative.
Examples of possible instances where we may engage a third party that refuses to execute a waiver include the engagement of a third party consultant whose particular expertise or skills are believed by management to be significantly superior to those of other consultants that would agree to execute a waiver or in cases where we are unable to find a service provider willing to execute a waiver. In addition, there is no guarantee that such entities will agree to waive any claims they may have in the future as a result of, or arising out of, any negotiations, contracts or agreements with us and will not seek recourse against the trust account for any reason. Upon redemption of our public shares, if we are unable to complete our initial business combination within the prescribed time frame, or upon the exercise of a redemption right in connection with our initial business combination, we will be required to provide for payment of claims of creditors that were not waived that may be brought against us within the 10 years following redemption. Accordingly, the per share redemption amount received by public stockholders could be less than the $10.00 per share initially held in the trust account, due to claims of such creditors.
The Sponsor has agreed that it will be liable to us if and to the extent any claims by a third party (other than our independent registered public accounting firm) for services rendered or products sold to us, or a prospective target business with which we have discussed entering into a transaction agreement, reduce the amount of funds in the trust account to below (1) $10.00 per public share or (2) such lesser amount per public share held in the trust account as of the date of the liquidation of the trust account due to reductions in the value of the trust assets, in each case net of the interest which may be withdrawn to pay taxes, except as to any claims by a third party
114
Table of Contents
who executed a waiver of any and all rights to seek access to the trust account and except as to any claim under our indemnity of the underwriters of the IPO against certain liabilities, including under the Securities Act. Moreover, in the event that an executed waiver is deemed to be unenforceable against a third party, the Sponsor will not be responsible to the extent of any liability for such third party claims. We have not independently verified whether the Sponsor has sufficient funds to satisfy its indemnity obligations and we have not asked the Sponsor to reserve for such indemnification obligations. As a result, if any such claims were successfully made against the trust account, the funds available for our initial business combination and redemptions could be reduced to less than $10.00 per public share. In such event, we may not be able to complete our initial business combination, and you would receive such lesser amount per share in connection with any redemption of your public shares. No member of our management team will indemnify us for claims by third parties including, without limitation, claims by vendors and prospective target businesses.
If, after we distribute the proceeds in the trust account to our public stockholders, we file a bankruptcy petition or an involuntary bankruptcy petition is filed against us that is not dismissed, a bankruptcy court may seek to recover such proceeds, and the members of our Board may be viewed as having breached their fiduciary duties to our creditors, thereby exposing the members of our Board and us to claims of punitive damages.
If, after we distribute the proceeds in the trust account to our public stockholders, we file a bankruptcy petition or an involuntary bankruptcy petition is filed against us that is not dismissed, any distributions received by stockholders could be viewed under applicable debtor/creditor and/or bankruptcy laws as either a preferential transfer or a fraudulent conveyance. As a result, a bankruptcy court could seek to recover some or all amounts received by our stockholders. In addition, our Board may be viewed as having breached its fiduciary duty to our creditors and/or having acted in bad faith by paying public stockholders from the trust account prior to addressing the claims of creditors, thereby exposing itself and us to claims of punitive damages.
If, before distributing the proceeds in the trust account to our public stockholders, we file a bankruptcy petition or an involuntary bankruptcy petition is filed against us that is not dismissed, the claims of creditors in such proceeding may have priority over the claims of our stockholders and the per-share amount that would otherwise be received by our stockholders in connection with our liquidation may be reduced.
If, before distributing the proceeds in the trust account to our public stockholders, we file a bankruptcy petition or an involuntary bankruptcy petition is filed against us that is not dismissed, the proceeds held in the trust account could be subject to applicable bankruptcy law, and may be included in our bankruptcy estate and subject to the claims of third parties with priority over the claims of our stockholders. To the extent any bankruptcy claims deplete the trust account, the per-share amount that would otherwise be received by our public stockholders in connection with our liquidation would be reduced.
Our stockholders may be held liable for claims by third parties against us to the extent of distributions received by them upon redemption of their shares.
Under the DGCL, stockholders may be held liable for claims by third parties against a corporation to the extent of distributions received by them in a dissolution. The pro rata portion of our trust account distributed to our public stockholders upon the redemption of our public shares in the event we do not complete our initial business combination within the required time period may be considered a liquidating distribution under Delaware law. If a corporation complies with certain procedures set forth in Section 280 of the DGCL intended to ensure that it makes reasonable provision for all claims against it, including a 60-day notice period during which any third-party claims can be brought against the corporation, a 90-day period during which the corporation may reject any claims brought, and an additional 150-day waiting period before any liquidating distributions are made to stockholders, any liability of stockholders with respect to a liquidating distribution is limited to the lesser of such stockholders pro rata share of the claim or the amount distributed to the stockholder, and any liability of the stockholder would be barred after the third anniversary of the dissolution. However, it is our intention to redeem
115
Table of Contents
our public shares as soon as reasonably possible following the required time period in the event we do not complete our initial business combination and, therefore, we do not intend to comply with the foregoing procedures.
Because we do not intend to comply with Section 280 of the DGCL, Section 281(b) of the DGCL requires us to adopt a plan, based on facts known to us at such time that will provide for our payment of all existing and pending claims or claims that may be potentially brought against us within the 10 years following our dissolution. However, because we are a blank check company, rather than an operating company, and our operations are limited to searching for prospective target businesses to acquire, the only likely claims to arise would be from our vendors (such as lawyers, investment bankers, consultants, etc.) or prospective target businesses. If our plan of distribution complies with Section 281(b) of the DGCL, any liability of stockholders with respect to a liquidating distribution is limited to the lesser of such stockholders pro rata share of the claim or the amount distributed to the stockholder, and any liability of the stockholder would likely be barred after the third anniversary of the dissolution. We cannot assure you that we will properly assess all claims that may be potentially brought against us. As such, our stockholders could potentially be liable for any claims to the extent of distributions received by them (but no more) and any liability of our stockholders may extend beyond the third anniversary of such date. Furthermore, if the pro rata portion of our trust account distributed to our public stockholders upon the redemption of our public shares in the event we do not complete our initial business combination within the required time period is not considered a liquidating distribution under Delaware law and such redemption distribution is deemed to be unlawful, then pursuant to Section 174 of the DGCL, the statute of limitations for claims of creditors could then be six years after the unlawful redemption distribution, instead of three years, as in the case of a liquidating distribution.
The trading price of our Class A common stock, warrants and units may be volatile.
Upon and following the consummation of the Business Combination, the trading price of our Class A common stock, warrants and units may highly volatile and subject to wide fluctuations due to a number of factors control. Some of the factors that could negatively affect the market price of our Class A common stock, warrants and units or result in significant fluctuations in price, regardless of our actual operating performance, include:
| | actual or anticipated variations in our quarterly operating results; |
| | results of operations that vary from the expectations of securities analysts and investors; |
| | changes in expectations as to our future financial performance, including financial estimates by securities analysts and investors; |
| | changes in market valuations of similar companies; |
| | changes in the markets in which we operate; |
| | announcements by us, our competitors or our vendors of significant contracts, acquisitions, joint marketing relationships, joint ventures or capital commitments; |
| | announcements by third parties of significant claims or proceedings against us; |
| | additions or departures of key personnel; |
| | actions by stockholders, including the sale by the PIPE Investors of any of their shares of our common stock; |
| | speculation in the press or investment community; |
| | general market, economic and political conditions, such as recessions, interest rates, trade wars, pandemics (such as COVID-19) and acts of war or terrorism; |
| | our operating performance and the performance of other similar companies; |
| | our ability to accurately project future results and our ability to achieve those and other industry and analyst forecasts; |
116
Table of Contents
| | litigation with third parties, including governmental entities; and |
| | new legislation or other regulatory developments that adversely affect us, our markets or our industry. |
Furthermore, in recent years, the stock market has experienced significant price and volume fluctuations. This volatility has had a significant impact on the market price of securities issued by many companies, including companies in Mirions industry, and often occurs without regard to the operating performance of the affected companies. Therefore, factors that have little or nothing to do with us could cause the price of our Class A common stock, warrants and units to fluctuate, and these fluctuations or any fluctuations related to our company could cause the market price of our Class A common stock, warrants and units to decline materially.
In the past, following periods of market volatility, stockholders have instituted securities class action litigation. If we were involved in securities litigation, it could have a substantial cost and divert resources and the attention of our management team from our business regardless of the outcome of such litigation.
Compliance obligations under the Sarbanes-Oxley Act may make it more difficult for us to effectuate the Business Combination, require substantial financial and management resources and increase the time and costs of completing a business combination.
The fact that we are a blank check company will make compliance with the requirements of the Sarbanes-Oxley Act particularly burdensome on us as compared to other public companies because Mirion is not currently subject to Section 404 of the Sarbanes-Oxley Act. The standards required for a public company under Section 404 of the Sarbanes-Oxley Act are significantly more stringent than those required of Mirion as a privately held company. Management may not be able to effectively and timely implement controls and procedures that adequately respond to the increased regulatory compliance and reporting requirements that will be applicable to us after the Business Combination. If we are not able to implement the requirements of Section 404, including any additional requirements once we are no longer an emerging growth company, in a timely manner or with adequate compliance, we may not be able to assess whether our internal controls over financial reporting are effective, which may subject us to adverse regulatory consequences and could harm investor confidence and the market price of our securities. Additionally, once we are no longer an emerging growth company, we will be required to comply with the independent registered public accounting firm attestation requirement on our internal control over financial reporting. We currently anticipate losing our emerging growth company status if the Business Combination is consummated, we currently anticipate losing our emerging growth company status at 2022 year end.
The obligations associated with being a public company will involve significant expenses and will require significant resources and management attention, which may divert from our business operations.
As a public company, we are subject to the reporting requirements of the Exchange Act and the Sarbanes-Oxley Act. The Exchange Act requires the filing of annual, quarterly and current reports with respect to a public companys business and financial condition. The Sarbanes-Oxley Act requires, among other things, that a public company establish and maintain effective internal control over financial reporting. As a result, the post-business combination company will incur significant legal, accounting and other expenses that Mirion did not previously incur. Mirions entire management team and many of its other employees will need to devote substantial time to compliance, and may not effectively or efficiently manage its transition into a public company.
These rules and regulations will result in the post-business combination company incurring substantial legal and financial compliance costs and will make some activities more time-consuming and costly. For example, these rules and regulations will likely make it more difficult and more expensive for the post-business combination company to obtain director and officer liability insurance, and it may be required to accept reduced policy limits and coverage or incur substantially higher costs to obtain the same or similar coverage. As a result, it may be difficult for the post-business combination company to attract and retain qualified people to serve on its Board of Directors, its board committees or as executive officers.
117
Table of Contents
We are currently an emerging growth company and a smaller reporting company within the meaning of the Securities Act, and to the extent we have taken advantage of certain exemptions from disclosure requirements available to emerging growth companies or smaller reporting companies, this could make our securities less attractive to investors and may make it more difficult to compare our performance with other public companies.
We are currently an emerging growth company within the meaning of the Securities Act, as modified by the JOBS Act, and we may take advantage of certain exemptions from various reporting requirements that are applicable to other public companies that are not emerging growth companies including, but not limited to, not being required to comply with the auditor attestation requirements of Section 404 of the Sarbanes-Oxley Act, reduced disclosure obligations regarding executive compensation in our periodic reports and proxy statements, and exemptions from the requirements of holding a nonbinding advisory vote on executive compensation and stockholder approval of any golden parachute payments not previously approved. As a result, our stockholders may not have access to certain information they may deem important. We cannot predict whether investors will find our securities less attractive because we will rely on these exemptions. If some investors find our securities less attractive as a result of our reliance on these exemptions, the trading prices of our securities may be lower than they otherwise would be, there may be a less active trading market for our securities and the trading prices of our securities may be more volatile.
Further, Section 102(b)(1) of the JOBS Act exempts emerging growth companies from being required to comply with new or revised financial accounting standards until private companies (that is, those that have not had a Securities Act registration statement declared effective or do not have a class of securities registered under the Exchange Act) are required to comply with the new or revised financial accounting standards. The JOBS Act provides that a company can elect to opt out of the extended transition period and comply with the requirements that apply to non-emerging growth companies but any such election to opt out is irrevocable. We have elected not to opt out of such extended transition period, which means that when a standard is issued or revised and it has different application dates for public or private companies, we, as an emerging growth company, can adopt the new or revised standard at the time private companies adopt the new or revised standard. This may make comparison of our financial statements with another public company, which is neither an emerging growth company nor an emerging growth company which has opted out of using the extended transition period difficult or impossible because of the potential differences in accounting standards used.
We will remain an emerging growth company until the earlier of: (1) the last day of the fiscal year (a) following the fifth anniversary of the closing of the IPO, (b) in which we have total annual gross revenue of at least $1.07 billion or (c) in which we are deemed to be a large accelerated filer, which means the market value of our common equity that is held by non-affiliates exceeds $700 million as of the end of the prior second fiscal quarter; and (2) the date on which we have issued more than $1.00 billion in non-convertible debt securities during the prior three-year period.
Our public stockholders will experience dilution as a consequence of, among other transactions, the issuance of Class A common stock as consideration in the Business Combination and the PIPE Investment. Having a minority share position may reduce the influence that our current stockholders have on the management of the post-business combination company.
The issuance of a significant number of shares of our Class A common stock in the Business Combination and in the PIPE Investment will dilute the equity interest of our existing stockholders and may adversely affect prevailing market prices for our public shares and/or public warrants.
It is anticipated that, upon completion of the Business Combination:
| | the PIPE Investors (including GSAM Holdings, assuming no syndication of its subscription) will own approximately 44% of the outstanding GSAH common stock; |
| | our public stockholders will own approximately 37% of the outstanding GSAH common stock; |
118
Table of Contents
| | the Sellers, other than current members of Mirion management, will own approximately 15% of outstanding GSAH common stock; |
| | Sellers who are current members of Mirion management will own approximately 4% of the outstanding GSAH common stock; and |
| | the GS Sponsor will own 0% of the outstanding GSAH common stock (assuming, for this purpose, that none of the founder shares performance vesting conditions have been satisfied at the time of completion of the Business Combination). |
These levels of ownership interest: (i) assume that no public shares are elected to be redeemed in connection with the Business Combination, (ii) exclude the 18,750,000 outstanding founder shares held by the GS Sponsor (all of which are subject to the performance vesting conditions and forfeiture as described herein), (iii) assume no exercise of any warrants to purchase Class A common stock that will remain outstanding immediately following the Business Combination, (iv) exclude the issuance of any shares under the Incentive Plan following the Business Combination and (v) assume that only members of Mirions management team will elect to receive shares of GSAH Class B common stock and that the other Sellers receiving shares of GSAH common stock elect to receive shares of GSAH Class A common stock. If the actual facts are different from these assumptions, the above levels of ownership interest will be different. For more information, see Summary of the Proxy Statement/Prospectus Ownership of the Company Following the Business Combination and Unaudited Pro Forma Condensed Combined Financial Information.
The issuance of additional shares of Class A common stock will significantly dilute the equity interests of existing holders of our securities and may adversely affect prevailing market prices for our units, public shares or public warrants.
Warrants will become exercisable for our Class A common stock, which would increase the number of shares eligible for future resale in the public market and result in dilution to our stockholders.
If the Business Combination is completed, outstanding warrants to purchase an aggregate of approximately 18,750,000 shares of our Class A common stock will become exercisable in accordance with the terms of the warrant agreement. These warrants will become exercisable 30 days after the completion of the Business Combination. The exercise price of these warrants will be $11.50 per share. To the extent such warrants are exercised, additional shares of our Class A common stock will be issued, which will result in dilution to the holders of our Class A common stock and increase the number of shares eligible for resale in the public market. Sales of substantial numbers of such shares in the public market or the fact that such warrants may be exercised could adversely affect the market price of our Class A common stock. However, there is no guarantee that the public warrants will ever be in the money prior to their expiration, and as such, the warrants may expire worthless. See Even if the Business Combination is consummated, the public warrants may never be in the money, and they may expire worthless and the terms of the warrants may be amended in a manner adverse to a holder if holders of at least 50% of the then outstanding public warrants approve of such amendment.
Even if the Business Combination is consummated, the public warrants may never be in the money, and they may expire worthless and the terms of the warrants may be amended in a manner adverse to a holder if holders of at least 50% of the then outstanding public warrants approve of such amendment.
The warrants were issued in registered form under a warrant agreement with Continental Stock Transfer & Trust Company, N.A., as warrant agent, and us. The warrant agreement provides that the terms of the warrants may be amended without the consent of any holder to cure any ambiguity or correct any defective provision, but requires the approval by the holders of at least 50% of the then outstanding public warrants to make any change that adversely affects the interests of the registered holders of public warrants. Accordingly, we may amend the terms of the public warrants in a manner adverse to a holder if holders of at least 50% of the then outstanding public warrants approve of such amendment. Although our ability to amend the terms of the public warrants with the
119
Table of Contents
consent of at least 50% of the then outstanding public warrants is unlimited, examples of such amendments could be amendments to, among other things, increase the exercise price of the warrants, convert the warrants into cash or stock (at a ratio different than initially provided), shorten the exercise period or decrease the number of shares of our Class A common stock purchasable upon exercise of a warrant.
We may redeem your unexpired warrants prior to their exercise at a time that is disadvantageous to you, thereby making your warrants worthless.
We have the ability to redeem outstanding warrants at any time after they become exercisable and prior to their expiration, at a price of $0.01 per warrant, provided that the last reported sales price of our Class A common stock equals or exceeds $18.00 per share (as adjusted for stock splits, stock dividends, reorganizations, recapitalizations and the like) for any 20 trading days within a 30 trading-day period ending on the third trading day prior to the date we send the notice of redemption to the warrant holders. If and when the warrants become redeemable by us, we may exercise our redemption right even if we are unable to register or qualify the underlying securities for sale under all applicable state securities laws. Redemption of the outstanding warrants could force you to: (1) exercise your warrants and pay the exercise price therefor at a time when it may be disadvantageous for you to do so (2) sell your warrants at the then-current market price when you might otherwise wish to hold your warrants; or (3) accept the nominal redemption price which, at the time the outstanding warrants are called for redemption, is likely to be substantially less than the market value of your warrants.
In addition, we may redeem your warrants after they become exercisable for a number of shares of Class A common stock determined based on the redemption date and the fair market value of our Class A common stock. Any such redemption may have similar consequences to a cash redemption described above. In addition, such redemption may occur at a time when the warrants are out-of-the-money, in which case you would lose any potential embedded value from a subsequent increase in the value of the Class A common stock had your warrants remained outstanding.
None of the private placement warrants will be redeemable by us so long as they are held by the Sponsor or its permitted transferees.
The NYSE may not list our securities on its exchange, which could limit investors ability to make transactions in our securities and subject us to additional trading restrictions.
In connection with the Business Combination, in order to continue to maintain the listing of our securities on the NYSE, we will be required to demonstrate compliance with the NYSEs initial listing requirements, which are more rigorous than the NYSEs continued listing requirements. We will seek to have the post-business combination companys securities listed on the NYSE upon consummation of the Business Combination. We cannot assure you that we will be able to meet all initial listing requirements. Even if the post-business combination companys securities are listed on the NYSE, we may be unable to maintain the listing of its securities in the future.
If we fail to meet the initial listing requirements and the post-business combination companys securities are not listed on the NYSE or on another national securities exchange, we could face significant material adverse consequences, including:
| | a limited availability of market quotations for our securities; |
| | reduced liquidity for our securities; |
| | a determination that our Class A common stock is a penny stock which will require brokers trading in our Class A common stock to adhere to more stringent rules and possibly result in a reduced level of trading activity in the secondary trading market for our securities; |
120
Table of Contents
| | a limited amount of news and analyst coverage; and |
| | a decreased ability to issue additional securities or obtain additional financing in the future. |
The National Securities Markets Improvement Act of 1996, which is a federal statute, prevents or preempts the states from regulating the sale of certain securities, which are referred to as covered securities. If the post-business combination companys securities were not listed on the NYSE, such securities would not qualify as covered securities and we would be subject to regulation in each state in which we offer our securities because states are not preempted from regulating the sale of securities that are not covered securities.
The coverage of our business or our securities by securities or industry analysts or the absence thereof could adversely affect our securities and trading volume.
The trading market for our securities will be influenced in part by the research and other reports that industry or securities analysts may publish about us or our business or industry from time to time. We do not control these analysts or the content and opinions included in their reports. As a former blank check company, we may be slow to attract equity research coverage, and the analysts who publish information about our securities will have had relatively little experience with our company, which could affect their ability to accurately forecast our results and make it more likely that we fail to meet their estimates. If no or few analysts commence equity research coverage of us, the trading price and volume of our securities would likely be negatively impacted. If analysts do cover us and one or more of them downgrade our securities, or if they issue other unfavorable commentary about us or our industry or inaccurate research, our stock price would likely decline. Furthermore, if one or more of these analysts cease coverage or fail to regularly publish reports on us, we could lose visibility in the financial markets. Any of the foregoing would likely cause our stock price and trading volume to decline.
We may be subject to certain ownership and voting power laws and regulations which may limit the ability of stockholders to acquire our Class A common stock and therefore limit demand for our Class A common stock. Our organizational and governing documents may include provisions to comply with such laws and regulations.
Under foreign direct investment and public interest laws, including in Germany, Finland, France, and the UK, and potentially other jurisdictions, certain acquisitions of our Class A common stock by investors are subject to government approval requirements. For example, in Germany, German foreign direct investment law may require foreign investors to obtain approval from the German Federal Ministry for Economic Affairs and Energy for the direct or indirect acquisition of shares of a German company if the acquirer directly or indirectly holds at least 10% of the voting rights of the company following the acquisition. Any acquisition in violation of the aforementioned provisions of German foreign direct investment law may be void. Any violation of the prohibition to consummate an acquisition without approval of the Ministry may be subject to sanctions. Similar foreign direct investment laws exist in other jurisdictions in which we have substantial operations. In Finland, government approvals are required if an investor holds at least 10% of the voting rights of the company following the investment. In France, the prior approval from the French Minister of Economy is required if a non-EU investor exceeds, directly or indirectly, 25% of the voting rights of the French entities of the company following the investment or, for an EU non-French investor, in case of acquisition of control, direct or indirect, of the French entities. The U.K. will have a 25% voting rights threshold for mandatory filings under the National Security and Investment Act 2021 when the new regime becomes operational on January 4, 2022. Accordingly, these restrictions on and approval requirements for the acquisition of a substantial shareholding in our share capital may restrict certain investments and limit demand for shares of our Class A common stock.
Anti-takeover provisions contained in the New Mirion Organizational Documents, as well as provisions of Delaware law, could impair a takeover attempt.
Assuming the passage of the Charter Proposal, the New Mirion Organizational Documents will contain provisions that may discourage unsolicited takeover proposals that stockholders may consider to be in their best
121
Table of Contents
interests. We are also subject to anti-takeover provisions under Delaware law, which could delay or prevent a change of control. Together, these provisions may make more difficult the removal of management and may discourage transactions that otherwise could involve payment of a premium over prevailing market prices for our securities. Certain of these provisions provide:
| | no cumulative voting in the election of directors, which limits the ability of minority stockholders to elect director candidates; |
| | the requirement that directors may only be removed from the Board for cause; |
| | the right of our Board to elect a director to fill a vacancy created by the expansion of our Board or the resignation, death or removal of a director in certain circumstances, which prevents stockholders from being able to fill vacancies on our Board; |
| | a prohibition on stockholder action by written consent, which forces stockholder action to be taken at an annual or special meeting of our stockholders; |
| | a prohibition on stockholders calling a special meeting and the requirement that a meeting of stockholders may only be called by members of our Board or the Chief Executive Officer of GSAH, which may delay the ability of our stockholders to force consideration of a proposal or to take action, including the removal of directors; and |
| | advance notice procedures that stockholders must comply with in order to nominate candidates to our Board or to propose matters to be acted upon at a meeting of stockholders, which may discourage or deter a potential acquirer from conducting a solicitation of proxies to elect the acquirers own slate of directors or otherwise attempting to obtain control of GSAH. |
The New Mirion Charter includes forum selection clauses, which could discourage claims or limit stockholders ability to make a claim against us, our directors, officers, other employees or stockholders.
The current GSAH Certificate of Incorporation includes, and the New Mirion Charter will also include, forum selection clauses. The New Mirion Charter will provide that, unless we consent in writing to the selection of an alternative forum, the Court of Chancery in the State of Delaware shall be the sole and exclusive forum for any stockholder (including a beneficial owner) to bring: (a) any derivative action or proceeding brought on behalf of the Company; (b) any claim or cause of action for breach of a fiduciary duty owed by any current or former director, officer or other employee of the Company, to the Company or the Companys stockholders; (c) any claim or cause of action against the Company or any current or former director, officer or other employee of the Company, arising out of or pursuant to any provision of the DGCL or our certificate of incorporation or bylaws; (d) any claim or cause of action seeking to interpret, apply, enforce or determine the validity of our certificate of incorporation or bylaws (as each may be amended from time to time, including any right, obligation, or remedy thereunder), (e) any claim or cause of action as to which the DGCL confers jurisdiction on the Court of Chancery of the State of Delaware; and (f) any claim or cause of action against the Company or any current or former director, officer or other employee of the Company, governed by the internal-affairs doctrine, in all cases to the fullest extent permitted by law and subject to the court having personal jurisdiction over the indispensable parties named as defendants. In addition, the New Mirion Charter will provide that, unless New Mirion consents in writing to the selection of an alternative forum, the federal district courts of the United States will be the exclusive forum for resolving any complaint asserting a cause of action arising under the Securities Act. Notwithstanding the foregoing, the Securities Act forum selection clause will not apply to suits brought to enforce any liability or duty created by the Exchange Act or any other claim for which the federal district courts of the United States of America shall be the sole and exclusive forum. These forum selection clauses may discourage claims or limit stockholders ability to submit claims in a judicial forum that they find favorable and may result in additional costs for a stockholder seeking to bring a claim. While we believe the risk of a court declining to enforce these forum selection clauses is low, if a court were to determine a forum selection clause to be inapplicable or unenforceable in an action, we may incur additional costs in conjunction with our efforts to resolve the dispute in an alternative jurisdiction, which could have a negative impact on our results of operations and financial condition.
122
Table of Contents
Our warrants are accounted for as derivative liabilities and the changes in the value of our warrants have had and may continue to have a material effect on our financial results.
Our warrants are included on our balance sheet as of December 31, 2020 as derivative liabilities. ASC 815 provides for the remeasurement of the fair value of such derivatives at each balance sheet date, with a resulting non-cash gain or loss related to the change in the fair value being recognized in earnings in the statement of operations. As a result of the recurring fair value measurement, our financial statements and results of operations have fluctuated and may continue to fluctuate quarterly, based on factors which are outside of our control. Due to the recurring fair value measurement, we expect that we will recognize non-cash gains or losses on our warrants each reporting period and that the amount of such gains or losses could be material.
We have identified a material weakness in our internal control over financial reporting, and we may experience additional material weaknesses or otherwise fail to design and maintain effective internal control over financial reporting, our ability to timely and accurately report our financial condition and operating results in compliance with reporting requirements applicable for public companies in the United States could be impaired, which may adversely affect investor confidence in us and, as a result, the value of our Class A common stock.
Following the reassessment of the accounting treatment of the warrants, we determined that it was appropriate to restate the Companys historical financial results for the Non-Reliance Periods, in each case to reflect the change in accounting treatment. In connection with the foregoing development, the Company identified a material weakness in the design and operation of the Companys internal control over financial reporting. A material weakness is a deficiency, or a combination of deficiencies, in internal control over financial reporting, such that there is a reasonable possibility that a material misstatement of the Companys annual or interim financial statements will not be prevented or detected on a timely basis. Specifically, the Companys management has concluded that our control around the interpretation and accounting for certain complex features of the Class A common stock and warrants issued by the Company was not effectively designed or maintained.
This material weakness resulted in the restatement of the Companys financial statements for the year ended December 31, 2020, its balance sheet as of July 2, 2020, and its interim financial statements for the quarter ended September 30, 2020. Additionally, this material weakness could result in a misstatement of the warrant liability, Class A common stock and related accounts and disclosures that would result in a material misstatement of the financial statements that would not be prevented or detected on a timely basis.
Effective internal controls are necessary for us to provide reliable financial reports and prevent fraud. If we are unable to develop and maintain effective internal control over financial reporting we may not be able to accurately report our financial results in a timely manner, which may cause us to be unable to comply with securities law or applicable stock exchange requirements, adversely affect investor confidence in us and/or materially and adversely affect our business and operating result, and our stock price may decline as a result. Any required remediation measures may be time consuming and costly and there is no assurance that any measures taken to date or any such measures taken in the future will ultimately have the intended effects, including to avoid potential future material weaknesses.
Risks Related to the Redemption
Public stockholders who wish to redeem their public shares for a pro rata portion of the trust account must comply with specific requirements for redemption that may make it more difficult for them to exercise their redemption rights prior to the deadline. If stockholders fail to comply with the redemption requirements specified in this proxy statement, they will not be entitled to redeem their public shares for a pro rata portion of the funds held in the trust account.
A public stockholder will be entitled to receive cash for any public shares to be redeemed only if such public stockholder: (1)(a) holds public shares, or (b) if the public stockholder holds public shares through units, the public stockholder elects to separate its units into the underlying public shares and warrants prior to exercising its
123
Table of Contents
redemption rights with respect to the public shares; (2) prior to 5:00 p.m. Eastern Time on [], 2021 (two business days before the scheduled date of the Special Meeting) submits a written request to Continental Stock Transfer & Trust Company, N.A., our transfer agent, that we redeem all or a portion of your public shares for cash, affirmatively certifying in your request if you ARE or ARE NOT acting in concert or as a group (as defined in Section 13d-3 of the Exchange Act) with any other stockholder with respect to shares of our common stock; and (3) delivers its public shares to our transfer agent physically or electronically through DTC. In order to obtain a physical share certificate, a stockholders broker or clearing broker, DTC and our transfer agent will need to act to facilitate this request. It is our understanding that stockholders should generally allot at least two weeks to obtain physical certificates from our transfer agent. However, because we do not have any control over this process or over DTC, it may take significantly longer than two weeks to obtain a physical stock certificate. If it takes longer than anticipated to obtain a physical certificate, public stockholders who wish to redeem their public shares may be unable to obtain physical certificates by the deadline for exercising their redemption rights and thus will be unable to redeem their shares.
If the Business Combination is consummated, and if a public stockholder properly exercises its right to redeem all or a portion of the public shares that it holds, including timely delivering its shares to our transfer agent, we will redeem such public shares for a per-share price, payable in cash calculated as of two business days prior to the consummation of the Business Combination, including interest (net of taxes payable). Please see the section entitled Special Meeting of GSAH StockholdersRedemption Rights for additional information on how to exercise your redemption rights.
If a public stockholder fails to receive notice of our offer to redeem public shares in connection with the Business Combination, or fails to comply with the procedures for tendering its shares, such shares may not be redeemed.
If, despite our compliance with the proxy rules, a public stockholder fails to receive our proxy materials, such public stockholder may not become aware of the opportunity to redeem his, her or its public shares. In addition, the proxy materials that we are furnishing to holders of public shares in connection with the Business Combination describe the various procedures that must be complied with in order to validly redeem the public shares. In the event that a public stockholder fails to comply with these procedures, its public shares may not be redeemed. Please see the section entitled Special Meeting of GSAH StockholdersRedemption Rights for additional information on how to exercise your redemption rights.
If you or a group of stockholders of which you are a part are deemed to hold an aggregate of more than 15% of the public shares, you (or, if a member of such a group, all of the members of such group in the aggregate) will lose the ability to redeem all such shares in excess of 15% of the public shares.
A public stockholder, together with any of his, her or its affiliates or any other person with whom it is acting in concert or as a group (as defined under Section 13 of the Exchange Act), will be restricted from redeeming in the aggregate his, her or its shares or, if part of such a group, the groups shares, in excess of 15% of the public shares. In order to determine whether a stockholder is acting in concert or as a group with another stockholder, we will require each public stockholder seeking to exercise redemption rights to certify to us whether such stockholder is acting in concert or as a group with any other stockholder. Such certifications, together with other public information relating to stock ownership available to us at that time, such as Section 13D, Section 13G and Section 16 filings under the Exchange Act, will be the sole basis on which we make the above-referenced determination. Your inability to redeem any such excess shares will reduce your influence over our ability to consummate the Business Combination and you could suffer a material loss on your investment in us if you sell such excess shares in open market transactions. Additionally, you will not receive redemption distributions with respect to such excess shares if we consummate the Business Combination. As a result, you will continue to hold that number of shares aggregating to more than 15% of the public shares and, in order to dispose of such excess shares, would be required to sell your stock in open market transactions, potentially at a loss. We cannot assure you that the value of such excess shares will appreciate over time following the Business Combination or that the market price of the public shares will exceed the per-share redemption price. Notwithstanding the foregoing,
124
Table of Contents
stockholders may challenge our determination as to whether a stockholder is acting in concert or as a group with another stockholder in a court of competent jurisdiction.
However, our stockholders ability to vote all of their shares (including such excess shares) for or against the Business Combination is not restricted by this limitation on redemption.
There is some uncertainty regarding the U.S. federal income tax consequences to holders of our Class A common stock who elect to exercise their redemption rights.
There is some uncertainty regarding the U.S. federal income tax consequences to holders of our Class A common stock who exercise their redemption rights. The uncertainty of tax consequences relates primarily to the individual circumstances of the taxpayer and include (i) whether the redemption results in a distribution or a sale taxable as capital gain, and (ii) whether such capital gain, if applicable, is long-term or short-term capital gain. Whether the redemption qualifies for sale treatment will depend largely on whether the holder owns (or is deemed to own) any shares of our Class A common stock following the redemption, and if so, the total number of shares of our Class A common stock held by the holder both before and after the redemption relative to all shares of our Class A common stock outstanding both before and after the redemption. The redemption generally will be treated as a sale, rather than a distribution, if the redemption (i) is substantially disproportionate with respect to the holder, (ii) results in a complete termination of the holders interest in us or (iii) is not essentially equivalent to a dividend with respect to the holder. Due to the personal and subjective nature of certain of such tests and the absence of clear guidance from the U.S. Internal Revenue Service (the IRS), there is uncertainty as to how a holder who elects to exercise its redemption rights will be taxed in connection with the exercise of redemption rights.
Furthermore, any redemption (or portion thereof) by a Non-U.S. Holder (as defined below) treated as a distribution that constitutes a dividend for U.S. federal income tax purposes will generally be subject to withholding tax at a rate of 30% of the gross amount of the dividend (unless the Non-U.S. Holder establishes that it is eligible for a reduced rate of withholding tax under an applicable income tax treaty or certain other exceptions apply). Because the determination as to whether a redemption is treated as a sale or a distribution is dependent on matters of fact, withholding agents may presume, for withholding purposes, that all amounts paid to Non-U.S. Holders in connection with a redemption are treated as distributions in respect of such Non-U.S. Holders shares of our Class A common stock. Accordingly, a Non-U.S. Holder should expect that a withholding agent will likely withhold U.S. federal income tax on the gross proceeds payable to it pursuant to a redemption at a rate of 30% unless the Non-U.S. Holder is eligible for a reduced rate of withholding tax under an applicable income tax treaty and provides proper certification of its eligibility for such reduced rate (usually on an IRS Form W-8BEN or W-8BEN-E, or other applicable IRS Form W-8).
Holders of our Class A common stock are urged to consult their own tax advisors regarding the U.S. federal income tax consequences of the redemption to them. For a more detailed explanation of the U.S. federal income tax consequences of the redemption, see the section entitled Proposal No. 1Approval of the Business CombinationUnited States Federal Income Tax Considerations to Stockholders Exercising Redemption Rights.
Risks if the Business Combination is not Consummated
If we are not able to complete the Business Combination with Mirion by July 2, 2022 (or if such date is extended at a duly called meeting of stockholders, such later date) nor able to complete another initial business combination by such date, we would cease all operations except for the purpose of winding up and we would redeem our public shares and liquidate, in which case our public stockholders may receive only $10.00 per share, or less than such amount in certain circumstances, and our warrants will expire worthless.
If we are not able to complete the Business Combination with Mirion by July 2, 2022 (or if such date is extended at a duly called meeting of stockholders, such later date) nor able to complete another initial business combination by such date, we will: (1) cease all operations except for the purpose of winding up; (2) as promptly
125
Table of Contents
as reasonably possible but not more than 10 business days thereafter, redeem the public shares, at a per share price, payable in cash, equal to the aggregate amount then on deposit in the trust account, including interest (which interest shall be net of taxes payable, and less up to $100,000 of interest to pay dissolution expenses), divided by the number of then outstanding public shares, which redemption will completely extinguish public stockholders rights as stockholders (including the right to receive further liquidating distributions, if any); and (3) as promptly as reasonably possible following such redemption, subject to the approval of our remaining stockholders and our Board, dissolve and liquidate, subject in each case to our obligations under Delaware law to provide for claims of creditors and the requirements of other applicable law. In such case, our public stockholders may receive only $10.00 per share, or less than $10.00 per share, on the redemption of their shares, and our warrants will expire worthless.
You will not have any rights or interests in funds from the trust account, except under certain limited circumstances. To liquidate your investment, therefore, you may be forced to sell your public shares or public warrants, potentially at a loss.
Our public stockholders will be entitled to receive funds from the trust account only upon the earlier to occur of: (1) the completion of our initial business combination, and then only in connection with those shares of Class A common stock that such stockholder properly elected to redeem, subject to the limitations described herein; (2) the redemption of any public shares properly tendered in connection with a stockholder vote to amend our amended and restated certificate of incorporation to modify the substance or timing of our obligation to redeem 100% of our public shares if we do not complete our initial business combination within the required time period; and (3) the redemption of all of our public shares if we are unable to complete our initial business combination by July 2, 2022 (or if such date is extended at a duly called meeting of stockholders, such later date), subject to applicable law and as further described herein. In addition, if we are unable to complete an initial business combination within the required time period for any reason, compliance with Delaware law may require that we submit a plan of dissolution to our then-existing stockholders for approval prior to the distribution of the proceeds held in our trust account. In that case, public stockholders may be forced to wait beyond the end of the required time period before they receive funds from our trust account. In no other circumstances will a public stockholder have any right or interest of any kind in the trust account. Holders of warrants will not have any right to the proceeds held in the trust account with respect to the warrants. Accordingly, to liquidate your investment, you may be forced to sell your public shares or public warrants, potentially at a loss.
If the funds not being held in the trust account are insufficient to allow us to operate until at least July 2, 2022, we may be unable to complete our initial business combination.
The funds available to us outside of the trust account may not be sufficient to allow us to operate until July 2, 2022, assuming that our initial business combination is not completed during that time. We expect to incur significant costs in pursuit of our acquisition plans. However, our affiliates are not obligated to make loans to us or invest in us in the future (other than the GS Sponsor Capital Commitment), and we may not be able to raise additional financing from unaffiliated parties necessary to fund our expenses. Any such event in the future may negatively impact the analysis regarding our ability to continue as a going concern at such time.
Of the funds available to us, we could use a portion of the funds available to us to pay fees to consultants to assist us with our search for a target business. We could also use a portion of the funds as a down payment or to fund a no-shop provision (a provision in letters of intent or Business Combination Agreements designed to keep target businesses from shopping around for transactions with other companies or investors on terms more favorable to such target businesses) with respect to a particular proposed business combination, although we do not have any current intention to do so. If we entered into a letter of intent or Business Combination Agreement where we paid for the right to receive exclusivity from a target business and were subsequently required to forfeit such funds (whether as a result of our breach or otherwise), we might not have sufficient funds to continue searching for, or conduct due diligence with respect to, a target business. If we are unable to complete our initial business combination, our public stockholders may receive only approximately $10.00 per share, or less in certain
126
Table of Contents
circumstances, on the liquidation of our trust account and our warrants will expire worthless. See Risks Related to the Business Combination and GSAHIf third parties bring claims against us, the proceeds held in the trust account could be reduced and the per share redemption amount received by stockholders may be less than $10.00 per share (which was the offering price per unit in our IPO) and other risk factors herein.
127
Table of Contents
UNAUDITED PRO FORMA CONDENSED COMBINED FINANCIAL INFORMATION
The following unaudited pro forma condensed combined balance sheet as of March 31, 2021 and the unaudited pro forma condensed combined statements of operations for the year ended December 31, 2020 and the three months ended March 31, 2021 present the historical financial statements of GSAH, adjusted to reflect the Business Combination. The Company and Mirion shall collectively be referred to herein as the Companies. The Companies, subsequent to the Business Combination, shall be referred to herein as the Combined Company. The unaudited condensed combined financial information presents the pro forma effects of the following transactions:
| | The Business Combination of Mirion with GSAH pursuant to the Business Combination Agreement and other agreements entered into as part of the Business Combination; |
| | Conversion of the GSAH Class B common stock outstanding prior to the Business Combination to GSAH Class A common stock; |
| | Execution of Subscription Agreements with the PIPE Investors pursuant to which the PIPE Investors have collectively subscribed for 90.0 million shares of the GSAH Class A common stock for an aggregate purchase price equal to $900 million (the PIPE Investment), $200 million of which has been subscribed for by GSAM Holdings (the Backstop Party) unless it chooses to syndicate such subscription. The PIPE Investment will be consummated substantially concurrently with the closing of the Business Combination; |
| | At the Closing, the Sellers (or the Mirion Sellers) will have the option to elect to receive equity consideration either in the form of shares of GSAH Class A common stock or shares of GSAH Class B common stock of the Company that will have voting rights but no economic interest in the Company, paired with shares of IntermediateCo Class B common stock (non-voting) of a newly formed subsidiary (IntermediateCo) (the Paired Interests). The Company will own 100% of the voting shares (Class A) of IntermediateCo but a portion of the economic interest of IntermediateCo will accrue to the management holders of IntermediateCo Class B common stock and GSAH Class B common stock in proportion to their ownership of GSAH Class B common stock, or voting interest, in the Company. As a result, the Company will recognize a noncontrolling interest for the portion of IntermediateCo that is not attributable to the Company. We have assumed only members of Mirions management team will elect to receive shares of Class B common stock (the Class B Holders) and that the remaining Mirion Sellers receiving shares of GSAH common stock will elect to receive GSAH Class A common stock; |
| | The transfer of a portion of the founder shares to executives and a board member of the Combined Company, to be forfeited if certain service and performance conditions are not met within five years of the Transaction Date. This transaction will be accounted for as stock compensation expense in the financial statements of the Combined Company; |
| | Repayment of Mirion third-party and related party notes and entering into a new term loan facility; and |
| | The pro forma impact of the acquisition by Mirion of the Sun Nuclear Corporation (SNC or Sun Nuclear) on December 18, 2020 (the Sun Acquisition) which was deemed a significant acquisition to Mirion under Regulation S-X Article 11, Proforma Financial Information.). |
The unaudited pro forma condensed combined balance sheet as of March 31, 2021 assumes that the Business Combination was completed on March 31, 2021 except with respect to the PIK Notes, as described below. The unaudited pro forma condensed combined statements of operations for the year ended December 31, 2020 and for the three months ended March 31, 2021 give pro forma effect to the Business Combination as if it had occurred on January 1, 2020. The consideration to be paid to the Mirion Sellers in connection with the Business Combination is allocated first in respect of the PIK Notes and second in respect of the Existing Mirion Shares. The PIK Notes accrue payment-in-kind (PIK) interest daily at a rate of 11.5% annually and are contemplated to be sold at the Closing for a price equal to the full outstanding principal amount together with all accrued but
128
Table of Contents
unpaid interest up to but excluding the Closing Date. See Certain Relationships and Related Persons TransactionsMirions Related Person TransactionsShareholder Notes. Accordingly, as the amount of accrued and unpaid interest increases, the holders of the PIK Notes will receive more consideration as compared to the holders of the Existing Mirion Shares. This has the effect over time of increasing the amount of shares of GSAH Class A Common Stock to be issued to the Mirion Sellers (excluding members of Mirion management) and reducing the number of shares of GSAH Class B common stock that will be issued to Mirion management (there is no incremental dilution to Public Stockholders). Accordingly, for purposes of estimating the amount of GSAH Class A Common Stock and GSAH Class B Common Stock to be outstanding, we have assumed an amount of principal and interest of the PIK Notes as if the Closing Date would be October 31, 2021, but for all other purposes have assumed the Closing Date will be March 31, 2021
GSAHs fiscal year ends on December 31, whereas Mirions fiscal year ends June 30. Due to this difference, the unaudited pro forma condensed combined statement of operations, which we refer to as the pro forma condensed combined statement of operations, for the year ended December 31, 2020, is derived from GSAHs audited consolidated statement of operations for the year ended December 31, 2020, and Mirions unaudited financial results for the twelve-month period from January 1, 2020 through December 31, 2020. Mirion arrived at the unaudited financial results for the twelve-month period ended December 31, 2020 by aggregating the results for each quarterly period in calendar year 2020 (i.e., quarters ending March 31, 2020, June 30, 2020, September 30, 2020, and December 31, 2020). The pro forma condensed combined statement of operations for the three months ended March 31, 2021, combines the unaudited consolidated statement of operations for both GSAH and Mirion during the same period. Mirions balances have been classified consistently with the Companys presentation.
On June 17, 2021, the Company entered into the Business Combination Agreement. After giving effect to the Business Combination, the Company is expected to own between 95% and 96% of Mirion and its subsidiaries, and the Charterhouse Parties and the other Sellers (including certain members of Mirion management) are expected to hold between 15% and 18% of the outstanding shares of GSAH Class A common stock (excluding the founder shares) and all of the outstanding shares of the GSAH Class B common stock. The pro forma condensed combined information contained herein assumes the Companys stockholders approve the proposed Business Combination. The Companys public stockholders may elect to redeem their shares of GSAH Class A common stock even if they approve the proposed Business Combination. The Company cannot predict how many of its public stockholders will elect to redeem their shares of GSAH Class A common stock for cash. As a result, the Company has provided pro forma condensed combined financial statements under two different redemption scenarios:
| | Assuming no redemptions: This presentation assumes that no shares of GSAH Class A common stock are redeemed. |
| | Assuming maximum redemptions: This presentation assumes that the maximum number of shares of GSAH Class A common stock are redeemed such that the remaining funds held in the trust account after the payment of the redeeming shares pro-rata allocation along with the proceeds from the PIPE Investment are sufficient to fund the Minimum Cash Condition (not less than $1,310 million available for use as cash consideration to the Sellers and to be retained on the balance sheet of the Combined Company). Based on the amount of $750.1 million in the trust account as of March 31, 2021, inclusive of accrued dividends, and considering the anticipated gross proceeds of approximately $900.0 million from the PIPE Investment, the aggregate commitment of $830 million from a first lien term facility pursuant to the Debt Commitment Letter and approximately $125.0 million from the Backstop Party, approximately 36,610,000 shares of GSAH Class A common stock may be redeemed and still enable us to have sufficient cash to satisfy the cash closing conditions in the Business Combination Agreement, including the debt refinancing of the Mirion 2019 Credit Facility. |
The actual redemptions will likely be within the scenarios described above; however, there can be no assurance regarding which scenario will be closest to the actual results. Under both scenarios, the Company is considered the accounting acquirer, as further discussed in Basis of the Pro Forma Presentation. The business
129
Table of Contents
combination is accounted for under the scope of Financial Accounting Standards Boards Accounting Standards Codification (ASC) Topic 805, Business Combinations (ASC 805). Pursuant to ASC 805, GSAH has been determined to be the accounting acquirer as GSAH is transferring cash via the use of funds in their trust account and proceeds from equity issuances to execute the business combination. The cash consideration to the sellers is equal to an amount greater than a majority of the total consideration exchanged.
The transfer of cash in exchange for the majority of the sellers equity supports the conclusion that GSAH is the accounting acquirer in the business combination. Mirion constitutes a business in accordance with ASC 805, and the business combination constitutes a change in control.
130
Table of Contents
GS ACQUISITION HOLDINGS CORP II
UNAUDITED PRO FORMA CONDENSED COMBINED BALANCE SHEET
MARCH 31, 2021
| Historical as of March 31, 2021 |
Pro Forma Financing Adjustments (Assuming No Redemptions) |
As of March 31, 2021 |
||||||||||||||||||||||||||
| ($ in millions) | GS Acquisition Holdings Corp II |
Mirion | Pro Forma Purchase Accounting Adjustments |
Notes | Notes | Pro Forma Combined (Assuming No Redemptions) |
||||||||||||||||||||||
| ASSETS |
||||||||||||||||||||||||||||
| Current assets: |
||||||||||||||||||||||||||||
| Cash and cash equivalents |
$ | 1.7 | $ | 86.8 | $ | 1.0 | (a) | $ | 900.0 | (b) | $ | 291.1 | ||||||||||||||||
| (908.5 | ) | (b) | 750.1 | (b) (d) | ||||||||||||||||||||||||
| (1,310.0 | ) | (b) | (11.8 | ) | (b) | |||||||||||||||||||||||
| (48.2 | ) | (b) | ||||||||||||||||||||||||||
| 830.0 | (b) | |||||||||||||||||||||||||||
| Accounts receivable, net |
| 127.1 | | | 127.1 | |||||||||||||||||||||||
| Costs in excess of billings |
| 59.7 | | | 59.7 | |||||||||||||||||||||||
| Inventories |
| 118.1 | 34.9 | (a) | | 153.0 | ||||||||||||||||||||||
| Other currents assets |
0.5 | 32.2 | (0.9 | ) | (a) | | 31.8 | |||||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||||||
| Total current assets |
2.2 | 423.9 | (2,183.5 | ) | 2,420.1 | 662.7 | ||||||||||||||||||||||
| Property, plant, and equipment, net |
| 89.0 | 18.8 | (a) | | 107.8 | ||||||||||||||||||||||
| Other assets: |
||||||||||||||||||||||||||||
| Cash and cash equivalents held in Trust |
750.1 | | | (750.1 | ) | (d) | | |||||||||||||||||||||
| Goodwill |
| 677.5 | 1,005.0 | (a) | | 1,682.5 | ||||||||||||||||||||||
| Intangible assets, net |
| 343.3 | 376.2 | (a) | | 719.5 | ||||||||||||||||||||||
| Other assets |
0.4 | 10.9 | (0.1 | ) | (a) | | 11.2 | |||||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||||||
| Total other assets |
750.5 | 1,031.7 | 1,381.1 | (750.1 | ) | 2,413.2 | ||||||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||||||
| Total assets |
$ | 752.7 | $ | 1,544.6 | $ | (783.6 | ) | $ | 1,670.0 | $ | 3,183.7 | |||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||||||
| LIABILITIES AND STOCKHOLDERS DEFICIT |
||||||||||||||||||||||||||||
| Current liabilities: |
||||||||||||||||||||||||||||
| Accounts payable |
$ | 1.4 | $ | 55.5 | $ | | $ | | $ | 56.9 | ||||||||||||||||||
| Deferred contract revenue |
| 49.9 | (16.2 | ) | (a) | | 33.7 | |||||||||||||||||||||
| Accrued offering costs |
0.3 | | | | 0.3 | |||||||||||||||||||||||
| Working capital note |
1.5 | | | | 1.5 | |||||||||||||||||||||||
| Warrant liability |
61.5 | | | | 61.5 | |||||||||||||||||||||||
| Notes payable to third-parties, current |
| 5.5 | (5.5 | ) | (a) (c) | 8.3 | (b) | 8.3 | ||||||||||||||||||||
| Accrued expenses and other current liabilities |
| 91.7 | | 7.4 | (e) | 99.1 | ||||||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||||||
| Total current liabilities |
64.7 | 202.6 | (21.7 | ) | 15.7 | 261.3 | ||||||||||||||||||||||
| Deferred underwriting discount |
26.3 | | | (26.3 | ) | (f) | | |||||||||||||||||||||
| Third-party notes payable, non-current, net |
| 885.5 | (885.5 | ) | (a) (c) | 821.7 | (b) | 803.3 | ||||||||||||||||||||
| (18.4 | ) | (e) | ||||||||||||||||||||||||||
| Related party notes payable, non-current, net |
| 1,202.5 | (1,202.5 | ) | (a) | | | |||||||||||||||||||||
| Deferred income taxes and other liabilities |
| 72.8 | 105.5 | (a) | | 178.3 | ||||||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||||||
| Total liabilities |
91.0 | 2,363.4 | (2,004.2 | ) | 792.7 | 1,242.9 | ||||||||||||||||||||||
| GSAH Class A common stock subject to redemption |
750.0 | | | (750.0 | ) | (g) | | |||||||||||||||||||||
| Stockholders deficit: |
||||||||||||||||||||||||||||
| A Ordinary shares |
| | | | (g) | | ||||||||||||||||||||||
| B Ordinary shares |
| 0.1 | (0.1 | ) | (a) | | (g) | | ||||||||||||||||||||
| Additional paid-in capital |
| 9.5 | (9.5 | ) | (a) | 900.0 | (b) | 1,954.3 | ||||||||||||||||||||
| 401.8 | (a) | 750.0 | (g) | |||||||||||||||||||||||||
| (11.8 | ) | (a) (b) | ||||||||||||||||||||||||||
| (85.7 | ) | (h) | ||||||||||||||||||||||||||
| Receivable from Employees for purchase of Stock |
| (2.5 | ) | 2.5 | (a) | | | |||||||||||||||||||||
| Accumulated (deficit) earnings |
(88.3 | ) | (860.6 | ) | 860.6 | (a) | (10.9 | ) | (e) | (99.2 | ) | |||||||||||||||||
| Noncontrolling interests |
| 2.1 | (2.1 | ) | (a) | 85.7 | (h) | 85.7 | ||||||||||||||||||||
| Accumulated other comprehensive income (loss) |
| 32.6 | (32.6 | ) | (a) | | | |||||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||||||
| Total stockholders equity (deficit) |
(88.3 | ) | (818.8 | ) | 1,220.6 | 1,627.3 | 1,940.8 | |||||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||||||
| Total liabilities and stockholders equity |
$ | 752.7 | $ | 1,544.6 | $ | (783.6 | ) | $ | 1,670.0 | $ | 3,183.7 | |||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||||||
131
Table of Contents
GS ACQUISITION HOLDINGS CORP II
UNAUDITED PRO FORMA CONDENSED COMBINED BALANCE SHEET
MARCH 31, 2021
| Historical as of March 31, 2021 |
As of March 31, 2021 |
|||||||||||||||||||||||||||
| ($ in millions) | GS Acquisition Holdings Corp II |
Mirion | Pro Forma Purchase Accounting Adjustments |
Notes | Pro Forma Financing Adjustments (Assuming Maximum Redemptions) |
Notes | Pro Forma Combined (Assuming Maximum Redemptions) |
|||||||||||||||||||||
| ASSETS |
||||||||||||||||||||||||||||
| Current assets: |
||||||||||||||||||||||||||||
| Cash and cash equivalents |
$ | 1.7 | $ | 86.8 | $ | 1.0 | (a) | $ | 900.0 | (b) | $ | 50.0 | ||||||||||||||||
| (908.5 | ) | (b) | 750.1 | (b) (d) | ||||||||||||||||||||||||
| (1,310.0 | ) | (b) | (11.8 | ) | (b) | |||||||||||||||||||||||
| (48.2 | ) | (b) | ||||||||||||||||||||||||||
| 830.0 | (b) | |||||||||||||||||||||||||||
| 125.0 | (b) | |||||||||||||||||||||||||||
| (366.1 | ) | (b) | ||||||||||||||||||||||||||
| Accounts receivable, net |
| 127.1 | | | 127.1 | |||||||||||||||||||||||
| Costs in excess of billings |
| 59.7 | | | 59.7 | |||||||||||||||||||||||
| Inventories |
| 118.1 | 34.9 | (a) | | 153.0 | ||||||||||||||||||||||
| Other currents assets |
0.5 | 32.2 | (0.9 | ) | (a) | | 31.8 | |||||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||||||
| Total current assets |
2.2 | 423.9 | (2,183.5 | ) | 2,179.0 | 421.6 | ||||||||||||||||||||||
| Property, plant, and equipment, net |
| 89.0 | 18.8 | (a) | | 107.8 | ||||||||||||||||||||||
| Other assets: |
||||||||||||||||||||||||||||
| Cash and cash equivalents held in Trust |
750.1 | | | (750.1 | ) | (d) | | |||||||||||||||||||||
| Goodwill |
| 677.5 | 1,005.0 | (a) | | 1,682.5 | ||||||||||||||||||||||
| Intangible assets, net |
| 343.3 | 376.2 | (a) | | 719.5 | ||||||||||||||||||||||
| Other assets |
0.4 | 10.9 | (0.1 | ) | (a) | | 11.2 | |||||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||||||
| Total other assets |
750.5 | 1,031.7 | 1,381.1 | (750.1 | ) | 2,413.2 | ||||||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||||||
| Total assets |
$ | 752.7 | $ | 1,544.6 | $ | (783.6 | ) | $ | 1,428.9 | $ | 2,942.6 | |||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||||||
| LIABILITIES AND STOCKHOLDERS DEFICIT |
||||||||||||||||||||||||||||
| Current liabilities: |
||||||||||||||||||||||||||||
| Accounts payable |
$ | 1.4 | $ | 55.5 | $ | | $ | | $ | 56.9 | ||||||||||||||||||
| Deferred contract revenue |
| 49.9 | (16.2 | ) | (a) | | 33.7 | |||||||||||||||||||||
| Accrued offering costs |
0.3 | | | | 0.3 | |||||||||||||||||||||||
| Working capital note |
1.5 | | | | 1.5 | |||||||||||||||||||||||
| Warrant liability |
61.5 | | | | 61.5 | |||||||||||||||||||||||
| Notes payable to third-parties, current |
| 5.5 | (5.5 | ) | (a) (c) | 8.3 | (b) | 8.3 | ||||||||||||||||||||
| Accrued expenses and other current liabilities |
| 91.7 | | 7.4 | (e) | 99.1 | ||||||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||||||
| Total current liabilities |
64.7 | 202.6 | (21.7 | ) | 7.4 | 261.3 | ||||||||||||||||||||||
| Deferred underwriting discount |
26.3 | | | (26.3 | ) | (f) | | |||||||||||||||||||||
| Third-party notes payable, non-current, net |
| 885.5 | (885.5 | ) | (a) (c) | 821.7 | (b) | 803.3 | ||||||||||||||||||||
| (18.4 | ) | (e) | ||||||||||||||||||||||||||
| Related party notes payable, non-current, net |
| 1,202.5 | (1,202.5 | ) | (a) | | | |||||||||||||||||||||
| Deferred income taxes and other liabilities |
| 72.8 | 105.5 | (a) | | 178.3 | ||||||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||||||
| Total liabilities |
91.0 | 2,363.4 | (2,004.2 | ) | 792.7 | 1,242.9 | ||||||||||||||||||||||
| GSAH Class A common stock subject to redemption |
750.0 | | | (750.0 | ) | (g) | | |||||||||||||||||||||
| Stockholders deficit: |
||||||||||||||||||||||||||||
| A Ordinary shares |
| | | | (g) | | ||||||||||||||||||||||
| B Ordinary shares |
| 0.1 | (0.1 | ) | (a) | | (g) | | ||||||||||||||||||||
| Additional paid-in capital |
| 9.5 | (9.5 | ) | (a) | 900.0 | (b) | 1,714.3 | ||||||||||||||||||||
| 401.8 | (a) | 750.0 | (g) | |||||||||||||||||||||||||
| (a) (b) | ||||||||||||||||||||||||||||
| 125.0 | (b) | |||||||||||||||||||||||||||
| (366.1 | ) | (b) | ||||||||||||||||||||||||||
| (84.6 | ) | (h) | ||||||||||||||||||||||||||
| Receivable from Employees for purchase of Stock |
| (2.5 | ) | 2.5 | (a) | | | |||||||||||||||||||||
| Accumulated (deficit) earnings |
(88.3 | ) | (860.6 | ) | 860.6 | (a) | (10.9 | ) | (e) | (99.2 | ) | |||||||||||||||||
| Noncontrolling interests |
| 2.1 | (2.1 | ) | (a) | 84.6 | (h) | 84.6 | ||||||||||||||||||||
| Accumulated other comprehensive income (loss) |
| 32.6 | (32.6 | ) | (a) | | | |||||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||||||
| Total stockholders equity (deficit) |
(88.3 | ) | (818.8 | ) | 1,220.6 | 1,386.2 | 1,699.7 | |||||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||||||
| Total liabilities and stockholders equity |
$ | 752.7 | $ | 1,544.6 | $ | (783.6 | ) | $ | 1,428.9 | $ | 2,942.6 | |||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||||||
132
Table of Contents
GS ACQUISITION HOLDINGS CORP II
UNAUDITED PRO FORMA CONDENSED COMBINED STATEMENT OF OPERATIONS
FOR THE THREE MONTHS ENDED MARCH 31, 2021
| Historical Financials |
||||||||||||||||||||||||||||||||
| ($ in millions, except shares outstanding and per share amounts) |
GS Acquisition Holdings Corp II |
Mirion | Pro Forma Purchase Accounting Adjustments |
Notes | Mirion Pro Forma |
Pro Forma Financing Adjustments (Assuming No Redemptions) |
Notes | Pro Forma Combined (Assuming No Redemptions) |
||||||||||||||||||||||||
| Revenues: |
||||||||||||||||||||||||||||||||
| Product |
$ | | $ | 126.6 | $ | | $ | 126.6 | $ | | $ | 126.6 | ||||||||||||||||||||
| Service |
| 39.6 | | 39.6 | | 39.6 | ||||||||||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||||||||
| Total Revenues |
| 166.2 | | 166.2 | | 166.2 | ||||||||||||||||||||||||||
| Costs and expenses: |
||||||||||||||||||||||||||||||||
| Cost of revenuesProduct |
| 82.7 | (1.6 | ) | (i | ) | 81.1 | | 81.1 | |||||||||||||||||||||||
| Cost of revenuesService |
| 21.0 | 0.7 | 21.7 | | 21.7 | ||||||||||||||||||||||||||
| Selling, general and administrative |
0.7 | 60.4 | 8.3 | (i) | 68.7 | 4.5 | (j) | 73.9 | ||||||||||||||||||||||||
| Research and development |
| 11.0 | | 11.0 | | 11.0 | ||||||||||||||||||||||||||
| Other deductions, net |
| (4.2 | ) | | (4.2 | ) | | (4.2 | ) | |||||||||||||||||||||||
| Change in fair value of warrant liability |
(10.2 | ) | | | | | (10.2 | ) | ||||||||||||||||||||||||
| Dividend expense (income) |
| | | | | | ||||||||||||||||||||||||||
| Interest expense (income), net |
| 43.1 | (43.1 | ) | (i) | | 7.9 | (k) | 7.9 | |||||||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||||||||
| Income (loss) before income taxes |
9.5 | (47.8 | ) | 35.7 | (12.1 | ) | (12.4 | ) | (15.0 | ) | ||||||||||||||||||||||
| Income tax expense (benefit) |
(0.1 | ) | 23.6 | 8.9 | (i) | 32.5 | (3.1 | ) | (l) | 29.3 | ||||||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||||||||
| Net income (loss) |
$ | 9.6 | $ | (71.4 | ) | $ | 26.8 | $ | (44.6 | ) | $ | (9.3 | ) | (44.3 | ) | |||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||||||||||
| Less: Income (loss) attributable to noncontrolling interests |
(m) | (1.9 | ) | |||||||||||||||||||||||||||||
|
|
|
|||||||||||||||||||||||||||||||
| Net income (loss) attributable to controlling interests |
$ | (42.4 | ) | |||||||||||||||||||||||||||||
|
|
|
|||||||||||||||||||||||||||||||
| Historical |
||||||||||||||||||||||||||||||||
| Weighted average common shares outstanding of Class A common stock |
75,000,000 | |||||||||||||||||||||||||||||||
| Basic and diluted net income per share, Class A |
$ | 0.10 | ||||||||||||||||||||||||||||||
| Weighted average common shares outstanding of Class B common stock |
18,750,000 | |||||||||||||||||||||||||||||||
| Basic and diluted net income per share, Class B |
$ | 0.10 | ||||||||||||||||||||||||||||||
| Earnings per shareno redemption scenario |
||||||||||||||||||||||||||||||||
| Pro Forma weighted average common shares of Class A common stock outstandingbasic and diluted |
195,050,000 | |||||||||||||||||||||||||||||||
| Pro Forma net income (loss) per share basic and diluted available to common stockholders, Class A |
(n) | $ | (0.22 | ) | ||||||||||||||||||||||||||||
133
Table of Contents
GS ACQUISITION HOLDINGS CORP II
UNAUDITED PRO FORMA CONDENSED COMBINED STATEMENT OF OPERATIONS
FOR THE THREE MONTHS ENDED MARCH 31, 2021
| Historical Financials | ||||||||||||||||||||||||||||||||
| ($ in millions, except shares outstanding and per share amounts) |
GS Acquisition Holdings Corp II |
Mirion Historical |
Pro Forma Purchase Accounting Adjustments |
Notes | Mirion Pro Forma |
Pro Forma Financing Adjustments (Assuming Maximum Redemptions) |
Notes | Pro Forma Combined (Assuming Maximum Redemptions) |
||||||||||||||||||||||||
| Revenues: |
||||||||||||||||||||||||||||||||
| Product |
$ | | $ | 126.6 | $ | | $ | 126.6 | $ | | $ | 126.6 | ||||||||||||||||||||
| Service |
| 39.6 | | 39.6 | | 39.6 | ||||||||||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||||||||
| Total Revenues |
| 166.2 | | 166.2 | | 166.2 | ||||||||||||||||||||||||||
| Costs and expenses: |
||||||||||||||||||||||||||||||||
| Cost of revenuesProduct |
| 82.7 | (1.6 | ) | (i | ) | 81.1 | | 81.1 | |||||||||||||||||||||||
| Cost of revenuesService |
| 21.0 | (0.7 | ) | 21.7 | | 21.7 | |||||||||||||||||||||||||
| Selling, general and administrative |
0.7 | 60.4 | 8.3 | (i | ) | 68.7 | 4.5 | (j) | 73.9 | |||||||||||||||||||||||
| Research and development |
| 11.0 | | 11.0 | | 11.0 | ||||||||||||||||||||||||||
| Other deductions, net |
| (4.2 | ) | | (4.2 | ) | | (4.2 | ) | |||||||||||||||||||||||
| Change in fair value of warrant liability |
(10.2 | ) | | | | | (10.2 | ) | ||||||||||||||||||||||||
| Dividend expense (income) |
| | | | | | ||||||||||||||||||||||||||
| Interest expense (income), net |
| 43.1 | (43.1 | ) | (i | ) | | 8.4 | (k) | 8.4 | ||||||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||||||||
| Income (loss) before income taxes |
9.5 | (47.8 | ) | 35.7 | (12.1 | ) | (12.9 | ) | (15.5 | ) | ||||||||||||||||||||||
| Income tax expense (benefit) |
(0.1 | ) | 23.6 | 8.9 | (i | ) | 32.5 | (3.2 | ) | (l) | 29.2 | |||||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||||||||
| Net income (loss) |
$ | 9.6 | $ | (71.4 | ) | $ | 26.8 | $ | (44.6 | ) | $ | (9.7 | ) | (44.7 | ) | |||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||||||||||
| Less: Income (loss) attributable to noncontrolling interests |
(m) | (2.2 | ) | |||||||||||||||||||||||||||||
|
|
|
|||||||||||||||||||||||||||||||
| Net income (loss) attributable to controlling interests |
$ | (42.5 | ) | |||||||||||||||||||||||||||||
|
|
|
|||||||||||||||||||||||||||||||
| Historical |
||||||||||||||||||||||||||||||||
| Weighted average common shares outstanding of Class A common stock |
75,000,000 | |||||||||||||||||||||||||||||||
| Basic and diluted net income per share, Class A |
$ | 0.10 | ||||||||||||||||||||||||||||||
| Weighted average common shares outstanding of Class B common stock |
18,750,000 | |||||||||||||||||||||||||||||||
| Basic and diluted net income per share, Class B |
$ | 0.10 | ||||||||||||||||||||||||||||||
| Earnings per sharemaximum redemption scenario |
||||||||||||||||||||||||||||||||
| Pro Forma weighted average common shares of Class A common stock outstandingbasic and diluted |
170,940,000 | |||||||||||||||||||||||||||||||
| Pro Forma net income (loss) per share basic and diluted available to common stockholders, Class A |
(n) | $ | (0.25 | ) | ||||||||||||||||||||||||||||
134
Table of Contents
GS ACQUISITION HOLDINGS CORP II
UNAUDITED PRO FORMA CONDENSED COMBINED STATEMENT OF OPERATIONS
FOR THE YEAR ENDED DECEMBER 31, 2020
| Historical Financials | ||||||||||||||||||||||||||||||||||||||||||||
| ($ in millions, except shares outstanding and per share amounts) |
GS Acquisition Holdings Corp II |
Historical Mirion |
Historical Sun Nuclear (1/1/20 12/18/20) |
Pro Forma Sun Nuclear Purchase Accounting Adjustments |
Notes | Mirion Pro Forma |
Pro Forma Purchase Accounting Adjustments |
Notes | Pro Forma Financing Adjustments (Assuming No Redemptions) |
Notes | Pro Forma Combined (Assuming No Redemptions) |
|||||||||||||||||||||||||||||||||
| Revenues: |
||||||||||||||||||||||||||||||||||||||||||||
| Product |
$ | | $ | 377.1 | $ | 75.7 | $ | (7.3 | ) | (o) | $ | 445.5 | $ | | $ | | $ | 445.5 | ||||||||||||||||||||||||||
| Service |
| 139.2 | 22.4 | (9.5 | ) | (o) | 152.1 | (12.9 | ) | (i | ) | | 139.2 | |||||||||||||||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||||||||||||||||
| Total Revenues |
| 516.3 | 98.1 | (16.8 | ) | 597.6 | (12.9 | ) | | 584.7 | ||||||||||||||||||||||||||||||||||
| Costs and expenses: |
||||||||||||||||||||||||||||||||||||||||||||
| Cost of revenuesProduct |
| 230.8 | 21.9 | 7.7 | (o) | 260.4 | 29.5 | (i | ) | | 289.9 | |||||||||||||||||||||||||||||||||
| Cost of revenuesService |
| 70.5 | 11.0 | | 81.5 | 0.7 | (i | ) | | 82.2 | ||||||||||||||||||||||||||||||||||
| Selling, general and administrative |
2.5 | 162.6 | 33.8 | 15.7 | (o) | 212.1 | 36.0 | (i | ) | 34.1 | (j) | 307.4 | ||||||||||||||||||||||||||||||||
| 22.7 | (p) | |||||||||||||||||||||||||||||||||||||||||||
| Research and development |
| 17.9 | 14.7 | | 32.6 | | | 32.6 | ||||||||||||||||||||||||||||||||||||
| Other deductions, net |
| 16.4 | (0.5 | ) | | 15.9 | | | 15.9 | |||||||||||||||||||||||||||||||||||
| Change in fair value of warrant liability |
43.1 | | | | | | | 43.1 | ||||||||||||||||||||||||||||||||||||
| Dividend expense (income) |
(0.1 | ) | | | | | | 0.1 | (q) | | ||||||||||||||||||||||||||||||||||
| Interest expense (income), net |
| 154.2 | 0.1 | 21.3 | (o) | 175.6 | (158.1 | ) | (i | ) | 31.7 | (k) | 49.2 | |||||||||||||||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||||||||||||||||
| Income (loss) before income taxes |
(45.5 | ) | (136.1 | ) | 17.1 | (61.5 | ) | (180.5 | ) | 79.0 | (88.6 | ) | (235.6 | ) | ||||||||||||||||||||||||||||||
| Income tax expense (benefit) |
(0.3 | ) | (15.7 | ) | | (11.1 | ) | (o) | (26.8 | ) | 19.8 | (i | ) | (22.2 | ) | (l) | (29.5 | ) | ||||||||||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||||||||||||||||
| Net income (loss) |
$ | (45.2 | ) | $ | (120.4 | ) | $ | 17.1 | $ | (50.4 | ) | $ | (153.7 | ) | $ | 59.2 | $ | (66.4 | ) | (206.1 | ) | |||||||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||||||||||||||||||
| Less: Income (loss) attributable to noncontrolling interests |
(m) | (9.0 | ) | |||||||||||||||||||||||||||||||||||||||||
|
|
|
|||||||||||||||||||||||||||||||||||||||||||
| Net income (loss) attributable to controlling interests |
$ | (197.1 | ) | |||||||||||||||||||||||||||||||||||||||||
|
|
|
|||||||||||||||||||||||||||||||||||||||||||
| Historical |
||||||||||||||||||||||||||||||||||||||||||||
| Weighted average common shares outstanding of Class A common stock |
37,397,260 | |||||||||||||||||||||||||||||||||||||||||||
| Basic and diluted net income per share, Class A |
$ | (0.79 | ) | |||||||||||||||||||||||||||||||||||||||||
| Weighted average common shares outstanding of Class B common stock |
19,597,603 | |||||||||||||||||||||||||||||||||||||||||||
| Basic and diluted net income per share, Class B |
$ | (0.79 | ) | |||||||||||||||||||||||||||||||||||||||||
| Earnings per share no redemption scenario |
||||||||||||||||||||||||||||||||||||||||||||
| Pro Forma weighted average common shares of Class A common stock outstandingbasic and diluted |
195,050,000 | |||||||||||||||||||||||||||||||||||||||||||
| Pro Forma net income (loss) per share basic and diluted available to common stockholders, Class A |
(n | ) | $ | (1.01 | ) | |||||||||||||||||||||||||||||||||||||||
135
Table of Contents
GS ACQUISITION HOLDINGS CORP II
UNAUDITED PRO FORMA CONDENSED COMBINED STATEMENT OF OPERATIONS
FOR THE YEAR ENDED DECEMBER 31, 2020
| Historical Financials | ||||||||||||||||||||||||||||||||||||||||||||
| ($ in millions, except shares outstanding and per share amounts) |
GS Acquisition Holdings Corp II |
Historical Mirion |
Historical Sun Nuclear (1/1/20 12/18/20) |
Pro Forma Sun Nuclear Purchase Accounting Adjustments |
Notes | Mirion Pro Forma |
Pro Forma Purchase Accounting Adjustments |
Notes | Pro Forma Financing Adjustments (Assuming Maximum Redemptions) |
Notes | Pro Forma Combined (Assuming Maximum Redemptions) |
|||||||||||||||||||||||||||||||||
| Revenues: |
||||||||||||||||||||||||||||||||||||||||||||
| Product |
$ | | $ | 377.1 | $ | 75.7 | $ | (7.3 | ) | (o) | $ | 445.5 | $ | | $ | | $ | 445.5 | ||||||||||||||||||||||||||
| Service |
| 139.2 | 22.4 | (9.5 | ) | (o) | 152.1 | (12.9 | ) | (i) | | 139.2 | ||||||||||||||||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||||||||||||||||
| Total Revenues |
| 516.3 | 98.1 | (16.8 | ) | 597.6 | (12.9 | ) | | 584.7 | ||||||||||||||||||||||||||||||||||
| Costs and expenses: |
||||||||||||||||||||||||||||||||||||||||||||
| Cost of revenuesProduct |
| 230.8 | 21.9 | 7.7 | (o) | 260.4 | 29.5 | (i) | | 289.9 | ||||||||||||||||||||||||||||||||||
| Cost of revenuesService |
| 70.5 | 11.0 | | 81.5 | 0.7 | (i) | | 82.2 | |||||||||||||||||||||||||||||||||||
| Selling, general and administrative |
2.5 | 162.6 | 33.8 | 15.7 | (o) | 212.1 | 36.0 | (i) | 34.1 | (j) | 307.4 | |||||||||||||||||||||||||||||||||
| 22.7 | (p) | |||||||||||||||||||||||||||||||||||||||||||
| Research and development |
| 17.9 | 14.7 | | 32.6 | | | 32.6 | ||||||||||||||||||||||||||||||||||||
| Other deductions, net |
| 16.4 | (0.5 | ) | | 15.9 | | | 15.9 | |||||||||||||||||||||||||||||||||||
| Change in fair value of warrant liability |
43.1 | | | | | | | 43.1 | ||||||||||||||||||||||||||||||||||||
| Dividend expense (income) |
(0.1 | ) | | | | | | 0.1 | (q) | | ||||||||||||||||||||||||||||||||||
| Interest expense (income), net |
| 154.2 | 0.1 | 21.3 | (o) | 175.6 | (158.1 | ) | (i) | 33.8 | (k) | 51.3 | ||||||||||||||||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||||||||||||||||
| Income (loss) before income taxes |
(45.5 | ) | (136.1 | ) | 17.1 | (61.5 | ) | (180.5 | ) | 79.0 | (90.7 | ) | (237.7 | ) | ||||||||||||||||||||||||||||||
| Income tax expense (benefit) |
(0.3 | ) | (15.7 | ) | | (11.1 | ) | (o) | (26.8 | ) | 19.8 | (i) | (22.7 | ) | (l) | (30.0 | ) | |||||||||||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||||||||||||||||
| Net income (loss) |
$ | (45.2 | ) | $ | (120.4 | ) | $ | 17.1 | $ | (50.4 | ) | $ | (153.7 | ) | $ | 59.2 | $ | (68.0 | ) | (207.7 | ) | |||||||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||||||||||||||||||
| Less: Income (loss) attributable to noncontrolling interests |
(m) | (10.3 | ) | |||||||||||||||||||||||||||||||||||||||||
|
|
|
|||||||||||||||||||||||||||||||||||||||||||
| Net income (loss) attributable to controlling interests |
$ | (197.4 | ) | |||||||||||||||||||||||||||||||||||||||||
|
|
|
|||||||||||||||||||||||||||||||||||||||||||
| Historical |
||||||||||||||||||||||||||||||||||||||||||||
| Weighted average common shares outstanding of Class A common stock |
37,397,260 | |||||||||||||||||||||||||||||||||||||||||||
| Basic and diluted net income per share, Class A |
$ | (0.79 | ) | |||||||||||||||||||||||||||||||||||||||||
| Weighted average common shares outstanding of Class B common stock |
19,597,603 | |||||||||||||||||||||||||||||||||||||||||||
| Basic and diluted net income per share, Class B |
$ | (0.79 | ) | |||||||||||||||||||||||||||||||||||||||||
| Earnings per sharemaximum redemption scenario |
||||||||||||||||||||||||||||||||||||||||||||
| Pro Forma weighted average common shares of Class A common stock outstandingbasic and diluted |
170,940,000 | |||||||||||||||||||||||||||||||||||||||||||
| Pro Forma net income (loss) per share basic and diluted available to common stockholders, Class A |
(n) | $ | (1.15 | ) | ||||||||||||||||||||||||||||||||||||||||
136
Table of Contents
NOTE 1Description of the Business Combination
On June 17, 2021, the Company, Mirion Technologies (TopCo), Ltd., the Charterhouse Parties and the other Sellers entered into the Business Combination Agreement. The Business Combination Agreement provides for, among other things, the combination of Mirion with a subsidiary of the Company in accordance with the terms and subject to the conditions of the Business Combination Agreement as more fully described elsewhere in this proxy statement. Following the closing of the Business Combination, (a) the Company is expected to own between 95% and 96% of the equity interests in Mirion and (b) the Mirion Sellers (including certain members of management) are expected to hold between 15% and 18% of the outstanding shares of GSAH Class A common stock and all of the outstanding shares of the GSAH Class B common stock.
The aggregate consideration for the Business Combination will include a combination of cash and stock consideration as follows (in millions):
| Assuming no redemptions |
Assuming maximum redemptions |
|||||||
| Shares transferred at closing(1) |
39,000,000 | 39,000,000 | ||||||
| Value per share(2) |
$ | 10.00 | $ | 10.00 | ||||
|
|
|
|
|
|||||
| Total share consideration |
390.0 | 390.0 | ||||||
| Plus: cash transferred |
1,310.0 | 1,310.0 | ||||||
|
|
|
|
|
|||||
| Total cash and share consideration at closing |
$ | 1,700.0 | $ | 1,700.0 | ||||
|
|
|
|
|
|||||
| (1) | Includes both shares of GSAH Class A common stock (30.1 million to the Mirion Sellers excluding management) and shares of GSAH Class B common stock (8.9 million) to Mirion management stockholders) on a pro forma basis. Note that the allocation of shares between Mirion management and non-management stockholders will change over time and will depend on when the Business Combination closes, as payment-in-kind interest on the outstanding related party notes payable will change the amount of consideration transferred to each party. |
| (2) | The value of shares transferred at closing is assumed to be $10.00 per share. |
Before the Closing of the Business Combination Agreement, the Sellers will have the option to elect to have their equity consideration exchanged for either shares of GSAH Class A common stock of the Company or Paired Interests. The Company will own 100% of the voting shares (Class A) of IntermediateCo and greater than 80% of the non-voting Class B shares. As a result, the Company will recognize a NCI for the portion of IntermediateCo that is not attributable to the Company. We have assumed that of the existing Mirion stockholders, only Mirion management will elect to receive shares of GSAH Class B common stock (initially to defer recognition of the Business Combination for U.S. tax purposes).
Concurrently with the execution of the Business Combination Agreement, we entered into Subscription Agreements with the PIPE Investors pursuant to which the PIPE Investors have collectively subscribed for 90.0 million shares of the GSAH Class A common stock for an aggregate purchase price equal to $900 million, $200 million of which has been subscribed for by the Backstop Party unless it chooses to syndicate such subscription. The PIPE Investment will be consummated substantially concurrently with the closing of the Business Combination. We received a Debt Commitment Letter for a $830 million term facility (to refinance existing Mirion third-party debt) and a $90 million revolving credit facility (for future operational purposes and not used to finance the Business Combination). In addition, a Backstop Agreement has been executed such that up to an additional 12,500,000 shares will be purchased by the Backstop Party (a related party of the Company) to cover redemptions by public stockholders to the extent redemptions exceed the cash available from PIPE investors, the Trust Account, and new debt financing after the payment of Mirion third-party debt (subject to the Minimum Cash Condition).
137
Table of Contents
The $900.0 million of gross proceeds from the sale of the GSAH Class A common stock to the PIPE Investors is included in the Cash Consideration. The remainder of the Cash Consideration will be provided by the funds held in the trust account. The following summarizes the pro forma GSAH Class A and GSAH Class B Common Stock ownership (as a percentage of outstanding common stock) under each scenario:
| Pro Forma Class A Share Ownership in the Company(1) | ||||||||||||||||
| No Redemptions | Maximum Redemptions(2) | |||||||||||||||
| Number of Shares (millions) |
Percentage of Outstanding Shares |
Number of Shares (millions) |
Percentage of Outstanding Shares |
|||||||||||||
| PIPE Investors(3) |
90.0 | 44 | % | 90.0 | 50 | % | ||||||||||
| Public Stockholders |
75.0 | 37 | % | 38.4 | 21 | % | ||||||||||
| Mirion Sellers (excluding Mirion Management)(4) |
30.0 | 15 | % | 30.0 | 17 | % | ||||||||||
| GS Backstop(5) |
| 0 | % | 12.5 | 7 | % | ||||||||||
| Pro Forma Class B Share Ownership in the Company | ||||||||||||||||
| No Redemptions | Maximum Redemptions(2) | |||||||||||||||
| Number of Shares (millions) |
Percentage of Outstanding Shares |
Number of Shares (millions) |
Percentage of Outstanding Shares |
|||||||||||||
| Mirion Management(4) |
9.0 | 4 | % | 9.0 | 5 | % | ||||||||||
| (1) | Excludes 18,750,000 founder shares that convert from shares of GSAH Class B common stock to shares of GSAH Class A common stock upon the closing of the Business Combination and are subject to certain vesting and forfeiture conditions described below. In addition, the consideration to be paid to the Mirion Sellers in connection with the Business Combination is allocated first in respect of the PIK Notes and second in respect of the Existing Mirion Shares. The PIK Notes accrue payment-in-kind (PIK) interest daily at a rate of 11.5% annually and are contemplated to be sold at the Closing for a price equal to the full outstanding principal amount together with all accrued but unpaid interest up to but excluding the Closing Date. See Certain Relationships and Related Persons TransactionsMirions Related Person TransactionsShareholder Notes. Accordingly, as the amount of accrued and unpaid interest increases, the holders of the PIK Notes will receive more consideration as compared to the holders of the Existing Mirion Shares. This has the effect over time of increasing the amount of shares of GSAH Class A Common Stock to be issued to the Mirion Sellers (excluding members of Mirion management) and reducing the number of shares of GSAH Class B common stock that will be issued to Mirion management (there is no incremental dilution to Public Stockholders). For purposes of the ownership levels described herein, we have assumed an amount of principal and interest of the PIK Notes as if the Closing Date will be October 31, 2021, but for all other purposes have assumed the Closing Date will be March 31, 2021. |
If the actual facts are different from these assumptions, the above levels of ownership interest will be different.
| (2) | Assumes that 36.6 million public shares (being our estimate of the maximum number of public shares that could be redeemed in connection with the Business Combination in order to satisfy the related Minimum Cash Condition contained in the Business Combination Agreement) are redeemed in connection with the Business Combination. |
| (3) | Includes 20 million GSAH Class A shares subscribed for by Sponsor-related PIPE Investors. See Proposal No. 1Approval of the Business CombinationRelated AgreementsSubscription Agreements. |
| (4) | Mirion Sellers have the option of receiving either shares of GSAH Class A common stock or Paired Interests at closing. We have assumed that all Mirion Sellers with the exception of members of management will elect to receive GSAH Class A shares. |
| (5) | The maximum redemptions scenario assumes there is a Cash Shortfall such that GSAM Holdings will purchase 12,500,000 shares of GSAH Class A common stock under the Backstop Agreement. |
The founder shares are subject to vesting in three equal tranches, based on the volume-weighted average price of the Companys GSAH Class A common stock being greater than or equal to $12.00, $14.00 and $16.00, respectively (each, a Founder Share Vesting Event), per share for any 20 trading days in any 30 consecutive
138
Table of Contents
trading day period. Holders of the founder shares are entitled to vote such founder shares and receive dividends and other distributions with respect to such founder shares prior to vesting, but such dividends and other distributions with respect to unvested founder shares will be set aside by the Company and shall only be paid to the holders of the founder shares upon the vesting of such founder shares. The founder shares will be forfeited to the Company for no consideration if they fail to vest within five years of the closing of the Business Combination.
In conjunction with the Business Combination Agreement, the Sponsor issued 3,200,000 membership interests to Thomas Logan, the Chief Executive Officer of Mirion, 700,000 membership interests to Brian Schopfer, the Chief Financial Officer of Mirion, and 4,200,000 membership interests to Lawrence Kingsley, who is expected to be Chairman of the Board of New Mirion (collectively, the Profits Interests). The Profits Interests are intended to be treated as profits interests for U.S. income tax purposes, pursuant to which Messrs. Logan, Schopfer and Kingsley will have an indirect interest in the founder shares held by the Sponsor. The Profits Interests are subject to service and performance vesting conditions, including the occurrence of the Closing, and do not fully vest until all of the applicable conditions are satisfied. In addition, the Profits Interests are subject to certain forfeiture conditions. See Certain Relationships and Related Persons TransactionsMirions Related Person TransactionsProfits Interests. Accordingly, these awards have been treated as compensation and reflected accordingly in the pro forma adjustments to the unaudited pro forma condensed combined statements of operations.
In the event that more than 36.6 million public shares are redeemed and Mirion and the Charterhouse Parties waive the Minimum Cash Condition, Cash Consideration will be reduced by $10.00 for each additional redemption and additional shares of GSAH common stock will be issued to the Sellers at an assumed value of $10.00 per share in lieu thereof. The following summarizes the pro forma GSAH Class A and GSAH Class B Common Stock ownership (as a percentage of outstanding common stock) under a scenario where 100% of the outstanding public shares were redeemed:
| Pro Forma Class A Share Ownership in the Company(1) |
||||||||
| 100% Redemptions | ||||||||
| Number of Shares (millions) |
Percentage of Outstanding Shares |
|||||||
| PIPE Investors(2) |
90.0 | 50 | % | |||||
| Public Stockholders |
| 0 | % | |||||
| Mirion Sellers (excluding Mirion Management)(3) |
68.3 | 38 | % | |||||
| GS Backstop(4) |
12.5 | 7 | % | |||||
| Pro Forma Class B Share Ownership in the Company |
||||||||
| 100% Redemptions | ||||||||
| Number of Shares (millions) |
Percentage of Outstanding Shares |
|||||||
| Mirion Management(3) |
9.0 | 5 | % | |||||
| (1) | Excludes 18,750,000 founder shares that convert from shares of GSAH Class B common stock to shares of GSAH Class A common stock upon the closing of the Business Combination and are subject to certain vesting and forfeiture conditions described below. |
| (2) | Includes 20 million GSAH Class A shares subscribed for by Sponsor-related PIPE Investors. See Proposal No. 1Approval of the Business CombinationRelated AgreementsSubscription Agreements. |
| (3) | Mirion Sellers have the option of receiving either shares of GSAH Class A common stock or Paired Interests at closing. We have assumed that all Mirion Sellers with the exception of members of management will elect to receive GSAH Class A shares. |
| (4) | The 100% redemptions scenario assumes there is a Cash Shortfall such that GSAM Holdings will purchase 12,500,000 shares of GSAH Class A common stock under the Backstop Agreement. |
139
Table of Contents
The Combined Company may issue incentive awards under the Equity Incentive Plan to the extent these plans are approved by the Companys shareholders. However, as the number of awards and terms are not yet known, a pro forma adjustment has not been reflected.
NOTE 2Description of the Sun Acquisition
On December 18, 2020, Mirion purchased 100% of the issued and outstanding shares of Sun Nuclear Corporation, global leader in radiation oncology quality assurance, delivering patient safety solutions for diagnostic imaging and radiation therapy centers around the world. Mirion acquired SNC for approximately $276.9 million of gross consideration. The Sun Acquisition was funded by proceeds from a $225.0 million extension of Mirions 2019 Credit Facility and $70.0 million of related party notes payable.
The Sun Acquisition was consummated on December 18, 2020 with purchase accounting adjustments recorded as of and for the period ended December 31, 2020. Therefore, the unaudited pro forma condensed combined balance sheet as of March 31, 2021 and the unaudited pro forma condensed combined statements of operations for the three months ended March 31, 2021 do not include pro forma adjustments for the Sun Acquisition as it is fully reflected in the results of Mirion. Refer to Pro Forma Adjustments for adjustments made to the unaudited pro forma condensed combined statements of operations for the twelve months ended December 31, 2020.
NOTE 3Basis of the Pro Forma Presentation
The unaudited pro forma condensed combined financial information has been prepared using the acquisition method of accounting in accordance with ASC 805, with GSAH as the accounting acquirer, using the fair value concepts defined in the Financial Accounting Standards Boards ASC Topic 820, Fair Value Measurement, and based on the historical financial information of GSAH and Mirion.
ASC 820 defines fair value, establishes a framework for measuring fair value, and sets forth a fair value hierarchy that prioritizes and ranks the level of observability of inputs used to develop the fair value measurements. Fair value is defined in ASC 820 as the price that would be received to sell an asset or paid to transfer a liability in an orderly transaction between market participants at the measurement date. This is an exit price concept for the valuation of the asset or liability. In addition, market participants are assumed to be buyers and sellers in the principal (or the most advantageous) market for the asset or liability. Fair value measurements for a non-financial asset assume the highest and best use by these market participants. Many of these fair value measurements can be highly subjective, and it is possible that other professionals applying reasonable judgment to the same facts and circumstances, could develop and support a range of alternative estimated amounts.
The unaudited pro forma condensed combined financial statements were prepared based on certain currently available information and certain assumptions and methodologies that the Company believes are reasonable under the circumstances. The unaudited condensed pro forma adjustments, which are described in the accompanying notes, may be revised as additional information becomes available and is evaluated. We are in the process of reviewing the estimated fair values of all assets acquired and liabilities assumed by the Company, including, among other things, obtaining final third-party valuations of certain tangible and intangible assets, as well as the fair value of certain contracts and the determination of certain tax balances; thus, the allocation of the purchase price is preliminary and subject to revision. Therefore, it is likely that the actual adjustments will differ from the pro forma adjustments, and it is possible the difference may be material. The Company believes that its assumptions and methodologies provide a reasonable basis for presenting all of the significant effects of the Business Combination based on information available to management at this time and that the pro forma adjustments give appropriate effect to those assumptions and are properly applied in the unaudited pro forma condensed combined financial information.
The unaudited pro forma condensed combined balance sheet as of March 31, 2021 assumes that the business combination, equity financing, and debt financing occurred on March 31, 2021. The unaudited pro forma
140
Table of Contents
condensed combined statements of operations for the three months ended March 31, 2021 and for the year ended December 31, 2020 present pro forma effect to the business combination, equity financing, and debt financing as if they had been completed on January 1, 2020. These periods are presented on the basis of GSAH being considered the accounting acquirer.
The unaudited pro forma condensed combined statements of operations are not necessarily indicative of what the actual results of operations would have been had the Business Combination taken place on the date indicated, nor are they indicative of the future consolidated results of operations of the Combined Company. They should be read in conjunction with the historical consolidated financial statements and notes thereto of the Companies.
Based on its initial analysis of the Companys and Mirions accounting policies, management did not identify any differences that would have a material impact on the unaudited pro forma condensed combined financial information. As a result, the unaudited pro forma condensed combined financial information does not assume any differences in accounting policies that would impact the financial statements of the Combined Company.
Note 4Pro Forma Adjustments
The unaudited pro forma condensed combined financial information has been prepared to illustrate the effect of the Business Combination and has been prepared for informational purposes only. The unaudited pro forma condensed combined statements of operations are not necessarily indicative of what the actual results of operations would have been had the Business Combination taken place on the date indicated, nor is it indicative of the future consolidated results of operations of the Combined Company. The unaudited pro forma condensed combined financial information is based upon the historical consolidated financial statements of the Company and should be read in conjunction with its historical financial statements.
The historical consolidated financial statements have been adjusted in the unaudited pro forma condensed combined financial information to give effect to the accounting required under U.S. GAAP for the Business Combination.
There were no significant intercompany balances or transactions between the Companies as of the dates and for the periods of these unaudited pro forma condensed combined financial statements.
The pro forma condensed combined income tax expense (benefit) does not necessarily reflect the amounts that would have resulted had the Companies filed consolidated income tax returns during the periods presented.
The pro forma basic and diluted earnings per share amounts presented in the unaudited pro forma condensed combined statements of operations are based upon the number of the Companys shares outstanding, assuming the Business Combination occurred on January 1, 2020.
141
Table of Contents
Adjustments to Unaudited Pro Forma Condensed Combined Balance Sheet
| (a) | Reflects purchase accounting adjustments for Mirion, the repayment of historical debt balances, the elimination of Mirions historical equity (including the settlement of receivables from employees for purchase of common stock in the amount of $2.5 million), and the resulting impacts on additional paid-in capital (dollars in millions). |
|
As of March 31, 2021 |
Transaction Adjustments |
Estimated Fair Value |
||||||||||||||
| Purchase consideration: |
||||||||||||||||
| Cash consideration |
$ | 1,310.0 | ||||||||||||||
| Equity consideration paid to existing owners of Mirion |
390.0 | |||||||||||||||
| Cash repayment of debt |
908.5 | |||||||||||||||
| Cash paid for seller transaction expenses |
11.8 | |||||||||||||||
|
|
|
|||||||||||||||
| Total |
$ | 2,620.3 | ||||||||||||||
|
|
|
|||||||||||||||
| Net assets and liabilities acquired: |
||||||||||||||||
| Goodwill |
677.5 | 1,005.0 | (1 | ) | 1,682.5 | |||||||||||
| Amortizable intangibles |
343.3 | 376.2 | (2 | ) | 719.5 | |||||||||||
| Cash and cash equivalents |
86.8 | 1.0 | (3 | ) | 87.8 | |||||||||||
| Accounts receivable, net |
127.1 | | 127.1 | |||||||||||||
| Costs in excess of billings on uncompleted contracts |
59.7 | | 59.7 | |||||||||||||
| Inventories |
118.1 | 34.9 | (2 | ) | 153.0 | |||||||||||
| Other current assets |
32.2 | (0.9 | ) | (3 | ) | 31.3 | ||||||||||
| Property, plant and equipment, net |
89.0 | 18.8 | (2 | ) | 107.8 | |||||||||||
| Other non-current assets |
10.9 | (0.1 | ) | (3 | ) | 10.8 | ||||||||||
| Accounts payable |
(55.5 | ) | | (55.5 | ) | |||||||||||
| Deferred contract revenue |
(49.9 | ) | 16.2 | (2 | ) | (33.7 | ) | |||||||||
| Accrued expenses and other current liabilities |
(91.7 | ) | | (91.7 | ) | |||||||||||
| Deferred income taxes and other non-current liabilities |
(72.8 | ) | (105.5 | ) | (2 | ) | (178.3 | ) | ||||||||
|
|
|
|
|
|
|
|||||||||||
| Total |
$ | 1,274.7 | $ | 1,345.6 | $ | 2,620.3 | ||||||||||
|
|
|
|
|
|
|
|||||||||||
| (1) | Reflects the net adjustment to goodwill as a result of Mirion purchase accounting adjustments. |
| (2) | Reflects the change in fair value of certain intangible assets, inventory, property, plant and equipment, deferred revenue, and deferred tax liabilities recognized in the purchase price allocation. |
| (3) | Represents the reclassification of restricted cash balances from other current assets ($0.9 million) and other assets ($0.1 million) to cash and cash equivalents as these balances are no longer restricted upon a change of control. |
|
As of March 31, 2021 |
Transaction Adjustments |
Adjusted Balance |
||||||||||||||
| Write-off of historical equity and pay-off of debt, net of cash on hand: |
||||||||||||||||
| Third-party notes payable, current, net |
$ | (5.5 | ) | $ | 5.5 | (4 | ) | $ | | |||||||
| Third-party notes payable, non-current, net |
(885.5 | ) | 885.5 | (4 | ) | | ||||||||||
| Related party notes payable, non-current, net |
(1,202.5 | ) | 1,202.5 | (4 | ) | | ||||||||||
| Class B common stock |
0.1 | (0.1 | ) | (5 | ) | | ||||||||||
| Additional paid-in capital |
9.5 | (9.5 | ) | (5 | ) | | ||||||||||
| Receivable from Employees for purchase of Common Stock |
(2.5 | ) | 2.5 | (5 | ) | | ||||||||||
| Accumulated (deficit) earnings |
(860.6 | ) | 860.6 | (5 | ) | | ||||||||||
| Noncontrolling interests |
2.1 | (2.1 | ) | (5 | ) | | ||||||||||
| Accumulated other comprehensive income (loss) |
32.6 | (32.6 | ) | (5 | ) | | ||||||||||
|
|
|
|
|
|
|
|||||||||||
| Total |
$ | (2,912.3 | ) | $ | (2,912.3 | ) | $ | | ||||||||
|
|
|
|
|
|
|
|||||||||||
142
Table of Contents
| (4) | Reflects the repayment of historical debt balances, net of cash and cash equivalents. |
| (5) | Represents the elimination of Mirions historical equity. This includes the settlement of receivables from employees for purchase of common stock in the amount of $2.5 million, which are assumed to be fully settled at closing. |
|
As of March 31, 2021 |
||||||||
| Adjustment to Additional Paid-in Capital |
||||||||
| Equity consideration to sellers |
$ | 390.0 | (6 | ) | ||||
| Payment of seller transaction expenses |
11.8 | (7 | ) | |||||
|
|
|
|||||||
| Total |
$ | 401.8 | ||||||
|
|
|
|||||||
| (6) | Reflects the net adjustment to additional paid-in capital for equity consideration issued to the selling equity holders. |
| (7) | Reflects the adjustment for the consideration paid to the sellers for certain transaction expenses. This adjustment is offset with a corresponding decrease to additional paid-in capital under the financing pro forma column (see note (b) for further details).additional paid-in capital |
| (b) | Reflects the net adjustment to cash associated with the PIPE Investment and Business Combination (dollars in millions). |
| Assuming No Redemptions |
Assuming Maximum Redemptions |
|||||||||||
| Sources: |
||||||||||||
| Cash inflow from PIPE Investment |
$ | 900.0 | $ | 900.0 | (1) | |||||||
| Cash inflow from Companys Trust Account |
750.1 | 750.1 | (2) | |||||||||
| Cash inflow from new debt |
830.0 | 830.0 | (3) | |||||||||
| Cash inflow from balance sheet |
89.5 | 89.5 | (4) | |||||||||
| Cash inflow from GS backstop |
| 125.0 | (5) | |||||||||
|
|
|
|
|
|||||||||
| Total sources |
2,569.6 | 2,694.6 | ||||||||||
| Uses: |
||||||||||||
| Paydown of Mirion third-party debt |
908.5 | 908.5 | (6) | |||||||||
| Payment to selling equity holders |
1,310.0 | 1,310.0 | (7) | |||||||||
| Payment to redeeming Company stockholders |
| 366.1 | (8) | |||||||||
| Cash to balance sheet |
291.1 | 50.0 | (9) | |||||||||
| Payment of seller transaction expenses |
11.8 | 11.8 | (10) | |||||||||
| Payment of other transaction expenses |
48.2 | 48.2 | (11) | |||||||||
|
|
|
|
|
|||||||||
| Total uses |
2,569.6 | 2,694.6 | ||||||||||
| Net pro forma cash flow |
$ | | $ | | ||||||||
|
|
|
|
|
|||||||||
| (1) | Represents the issuance of 90 million shares of GSAH Class A common stock through the PIPE Investment at a par value of $0.0001 per share and an assumed fair value of $10.00 per share. |
| (2) | Reflects the reclassification of cash equivalents held in the trust account and to reflect that the cash equivalents are available to effectuate the Business Combination or to pay redeeming Company stockholders. |
| (3) | Represents the assumed issuance of $830.0 million of new debt as part of the transaction. We have assumed that a 1% minimum of the original principal amount will be due annually and have classified $8.3 million of the new debt as current and $821.7 million as noncurrent. |
143
Table of Contents
| (4) | Represents the cash held by GSAH (outside of the trust account) and Mirion (including restricted cash balances that are no longer restricted upon a change in control) as of March 31, 2021. |
| (5) | Represents the cash inflow from the exercise of the option under the Backstop Agreement to require the Backstop Party to purchase up to 12.5 million shares of Class A common stock in the maximum redemption scenario and no exercise of such option in the no redemption scenario. |
| (6) | Reflects the cash used to effect the debt refinancing under Mirions 2019 Credit Facility. |
| (7) | Reflects the net cash consideration paid to or on behalf of the Mirion Sellers under the terms of the Business Combination Agreement. This includes the repayment of outstanding notes payable to the Mirion Sellers. |
| (8) | Reflects the maximum payment that could be made to redeeming Company stockholders which would leave sufficient cash to satisfy the Minimum Cash Condition. The maximum amount of redemptions assumed is 36.6 million shares at a price of $10.00 per share. |
| (9) | Reflects the net amount of cash estimated to be retained on the balance sheet under each scenario. |
| (10) | Represents the payment of estimated seller transaction and transaction advisor fees and expenses. |
| (11) | Represents the payment of deferred underwriter discounts and commissions of $26.3 million and an estimated $21.9 million of other acquisition-related transaction and transaction advisor fees and expenses. Acquisition-related transaction expenses and related charges are not included as a component of consideration to be transferred but are reflected as a period cost. The unaudited pro forma condensed balance sheet reflects these costs as a reduction of cash with a corresponding adjustment to deferred underwriting fees and accumulated (deficit) earnings. See (e) for details of the adjustment to accumulated (deficit) earnings. |
| (c) | Represents funds from equity and debt issuances as part of the Business Combination used to repay Mirions 2019 Credit Facility and other third-party borrowings under the terms of the Business Combination Agreement (dollars in millions). |
| As of March 31, 2021 |
||||
| Third-party debt, reduction of principal |
$ | 908.5 | ||
| Accelerated amortization of debt issuance costs and discount |
(17.5 | ) | ||
|
|
|
|||
| Total reduction of third-party debt |
$ | 891.0 | ||
|
|
|
|||
| Third party debt: |
||||
| Current |
$ | 5.5 | ||
| Non-current |
885.5 | |||
|
|
|
|||
| Total |
$ | 891.0 | ||
|
|
|
|||
| (d) | Represents the release of restrictions on the investments and cash held in the Trust Account upon consummation of the Business Combination. |
| (e) | Represents the accrual for estimated transaction expenses exceeding payment of transaction expenses from debt and equity issuances in conjunction with the Business Combination, and the resulting impact on accumulated (deficit) earnings. |
144
Table of Contents
| Assuming No Redemptions |
Assuming Maximum Redemptions |
|||||||
| Total estimated transaction expenses |
$ | 70.0 | $ | 70.0 | ||||
| Less: Payment of transaction expenses from consideration |
(60.0 | ) | (60.0 | ) | ||||
| Less: Mirion transaction expenses recognized historically |
(2.6 | ) | (2.6 | ) | ||||
|
|
|
|
|
|||||
| Total accrual for transaction expenses |
$ | 7.4 | $ | 7.4 | ||||
|
|
|
|
|
|||||
| Total estimated transaction expenses |
$ | 70.0 | $ | 70.0 | ||||
| Less: Deferred underwriting discount |
(26.3 | ) | (26.3 | ) | ||||
| Less: Debt issuance costs on new debt |
(18.4 | ) | (18.4 | ) | ||||
| Less: Mirion transaction expenses recognized historically |
(2.6 | ) | (2.6 | ) | ||||
| Less: Transaction expenses paid on behalf of seller |
(11.8 | ) | (11.8 | ) | ||||
|
|
|
|
|
|||||
| Total adjustment to accumulated (deficit) earnings |
$ | 10.9 | $ | 10.9 | ||||
|
|
|
|
|
|||||
| (f) | Represents the $26.3 million payment of underwriting costs incurred as part of the Companys IPO and committed to be paid upon the consummation of a business combination. |
| (g) | Represents the reclassification of 75,000,000 shares of GSAH Class A common stock subject to possible redemption to permanent equity at a par value of $0.0001 per share. |
| (h) | Represents the recording of a noncontrolling interest (NCI) for the shares of GSAH Class B common stock issued to certain existing Mirion Sellers. At closing of the Business Combination Agreement, equity holders of Mirion will have the option to elect to have their rollover equity in Mirion exchanged for either shares of GSAH Class A common stock of the Company or Paired Interests. The Combined Company will own 100% of the voting shares (Class A) of IntermediateCo and greater than 80% of the non-voting shares of IntermediateCo Class B common stock. As a result, the Combined Company will recognize a NCI for the portion of IntermediateCo that is not attributable to the Combined Company. We have assumed that of the existing Mirion stockholders, only Mirion management will elect to receive Paired Interests. See the table below for the expected interest to be held by Mirion management in either the no redemption or maximum redemption scenarios. |
| Assuming No Redemptions |
Assuming Maximum Redemptions |
|||||||
| Noncontrolling interest: |
||||||||
| Percentage |
4.4 | % | 5.0 | % | ||||
| At March 31, 2021 (in millions) |
$ | 85.7 | $ | 84.6 | ||||
145
Table of Contents
Adjustments to Unaudited Pro Forma Condensed Statements of Operations
| (i) | Reflects the impact of Mirion purchase accounting adjustments on the operating results for the 3 months ending March 31, 2021 and for the year ending December 31, 2020. |
| For the 3 months ending March 31, 2021 |
(1) | (2) | (3) | (4) | (5) | (6) | Total | |||||||||||||||||||||
| Revenue |
||||||||||||||||||||||||||||
| Product |
$ | | $ | | $ | | $ | | $ | | $ | | $ | | ||||||||||||||
| Service |
| | | | | | | |||||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||
| Total revenues |
| | | | | | | |||||||||||||||||||||
| Costs and expenses |
||||||||||||||||||||||||||||
| Cost of revenuesProduct |
(1.7 | ) | 0.1 | | | | | (1.6 | ) | |||||||||||||||||||
| Cost of revenuesService |
0.7 | | | | | | 0.7 | |||||||||||||||||||||
| Selling, general and administrative |
9.3 | (1.0 | ) | | | | | 8.3 | ||||||||||||||||||||
| Research and development |
| | | | | | | |||||||||||||||||||||
| Other deductions, net |
| | | | | | | |||||||||||||||||||||
| Change in fair value of warrant liability |
| | | | | | | |||||||||||||||||||||
| Dividend (income) expense |
| | | | | | | |||||||||||||||||||||
| Interest expense, net |
| | | | (43.1 | ) | | (43.1 | ) | |||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||
| Income (loss) before income taxes |
(8.3 | ) | 0.9 | | | 43.1 | | 35.7 | ||||||||||||||||||||
| Income tax (benefit) expense |
| | | | | 8.9 | 8.9 | |||||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||
| Net income (loss) |
$ | (8.3 | ) | $ | 0.9 | $ | | $ | | $ | 43.1 | $ | (8.9 | ) | $ | 26.8 | ||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||
| For the 12 months ending December 31, 2020 |
(1) | (2) | (3) | (4) | (5) | (6) | Total | |||||||||||||||||||||
| Revenue |
||||||||||||||||||||||||||||
| Product |
$ | | $ | | $ | | $ | | $ | | $ | | $ | | ||||||||||||||
| Service |
| | (12.9 | ) | | | | (12.9 | ) | |||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||
| Total revenues |
| | (12.9 | ) | | | | (12.9 | ) | |||||||||||||||||||
| Costs and expenses |
||||||||||||||||||||||||||||
| Cost of revenuesProduct |
| (0.2 | ) | | 29.7 | | | 29.5 | ||||||||||||||||||||
| Cost of revenuesService |
0.9 | (0.2 | ) | | | | | 0.7 | ||||||||||||||||||||
| Selling, general and administrative |
36.1 | (0.1 | ) | | | | | 36.0 | ||||||||||||||||||||
| Research and development |
| | | | | | | |||||||||||||||||||||
| Other deductions, net |
| | | | | | | |||||||||||||||||||||
| Change in fair value of warrant liability |
| | | | | | | |||||||||||||||||||||
| Dividend (income) expense |
| | | | | | | |||||||||||||||||||||
| Interest expense, net |
| | | | (158.1 | ) | | (158.1 | ) | |||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||
| Income (loss) before income taxes |
(37.0 | ) | 0.5 | (12.9 | ) | (29.7 | ) | 158.1 | | 79.0 | ||||||||||||||||||
| Income tax (benefit) expense |
| | | | | 19.8 | 19.8 | |||||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||
| Net income (loss) |
$ | (37.0 | ) | $ | 0.5 | $ | (12.9 | ) | $ | (29.7 | ) | $ | 158.1 | $ | (19.8 | ) | $ | 59.2 | ||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||
| (1) | Reflects the change in amortization related to the change in fair value of certain Mirion intangible assets as if Mirion was acquired on January 1, 2020. |
| (2) | Reflects the change in depreciation related to the change in fair value of certain Mirion property, plant and equipment as if Mirion was acquired on January 1, 2020. |
| (3) | Reflects the impact of acquisition accounting adjustments related to reducing deferred revenue to its estimated fair value as of the acquisition date as if Mirion was acquired on January 1, 2020. |
| (4) | Reflects the increase to product cost of revenues from the acquisition accounting increase in fair value of inventory that is expected to be sold within one year of the acquisition date as if Mirion was acquired on |
146
Table of Contents
| January 1, 2020. The increase in fair value was determined based on the estimated selling price of the inventory, less the remaining manufacturing and selling costs and a normal profit margin on those manufacturing and selling efforts. These expenses will not affect the Companys statement of operations beyond 12 months after the acquisition date. |
| (5) | Reflects the elimination of interest expense on debt assumed settled as of January 1, 2020 ($175.6 million), net of $17.5 million accelerated amortization of debt issuance costs and discount on historical debt. |
| (6) | Represents the income tax effect of the above pro forma adjustments based on an estimated blended statutory rate of 25%. |
| (j) | Reflects share-based compensation expense estimated for 8.1 million Profits Interests issued to Messrs. Logan, Schopfer and Kingsley. The Profits Interests are subject to service vesting conditions (50% of the Profits Interests granted to each of Messrs. Logan and Schopfer service-vest on each of the second and third anniversaries of the Closing, and fifty percent (50%) of the Profits Interests granted to Mr. Kingsley service-vest on each of the first and second anniversaries of the Closing) and performance vesting conditions (the share price must meet or exceed certain established thresholds for 20 out of 30 trading days before the fifth anniversary of the closing date). Of the Profits Interests, 3.2 million have a threshold price of $12 per share, 2.0 million have a threshold price of $14 per share, and 3.0 million have a threshold price of $16 per share. Based upon a valuation model using Monte Carlo simulations, a fair value per share of $8.03, $6.83, and $5.74 has been estimated for the $12, $14, and $16 per share performance vesting conditions, respectively. The expense will be recognized on a straight-line basis over the related service period. As the Profits Interests include as a vesting condition the completion of the Business Combination, the expense that accumulates prior to the Business Combination will not be recorded until it occurs. |
| (k) | Represents the interest expense and amortization of debt issuance costs related to new debt issued in the amount of $830.0 million under the no redemption scenario assuming an indicative 3.20% interest rate (LIBOR + 3.00%) and $830.0 million under the maximum redemption scenario assuming an indicative 3.45% interest rate (LIBOR + 3.25%). The higher interest rate in the maximum redemption scenario has been assumed given the higher leverage of the Combined Company under that scenario. Debt issuance costs have been estimated to be approximately $18.4 million; a 1% change in the debt issuance costs would impact the total debt issuance costs by $9 million. Note that actual interest rates and debt issuance costs, including any upfront fees or OID, will vary depending upon a variety of factors including the timing of the debt financing marketing and market conditions existing at such time. The following table details the pro forma impact of a net increase/decrease in the interest rate of 1/8th of a percentage point and the pro forma impact of a 1% increase/decrease in the debt issuance costs as a percentage of debt (dollars in millions). |
| Assuming No Redemptions | Assuming Maximum Redemptions | |||||||||||||||
|
Three months ended March 31, 2021 |
Year ended December 31, 2020 |
Three months ended March 31, 2021 |
Year ended December 31, 2020 |
|||||||||||||
| Increase in interest expense due to a rate increase of 1/8th of a percentage point |
0.3 | 1.0 | 0.3 | 1.0 | ||||||||||||
| Decrease in interest expense due to a rate decrease of 1/8th of a percentage point |
(0.3 | ) | (1.0 | ) | (0.3 | ) | (1.0 | ) | ||||||||
| Increase in interest expense due to an increase in the percentage for debt issuance costs of 1% |
0.6 | 2.6 | 0.6 | 2.6 | ||||||||||||
| Decrease in interest expense due to an decrease in the percentage for debt issuance costs of 1% |
(0.6 | ) | (2.6 | ) | (0.6 | ) | (2.6 | ) | ||||||||
| (l) | Reflects adjustments to income tax expense due to the tax impact on the pro forma adjustments at the estimated statutory rate of 25%. |
| (m) | Represents the recording of a non-controlling interest. See (h) above for further details. |
| (n) | Pro forma earnings per share (amounts rounded and in millions except share and per share)(1): |
147
Table of Contents
| Assuming No Redemptions | Assuming Maximum Redemptions | |||||||||||||||
|
Three months ended March 31, 2021 |
Year ended December 31, 2020 |
Three months ended March 31, 2021 |
Year ended December 31, 2020 |
|||||||||||||
| Pro forma net income (loss) available to common stockholders |
$ | (42.4 | ) | $ | (197.1 | ) | $ | (42.5 | ) | $ | (197.4 | ) | ||||
|
|
|
|
|
|
|
|
|
|||||||||
| Shares of Class A Common Stock: |
||||||||||||||||
| Class A common stock outstanding |
75,000,000 | 75,000,000 | 75,000,000 | 75,000,000 | ||||||||||||
| Class A common stock issued to Mirion Sellers, excluding Mirion management |
30,050,000 | 30,050,000 | 30,050,000 | 30,050,000 | ||||||||||||
| Class A common stock issued to PIPE Investors |
90,000,000 | 90,000,000 | 90,000,000 | 90,000,000 | ||||||||||||
| Class A common stock issued to Backstop Party |
| | 12,500,000 | 12,500,000 | ||||||||||||
| Class A redemptions |
| | (36,610,000 | ) | (36,610,000 | ) | ||||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Pro forma weighted average number shares outstanding, Class A |
195,050,000 | 195,050,000 | 170,940,000 | 170,940,000 | ||||||||||||
| Pro forma net income (loss) per share of common stockbasic and diluted, Class A(2)(3) |
$ | (0.22 | ) | $ | (1.01 | ) | $ | (0.25 | ) | $ | (1.15 | ) | ||||
| (1) | Class B common stock of the Combined Company will have voting rights but no economic interest in the Combined Company and therefore have been excluded from the calculation of basic earnings per share. |
| (2) | At March 31, 2021, the Company had outstanding warrants to purchase up to 27,250,000 shares of Class A common stock. One whole warrant entitles the holder thereof to purchase one share of GSAH Class A common stock at a price of $11.50 per share. The Companys warrants are anti-dilutive due to pro forma net losses and have been excluded from the diluted number of the Combined Companys Shares outstanding. |
| (3) | Excludes 18,750,000 founder shares that are subject to forfeiture if a Founder Share Vesting Event does not occur within five years of the closing of the Business Combination. The founder shares are subject to certain Founder Share Vesting Events. Holders of the founder shares are entitled to vote such founder shares and receive dividends and other distributions with respect to such founder shares prior to vesting, but such dividends and other distributions with respect to unvested founder Shares will be set aside by the Combined Company and shall only be paid to the holders of the founder shares upon the vesting of such founder shares. |
As the holders of the founder shares are not entitled to participate in earnings unless the vesting conditions are met, the founders shares have been excluded from the calculation of basic earnings per share. The founders shares are also excluded from the calculation of diluted earnings per share because their inclusion would be anti-dilutive.
148
Table of Contents
| (o) | Reflects the impact of Sun purchase accounting adjustments on the operating results for the year ending December 31, 2020 assuming the acquisition occurred on January 1, 2020 rather than the date acquired by Mirion (December 18, 2020). |
| For the 12 months ending December 31, 2020 |
(1) | (2) | (3) | (4) | (5) | (6) | (7) | (8) | Total | |||||||||||||||||||||||||||
| Revenue |
||||||||||||||||||||||||||||||||||||
| Product |
$ | | $ | | $ | (4.7 | ) | $ | | $ | | $ | (2.6 | ) | $ | | $ | | $ | (7.3 | ) | |||||||||||||||
| Service |
| | (9.5 | ) | | | | | | (9.5 | ) | |||||||||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||||||
| Total revenues |
| | (14.2 | ) | | | (2.6 | ) | | | (16.8 | ) | ||||||||||||||||||||||||
| Costs and expenses |
||||||||||||||||||||||||||||||||||||
| Cost of revenues Product |
4.1 | (0.1 | ) | | 4.7 | | (1.0 | ) | | | 7.7 | |||||||||||||||||||||||||
| Cost of revenues Service |
| | | | | | | | | |||||||||||||||||||||||||||
| Selling, general and administrative |
15.7 | (0.5 | ) | | | 1.6 | (1.1 | ) | | | 15.7 | |||||||||||||||||||||||||
| Research and development |
| | | | | | | | | |||||||||||||||||||||||||||
| Other deductions, net |
| | | | | | | | | |||||||||||||||||||||||||||
| Change in fair value of warrant liability |
| | | | | | | | | |||||||||||||||||||||||||||
| Dividend (income) expense |
| | | | | | | | | |||||||||||||||||||||||||||
| Interest expense, net |
| | | | | | 21.3 | | 21.3 | |||||||||||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||||||
| Income (loss) before income taxes |
(19.8 | ) | 0.6 | (14.2 | ) | (4.7 | ) | (1.6 | ) | (0.5 | ) | (21.3 | ) | | (61.5 | ) | ||||||||||||||||||||
| Income tax (benefit) expense |
| | | | | | | (11.1 | ) | (11.1 | ) | |||||||||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||||||
| Net income (loss) |
$ | (19.8 | ) | $ | 0.6 | $ | (14.2 | ) | $ | (4.7 | ) | $ | (1.6 | ) | $ | (0.5 | ) | $ | (21.3 | ) | $ | 11.1 | $ | (50.4 | ) | |||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||||||
| (1) | Reflects the incremental amortization of related to the additional intangible assets recognized in the purchase price allocation as well as the increase in fair value of certain intangible assets. |
| (2) | Reflects the elimination of depreciation expense of related to the reduction in fair value of Sun Nuclears property and equipment as of the acquisition date. |
| (3) | Reflects the reduction in revenue related to the reduction in the fair value of Sun Nuclears deferred revenue as of the acquisition date. The reduction in revenue represents the difference between prepayments related to the extended maintenance and software arrangements and the fair value of the assumed performance obligations. |
| (4) | Reflects the increase to product cost of revenues from the increase in fair value of Sun Nuclears inventory that is expected to be sold within one year of the acquisition date. The increase in fair value was determined based on the estimated selling price of the inventory, less the remaining manufacturing and selling costs and a normal profit margin on those manufacturing and selling efforts. These expenses will not affect the Combined Companys statement of operations beyond 12 months after the acquisition date. |
| (5) | Reflects the increase in rent expense from the de-consolidation of affiliates that will no longer qualify for consolidation as a result of the Sun Acquisition. |
| (6) | Reflects the elimination of the Radon business distributed to the Sun Nuclear shareholders prior to the Sun Acquisition. |
| (7) | Reflects the incremental interest expense of $21.3 million, including the amortization of related debt issuance costs, related to financing the Sun Acquisition with a draw of $225 million on the 2019 Credit Facility and increase of $70 million in shareholder loans. The interest rate on the 2019 Credit Facility is based upon the lessor of LIBOR or 0% plus 4%. An increase in this interest rate of 1/8th of a percentage point would result in $0.3 million in additional interest expense; a decrease of 1/8th of a percentage point would result in $0.2 million less interest expense. |
| (8) | Represents the income tax effect of the above pro forma adjustments for the year ended December 31, 2020 based on the U.S. statutory income tax rate of 25%. |
149
Table of Contents
| (p) | Represents the recognition of additional transaction expenses estimated to be incurred in conjunction with the Business Combination. These costs will not affect the Combined Companys statement of operations beyond 12 months after the acquisition date. |
| Assuming No Redemptions |
Assuming Maximum Redemptions |
|||||||
| Total estimated transaction expenses |
$ | 70.0 | $ | 70.0 | ||||
| Less: |
||||||||
| Deferred underwriting discount |
(26.3 | ) | (26.3 | ) | ||||
| Transaction expenses recognized historically by Mirion |
(2.6 | ) | (2.6 | ) | ||||
| Debt issuance costs on new debt |
(18.4 | ) | (18.4 | ) | ||||
|
|
|
|
|
|||||
| Total additional expense to recognize |
$ | 22.7 | $ | 22.7 | ||||
|
|
|
|
|
|||||
| (q) | To eliminate the Companys dividend income on the trust account. |
150
Table of Contents
SPECIAL MEETING OF GSAH STOCKHOLDERS
This proxy statement/prospectus is being provided to GSAHs stockholders as part of a solicitation of proxies by the Board for use at the Special Meeting of Stockholders to be held on [●], 2021, and at any adjournment or postponement thereof. This proxy statement/prospectus contains important information regarding the Special Meeting, the proposals on which you are being asked to vote and information you may find useful in determining how to vote and voting procedures.
This proxy statement/prospectus is being first mailed on or about [●], 2021 to all stockholders of record of the Company as of [●], 2021, the record date for the Special Meeting. Stockholders of record who owned our common stock at the close of business on the record date are entitled to receive notice of, attend and vote at, the Special Meeting. On the record date, there were [●] shares of our common stock outstanding.
Date, Time and Place of Special Meeting
The Special Meeting will be held via live webcast on [●], 2021 at [●] a.m. [Eastern Time], or at such other time, on such other date and at such other place to which the meeting may be adjourned or postponed. The special meeting can be accessed by visiting [●], where you will be able to listen to the meeting live and vote during the meeting.
Purpose of the GSAH Special Meeting
At the Special Meeting, Company stockholders will vote on the following proposals:
| | Business Combination Proposal To approve and adopt the Business Combination Agreement and approve the Business Combination; |
| | NYSE ProposalTo approve, for purposes of complying with applicable listing rules of the NYSE, (a) the issuance of more than 20% of the Companys outstanding Class A common stock in connection with the Business Combination, including the PIPE Investment, and (b) the issuance of shares of the GSAH Class A common stock and the GSAH Class B common stock to a Related Party (as defined in Section 312.03 of the NYSEs Listed Company Manual) in connection with the Business Combination; |
| | Charter ProposalTo consider and act upon a proposal relating to adopting the New Mirion Charter in the form attached hereto as Annex B, which, if approved, would take effect upon the closing of the Business Combination; |
| | Governance ProposalTo consider and vote upon a proposal to approve, on a non-binding advisory basis, certain governance provisions in New Mirion Charter, presented separately in accordance with SEC requirements; |
| | Director Election ProposalTo elect nine directors to serve, effective upon the closing of the Business Combination, on our Board of Directors until the 2022 annual meeting of stockholders, and until their respective successors are duly elected and qualified, or until their earlier resignation, removal or death; |
| | Incentive Plan Proposal To approve Incentive Plan, including the authorization of the initial share reserve under the Incentive Plan; and |
| | Adjournment ProposalTo approve the adjournment of the Special Meeting to a later date or dates, if necessary, to permit further solicitation and vote of proxies in the event that there are insufficient votes for, or for any other reason in connection with, the approval of one or more of the other proposals at the Special Meeting. |
151
Table of Contents
Recommendation to GSAH Stockholders
Our Board believes that the Business Combination Proposal and the other proposals to be presented at the Special Meeting are in the best interest of GSAHs stockholders and unanimously recommends that its stockholders vote FOR the Business Combination Proposal, FOR the NYSE Proposal, FOR the Charter Proposal, FOR the Governance Proposal, FOR each nominee in the Director Election Proposal, FOR the Incentive Plan Proposal and FOR the Adjournment Proposal, in each case, if presented to the Special Meeting.
The existence of financial and personal interests of one or more of GSAHs directors may result in a conflict of interest on the part of such director(s) between what he or they may believe is in the best interests of GSAH and its stockholders and what he or they may believe is best for himself or themselves in determining to recommend that stockholders vote for the proposals. See Proposal No. 1Approval of the Business CombinationInterests of Goldman Sachs Parties and Certain Other Persons in the Business Combination for a further discussion of these considerations.
Vote of the Sponsor and GS Employee Participation
The Sponsor and GS Employee Participation have each agreed to, among other things, vote in favor of the Business Combination Proposal and the other proposals described herein to be presented at the Special Meeting. As of the date of this proxy statement/prospectus, the Initial Stockholders own approximately 20% of the outstanding shares of our common stock.
The Sponsor and GS Employee Participation have also agreed to waive their redemption rights in connection with the consummation of the Business Combination with respect to any shares held by them. The founder shares will be excluded from the pro rata calculation used to determine the per-share redemption price. However, the Sponsor and any other holder of GSAH Class B common stock will be entitled to liquidating distributions from the trust account with respect to any public shares they hold if we fail to complete our initial business combination within the allotted time period.
Voting Power; Record Date
GSAH stockholders will be entitled to vote or direct votes to be cast at the Special Meeting if they owned shares of Common Stock at the close of business on [●], 2021 which is the record date for the Special
Meeting. Stockholders are entitled to one vote on each proposal presented at the Special Meeting for each share of our common stock held of record as of on [●], 2021 the record date for the Special Meeting. If your shares are held in street name or are in a margin or similar account, you should contact your broker, bank or other nominee to ensure that votes related to the shares you beneficially own are properly counted. GSAHs warrants do not have voting rights with respect to the proposals to be presented at the Special Meeting. As of the close of business on the record date, there were [●] outstanding shares of our common stock.
Quorum and Required Vote for Proposals for the Special Meeting
A quorum of Company stockholders is necessary to hold a valid meeting. A quorum will be present at the Special Meeting if a majority of the outstanding shares of our common stock entitled to vote as of the record date at the Special Meeting are present at the special meeting by attendance via the virtual meeting website or represented by proxy. Abstentions will be counted as present for the purpose of determining a quorum but broker non-votes will not. Our Initial Stockholders, who currently own approximately 20% of our outstanding shares of Common Stock, will count towards this quorum.
The following votes are required for each proposal at the Special Meeting:
| | Business Combination Proposal: The approval of the Business Combination Proposal requires the affirmative vote of a majority of the votes cast by holders of our outstanding shares of |
152
Table of Contents
| common stock represented at the special meeting by attendance via the virtual meeting website or by proxy and entitled to vote thereon at the Special Meeting. Generally, only votes FOR or AGAINST are considered to be votes cast. Accordingly, a Company stockholders failure to vote by proxy or to vote during the Special Meeting, as well as an abstention from voting and a broker non-vote with regard to the Business Combination Proposal will have no effect on the Business Combination Proposal. Abstentions will be counted in connection with the determination of whether a valid quorum is established but will have no effect on the Business Combination Proposal. |
| | NYSE Proposal: The approval of the NYSE Proposal requires the affirmative vote of holders of a majority of the votes cast by holders of our outstanding shares of common stock represented at the special meeting by attendance via the virtual meeting website or by proxy and entitled to vote thereon at the Special Meeting. A Company stockholders failure to vote by proxy or to vote during the Special Meeting and a broker non-vote with regard to the NYSE Proposal will have no effect on the NYSE Proposal. The NYSE considers abstentions to be votes cast and included in the number of shares of which a majority is required to vote in favor. Accordingly, abstentions will have the same effect as a vote AGAINST the NYSE Proposal. |
| | Charter Proposal: The approval of the Charter Proposal requires the affirmative vote of holders of a majority of our outstanding shares of common stock entitled to vote thereon at the Special Meeting. Accordingly, a Company stockholders failure to vote by proxy or to vote during the Special Meeting, as well as an abstention from voting and a broker non-vote with regard to any of the Charter Proposal will have the same effect as a vote AGAINST the Charter Proposal. |
| | Governance Proposal: The approval of the Governance Proposal will require the affirmative vote of a majority of the votes cast by holders of our outstanding shares of common stock represented at the special meeting by attendance via the virtual meeting website or by proxy and entitled to vote at the special meeting. Accordingly, a Company stockholders failure to vote by proxy or to vote during the Special Meeting, as well as an abstention from voting and a broker non-vote with regard to the Governance Proposal, will have no effect on the Governance Proposal. Abstentions will be counted in connection with the determination of whether a valid quorum is established but will have no effect on the Governance Proposal. |
| | Director Election Proposal: Directors are elected by a plurality of all of the votes cast by holders of shares of common stock represented at the special meeting by attendance via the virtual meeting website or by proxy and entitled to vote thereon at the Special Meeting. This means that the nine director nominees who receive the most affirmative votes will be elected. Stockholders may not cumulate their votes with respect to the election of directors. Assuming a valid quorum is established, abstentions and broker non-votes will have no effect on the election of directors. |
| | Incentive Plan Proposal: The approval of the Incentive Plan Proposal requires the affirmative vote of a majority of the votes cast by holders of our outstanding shares of common stock represented holders of our outstanding shares of common stock represented at the Special Meeting by attendance via the virtual meeting website or by proxy and entitled to vote at the Special Meeting or by proxy and entitled to vote thereon at the Special Meeting. A Company stockholders failure to vote by proxy or to vote during the Special Meeting and a broker non-vote with regard to the Incentive Plan Proposal will have no effect on the Incentive Plan Proposal. The NYSE considers abstentions to be votes cast and included in the number of shares of which a majority is required to vote in favor. Accordingly, abstentions will have the same effect as a vote AGAINST the Incentive Plan Proposal. |
| | Adjournment Proposal: The approval of the Adjournment Proposal requires the affirmative vote of a majority of the votes cast by holders of our outstanding shares of common stock represented holders of our outstanding shares of common stock represented at the Special Meeting by attendance via the virtual meeting website or by proxy and entitled to vote at the Special Meeting or by proxy and entitled to vote thereon at the Special Meeting. Generally, only votes FOR or |
153
Table of Contents
| AGAINST are considered to be votes cast. Accordingly, a Company stockholders failure to vote by proxy or to vote during the Special Meeting, as well as an abstention from voting and a broker non-vote will have no effect on the Adjournment Proposal. Abstentions will be counted in connection with the determination of whether a valid quorum is established but will have no effect on the Adjournment Proposal. |
The Business Combination is conditioned on the approval of each of the Condition Precedent Proposals at the Special Meeting. Each of the Condition Precedent Proposals is cross-conditioned on the approval of each other. The Adjournment Proposal is not conditioned upon the approval of any other proposal. Each of these proposals is more fully described in this proxy statement/prospectus, which each stockholder is encouraged to read carefully and in its entirety.
It is important for you to note that in the event that the Condition Precedent Proposals do not receive the requisite vote for approval, we will not consummate the Business Combination. If we do not consummate the Business Combination and fail to complete an initial business combination by July 2, 2022 (or if such date is extended at a duly called meeting of our stockholders, such later date), we will be required to dissolve and liquidate our trust account by returning the then remaining funds in such account to our public stockholders.
Abstentions and Broker Non-Votes
Abstentions are considered present for the purposes of establishing a quorum.
In general, if your shares are held in street name and you do not instruct your broker, bank or other nominee on a timely basis on how to vote your shares, your broker, bank or other nominee, in its sole discretion, may either leave your shares unvoted or vote your shares on routine matters, but not on any non-routine matters. We believe all of the proposals presented to our stockholders will be considered non-discretionary and therefore your broker, bank or other nominee cannot vote your shares without your instruction.
In connection with (i) the Business Combination Proposal and the Adjournment Proposal, abstentions and broker non-votes will have no effect, (ii) the NYSE Proposal and the Incentive Plan Proposal, abstentions will be counted as a vote cast at the Special Meeting and will have the same effect as a vote AGAINST the proposal but broker non-votes will have no effect and (iii) the Charter Proposal, abstentions and broker non-votes will have the same effect as voting AGAINST the proposal.
Voting Your SharesStockholders of Record
If you are a holder of record of shares of our common stock on the record date for the Special Meeting, you may vote during the Special Meeting via the virtual meeting website or by submitting a proxy for the Special Meeting. Our stockholders are entitled to one vote on each proposal presented at the Special Meeting for each share of our common stock held of record as of the record date for the Special Meeting. Your one or more proxy cards show the number of shares of our Common Stock that you own.
Voting by Mail. You may submit your proxy by completing, signing, dating and returning the enclosed proxy card in the accompanying pre-addressed postage-paid envelope. By signing the proxy card and returning it, you are authorizing the individuals named on the proxy card to vote your shares at the Special Meeting in the manner you indicate. We encourage you to complete, sign, date and return the proxy card even if you plan to attend the Special Meeting so that your shares will be voted if you are unable to attend the Special Meeting. If you receive more than one proxy card, it is an indication that your shares are held in multiple accounts. Please complete, sign, date and return all proxy cards to ensure that all of your shares are voted. If you sign and return your proxy card without indicating how you wish to vote, your proxy will be voted as recommended by our Board. Our Board recommends voting FOR the Business Combination Proposal, FOR the NYSE Proposal, FOR the Charter Proposal, FOR the Governance Proposal, FOR each nominee in the Director Election Proposal, FOR the
154
Table of Contents
Incentive Plan Proposal and FOR the Adjournment Proposal. Votes submitted by mail must be received by [●] a.m. [Eastern Time] on [●], 2021.
Voting in Person at the Meeting. If you attend the Special Meeting and plan to vote during the meeting via the virtual meeting website, you may do so by following the instructions on your proxy card. If your shares are registered directly in your name, you are considered the stockholder of record and you have the right to vote during the Special Meeting.
Voting Your SharesBeneficial Owners
If your shares are held in a stock brokerage account or by a bank or other nominee, you are considered the beneficial holder of the shares held for you in what is known as street name. If this is the case, this proxy statement/prospectus may have been forwarded to you by your broker, bank or other nominee, or its agent. The broker, bank or other nominee holding your account is considered to be the stockholder of record for purposes of voting at the Special Meeting. As the beneficial holder, you have the right to direct your broker, bank or other nominee as to how to vote your shares and you should instruct your broker, bank or other nominee to vote your shares in accordance with directions you provide. As a beneficial owner, if you wish to vote at the Special Meeting, you will need to bring to the Special Meeting a legal proxy from your broker, bank or other nominee authorizing you to vote those shares. That is the only way we can be sure that the broker, bank or other nominee has not already voted your shares of Common Stock. See Attending the Special Meeting below for more details.
Attending the Special Meeting
Only holders of record of shares of our common stock on the record date for the Special Meeting or their legal proxy holders may attend the Special Meeting. To be admitted to the Special Meeting, you will need a form of photo identification and valid proof of ownership of our common stock or a valid legal proxy. If you have a valid legal proxy from a stockholder of record, you must bring a form of photo identification and the legal proxy to the Special Meeting. If you have a legal proxy from a street name stockholder, you must bring a form of photo identification, a legal proxy from the record holder (that is, the broker, bank or other holder of record) to the street name stockholder that is assignable, and the legal proxy from the street name stockholder to you. Stockholders may appoint only one proxy holder to attend the Special Meeting on their behalf.
Revoking Your Proxy
If you give a proxy, you may revoke it at any time before the Special Meeting or at the Special Meeting by doing any one of the following:
| | you may send a later-dated, signed proxy card; |
| | you may notify the Companys Secretary in writing to GS Acquisition Holdings Corp II, 200 West Street, New York, NY 10282, before the Special Meeting that you have revoked your proxy; or |
| | you may attend the Special Meeting, revoke your proxy, and vote during the Special Meeting via the virtual meeting website, as indicated above. |
No Additional Matters
The Special Meeting has been called only to consider the approval of the Business Combination Proposal, the NYSE Proposal, the Charter Proposal, the Governance Proposal, the Director Election Proposal, the Incentive Plan Proposal and the Adjournment Proposal. Under our bylaws, no other matters may be considered at the Special Meeting if they are not included in this proxy statement/prospectus, which serves as the notice of the Special Meeting.
155
Table of Contents
Who Can Answer Your Questions About Voting
If you have any questions about how to vote or direct a vote in respect of your shares of our common stock, you may call Innisfree M&A Incorporated, our proxy solicitor, at (877) 456-3463 (toll free), or Banks and Brokers may call collect: (212) 750-5833.
Redemption Rights
Pursuant to the GSAH Certificate of Incorporation, our public stockholders may request that we redeem all or a portion of such stockholders public shares if the Business Combination is consummated for a per-share price, payable in cash, equal to the pro rata portion of the trust account, calculated as of two business days prior to the consummation of the Business Combination, including interest (net of taxes payable). For illustrative purposes, as of [●], 2021, this would have amounted to approximately $[●] per outstanding public share. If a public stockholder properly exercises its redemption rights in full, then it will be electing to exchange all of its public shares for cash and will not own any public shares of the post-business combination company. Holders of our outstanding warrants do not have redemption rights in connection with the Business Combination. Public stockholders may elect to redeem their public shares even if they vote for the Business Combination Proposal or any other proposal.
In order to exercise your redemption rights, you must:
| | (a) hold public shares, or (b) if you hold public shares through units, elect to separate your units into the underlying public shares and warrants prior to exercising your redemption rights with respect to the public shares; |
| | prior to 5:00 p.m. Eastern Time on [], 2021 (two business days before the scheduled date of the Special Meeting) submit a written request to Continental Stock Transfer & Trust Company, N.A., our transfer agent, that we redeem all or a portion of your public shares for cash, affirmatively certifying in your request if you ARE or ARE NOT acting in concert or as a group (as defined in Section 13d-3 of the Exchange Act) with any other stockholder with respect to shares of our common stock; and |
| | deliver your public shares either physically or electronically through DTCs DWAC system to our transfer agent prior to 5:00 p.m. Eastern Time on [●], 2021 (two business days before the scheduled date of the Special Meeting). Stockholders seeking to exercise their redemption rights and opting to deliver physical certificates should allot sufficient time to obtain physical certificates from our transfer agent and time to effect delivery. It is our understanding that stockholders should generally allot at least two weeks to obtain physical certificates from our transfer agent. However, because we do not have any control over this process or over DTC, it may take significantly longer than two weeks to obtain a physical stock certificate. Stockholders who hold their shares in street name will have to coordinate with their broker, bank or other nominee to have the shares certificated or delivered electronically. If you do not submit a written request and deliver your public shares as described above, your shares will not be redeemed. |
Stockholders seeking to exercise their redemption rights, whether they are record holders or hold their shares in street name are required to either tender their certificates to our transfer agent prior to 5:00 p.m. Eastern Time on [●], 2021 (two business days before the scheduled date of the Special Meeting), or to deliver their shares to the transfer agent electronically using DTCs DWAC system, at such stockholders option.
Holders of outstanding units must separate the units into the underlying public shares and warrants prior to exercising redemption rights with respect to such public shares.
If you hold units registered in your own name, you must deliver the certificate for such units to our transfer agent with written instructions to separate such units into public shares and warrants. If you hold your units through a broker, bank or other nominee, you must notify your broker, bank or other nominee that you elect to separate the
156
Table of Contents
units into the underlying public shares and warrants. Your nominee must send written instructions by facsimile to our transfer agent. Such written instructions must include the number of units to be split and the nominee holding such units. Your nominee must also initiate electronically, using DTCs DWAC system, a withdrawal of the relevant units and a deposit of an equal number of public shares and warrants. This must be completed far enough in advance to permit your nominee to exercise your redemption rights upon the separation of the public shares from the units. While this is typically done electronically on the same business day, you should allow at least one full business day to accomplish the separation. If you fail to cause your public shares to be separated in a timely manner, you will likely not be able to exercise your redemption rights.
Notwithstanding the foregoing, a public stockholder, together with any affiliate of such public stockholder or any other person with whom such public stockholder is acting in concert or as a group (as defined in Section 13(d)(3) of the Exchange Act), will be restricted from redeeming its public shares with respect to more than an aggregate of 15% of the public shares. Accordingly, if a public stockholder, alone or acting in concert or as a group, seeks to redeem more than 15% of the public shares, then any such shares in excess of that 15% limit would not be redeemed for cash. In addition, pursuant to the GSAH Certificate of Incorporation, in no event will we redeem public shares in an amount that would cause our net tangible assets (as determined in accordance with Rule 3a51-1(g)(1) of the Exchange Act) to be less than $5,000,001. In such case, we would not proceed with the redemption of our public shares and the Business Combination, and instead may search for an alternate initial business combination.
The closing price of public shares on June 28, 2021, the most recent practicable date prior to the date of this proxy statement/prospectus, was $10.20. As of [●], 2021, funds in the trust account totaled $[●], which amount does not include any accrued dividends post- [●], 2021, or approximately $[●] per outstanding public share.
Prior to exercising redemption rights, public stockholders should verify the market price of the public shares as they may receive higher proceeds from the sale of their public shares in the public market than from exercising their redemption rights if the market price per share is higher than the redemption price. We cannot assure our stockholders that they will be able to sell their public shares in the open market, even if the market price per share is higher than the redemption price stated above, as there may not be sufficient liquidity in its securities when its stockholders wish to sell their shares.
Any request for redemption, once made by a holder of public shares, may be withdrawn at any time up to the deadline for exercising redemption requests (being two business days before the scheduled date of the Special Meeting) and thereafter, with our consent, until the consummation of the Business Combination. If you deliver your shares for redemption to our transfer agent, and later decide prior to such deadline not to elect redemption, you may request that our transfer agent return the shares (physically or electronically) to you. You may make such request by contacting our transfer agent at the phone number or address listed at the end of this section.
If the Business Combination is not consummated, the public shares will be returned to the respective holder, broker, bank or other nominee.
Appraisal Rights
Neither our stockholders nor our warrant holders have appraisal rights in connection with the Business Combination under the DGCL.
Proxy Solicitation Costs
We are soliciting proxies on behalf of our Board. This proxy solicitation is being made by mail, but also may be made by telephone or in person. We have engaged Innisfree to assist in the solicitation of proxies for the Special Meeting. The Company and its directors, officers and employees may also solicit proxies in person. The
157
Table of Contents
Company will ask banks, brokers and other institutions, nominees and fiduciaries to forward the proxy materials to their principals and to obtain their authority to execute proxies and voting instructions.
We will pay the cost of soliciting proxies for the Special Meeting. GSAH has engaged Innisfree to assist in the solicitation of proxies for the Special Meeting. GSAH has agreed to pay Innisfree a fee of $40,000, plus disbursements. GSAH will reimburse Innisfree for reasonable out-of-pocket expenses and will indemnify Innisfree and its affiliates against certain claims, liabilities, losses, damages and expenses. GSAH will also reimburse banks, brokers and other custodians, nominees and fiduciaries representing beneficial owners of our common stock for their expenses in forwarding soliciting materials to beneficial owners of our common stock and in obtaining voting instructions from those owners. GSAHs management team may also solicit proxies by telephone, by facsimile, by mail, on the Internet or in person. They will not be paid any additional amounts for soliciting proxies.
158
Table of Contents
PROPOSAL NO. 1APPROVAL OF THE BUSINESS COMBINATION
Overview
We are asking our stockholders to approve and adopt the Business Combination Agreement and approve the Business Combination. Our stockholders should read carefully this proxy statement/prospectus in its entirety for more detailed information concerning the Business Combination Agreement, which is attached as Annex A to this proxy statement/prospectus. See The Business Combination Agreement below, for additional information and a summary of certain terms of the Business Combination Agreement. You are urged to read carefully the Business Combination Agreement in its entirety before voting on this proposal.
We may consummate the Business Combination only if it is approved by the affirmative vote of a majority of the votes cast by holders of our outstanding shares of common stock represented at the Special Meeting by attendance via the virtual meeting website or by proxy and entitled to vote thereon at the Special Meeting.
The Business Combination Agreement
This subsection of the proxy statement/prospectus describes the material provisions of the Business Combination Agreement, but does not purport to describe all of the terms of the Business Combination Agreement. The following summary is qualified in its entirety by reference to the complete text of the Business Combination Agreement, a copy of which is attached as Annex A to this proxy statement/prospectus. You are urged to read the Business Combination Agreement in its entirety because it is the primary legal document that governs the Business Combination.
The Business Combination Agreement contains representations, warranties and covenants that the respective parties made to each other as of the date of the Business Combination Agreement or other specific dates. The assertions embodied in those representations, warranties and covenants were made for purposes of the contract among the respective parties and are subject to important qualifications and limitations agreed to by the parties in connection with negotiating the Business Combination Agreement. The representations, warranties and covenants in the Business Combination Agreement are also modified in part by the underlying disclosure letters (the disclosure letters), which are not filed publicly and which are subject to a contractual standard of materiality different from that generally applicable to stockholders and were used for the purpose of allocating risk among the parties rather than establishing matters as facts. We do not believe that the disclosure letters contain information that is material to an investment decision. Additionally, the representations and warranties of the parties to the Business Combination Agreement may or may not have been accurate as of any specific date and do not purport to be accurate as of the date of this proxy statement/prospectus. Accordingly, no person should rely on the representations and warranties in the Business Combination Agreement or the summaries thereof in this proxy statement/prospectus as characterizations of the actual state of facts about GSAH, Mirion or any other matter.
General Description of the Business Combination Agreement
The Business Combination Agreement
This subsection of the proxy statement describes the material provisions of the Business Combination Agreement, but does not purport to describe all of the terms of the Business Combination Agreement. The following summary is qualified in its entirety by reference to the complete text of the Business Combination Agreement, a copy of which is attached as Annex A to this proxy statement. You are urged to read the Business Combination Agreement in its entirety because it is the primary legal document that governs the Business Combination.
The Business Combination Agreement contains representations, warranties and covenants that the respective parties made to each other as of the date of the Business Combination Agreement or other specific dates. The assertions embodied in those representations, warranties and covenants were made for purposes of the contract
159
Table of Contents
among the respective parties and are subject to important qualifications and limitations agreed to by the parties in connection with negotiating the Business Combination Agreement. The representations, warranties and covenants in the Business Combination Agreement are also modified in part by the underlying disclosure letters (the disclosure letters), which are not filed publicly and which are subject to a contractual standard of materiality different from that generally applicable to stockholders and were used for the purpose of allocating risk among the parties rather than establishing matters as facts. We do not believe that the disclosure letters contain information that is material to an investment decision. Additionally, the representations and warranties of the parties to the Business Combination Agreement may or may not have been accurate as of any specific date and do not purport to be accurate as of the date of this proxy statement. Accordingly, no person should rely on the representations and warranties in the Business Combination Agreement or the summaries thereof in this proxy statement as characterizations of the actual state of facts about GSAH, Mirion or any other matter.
General Description of the Business Combination Agreement
On June 17, 2021, GSAH entered into the Business Combination Agreement with Mirion, the Charterhouse Parties the other parties thereto, pursuant to which, among other things and subject to the terms and conditions contained in the Business Combination Agreement, GSAH will acquire control of Mirion. After giving effect to the Business Combination, Mirion will continue as a subsidiary of GSAH.
Consideration to the Sellers in the Business Combination
Subject to the terms of the Business Combination Agreement and adjustments set forth therein, the consideration to be paid in connection with the Business Combination is $1,700,000,000 (the Total Consideration) and will be paid in a combination of equity and cash consideration. The cash consideration will be an amount equal to $1,310,000,000; provided, that if the Minimum Cash Condition is not met, and Mirion and the Charterhouse Parties elect to waive the Minimum Cash Condition, then the cash consideration will be equal to $1,310,000,000 less the amount by which $1,310,000,000 exceeds the Available Closing Cash. In exchange for the A Ordinary Shares of $0.01 each in the capital of Mirion, the B Ordinary Shares of $0.01 each in the capital of Mirion and certain loan notes due 2026 issued by Mirion Technologies (HoldingSub1), Ltd, each Seller may elect to receive cash or equity consideration or a combination thereof, which equity consideration shall be in the form of either shares of GSAHs Class A common stock or shares of GSAHs Class B common stock combined with shares of Class B common stock of a to be formed Delaware corporation that will be majority owned and controlled by GSAH. The Available Closing Cash will be an amount equal to (i) the amount of funds contained in GSAHs trust account (after reduction for the aggregate amount of payments required to be made in connection with any valid stockholder redemptions), plus (ii) the aggregate amount of cash that has been funded to and remains with GSAH pursuant to the Subscription Agreements as of immediately prior to the closing of the Business Combination (the Closing), plus (iii) the amounts delivered pursuant to the Debt Financing (as defined in the Agreement), plus (iv) the cash and cash equivalents of Mirion and its subsidiaries on a consolidated basis as of the date of the Closing (the Closing Date), plus (v) the proceeds, if any, from the sale by GSAH to GSAM Holdings LLC (GSAM Holdings) of shares of the GSAH Class A common stock, pursuant to the Backstop Agreement, less (vi) the total amount required to be paid to fully satisfy all obligations related to Mirions credit agreement as of the Closing Date, less (vii) certain transaction expenses, less (viii) $50,000,000 (collectively, the Available Closing Cash).
Mirion Material Adverse Effect
Under the Business Combination Agreement, certain representations and warranties of Mirion are qualified in whole or in part by a material adverse effect standard for purposes of determining whether a breach of such representations and warranties has occurred. In addition, it is a condition to the performance of GSAHs obligations to close the transactions contemplated by the Business Combination Agreement that no Material Adverse Effect occurs between signing and Closing. Pursuant to the Business Combination Agreement, a Material Adverse Effect means any state of facts, development, effect, change, circumstance, event or occurrence, that, individually
160
Table of Contents
or in the aggregate, has had, or would reasonably be expected to have, a material adverse effect on the business, assets, results of operations or financial condition of Mirion and its subsidiaries, taken as a whole; provided, however, that in no event would any of the following (or the effect of any of the following), alone or in combination, be deemed to constitute a Material Adverse Effect or be taken into account in determining whether a Material Adverse Effect has occurred or would reasonably be expected to occur: (i) any change in law or GAAP or any interpretation or enforcement thereof, (ii) any change in interest rates or economic, political, business, financial, commodity, currency or market conditions generally, (iii) any national or international political or social conditions in countries in which, or in the proximate geographic region in which, Mirion or any of its subsidiaries operates, including the engagement by the United States or such other countries in hostilities or the escalation thereof, whether or not pursuant to the declaration of a national emergency or war, or the occurrence or the escalation of any military or terrorist attack upon the United States or such other country, or any territories, possessions, or diplomatic or consular offices of the United States or such other countries or upon any military installation, equipment or personnel of the United States or such other country, (iv) any earthquake, hurricane, tsunami, tornado, flood, mudslide, wild fire or other natural disaster, act of God or other force majeure event, (v) any change generally affecting any of the industries or markets in which Mirion or any of its subsidiaries operates, (vi) the announcement or execution of the Business Combination Agreement, or the pendency, performance or consummation of the transactions contemplated thereby, including the impact thereof on relationships, contractual or otherwise, with customers, suppliers, distributors, partners, licensors, providers or employees, (vii) the taking of any action required by the terms of Business Combination Agreement or with the prior written consent of GSAH, (viii) any failure of Mirion and its subsidiaries, taken as a whole, to meet any projections, forecasts or budgets (provided that clause (viii) shall not prevent or otherwise affect a determination that any change or effect underlying such failure to meet projections or forecasts has resulted in, or contributed to, or would reasonably be expected to result in, a Material Adverse Effect, to the extent that such change or effect is not otherwise excluded from this definition of Material Adverse Effect) and (ix) COVID-19 or any other epidemic, pandemic or disease outbreak, or any law, directive, pronouncement or guideline issued by a governmental authority, the Centers for Disease Control and Prevention (or similar national or international organization), the World Health Organization or industry group providing for business closures, changes to business operations, sheltering-in-place or other restrictions that relate to, or arise out of, an epidemic, pandemic or disease outbreak (including the COVID-19 pandemic) or any change in such law, directive, pronouncement or guideline or interpretation thereof following the date of the Business Combination Agreement or Mirions or any of its subsidiaries compliance therewith; provided that, in the case of clauses (i)-(v) and (ix), such changes may be taken into account to the extent that such changes have had a disproportionate impact on Mirion and its subsidiaries, taken as a whole, as compared to other competitors or similarly situated companies operating in the industries or markets in which the Mirion and its subsidiaries operate.
Seller Material Adverse Effect
Under the Business Combination Agreement, certain representations and warranties of the Sellers are qualified in whole or in part by a material adverse effect standard for purposes of determining whether a breach of such representations and warranties has occurred. Pursuant to the Business Combination Agreement, a Seller Material Adverse Effect means, with respect to the Sellers, any state of facts, development, effect, change, circumstance, event or occurrence that, individually or in the aggregate, has prevented, materially delayed or materially impaired, or would reasonably be expected to prevent, materially delay or materially impair, the ability of the Sellers, considered as a group, to consummate the transactions contemplated by the Business Combination Agreement.
SPAC Material Adverse Effect
Under the Business Combination Agreement, certain representations and warranties of GSAH are qualified in whole or in part by a material adverse effect standard for purposes of determining whether a breach of such representations and warranties has occurred. In addition, it is a condition to the performance of Mirion and the Sellers obligations to close the transactions contemplated by the Business Combination Agreement that no SPAC Material Adverse
161
Table of Contents
Effect occurs between signing and Closing. Pursuant to the Business Combination Agreement, a SPAC Material Adverse Effect means, with respect to the SPAC, any state of facts, development, effect, change, circumstance, event or occurrence that, individually or in the aggregate, has prevented, materially delayed or materially impaired, or would reasonably be expected to prevent, materially delay or materially impair, the ability of the SPAC to consummate the transactions contemplated by the Business Combination Agreement.
Closing of the Business Combination
The Closing of the Business Combination is expected to occur by electronic exchange of documents at 10:00 a.m. Eastern Time, which will not be later than the third business day after the satisfaction or waiver of the conditions described below under the subsection Conditions to Closing of the Business Combination. Notwithstanding the foregoing, the Closing of the Business Combination may occur at such other time, date and location as may be mutually agreed upon in writing by GSAH, Mirion and the Charterhouse Parties.
Conditions to Closing of the Business CombinationConditions to Each Partys Obligations
The respective obligations of the parties to the Business Combination Agreement to effect the Closing and the other transactions contemplated by the Business Combination Agreement are subject to the satisfaction, any of which may be waived, in writing, by mutual agreement of GSAH, Mirion and the Charterhouse Parties at or prior to the Closing of the Business Combination of each of the following conditions:
| | No provision of law, and no judgment, injunction, order or decree of any applicable governmental authority, will prohibit the consummation of the Closing. |
| | Any applicable waiting period under the HSR Act (and any extensions thereof or any timing agreements, understandings or commitments obtained by request or other action of the United States Federal Trade Commission or the Antitrust Division of the United States Department of Justice, as applicable) will have expired or been terminated. |
| | The parties to the Business Combination Agreement will have received specified pre-Closing authorizations, consents, clearances, waivers and approvals of certain governmental authorities in connection with the execution, delivery and performance of the Business Combination Agreement and the transactions contemplated thereunder. |
| | The Registration Statement will have become effective in accordance with the Securities Act, no stop order shall have been issued by the SEC with respect to the Registration Statement and no action seeking such stop order shall have been threatened or initiated. |
| | The required vote of GSAHs stockholders to approve the Business Combination Proposal, the NYSE Proposal, the Charter Proposal, the Director Election Proposal, the Incentive Plan Proposal and the Adjournment Proposal shall have been duly obtained in accordance with the DGCL, the GSAH Certificate of Incorporation and bylaws, and the rules and regulations of the NYSE; |
| | GSAH will have at least $5,000,001 of net tangible assets following the exercise of any redemption rights by the Companys holders of Class A common stock in accordance with the GSAH Certificate of Incorporation and bylaws. |
Conditions to Closing of the Business CombinationConditions to GSAHs Obligations
The obligation of GSAH to consummate the Closing is subject to the satisfaction, at or prior to the Closing of the Business Combination, of each of the following conditions, any of which may be waived, in writing, by GSAH:
| | Mirion and the Sellers will have performed in all material respects all of their respective obligations under the Business Combination Agreement required to be performed thereby on or prior to the Closing Date. |
162
Table of Contents
| | (i) The representations and warranties of Mirion contained in Section 4.09(b) of the Business Combination Agreement must be true and correct in all respects as of the date of the Business Combination Agreement and as of the Closing Date, as if made at and as of the Closing Date; (ii) the fundamental representations and warranties of Mirion (i.e., representations related to organization and qualification, capitalization, authority and brokers) must be true and correct in all but de minimis respects (without giving effect to any limitations as to materiality or Material Adverse Effect or any similar limitation set forth therein) as of the date of the Business Combination Agreement and as of the Closing Date, as if made at and as of the Closing Date (except to the extent such representations and warranties expressly relate to an earlier date, and in such case, shall be so true and correct in all but de minimis respects at and as of such earlier date); and (iii) each other representation and warranty of Mirion contained in Article 4 of the Business Combination Agreement must be true and correct (without giving effect to any limitations as to materiality or Material Adverse Effect or any similar limitation set forth therein) as of the date of the Business Combination Agreement and as of the Closing Date, as if made at and as of the Closing Date (except to the extent such representations and warranties expressly relate to an earlier date, and in such case, shall be true and correct at and as of such earlier date), except, in the case of this clause (iii), where the failure to be so true and correct, individually or in the aggregate, has not had, and would not reasonably be expected to have, a Material Adverse Effect. |
| | The representations and warranties of the Sellers contained in Article 3 of the Business Combination Agreement must be true and correct (without giving effect to any limitations as to materiality or Material Adverse Effect or any similar limitation set forth therein) as of the date of the Business Combination Agreement and as of the Closing Date, as if made at and as of such date (except to the extent such representations and warranties expressly relate to an earlier date, and in such case, shall be true and correct at and as of such earlier date), except where the failure to be so true and correct, individually or in the aggregate, has not had, and would not reasonably be expected to have, a Seller Material Adverse Effect. |
| | GSAH will have received a certificate signed by an officer of Mirion, dated the Closing Date, certifying that the conditions specified in the three immediately preceding bullet points have been fulfilled. |
| | A majority of the holders of the A Ordinary Shares and the B Ordinary Shares of Mirion will have delivered the drag along notice. |
| | No Material Adverse Effect will have occurred since the date of the Business Combination Agreement. |
| | Mirion and its subsidiaries will have issued notice of suspension or termination of any contracts to certain sales channel partners and otherwise ceased doing business with such sales channel partners as requested in writing by GSAH. |
Conditions to Closing of the Business CombinationConditions to Mirions and the Sellers Obligations
The obligation of Mirion and the Sellers to consummate the Closing are subject to the satisfaction, at or prior to the Closing of the Business Combination, of each of the following conditions, any of which may be waived, in writing, by Mirion and the Charterhouse Parties:
| | GSAH will have performed in all material respects all of its obligations under the Business Combination Agreement required to be performed by it on or prior to the Closing Date. |
| | (i) The representations and warranties of GSAH contained in Section 5.09(b) of the Business Combination Agreement must be true and correct in all respects as of the Closing Date, as if made at and as of such date; (ii) the fundamental representations and warranties of GSAH (i.e., representations related to organization and qualification, capitalization, authority and brokers) must be true and correct (without giving effect to any limitations as to materiality or material adverse effect or any similar limitation set forth therein) in all but de minimis respects as of the Closing Date, as if made at and as of |
163
Table of Contents
| such date (except to the extent such representations and warranties expressly relate to an earlier date, and in such case, shall be so true and correct in all but de minimis respects at and as of such earlier date); and (iii) each other representation and warranty of GSAH contained in Article 5 of the Business Combination Agreement shall be true and correct (without giving effect to any limitations as to materiality or material adverse effect or any similar limitation set forth therein) as of the Closing Date, as if made at and as of such date (except to the extent such representations and warranties expressly relate to an earlier date, and in such case, shall be true and correct at and as of such earlier date), except, in the case of this clause (iii), where the failure to be so true and correct has not had, and would not reasonably be expected to have, a SPAC Material Adverse Effect. |
| | Mirion will have received a certificate signed by an officer of GSAH, dated the Closing Date, certifying that the conditions specified in the two immediately preceding bullets have been fulfilled. |
| | The Available Closing Cash will not be less than $1,310,000,000. |
| | Since the date of the Business Combination Agreement, there will have been no development, effect, change, circumstance, event or occurrence, that, individually or in the aggregate, has had, or would reasonably be expected to have a SPAC Material Adverse Effect. |
Representations and Warranties
Under the Business Combination Agreement, the Sellers made customary representations and warranties about such Seller relating to: existence and power; authorization; governmental authorization; noncontravention; ownership; and brokers fees.
Under the Business Combination Agreement, Mirion made customary representations and warranties about it relating to: existence and power; authorization; governmental authorization; noncontravention; capitalization; subsidiaries; financial statements; absence of certain changes; undisclosed material liabilities; material contracts; litigation; compliance with laws and court orders; real property; intellectual property; insurance coverage; employees; employee benefit plans; taxes; environmental matters; affiliate transactions; brokers fees; anti-bribery; anti-corruption; international trade; sanctions; data privacy; customers and suppliers; product liabilities and recalls; PPP loans; and debt financing.
Under the Business Combination Agreement, GSAH made customary representations and warranties about it relating to: corporate existence and power; authorization; governmental authorization; noncontravention; capital structure; SEC documents; controls; listing; registration statement and proxy statement; absence of certain changes; litigation; compliance with applicable laws; taxes; employees and employee benefit plans; affiliate transactions; properties; brokers fees; financial ability; PIPE financing; and trust account.
Covenants of the Parties
Covenants of Mirion
Mirion made certain covenants under the Business Combination Agreement, including, among other things, the covenants set forth below.
| | Mirion has agreed to, and to cause its subsidiaries to, from the date of the Business Combination Agreement until the earlier of the Closing or the termination of the Business Combination Agreement in accordance with its terms, operate in the ordinary course of business consistent with past practice. |
| | Subject to certain exceptions, from the date of the Business Combination Agreement until the earlier of the Closing or the termination of the Business Combination Agreement in accordance with its terms, Mirion will not, and will cause its subsidiaries not to: |
| | change or amend its organizational documents; |
164
Table of Contents
| | make, declare, set aside, establish a record date for or pay any dividend or distribution; |
| | enter into, assume, assign, partially or completely amend any material term of, modify any material term of or terminate any collective bargaining or similar agreement (including agreements with works councils and trade unions and side letters) to which Mirion or its subsidiaries is a party or by which it is bound, other than in the ordinary course of business consistent with past practice; |
| | (i) issue, deliver, sell, transfer, pledge, dispose of or place any lien (other than a permitted lien) on any shares or any other equity or voting securities of Mirion or any of its subsidiaries or (ii) issue any securities (including any shares, voting securities or loan capital) or grant any options, appreciation rights, share units, profits interests, warrants or other rights to purchase or obtain any shares or any other equity or voting securities or loan capital of Mirion and/or any of its subsidiaries; |
| | sell, assign, transfer, convey, abandon, subject to a lien, or otherwise dispose of any owned real property; |
| | sell, assign, transfer, convey, lease, license, abandon, allow to lapse or expire, subject to or grant any lien (other than permitted liens) on, or otherwise dispose of, any material assets, rights or properties (including leased real property) of Mirion and its subsidiaries, taken as a whole, other than in the ordinary course of business; |
| | waive, release, compromise, settle or satisfy any pending or threatened claim or compromise or settle any liability, (i) if such settlement would require payment by Mirion in an amount greater than $1,000,000 individually or in the aggregate, (ii) to the extent such settlement includes an agreement to accept or concede material injunctive relief or (iii) to the extent such settlement involves a governmental authority or alleged criminal wrongdoing; |
| | agree to modify in any respect materially adverse to Mirion and its subsidiaries any confidentiality or similar contract or agreement to which Mirion or any of its subsidiaries are a party; |
| | directly or indirectly acquire by merging or consolidating with, or by purchasing a substantial portion of the assets of, or by purchasing all of or a substantial equity interest in, or by any other manner, any business or any corporation, partnership, limited liability company, joint venture, association or other entity or person or division thereof; |
| | make any loans or advance any money or other property; |
| | redeem, purchase or otherwise acquire any shares (or other equity interests) of Mirion or any of its subsidiaries or any securities or obligations convertible (whether currently convertible or convertible only after the passage of time or the occurrence of certain events) into or exchangeable for any shares of capital stock (or other equity interests) of Mirion or any of its subsidiaries; |
| | adjust, split, combine, subdivide, recapitalize, reclassify or otherwise effect any change in respect of any shares or other equity interests or securities of Mirion; |
| | make any change in its customary accounting principles or methods of accounting materially affecting the reported consolidated assets, liabilities or results of operations of Mirion and its subsidiaries; |
| | adopt or enter into a plan of complete or partial liquidation, dissolution, merger, consolidation or recapitalization; |
| | make, change or revoke any material tax election, adopt or change any material accounting method with respect to taxes, file any amended material tax return, settle or compromise any material tax liability, surrender any right to claim a material refund of taxes, consent to any extension or waiver of the limitations period applicable to any material tax claim or assessment (other than any extension pursuant to an extension to file any tax return), change its tax residence or start to trade to a material extent through a permanent establishment or other taxable presence, or enter into any material closing agreement with respect to any tax; |
165
Table of Contents
| | (i) increase or grant any increase in the compensation, bonus, or other benefits (other than de minimis fringe or other benefits) of, or pay, grant or promise any bonus to, certain key employees; (ii) grant or pay any severance or change in control pay or benefits to, or otherwise increase the severance or change in control pay or benefits of, any current or former service provider; (iii) enter into, amend (other than immaterial amendments) or terminate any employee benefit plan, policy, program, agreement, trust or arrangement; (iv) take any action to accelerate the vesting or payment of, or otherwise fund or secure the payment of, any compensation or non-de minimis benefits under any employee plan; (v) grant any equity or equity based compensation awards; or (vi) hire or terminate (other than for cause) certain key employees; |
| | voluntarily fail to maintain in full force and effect material insurance policies covering the Mirion and its subsidiaries and their respective properties, assets and businesses in a form and amount consistent with past practices; |
| | enter into any transaction or amend in any material respect any existing agreement with any related party; |
| | enter into any agreement that materially restricts the ability of Mirion or its subsidiaries to engage or compete in any line of business or enter into a new line of business; |
| | make any capital expenditure that in the aggregate exceeds $2,500,000; |
| | receive, collect, compile, use, store, process, share, safeguard, secure (technically, physically or administratively), dispose of, destroy, disclose, or transfer (including cross-border) any personal information (or fail to do any of the foregoing, as applicable) in violation of any (i) applicable privacy laws, (ii) external-facing privacy policies or notices of, or (iii) contractual obligations with respect to any personal information; |
| | incur, guarantee or otherwise become liable for (whether directly, contingently or otherwise) any indebtedness; |
| | enter into, assume, assign, partially or completely amend any material term of, modify any material term of or terminate certain material contracts for which payments to or from Mirion or any of its subsidiaries would be expected to exceed $5,000,000 annually or $20,000,000 in the aggregate. |
| | Subject to confidentiality obligations and certain other restrictions, Mirion will, and will cause its subsidiaries to, afford to GSAH reasonable access to Mirions properties, books, contracts, commitments, records and appropriate officers and employees, and will use its commercially reasonable efforts to furnish GSAH with financial and operating data and other information concerning the affairs of Mirion and its subsidiaries as GSAH may reasonably request solely for purposes of consummating the transactions contemplated by the Business Combination Agreement. |
| | Mirion and each Seller (on behalf of itself and its respective controlled affiliates) waives any past, present or future claim of any kind against, and any right to access, the Trust Account, the trustee and GSAH, or to collect from the Trust Account any monies that may be owed to them by GSAH or any of its affiliates for any reason whatsoever, and will not seek recourse against the Trust Account at any time for any reason whatsoever. |
| | The Charterhouse Parties will: (i) cause the holders of a majority of Mirions A Ordinary Shares and B Ordinary Shares to deliver a duly executed joinder agreement and a duly executed election agreement and (ii) cause the applicable shareholders of Mirion to provide a drag along notice to certain other shareholders of Mirion. Promptly following the provision of the drag along notice, the Charterhouse Parties shall cause all called shareholders of Mirion to deliver, at least five (5) Business Days prior to the Closing Date, a duly executed joinder agreement and a duly executed election agreement. |
| | Mirion, the Charterhouse Parties and GSAH will use their reasonable best efforts to agree on a final steps plan regarding certain pre-Closing transactions. Prior to the Closing, Mirion will, and will cause |
166
Table of Contents
| its subsidiaries to, to take, or cause to be taken, all actions and to do, or cause to be done, all things necessary to effectuate the agreed-upon pre-Closing step plan, with such amendments, modifications, re-orderings and the like as Mirion may determine to be reasonably necessary and desirable to effect the transactions contemplated by the Business Combination Agreement, subject to certain consent rights of GSAH and the Charterhouse Parties. |
| | Mirion will take certain actions to supplement existing policies, procedures and practices concerning sales channel partners, including ceasing to do business with channel partners in certain jurisdictions, if requested by GSAH. |
| | Mirion and the Sellers will settle and terminate certain related party agreements. |
| | Mirion will reasonably cooperate with GSAH and GSAHs lender to obtain, owners and lenders title insurance policies with respect to owned real property, dated as of the Closing Date, issued from a title insurance company and in amounts reasonably satisfactory to GSAH, including to deliver such customary affidavits from officers of Mirion and its subsidiaries as reasonably requested by the title insurance company , including any affidavit required by the title company in order to issue a non-imputation endorsement. |
| | Neither Mirion, any Seller nor any of their respective controlled affiliates, directly or indirectly, will engage in any transactions involving the securities of GSAH prior to the time of the making of a public announcement regarding all of the material terms of the transactions contemplated by the Business Combination Agreement. |
| | No later than ten (10) Business Days prior to the Closing, Mirion will take such action as may be necessary such that, as of the Closing, (i) certain loan agreements between Mirion and certain of its officers will be terminated and of no further continued force or effect without any obligations or liabilities surviving the Closing, and (ii) all accounts payable to either party to such agreements will be settled and fully discharged with no further obligation or liability to either party. |
| | Mirion will cause Mirion Technologies (HoldingSub2) Ltd., a limited liability company incorporated in England and Wales with a company number 09299632 (the DCL Beneficiary) to use its reasonable best efforts to, and will cause each of its subsidiaries to use its reasonable best efforts to, arrange and obtain debt financing on terms and conditions not less favorable than (taken as a whole) those set forth in the debt commitment letter delivered by Mirion in connection with the execution of the Business Combination Agreement (or such other terms as are reasonably acceptable to the DCL Beneficiary and GSAH). |
| | Prior to the Closing, Mirion will, or shall cause its applicable subsidiaries to, repay in full any loans applied for or received by Mirion or any of its subsidiaries pursuant to the Paycheck Protection Program (PPP Loans) outstanding as of the date of the Business Combination Agreement (including any PPP Loans that as of the date of the Business Combination Agreement are subject to (i) an application for forgiveness, (ii) an audit by the U.S. Small Business Administration or the lender of such PPP Loan, or (iii) an escrow agreement). |
| | Neither Mirion nor any Seller will take, nor will Mirion or any Seller permit any of their respective affiliates or representatives to take, whether directly or indirectly, any action to solicit, initiate or engage in discussions or negotiations with, or enter into any agreement with, or encourage, or provide information to, any person or entity concerning any purchase of any of Mirions equity securities or the issuance and sale of any securities of, or membership interests in, Mirion or its subsidiaries or any merger or sale of substantial assets involving Mirion or its subsidiaries, other than immaterial assets or assets sold in the ordinary course of business. |
Covenants of GSAH
GSAH made certain covenants under the Business Combination Agreement, including, among other things, the covenants set forth below.
167
Table of Contents
| | Subject to certain exceptions, from the date of the Business Combination Agreement until the earlier of the Closing or the termination of the Business Combination Agreement in accordance with its terms, GSAH will not: |
| | change, modify or amend the Trust Agreement, or organizational documents; |
| | declare, set aside or pay any dividends on, or make any other distribution in respect of any outstanding capital stock of, or other equity interests in, GSAH; split, combine or reclassify any capital stock of, or other equity interests in, GSAH; or repurchase, redeem or otherwise acquire, or offer to repurchase, redeem or otherwise acquire, any capital stock of, or other equity interests in, GSAH; |
| | make, change or revoke any material tax election, adopt or change any material accounting method with respect to taxes, file any amended material tax return, settle or compromise any material tax liability, surrender any right to claim a material refund of taxes, consent to any extension or waiver of the limitations period applicable to any material tax claim or assessment (other than any extension pursuant to an extension to file any tax return), change its tax residence or start to trade to a material extent through a permanent establishment other taxable presence, or enter into any material closing agreement with respect to any tax; |
| | enter into, renew or amend in any material respect any transaction or contract with a related party of GSAH; |
| | waive, release, compromise, settle or satisfy any pending or threatened material claim or compromise or settle any liability; |
| | incur, guarantee or otherwise become liable for (whether directly, contingently or otherwise) any indebtedness; |
| | offer, issue, deliver, grant or sell, or authorize or propose to offer, issue, deliver, grant or sell, any capital stock of, other equity interests, equity equivalents, stock appreciation rights, phantom stock ownership interests or similar rights in, GSAH or any securities convertible into, or any rights, warrants or options to acquire, any such capital stock or equity interests; |
| | amend, modify or waive any of the terms or rights set forth in any private placement warrant; or |
| | merge or consolidate, restructure, reorganize or completely or partially liquidate or dissolve, or adopt or enter into a plan of complete or partial liquidation, dissolution, restructuring, recapitalization or other reorganization of GSAH; |
| | GSAH will take all necessary action to cause the Board of Directors GSAH as of immediately following the Closing to consist of nine (9) directors, of whom one (1) shall be the Chief Executive Officer of GSAH upon the Closing (i.e., the Chief Executive Officer of Mirion immediately prior to the Closing), two (2) shall be named by the Sponsor, one (1) shall be named by the Charterhouse Parties and the remainder shall be mutually agreed by the Charterhouse Parties, Mirion and GSAH prior to the Closing; |
| | Prior to the Closing, GSAH will file an amendment to its certificate of incorporation with the Secretary of State of the State of Delaware in the form agreed by the parties to the Business Combination Agreement; |
| | Concurrently with the Closing, GSAH shall cause the existing Registration Rights Agreement, dated June 29, 2020, to be amended and restated in the form of the Amended and Restated Registration Rights Agreement agreed by the parties to the Business Combination Agreement. |
| | GSAH will use reasonable best efforts to ensure that it remains listed as a public company, and that shares of Class A common stock of GSAH remain listed, on the NYSE. |
| | GSAH shall use reasonable best efforts to keep current and timely file all reports required to be filed or furnished with the SEC and otherwise comply in all material respects with its reporting obligations under applicable law. |
168
Table of Contents
| | Unless otherwise approved in writing by Mirion and the Charterhouse Parties, GSAH will not permit any material amendment or modification to be made to, or any waiver of any provision or remedy under, or any replacements or terminations of, the Subscription Agreements in any manner adverse to Mirion or the Charterhouse Parties. GSAH will use its reasonable best efforts to take, or cause to be taken, all actions and do, or cause to be done, all things necessary, proper or advisable to consummate the transactions contemplated by the PIPE Subscription Agreements on the terms and conditions described therein, including using its reasonable best efforts to enforce its rights under the PIPE Subscription Agreements to cause the PIPE Investors to pay the applicable purchase price under each PIPE Investors applicable Subscription Agreement in accordance with its terms. |
| | Unless otherwise approved in writing by Mirion and the Charterhouse Parties, GSAH will not permit any material amendment or modification to be made to, or any waiver of any provision or remedy under, or any replacement or termination of, the Backstop Agreement in any manner adverse to Mirion or the Charterhouse Parties. |
| | Upon the satisfaction (or waiver by GSAH) of the conditions set forth in Article 11 of the Business Combination Agreement, and in accordance with and pursuant to the Trust Agreement at the Closing, (A) GSAH will cause the documents, opinions and notices required to be delivered to the trustee pursuant to the Trust Agreement to be so delivered and (B) GSAH will make arrangements to cause the trustee to (1) pay as and when due all amounts payable to all holders, as of the date of the Business Combination Agreement, of Class A common stock and Class B common stock of GSAH who shall have previously validly elected to redeem their shares pursuant to a redemption of such stockholders shares and (2) promptly thereafter, pay all remaining amounts then available in the Trust Account in accordance with the Business Combination Agreement and the Trust Agreement. |
| | GSAH will use its reasonable best efforts to, and will cause its respective representatives to use their reasonable best efforts to, provide all cooperation in connection with the arrangement of the debt financing as may be reasonably requested by Mirion that is necessary or customary for financings of the type contemplated by the debt commitment letter. |
| | Subject to certain limitations, GSAH agrees to take all steps necessary or advisable to eliminate impediments under any antitrust, competition, or other applicable law (including mitigation measures imposed by CFIUS, ITAR or MINEFI) that are asserted by any governmental authority or any other party having jurisdiction over the transactions contemplated by the Business Combination Agreement. |
| | GSAH will not take, nor will it permit any of its affiliates or representatives to take, whether directly or indirectly, any action to solicit, initiate, continue or engage in discussions or negotiations with, or enter into any agreement with, or encourage, respond, provide information to or commence due diligence with respect to, any person or entity, concerning, relating to or which is intended or is reasonably likely to give rise to or result in, any offer, inquiry, proposal or indication of interest, written or oral relating to any business combination. |
Joint Covenants
| | GSAH, the Sellers and Mirion will use their respective reasonable best efforts to take, or cause to be taken, all actions and to do, or cause to be done, all things necessary or advisable under applicable law to consummate the transactions contemplated by the Business Combination Agreement. |
| | The Sellers, GSAH and Mirion will cooperate with one another (i) in determining whether any action by or in respect of, or filing with, any governmental authority is required, or any actions, consents, approvals or waivers are required to be obtained from parties to any material contracts, in connection with the consummation of the transactions contemplated by the Business Combination Agreement and (ii) in using their respective reasonable best efforts to take such actions or make any such filings, furnishing information required in connection therewith and seeking timely to obtain any such actions, consents, approvals or waivers. |
169
Table of Contents
| | The parties to the Business Combination Agreement will negotiate in good faith to establish a directors and officers liability insurance policy, to be in place as of the Closing, that includes full prior acts coverage and continuity. |
| | The parties to the Business Combination Agreement will consult with each other before issuing any press release or making any public statement with respect to the Business Combination Agreement or the transactions contemplated thereby and will not issue any such press release or make any such public statement prior to such consultation. |
| | Each party to the Business Combination Agreement shall give prompt notice to the other parties of (a) certain actions or investigations; (b) the occurrence or non-occurrence of any event whose occurrence or non-occurrence, as the case may be, could reasonably be expected to cause any condition set forth in Section 11.02 or Section 11.03 of the Business Combination Agreement not to be satisfied at any time from the date the Business Combination Agreement to the Closing; (c) any notice or other communication from any third-party alleging that the consent of such third-party is or may be required in connection with the transactions contemplated by the Business Combination Agreement; and (d) any regulatory notice, report or results of inspection from a governmental authority in respect of the transactions contemplated by the Business Combination Agreement. |
Survival of Representations, Warranties and Covenants; No Indemnification
None of the representations, warranties, covenants and agreements in the Business Combination Agreement or in any instrument, document or certificate delivered pursuant to the Business Combination Agreement survive the Closing, except for those covenants and agreements contained therein which by their terms expressly apply in whole or in part after the Closing and then only to such extent until such covenants and agreements have been fully performed. Neither Mirion nor the Sellers will have any indemnification obligations pursuant to the Business Combination Agreement. None of the foregoing will limit the rights of GSAH to pursue recoveries under any buyer-side representations and warranties insurance policy.
Termination
The Business Combination Agreement may be terminated at any time prior to the Closing as follows:
| | by mutual written agreement of GSAH, Mirion and the Charterhouse Parties; |
| | by GSAH, on the one hand, and Mirion and the Charterhouse Parties, on the other hand, if the Closing has not been consummated on or before November 30, 2021 (as may be extended under the Business Combination Agreement, or by mutual agreement of the parties, the End Date); provided, however, that if the conditions specified in Sections 11.01(b) and 11.01(c) of the Business Combination Agreement have not been satisfied by November 30, 2021, either GSAH, on the one hand, or Mirion and the Charterhouse Parties, on the other hand, may by written notice to the other party, extend the End Date to January 31, 2022; provided, further however, that if, following such extension of the End Date, the conditions specified in Sections 11.01(b) and 11.01(c) of the Business Combination Agreement have not been satisfied by January 31, 2022 because of a failure to receive the specified approval or approvals noted on Section 11.01(c) of the Company Disclosure Schedule, either GSAH, on the one hand, or Mirion and the Charterhouse Parties, on the other hand, may by written notice to the other party, extend the End Date to March 31, 2022; |
| | by written notice from either Mirion and the Charterhouse Parties, on the one hand, or GSAH, on the other hand, to the other(s) if consummation of the transactions contemplated by the Business Combination Agreement would violate any nonappealable final order, decree or judgment of any court or governmental authority having competent jurisdiction; |
| | by written notice to Mirion and the Charterhouse Parties from GSAH if there is any breach of any representation, warranty or covenant on the part of Mirion or the Sellers set forth in the Business |
170
Table of Contents
| Combination Agreement, such that the conditions specified in Sections 11.02(a), 11.02(b) and 11.02(c) of the Business Combination Agreement would not be satisfied at the Closing, subject to applicable cure periods as specified in the Business Combination Agreement; |
| | by written notice to GSAH from Mirion and the Charterhouse Parties if there is any breach of any representation, warranty or covenant on the part of GSAH set forth the Business Combination Agreement, such that the conditions specified in Sections 11.03(a) and 11.03(b) of the Business Combination Agreement would not be satisfied at the Closing, subject to applicable cure periods as specified in the Business Combination Agreement; |
| | by written notice from either Mirion and the Charterhouse Parties, on the one hand, GSAH, on the other hand, to the other(s) if the approval of the Business Combination Proposal, the NYSE Proposal, the Charter Proposal, the Director Election Proposal, the Incentive Plan Proposal and the Adjournment Proposal by the required vote of GSAHs stockholders is not obtained; or |
| | by written notice from either Mirion and the Charterhouse Parties, on the one hand, or GSAH, on the other hand, to the other(s) if the Minimum Cash Condition is incapable of being satisfied and Mirion and the Charterhouse Parties have not waived the Minimum Cash Condition within fifteen (15) Business Days of receipt by Mirion and the Charterhouse Parties of a written notice from GSAH that the Minimum Cash Condition has not been satisfied and is incapable of being satisfied. |
If the Business Combination Agreement is terminated, the Business Combination Agreement will become void and have no effect, without any liability on the part of any party thereto or its respective affiliates, shareholders, stockholders, directors, officers, employees, agents, consultants or representatives to the other parties to the Business Combination Agreement; provided that if such termination shall result from (i) fraud, (ii) the knowing and willful failure of a party to fulfill a condition to the performance of the obligations of the other party, (iii) the knowing and willful failure of a party to perform a covenant of the Business Combination Agreement or (iv) the knowing and willful breach by a party thereto of any representation or warranty or agreement contained herein, such party will, subject to the terms of the Business Combination Agreement, be fully liable for any and all losses, damages, claims, costs or expenses incurred or suffered by the other parties as a result of such failure or breach.
Amendments
Any provision of the Business Combination Agreement may be amended if such amendment is in writing and is signed by each of Mirion, GSAH and the Charterhouse Parties, on behalf of the Sellers.
The foregoing description of the Business Combination Agreement does not purport to be complete and is qualified in its entirety by the terms and conditions of the Amended and Restated Sponsor Agreement, a copy of which is attached hereto as Annex A and is incorporated herein by reference.
Related Agreements
This section describes certain additional agreements entered into or to be entered into pursuant to the Business Combination Agreement, but does not purport to describe all of the terms thereof. The following summary is qualified in its entirety by reference to the complete text of each of the agreements. Each of the Amended and Restated Sponsor Agreement, the Subscription Agreements, the Amended and Restated Registration Rights Agreement, the Backstop Agreement and the Option Agreement (or forms thereof) are attached hereto as Annex D, Annex E, Annex F, Annex G and Annex H, respectively. You are urged to read such agreements in their entirety prior to voting on the proposals presented at the Special Meeting.
Amended and Restated Sponsor Agreement
In connection with the execution of the Agreement, the Company amended and restated that certain letter, dated June 29, 2020, by and among the Company, the Sponsor, GSAM Holdings and GS Employee Participation
171
Table of Contents
(collectively, the Insiders), pursuant to which, among other things, the Company and the Insiders agreed (i) to vote any shares of the Companys securities in favor of the Business Combination and other Business Combination proposals, (ii) not to redeem any shares of the GSAH Class A common stock or the founder shares, in connection with the optional stockholder redemption, (iii) not to transfer any founder shares until the earlier of (x) the one year anniversary of the Closing Date and (y) the day following the trading date when the last reported sale price of the GSAH Class A common stock first equals or exceeds $12.00 per share (as adjusted for stock splits, stock dividends, reorganizations, recapitalizations and the like) for any 20 trading days within any 30 consecutive trading day period commencing at least 150 days after the Closing Date, subject to the clear market provisions in the Amended and Restated Registration Rights Agreement, (iv) not to transfer any shares issued to GSAM Holdings as part of the PIPE Investment if such shares are retained by GSAM Holdings or its affiliates (but, for the avoidance of doubt, not if distributed to GSAM Holdings or its permitted transferees employees, investment partners or clients) for a period of 180 days after the Closing, subject to the clear market provisions in the Amended and Restated Registration Rights Agreement, and (v) to be bound to certain other obligations as described in the Amended and Restated Sponsor Agreement.
Additionally, the founder shares will be subject to vesting in three equal tranches, based on the volume weighted average price of the GSAH Class A common stock being greater than or equal to $12.00, $14.00 and $16.00, respectively, per share for any 20 trading days in any 30 consecutive trading day period. Such founder shares will be forfeited to the Company for no consideration if they fail to vest within five years of the Closing. The Sponsor has issued membership interests intended to be treated as profits interests for U.S. income tax purposes to each of Lawrence D. Kingsley, who will serve as Chairman of the Board of the Company when the Business Combination closes, Thomas Logan, Chief Executive Officer of Mirion, and Brian Schopfer, Chief Financial Officer of Mirion, whereby such individuals will have an indirect interest in the founder shares held by the Sponsor, subject to vesting upon the satisfaction of service, performance and other conditions, including the Closing.
Each of the holders of the founder shares have agreed to waive the anti-dilution adjustments provided for in the Companys Amended and Restated Certificate of Incorporation applicable to the founder shares in connection with the Business Combination, including the PIPE Investment. As a result of such waiver, the 18,750,000 founder shares will automatically convert into shares of the GSAH Class A common stock on a one-for-one basis upon the consummation of the Business Combination.
The foregoing description of the Amended and Restated Sponsor Agreement does not purport to be complete and is qualified in its entirety by the terms and conditions of the Amended and Restated Sponsor Agreement, a copy of which is attached hereto as Annex D and is incorporated herein by reference.
Subscription Agreements
Concurrently with the execution of the Agreement, the Company entered into Subscription Agreements with the PIPE Investors, pursuant to, and on the terms and subject to the conditions of which, the PIPE Investors have collectively subscribed for 90,000,000 shares of the GSAH Class A common stock for an aggregate purchase price equal to $900,000,000. The PIPE Investment will be consummated substantially concurrently with the Closing.
The Subscription Agreements for the PIPE Investors (other than GSAM Holdings, whose registration rights are governed by the Amended and Restated Registration Rights Agreement) provide for certain registration rights. In particular, the Company is required to, as soon as practicable but no later than (i) 30 calendar days following the Closing Date, file with the SEC (at the Companys sole cost and expense) a registration statement registering the resale of such PIPE Shares, and use its commercially reasonable efforts to have such registration statement declared effective as soon as practicable after the filing thereof, but no later than the earlier of (x) the 60th calendar day (or 90th calendar day if the SEC notifies the Company that it will review such registration statement) following the Closing and (y) the 10th business day after the date the Company is notified (orally or in
172
Table of Contents
writing, whichever is earlier) by the SEC that such registration statement will not be reviewed or will not be subject to further review. Such registration statement is required to be kept effective until the earliest of (i) the date the PIPE Shares thereunder have been sold by the non-GSAM PIPE Investors, (ii) the date the PIPE Shares may be sold without restrictions under Rule 144 under the Securities Act, including without limitation, any volume and manner of sale restrictions that may be applicable to affiliates under Rule 144 and without the requirement for the Company to be in compliance with the current public information required under Rule 144(c)(1) (or Rule 144(i)(2), if applicable) and (iii) three years after effectiveness of such registration statement.
The Subscription Agreements will terminate with no further force and effect upon the earliest to occur of: (i) such date and time as the Business Combination Agreement is terminated in accordance with its terms; (ii) upon the mutual written agreement of the parties to such Subscription Agreement; (iii) the conditions contained in the Subscription Agreement not being satisfied or waived prior to the Closing; and (iv) the first anniversary of the date of the Subscription Agreement if the closing pursuant to the Subscription Agreement has not yet occurred.
The foregoing description of the Subscription Agreements does not purport to be complete and is qualified in its entirety by the terms and conditions of the form of Subscription Agreement, a copy of which is attached hereto as Annex E and is incorporated herein by reference.
Amended and Restated Registration Rights Agreement
At the Closing, the Company will enter into the Amended and Restated Registration Rights Agreement with the GS Holders and the Sellers (collectively, with each other person who has executed and delivered a joinder thereto, the RRA Parties), pursuant to which the RRA Parties will be entitled to registration rights in respect of certain shares of the GSAH Class A common stock and certain other equity securities of the Company that are held by the RRA Parties from time to time. The Company will be required to register up to approximately 150.0 million shares (including 12.5 million shares in the event GSAM Holdings exercises the Backstop Agreement and/or the Option Agreement in full and excluding 18.75 million founder shares subject to vesting conditions).
The Amended and Restated Registration Rights Agreement provides that the Company will use commercially reasonable efforts to file with the SEC a shelf registration statement pursuant to Rule 415 under the Securities Act registering the resale of certain shares of the GSAH Class A common stock and certain other equity securities of the Company held by the RRA Parties as soon as reasonably practicable but no later than 30 calendar days following the consummation of the Business Combination and use its commercially reasonably efforts to have such shelf registration statement declared effective as soon as reasonably practicable after the filing thereof and no later than the earlier of (x) the 90th calendar day following the filing date if the SEC notifies the Company that it will review such shelf registration statement and (y) the 10th business day after the date the Company is notified in writing by the SEC that such shelf registration statement will not be reviewed or will not be subject to further review.
Each of (i) the Charterhouse Parties, (ii) the GS Holders or (iii) the holders of at least thirty percent (30%) in interest of the then outstanding registrable securities (each of (i), (ii) or (iii), the Demanding Holders) will be entitled to demand registration rights in connection with an underwritten offering. The Charterhouse Parties have an exclusive right for a 90-day period beginning on the 181st day after the Closing (the Charterhouse Demand Period) to exercise a single demand right. The Demanding Holders will be, at any time and from time to time on or after the date the Charterhouse Demand Period ends, entitled to demand registrations of all or part of their registrable securities. Such demand registrations are subject to certain offering thresholds, applicable lock-up restrictions and certain other conditions.
In addition, the RRA Parties have certain piggy-back registration rights. The Amended and Restated Registration Rights Agreement includes customary indemnification and confidentiality provisions. The Company
173
Table of Contents
will bear the expenses incurred in connection with the filing of any registration statements filed pursuant to the terms of the Amended and Restated Registration Rights Agreement.
The foregoing description of the Amended and Restated Registration Rights Agreement does not purport to be complete and is qualified in its entirety by the terms and conditions of the form of Amended and Restated Registration Rights Agreement, a copy of which is attached hereto as Annex F and is incorporated herein by reference.
Backstop Agreement
In connection with the execution of the Agreement, GSAM Holdings and the Company have entered into the Backstop Agreement pursuant to which GSAM Holdings has committed to purchase from the Company up to 12,500,000 shares of the GSAH Class A common stock at a price per share equal to $10.00 immediately prior to (and contingent upon) the Closing, solely to the extent necessary to fund any valid redemptions by the Companys stockholders that results in the Cash Shortfall being greater than zero dollars, contingent upon the terms and subject to the conditions set forth in the Backstop Agreement.
The foregoing description of the Backstop Agreement does not purport to be complete and is qualified in its entirety by the terms and conditions of the Backstop Agreement, a copy of which is attached hereto as Annex G and is incorporated herein by reference.
Option Agreement
In connection with the execution of the Agreement, at the Closing, GSAM Holdings and Sellers that elect to receive cash for their Existing Mirion Shares will enter into the Option Agreement pursuant to which such Sellers will agree to, at the option of GSAM Holdings and subject to there being a partial exercise of the Backstop Agreement as described above, sell to GSAM Holdings up to 12,500,000 shares of the GSAH Class A common stock to be received by such Sellers pursuant to the Business Combination Agreement at the Closing at a price per share equal to $10.00 in cash, on the terms and subject to the conditions set forth in the Option Agreement. In the event there are sufficient redemptions of public shares such that there is a Cash Shortfall, the Company may exercise its rights under the Backstop Agreement to require GSAM Holdings to purchase up to 12,500,000 shares of GSAH Class A common stock to cover such Cash Shortfall as described above. If the Company exercises its rights under the Backstop Agreement for less than 12,500,000 shares of GSAH Class A common stock, GSAM Holdings has the right, but not the obligation, under the Option Agreement to purchase up to the difference of 12,500,000 shares of GSAH Class A common stock and the amount of shares purchased under the Backstop Agreement from the Sellers party to the Option Agreement.
The foregoing description of the Option Agreement does not purport to be complete and is qualified in its entirety by the terms and conditions of the form of Option Agreement, a copy of which is attached hereto as Annex H and is incorporated herein by reference.
Background of the Business Combination
The Company is a blank check company incorporated as a Delaware corporation on May 31, 2018, and formed for the purpose of effecting a merger, capital stock exchange, asset acquisition, stock purchase, reorganization or similar business combination with one or more businesses. The Business Combination was the result of an extensive search for a potential transaction that leveraged Mr. Knotts industry experience and operating capabilities, along with Goldman Sachs unique sourcing infrastructure and experience investing in public and private markets. The terms of the Business Combination were the result of extensive negotiations between our representatives, representatives of Mirion and representatives of the Charterhouse Parties.
Prior to the consummation of our IPO, neither the Company, nor anyone on its behalf contacted any prospective target business or had discussion, formal or otherwise, with respect to a transaction with the Company.
174
Table of Contents
On June 29, 2020, the Company completed its IPO. After the completion of our IPO, the Company commenced an active search for business combination candidates using the Companys network of investment bankers, private equity firms, consulting firms, and numerous other business relationships. During this period, the Company retained the Goldman Sachs Investment Banking Division to act as financial advisor to the Company and will receive compensation in connection therewith. In selecting Goldman Sachs as financial advisor (including as placement agent in connection with the PIPE Investment) the Company considered, among other things, (1) Goldman Sachs familiarity with the Company, including as a result of Goldman Sachs acting as book-running manager of the Companys IPO, (2) the fee to be paid to Goldman Sachs compared to the fees that the Company believed would be typical in connection with an investment bank serving as financial advisor for a business combination valued at over $2.5 billion (and as placement agent in an approximately $900 million private placement) as well as the other terms of the engagement letter, (3) the Companys belief that, as an internationally recognized investment banking firm with substantial experience in similar transactions, Goldman Sachs was well equipped to help the Company execute the Business Combination (and the PIPE Investment), and (4) that the Company previously disclosed, among other potential conflicts, that it may engage Goldman Sachs in connection with the Companys initial business combination in its prospectus related to its IPO and its Annual Report on Form 10-K for the year ended December 31, 2020.
The Company considered numerous potential target businesses with the objective of consummating its initial business combination. Representatives of the Company contacted and were contacted by numerous individuals and entities who presented ideas for business combination opportunities, including financial advisors and companies in the aerospace, specialty chemical and general industrial sectors.
In evaluating potential acquisition targets, the Company considered, among other things, businesses it believed had: (i) an enterprise value greater than $2.0 billion, (ii) advantageous positions in their respective industries, (ii) the ability to differentiate with technology, products and services, (iii) pathways for organic and inorganic growth, (iv) a capable, engaged and experienced management team, (iv) attractive margins and free cash flow characteristics, and (v) opportunities for margin expansion to be realized with the assistance of Mr. Knott, the Chief Executive Officer of the Company or industry contacts recruited by the Company. The Company also sought to identify companies that it believed would benefit from being a publicly-held entity, particularly with respect to access to capital for both organic growth and for use in acquisitions.
Throughout the evaluation process, the Company evaluated over 800 potential acquisition targets and made contact with representatives of over 50 of such potential targets to discuss a potential business combination transaction, ultimately receiving detailed information on 20 such potential targets (such other potential targets, the Other Targets) and entering into non-disclosure agreements with 16 of the Other Targets, in addition to Mirion. The company submitted a proposal on six of the Other Targets. The Company did not enter into exclusivity, nor did the Company agree to terms, with any of the potential targets other than Mirion and Company A, as described below.
The Other Targets included (i) a company in the cleaning chemicals industry (Company A), (ii) a company in the global packaging industry (Company B), (iii) a company in the software industry (Company C), (iv) a company in the household care industry (Company D), (v) a company in the healthcare services industry (Company E), (vi) a company in the media industry (Company F), (vii) a company in the financial services industry (Company G), (viii) a company in the investment management industry (Company H), (ix) a company in the alternative asset management industry (Company I), (x) a company in the media industry (Company J), (xi) a company in the battery technology industry (Company K), (xii) a company in the automotive industry (Company L), (xiii) a company in the specialty chemicals industry (Company M), (xiv) a company in the financial services industry (Company N), (xv) a company in the healthcare services industry (Company O) and (xvi) a company in the industrial services industry (Company P).
Each of Companies A, B, C, E, M, P had an enterprise value in the range of $5.0 to $10.0 billion. Each of the remaining Other Targets had an enterprise value in the range of $2.0 to $5.0 billion.
175
Table of Contents
The Company engaged in discussions with Company A from August 2020 through November 2020 but could not reach a mutually acceptable valuation with Company A as a result of concerns about Company As competitive position and future outlook. In connection with the Companys evaluation of Company A, in October 2020, the Company discussed a potential partnership with Lawrence D. Kingsley. Mr. Knott and Mr. Kingsley were in regular communication regarding the opportunity with Company A.
The Company engaged in discussions with Company B from November 2020 through December 2020 and submitted an initial indication of interest, but could not reach a mutually acceptable valuation.
The Company engaged in discussions with Company C from October 2020 through December 2020 and submitted an indication of interest, but developed concerns around Company Cs on-going product transitions and could not reach a mutually acceptable valuation.
The Company engaged in discussions with Company D in August 2020, but Company D halted the discussions after an accident at one of their facilities significantly impacted their operations.
The Company engaged in discussions with Company E from August 2020 to September 2020, but could not reach a mutually acceptable valuation.
The Company engaged in discussions with Company F from September 2020 through November 2020 and then in February 2021, but ultimately terminated the discussions with Company F when discussions with Mirion progressed to the point that Company determined to focus its efforts on a transaction with Mirion.
The Company engaged in discussions with Company G, H and I from September 2020 through October 2020, and also explored a potential business combination, but ended the discussions upon further due diligence.
The Company engaged in discussions with Company M in April 2021, but halted the discussions when discussions with Mirion progressed to the point that Company determined to focus its efforts on a transaction with Mirion.
The Company terminated discussions with each of Companies J, K, L, N, O and P after initial due diligence when it determined that the relevant company did not meet the key investment criteria discussed above.
Beginning in January 2021, representatives of Goldman Sachs Investment Banking Division, the Companys financial advisor, contacted representatives of Mirion, a portfolio company of Charterhouse Capital Partners LLP (Charterhouse) to discuss possible business combination opportunities for the Company. They subsequently arranged a telephone call for January 25, 2021, to introduce representatives of GSAM who were working with the Company (and for purposes of this section any references to representatives of the Company shall include representatives of GSAM that were working with the Company) in connection with the Companys search for business combination candidates, to Mirion. Mr. Knott discussed the potential opportunity with Mr. Kingsley as part of their ongoing review of targets for a potential business combination.
The following day, on January 26, 2021, representatives of Goldman Sachs Investment Banking Division and the Company met with Mr. Thomas Logan, Founder and Chief Executive Officer of Mirion and Mr. Brian Schopfer, Chief Financial Officer of Mirion, to discuss the possibility of a business combination.
On January 27, 2021, Mr. Knott and Mr. Logan met again to discuss the Companys interest in a potential business combination between the Company and Mirion. Mr. Logan indicated that Mr. Knott and other representatives of the Company should meet with representatives of Charterhouse as a next step in the process. During this period of time, Mr. Knott spoke to members of Companys Board of Directors regarding Mirion, its business model and potential for a transaction.
On February 2, 2021, Mr. Knott spoke in detail with Mr. Kingsley regarding the transaction and a potential partnership with Mr. Kingsley whereby Mr. Kingsley would assist in the Companys due diligence of Mirion. Mr. Knott and Mr. Kingsley were in regular communication throughout the Companys due diligence and negotiations. This regular communication continued throughout the transaction process.
176
Table of Contents
On February 3, 2021, representatives of the Company submitted a preliminary indication of interest to representatives of Charterhouse (the February 3 Letter). The February 3 Letter described the Companys interest in a business combination and contemplated a 100% acquisition of Mirion by the Company ascribing an enterprise value of $2.4 billion, and proposed a 30-day exclusivity period.
On February 4, 2021, representatives of Charterhouse contacted representatives of the Company to discuss the February 3 Letter and the potential advantages of effecting a transaction with a special purpose acquisition company, and in particular the Company, as compared to various alternative transaction structures.
On February 8, 2021, representatives of Charterhouse responded to the February 3 Letter, requesting that the Company clarify its position on value, transaction execution, timing and the due diligence process. Later, on February 8, 2021, representatives of Goldman Sachs Investment Banking Division spoke with representatives of Charterhouse regarding the February 3 Letter, and reiterated the Companys interest in pursuing a potential business combination with Mirion.
On February 11, 2021, representatives of the Company sent a written response to Charterhouse addressing the questions Charterhouse posed on February 8, 2021, which the parties discussed in detail on a call the following day.
On February 12, 2021, representatives of the Company held a call with representatives of Charterhouse to review the February 3 Letter. In response to Charterhouses request, representatives of the Company indicated that, subject to further due diligence, it may be willing to increase the valuation to Charterhouse. Additionally, the Company described that the focus of its financial diligence efforts would be on understanding Mirions expected financial performance, durability during periods of economic volatility, M&A opportunities and potential for continued operational improvement. The Company also provided a detailed execution timeline.
On February 15, 2021, representatives of Charterhouse sent a draft confidentiality agreement to representatives of the Company. Between February 15, 2021 and February 18, 2021, representatives of the Company and representatives of Charterhouse negotiated the confidentiality agreement covering confidential and proprietary information of Mirion and its affiliates. On February 18, 2021, the Company entered into the confidentiality agreement with Mirion.
On February 18, 2021, representatives of Charterhouse provided overview materials regarding Mirion to representatives of the Company in order to familiarize them with the nature of Mirions business and its financial profile as well as a preliminary term sheet in connection with a proposed business combination between the Company and Mirion. The term sheet contemplated total transaction consideration of $1.7 billion in cash consideration to Mirion equityholders, based on an enterprise value of Mirion of $2.5 billion, debt and equity financing of the transaction and an exclusivity period of 21 days.
From February 18, 2021 through February 25, 2021, representatives of Charterhouse and representatives of the Company exchanged drafts of the proposed term sheet and held telephonic discussions and negotiations regarding the terms. The key areas of discussion and negotiation included: management equity rollover, size of the PIPE Investment, treatment of transaction expenses, post-closing adjustments, transaction closing conditions (including with respect to regulatory consents) and exclusivity period.
On February 24, 2021, Mr. Knott discussed with the members of the Companys Board the potential transaction opportunity, preliminary terms negotiated in the term sheet, anticipated relative valuation and Mirions historical financial performance as well as its business model. In addition, Mr. Knott discussed and received approval for signing a preliminary term sheet with Charterhouse and agreeing to an exclusivity period to continue the diligence process. Finally, Mr. Knott discussed economic terms of the potential partnership with Mr. Kingsley for the transaction opportunity, including the possibility of Mr. Kingsley serving as Chairman of the Board of Mirion.
177
Table of Contents
On February 25, 2021, Mirion and the Company exchanged emails with a final version of a non-binding term sheet which contemplated transaction consideration based on an equity value of Mirion of $2.5 billion, comprised of $1.599 billion in cash to Charterhouse and its co-investors and shares of the Companys common stock to be issued to Mirion management, the expectation that the Company would raise at least $1.79 billion in equity financing and the expectation of an exclusivity period of 21 days from execution. The valuation was developed through consultation with the Company management and Mr. Kingsley, preliminary review of Mirions operating plan and financial forecast (including the achievability of such forecast) and consideration of certain customary valuation methodologies, including discounted cash flow analysis, relative valuation as compared to Mirions peers and relative free cash flow yield.
Following the agreement on the term sheet, the Company, Mr. Kingsley and its legal and financial advisors, commenced an in depth business and financial due diligence review of Mirion and continued to negotiate a more detailed term sheet and exclusivity agreement.
On February 26, 2021, the Company and its representatives were granted access to an electronic data room to facilitate their due diligence review of Mirion.
On March 2, 2021, Messrs. Knott, Logan, Kingsley and Jo Natauri, Global Head of Healthcare Investing within Goldman Sachs Asset Management, had a dinner meeting during which they discussed the potential business combination and the merits of such a transaction.
On March 3, 2021, representatives of Mirion held a full day meeting with representatives of the Company which began a detailed due diligence review of Mirion. In addition to the diligence session completed that day, the Company developed a detailed diligence process and workplan.
From March 3, 2021 to March 5, 2021, Mr. Knott discussed the opportunity, diligence findings to date and outstanding diligence items with the Companys Board.
On March 9, 2021, the Company spoke with a financial accounting firm and a third party consulting firm regarding market and financial due diligence. Both had been previously engaged by Mirion starting in December 2020 to conduct financial and tax vendor due diligence. The third party consulting firm reviewed Mirions competitive positioning and customer perception and conducted market due-diligence in connection with Mirions acquisition of Sun Nuclear. The financial accounting firms and consulting firms assessments were reviewed with the Company, and the Companys management reviewed such assessments with the Board.
Throughout the month of March 2021, in connection with the Companys due diligence review of Mirion, representatives of each party and their respective advisors (acting at the direction of the respective party) held several dozen calls and meetings in furtherance of that review, including in person management meetings in Atlanta, Georgia on March 16 and 17, 2021.
On March 23, 2021 and March 24, 2021, Mr. Knott, Mr. Kingsley, Mr. Logan and representatives of Mirion visited facilities in Meriden, Connecticut; Melbourne, Florida and Oak Ridge, Tennessee.
On March 29, 2021 and March 30, 2021, representatives of the Company held full day onsite meetings in Atlanta with representatives of Mirion to continue its diligence review.
On March 26, 2021, the Company held a meeting of its board of directors. Mr. Knott discussed the diligence finding to date, site visits and findings and Mr. Kingsleys perspective with the Board of Directors. Mr. Knott also discussed Companys intention to revise its initial proposal to Charterhouse with two options relating to amount of rollover equity or reduced enterprise value.
178
Table of Contents
On March 31, 2021, the Company sent a revised proposal to representatives of Charterhouse which reflected two options: one proposal which contemplated reducing the enterprise value of Mirion from $2.5 billion to $2.4 billion, which would be offset by the Company including an additional $15 million in expenses related to securing committed financing, without a rollover of equity, and an alternative proposal that maintained Mirions enterprise value of $2.5 billion, but would require the Charterhouse parties to roll $300 million in equity, which would be subject to a 6 month lock-up.
From March 31, 2021 to April 8, 2021, representatives of the Company and representatives of Charterhouse had numerous discussions regarding the two proposals.
On April 8, 2021, representatives of the Company sent a revised term sheet describing $2.5 billion enterprise value to Mirion with $300 million of rollover equity and a form of exclusivity agreement to representatives of Charterhouse based on conversations since March 31, 2021.
On April 9, 2021, the Company, Charterhouse, Mirion and their respective representatives held an all-hands call to discuss the negotiation of exclusivity, process and timeline for the potential business combination of the Company and Mirion including with respect to, execution risk, feedback from initial discussions with potential PIPE Investors, financing and the impact of current market conditions on a potential transaction. Following the meeting, the parties reaffirmed their interest in pursuing a potential business combination.
On April 9-12, 2021, Mr. Knott discussed the transaction proposal with the members of the Board as well as the proposed exclusivity. Mr. Knott also discussed the proposed transaction structure and relative valuation as well as the remaining items being pursued in diligence prior to the PIPE capital raise.
On April 13, 2021, the members of the Companys Board determined to continue the discussions regarding a potential acquisition of Mirion and approved the exclusivity agreement and the revised term sheet specifying $2.5 billion of enterprise value and $300 million of equity rollover by Charterhouse.
On April 14, 2021, Charterhouse, Mirion and the Company entered into an exclusivity agreement and term sheet providing for exclusive negotiations through May 5, 2021.
On April 14, 2021, Representatives of GSAM spoke with members of Charterhouse regarding transaction structure, expected timeline and the Companys commitment to the process.
On April 15, 2021, Weil and certain other legal counsel to the Company began their legal due diligence review of Mirion on behalf of the Company.
From April 19, 2021 through June 11, 2021, representatives of the Company, Mirion, and Goldman Sachs Investment Banking Division hosted numerous discussions with potential investors regarding the PIPE Investment. During such time period, representatives of the Company, Mirion and certain of their advisors, acting at the direction of the Company and Mirion, respectively, had a number of calls to discuss, among other things, the terms and status of the potential PIPE Investment and the composition of the investor base to participate in the PIPE Investment. On April 29, 2021, the Company held a meeting with its Board of Directors. Mr. Knott provided an update to Board regarding diligence findings, preliminary feedback from investors meetings as well as diligence items under review.
From May 1, 2021 to May 15, 2021, Messrs. Knott and Kingsley discussed with management of Mirion profits interest to enable participation in Founder Shares in connection with the potential business combination. The terms of these grants were discussed extensively between Weil and counsel to Messrs. Logan, Kingsley and Schopfer on May 8 and May 9 and then until the signing of the Business Combination Agreement.
179
Table of Contents
On May 4, 2021, the exclusivity period expired and the parties commenced negotiation of an updated term sheet without exclusivity.
On May 11, 2021, Davis Polk & Wardwell LLP, counsel to Mirion (Davis Polk) sent an initial draft Business Combination Agreement to Weil.
On May 13, 2021, the Company held a Board of Directors meeting. Mr. Knott discussed the exploration of exclusivity, current transaction dynamics, investor feedback and ongoing diligence items.
Between May 10 and May 20, 2021, representatives of Weil and Mirion held telephonic discussions regarding due diligence of certain sales channel partners of Mirion and developed a work plan in respect of such due diligence, including engaging third parties to assist in such due diligence.
On May 17, 2021, Weil provided a revised draft of the Business Combination Agreement to Davis Polk. In this draft the following material changes to the draft provided by Davis Polk were proposed: (i) the definition of Material Adverse Effect was revised to remove certain elements of the definition, and included certain carve-outs to other elements; (ii) inclusion of a right of GSAH to participate in decisions with respect to, and having certain consent rights over, certain pre-Closing restructuring transactions; (iii) cooperation amongst the parties with respect to obtaining debt financing; (iv) removal of the concept of a classified board of GSAH following the Closing; (v) the removal of certain actions required by GSAH in connection with certain regulatory filings; (vi) the scope of the representations and warranties and covenants of the parties; (vii) the inclusion of an exclusivity provision pursuant to which all parties would be bound until the Closing or termination of the Business Combination Agreement; and (viii) revisions to the Closing bring down of Mirions representations and warranties made under the Business Combination Agreement, as well as inclusion of a bring down of Seller representations and warranties.
On May 28, 2021 Weil, Davis Polk and Sullivan & Cromwell LLP (S&C), legal advisor to Goldman Sachs & Co. LLC as lead placement agent, signed off on the form of PIPE Subscription Agreement which was subsequently uploaded to the data room that was set up specifically for the PIPE investors.
On June 1, 2021, Davis Polk provided a revised draft of the Business Combination Agreement to Weil generally rejecting the changes in the May 17, 2021 draft provided by Weil, but accepting the inclusion of the exclusivity provision, a covenant regarding the termination of loans between Mirion and certain officers and directors, and the bring down of Seller representations and warranties. Additionally, in this draft the following material changes to the draft provided by Weil were proposed: (i) removal of a Closing deliverable related to the termination of certain outstanding employee loans; and (ii) the Outside Date was revised from October 31, 2021 to December 31, 2021.
From June 4, 2021 to June 12, 2021, the parties and their advisors exchanged various drafts of the Business Combination Agreement and related transaction documents. Significant areas of discussion and negotiation included: (i) the level of conditionality in the Business Combination Agreement; (ii) the allocation of transaction expenses related to the Transactions, (iii) the scope of the representations and warranties and covenants of the parties, (iv) certain interim operating covenants relating to the operation of Mirions business in between signing and Closing, and (v) the Outside Date, which was ultimately agreed to be November 30, 2021, subject to extension to January 31, 2022 and March 31, 2022 in the event that certain regulatory approvals are not obtained.
On June 5, 2021, Weil provided a consolidated set of disclosure schedules with respect to the Business Combination Agreement to Davis Polk.
On June 6, 2021, Davis Polk provided a consolidated set of disclosure schedules with respect to the Business Combination Agreement to Weil.
180
Table of Contents
On June 7, 2021, the Company held a Board of Directors meeting and Mr. Knott discussed ongoing negotiations with Charterhouse, investor feedback and remaining open items related to the transaction.
On June 9, 2021, the Board held a call to discuss the status of the transaction, open items and the Boards fiduciary duties. Representatives from Weil participated in this call.
From June 8, 2021 to June 16, 2021, representatives of the Company had multiple discussions with representatives of Charterhouse regarding the PIPE capital raise, process related items, segment level financials and several topics related to the Business Combination Agreement.
On June 12, Weil provided an initial draft of the Backstop Agreement which provided the Company with the right to require GSAM Holdings to fund up to $125,000,000 at the Closing in the event that our redemptions were to exceed $250,000,000. Such requirement of GSAM Holdings to fund this amount would be subject to, among other conditions: (i) the Transaction being consummated immediately following the purchase of the backstop shares, (ii) GSAH redemptions being greater than $250,000,000, and (iii) the minimum cash condition being waived by Mirion and the Charterhouse Parties.
On June 13, Davis Polk provided a revised draft of the Backstop Agreement to Weil. In this draft, the following material changes to the draft provided to Davis Polk were proposed: (i) a number of new representations and warranties regarding knowledge, skill and investigation of GSAM Holdings were added and (ii) the conditions set forth in the immediately preceding paragraph were deleted. On the same date, Weil provided a revised draft of the Backstop Agreement to Davis Polk, reinserting the conditions that were deleted. Later that day, Davis Polk provided a further revised draft of the Backstop Agreement which deleted the conditions that were re-inserted by Weil, and also revised the agreement to provide the Company with the right to require GSAM Holdings to fund up to $125,000,000 at the Closing in the event that the Cash Shortfall amount was greater than $0.
On June 14, 2021, Weil provided a revised draft of the Business Combination Agreement to Davis Polk. In this draft the following material changes to the draft provided by Davis Polk were proposed: (i) certain obligations of Mirion were proposed, requiring Mirion to supplement existing policies, procedures and practices concerning sales channel partners; (ii) a requirement that Mirion and its subsidiaries repay all PPP Loans outstanding as of the date of the Business Combination Agreement (and not just those outstanding as of the Closing); (iii) inclusion of a condition to Closing regarding the suspension or termination of contracts with certain sales channel partners and (iv) removal of covenant requiring GSAH to arrange alternative financing in the event that the debt financing contemplated by the Business Combination Agreement becomes unavailable.
On June 15, 2021, representatives of the Company discussed the remaining open items related to the Business Combination Agreement with representatives of Mirion and Charterhouse and resolved the majority of remaining open items including: (i) certain conditions to closing and covenants with respect to compliance related matters in the Business Combination Agreement, (ii) final revisions to be made to the form of PIPE Subscription Agreement, and (iii) final revisions to be made to the form of Registration Rights Agreement.
On the afternoon of June 15, 2021, the Company held a telephonic meeting of the Board. At the meeting, representatives of the Company and Weil provided an update to the directors regarding the resolution of the final open issues in the transaction documents, subject to the Boards approval. Weil gave the Board a presentation describing their fiduciary duties in connection with evaluating a transaction, as well as a summary of the terms of the transaction and a description of certain conflicts regarding GSAHs relationships with certain related parties (See section entitled Certain Relationships and Related Person Transactions. Representatives of the Company reviewed with the Board the Companys perspective on Mirions valuation implied by the terms of the transaction and how that valuation compared to similar companies based on certain financial metrics. The relative valuation analysis was based on the enterprise value as a multiple of EBITDA of selected publicly-traded companies in the industrial technology, life science tools and healthcare equipment markets with above average growth and high-adjusted EBITDA margins. The Company identified a peer set of the following publicly traded
181
Table of Contents
companies: Badger Meter, Bruker Corporation, Halma Plc, IDEX Corporation, Keysight Technologies, Mettler Toledo, MSA Safety and Teledyne Technologies. The Company had also identified FLIR Systems as a peer, but excluded them from the analysis due to the impact on its trading multiples as a result of its pending acquisition by Teledyne Technologies. While these companies may share certain characteristics that are similar to those of Mirion, the Board recognized that no company was identical in nature to Mirion and did not assign relative weights to such companies. As a result of the relative valuation on an EV/EBITDA basis, the Company concluded that Mirions enterprise value was at a discount to most of its peers.
The Board then discussed the proposed transaction and the benefits of the proposed transaction to the Companys shareholders. Following this discussion and upon a motion duly made and seconded, the Board unanimously approved, among other things, the relationships described in Certain Relationships and Related Person Transactions, the Business Combination Agreement and the form of Subscription Agreement, in each case substantially in the form presented to the directors, and adopted a resolution recommending the Business Combination be adopted by the Companys stockholders. The Board also unanimously approved the Business Combination, the issuance of shares in the transaction and to the PIPE Investors, the amendment of the charter and bylaws of the Company in connection with the transaction and certain post-closing incentive equity arrangements and certain management compensation matters.
On June 16, Weil provided a revised draft of the Backstop Agreement to Davis Polk. In this draft, the following material changes to the draft provided to Davis Polk were proposed: (i) the Company would only have the right to require GSAM Holdings to fund up to $125,000,000 at the Closing in the event that our redemptions were to exceed $250,000,000, and (ii) various conditions were inserted, including the requirement for the minimum cash condition to be waived by Mirion and the Charterhouse Parties. On the same date, David Polk provided a revised draft of the Backstop Agreement to Weil: (i) providing the Company with the right to require GSAM Holdings to fund up to $125,000,000 at the Closing in the event that the Cash Shortfall amount was greater than $0 and, (ii) deleting the additional conditions that were inserted in the prior Weil draft.
On June 16, 2021, Davis Polk and Weil exchanged revised drafts of the disclosure schedules.
Following conclusion of the Company Board meeting, the Company, Mirion and Charterhouse and their respective counsel finalized the transaction documentation and the PIPE Investors executed their respective subscription agreements and other documentation related thereto. On the morning of June 17, 2021, the parties executed the Business Combination Agreement and other transaction documents. See The Business Combination Agreement for a discussion of the terms of the Business Combination Agreement. Promptly following the execution of such documentation, Mirion and the Company issued a joint press release announcing the transaction.
GSAHs Board of Directors Reasons for the Approval of the Business Combination
On June 15, 2021 our Board unanimously (i) approved the Business Combination Agreement and the transactions contemplated thereby, (ii) determined that the Business Combination is in the best interests of GSAH and its stockholders and (iii) recommended that GSAHs stockholders approve the Business Combination Proposal and the other proposals described herein.
In evaluating the Business Combination and making these determinations and this recommendation, our Board consulted with the Companys management and its legal counsel, financial advisor and other advisors and considered a number of factors, including, but not limited to, the factors discussed below. In light of the wide number and complexity of the factors considered in connection with its evaluation of the Business Combination, the Board did not consider it practicable to, and did not attempt to, quantify or otherwise assign relative weights to the specific factors that it considered in reaching its determination and supporting its decision. The Board viewed its decision as being based on all of the information available and the factors presented to and considered by it. In addition, individual directors may have given different weight to different factors. This explanation of the Companys reasons for the Business Combination and all other information presented in this section is
182
Table of Contents
forward-looking in nature and, therefore, should be read in light of the factors discussed under Cautionary Statement Regarding Forward-Looking Statements beginning on page [●] of this proxy statement.
Our Board considered a number of factors pertaining to the Business Combination as generally supporting its decision to approve the entry into the Business Combination Agreement and the transactions contemplated thereby, including but not limited to, the following material factors:
| | Mirions Highly Attractive Business Model. Mirion is a global leader in the design and manufacturing of radiation detection, measurement and monitoring technology, offering a broad range of products to the nuclear, medical, defense and research end markets. Mirion has driven further growth by expanding into attractive medical markets with heightened growth opportunities, adding incremental profitable and recurring products to its broad portfolio. Mirion benefits from defensible barriers to entry due to its technology leadership, rigorous regulatory requirements and demanding design & operating standards. Further, Mirions broad customer base, across the detection & measurement technology, product offering, software and end markets verticals, combined with its broad geographic reach, provides the potential for a resilient business model. |
| | Mirions Deep Relationships with a Diverse, Loyal Customer Base. Mirion has a highly diversified customer base across markets and geographies, with the top 10 customers representing approximately 30% of total customer revenue for the fiscal year ended June 30, 2020. These customers span a wide array of industries and verticals, including the detection & measurement technology, product offering, software and end markets verticals, with many of Mirions key customer relationships spanning multiple decades. Mirions customer base demonstrates high loyalty because of the collaborative working relationship between Mirion and its customers and the critical necessity of Mirions products and the high technical expertise required to produce them. |
| | Mirions Strong Expected Revenue. Mirion has revenue that is expected to be predictable, in part due to the nature of the services and products it provides, with products from the research end markets having known replacement cycles and customers from Mirions nuclear and defense end markets procuring products and builds on multi-year horizons. Mirions practice of entering into multi-year agreements with its top customers and its status as a top competitor in each of its end markets provide further stability to its business model. The Board validated Mirions leading position in each of its end markets based on market research, competitor analysis, customer checks by third party consultants and research by the Company. |
| | Mirions Experienced and Proven Management Team. The Board considered the fact that, post-Business Combination, Mirion will be led by a senior management team with significant industry experience and a proven track record of operational excellence and financial performance at Mirion. In addition, the Board considered the fact that Mr. Kingsley, with his significant leadership experience and extensive management and investment experience, including with multinational high-technology growth companies, would serve as Chairman of the Board of the post-Business Combination Mirion. |
| | Mirions Strong Balance Sheet. The Boards considered the fact that the Business Combination will provide significant primary capital and financial flexibility for Mirion to pursue value-additive growth opportunities. Mirions strong initial balance sheet combined with its financial performance and access to the public markets through the Business Combination should enable Mirion to expedite its growth strategy through both organic growth and strategic acquisitions. |
| | Other Alternatives. The Boards belief, after a thorough review of other business combination opportunities reasonably available to the Company, that the proposed Business Combination represents the best potential business combination for the Company based upon the process utilized to evaluate and assess other potential acquisition targets. |
| | Terms of the Business Combination Agreement. The Board considered the terms and conditions of the Business Combination Agreement and the transactions contemplated thereby, including each partys |
183
Table of Contents
| representations, warranties and covenants, the conditions to each partys obligation and the termination provisions as well as the strong commitment by both Mirion and the Company to complete the Transactions. |
| | Reasonableness of Aggregate Consideration. Following a review of the financial data provided to the Company, including Mirions historical financial statements and certain unaudited prospective financial information, as well as the Companys due diligence review of Mirions business and the views of the Companys consultants and financial and other advisors, the Board considered the aggregate consideration to be paid and determined that the aggregate consideration was reasonable in light of such data and financial information. |
| | Mirion Being an Attractive Target. The Board considered the fact that Mirion (i) is of a size relevant to the public marketplace, (ii) has a strong existing management team, (iii) has a significant total addressable market and growth expansion opportunities, (iv) provides an opportunity for operational improvement, and (v) would benefit from the consummation of the Transactions as a result of becoming a public company and de-leveraging, which the Board believed would improve Mirions ability to grow, including through acquisitions. |
| | Continued Ownership By Sellers. The Board considered that Mirion stockholders would be receiving a significant amount of Mirion common stock as part of its consideration following the Business Combination, including the fact that, in connection with entry into the Business Combination Agreement, Mirions senior management team delivered election agreements pursuant to which each person elected to receive one hundred percent (100%) of their consideration in Mirion common stock. The Board considered this a strong sign of Mirions Stockholders confidence in Mirion and the benefits to be realized as a result of the Business Combination. |
| | Vesting of Founder Shares. The Board considered that in connection with the Closing, the Sponsors founder shares will be subject to vesting in three equal tranches, based on the volume weighted average price of the GSAH Class A common stock being greater than or equal to $12.00, $14.00 and $16.00, respectively, per share for any 20 trading days in any 30 consecutive trading day period. Such founder shares will be forfeited to the Company for no consideration if they fail to vest within five years of the Closing. Subjecting the founder shares to such vesting provisions is an indication of our Sponsors belief in the long-term growth trajectory of Mirion, and aligns our Sponsors interests with the interests of our shareholders. |
| | The Role of the Independent Directors. The Board is comprised of a majority of independent directors who are not affiliated with our Sponsor and its affiliates, including Goldman Sachs. In connection with the Business Combination, our independent directors, Messrs. William Frist, Steven S. Reinemund and David Robinson and Ms. Martha Sullivan, evaluated the proposed terms of the Business Combination, including the Business Combination Agreement, the Related Agreements and the amendments to our current certificate of incorporation to take effect upon the completion of the Business Combination. Our independent directors evaluated and unanimously approved, as members of the Board, the Business Combination Agreement and the transactions contemplated therein, including the Business Combination. |
The Board also considered a variety of uncertainties and risks and other potentially negative factors concerning the Business Combination, including, but not limited to, the following:
| | Macroeconomic Risks. Macroeconomic uncertainty, including the potential impact of the COVID-19 pandemic, and the effects they could have on the combined companys revenues. |
| | Benefits May Not Be Achieved. The risk that the potential benefits of the Business Combination may not be fully achieved, or may not be achieved within the expected timeframe. |
| | Growth Initiatives May Not be Achieved. The risk that the growth initiatives may not be fully achieved or may not be achieved within the expected timeframe. |
184
Table of Contents
| | Reduction in Majority Shareholder Ownership. The risk of investors perceiving negatively the reduction in ownership by Charterhouse of Mirion as part of the Business Combination. |
| | Indebtedness. The risk that Mirions levels of outstanding indebtedness could adversely affect its and the Companys financial condition following the closing of the Business Combination. |
| | No Third-Party Valuation. The risk that the Company did not obtain a third-party valuation or fairness opinion in connection with the Transactions. |
| | Liquidation of the Company. The risks and costs to the Company if the Business Combination is not completed, including the risk of diverting our officers and directors focus and resources from other businesses combination opportunities, which could result in the Company being unable to effect a business combination by July 2, 2022 and force the Company to liquidate and the warrants to expire worthless. |
| | Exclusivity. The fact that the Business Combination Agreement includes an exclusivity provision that prohibits the Company from soliciting other business combination proposals. |
| | Stockholder Vote. The risk that the Companys stockholders may fail to provide the respective votes necessary to effect the Business Combination. |
| | Closing Conditions. The fact that completion of the Business Combination is conditioned on the satisfaction of certain closing conditions that are not within the Companys control. |
| | Regulatory Approval. The fact that completion of the Business Combination is conditioned on obtaining regulatory approvals (both foreign and domestic) that are not within the Companys control and can take a significant amount of time to obtain, which provides the parties with the ability to extend the Outside Date and delay the consummation of the transactions contemplated by the Business Combination until certain of such approvals have been obtained. |
| | Litigation. The possibility of litigation challenging the Business Combination or that an adverse judgment granting permanent injunctive relief could indefinitely enjoin consummation of the Business Combination. |
| | Fees and Expenses. The fees and expenses associated with completing the Business Combination. |
| | Other Risks. Various other risks associated with the Business Combination, the business of the Company and the business of Mirion described under Risk Factors beginning on page [●] of this proxy statement/prospectus. |
In addition to considering the factors described above, the Board also considered that:
| | Interests of Goldman Sachs Parties and Certain Other Persons. The officer and some of the directors of the Company may have interests in the Business Combination as individuals that are in addition to, and that may be different from, the interests of the Companys stockholders (see Certain Relationships and Related Persons Transactions). Our independent directors reviewed and considered these interests during their evaluation of the Business Combination and in unanimously approving, as members of the Board, the Business Combination Agreement and the transactions contemplated therein, including the Business Combination. |
The Board concluded that the potential benefits that it expected the Company and its stockholders to achieve as a result of the Business Combination outweighed the potentially negative factors associated with the Business Combination. Accordingly, the Board unanimously determined that the Business Combination Agreement and the transactions contemplated thereby, including the Business Combination, were advisable, fair to, and in the best interests of, the Company and its stockholders.
Certain Unaudited Projected Financial Information
Mirion provided the Company with its internally prepared financial projections for each of the years in the three year period ending June 30, 2023. Mirion does not, as a matter of general practice, publicly disclose long-term
185
Table of Contents
forecasts or internal projections of their future performance, revenue, financial condition or other results. The prospective financial information was prepared solely for internal use and not with a view toward compliance with published guidelines of the SEC regarding projections or the guidelines established by the American Institute of Certified Public Accountants for preparation and presentation of prospective financial information, but, in the view of the Companys management, was prepared on a reasonable basis, reflects the best currently available estimates and judgments, and presents, to the best of managements knowledge and belief, the expected course of action and the expected future financial performance of the Company. The financial projections were requested by, and disclosed to, the Company for use as a component in its overall evaluation of Mirion, and are included in this proxy statement/prospectus because they were provided to the Board for its evaluation of the Business Combination.
The financial projections were developed by Mirion management and considered various material assumptions, set forth below:
| | The growth rate in the end markets that Mirion serves (based on a weighted-average of Mirions revenue per end market) will be in the range of 4% to 6% annually through fiscal year 2023 (such forecasted growth rate was estimated in partnership with a global consulting firm), and no significant change in end market trends; |
| | No major changes in the dynamics of the end markets that Mirion serves (e.g., no positive or negative changes in the nuclear or medical end markets globally); |
| | Spending in the Industrial and Medical end markets (excluding any COVID-19 specific activity) returns to pre-COVID-19 pandemic levels beginning in fiscal year 2022; |
| | No significant supply chain interruptions; |
| | The conversion of Mirions backlog of $766.5 million as of March 31, 2021 to revenue in accordance with contracts that Mirion has been awarded with no changes to customer-communicated project timelines, primarily in the Industrial segment; |
| | Investments in research and development for new products, such as Mirions investments in the SaaS SunCheck platform in Mirions Medical segment and investments in corporate functions primarily related to the integration of acquired companies as well as corresponding increases in finance and accounting and legal and compliance costs as a public company; |
| | Mirion maintains and expands its leading position relative to its competitors; |
| | No material acquisitions or divestitures by Mirion; |
| | No changes to foreign exchange rates from those as of December 2020; and |
| | Performance inline with Mirions historical performance and no significant changes to the general political and economic environment, including with respect to inflation. |
Mirions management, with the assistance of a global consulting firm, conducted market research and surveyed companies across Mirions end markets, although there was no specific company or group of companies that Mirions management and the consulting firm relied on. Based on the foregoing process and managements judgment, Mirions management estimated the approximate market sizes and growth rate ranges as set forth in the table below and calculated a projected growth rate range of 4% to 6% using the average of such growth rates, weighted by Mirions percentage of sales in such end markets.
| End Market |
Approximate Market Size(1) |
Estimated Growth Rate(2) |
Percentage of Sales(3) |
|||||||||
| Medical |
$ | 1.4 billion | 5 7 | % | 33 | % | ||||||
| Labs |
$ | 0.2 billion | 3 5 | % | 11 | % | ||||||
| Diversified Industrial |
$ | 0.7 billion | 3 5 | % | 17 | % | ||||||
| Nuclear |
$ | 2.0 billion | 2 4 | % | 39 | % | ||||||
| Total |
$ | 4.3 billion | 4 6 | % | 100 | % | ||||||
186
Table of Contents
| (1) | Market size for CY 2026. |
| (2) | Represents CY 2020 to CY 2026. |
| (3) | Estimated based on projections for prospective adjusted revenue for fiscal year 2021. |
The foregoing estimates and assumptions with respect to general business, economic, regulatory, market and financial conditions and other future events, as well as matters specific to Mirions business, are inherently uncertain, difficult to predict and are beyond Mirions and the Companys control. The inclusion of financial projections in this proxy statement/prospectus should not be regarded as an indication that the Company, our Board, or their respective affiliates, advisors or other representatives considered, or now considers, such financial projections necessarily to be predictive of actual future results or to support or fail to support your decision whether to vote for or against the Business Combination Proposal. The financial projections are not fact and should not be relied upon as being necessarily indicative of future results, and readers of this proxy statement/prospectus, including investors or holders, are cautioned not to place undue reliance on this information. You are cautioned not to rely on the financial projections in making a decision regarding the transaction, as the financial projections may be materially different than actual results. We will not refer back to the financial projections in our future periodic reports filed under the Exchange Act.
The financial projections are forward looking statements that are inherently subject significant uncertainties and contingencies, many of which are beyond Mirions and the Companys control. The various risks and uncertainties include those set forth in the Risk Factors, Mirions Managements Discussion and Analysis of Financial Condition and Results of Operations and Cautionary Statement Regarding Forward-Looking Statements sections of this proxy statement/prospectus, respectively. As a result, there can be no assurance that the financial projections described below will be realized or that actual results will not be significantly higher or lower than projected. Since the financial projections cover multiple years, such information by its nature becomes less reliable with each successive year. These financial projections are subjective in many respects and thus are susceptible to multiple interpretations and periodic revisions based on actual experience and business developments.
The financial projections included in this proxy statement/prospectus have been prepared by, and are the responsibility of, Mirions management. Neither PricewaterhouseCoopers LLP nor Deloitte & Touche LLP has audited, reviewed, examined, compiled nor applied any procedures with respect to the accompanying financial projections and accordingly, neither PricewaterhouseCoopers LLP nor Deloitte & Touche LLP expresses an opinion or any other form of assurance with respect thereto, and assume no responsibility for, and disclaim any association with, the prospective financial information. The PricewaterhouseCoopers LLP report included in this proxy statement/prospectus relates to the Companys previously issued financial statements and the Deloitte & Touche LLP report included in this proxy statement/prospectus relates to Mirions previously issued financial statements. Such reports do not extend to the financial projections and should not be read to do so.
Furthermore, the financial projections do not take into account any circumstances or events occurring after the date they were prepared, which was in December 2020. None of Mirions independent registered accounting firm, the Companys independent registered accounting firm or any other independent accountants, have compiled, examined or performed any procedures with respect to the financial projections included below, nor have they expressed any opinion or any other form of assurance on such information or their achievability, and they assume no responsibility for, and disclaim any association with, the financial projections. Nonetheless, a summary of the financial projections is provided in this proxy statement/prospectus because they were made available to the Company and our Board in connection with their review of the proposed Business Combination.
EXCEPT TO THE EXTENT REQUIRED BY APPLICABLE FEDERAL SECURITIES LAWS, BY INCLUDING IN THIS PROXY STATEMENT/PROSPECTUS A SUMMARY OF THE FINANCIAL PROJECTIONS FOR MIRION, THE COMPANY UNDERTAKES NO OBLIGATIONS AND EXPRESSLY DISCLAIMS ANY RESPONSIBILITY TO UPDATE OR REVISE, OR PUBLICLY DISCLOSE ANY
187
Table of Contents
UPDATE OR REVISION TO, THESE FINANCIAL PROJECTIONS TO REFLECT CIRCUMSTANCES OR EVENTS, INCLUDING UNANTICIPATED EVENTS, THAT MAY HAVE OCCURRED OR THAT MAY OCCUR AFTER THE PREPARATION OF THESE FINANCIAL PROJECTIONS, EVEN IN THE EVENT THAT ANY OR ALL OF THE ASSUMPTIONS UNDERLYING THE FINANCIAL PROJECTIONS ARE SHOWN TO BE IN ERROR OR CHANGE.
The key elements of the projections provided by management of Mirion to the Company are summarized in the table below. The forecasts contain certain non-GAAP financial measures, including Prospective Adjusted Revenue, Prospective Adjusted EBITDA, Prospective Adjusted EBITDA less Capital Expenditures and Average Cash Conversion which are summarized below the table.
| ($ in millions) | FY 2021E | FY 2022E | FY 2023E | |||||||||
| Prospective Adjusted Revenue |
$ | 688.7 | $ | 722.6 | $ | 761.9 | ||||||
| % Organic Growth |
5.9 | % | 4.9 | % | 5.4 | % | ||||||
| Prospective Adjusted EBITDA |
172.4 | 178.7 | 205.1 | |||||||||
| % Margin |
25.0 | % | 24.7 | % | 26.9 | % | ||||||
| Prospective Adjusted EBITDA less Capital Expenditures |
140.4 | 142.4 | 168.6 | |||||||||
| % Prospective Adjusted Revenue |
20.4 | % | 19.7 | % | 22.1 | % | ||||||
| Average Cash Conversion |
95.7 | % | 94.3 | % | 95.4 | % | ||||||
Non-GAAP Financial Measures
The forecasts contain certain non-GAAP financial measures, including Prospective Adjusted Revenue, Prospective Adjusted EBITDA, Prospective Adjusted EBITDA less Capital Expenditures and Average Cash Conversion. Mirion and the Company believe these non-GAAP measures provide useful information to management and investors regarding certain financial and business trends relating to Mirions financial condition and results of operations. However, the use of non-GAAP measures instead of GAAP measures has limitations as an analytical tool, and you should not consider the non-GAAP measures in isolation, or as a substitute for, comparable measures reported under GAAP. Mirions definitions of and methods of calculating these non-GAAP financial measures vary from the definitions and methods used by other companies, which may limit their usefulness as comparative measures.
Prospective Adjusted Revenue is defined as GAAP revenue adjusted for impacts from purchase accounting, prospective adjustments from acquisitions and impact of foreign exchange conversion. Prospective adjustment from acquisitions represents all acquisitions, including those not deemed significant under Article 11, Pro Forma Financials. EBITDA is defined as net income before interest expense, income tax expense, depreciation and amortization. Average Cash Conversion is measured as Adjusted EBITDA less Maintenance Capital Expenditures divided by Adjusted EBITDA. These measures are not measurements of Mirions financial performance under GAAP and should not be considered in isolation or as alternatives to net income, net cash flows provided by operating activities, total net cash flows or any other performance measures derived in accordance with GAAP or as alternatives to net cash flows from operating activities or total net cash flows as measures of Mirions operating activity. Management does not consider EBITDA in isolation or as alternatives to financial measures determined in accordance with GAAP.
Set forth below are reconciliations of Prospective Adjusted Revenue and Prospective Adjusted EBITDA to GAAP revenue and net income, in each case the most directly comparable GAAP measures. In addition, set forth below is a calculation of cash conversion.
188
Table of Contents
Reconciliation of Prospective Adjusted Revenue to Revenue
The following table sets forth the reconciliation of Prospective Adjusted Revenue to GAAP revenue, the most directly comparable GAAP measure.
| ($ in millions) | FY2021E | FY2022E | FY2023E | |||||||||
| Revenue (GAAP basis) |
$ | 616.6 | $ | 715.8 | $ | 761.9 | ||||||
| Deferred revenue purchase accounting adjustments |
8.0 | 6.8 | | |||||||||
| Prospective adjustments from acquisitions(1) |
60.0 | | | |||||||||
| Impact of foreign exchange conversion |
4.0 | | | |||||||||
| Prospective Adjusted Revenue |
$ | 688.7 | $ | 722.6 | $ | 761.9 | ||||||
| (1) | Includes Biodex, Dosimetrics and Sun Nuclear acquisitions. |
Reconciliation of Prospective Adjusted EBITDA to Net Income (Loss)
The following table sets forth the reconciliation of Prospective Adjusted EBITDA to Net income (loss), the most directly comparable GAAP measure.
| ($ in millions) | FY2021E | FY2022E | FY2023E | |||||||||
| Net income (loss) (GAAP) |
$ | (146.9 | ) | $ | 37.0 | $ | 82.7 | |||||
| Minority interest |
(0.0 | ) | | | ||||||||
| Income taxes |
(10.3 | ) | 13.0 | 29.1 | ||||||||
| Other (income) / expense |
(0.3 | ) | | | ||||||||
| Loss on debt extinguishment |
| | | |||||||||
| Foreign currency (gain) loss, net |
16.3 | | | |||||||||
| Net interest expense(1) |
165.5 | 17.1 | 15.2 | |||||||||
| Amortization of acquired intangibles |
60.8 | 62.5 | 55.1 | |||||||||
| Depreciation |
21.6 | 21.3 | 15.1 | |||||||||
| Stock-based compensation |
0.2 | | | |||||||||
| Other non-operating costs |
44.3 | 27.7 | 10.3 | |||||||||
| Other adjustments |
0.3 | 0.0 | (2.6 | ) | ||||||||
| Adjusted EBITDA (Before Prospective Adjustment) |
$ | 151.5 | $ | 178.7 | $ | 205.1 | ||||||
| Prospective adjustments from acquisitions |
20.9 | | | |||||||||
| Prospective adjusted EBITDA |
$ | 172.4 | $ | 178.7 | $ | 205.1 | ||||||
| Prospective adjustments from acquisitions |
||||||||||||
| Acquisitions(2) |
$ | 19.2 | | | ||||||||
| Foreign currency impact from acquisitions |
1.7 | | | |||||||||
| Total prospective adjustments from acquisitions |
$ | 20.9 | $ | 0.0 | $ | 0.0 | ||||||
Source: Mirion management
| (1) | FY 2021E net interest expense includes non-cash interest expense related to PIK interest. |
| (2) | Includes Biodex, Dosimetrics and Sun Nuclear acquisitions. |
Average Cash Conversion
The following table sets forth the calculations used to derive Average Cash Conversion, which is calculated as Adjusted EBITDA less Maintenance Capital Expenditures divided by Adjusted EBITDA.
| Average Cash Conversion | ||||||||||||
| ($ in millions) | FY2021 | FY2022 | FY2023 | |||||||||
| Adjusted EBITDA |
$ | 151.5 | $ | 178.7 | $ | 205.1 | ||||||
| (-) Maintenance Capital Expenditures |
(6.5 | ) | (10.3 | ) | (9.4 | ) | ||||||
| Adjusted EBITDA Less Maintenance CapEx |
$ | 145.0 | $ | 168.4 | $ | 195.7 | ||||||
| Cash Conversion |
95.7 | % | 94.3 | % | 95.4 | % | ||||||
189
Table of Contents
Satisfaction of 80% Test
The NYSE rules require that our initial business combination must be with one or more operating businesses or assets with a fair market value equal to at least 80% of the net assets held in the trust account (net of amounts disbursed to management for working capital purposes, if permitted, and excluding the amount of any deferred underwriting discount). Based on the financial analysis of Mirion generally used to approve the transaction, our Board determined that this requirement was met. Our Board determined that the consideration being paid in the Business Combination, which amount was negotiated at arms-length, was fair to and in the best interests of GSAH and its stockholders and appropriately reflected Mirions value. In reaching this determination, our Board concluded that it was appropriate to base such valuation in part on qualitative factors such as management strength and depth, competitive positioning, customer relationships, and technical skills, as well as quantitative factors such as its potential for future growth in revenue and profits. Our Board believes that the financial skills and background of its members qualify it to conclude that the acquisition of Mirion met this requirement.
Interests of Goldman Sachs Parties and Certain Other Persons
When you consider the recommendation of our Board in favor of approval of the Business Combination Proposal and the other proposals included herein, you should keep in mind that the Sponsor and our directors have interests in such proposal that are different from, or in addition to, those of our stockholders and warrant holders generally. Our Board was aware of and considered these interests, among other matters, in evaluating and negotiating the Business Combination and transaction agreements and in recommending to our stockholders that they vote in favor of the proposals presented at the Special Meeting, including the Business Combination Proposal. GSAH stockholders should take these interests into account in deciding whether to approve the proposals presented at the Special Meeting, including the Business Combination Proposal. These interests include, among other things:
| | If we do not consummate a business combination by July 2, 2022 (or if such date is extended at a duly called meeting of our stockholders, such later date), we would (1) cease all operations except for the purpose of winding up; (2) as promptly as reasonably possible but not more than ten business days thereafter, redeem the public shares, at a per share price, payable in cash, equal to the aggregate amount then on deposit in the trust account, including interest (which interest shall be net of taxes payable, and less up to $100,000 of interest to pay dissolution expenses), divided by the number of then outstanding public shares, which redemption will completely extinguish public stockholders rights as stockholders (including the right to receive further liquidating distributions, if any); and (3) as promptly as reasonably possible following such redemption, subject to the approval of our remaining stockholders and our Board, dissolve and liquidate, subject in each case to our obligations under Delaware law to provide for claims of creditors and the requirements of other applicable law. In such event, the 18,750,000 shares of Class B common stock owned by our Initial Stockholders, including the Sponsor, would be worthless because following the redemption of the public shares, we would likely have few, if any, net assets and because the Sponsor and each of our officers and directors have agreed to waive their rights to liquidating distributions from the trust account with respect to such shares if we fail to complete a business combination within the required period. Additionally, in such event, the 8,500,000 private placement warrants that the Sponsor paid $17 million for will expire worthless. The [-] shares of Class A common stock that the Initial Stockholders will hold following the Business Combination, if unrestricted and freely tradable, would have had aggregate market value of approximately $[-] million based upon the closing price of $[-] per share of Class A common stock on the NYSE on [-], 2021, the most recent practicable date prior to the date of this proxy statement. Given such shares of our common stock will be subject to certain restrictions, we believe such shares have less value. The [-] private placement warrants that the Sponsor will hold following the Business Combination, if unrestricted and freely tradable, would have had an aggregate market value of approximately $[-] million based upon the closing price of $[-] per warrant on the NYSE on [-], 2021, the most recent practicable date prior to the date of this proxy statement. The following table sets forth the implied ownership levels by and |
190
Table of Contents
| returns to the GSAH stockholders (including the Sponsor) at various prices based on the assumptions described under Summary of the Proxy Statement / ProspectusOwnership of the Company Following the Business Combination. |
Implied Ownership and Returns at Various Prices(1)
| Share Price: |
$ | 6.00 | $ | 8.00 | $ | 10.00 | $ | 12.00 | $ | 14.00 | $ | 16.00 | $ | 18.00 | $ | 20.00 | ||||||||||||||||
| Public Shares |
75 | 75 | 75 | 75 | 75 | 75 | 75 | 75 | ||||||||||||||||||||||||
| Public Warrants |
| | | 1 | 3 | 5 | 7 | 7 | ||||||||||||||||||||||||
| Founder Shares(2) |
| | | 6 | 13 | 19 | 19 | 19 | ||||||||||||||||||||||||
| Private Placement Warrants |
| | | 0 | 2 | 2 | 3 | 4 | ||||||||||||||||||||||||
| PIPE Investors |
90 | 90 | 90 | 90 | 90 | 90 | 90 | 90 | ||||||||||||||||||||||||
| Previous Owners and Management Rollover Equity |
39 | 39 | 39 | 39 | 39 | 39 | 39 | 39 | ||||||||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||||
| Post-Money Equity Value |
$ | 1,224 | $ | 1,632 | $ | 2,040 | $ | 2,537 | $ | 3,099 | $ | 3,687 | $ | 4,187 | $ | 4,663 | ||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||||
| Implied Returns ($ mm): |
||||||||||||||||||||||||||||||||
| Illustrative IPO Investor 1-Year Return (%)(3),(4) |
(40 | )% | (20 | )% | 0 | % | 21 | % | 46 | % | 71 | % | 96 | % | 118 | % | ||||||||||||||||
| Illustrative PIPE Investor 1-Year Return (%)(3) |
(40 | )% | (20 | )% | 0 | % | 20 | % | 40 | % | 60 | % | 80 | % | 100 | % | ||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||||
| Sponsor Gain ($) (excl. PIPE Investment)(2),(5) |
$ | (17 | ) | $ | (17 | ) | $ | (17 | ) | $ | 62 | $ | 179 | $ | 321 | $ | 376 | $ | 430 | |||||||||||||
| Illustrative Founder 1-Year Return (%) (excl. PIPE Investment)(2),(5) |
(100 | )% | (100 | )% | (100 | )% |
|
366 |
% |
1054 | % | 1890 | % | 2210 | % | 2531 | % | |||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||||
| Sponsor Gain ($) (incl. PIPE Investment)(2),(6) |
$ | (97 | ) | $ | (57 | ) | $ | (17 | ) | $ | 102 | $ | 259 | $ | 441 | $ | 536 | $ | 630 | |||||||||||||
| Illustrative Sponsor 1-Year Return (%) (incl. PIPE Investment)(2),(6) |
(45 | )% | (26 | )% | (8 | )% | 47 | % | 119 | % | 203 | % | 247 | % | 290 | % | ||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||||
| Implied Ownership: |
$ | 6.00 | $ | 8.00 | $ | 10.00 | $ | 12.00 | $ | 14.00 | $ | 16.00 | $ | 18.00 | $ | 20.00 | ||||||||||||||||
| Public Stockholders |
36.8 | % | 36.8 | % | 36.8 | % | 35.8 | % | 35.4 | % | 34.8 | % | 35.2 | % | 35.1 | % | ||||||||||||||||
| Sponsor (excl. PIPE Investment)(2) |
| | | 3.1 | 6.3 | 9.2 | 9.4 | 9.6 | ||||||||||||||||||||||||
| PIPE Investors |
44.1 | 44.1 | 44.1 | 42.6 | 40.7 | 39.1 | 38.7 | 38.6 | ||||||||||||||||||||||||
| of which is Founder PIPE Investment |
9.8 | 9.8 | 9.8 | 9.5 | 9.0 | 8.7 | 8.6 | 8.6 | ||||||||||||||||||||||||
| Previous Owners and Management(7) |
19.1 | 19.1 | 19.1 | 18.4 | 17.6 | 16.9 | 16.8 | 16.7 | ||||||||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||||
| Total |
100.0 | % | 100.0 | % | 100.0 | % | 100.0 | % | 100.0 | % | 100.0 | % | 100.0 | % | 100.0 | % | ||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||||
| Implied Dilution from Founder Shares and Private Placement Warrants |
0.0 | % | 0.0 | % | 0.0 | % | 3.1 | % | 6.3 | % | 9.2 | % | 9.4 | % | 9.6 | % | ||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
| | In conjunction with the Business Combination Agreement, the Sponsor issued 3,200,000 membership interests to Thomas Logan, the Chief Executive Officer of Mirion, 700,000 membership interests to Brian Schopfer, the Chief Financial Officer of Mirion, and 4,200,000 membership interests to Lawrence Kingsley, who is expected to be Chairman of the Board of New Mirion. The Profits Interests are intended to be treated as profits interests for U.S. income tax purposes, pursuant to which Messrs. Logan, Schopfer and Kingsley will have an indirect interest in the founder shares held by the Sponsor. The Profits Interests are subject to service and performance vesting conditions, including the occurrence of the Closing, and do not fully vest until all of the applicable conditions are satisfied. In addition, the Profits Interests are subject to certain forfeiture conditions. |
191
Table of Contents
| (1) | Does not contemplate any incentive awards under the Equity Incentive Plan as the number of awards and terms of any such awards are not yet known. |
| (2) | The founder shares will be subject to vesting after the Closing in three equal tranches, based on the volume weighted average price of the GSAH Class A common stock being greater than or equal to $12.00, $14.00 and $16.00, respectively, per share for any 20 trading days in any 30 consecutive trading day period. Such founder shares will be forfeited to the Company for no consideration if they fail to vest within five years of the Closing. Includes portion of founders shares allocated to Lawrence Kingsley and members of Mirion management. |
| (3) | Assumes investor entry price of $10/share. |
| (4) | Includes public common shares and public warrants. |
| (5) | Assumes at risk capital of $17mm. |
| (6) | Assumes PIPE Investment of $200mm and at risk capital of $17mm. |
| (7) | Founder shares portion of management ownership (see footnote 2) not included in management ownership. |
| | Our existing management team members and directors will be eligible for continued indemnification and continued coverage under a directors and officers liability insurance policy after the Business Combination and pursuant to the Business Combination Agreement. |
| | In order to protect the amounts held in the trust account, the Sponsor has agreed that it will be liable to us if and to the extent any claims by a third party (other than our independent registered public accounting firm) for services rendered or products sold to us, or a prospective target business with which we have discussed entering into a transaction agreement, reduce the amount of funds in the trust account to below: (1) $10.00 per public share; or (2) such lesser amount per public share held in the trust account as of the date of the liquidation of the trust account due to reductions in the value of the trust assets, in each case, net of the interest which may be withdrawn to pay taxes, except as to any claims by a third party who executed a waiver of any and all rights to seek access to the trust account and except as to any claims under our indemnity of the underwriters of our IPO against certain liabilities, including liabilities under the Securities Act. |
| | Upon completion of the Business Combination, an aggregate amount of approximately $33,000,000 in deferred underwriting discount, advisory fees and placement agent fees, will be payable to Goldman Sachs & Co. LLC, an affiliate of us and the Sponsor. In addition, Goldman Sachs will receive a committed financing fee of $18,400,000 in connection with the Debt Financing. |
| | Tom Knott, our Chief Executive Officer, Chief Financial Officer and Secretary, is a Managing Director of Goldman Sachs and Raanan A. Agus, one of our directors, is a Participating Managing Director of Goldman Sachs. |
| | Goldman Sachs Private Credit Funds, affiliates of GSAH, are a current lender to Mirion, holding $137.6mm of the USD Term Loan and 122.8mm of the EUR Term Loan under the Mirion Credit Agreement. GSAH intends to use a portion of the proceeds from the Business Combination, including the PIPE Investment, to repay the outstanding Mirion Credit Agreement and, as a result, such affiliates would receive their pro rata portion of such proceeds. |
| | GSAM Holdings has subscribed for $200 million of the PIPE Investment, for which it will receive up to 20 million shares of our Class A common stock, unless it chooses to syndicate such subscription prior to Closing. See Proposal No. 1Approval of the Business CombinationRelated Agreements Subscription Agreements. |
| | GSAM Holdings and GSAH have entered into the Backstop Agreement pursuant to which GSAM Holdings has committed to purchase from GSAH up to 12,500,000 shares of GSAHs Class A common stock at a price per share equal to $10.00 immediately prior to (and contingent upon) the Closing, contingent upon the terms and subject to the conditions set forth in the Backstop Agreement. For additional information, see Proposal No. 1Approval of the Business CombinationRelated AgreementsBackstop Agreement. |
192
Table of Contents
| | GSAM Holdings and Sellers that elect to receive cash for their Existing Mirion Shares at Closing will enter into the Option Agreement, pursuant to which such Sellers will agree to, at the option of GSAM Holdings and subject to the conditions set forth in the Option Agreement, sell to GSAM Holdings up to 12,500,000 shares of the GSAH Class A common stock to be received by such Sellers pursuant to the Business Combination Agreement at the Closing at a price per share equal to $10.00 in cash, on the terms and subject to the conditions set forth in the Option Agreement, see Proposal No. 1Approval of the Business CombinationRelated AgreementsOption Agreement. |
| | Pursuant to the Amended and Restated Registration Rights Agreement, the Sponsor, GS Employee Participation and GSAM Holdings will have customary registration rights, including demand and piggy-back rights, subject to cooperation and cut-back provisions with respect to the shares of Class A common stock and warrants held by such parties. See Proposal No. 1Approval of the Business CombinationRelated Agreements Amended and Restated Registration Rights Agreement. |
| | The GS Director Nomination Agreement will grant the Sponsor the ongoing right (but not the obligation) to appoint or nominate to the Board of Directors two (2) individuals, or the GS Sponsor Directors, to serve as director of New Mirion. |
| | The Sponsor and GS Employee Participation have agreed to, among other things, vote in favor of the Business Combination Proposal and the other proposals described herein to be presented at the Special Meeting. See Proposal No. 1Approval of the Business CombinationRelated Agreements Amended and Restated Sponsor Agreement. As of the date of this proxy statement, the Initial Stockholders own approximately 20% of the outstanding shares of our common stock. |
The existence of financial and personal interests of one or more of GSAHs directors may result in a conflict of interest on the part of such director(s) between what he or they may believe is in the best interests of GSAH and its stockholders and what he or they may believe is best for himself or themselves in determining to recommend that stockholders vote for the proposals.
At any time at or prior to the Business Combination, subject to applicable securities laws (including with respect to material nonpublic information), the Sponsor, the Mirion Stockholder or our or their respective directors, officers, advisors or respective affiliates may (1) purchase public shares from institutional and other investors who vote, or indicate an intention to vote, against any of the Condition Precedent Proposals, or elect to redeem, or indicate an intention to redeem, public shares, (2) execute agreements to purchase such shares from such investors in the future, or (3) enter into transactions with such investors and others to provide them with incentives to acquire public shares, vote their public shares in favor of the Condition Precedent Proposals or not redeem their public shares. Such a purchase may include a contractual acknowledgement that such stockholder, although still the record holder of GSAHs shares, is no longer the beneficial owner thereof and therefore agrees not to exercise its redemption rights. In the event that the Sponsor, the Mirion stockholders or our or their respective directors, officers, advisors or respective affiliates purchase shares in privately negotiated transactions from public stockholders who have already elected to exercise their redemption rights, such selling stockholder would be required to revoke their prior elections to redeem their shares. The purpose of such share purchases and other transactions would be to (1) increase the likelihood of approving the Condition Precedent Proposals and (2) limit the number of public shares electing to redeem, including to satisfy any redemption threshold.
Entering into any such arrangements may have a depressive effect on our common stock (e.g., by giving an investor or holder the ability to effectively purchase shares at a price lower than market, such investor or holder may therefore become more likely to sell the shares he or she owns, either at or prior to the Business Combination). If such transactions are effected, the consequence could be to cause the Business Combination to be consummated in circumstances where such consummation could not otherwise occur. Purchases of shares by the persons described above would allow them to exert more influence over the approval of the proposals to be presented at the Special Meeting and would likely increase the chances that such proposals would be approved. We will file or submit a Current Report on Form 8-K to disclose any material arrangements entered into or significant purchases made by any of the aforementioned persons that would affect the vote on the proposals to be put to the Special Meeting or the redemption threshold. Any such report will include descriptions of any arrangements entered into or significant purchases by any of the aforementioned persons.
193
Table of Contents
The existence of financial and personal interests of one or more of GSAHs directors may result in a conflict of interest on the part of such director(s) between what he or they may believe is in the best interests of GSAH and its stockholders and what he or they may believe is best for himself or themselves in determining to recommend that stockholders vote for the proposals.
Appraisal Rights
Neither our stockholders nor our warrant holders have appraisal rights in connection with the Business Combination under the DGCL.
Expected Accounting Treatment
The Business Combination is accounted for under the scope of Financial Accounting Standards Boards Accounting Standards Codification (ASC) Topic 805, Business Combinations (ASC 805). Pursuant to ASC 805, GSAH has been determined to be the accounting acquirer. Mirion constitutes a business in accordance with ASC 805 and the business combination constitutes a change in control. Accordingly, the business combination will be accounted for using the acquisition method. Under this method of accounting, Mirion will be treated as the acquired company for financial reporting purposes and the net assets of the post-business combination company will be stated at fair value, with goodwill or other intangible assets recorded.
United States Federal Income Tax Considerations to Stockholders Exercising Redemption Rights
The following discussion is a summary of certain U.S. federal income tax considerations generally applicable to holders of GSAH Class A common stock (including holders of units that elect to separate such units in order to exercise redemption rights) that elect to have their Class A common stock redeemed for cash if the Business Combination is not completed. This discussion applies only to Class A common stock that is held as a capital asset within the meaning of Section 1221 of the Code and the Treasury regulations thereunder (Treasury Regulations) (generally, property held for investment). This discussion does not address all of the U.S. federal income tax consequences that may be relevant to the holders or prospective holders of Class A common stock in light of their particular circumstances, including the alternative minimum tax, the Medicare contribution tax on net investment income and the different consequences that may apply to holders of Class A common stock that are subject to special treatment under the Code, such as:
| | banks and financial institutions; |
| | insurance companies; |
| | brokers and dealers in securities, currencies or commodities; |
| | dealers or traders in securities subject to a mark-to-market method of tax accounting with respect to shares of GSAH Class A common stock; |
| | individual retirement and other deferred accounts; |
| | real estate investment trusts; |
| | regulated investment companies; |
| | controlled foreign corporations; |
| | passive foreign investment companies; |
| | governmental organizations and qualified foreign pension funds; |
| | persons holding Class A common stock as part of a straddle, hedge, conversion transaction, synthetic security, constructive sale transaction, integrated transaction or similar transaction; |
| | persons holding (directly, indirectly or constructively) 5% or more of our stock; |
194
Table of Contents
| | U.S. Holders (as defined below) whose functional currency is not the U.S. dollar; |
| | persons that receive Class A common stock in connection with services provided; |
| | investors holding Class A common stock in connection with a trade or business conducted outside of the U.S.; |
| | partnerships, S-corporations, or other pass-through entities for U.S. federal income tax purposes (and investors in such entities); |
| | certain former citizens or long-term residents of the United States; |
| | former U.S. citizens or lawful permanent residents living abroad; and |
| | tax-exempt organizations (including private foundations). |
If a partnership (or other entity or arrangement treated as a partnership for U.S. federal income tax purposes) holds shares of GSAH Class A common stock, the U.S. federal income tax treatment of the partners in the partnership will generally depend on the status of the partners and the activities of the partnership. Partnerships and partners in partnerships holding shares of GSAH Class A common stock should consult their tax advisors regarding the U.S. federal income tax consequences to them.
This discussion is based on the Code and administrative pronouncements, judicial decisions and final, temporary and proposed Treasury Regulations as of the date hereof, changes to any of which subsequent to the date of this proxy statement/prospectus may affect the tax consequences described herein. This discussion does not address any aspect of state, local or non-U.S. taxation, or any U.S. federal taxes other than income taxes (such as gift and estate taxes). No assurance can be given that the Internal Revenue Service (the IRS) would not assert, or that a court would not sustain, a position contrary to any of the tax considerations described below. No advance ruling has been or will be sought from the IRS regarding any matter discussed in this summary.
THE U.S. FEDERAL INCOME TAX TREATMENT OF THE TRANSACTIONS DISCUSSED HEREIN TO ANY PARTICULAR STOCKHOLDER WILL DEPEND ON THE STOCKHOLDERS PARTICULAR TAX CIRCUMSTANCES. YOU ARE URGED TO CONSULT YOUR TAX ADVISOR REGARDING THE U.S. FEDERAL, STATE, LOCAL, AND NON-U.S. INCOME AND OTHER TAX CONSEQUENCES TO YOU, IN LIGHT OF YOUR PARTICULAR INVESTMENT OR TAX CIRCUMSTANCES, OF A REDEMPTION OF YOUR CLASS A COMMON STOCK
Redemption of GSAH Class A Common Stock
In the event that a stockholders shares of GSAH Class A common stock are redeemed pursuant to the redemption provisions described in this proxy statement/prospectus under the section entitled Special Meeting of GSAH StockholdersRedemption Rights, the treatment of the redemption for U.S. federal income tax purposes will depend on whether the redemption qualifies as a sale (or other taxable exchange) of shares under Section 302 of the Code. If the redemption qualifies as a sale of shares, a U.S. Holder (as defined below) will be treated as described below under the section entitled U.S. HoldersGain or Loss on Sale, Taxable Exchange or Other Taxable Disposition of GSAH Class A Common Stock, and a Non-U.S. Holder (as defined below) will be treated as described under the section entitled Non-U.S. HoldersGain on Sale, Taxable Exchange or Other Taxable Disposition of GSAH Class A Common Stock. If the redemption does not qualify as a sale of shares, a stockholder will be treated as receiving a corporate distribution with the tax consequences to a U.S. Holder described below under the section entitled U.S. HoldersTaxation of Distributions, and the tax consequences to a Non-U.S. Holder described below under the section entitled Non-U.S. HoldersTaxation of Distributions.
Whether a redemption of shares of GSAH Class A common stock qualifies for sale treatment will depend largely on the total number of shares of our stock treated as held by the redeemed stockholder before and after the
195
Table of Contents
redemption (including any stock constructively owned by the stockholder as a result of owning warrants and any of our stock that a stockholder would directly or indirectly acquire pursuant to the Business Combination or PIPE Investment) relative to all of our shares outstanding both before and after the redemption. The redemption of GSAH Class A common stock generally will be treated as a sale of stock (rather than as a corporate distribution) if the redemption (i) is substantially disproportionate with respect to the stockholder, (ii) results in a complete termination of the stockholders interest in us or (iii) is not essentially equivalent to a dividend with respect to the stockholder. These tests are explained more fully below.
In determining whether any of the foregoing tests result in a redemption qualifying for sale treatment, a stockholder takes into account not only shares of our stock actually owned by the stockholder, but also shares of our stock that are constructively owned by it under certain attribution rules set forth in the Code. A stockholder may constructively own, in addition to stock owned directly, stock owned by certain related individuals and entities in which the stockholder has an interest or that have an interest in such stockholder, as well as any stock that the stockholder has a right to acquire by exercise of an option, which would generally include Class A common stock which could be acquired pursuant to the exercise of the warrants. Moreover, any of our stock that a stockholder directly or constructively acquires pursuant to the Business Combination or the PIPE Investment should be included in determining the U.S. federal income tax treatment of the redemption.
In order to meet the substantially disproportionate test, the percentage of our outstanding voting stock actually and constructively owned by the stockholder immediately following the redemption of shares of GSAH Class A common stock must, among other requirements, be less than eighty percent (80%) of the percentage of our outstanding voting stock actually and constructively owned by the stockholder immediately before the redemption (taking into account both redemptions by other holders of GSAH Class A common stock and the Class A common stock to be issued pursuant to the Business Combination and the PIPE Investment) and such stockholder immediately following the redemptions must actually and constructively own less than 50 percent of our total combined voting power. Because holders of GSAH Class A common stock are not entitled to elect directors until after the completion of the Business Combination (and are not voting to elect directors at the Special Meeting pursuant to Proposal 4, as described elsewhere in this proxy statement/prospectus), the Class A common stock may not be treated as voting stock for this purpose and, consequently, this substantially disproportionate test may not be applicable to stockholders exercising redemption rights. There will be a complete termination of a stockholders interest if either (i) all of the shares of our stock actually and constructively owned by the stockholder are redeemed or (ii) all of the shares of our stock actually owned by the stockholder are redeemed and the stockholder is eligible to waive, and effectively waives in accordance with specific rules, the attribution of stock owned by certain family members and the stockholder does not constructively own any other stock (including any stock constructively owned by the stockholder as a result of owning warrants).
The redemption of GSAH Class A common stock will not be essentially equivalent to a dividend if the redemption results in a meaningful reduction of the stockholders proportionate interest in us. Whether the redemption will result in a meaningful reduction in a stockholders proportionate interest in us will depend on the particular facts and circumstances. However, the IRS has indicated in a published ruling that even a small reduction in the proportionate interest of a small minority stockholder in a publicly held corporation who exercises no control over corporate affairs may constitute such a meaningful reduction.
If none of the foregoing tests is satisfied, then the redemption of shares of GSAH Class A common stock will be treated as a corporate distribution to the redeemed stockholder, and the tax effects to a stockholder will be as described below under the section entitled U.S. HoldersTaxation of Distributions if such stockholder is a U.S. Holder or as described below under the section entitled Non-U.S. HoldersTaxation of Distributions if such stockholder is a Non-U.S. Holder. After the application of those rules, any remaining tax basis of the stockholder in the redeemed Class A common stock will be added to the stockholders adjusted tax basis in its remaining stock, or, if it has none, to the holders adjusted tax basis in its warrants or possibly in other stock constructively owned by it.
196
Table of Contents
All holders of Class A common stock should consult with their own tax advisors as to the tax consequences of a redemption to them.
U.S. Holders
This section applies to you if you are a U.S. Holder. A U.S. Holder is a beneficial owner of shares of GSAH Class A common stock who or that is, for U.S. federal income tax purposes:
| | an individual who is a citizen or resident of the United States; |
| | a corporation (or other entity taxable as a corporation for U.S. federal income tax purposes) organized in or under the laws of the United States, any state thereof or the District of Columbia; |
| | an estate the income of which is subject to U.S. federal income tax purposes regardless of its source; or |
| | a trust, if (A) a court within the United States is able to exercise primary supervision over the administration of such trust and one or more United States persons (within the meaning of the Code) have the authority to control all substantial decisions of the trust or (B) the trust validly elected to be treated as a United States person for U.S. federal income tax purposes. |
Gain or Loss on Sale, Taxable Exchange or Other Taxable Disposition of GSAH Class A Common Stock.
If our redemption of a U.S. Holders shares of GSAH Class A common stock is treated as a sale or other taxable disposition, as discussed above under the section entitled Redemption of GSAH Class A Common Stock, a U.S. Holder generally will recognize capital gain or loss in an amount equal to the difference between the amount realized and the U.S. Holders adjusted tax basis in the shares of GSAH Class A common stock treated as sold. Any such capital gain or loss generally will be long-term capital gain or loss if the U.S. Holders holding period for the GSAH Class A common stock so disposed of exceeds one year. It is unclear, however, whether the redemption rights with respect to the GSAH Class A common stock described in this proxy statement/prospectus may suspend the running of the applicable holding period for this purpose. It is unclear, however, whether the redemption rights with respect to the GSAH Class A common stock described in this proxy statement/prospectus may suspend the running of the applicable holding period for this purpose. Under tax law currently in effect, long-term capital gains recognized by non-corporate U.S. Holders will be eligible to be taxed at reduced rates. The deductibility of capital losses is subject to various limitations. U.S. Holders who hold different blocks of GSAH Class A common stock (shares of GSAH Class A common stock purchased or acquired on different dates or at different prices) should consult their tax advisors to determine how the above rules apply to them.
Taxation of Distributions.
If our redemption of a U.S. Holders shares of GSAH Class A common stock is treated as a distribution, as discussed above under the section entitled Redemption of GSAH Class A Common Stock, such distribution generally will constitute a dividend for U.S. federal income tax purposes to the extent paid from our current or accumulated earnings and profits, as determined under U.S. federal income tax principles. Distributions in excess of current and accumulated earnings and profits will constitute a return of capital that will be applied against and reduce (but not below zero) the U.S. Holders adjusted tax basis in GSAH Class A common stock. Any remaining excess will be treated as gain realized on the sale or other disposition of the Class A common stock and will be treated as described above under the section entitled Gain or Loss on Sale, Taxable Exchange or Other Taxable Disposition of GSAH Class A Common Stock.
Dividends we pay to a U.S. Holder that is a taxable corporation generally will qualify for the dividends received deduction if the requisite holding period is satisfied. With certain exceptions (including, but not limited to, dividends treated as investment income for purposes of investment interest deduction limitations), and provided certain holding period requirements are met, dividends we pay to a non-corporate U.S. Holder generally will constitute qualified dividends that will be subject to tax at the maximum tax rate accorded to long-term capital
197
Table of Contents
gains. It is unclear whether the redemption rights with respect to the Class A common stock described in this proxy statement/prospectus may prevent a U.S. Holder from satisfying the applicable holding period requirements with respect to the dividends received deduction or the preferential tax rate on qualified dividend income, as the case may be. U.S. Holders should consult their tax advisors to determine how the above rules apply to them.
Non-U.S. Holders
This section applies to you if you are a Non-U.S. Holder. A Non-U.S. Holder is a beneficial owner of GSAH Class A common stock who or that is, for U.S. federal income tax purposes:
| | a non-resident alien individual, other than certain former citizens and residents of the United States subject to U.S. tax as expatriates; |
| | a foreign corporation; or |
| | an estate or trust that is not a U.S. Holder; |
but does not include an individual who is present in the United States for 183 days or more in the taxable year of disposition of Company Class A common stock. If you are such an individual, you should consult your tax advisor regarding the U.S. federal income tax consequences of a redemption.
Gain on Sale, Taxable Exchange or Other Taxable Disposition of GSAH Class A Common Stock.
If our redemption of a Non-U.S. Holders shares of GSAH Class A common stock is treated as a sale or other taxable disposition as discussed above under the section entitled Redemption of GSAH Class A Common Stock, subject to the discussions of FATCA (defined below) and backup withholding below, a Non-U.S. Holder generally will not be subject to U.S. federal income or withholding tax in respect of gain recognized on a sale, taxable exchange or other taxable disposition of GSAH Class A common stock, unless:
| | the gain is effectively connected with the conduct of a trade or business by the Non-U.S. Holder within the United States (and, under certain applicable income tax treaties, is attributable to a United States permanent establishment or fixed base maintained by the Non-U.S. Holder); or |
| | we are or have been a United States real property holding corporation for U.S. federal income tax purposes at any time during the shorter of the five-year period ending on the date of disposition or the period that the Non-U.S. Holder held GSAH Class A common stock, and, in the case where shares of GSAH Class A common stock are regularly traded on an established securities market, the Non-U.S. Holder has owned, directly or constructively, more than 5% of GSAH Class A common stock at any time within the shorter of the five-year period preceding the disposition or such Non-U.S. Holders holding period for the shares of GSAH Class A common stock. |
Unless an applicable treaty provides otherwise, gain described in the first bullet point above will be subject to tax at generally applicable U.S. federal income tax rates as if the Non-U.S. Holder were a U.S. resident. In the event the Non-U.S. Holder is a corporation for U.S. federal income tax purposes, such gain may also be subject to an additional branch profits tax at a thirty percent (30%) rate (or such lower rate specified by an applicable tax treaty).
If the second bullet point above applies to a Non-U.S. Holder, gain recognized by such Non-U.S. Holder on the redemption treated as a sale of GSAH Class A common stock will be subject to tax at generally applicable U.S. federal income tax rates. In addition, unless GSAH Class A common stock is treated as regularly traded on an established securities market, a buyer of GSAH Class A common stock (we would be treated as a buyer with respect to a redemption of GSAH Class A common stock) may be required to withhold U.S. federal income tax at a rate of fifteen percent (15%) of the amount realized upon such redemption. There can be no assurance that
198
Table of Contents
GSAH Class A common stock will be treated as regularly traded on an established securities market. We believe that we are not and have not been at any time since our formation a United States real property holding company and we do not expect to be a United States real property holding corporation immediately after the Business Combination is completed.
Taxation of Distributions.
If our redemption of a Non-U.S. Holders shares of GSAH Class A common stock is treated as a distribution, as discussed above under the section entitled Redemption of GSAH Class A Common Stock, to the extent paid out of our current or accumulated earnings and profits (as determined under U.S. federal income tax principles), such distribution will constitute a dividend for U.S. federal income tax purposes and, provided such dividend is not effectively connected with the Non-U.S. Holders conduct of a trade or business within the United States, and subject to the discussion below, we or an applicable withholding agent may be required to withhold tax from the gross amount of the dividend at a rate of thirty percent (30%), unless such Non-U.S. Holder is eligible for a reduced rate of withholding tax under an applicable income tax treaty and timely provides proper certification of its eligibility for such reduced rate (usually on an IRS Form W-8BEN or W-8BEN-E, as applicable). Any distribution not constituting a dividend will be treated first as reducing (but not below zero) the Non-U.S. Holders adjusted tax basis in its shares of GSAH Class A common stock redeemed and, to the extent such distribution exceeds the Non-U.S. Holders adjusted tax basis, as gain realized from the sale or other disposition of the Class A common stock, which will be treated as described above under the section entitled Gain on Sale, Taxable Exchange or Other Taxable Disposition of GSAH Class A Common Stock.
Because it may not be certain at the time a Non-U.S. Holder is redeemed whether such Non-U.S. Holders redemption will be treated as a sale of shares or a distribution constituting a dividend, and because such determination will depend in part on a Non-U.S. Holders particular circumstances, we or the applicable withholding agent may not be able to determine whether (or to what extent) a Non-U.S. Holder is treated as receiving a dividend for U.S. federal income tax purposes. Therefore, we or the applicable withholding agent may withhold tax at a rate of thirty percent (30%) (or such lower rate as may be specified by an applicable income tax treaty) on the gross amount of any consideration paid to a Non-U.S. Holder in redemption of such Non-U.S. Holders Class A common stock, unless (i) we or the applicable withholding agent have established special procedures allowing Non-U.S. Holders to certify that they are exempt from such withholding tax and (ii) such Non-U.S. Holder is able to certify that it meets the requirements of such exemption (e.g., because such Non-U.S. Holder is not treated as receiving a dividend under the Section 302 tests described above under the section entitled Redemption of GSAH Class A Common Stock). However, there can be no assurance that we or any applicable withholding agent will establish such special certification procedures. If we or an applicable withholding agent withhold excess amounts from the amount payable to a Non-U.S. Holder, such Non-U.S. Holder generally may obtain a refund of any such excess amounts by timely filing an appropriate claim for refund with the IRS. Non-U.S. Holders should consult their own tax advisors regarding the application of the foregoing rules in light of their particular facts and circumstances and any applicable procedures or certification requirements.
Additionally, the withholding tax described above does not apply to dividends paid to a Non-U.S. Holder who provides an IRS Form W-8ECI, certifying that the dividends are effectively connected with the Non-U.S. Holders conduct of a trade or business within the United States. Instead, the effectively connected dividends will be subject to regular U.S. federal income tax as if the Non-U.S. Holder were a U.S. resident, subject to an applicable income tax treaty providing otherwise. A Non-U.S. Holder that is a corporation for U.S. federal income tax purposes and is receiving effectively connected dividends may also be subject to an additional branch profits tax imposed at a rate of thirty percent (30%) (or a lower applicable treaty rate).
FACTA Withholding Taxes
Sections 1471 through 1474 of the Code and the Treasury Regulations and administrative guidance promulgated thereunder (commonly referred as the Foreign Account Tax Compliance Act or FATCA) generally impose
199
Table of Contents
withholding at a rate of thirty (30%) in certain circumstances on dividends (including amounts treated as dividends received pursuant to a redemption of GSAH Class A common stock) in respect of GSAH Class A common stock and, subject to the discussion of certain proposed Treasury Regulations below, on the gross proceeds from a sale of disposition of our Class A common stock, in each case if paid to foreign financial institutions (which is broadly defined for this purpose and in general includes including investment funds), unless any such institution (1) enters into, and complies with, an agreement with the IRS to report, on an annual basis, information with respect to interests in, and accounts maintained by, the institution that are owned by certain U.S. persons and by certain non-U.S. entities that are wholly or partially owned by U.S. persons and to withhold on certain payments, or (2) if required under an intergovernmental agreement between the United States and an applicable foreign country, reports such information to its local tax authority, which will exchange such information with the U.S. authorities. An intergovernmental agreement between the United States and an applicable foreign country may modify these requirements. Accordingly, the entity through which GSAH Class A common stock is held will affect the determination of whether such withholding is required. Similarly, dividends (including amounts treated as dividends received pursuant to a redemption of GSAH Class A common stock) in respect of, or the gross proceeds from a sale or other disposition of, GSAH Class A common stock held by an investor that is a non-financial non-U.S. entity that does not qualify under certain exceptions will generally be subject to withholding at a rate of thirty (30%), unless such entity either (1) certifies to us or the applicable withholding agent that such entity does not have any substantial United States owners or (2) provides certain information regarding the entitys substantial United States owners, which will in turn be provided to the U.S. Department of Treasury.
The IRS released proposed Treasury Regulations that, if finalized in their present form, would eliminate the U.S. federal withholding tax of thirty (30%) applicable to the gross proceeds of a sale or other disposition of Class A common stock. In its preamble to such proposed Treasury Regulations, the IRS stated that taxpayers may generally rely on the proposed Treasury Regulations until final Treasury Regulations are issued.
All stockholders should consult their own tax advisors regarding the possible implications of FATCA on an exercise of redemption rights.
Information Reporting and Backup Withholding
In general, information reporting requirements may apply to dividends received by U.S. Holders of Class A common stock, and the proceeds received on the sale, exchange or other disposition of Class A common stock effected within the U.S. (and, in certain cases, outside the U.S.), in each case other than U.S. Holders that are exempt recipients (such as corporations). Backup withholding (currently at a rate of 24%) may apply to such amounts if the U.S. holder fails to provide an accurate taxpayer identification number (generally on an IRS Form W-9 provided to the paying agent of the U.S. Holders broker) or is otherwise subject to backup withholding. Any redemptions treated as dividend payments with respect to Class A common stock and proceeds from the sale, exchange, or other disposition of Class A common stock may be subject to information reporting to the IRS and possible U.S. backup withholding. U.S. Holders should consult their tax advisors regarding the application of the U.S. information reporting and backup withholding rules.
Information returns may be filed with the IRS in connection with, and Non-U.S. Holders may be subject to backup withholding on, amounts received in respect of their Class A common stock, unless the Non-U.S. Holder furnishes to the applicable withholding agent the required certification as to its non-U.S. status, such as by providing a valid IRS Form W-8BEN, IRS Form W-8BEN- E or IRS Form W-8ECI, as applicable, or the Non-U.S. Holder otherwise establishes an exemption. Dividends paid with respect to Class A common stock and proceeds from the sale, exchange or other disposition of Class A common stock received in the U.S. by a Non-U.S. Holder through certain U.S.-related financial intermediaries may be subject to information reporting and backup withholding unless such Non-U.S. Holder provides proof of an applicable exemption or complies with certain certification procedures described above, and otherwise complies with the applicable requirements of the U.S. information reporting and backup withholding rules.
200
Table of Contents
Backup withholding is not an additional tax. Amounts withheld as backup withholding may be credited against the U.S. Holders U.S. federal income tax liability, and a U.S. Holder may obtain a refund of any excess amounts withheld under the backup withholding rules by timely filing the appropriate claim for a refund with the IRS and furnishing any required information. The amount of any backup withholding from a payment to a Non-U.S. Holder will be allowed as a credit against such Non-U.S. Holders U.S. federal income tax liability and may entitle such holder to a refund, provided that the required information is timely furnished to the IRS.
THE CONCLUSIONS EXPRESSED ABOVE ARE BASED ON CURRENT LAW. FUTURE LEGISLATIVE, ADMINISTRATIVE OR JUDICIAL CHANGES OR INTERPRETATIONS, WHICH CAN APPLY RETROACTIVELY, COULD AFFECT THE ACCURACY OF THOSE CONCLUSIONS. THIS DISCUSSION IS INTENDED TO PROVIDE ONLY A SUMMARY OF CERTAIN U.S. FEDERAL INCOME TAX CONSEQUENCES OF THE REDEMPTION OF HOLDERS OF CLASS A COMMON STOCK. IT DOES NOT ADDRESS TAX CONSEQUENCES THAT MAY VARY WITH, OR ARE CONTINGENT ON, YOUR INDIVIDUAL CIRCUMSTANCES.
Regulatory Matters
Under the HSR Act and the rules that have been promulgated thereunder by the FTC, certain transactions may not be consummated unless information has been furnished to the Antitrust Division and the FTC and certain waiting period requirements have been satisfied. The Business Combination is subject to these requirements and may not be completed until the expiration of a 30-day waiting period and any extensions thereto following the filing of the required Notification and Report Forms with the Antitrust Division and the FTC or until early termination is granted. GSAH and Mirion each filed a Notification and Report Form under the HSR Act on July 9, 2021, and the HSR waiting period expired on August 9, 2021.
At any time before or after consummation of the Business Combination, notwithstanding the expiration of the waiting period under the HSR Act, the applicable competition authorities could take such action under applicable antitrust laws as each deems necessary or desirable in the public interest, including seeking to enjoin the consummation of the Business Combination. Private parties may also seek to take legal action under the antitrust laws under certain circumstances. Even following the termination or expiration of the waiting period under the HSR Act, we cannot assure you that the Antitrust Division, the FTC, any state attorney general, or any other government authority will not attempt to challenge the Business Combination on antitrust grounds, and, if such a challenge is made, we cannot assure you as to its result.
Additionally, under applicable foreign direct investment (FDI) laws in certain foreign jurisdictions, certain transactions may not be consummated until approval is granted or applicable waiting periods expire or terminate. The Business Combination is subject to these requirements in Finland, France, Germany and possibly the United Kingdom, and may not be completed until such approvals are obtained or the applicable waiting periods have expired.
Finnish FDI
Under the Finnish FDI regime (the Finnish Act on the Monitoring of Foreign Corporate Acquisitions (172/2012, as amended, the Monitoring Act)), all corporate acquisitions by foreign acquirers concerning the defense and dual-use sectors require advance approval by Finnish authorities. Advance approval must also be received for corporate acquisitions concerning companies operating in the security sector that provide products or services to authorities that are deemed vital for the security of the society. The Business Combination is subject to such monitoring process and may not be completed until the Finnish Ministry of Economic Affairs and Employment (the Ministry) has approved the transaction. GSAH submitted a request for approval to the Ministry on July 2, 2021.
If the Ministry finds that the Business Combination may endanger key national interests, it transfers the matter to the governments plenary session for resolution. The governments plenary session then makes the decision
201
Table of Contents
about whether to restrict or approve the Business Combination, depending on whether it believes the Business Combination poses a threat to the national interest. The Ministry may also impose conditions on the completion of the transaction that are necessary to secure very important national interest. However, if the Ministry considers that the Business Combination does not endanger a key national interest, it must approve the transaction. It is presently contemplated that the Ministry will approve the Business Combination. There can be no assurance, however, that such approval will be obtained or that no additional conditions will be imposed on the completion of the Business Combination.
French FDI
Under French FDI rules (Articles L. 151-3 and R. 151-1 et seq. of the French Monetary and Financial Code) investments made by non-French investors in French companies active in certain sensitive sectors listed in Article R. 151-3 of the Code are subject to the prior authorization of the French Minister for Economy. The authorization procedure has a suspensory effect on the consummation of the transaction.
Since 1 April 2020, a two-stage process applies to foreign investment applications filed with the French Treasury, which is responsible for handling the review of foreign investments:
| | first, within 30 business days following receipt of an application for authorization, the French Treasury either (i) issues a decision stating that (a) the transaction does not fall within the scope of the foreign investment control regime and is therefore not subject to an authorization, (b) the transaction falls within the scope of the regime and is authorized without any conditions or (c) the transaction falls within the scope of the regime but further examination is necessary to determine whether the preservation of national interests can be ensured by attaching conditions to the authorization or (ii) fails to issue such a decision, in which case the authorization is deemed to be refused; |
| | second, within 45 business days following receipt by the investor of the decision mentioned in (c) above, the French Treasury either (i) issues a decision refusing or granting the necessary authorization, with or without conditions, or (ii) fails to issue such a decision, in which case the authorization is deemed to be refused. |
If the French Treasury considers that the approval can be granted but requires undertakings from the foreign investor prior to approving the transaction, a commitment letter has to be negotiated between the investor and the ministries concerned.
Mirions French subsidiaries are active in certain business segments that qualify as sensitive within the meaning of Article R. 151-3 of the French Monetary and Financial Code such as the defense sector and the energy (nuclear) sector. Therefore, the indirect acquisition by GSAH of Mirion Technologies (France) SAS, Mirion Technologies (Canberra) SAS, Mirion Technologies (MGPI) SAS, Mirion Technologies (Premium Analyse) SAS and Mirion Technologies (IST France) will require a prior foreign investment clearance and the entering into a commitment letter setting out the commitments of GSAH in respect of the sensitive activities of these companies.
GSAH submitted a request for approval to the French Treasury on June 28, 2021. It is presently expected that the French Treasury will confirm that the transaction falls within the scope of the FDI regime and that further examination is necessary to determine and negotiate the conditions that will be attached to the foreign investment authorization.
It is presently contemplated that the French Minister for Economy will approve the Business Combination. There can be no assurance, however, that such approval will be obtained or that no additional conditions (other than the conditions set out in the above-mentioned commitment letter) will be imposed on the completion of the Business Combination.
German FDI
Under the German FDI regime, any acquisition of significant participations of 10% or more of the voting rights in domestic target companies may be reviewed and ultimately be prohibited by the Germany authorities. This
202
Table of Contents
applies where a domestic target company develops or manufactures goods or provides services of certain highly sensitive industries specified in more detail in the German Foreign Trade and Payments Ordinance, including a target company that, inter alia, develops, manufactures, modifies or has effective control over military. Within the framework of the sector-specific FDI regime, the competent German authority reviews whether the acquisition of a domestic company by a foreigner is likely to impair essential security interests of Germany or of, or within, the EU.
Irrespective of the relevant industry, the competent German authority may also examine, under the cross-sector FDI, whether the direct or indirect acquisition of voting rights or shareholdings by a non-European Union investor in a German company above a certain threshold results in a probable impediment of the public order or security of Germany. There are different thresholds within the cross-sector FDI regime: In principle, any acquisition of at least 25% of the voting rights of a company resident in Germany by acquirers located outside the territory of the EU region, is subject to the cross-sectoral FDI regime. Foreign investments in German target companies active in certain civilian security relevant industries listed as Catalogue Activities, such as critical infrastructures, can be reviewed by the German authority if 10% or more or in some cases 20% or more of the voting rights are acquired.
Based on the latest amendments to the law, also the granting of supervisory or management board seats in the domestic target companies, of veto rights on strategic business or personnel decisions of the domestic target companies or of the right to obtain particularly sensitive technically critical information from the domestic target companies may trigger the sector-specific or cross-sectoral FDI regime if such granting of rights would give the acquirer a control right equivalent to that of a holder 10% of the voting rights under the sector-specific FDI regime or 10%, 20% or 25% of the voting rights under cross-sectoral FDI regime (atypical acquisition of control).
Mirion German subsidiary Mirion Technologies (Canberra) GmbHs product portfolio used to include two products related to the defense industry. However, the sector-specific FDI regime also applies to companies which have developed, produced, modified or had effective control over such goods in the past and which still have knowledge of or other access to the technology underlying such goods.
Furthermore, Mirion German subsidiary Mirion Technologies (MGPI H&B) GmbH and Mirion Technologies (Canberra) GmbH are active in certain business segments related to the defense industry. The acquisition by GSAH of Mirion under the Business Combination therefore requires clearance under the German FDI regime by way of a sector-specific review and cross-sectoral review, and accordingly a filing has been made for such clearance. Further, the acquisition by GSAH of Mirion German subsidiaries Mirion Technologies (AWST) GmbH and Sun Nuclear GmbH could be subject to a review by the competent German authority under the less strict cross-sectoral FDI regime.
On July 1, 2021, a filing under the German FDI regime was submitted to the Federal Ministry for Economic Affairs and Energy (Bundesministerium für Wirtschaft und Energie; BMWi) by GSAH requesting approval of the Business Combination. In light of the clearance practice of the BMWi, it is currently expected that the Business Combination will obtain clearance. There can be no assurance, however, that such approval will be obtained or that no additional conditions will be imposed on the completion of the Business Combination.
Vote Required for Approval
The approval of this Business Combination Proposal requires the affirmative vote of a majority of the votes cast by holders of our outstanding shares of common stock represented at the Special Meeting by attendance via the virtual meeting website or by proxy and entitled to vote thereon at the Special Meeting. Accordingly, a Company stockholders failure to vote by proxy or to vote during the Special Meeting, as well as an abstention from voting and a broker non-vote with regard to this Business Combination Proposal will have no effect on this Business Combination Proposal. Abstentions will be counted in connection with the determination of whether a valid quorum is established but will have no effect on this Business Combination Proposal.
203
Table of Contents
The Business Combination is conditioned on the approval of this Business Combination Proposal as well as the other Condition Precedent Proposals. If the other Condition Precedent Proposals are not approved, this Business Combination Proposal will have no effect, even if approved by our stockholders.
The Sponsor and GS Employee Participation have each agreed to, among other things, vote in favor of this Business Combination Proposal and the transactions contemplated under the Business Combination Agreement.
Recommendation of the Board of Directors
OUR BOARD OF DIRECTORS UNANIMOUSLY RECOMMENDS THAT OUR STOCKHOLDERS VOTE FOR THE BUSINESS COMBINATION PROPOSAL.
204
Table of Contents
PROPOSAL NO. 2THE NYSE PROPOSAL
Overview
Assuming the Business Combination Proposal is approved, we are asking our stockholders to approve, for purposes of complying with applicable listing rules of the NYSE, the issuance of more than 20% of the Companys outstanding common stock in connection with the Business Combination, including:
| | 90,000,000 shares of our Class A common stock as part of the PIPE Investment |
| | 38,999,986 shares of our common stock to the Sellers (which may be in the form of our Class A common stock or Class B common stock) |
In addition, in the event we waive the Minimum Cash Condition, we could issue up to an additional 131,000,000 shares in lieu of the $1,310,000,000 cash consideration (assuming a $10.00 per share value). We are also asking our stockholders to approve the issuance of the shares of Class A common stock issuable upon the exchange of shares of the Class B common stock.
In addition, we will issue 20,000,000 of the shares of our Class A common stock as part of the PIPE Investment to GSAM Holdings, which may be deemed to be an affiliate of the Company. In connection with the Backstop Agreement and the Option Agreement we may also issue up to an additional 12,500,000 of shares of our Class A common stock to GSAM Holdings.
Each of the holders of GSAH Class B common stock has agreed to waive the anti-dilution adjustments provided for in the GSAH Certificate of Incorporation applicable to GSAH Class B common stock in connection with the Business Combination, including the PIPE Investment. As a result of such waiver, the 18,750,000 shares of GSAH Class B common stock will automatically convert into shares of GSAH Class A common stock on a one-for-one basis upon the consummation of the Business Combination.
Additionally, as contemplated by the Incentive Plan Proposal, we intend to reserve 10% of the shares of our common stock to be outstanding immediately following the closing of the Business Combination for grants of awards under the Incentive Plan, which amount automatically increases annually as described in the Incentive Plan Proposal. For more information on the Incentive Plan and the Incentive Plan Proposal, See Proposal No. 6The Incentive Plan Proposal.
Why the Company Needs Stockholder Approval
Pursuant to Section 312.03(c) of the NYSEs Listed Company Manual, stockholder approval is required prior to the issuance of common stock, or of securities convertible into or exercisable for common stock, in any transaction or series of related transactions if: (1) the common stock has, or will have upon issuance, voting power equal to or in excess of 20 percent of the voting power outstanding before the issuance of such stock or of securities convertible into or exercisable for common stock or (2) the number of shares of common stock to be issued is, or will be upon issuance, equal to or in excess of 20 percent of the number of shares of common stock outstanding before the issuance of the common stock or of securities convertible into or exercisable for common stock. For further details, see Proposal No. 6The Incentive Plan Proposal.
Accordingly, the aggregate number of shares of GSAH Class A common stock that we will issue in connection with the Business Combination will exceed 20% of both the voting power and the shares of common stock outstanding before such issuance, and for this reason, we are seeking the approval of our stockholders for the issuance of shares of GSAH Class A common stock pursuant to the Business Combination Agreement and the PIPE Investment.
205
Table of Contents
Additionally, pursuant to Section 312.03(b) of the NYSEs Listed Company Manual, stockholder approval is required prior to the issuance of common stock, or of securities convertible into or exercisable for common stock, in any transaction or series of related transactions, to (1) a director, officer or substantial security holder of the company (each a Related Party), (2) a subsidiary, affiliate or other closely related person of a Related Party or (3) any company or entity in which a Related Party has a substantial direct or indirect interest, in each case, if the number of shares of common stock to be issued, or if the number of shares of common stock into which the securities may be convertible or exercisable, exceeds either one percent of the number of shares of common stock or one percent of the voting power outstanding before the issuance.
As part of the PIPE Investment, we expect to issue an estimated 20 million shares of GSAH Class A common stock to GSAM Holdings in accordance with the terms and subject to the conditions of the Subscription Agreements. For further details, see Proposal No. 1Approval of the Business CombinationRelated AgreementsSubscription Agreements.
Additionally, members of Mirion management will receive shares of GSAH Class B common stock as Transaction Consideration.
Accordingly, the aggregate number of shares of GSAH Class A common stock and GSAH Class B common stock that we will issue to a Related Party in the Business Combination may exceed 1% of the shares of our common stock outstanding before such issuance, and for this reason, we are seeking the approval of our stockholders for the issuance of (x) shares of GSAH Class A common stock pursuant to the PIPE Investment and (y) shares of GSAH Class B common stock to members of Mirion management as Transaction Consideration.
In the event that this proposal is not approved by GSAH stockholders, the Business Combination cannot be consummated. In the event that this proposal is approved by GSAH stockholders, but the Business Combination Agreement is terminated (without the Business Combination being consummated) prior to the issuance of shares of GSAH Class A common stock pursuant to the Business Combination Agreement or the PIPE Investment, such shares of GSAH Class A common stock will not be issued.
Vote Required for Approval
The approval of this NYSE Proposal requires the affirmative vote of holders of a majority of the votes cast by the holders of our outstanding shares of common stock represented at the Special Meeting by attendance via the virtual meeting website or by proxy and entitled to vote thereon at the Special Meeting. Accordingly, a Company stockholders failure to vote by proxy or to vote during the Special Meeting and a broker non-vote with regard to the NYSE Proposal will have no effect on the NYSE Proposal. The NYSE considers abstentions to be votes cast and included in the number of shares of which a majority is required to vote in favor. Accordingly, abstentions will have the same effect as a vote AGAINST the NYSE Proposal.
The Business Combination is conditioned on the approval of this NYSE Proposal as well as the other Condition Precedent Proposals. This NYSE Proposal is conditioned upon the approval of the other Condition Precedent Proposals. If the other Condition Precedent Proposals are not approved, this NYSE Proposal will have no effect, even if approved by our stockholders.
The Sponsor and GS Employee Participation have each agreed to, among other things, vote in favor of this NYSE Proposal. As of the date of this proxy statement/prospectus, the Initial Stockholders own approximately 20% of the outstanding shares of our common stock.
Recommendation of the Board of Directors
OUR BOARD OF DIRECTORS UNANIMOUSLY RECOMMENDS THAT OUR STOCKHOLDERS VOTE FOR THE NYSE PROPOSAL.
206
Table of Contents
PROPOSAL NO. 3THE CHARTER PROPOSAL
Assuming the Business Combination Proposal and the NYSE Proposal are approved, we are asking our stockholders to adopt the New Mirion Charter in the form attached hereto as Annex B, which, in the judgment of our Board, is necessary to adequately address the needs of the Company following the consummation of the Transactions.
The New Mirion Charter differs materially from the GSAH Certificate of Incorporation. The following is a summary of the principal changes proposed between the GSAH Certificate of Incorporation and the New Mirion Charter. This summary is qualified by reference to the complete text of the New Mirion Charter, a copy of which is attached to this proxy statement/prospectus as Annex B. All stockholders are encouraged to read the New Mirion Charter in its entirety for a more complete description of its terms.
| | change the purpose of the Company to engage in any lawful act or activity for which corporations may be organized under the General Corporation Law of the State of Delaware; |
| | increase the total number of shares of our capital stock from 555,000,000 shares to 2,200,000,000 shares, which would consist of increasing (i) the GSAH Class A common stock from 500,000,000 shares to 2,000,000,000, (ii) the GSAH Class B common stock from 50,000,000 shares to 100,000,000 shares, and (iii) the GSAH preferred stock from 5,000,000 to 100,000,000 shares; |
| | delete the prior Article IX to eliminate provisions specific to our status as a blank check company and to make conforming changes; |
| | remove the conversion rights relating to the GSAH Class B common stock; and |
| | provide that the affirmative vote of holders of not less than 66 2/3% of the total voting power of all outstanding securities of the Company generally entitled to vote in the election of directors, voting together as a single class will be required to amend, alter, change or repeal specified provisions of the New Mirion Charter, including those relating to the terms of the New Mirion common stock, actions by written consent of stockholders, calling of special meetings of stockholders, election and removal of directors, certain indemnification and corporate opportunity matters, and the required vote to amend the New Mirion Charter and New Mirion Bylaws. |
Reasons for the Amendments
Each of the amendments was negotiated as part of the Transactions. The Boards reasons for proposing each of these amendments to the certificate of incorporation are set forth below:
| | Amending prior Article II to provide that the purpose of the Company is to engage in any lawful act or activity for which corporations may be organized under the General Corporation Law of the State of Delaware. The Board believes this change is appropriate to remove language applicable to a blank check company. |
| | Amending prior Section 4(a)1 to increase our total number of authorized shares of capital stock. The amendment provides for the issuance of shares of the GSAH Class A common stock necessary to consummate the Transactions including, without limitation, the PIPE Investment and also provides flexibility for future issuances of common stock if determined by the Board to be in the best interests of the Company without incurring the risk, delay and potential expense incident to obtaining stockholder approval for a particular issuance. |
| | Deleting the prior Article IX to eliminate provisions specific to our status as a blank check company and to make conforming changes. These revisions are desirable because they will serve no purpose following the Transactions. |
| | Revising the supermajority voting provisions to provide that the affirmative vote of holders of not less than 66 2/3% of the total voting power of all outstanding securities of the Company generally entitled to vote in |
207
Table of Contents
| the election of directors, voting together as a single class will be required to amend, alter, change or repeal specified provisions of the New Mirion Charter, including those relating to the terms of the New Mirion common stock, actions by written consent of stockholders, calling of special meetings of stockholders, election and removal of directors, certain indemnification and corporate opportunity matters, and the required vote to amend the New Mirion Charter and New Mirion Bylaws. The Board believes this amendment protects key provisions of the proposed New Mirion Charter and New Mirion Bylaws from arbitrary amendment and prevents a simple majority of stockholders from taking actions that may be harmful to other stockholders or making changes to provisions that are intended to protect all stockholders. |
Vote Required for Approval
The approval of the Charter Proposal requires the affirmative vote of holders of a majority of our outstanding shares of common stock entitled to vote thereon at the Special Meeting. Accordingly, a Company stockholders failure to vote by proxy or to vote during the Special Meeting, as well as an abstention from voting and a broker non-vote with regard to the Charter Proposal will have the same effect as a vote AGAINST the Charter Proposal.
The Charter Proposal is conditioned upon the approval of the other Condition Precedent Proposals. If the other Condition Precedent Proposals are not approved, the Charter Proposal will have no effect, even if approved by our stockholders, and the GSAH Certificate of Incorporation will remain in effect.
The Business Combination is conditioned on the approval of the Charter Proposal as well as the other Condition Precedent Proposals. The Sponsor and GS Employee Participation have each agreed to, among other things, vote in favor of the Charter Proposal. As of the date of this proxy statement/prospectus, the Initial Stockholders own approximately 20% of the outstanding shares of our common stock.
Recommendation of the Board of Directors
OUR BOARD OF DIRECTORS UNANIMOUSLY RECOMMENDS THAT OUR STOCKHOLDERS VOTE FOR THE CHARTER PROPOSAL.
208
Table of Contents
PROPOSAL NO. 4THE GOVERNANCE PROPOSAL
Overview
Our stockholders are also being asked to vote on the governance provisions referred to below, which are included in the New Mirion Charter. In accordance with SEC guidance, this proposal is being presented separately and will be voted upon on a non-binding advisory basis.
In the judgment of our Board, these provisions are necessary to adequately address the needs of the Company and its stockholders following the consummation of the Transactions. Accordingly, regardless of the outcome of the non-binding advisory vote on these proposals, the Company intends that the New Mirion Charter in the form set forth on Annex B will take effect at consummation of the business combination, assuming adoption of the Charter Proposal.
Proposal No. 4A Change in Authorized Shares
Description of Amendment
The amendment would increase the total number of shares of our capital stock from 555,000,000 shares to 2,200,000,000 shares, which would consist of increasing (i) the GSAH Class A common stock from 500,000,000 shares to 2,000,000,000, (ii) the GSAH Class B common stock from 50,000,000 shares to 100,000,000 shares, and (iii) the Companys preferred stock from 5,000,000 to 100,000,000 shares.
Reasons for the Amendment
The amendment provides for the increase necessary to consummate the Transactions including, without limitation, the PIPE Investment and also provides flexibility for future issuances of common stock if determined by the Board to be in the best interests of the Company without incurring the risk, delay and potential expense incident to obtaining stockholder approval for a particular issuance.
Proposal No. 4B Super Majority Voting Provisions
The amendment would provide that the affirmative vote of holders of not less than 66 2/3% of the total voting power of all outstanding securities of the Company generally entitled to vote in the election of directors, voting together as a single class will be required to amend, alter, change or repeal specified provisions of the New Mirion Charter, including those relating to the terms of the New Mirion common stock, actions by written consent of stockholders, calling of special meetings of stockholders, election and removal of directors, certain indemnification and corporate opportunity matters, and the required vote to amend the New Mirion Charter and New Mirion Bylaws.
Reasons for the Amendment
The amendment protects key provisions of the proposed New Mirion Charter and New Mirion Bylaws from arbitrary amendment and prevents a simple majority of stockholders from taking actions that may be harmful to other stockholders or making changes to provisions that are intended to protect all stockholders.
Proposal No. 4C Corporate Opportunity
The amendment provides that certain potential transactions are not corporate opportunities and that any member of the Board, who is not an employee of the Company or its subsidiaries, or any employee or agent of such member, other than someone who is an employee of the Company or its subsidiaries (collectively, the Covered Persons), are not subject to the doctrine of corporate opportunity, except with respect to business opportunity matters, potential transactions or interests that a Covered Person obtains expressly and solely in connection with the individuals services as a member of the Board.
209
Table of Contents
Reasons for the Amendment
The amendment is intended to provide that certain transactions are not corporate opportunities and that each Covered Person is not subject to the doctrine of corporate opportunity and does not have any fiduciary duty to refrain from engaging directly or indirectly in the same or similar business activities or lines of business as the Company or any of its subsidiaries. The prior Article X provided that the doctrine of corporate opportunity, or any other analogous doctrine, does not apply with respect to the Company or any of its officers or directors in circumstances where the application of any such doctrine to a corporate opportunity would conflict with any fiduciary duties or contractual obligations. The Board believes the amendment is appropriate because members of the Board and their affiliates should not be restricted from investing in or operating similar businesses unless such opportunities are presented to such individuals in connection with his or her service on the Board.
Vote Required for Approval
The approval of the Governance Proposal requires the affirmative vote of a majority of the votes cast by holders of outstanding shares of the Companys common stock represented at the Special Meeting by attendance via the virtual meeting website or by proxy and entitled to vote at the Special Meeting. Accordingly, if a valid quorum is established, a Churchill stockholders failure to vote by proxy or to vote at the special meeting, abstentions and broker non-votes with regard to the Governance Proposal will have no effect on such proposal. Abstentions will be counted in connection with the determination of whether a valid quorum is established, but will have no effect on the outcome of the Governance Proposal.
As discussed above, a vote to approve the Governance Proposal is an advisory vote, and therefore, is not binding on the Company, Mirion or their respective boards of directors. Accordingly, regardless of the outcome of the non-binding advisory vote, the Company and Mirion intend that the Mirion New Charter, in the form set forth on Annex B and containing the provisions noted above, will take effect at consummation of the business combination, assuming adoption of the Charter Proposal.
The Business Combination is conditioned on the approval of the Condition Precedent Proposals. The Sponsor and GS Employee Participation have each agreed to, among other things, vote in favor of the Charter Proposal. As of the date of this proxy statement/prospectus, the Initial Stockholders own approximately 20% of the outstanding shares of our common stock.
Recommendation of the Board of Directors
OUR BOARD OF DIRECTORS UNANIMOUSLY RECOMMENDS THAT OUR STOCKHOLDERS VOTE FOR THE CHARTER PROPOSAL.
210
Table of Contents
PROPOSAL NO. 5THE DIRECTOR ELECTION PROPOSAL
Overview
Assuming the Business Combination Proposal, the NYSE Proposal and the Charter Proposal are approved, we are asking holders of our common stock to elect nine directors, effective upon the closing of the Business Combination, with each director on our Board having a term that expires at the post-business combination companys annual meeting of stockholders in 2022, and until their respective successors are duly elected and qualified, or until their earlier resignation, removal or death.
Following consummation of the Business Combination, our Board will consist of nine directors. Accordingly, our Board has nominated each of Thomas D. Logan, Lawrence D. Kingsley, Jyothsna (Jo) Natauri, Christopher Warren, Steven Etzel, [●], [●], [●] and [●] to serve as our directors upon the consummation of the Business Combination, with Mr. Kingsley to serve as the Chairman of the Board of Directors. For more information on the experience of each of these director nominees, see the section titled Management of New Mirion Following the Business Combination of this proxy statement/prospectus.
Vote Required for Approval
Directors are elected by a plurality of all of the votes cast by holders of shares of common stock represented at the special meeting by attendance via the virtual meeting website or by proxy and entitled to vote thereon at the Special Meeting. This means that the nine director nominees who receive the most affirmative votes will be elected. Stockholders may not cumulate their votes with respect to the election of directors. Assuming a valid quorum is established, abstentions and broker non-votes will have no effect on the election of directors.
The Business Combination is conditioned on the approval of this Director Election Proposal as well as the other Condition Precedent Proposals. This Director Election Proposal is conditioned upon the approval of the other Condition Precedent Proposals. If the other Condition Precedent Proposals are not approved, this Director Election Proposal will have no effect, even if approved by our stockholders.
The Sponsor and GS Employee Participation have each agreed to, among other things, vote in favor of this Director Election Proposal.
Recommendation of the Board of Directors
OUR BOARD OF DIRECTORS UNANIMOUSLY RECOMMENDS THAT OUR STOCKHOLDERS VOTE FOR THE ELECTION OF EACH OF THE NINE DIRECTOR NOMINEES TO THE BOARD OF DIRECTORS IN THE DIRECTOR ELECTION PROPOSAL
211
Table of Contents
PROPOSAL NO. 6THE INCENTIVE PLAN PROPOSAL
Overview
Assuming that the Business Combination is approved, our stockholders are also being asked to approve and adopt the Mirion Technologies, Inc. 2021 Omnibus Incentive Plan (the Incentive Plan). Our board of directors has approved the Incentive Plan, subject to receiving stockholder approval. A summary of the principal features of the Incentive Plan is provided below. This summary does not purport to be complete and is subject to, and qualified in its entirety by, the complete text of the Incentive Plan. A copy of the Incentive Plan is attached to this proxy statement/prospectus as Annex I. If the Business Combination closes and the Incentive Plan is approved by our stockholders, the Incentive Plan will be administered by the Compensation Committee of our board of directors, which will have the authority to make awards under the Incentive Plan.
If the Business Combination closes and the Incentive Plan is not approved by stockholders, we will be unable to make equity grants to our employees, consultants and directors, and therefore we will be at a significant competitive disadvantage in attracting, retaining and motivating talented individuals who contribute to our success.
Considerations for the Approval of the Plan
The Incentive Plan incorporates corporate governance best practices to align our equity compensation program with the interests of our stockholders. Certain of the corporate governance best practices included in our Plan are as follows:
| | Restricted dividends on awards. The Incentive Plan prohibits the payment of dividends in respect of an award (other than awards of restricted stock) prior to the time such award (or the applicable portion thereof) vests (and, in the case of performance awards, the applicable performance condition is achieved). |
| | No repricing. Repricing of stock options and stock appreciation rights (SARs) is not permitted without stockholder approval, except for adjustments with respect to certain specified extraordinary corporate transactions. |
| | No liberal change in control definition. The change in control definition under the Incentive Plan is only triggered in those instances where an actual change in control occurs, such as a 50% or greater change in beneficial ownership (see Change in Control, below). |
| | Clawback of awards. The Incentive Plan provides that awards granted thereunder are subject to any clawback or recoupment policies that we have in effect from time to time. |
Summary of the Incentive Plan
Purpose
The purpose of the Incentive Plan is to enable us to offer our employees, directors and other individual service providers long-term equity-based incentives in us, thereby attracting, retaining and rewarding such individuals, and strengthening the mutuality of interests between such individuals and our stockholders.
Eligibility
Our employees, non-employee directors, individual consultants, and advisors are eligible to receive awards under the Incentive Plan based on the Compensation Committees determination, in its sole discretion, that an award to such individual will further the Incentive Plans stated purpose (as described above). Awards of incentive stock options will be limited to our employees or certain of our affiliates. As of March 31, 2021, there are approximately 2,575 employees and 135 individual consultants, directors, advisers and other service providers eligible to receive awards under the Incentive Plan.
212
Table of Contents
Authorized Shares
Subject to adjustment (as described below), the number of shares of our common stock that may initially be subject to awards granted under the Incentive Plan will be 10% of the shares of our common stock outstanding immediately following the closing of the Business Combination, and up to 20,000,000 of such shares may be issued in the form of incentive stock options under the Incentive Plan. The total number of shares of our common stock available for issuance under the Incentive Plan will, following the effective date of the Incentive Plan, automatically increase on the first day of each New Mirion fiscal year for the duration of the term of the Incentive Plan in an amount equal to the lesser of (i) 3% of the total number of shares of our common stock outstanding on the last day of the immediately preceding fiscal year, (ii) 5% of the shares of our common stock outstanding immediately following the closing of the Business Combination and (iii) such number of shares of our common stock as determined by the Compensation Committee in its discretion. If an award expires or is cancelled or forfeited, or is otherwise settled without the issuance of shares, the shares covered by the award will again be available for issuance under the Incentive Plan. Shares tendered or withheld to pay or satisfy the exercise price of a stock option or SAR or to pay taxes in respect of any stock option or SAR, will again be available for issuance under the Incentive Plan. Shares underlying replacement awards (i.e., awards granted as replacements for awards granted by a company that we acquire or with which we combine) will not reduce the number of shares available for issuance under the plan. The Incentive Plan limits non-employee director compensation, including cash fees and incentive equity awards (based on their grant-date fair value), to a maximum of (i) $750,000 during the initial annual period following a non-employee directors appointment or election to our board of directors and (ii) $500,000 per each subsequent calendar year, in each case, in respect of their service as non-employee directors. The limitation on non-employee director compensation applies beginning the first calendar year following the effective date of the Incentive Plan.
Administration
The Incentive Plan is administered by the Compensation Committee.
The Compensation Committee has authority under the Incentive Plan to:
| | designate participants; |
| | determine the types of awards to grant, the number of shares to be covered by awards, the terms and conditions of awards, the circumstances under which awards may be canceled, forfeited or suspended, and whether awards may be deferred; |
| | amend the terms of any outstanding awards; |
| | correct any defect, supply any omission or reconcile any inconsistency in the Incentive Plan or any award agreement, in the manner and to the extent it shall deem desirable to carry the Incentive Plan into effect; |
| | interpret and administer the Incentive Plan and any instrument or agreement relating to, or award made under, the Incentive Plan; and |
| | make any other determination and take any other action that it deems necessary or desirable to administer the Incentive Plan, in each case, as it deems appropriate for the proper administration of the Incentive Plan and compliance with applicable law, stock market or exchange rules and regulations or accounting or tax rules and regulations. |
The Compensation Committee may delegate some or all of its authority under the Incentive Plan, to the extent permitted by applicable law, to (i) one or more of our officers (except that such delegation will not be applicable to grant awards to a person then covered by Section 16 of the Securities Exchange Act of 1934 (the Exchange Act)) and (ii) one or more committees of our board of directors.
Establishment of Sub-plans
Our board of directors has the authority to establish one or more sub-plans under the Incentive Plan to facilitate the local administration of the Incentive Plan in any jurisdiction in which we and our affiliates operate and to
213
Table of Contents
conform the Incentive Plan to the legal requirements of any such jurisdiction or to allow for favorable tax treatment under any applicable provision of tax law. Our board of directors may establish such sub-plans by adopting supplements setting forth (i) such limitations on the Committees discretion under the Incentive Plan as the board of directors deems necessary or desirable and (ii) such additional terms and conditions not otherwise inconsistent with the Incentive Plan as the board of directors deems necessary or desirable. All sub-plans adopted by the board of directors will be deemed to be part of the Incentive Plan, but each such sub-plan will only apply to participants within the affected jurisdiction.
Types of Awards
The Incentive Plan provides for grants of stock options (both nonqualified and incentive stock options), SARs, restricted stock, restricted stock units, performance awards and other cash-based or stock-based awards. Any award may be granted alone or in tandem with other awards, and may be granted in addition to, or in substitution for, other types of awards.
Stock Options. A stock option is a contractual right to purchase shares at a future date at a specified exercise price. The per share exercise price of a stock option will be determined by the Compensation Committee and may not be less than the fair market value of a share of our common stock on the grant date. The Compensation Committee will determine the date after which each stock option may be exercised, the method and form by which each option is to be exercised, and the expiration date of each option, provided that no option will be exercisable more than ten years after the grant date. Options intended to be incentive stock options under Section 422 of the Code may not be granted to any person who is not an employee of us or any parent or subsidiary, as defined in Section 424 of the Code. There have not yet been any options granted under the Incentive Plan, and so there are no options outstanding under the Incentive Plan.
Stock Appreciation Rights. Stock appreciation rights (SARs) represent a contractual right to receive, in cash or shares, an amount equal to the appreciation of one share from the grant date. The terms and conditions applicable to stock options also apply to SARs.
Restricted Stock. Restricted stock is an award of shares that are subject to restrictions on transfer and a substantial risk of forfeiture. Recipients of restricted stock generally have the rights and privileges of a stockholder, including the right to vote such shares of restricted stock and receive dividends.
Restricted Stock Units. A restricted stock unit award is a right to receive a specified number of shares of our common stock (or the fair market value thereof in cash, other property or any combination thereof, as determined by the Committee), subject to the expiration of a specified restriction period and/or the achievement of any performance measures selected by the Committee, consistent with the terms of the Incentive Plan. The restricted stock unit award agreement will specify whether the award recipient is entitled to receive dividend equivalents with respect to the number of shares of our common stock subject to the award. Prior to the settlement of a restricted stock unit award in our common stock, the award recipient will have no rights or privileges as a stockholder of us with respect to our common stock subject to the award.
Performance Awards. Performance awards, which may be denominated in cash, shares or units (including restricted stock units), will be earned on the satisfaction of performance goals specified by the Compensation Committee. With respect to any performance award that becomes settled in our common stock upon achievement or satisfaction of the applicable performance conditions, prior to such settlement the award recipient will have no rights or privileges as a stockholder of us with respect to our common stock subject to the award.
Other Cash-Based and Other Stock-Based Awards. The Compensation Committee is authorized to grant other cash-based and other stock-based awards that are payable in cash or our common stock (or a combination thereof), and may be granted either independently or as an element of or supplement to any other award under the Incentive Plan. Other stock-based awards are valued in whole or in part by reference to such stock, including restricted stock units, phantom stock and similar units.
214
Table of Contents
Dividends and Dividend Equivalents
Other than with respect to awards of restricted stock, awards granted under the Incentive Plan may not provide for any dividend to be payable to the participant in respect of such award prior to the time such award (or the applicable portion thereof) vests (and, in the case of performance awards, the applicable performance condition is achieved). The Compensation Committee may, in its discretion, provide for dividend equivalents on awards of restricted stock units.
Adjustments
In the event the Compensation Committee determines that, as a result of any dividend or other distribution (other than an ordinary dividend or distribution), recapitalization, stock split, reverse stock split, reorganization, merger, amalgamation, consolidation, separation, rights offering, split-up, spin-off, combination, repurchase or exchange of shares of our common stock or other securities, or other similar corporate transaction or event affecting our common stock or of changes in applicable laws, regulations or accounting principles, an adjustment is necessary to prevent dilution or enlargement of the benefits or potential benefits intended to be made available under the Incentive Plan, the Compensation Committee will adjust equitably any or all of: (i) the number and type of shares or other securities that thereafter may be made the subject of awards, including the aggregate limit under the Incentive Plan; (ii) the number and type of shares or other securities subject to outstanding awards; (iii) the grant, purchase, exercise or hurdle price for any award or, if deemed appropriate, make provision for a cash payment to the holder of an outstanding award; and (iv) the terms and conditions of any outstanding awards, including the performance criteria of any performance awards.
Change in Control
In the event of a change in control, except as otherwise provided in the applicable award agreement, the Committee may provide for:
| | continuation or assumption of outstanding awards under the Incentive Plan by us (if we are the surviving corporation) or by the surviving corporation or its parent; |
| | substitution or replacement or any outstanding award for a cash payment; |
| | acceleration of the vesting (including the lapse of any restriction) and exercisability of outstanding awards, in each case, either (i) immediately prior to or as of the date of the change in control, (ii) upon a participants involuntary termination of service on or within a specified period following the change in control, or (iii) upon the failure of the successor or surviving corporation (or its parent) to continue or assume such outstanding awards; |
| | in the case of a performance award, determination of the level of attainment of the applicable performance conditions; and |
| | cancellation of outstanding awards under the Incentive Plan in consideration of a payment, with the form, amount and timing of such payment to be determined by the Committee in its sole discretion, provided that (i) such payment is made in cash, securities, rights and/or other property, (ii) the amount of such payment equals the value of the award, as determined by the Committee in its sole discretion (provided that the Committee may cancel out-of-the-money options or SARs for no consideration) and (iii) such payment will be made promptly following the change in control, in compliance with Section 409A of the Code. |
A change in control generally means (i) the acquisition of 50% or more of our common stock or combined voting power of voting securities; (ii) a change in the composition of our board of directors such that, during any 12-month period, the individuals who as of the beginning of such period constitute our board of directors cease for any reason to constitute at least 50% of our board of directors (provided that any individual becoming a member of our board of directors after the beginning of such 12-month period whose election or nomination for election by our stockholders was approved by a vote of at least a majority of the directors immediately prior to
215
Table of Contents
the date of such appointment or election will be considered as though such individual were a member of our board of directors at the beginning of such 12-month period); (iii) our merger or consolidation with another entity after which our voting securities outstanding immediately prior to such transaction do not continue to represent 50% or more of the total voting power of our stock or of the surviving entity or parent entity thereof (if we are not the surviving entity in such merger or consolidation); or (iv) a disposition of all or substantially all of our assets.
Amendment and Termination
Our board of directors may amend, modify, suspend, discontinue or terminate the Incentive Plan (or any portion thereof) at any time. However, no such action may, without the consent of the participant, materially adversely affect the rights of such participant under any award previously granted (other than to apply with applicable law or to impose any clawback or recoupment provisions on any awards). Additionally, no such action may be made without our stockholder approval, if such approval is required by applicable law or by the rules of the stock market or exchange on which our common shares are principally quoted or traded. No award may be granted pursuant to the Incentive Plan after the tenth anniversary of the date on which the Incentive Plan was approved by our stockholders.
Prohibition on Repricing
Subject to the adjustment provision described above, the Compensation Committee may not directly or indirectly, through cancellation or re-grant or any other method, reduce, or have the effect of reducing, the exercise or hurdle price of any award established at the time of grant without approval of our stockholders.
Cancellation or Clawback of Awards
The Compensation Committee may, to the extent permitted by applicable law and stock exchange rules or by any of our policies (including any recoupment policy we may adopt from time to time or pursuant to the recoupment provisions in any award agreement), cancel or require reimbursement of any awards granted, shares issued or cash received upon the vesting, exercise or settlement of any awards granted under the Plan or the sale of shares underlying such awards.
Term
The Plan expires 10 years after the date on which the Business Combination is consummated, unless earlier terminated (x) upon the maximum number of shares of common stock available for issuance under the Incentive Plan having been issued or (y) by the board of directors at its discretion (and in accordance with the terms of the Incentive Plan).
U.S. Federal Income Tax Consequences of Equity Awards
The following is a general summary under current law of certain United States federal income tax consequences to us and participants who are citizens or individual residents of the United States relating to awards granted under the Incentive Plan. This summary deals with the general tax principles that apply to such awards and is provided only for general information. Certain kinds of taxes, such as foreign taxes, state and local income taxes, payroll taxes and the alternative minimum tax, are not discussed. This summary is not tax advice and it does not discuss all aspects of federal taxation that may be relevant to us and participants. Accordingly, we urge each participant to consult his or her own tax advisor as to the specific tax consequences of participation in the Plan under federal, state, local and other applicable laws. In addition, we may be subject to limits on tax deductibility relating to compensation described herein under certain statutory provisions, including Sections 162(m) and 280G of the Code.
216
Table of Contents
Non-Qualified Stock Options
A non-qualified stock option is an option that does not meet the requirements of Section 422 of the Code. A participant generally will not recognize taxable income when granted a non-qualified stock option. When the participant exercises the stock option, he or she generally will recognize taxable ordinary income equal to the excess of the fair market value of the shares received on the exercise date over the aggregate exercise price of the shares. The participants tax basis in the shares acquired on exercise of the option will be increased by the amount of such taxable income. We generally will be entitled to a corresponding federal income tax deduction. When the participant sells the shares acquired on exercise, the participant generally will realize long-term or short-term capital gain or loss, depending on whether the participant holds the shares for more than one year before selling them.
Incentive Stock Options
An incentive stock option or ISO is an option that meets the requirements of Section 422 of the Code. A participant will not have taxable income when granted an ISO or when exercising an ISO. If a participant exercises an ISO and does not dispose of the shares until the later of two years after the grant date and one year after the exercise date, the entire gain, if any, realized when the participant sells the shares will be taxable as long-term capital gain. However, even though a participant will not have taxable income when exercising an ISO, the exercise of an ISO is taken into account for purposes of determining whether the participant has any alternative minimum tax liability (described below). We generally will not be entitled to a corresponding federal income tax deduction.
If a participant disposes of the shares received upon exercise of an ISO within the one-year or two-year periods described above, it will be considered a disqualifying disposition. Under such circumstances, the participant generally will realize ordinary income in the year of the disposition, and we generally will be entitled to a corresponding federal income tax deduction. The amounts of the participants ordinary income and our deduction will equal the excess of the lesser of the amount, if any, realized on the disposition and the fair market value of the shares on the exercise date over the aggregate exercise price of the ISO. Any additional gain or loss that the participant realizes on the disposition will be long-term or short-term capital gain or loss, depending on whether the participant holds the shares for more than one year before selling them.
If a participant exercises an ISO more than three months after the participants employment with us terminates, the option will be treated as a non-qualified stock option for federal income tax purposes. If a participant is disabled and terminates employment because of his or her disability, the three-month period is extended to one year. The three-month period does not apply in the case of a participants death.
SARs
A participant does not recognize income at the time a SAR is granted. A participant will recognize income at the time cash or stock representing the amount of the appreciation is transferred to the participant pursuant to exercise of a SAR. The amount of income will equal the amount of cash or fair market value of shares paid or transferred to the participant and will be ordinary income. We generally will be entitled to a corresponding federal income tax deduction.
Restricted Stock
Unless a participant makes an election to accelerate recognition of the income to the date of grant as described below, the participant generally will not recognize income, and we generally will not be entitled to a corresponding federal income tax deduction at the time restricted stock is granted. When the restrictions lapse, the participant generally will recognize ordinary income equal to the fair market value of the shares as of that date, less any amount paid for the restricted stock, and we generally will be entitled to a corresponding federal
217
Table of Contents
income tax deduction at that time. If the participant files an election under Section 83(b) of the Code within 30 days after the date of grant of the restricted stock, the participant generally will recognize ordinary income as of the date of grant equal to the fair market value of the common shares as of that date, less any amount the participant paid for the restricted stock, and we generally will be entitled to a corresponding federal income tax deduction at that time. Any future appreciation in the shares generally will be taxable to the participant at capital gains rates. However, if the restricted stock is later forfeited, the participant generally will not be able to recover the tax previously paid pursuant to his Section 83(b) election.
Restricted Stock Units
A participant does not recognize taxable income at the time of grant of a restricted stock unit, and we are not entitled to a tax deduction at that time. The participant will recognize compensation taxable as ordinary income (and subject to income tax withholding), however, at the time of the settlement of the award, equal to the fair market value of any shares delivered and the amount of cash paid by us. We will be entitled to a corresponding deduction, except to the extent that the deduction limits of Section 162(m) of the Code apply.
Registration With the SEC
If our stockholders approve the Incentive Plan, we plan to file a registration statement on a Form S-8 with the SEC, as soon as reasonably practicable following the 60th day after Closing, to register the shares available for issuance under the Incentive Plan.
New Plan Benefits
As described above, the Compensation Committee, in its discretion, will select the participants who receive awards and the size and types of those awards under the Incentive Plan, if the Incentive Plan is approved by our stockholders. Therefore, the awards (or associated benefits or amounts) that will be made to particular individuals or groups of individuals in the future under the Plan are not currently determinable.
Vote Required for the Incentive Plan Proposal Approval
The Incentive Plan Proposal is conditioned on the approval of the Business Combination Proposal at the Special Meeting.
The approval of the Incentive Plan Proposal requires the affirmative vote of a majority of the votes cast by holders of our outstanding shares of common stock represented at the Special Meeting by attendance via the virtual meeting website or by proxy and entitled to vote thereon at the Special Meeting. A Company stockholders failure to vote by proxy or to vote during the Special Meeting and a broker non-vote with regard to the Incentive Plan Proposal will have no effect on the Incentive Plan Proposal. The NYSE considers abstentions to be votes cast and included in the number of shares of which a majority is required to vote in favor. Accordingly, abstentions will have the same effect as a vote AGAINST the Incentive Plan Proposal.
Recommendation of the Board of Directors
OUR BOARD OF DIRECTORS RECOMMENDS THAT OUR STOCKHOLDERS VOTE FOR THE APPROVAL OF THE INCENTIVE PLAN PROPOSAL.
Recommendation of the Board of Directors
OUR BOARD OF DIRECTORS UNANIMOUSLY RECOMMENDS THAT OUR STOCKHOLDERS VOTE FOR THE INCENTIVE PLAN PROPOSAL.
218
Table of Contents
PROPOSAL NO. 7THE ADJOURNMENT PROPOSAL
Overview
The Adjournment Proposal, if adopted, will allow our Board to adjourn the Special Meeting to a later date or dates to permit further solicitation of proxies. The Adjournment Proposal will only be presented to our stockholders in the event that there are insufficient votes for, or for any other reason in connection with, the approval of the Condition Precedent Proposals.
Consequences if the Adjournment Proposal is Not Approved
If the Adjournment Proposal is not approved by our stockholders, our Board may not have the ability to adjourn the Special Meeting to a later date and, therefore, will not have more time to solicit votes to approve the Condition Precedent Proposals. In such events, the Business Combination would not be completed and there is a chance we may not consummate an alternative initial business combination.
Vote Required for Approval
The approval of the Adjournment Proposal requires the affirmative vote of a majority of the votes cast by holders of our outstanding shares of common stock represented at the Special Meeting by attendance via the virtual meeting website or by proxy and entitled to vote thereon at the Special Meeting. Accordingly, a Company stockholders failure to vote by proxy or to vote during the Special Meeting, as well as an abstention from voting and a broker non-vote will have no effect on the Adjournment Proposal. Abstentions will be counted in connection with the determination of whether a valid quorum is established but will have no effect on the Adjournment Proposal.
The Sponsor and GS Employee Participation have each agreed to, among other things, vote in favor of this Adjournment Proposal. As of the date of this proxy statement/prospectus, the Initial Stockholders own approximately 20% of the outstanding shares of our common stock.
Recommendation of the Board of Directors
OUR BOARD OF DIRECTORS UNANIMOUSLY RECOMMENDS THAT OUR STOCKHOLDERS VOTE FOR THE ADJOURNMENT PROPOSAL.
219
Table of Contents
Unless the context otherwise requires, all references in this section to the Company, we, us or our refer to GSAH prior to the consummation of the Business Combination.
General
We are a blank check company incorporated on May 31, 2018 as a Delaware corporation and formed for the purpose of effecting a merger, capital stock exchange, asset acquisition, stock purchase, reorganization or similar business combination with one or more businesses, which we refer to as a business combination. We have neither engaged in any operations nor generated any operating revenue to date. Based on our business activities, we are a shell company as defined under the Exchange Act because we have no operations and nominal assets consisting almost entirely of cash.
The Sponsor is an affiliate of Goldman Sachs and is managed by the Permanent Capital Strategies team within GSAM, a business within Goldman Sachs Asset Management. Prior to our entering into the Business Combination Agreement, our strategy was to identify and complete our initial business combination with a company in the Diversified Industrial, Healthcare, Technology, Media and Telecom, or Alternative Asset Management sector. Specifically, many companies in these sectors tend to be cash generative businesses that are growing at rates higher than U.S. gross domestic product. In addition to these fundamentals, the sectors are fragmented and contain a large number of privately-held and sponsor-owned businesses that we believe could benefit from deleveraging, accelerating revenue growth, expanding margins, and improving capital allocation decision-making. In addition to independent privately- and sponsor-held middle market businesses, we believe many larger companies in the sector are in the process of evaluating their portfolios of businesses and reviewing candidates for potential divestitures, which we believe may also prove to be attractive business combination targets.
On July 2, 2020, we closed the IPO of 75,000,000 units, including 5,000,000 units issued pursuant to the exercise by the underwriters of their option to purchase additional units in full, at a price of $10.00 per unit, generating gross proceeds to the Company of $750,000,000 before underwriting discounts and expenses. Each unit consists of one share of GSAH Class A common stock of our company, $0.0001 par value per share, and onequarter of one redeemable warrant, with each whole warrant exercisable for one share of GSAH Class A common stock at a price of $11.50 per share. Substantially concurrently with the closing of the IPO, we closed the private placement of an aggregate of 8,500,000 warrants, each exercisable to purchase one share of GSAH Class A common stock, par value $0.0001 per share, at an exercise price of $11.50 per share, to the Sponsor, at a price of $2.00 per private placement warrant, generating proceeds of $17,000,000. Each warrant and private placement warrant will become exercisable 30 days after the completion of our initial business combination, and will expire at 5:00 p.m., New York City time, five years after the completion of our initial business combination or earlier upon redemption or liquidation.
On the date the IPO was consummated, we placed $750,000,000 of proceeds (including $26,250,000 of deferred underwriting discount) from the IPO and the sale of the private placement warrants into the trust account and held $2,000,000 (net of offering expenses, other than underwriting discounts, paid upon the consummation of the IPO) of such proceeds outside the trust account. Except with respect to interest earned on the funds held in the trust account that may be released to the Company to pay its taxes, if any, the funds held in the trust account will not be released from the trust account until the earliest of: (1) the completion of the Companys initial business combination; (2) the redemption of any public shares properly submitted in connection with a stockholder vote to amend the Companys amended and restated certificate of incorporation (A) to modify the substance or timing of the Companys obligation to allow redemptions in connection with its initial business combination or to redeem 100% of its public shares if the Company does not complete its initial business combination by July 2, 2022 from the closing of the IPO or (B) with respect to any other provision relating to stockholders rights or pre-initial business combination activity; and (3) the redemption of all of the Companys public shares if it is unable to complete its initial business combination by July 2, 2022 from the closing of the IPO, subject to applicable law.
220
Table of Contents
Initial Business Combination
The NYSE rules require that our initial business combination must be with one or more operating businesses or assets with a fair market value equal to at least 80% of the net assets held in the trust account (net of amounts disbursed to management for working capital purposes, if permitted, and excluding the amount of any deferred underwriting discount). Our Board believes that the financial skills and background of its members qualify it to conclude that the acquisition of Mirion met this requirement.
Liquidation if No Business Combination
Our amended and restated certificate of incorporation provides that we will have until July 2, 2022 to complete our initial business combination. If we have not completed our initial business combination within such period or during any Extension Period, we will: (1) cease all operations except for the purpose of winding up; (2) as promptly as reasonably possible but not more than ten business days thereafter, redeem the public shares, at a per-share price, payable in cash, equal to the aggregate amount then on deposit in the trust account, including interest (which interest shall be net of taxes payable, and less up to $100,000 of interest to pay dissolution expenses), divided by the number of then outstanding public shares, which redemption will completely extinguish public stockholders rights as stockholders (including the right to receive further liquidating distributions, if any); and (3) as promptly as reasonably possible following such redemption, subject to the approval of our remaining stockholders and our board of directors, dissolve and liquidate, subject in each case to our obligations under Delaware law to provide for claims of creditors and the requirements of other applicable law. There will be no redemption rights or liquidating distributions with respect to our warrants, which will expire worthless if we fail to complete our initial business combination within the prescribed time period.
Our initial stockholders, officers and directors have entered into a letter agreement with us, pursuant to which they have waived their rights to liquidating distributions from the trust account with respect to any founder shares held by them if we fail to complete our initial business combination within the prescribed time period. However, if our sponsor or any of our officers, directors or any of their respective affiliates then hold any public shares, they will be entitled to liquidating distributions from the trust account with respect to such public shares if we fail to complete our initial business combination within the allotted time frame to complete our initial business combination.
Our sponsor, officers and directors have agreed, pursuant to a written agreement with us, that they will not propose any amendment to our amended and restated certificate of incorporation (A) to modify the substance or timing of our obligation to allow redemptions in connection with our initial business combination or to redeem 100% of our public shares if we do not complete our initial business combination by July 2, 2022 or (B) with respect to any other provision relating to stockholders rights or pre-initial business combination activity, unless we provide our public stockholders with the opportunity to redeem their shares of GSAH Class A common stock upon approval of any such amendment at a per-share price, payable in cash, equal to the aggregate amount then on deposit in the trust account, including interest (which interest shall be net of taxes payable), divided by the number of then outstanding public shares. However, we may not redeem our public shares in an amount that would cause our net tangible assets to be less than $5,000,001 following such redemptions.
We expect that all costs and expenses associated with implementing our plan of dissolution, as well as payments to any creditors, will be funded from amounts remaining out of the estimated $1,000,000 of proceeds held outside the trust account, although we cannot assure you that there will be sufficient funds for such purpose. However, if those funds are not sufficient to cover the costs and expenses associated with implementing our plan of dissolution, to the extent that there is any interest accrued in the trust account not required to pay taxes, we may request the trustee to release to us an additional amount of up to $100,000 of such accrued interest to pay those costs and expenses.
If we were to expend all of the net proceeds of the Business Combination and the sale of the private placement warrants, other than the proceeds deposited in the trust account, and without taking into account interest, if any,
221
Table of Contents
earned on the trust account and any tax payments or expenses for the dissolution of the trust, the per-share redemption amount received by stockholders upon our dissolution would be $10.00. The proceeds deposited in the trust account could, however, become subject to the claims of our creditors which would have higher priority than the claims of our public stockholders. We cannot assure you that the actual per-share redemption amount received by stockholders will not be substantially less than $10.00.
Although we will seek to have all vendors, service providers (other than our independent registered public accounting firm), prospective target businesses and other entities with which we do business execute agreements with us waiving any right, title, interest or claim of any kind in or to any monies held in the trust account for the benefit of our public stockholders, there is no guarantee that they will execute such agreements or even if they execute such agreements that they would be prevented from bringing claims against the trust account including but not limited to fraudulent inducement, breach of fiduciary responsibility or other similar claims, as well as claims challenging the enforceability of the waiver, in each case in order to gain an advantage with respect to a claim against our assets, including the funds held in the trust account. If any third party refuses to execute an agreement waiving such claims to the monies held in the trust account, our management will perform an analysis of the alternatives available to it and will only enter into an agreement with a third party that has not executed a waiver if management believes that such third partys engagement would be significantly more beneficial to us than any alternative. Examples of possible instances where we may engage a third party that refuses to execute a waiver include the engagement of a third-party consultant whose particular expertise or skills are believed by management to be significantly superior to those of other consultants that would agree to execute a waiver or in cases where we are unable to find a service provider willing to execute a waiver. In addition, there is no guarantee that such entities will agree to waive any claims they may have in the future as a result of, or arising out of, any negotiations, contracts or agreements with us and will not seek recourse against the trust account for any reason. In order to protect the amounts held in the trust account, our sponsor has agreed that it will be liable to us if and to the extent any claims by a third party for services rendered or products sold to us, or a prospective target business with which we have discussed entering into a transaction agreement, reduce the amount of funds in the trust account to below (1) $10.00 per public share; or (2) such lesser amount per public share held in the trust account as of the date of the liquidation of the trust account due to reductions in the value of the trust assets, in each case net of the amount of interest which may be withdrawn to pay taxes, except as to any claims by a third party who executed a waiver of any and all rights to seek access to the trust account and except as to any claims under our indemnity of the underwriters of the IPO against certain liabilities, including liabilities under the Securities Act. Moreover, in the event that an executed waiver is deemed to be unenforceable against a third party, our sponsor will not be responsible to the extent of any liability for such third-party claims. We have not independently verified whether our sponsor has sufficient funds to satisfy its indemnity obligations and believe that our sponsors only assets are securities of our company and, therefore, our sponsor may not be able to satisfy those obligations. We have not asked our sponsor to reserve for such obligations. Therefore, we cannot assure you that our sponsor would be able to satisfy those obligations. As a result, if any such claims were successfully made against the trust account, the funds available for our initial business combination and redemptions could be reduced to less than $10.00 per public share. In such event, we may not be able to complete our initial business combination, and you would receive such lesser amount per share in connection with any redemption of your public shares. None of our officers or directors will indemnify us for claims by third parties including, without limitation, claims by vendors and prospective target businesses.
In the event that the proceeds in the trust account are reduced below: (1) $10.00 per public share; or (2) such lesser amount per public share held in the trust account as of the date of the liquidation of the trust account due to reductions in the value of the trust assets, in each case net of the amount of interest which may be withdrawn to pay taxes, and our sponsor asserts that it is unable to satisfy its indemnification obligations or that it has no indemnification obligations related to a particular claim, our independent directors would determine whether to take legal action against our sponsor to enforce its indemnification obligations. While we currently expect that our independent directors would take legal action on our behalf against our sponsor to enforce its indemnification obligations to us, it is possible that our independent directors in exercising their business judgment may choose not to do so in certain instances. For example, the cost of such legal action may be deemed by the independent
222
Table of Contents
directors to be too high relative to the amount recoverable or the independent directors may determine that a favorable outcome is not likely. Accordingly, we cannot assure you that due to claims of creditors the actual value of the per-share redemption price will not be substantially less than $10.00 per public share.
We will seek to reduce the possibility that our sponsor will have to indemnify the trust account due to claims of creditors by endeavoring to have all vendors, service providers (other than our independent registered public accounting firm), prospective target businesses and other entities with which we do business execute agreements with us waiving any right, title, interest or claim of any kind in or to monies held in the trust account. Our sponsor will also not be liable as to any claims under our indemnity of the underwriters of our IPO against certain liabilities, including liabilities under the Securities Act. We will have access to up to an estimated $1,000,000 from the proceeds of the IPO and the sale of the private placements warrants with which to pay any such potential claims (including costs and expenses incurred in connection with our liquidation, currently estimated to be no more than approximately $100,000). In the event that we liquidate and it is subsequently determined that the reserve for claims and liabilities is insufficient, stockholders who received funds from our trust account could be liable for claims made by creditors. In the event that our offering expenses exceed our estimate of $1,000,000, we may fund such excess with funds from the funds not to be held in the trust account. In such case, the amount of funds we intend to be held outside the trust account would decrease by a corresponding amount. Conversely, in the event that the offering expenses are less than our estimate of $1,000,000, the amount of funds we intend to be held outside the trust account would increase by a corresponding amount.
Under the DGCL, stockholders may be held liable for claims by third parties against a corporation to the extent of distributions received by them in a dissolution. The pro rata portion of our trust account distributed to our public stockholders upon the redemption of our public shares in the event we do not complete our initial business combination within the required time period may be considered a liquidating distribution under Delaware law. If the corporation complies with certain procedures set forth in Section 280 of the DGCL intended to ensure that it makes reasonable provision for all claims against it, including a 60-day notice period during which any third-party claims can be brought against the corporation, a 90-day period during which the corporation may reject any claims brought, and an additional 150-day waiting period before any liquidating distributions are made to stockholders, any liability of stockholders with respect to a liquidating distribution is limited to the lesser of such stockholders pro rata share of the claim or the amount distributed to the stockholder, and any liability of the stockholder would be barred after the third anniversary of the dissolution.
Furthermore, if the pro rata portion of our trust account distributed to our public stockholders upon the redemption of our public shares in the event we do not complete our initial business combination within the required time period, is not considered a liquidating distribution under Delaware law and such redemption distribution is deemed to be unlawful, then pursuant to Section 174 of the DGCL, the statute of limitations for claims of creditors could then be six years after the unlawful redemption distribution, instead of three years, as in the case of a liquidating distribution. If we have not completed our initial business combination by July 2, 2022 or during any Extension Period, we will: (1) cease all operations except for the purpose of winding up; (2) as promptly as reasonably possible but not more than ten business days thereafter, redeem the public shares, at a per-share price, payable in cash, equal to the aggregate amount then on deposit in the trust account, including interest (which interest shall be net of taxes payable, and less up to $100,000 of interest to pay dissolution expenses), divided by the number of then outstanding public shares, which redemption will completely extinguish public stockholders rights as stockholders (including the right to receive further liquidating distributions, if any); and (3) as promptly as reasonably possible following such redemption, subject to the approval of our remaining stockholders and our board of directors, dissolve and liquidate, subject in each case to our obligations under Delaware law to provide for claims of creditors and the requirements of other applicable law. Accordingly, it is our intention to redeem our public shares as soon as reasonably possible following the end of our acquisition period and, therefore, we do not intend to comply with those procedures. As such, our stockholders could potentially be liable for any claims to the extent of distributions received by them (but no more) and any liability of our stockholders may extend well beyond the third anniversary of such date.
223
Table of Contents
Because we will not be complying with Section 280, Section 281(b) of the DGCL requires us to adopt a plan, based on facts known to us at such time that will provide for our payment of all existing and pending claims or claims that may be potentially brought against us within the subsequent ten years. However, because we are a blank check company, rather than an operating company, and our operations will be limited to searching for prospective target businesses to acquire, the only likely claims to arise would be from our vendors (such as lawyers, investment bankers, etc.) or prospective target businesses. As described above, pursuant to the obligation contained in our underwriting agreement, we will seek to have all vendors, service providers (other than our independent registered public accounting firm), prospective target businesses and other entities with which we do business execute agreements with us waiving any right, title, interest or claim of any kind in or to any monies held in the trust account.
As a result of this obligation, the claims that could be made against us are significantly limited and the likelihood that any claim that would result in any liability extending to the trust account is remote. Further, our sponsor may be liable only to the extent necessary to ensure that the amounts in the trust account are not reduced below: (1) $10.00 per public share; or (2) such lesser amount per public share held in the trust account as of the date of the liquidation of the trust account due to reductions in the value of the trust assets, in each case net of the amount of interest which may be withdrawn to pay taxes, except as to any claims by a third party who executed a waiver of any and all rights to seek access to the trust account and except as to any claims under our indemnity of the underwriters of this offering against certain liabilities, including liabilities under the Securities Act. In the event that an executed waiver is deemed to be unenforceable against a third party, our sponsor will not be responsible to the extent of any liability for such third-party claims.
If we file a bankruptcy petition or an involuntary bankruptcy petition is filed against us that is not dismissed, the proceeds held in the trust account could be subject to applicable bankruptcy law, and may be included in our bankruptcy estate and subject to the claims of third parties with priority over the claims of our stockholders. To the extent any bankruptcy claims deplete the trust account, we cannot assure you we will be able to return $10.00 per share to our public stockholders. Additionally, if we file a bankruptcy petition or an involuntary bankruptcy petition is filed against us that is not dismissed, any distributions received by stockholders could be viewed under applicable debtor/creditor and/or bankruptcy laws as either a preferential transfer or a fraudulent conveyance. As a result, a bankruptcy court could seek to recover some or all amounts received by our stockholders. Furthermore, our board of directors may be viewed as having breached its fiduciary duty to our creditors and/or may have acted in bad faith, and thereby exposing itself and our company to claims of punitive damages, by paying public stockholders from the trust account prior to addressing the claims of creditors. We cannot assure you that claims will not be brought against us for these reasons.
A public stockholder will be entitled to receive funds from the trust account only upon the earliest to occur of: (1) the completion of our initial business combination and then, only in connection with those public shares that such stockholder has properly elected to redeem, subject to the limitations described in this prospectus; (2) the redemption of any public shares properly submitted in connection with a stockholder vote to amend our amended and restated certificate of incorporation (A) to modify the substance or timing of our obligation to allow redemptions in connection with our initial business combination or to redeem 100% of our public shares if we do not complete our initial business combination by July 2, 2022 or (B) with respect to any other provision relating to stockholders rights or pre-initial business combination activity; and (3) the redemption of all of our public shares if we have not completed our initial business combination by July 2, 2022] subject to applicable law. In no other circumstances will a public stockholder have any right or interest of any kind to or in the trust account. In the event we seek stockholder approval in connection with our initial business combination, a stockholders voting in connection with our initial business combination alone will not result in a stockholders redeeming its shares to us for an applicable pro rata share of the trust account. Such stockholder must have also exercised its redemption rights described above. Holders of warrants will not have any rights of proceeds held in the trust account with respect to the warrants.
224
Table of Contents
Facilities
We currently maintain our executive offices at 200 West Street, New York, New York 10282. The cost for this space is included in the $10,000 per month fee that we will pay an affiliate of our sponsor for office space, administrative and support services. We consider our current office space adequate for our current operations.
Upon consummation of the Business Combination, our principal executive offices will be located at [●].
Employees
We currently have two officers and do not intend to have any full-time employees prior to the completion of our initial business combination. Members of our management team are not obligated to devote any specific number of hours to our matters but they intend to devote as much of their time as they deem necessary to our affairs until we have completed our initial business combination. The amount of time that any such person will devote in any time period to our company will vary based on whether a target business has been selected for our initial business combination and the current stage of the business combination process.
Legal Proceedings
There is no material litigation, arbitration or governmental proceeding currently pending against us or any members of our management team in their capacity as such, and we and the members of our management team have not been subject to any such proceeding in the 12 months preceding the date of this prospectus.
GSAH Management
Directors, Director Nominees and Executive Officers
| Name |
Age | Title |
||
| Raanan A. Agus |
53 | Chairman of the Board of Directors | ||
| Tom Knott |
34 | Chief Executive Officer, Chief Financial Officer and Secretary | ||
| Senator William Frist |
69 | Director | ||
| Steven S. Reinemund |
73 | Director | ||
| David Robinson |
55 | Director | ||
| Martha Sullivan |
64 | Director |
The directors and officers of the Company are as follows:
Raanan A. Agus has been the Chairman of our Board of Directors since June 2020 (previously serving as our Co-Chief Executive Officer and a director in April and May 2020). Mr. Agus serves as global co-head and co-chief investment officer of the Alternative Investments & Manager Selection (AIMS) Group of Goldman Sachs Asset Management. He also oversees GSAMs Energy and Infrastructure business. Previously, Mr. Agus served as head of Direct Alternatives for GSAM and global co-head of Goldman Sachs Investment Partners within GSAM, a position he held from the groups inception in 2007 until December 2019. Prior to that, he was co-head of the Goldman Sachs Principal Strategies Group beginning in 2003; he later became sole head of the group until 2007. Mr. Agus joined Goldman Sachs in 1993 as an associate in Equities Arbitrage. He was named managing director in 1999 and partner in 2000. Mr. Agus served as a director of GSAH I from April 2018 until the consummation of its business combination with Vertiv in February 2020. Mr. Agus earned an A.B. in economics, summa cum laude, phi beta kappa, from Princeton University in 1989 and a JD/MBA, Stone scholar, beta gamma sigma, specializing in Finance from Columbia University in 1993.
Tom Knott has been our Chief Executive Officer, Chief Financial Officer, Secretary and one of our directors since June 2020 (previously serving as our Co-Chief Executive Officer and Chairman of our Board of
225
Table of Contents
Directors in April and May 2020). Mr. Knott has served as the head of the Permanent Capital Strategies (PCS) Group of Goldman Sachs Asset Management since March 2018. Mr. Knott led all aspects of Goldman Sachs co-sponsorship of GSAH I from its IPO in June 2018 to its merger with Vertiv in February 2020. Prior to his role in the Permanent Capital Strategies Group, Mr. Knott worked within the Credit Alternatives Group in the Consumer and Investment Management Division of Goldman Sachs beginning in 2014. Mr. Knott earned a B.A. in history from Wake Forest University in 2009 and a MA in Management from Wake Forest University in 2010.
Senator William Frist has served on our board since the completion of the Initial Public Offering. Since 2007, Dr. Frist has served as a partner in Cressey & Company, a private equity firm focused exclusively on investing in and building leading healthcare businesses. He is chairman of the Cressey Distinguished Executive Council. Dr. Frist is also a co-founder and partner at Frist Cressey Ventures, a venture capital firm specializing in healthcare investments. As a U.S. Senator, Dr. Frist represented Tennessee for 12 years where he served on both the Finance and HELP committees responsible for writing all health legislation. He served as U.S. Senate Majority Leader from 2003 to 2007. Prior to the Senate, Dr. Frist was a heart and lung transplant surgeon. He spent 20 years in clinical medicine, completing surgical training at Harvards Massachusetts General Hospital and Stanford University, and he subsequently founded the Vanderbilt Multi-Organ Transplant Center. Dr. Frist serves as an adjunct professor of Cardiac Surgery at Vanderbilt University School of Medicine. Dr. Frist currently serves as a director of the publicly held companies Teladoc Health, Inc. (NYSE: TDOC), Select Medical Holdings Corporation (NYSE: SEM) and SmileDirectClub, Inc. (Nasdaq: SCX). His current board services include the Robert Wood Johnson Foundation, NashvilleHealth, SCORE and The Nature Conservancy (Global Board). His previous board service includes Princeton University, the Smithsonian Institution, AECOM, URS Corporation and Third National Bank. Dr. Frist earned his B.A. from Princeton University and M.D. from Harvard Medical School.
Steven S. Reinemund has served on our board since the completion of the Initial Public Offering. Mr. Reinemund served as Dean at Wake Forest University School of Business from July 2008 to June 2014, an organization he joined after a 23-year career with PepsiCo, Inc. (Nasdaq: PEP) (PepsiCo). At PepsiCo, Mr. Reinemund served as Executive Chairman from October 2006 to May 2007, and as Chairman and Chief Executive Officer from May 2001 to October 2006. Prior to being Chief Executive Officer, he was PepsiCo, Inc.s president and chief operating officer from September 1999 to May 2001. Mr. Reinemund began his career with PepsiCo, Inc. in 1984 at Pizza Hut, Inc. and held other positions until he became president and Chief Executive Officer of Frito-Lays North American snack division in 1992. He became chairman and Chief Executive Officer of Frito-Lays worldwide operations in 1996. Mr. Reinemund was a director of Johnson & Johnson (NYSE: JNJ) from 2003 to 2008, of American Express Company (NYSE: AXP) from 2007 to 2015, of Exxon Mobil Corporation (NYSE: XOM) from 2007 to May 2020 and Marriott International, Inc. (Nasdaq: MAR) from 2007 to May 2020. Mr. Reinemund currently serves as a director of Vertiv (and served as a director of GSAH I prior to its business combination with Vertiv), Walmart Inc. (NYSE: WMT) and Chick-fil-A, Inc. He also serves on the Board of Directors of the United States Naval Academy Foundation. Mr. Reinemund is a graduate of the United States Naval Academy in 1970 and served five years as an officer in the United States Marine Corps, achieving the rank of Captain. He also earned an MBA from the University of Virginia, and has been awarded honorary doctorate degrees by Johnson and Wales University and Bryant University.
David Robinson has served on our board since the completion of the Initial Public Offering. Mr. Robinson has served as co-founder of the Admiral Capital Group, a real estate and private equity firm, since 2009. Mr. Robinson has also served as the founder of the Carver Academy, a public charter school located in San Antonio, Texas, since 2001. Mr. Robinson spent 14 years in the NBA with the San Antonio Spurs and was introduced into the Naismith Memorial Basketball Hall of Fame in September 2009 among other honors, won the NBAs Most Valuable Player, two NBA championships and two Olympic Gold Medals. Mr. Robinsons philanthropic efforts led to the NBA naming its community service award the David Robinson Plaque which recognizes current NBA players for their community engagement, philanthropic activity and charity work. He is a graduate of the U.S. Naval Academy with a degree in mathematics.
226
Table of Contents
Martha Sullivan has served on our board since the completion of the Initial Public Offering. Ms. Sullivan has served as a director of Sensata Technologies Holding plc (NYSE: ST) (Sensata), a supplier of sensors and controls, since January 1, 2013, where she was Chief Executive Officer from January 1, 2013 until her retirement effective March 1, 2020. She previously served as President of Sensata from September 2010 until January 2019, and was also its Chief Operating Officer from September 2010 until July 2012. Prior to that, Ms. Sullivan held a variety of other positions with Sensata, its primary U.S. operating subsidiary and Texas Instruments (Sensatas preceding business unit) since she joined Texas Instruments in 1984. Ms. Sullivan has served as a director of Avery Dennison Corporation (NYSE: AVY), an adhesive manufacturing company, since 2013. Past and present external positions also include the Key Executive Council at Rensselaer Polytechnic Institute, Presidents Alumni Council at Michigan Technological University, and Ford International Supplier Advisory Council. She has been inducted into the Academy of Mechanical Engineering at Michigan Technological University and holds an Honorary Doctorate in Philosophy from that institution. She earned a B.S. in Mechanical Engineering from Michigan Technological University in 1980.
Number and Terms of Office of Officers and Directors
Our board of directors consists of six members. Holders of GSAH Class B common stock have the right to elect all of our directors and remove members of our board of directors for any reason prior to consummation of our initial business combination. Holders of our public shares do not have the right to vote on the election of directors during such time. These provisions of our amended and restated certificate of incorporation may only be amended if approved by holders of a majority of at least 90% of the outstanding shares of our common stock voting at a stockholder meeting. Approval of our initial business combination will require the affirmative vote of a majority of our board directors, which must include a majority of our independent directors and the two director nominees of our sponsor, initially Raanan A. Agus and Tom Knott. Subject to any other special rights applicable to the stockholders, prior to our initial business combination, any vacancies on our board of directors may be filled by the affirmative vote of a majority of the directors present and voting at the meeting of our board of directors that includes any directors representing our sponsor then on our board of directors, or by holders of a majority of the outstanding shares of GSAH Class B common stock.
Our officers are elected by the board of directors and serve at the discretion of the board of directors, rather than for specific terms of office. Our board of directors is authorized to appoint persons to the offices set forth in our bylaws as it deems appropriate. Our bylaws will provide that our officers may consist of a Chief Executive Officer, a President, a Chief Financial Officer, Vice Presidents, a Secretary, Assistant Secretaries, a Treasurer, Assistant Treasurers and such other offices as may be determined by the board of directors (including interim officers as it deems appropriate).
Committees of the Board of Directors
Our board of directors has three standing committees: an audit committee, a compensation committee and a nominating and corporate governance committee, each of which are composed solely of independent directors. Each committee operates under a charter that was approved by our board of directors and has the composition and responsibilities described below. The charter of each committee is available on our website.
Audit Committee
The members of our audit committee are Mr. Reinemund, Mr. Robinson and Ms. Sullivan. Ms. Sullivan serves as chairman of the audit committee. Each member of the audit committee is financially literate and our board of directors has determined that Mr. Reinemund qualifies as an audit committee financial expert as defined in applicable SEC rules and has accounting or related financial management expertise.
227
Table of Contents
We have adopted an audit committee charter, which details the purpose and principal functions of the audit committee, including:
| | assisting board oversight of (1) the integrity of our financial statements, (2) our compliance with legal and regulatory requirements, (3) our independent auditors qualifications and independence, and (4) the performance of our internal audit function and independent auditors; |
| | the appointment, compensation, retention, replacement, and oversight of the work of the independent auditors and any other independent registered public accounting firm engaged by us; |
| | pre-approving all audit and non-audit services to be provided by the independent auditors or any other registered public accounting firm engaged by us, and establishing pre-approval policies and procedures; |
| | reviewing and discussing with the independent auditors all relationships the auditors have with us in order to evaluate their continued independence; |
| | setting clear hiring policies for employees or former employees of the independent auditors; |
| | setting clear policies for audit partner rotation in compliance with applicable laws and regulations; |
| | obtaining and reviewing a report, at least annually, from the independent auditors describing (1) the independent auditors internal quality-control procedures and (2) any material issues raised by the most recent internal quality-control review, or peer review, of the audit firm, or by any inquiry or investigation by governmental or professional authorities, within the preceding five years respecting one or more independent audits carried out by the firm and any steps taken to deal with such issues; |
| | meeting to review and discuss our annual audited financial statements and quarterly financial statements with management and the independent auditor, including reviewing our specific disclosures under GSAHs Managements Discussion and Analysis of Financial Condition and Results of Operations; |
| | reviewing and approving any related party transaction required to be disclosed pursuant to Item 404 of Regulation S-K promulgated by the SEC prior to us entering into such transaction; and |
| | reviewing with management, the independent auditors, and our legal advisors, as appropriate, any legal, regulatory or compliance matters, including any correspondence with regulators or government agencies and any employee complaints or published reports that raise material issues regarding our financial statements or accounting policies and any significant changes in accounting standards or rules promulgated by the Financial Accounting Standards Board, the SEC or other regulatory authorities. |
Compensation Committee
The members of our Compensation Committee are Dr. Frist, Mr. Robinson and Ms. Sullivan. Mr. Robinson serves as chairman of the compensation committee. We have adopted a compensation committee charter, which details the purpose and responsibility of the compensation committee, including:
| | reviewing and approving on an annual basis the corporate goals and objectives relevant to our Chief Executive Officers compensation, evaluating our Chief Executive Officers performance in light of such goals and objectives and determining and approving the remuneration (if any) of our Chief Executive Officer based on such evaluation; |
| | reviewing and making recommendations to our board of directors with respect to the compensation, and any incentive-compensation and equity-based plans that are subject to board approval of all of our other officers; |
| | reviewing our executive compensation policies and plans; |
| | implementing and administering our incentive compensation equity-based remuneration plans; |
| | assisting management in complying with our proxy statement/prospectus and annual report disclosure requirements; |
228
Table of Contents
| | approving all special perquisites, special cash payments and other special compensation and benefit arrangements for our officers and employees; |
| | producing a report on executive compensation to be included in our annual proxy statement/prospectus; and |
| | reviewing, evaluating and recommending changes, if appropriate, to the remuneration for directors. |
The charter also provides that the compensation committee may, in its sole discretion, retain or obtain the advice of a compensation consultant, independent legal counsel or other adviser and is directly responsible for the appointment, compensation and oversight of the work of any such adviser. However, before engaging or receiving advice from a compensation consultant, external legal counsel or any other adviser, the compensation committee will consider the independence of each such adviser, including the factors required by the NYSE and the SEC.
Nominating and Corporate Governance Committee
The members of our nominating and corporate governance committee are Dr. Frist, Mr. Robinson and Ms. Sullivan. Ms. Sullivan serves as chair of the nominating and corporate governance committee.
We have adopted a nominating and corporate governance committee charter, which details the purpose and responsibilities of the nominating and corporate governance committee, including:
| | identifying, screening and reviewing individuals qualified to serve as directors, consistent with criteria approved by the board, and recommending to the board of directors candidates for nomination for election at the annual meeting of stockholders or to fill vacancies on the board of directors; |
| | developing and recommending to the board of directors and overseeing implementation of our corporate governance guidelines; |
| | coordinating and overseeing the annual self-evaluation of the board of directors, its committees, individual directors and management in the governance of the company; and |
| | reviewing on a regular basis our overall corporate governance and recommending improvements as and when necessary. |
The charter also provides that the nominating and corporate governance committee may, in its sole discretion, retain or obtain the advice of, and terminate, any search firm to be used to identify director candidates, and is directly responsible for approving the search firms fees and other retention terms.
We have not formally established any specific, minimum qualifications that must be met or skills that are necessary for directors to possess. In general, in identifying and evaluating nominees for director, the board of directors considers educational background, diversity of professional experience, knowledge of our business, integrity, professional reputation, independence, wisdom, and the ability to represent the best interests of our stockholders. Prior to our initial business combination, holders of our public shares will not have the right to recommend director candidates for nomination to our board of directors.
Director Independence
The rules of the NYSE require that a majority of our board of directors be independent within one year of our IPO. An independent director is defined generally as a person that, in the opinion of the companys board of directors, has no material relationship with the listed company (either directly or as a partner, shareholder or officer of an organization that has a relationship with the company). We have four independent directors as defined in the NYSE rules and applicable SEC rules. Our board of directors has determined that each of Dr. Frist, Mr. Reinemund, Mr. Robinson and Ms. Sullivan is an independent director under applicable SEC and NYSE rules. Our independent directors have regularly scheduled meetings at which only independent directors are present.
229
Table of Contents
Executive and Director Compensation
None of our officers or directors have received any compensation for services rendered to us. Our sponsor, officers, directors and their respective affiliates will be reimbursed for any out-of-pocket expenses incurred in connection with activities on our behalf such as identifying potential target businesses and performing due diligence on suitable business combinations. Our audit committee will review on a quarterly basis all payments that were made by us to our sponsor, officers, directors or our or any of their respective affiliates. Directors or members of our management team may be paid consulting, management or other compensation from the combined company.
We are not party to any agreements with our officers and directors that provide for benefits upon termination of employment. The existence or terms of any such employment or consulting arrangements may influence our managements motivation in identifying or selecting a target business, and we do not believe that the ability of our management to remain with us after the consummation of our initial business combination should be a determining factor in our decision to proceed with any potential business combination.
Limitation on Liability and Indemnification of Officers and Directors
Our amended and restated certificate of incorporation provides that our officers and directors will be indemnified by us to the fullest extent authorized by Delaware law, as it now exists or may in the future be amended. In addition, our amended and restated certificate of incorporation provides that our directors will not be personally liable for monetary damages to us or stockholders for breaches of their fiduciary duty as directors, except to the extent such exemption from liability or limitation thereof is not permitted by the DGCL.
We have entered into agreements with our officers and directors to provide contractual indemnification in addition to the indemnification provided for in our amended and restated certificate of incorporation. Our bylaws also permit us to maintain insurance on behalf of any officer, director or employee for any liability arising out of his or her actions, regardless of whether Delaware law would permit such indemnification. We have obtained a policy of directors and officers liability insurance that insures our officers and directors against the cost of defense, settlement or payment of a judgment in some circumstances and insures us against our obligations to indemnify our officers and directors.
A stockholders investment may be adversely affected to the extent we pay the costs of settlement and damage awards against officers and directors pursuant to these indemnification provisions.
We believe that these provisions, the insurance and the indemnity agreements are necessary to attract and retain talented and experienced officers and directors.
Insofar as indemnification for liabilities arising under the Securities Act may be permitted to directors, officers or persons controlling us pursuant to the foregoing provisions, we have been informed that in the opinion of the SEC such indemnification is against public policy as expressed in the Securities Act and is therefore unenforceable.
230
Table of Contents
GSAHS MANAGEMENTS DISCUSSION AND ANALYSIS OF FINANCIAL CONDITION AND RESULTS OF OPERATIONS
Unless the context otherwise requires, all references in this section to the we, us, our, the Company or GSAH refer to GSAH prior to the consummation of the Business Combination. The following discussion and analysis of GSAHs financial condition and results of operations should be read in conjunction with GSAHs condensed financial statements and notes to those statements included in this proxy statement/prospectus. Certain information contained in the discussion and analysis set forth below includes forward-looking statements that involve risks and uncertainties. GSAHs actual results may differ materially from those anticipated in these forward-looking statements as a result of many factors. See Cautionary Statement Regarding Forward-Looking Statements and Risk Factors in this proxy statement/prospectus.
Overview
We are a blank check company incorporated as a Delaware corporation and formed for the purpose of effecting a merger, capital stock exchange, asset acquisition, stock purchase, reorganization or similar business combination with one of more businesses. We intend to effectuate the Business Combination using cash from the proceeds of our IPO that closed on July 2, 2020 and the private placement of warrants to purchase shares of GSAH Class A common stock that closed on July 2, 2020 and from additional issuances of, if any, our capital stock and our debt, or a combination of cash, stock and debt.
At March 31, 2021, we had current assets of $2,245,581 and current liabilities of $64,746,945. Further, we expect to continue to incur significant costs in the pursuit of our acquisition plans. We have reviewed, and continue to review, a number of opportunities to enter into an initial business combination with operating businesses, but we are not able to determine at this time whether we will complete an initial business combination with any of the target businesses that we have reviewed or with any other target business.
Results of Operations
For the three months ended March 31, 2021 and 2020, we had net income of $9,682,349, of which $10,200,787 is related to the change in the fair value of the warrant liability and $0, respectively. Our business activities from inception to March 31, 2021 consisted primarily of our formation and completing our IPO, and since the offering, our activity has been limited to identifying and evaluating prospective acquisition targets for an initial business combination.
Liquidity and Capital Resources
Prior to the closing of our IPO, our only source of liquidity was an initial sale of shares of GSAH Class B common stock, par value $0.0001 per share, to the Sponsor and the proceeds of a promissory note from an affiliate of our Sponsor, in the amount of $300,000. The note was repaid upon the closing of our IPO.
The registration statement relating to our IPO was declared effective by the SEC on June 29, 2020. On June 30, 2020, the underwriters exercised a portion of their option to purchase additional units. Our IPO of 75,000,000 units, including 5,000,000 units pursuant to the underwriters partial exercise of such option, closed on July 2, 2020. Simultaneously with the closing of our IPO, we closed the private placement of an aggregate of 8,500,000 warrants, each exercisable to purchase one share of GSAH Class A common stock, par value $0.0001 per share, at an exercise price of $11.50 per share, to the Sponsor, at a price of $2.00 per private placement warrant, generating proceeds of $17,000,000. On July 2, 2020, we placed $750,000,000 of proceeds (including $26,250,000 of deferred underwriting discount) from the IPO and the private placement warrants into a U.S.-based trust account, with Continental Stock Transfer & Trust Company acting as trustee and held $2,000,000 of such proceeds outside the trust account.
At March 31, 2021, we had cash held in a custodian account of $1,740,897 and working capital of ($62,501,364).
231
Table of Contents
We do not believe we will need to raise additional funds in order to meet the expenditures required for operating our business prior to our initial business combination, due to the new Working Capital Note (as defined below). However, if our estimates of the costs of identifying a target business, undertaking in-depth due diligence and negotiating an initial business combination are less than the actual amount necessary to do so, we may have insufficient funds available to operate our business prior to our initial business combination. Moreover, we may need to obtain additional financing either to complete our initial business combination or because we become obligated to redeem a significant number of our shares of GSAH Class A common stock upon completion of our initial business combination, in which case we may issue additional securities or incur debt in connection with such Business Combination (including from our affiliates or affiliates of our Sponsor).
On November 12, 2020, the Sponsor agreed to loan us up to an aggregate of $2,000,000 pursuant to the working capital note (the Working Capital Note). Any amounts borrowed under the Working Capital Note are non-interest bearing, unsecured and are due at the earlier of the date we are required to complete our initial business combination pursuant to our amended and restated certificate of incorporation, as amended from time to time, and the closing of the initial business combination. On March 12, 2021, we borrowed $1,500,000 under the Working Capital Note.
Off-Balance Sheet Arrangements
We have no obligations, assets or liabilities which would be considered off-balance sheet arrangements. We do not participate in transactions that create relationships with unconsolidated entities or financial partnerships, often referred to as variable interest entities, which would have been established for the purpose of facilitating off-balance sheet arrangements.
We have not entered into any off-balance sheet financing arrangements, established any special purpose entities, guaranteed any debt or commitments of other entities, or entered into any non-financial agreements involving assets.
Contractual Obligations
At March 31, 2021, we did not have any long-term debt, capital lease obligations, operating lease obligations or long-term liabilities. On June 29, 2020, we entered into an administrative support agreement pursuant to which we have agreed to pay an affiliate of our Sponsor a total of $10,000 per month for office space, administrative and support services. Upon the earlier of the completion of the initial business combination and our liquidation, we will cease paying these monthly fees. For the three months ended March 31, 2021, we incurred expenses of $30,000 under this agreement.
The underwriters of our IPO are entitled to underwriting discounts and commissions of 5.5%, of which 2.0% ($15,000,000) was paid at the closing of our IPO and 3.5% ($26,250,000) was deferred. The deferred underwriting discount will be paid to the underwriters upon the completion of the initial business combination.
Critical Accounting Policies/Estimates
The preparation of financial statements and related disclosures in conformity with accounting principles generally accepted in the United States requires management to make estimates and assumptions that affect the reported amounts of assets and liabilities, disclosure of contingent assets and liabilities at the date of the condensed financial statements, and expenses during the periods reported. Actual results could materially differ from those estimates. We have identified the following as our critical accounting policies:
Net Income Per Common Share
Net income per share of common stock is computed by dividing net income by the weighted average number of common shares outstanding during the period. We apply the two-class method in calculating earnings per share.
232
Table of Contents
Accretion associated with the redeemable shares of GSAH Class A common stock is excluded from earnings per share as the redemption value approximates fair value.
As of March 31, 2021, we had outstanding warrants to purchase of up to 27,250,000 shares of GSAH Class A common stock. The weighted average of these shares was excluded from the calculation of diluted net income per share of common stock since the exercise of the warrants is contingent upon the occurrence of future events. As of March 31, 2021, we did not have any dilutive securities or other contracts that could, potentially, be exercised or converted into shares of common stock and then share in our earnings. As a result, diluted net income per common share is the same as basic net income per common share for the periods.
Redeemable Shares of GSAH Class A Common Stock
All of the 75,000,000 shares of GSAH Class A common stock sold as parts of the units in our IPO contain a redemption feature. In accordance with the Accounting Standards Codification 480-10-S99-3A (ASC 480), Classification and Measurement of Redeemable Securities, redemption provisions not solely within our control require the security to be classified outside of permanent equity. Ordinary liquidation events, which involve the redemption and liquidation of all of the entitys equity instruments, are excluded from the provisions of ASC 480. We classify all shares of GSAH Class A common stock as redeemable.
Warrant Liability
We account for the warrants in accordance with the guidance contained in Accounting Standards Codification 815 (ASC 815), Derivatives and Hedging, under which the warrants do not meet the criteria for equity treatment and must be recorded as derivative liabilities. Accordingly, we classify the warrants as liabilities at their fair value and adjusts the warrants to fair value at each reporting period. This liability is subject to re-measurement at each balance sheet date until the warrants are exercised, and any change in fair value is recognized in our statement of operations. The fair value of the private placement warrants have been estimated using a Black-Scholes-Merton model and the fair value of public warrants issued in connection with our IPO have been measured based on the listed market price of such public warrants.
Recent Accounting Pronouncements
Management does not believe that any recently issued, but not yet effective, accounting pronouncements, if currently adopted, would have a material effect on our financial statements.
233
Table of Contents
Unless the context otherwise requires, all references in this subsection to the Company, we, us or our refer to the business of the Mirion prior to the consummation of the Business Combination, which will be the business of the post-business combination company following the consummation of the Business Combination.
Business Overview
Mirion Technologies provides products, services and software that allow our customers to safely leverage the power of ionizing radiation for the greater good of humanity. Our solutions have critical applications in the medical, nuclear energy and defense markets, as well as in laboratories and scientific research, analysis and space exploration. Many of our markets are characterized by the need to meet rigorous regulatory standards, design qualifications and operating requirements. Throughout our history, we have successfully leveraged the strength of our expertise in ionizing radiation to continually drive innovation and expand the commercial applications of our core technology competencies. Through our facilities in 12 countries, we supply our solutions in the Americas, Europe, Africa, the Middle East and Asia Pacific regions.
We have two reportable business segments: Medical and Industrial. Our Medical segment supports applications in medical diagnostics, cancer treatment, practitioner safety and rehabilitation. Our products in these fields are focused on enhancing the effectiveness and safety of life-saving procedures. Our Industrial segment is focused on addressing critical radiation safety, measurement and analysis applications across nuclear energy, defense, laboratories and research and other industrial markets. Our products and solutions include: dosimetry services (environmental radiation monitoring dose of records services), cancer diagnostics and therapy quality assurance, or QA, nuclear medicine, dosimeters (wearable devices that measure exposure to ionizing radiation), contamination and clearance monitors, detection and identification instruments, radiation monitoring systems, electrical penetrations, reactor instrumentation and control equipment and systems, medical and industrial imaging systems and related accessories, software and services, alpha spectroscopy instruments (instruments that quantify and identify alpha-emitting nuclides), alpha/beta counting instruments (instruments for quantification of alpha and beta radiation) and gamma spectroscopy detector systems (instruments for qualification and quantification of gamma emitting nuclides) and software (related software to support our product and solution offerings).
For more than 60 years, we and our predecessor companies have delivered products and services that enable our customer to harness ionizing radiation for applications that benefit the health, safety, vitality and technological progress of humanity. We believe the breadth and proven performance of our solutions support our longstanding strategic customer partnerships across diverse end markets. Our products, software and services have been sold directly and indirectly to a variety of end-use customers, including, medical service providers, the vast majority of the U.S. nuclear power producers and the addressable global installed base of active nuclear power reactors, many of the leading nuclear reactor design firms, universities, numerous international government and supranational agencies, 19 of the 28 NATO militaries, national laboratories, environmental laboratories, research institutes and industrial companies.
Our broad product and services portfolio of medical, search, measurement, scientific analysis and reactor safety and control systems are supported by our engineering and research and development organization of 376 scientists, engineers and technicians, who represented approximately 14% of our workforce as of March 31, 2021. We possess numerous product qualifications, trade secrets and patents that support our market position and our ability to deliver next generation products and services. In addition, we maintain design, manufacturing and sales capabilities across 12 countries in America, Europe and Asia, enabling us to capitalize on growth opportunities, including the ongoing growth in spending for medical, defense and homeland security and the ongoing growth for nuclear power.
Our financial performance is driven by the replacement of products and the recurring provision of services into our core end markets, as well as the construction of new facilities like nuclear power plants, or NPPs, globally.
234
Table of Contents
Many of our products and services are ordered in advance of the anticipated shipment date and secured in backlog, which provides visibility into future revenue. For the nine months ended March 31, 2021 we generated revenue of $431.6 million, as compared to $337.0 million for the nine months ended March 31, 2020, an increase of $94.6 million or 28.1%. Adjusted revenues, excluding the acquisition accounting impact for deferred revenue was $435.9 million and $337.0 million for the nine months ended March 31, 2021 and 2020, respectively, an increase of $98.9 million or 29.3%. For fiscal 2020, we generated revenue of $478.2 million, as compared to $440.1 million for fiscal 2019, an increase of $38.2 million or 8.7%. For the nine months ended March 31, 2021, our net loss was $131.0 million and our Adjusted EBITDA was $102.0 million. In fiscal 2020 our net loss was $119.1 million and our Adjusted EBITDA was $114.6 million. At March 31, 2021, and June 30, 2020, Mirion had $908.5 million and $724.3 million, respectively, of third-party debt outstanding (excluding deferred financing costs). At March 31, 2021, and June 30, 2020, Mirion had $1,170.3 million and $987.1 million, respectively, of related-party debt outstanding (excluding accrued interest).
Industry Overview
We have two reportable business segments: Medical and Industrial. Our Medical segment is based around our sales, products and services to customers in the medical market. The Industrial segment is primarily based around the nuclear energy, defense, laboratories and scientific research markets as well other industrial markets.
Medical
Mirions medical market is comprised of rapidly growing product applications in cancer diagnostics and therapeutics, nuclear medicine, dosimetry services and rehabilitation. We offer products, software and services in each of these areas that enhance the effectiveness and safety of life-saving procedures in these areas. According to the World Nuclear Association, or WNA, as of April 2021, there are over 10,000 hospitals worldwide using radioisotopes in medicine, with about 90% of the procedures for diagnostics, and more than 40 million procedures are performed globally every year, 20 million being in the United States and 10 million in Europe. The WNA also estimates that, as of April 2021, the use of radioactive substances, or radiopharmaceuticals, in diagnosis is growing at over 10% per year. In the radiotherapy market, demand is driven by replacement of the underlying linear accelerator, or Linac, installed base. As of 2019 there were approximately 14,000 Linacs deployed worldwide, and it is estimated that this will grow to approximately 16,500 Linacs worldwide by 2024, according to a global consulting firm.
Nuclear medicine is a medical specialty that uses radiopharmaceuticals to diagnose, monitor and treat disease. Mirions products address the complicated lifecycle of radiopharmaceuticals from radiopharmaceutical production and handling through patient dosing, imaging, diagnosis and therapy with our line of dose calibrators, thyroid uptake systems, shielding systems, management software and supporting accessories.
Radiotherapy (also known as radiation therapy or radiation oncology), uses radiation in the form of X-rays, protons and electrons to destroy cancer cells and shrink tumors. Mirion provides both hardware and software products, as well as services to accomplish the critical task of performing independent quality management in the diagnosis and treatment of cancer. Our suite of patient, machine and diagnostic QA solutions are relied on in the field to mitigate errors, reduce inefficiencies, validate technologies and techniques, and elevate the quality of clinical care.
Medical imaging encompasses a number of technologies (MRI, Ultrasound, X-ray) that are used to view the human body to diagnose, monitor or treat medical conditions. Mirion provides support for these imaging techniques through our array of C-Arm and ultrasound tables, MRI accessories, positioners and radiation protection accessories.
As a result of the proliferation of radiological medical technologies, hospitals, clinics, and small dental and veterinary facilities rely on occupational dosimetry systems and services to ensure the safety of both medical personnel and patients. Mirions dosimetry services products like Instadose, provide instant dose measurement
235
Table of Contents
results when connected to any computer or mobile device via Bluetooth and ensure that radiation safety programs run smoothly and are easy to administrate.
Laboratories and Research
The laboratory and research market includes different types of facilities like environmental radiochemistry laboratories, research laboratories, research reactors and education laboratories. All these facilities analyze nuclear samples or monitor experiments to identify the chemical composition of the material involved or understand the basic structure of matter.
The environmental radiochemistry laboratories, or counting labs, monitor the environment by analyzing samples, measuring their radiation and identifying the source and the nature of contamination, if any. The laboratories can be governmental (e.g., health or environment institutions, safety authorities) or private (e.g., facility bio assay, process labs). We believe there are over 500 environmental laboratories worldwide based on our estimates as of December 2020.
Research centers include national laboratories and research institutions conducting research in the areas of space, underground studies, physics around synchrotrons and accelerators. Radiation measurement systems are used in research for the discovery of elements, to study the formation of matter after the big bang or in the deepest underground laboratories in the world to perform dark matter experiments. They are also used in space, mounted in satellites or robots, landing on planets (Mars Rover) or orbiting around Earth (STEREO), Saturn (Cassini), Venus and Mercury (Messenger), Pluto (New Horizons), Mars (MSL-Rad), and Jupiter (JUNO).
Research reactors are used for research and training, materials testing, medicine (like the production of radioisotopes) and industrial functions. According to the WNA, there were 220 operational nuclear research reactors in 53 countries, with 11 more under construction and 16 planned to be built, as of June 2021.
Education laboratories are located in universities and offer programs in nuclear engineering, health physics, radioprotection, nuclear physics or nuclear science and technology. We believe there are more than 600 colleges worldwide, universities and degree-granting institutions that are equipped with nuclear measurement products.
Nuclear
The nuclear end market spans the entire nuclear fuel cycle, including mining, enrichment, fuel manufacturing, nuclear power generation, waste management and fuel reprocessing. Key nuclear installations include mines, fuel fabrication facilities, commercial nuclear power reactors, reprocessing facilities, research facilities, military facilities and ships, weapons facilities and waste storage facilities.
We sell products and services for use in each of these types of installations at any stage of their life (construction, operation, decommissioning and dismantling), with commercial nuclear power reactors representing the majority of our sales into the nuclear end market. This market is segmented between new builds, installed base requesting upgrades/uprates/relicensing, and decommissioning and dismantling.
Driven by increasing demand for electricity and reliable and carbon-free energy, the nuclear power market is forecasted to grow in the near and long term, which presents opportunities for Mirion. These trends are further driven by global decarbonization goals, which are likely to increase the demand for nuclear power.
Despite some challenges in certain regions of the world (e.g., Germany, Sweden, Japan), the new build market is expected to be very dynamic with 55 reactors in construction and 425 planned or proposed as of May 2021 (according to the WNA). The installed base market presents opportunities while nuclear power plants, or NPPs, are being relicensed with extended life time and upgraded. Meanwhile, we believe that more than 50 reactors will be shut down by 2030, growing the demand for radiation equipment used in decommissioning and dismantling of nuclear facilities.
236
Table of Contents
Defense
Our global defense end market is driven by a combination of military, civil defense and event-driven security spending. The proliferation of global security threats has reached an unprecedented level, driven by an unstable geopolitical climate, the emergence and expansion of terrorist organizations, the development of nuclear weapons in non-nuclear countries and the proliferation of radiological and nuclear technologies. Taken together, these threats have the potential to cause significant human casualties and economic loss. As a result, militaries, civil defense and other security organizations have bolstered investment in the prevention and detection of radiological threats as well as in technologies capable of detecting and monitoring radiation levels in the aftermath of radiological attack.
Militaries throughout the world utilize radiation detection technologies for troop security. Spending on personnel protection and detection of radiological threats is a priority for both NATO and non-NATO militaries and, as such, has led many countries to provide dosimeters to military personnel on a standard-issue basis. We believe that spending on these technologies will remain a high priority among armed forces globally.
Spending within the global civil defense, or homeland security, market has rapidly expanded in recent years based on increased threats presented by terrorist organizations. As a result, civil defense, first responder and other security organizations are investing in technologies and services designed both to protect civil defense personnel, civilians and domestic infrastructure from radiological threats and to detect and monitor radiation levels following a radiological incident, such as the release of a nuclear or other radiological device.
In addition, homeland security organizations are increasingly focused on enhancing radiological detection capabilities at critical points of entry, such as airports, ports and borders. Large-scale public meeting events have also greatly increased security measures at facilities, including rapid adoption of radiological detection technologies to address the increased threat of radiological attacks, due to their profile as high visibility targets.
Industrial
Other end markets include industrial facilities such as cement kilns, pulp and paper mills and coal/gas fired power boilers that utilize high-temperature industrial processes. These high-temperature processes are critical to plant operation and must be accurately monitored to ensure optimal operating conditions. Imaging equipment capable of withstanding the high temperatures and environmental conditions found in these facilities is employed to monitor and optimize process efficiency. These imaging systems require routine replacement or upgrades.
Other end markets also include original equipment manufacturers, or OEMs for general industrial market or medical applications, using radiation measurement detectors to sort material or precisely locate some radioisotopes.
Our Market Opportunity
We believe that significant opportunities for growth exist within each of our primary end markets.
Medical
Radiological procedure growth. The use of radiodiagnostic and radiotherapeutic procedures is expanding globally due to aging population demographics, technological advancements and emerging middle classes in developing economies. As the use of radiological procedures increases in the medical industry, so does our associated market opportunity. According to a global consulting firm, we believe the global nuclear medicine market is expected to grow 7% per year from 2020 through 2030, primarily driven by the increase in the prevalence and incidences of cancer worldwide. Likewise, the global radiotherapy market is expected to grow 6% per year from 2020 through 2030, primarily driven by factors including growing awareness about the benefits of radiotherapy for cancer control and eradication, increasing incidence and prevalence of cancer, and
237
Table of Contents
technological advancements in the field of radiotherapy. Mirion plays in select sub-segments of the global nuclear medicine and radiotherapy markets. The growth trajectories in these markets represent significant market opportunities for Mirion products that are deployed in hospitals, clinics, and other diagnostic and therapeutic centers around the world.
Dosimetry outsourcing. In some regions outside the United States, dosimetry services for health care practitioners historically have been provided by government agencies. We believe that more government agencies are outsourcing dosimetry services to private providers due to favorable cost dynamics in some regions. This provides a market opportunity where we can leverage our technical expertise and North American service experience to expand into other regions as we have done through our acquisitions of state-owned dosimetry services businesses in the Netherlands and Germany. According to a global leading consulting firm, we believe Mirions core dosimetry market is expected to grow 3 to 4% per year from 2020 through 2026, primarily driven by volume increase in number of healthcare workers exposed to radiation and standard annual price increases. In addition, through the differentiating factors behind the innovative Instadose product line, we believe that we have the right product ecosystem to maximize this opportunity.
Laboratories and Research
Customer loyalty. Loyalty is driven by long standing relationships, customer hesitancy to switch suppliers, high switching costs and limited competition globally. We believe we can benefit from price growth in most of our markets. In addition, our business is well protected by consistent replacement cycles on installed base.
Nuclear
Our legacy in the nuclear industry positions us to capitalize on the growth in demand for radiation detection, measurement, analysis and monitoring products and services in each phase of the nuclear life cycle, as outlined in the chart below.
Mirion provides essential products and services to NPPs throughout the entire life cycle of a plant: from construction and operation to decommissioning and decontamination. For example, we provide:
| | Radiation measurement and monitoring solutions, such as detection portals, environmental monitors and dosimetry systems that are typically installed in nuclear facilities during construction and are replaced or upgraded during the entire lifetime of the reactors, in particular upon life extensions. This provides recurring revenue opportunities as customers must replace and upgrade components and services during this timeframe, |
| | Reactor instrumentation and control detectors that are typically installed in nuclear facilities during construction and are replaced or upgraded regularly. In addition, there are opportunities to provide |
238
Table of Contents
| more comprehensive upgrades of reactor instrumentation and control detector systems in certain existing reactors to facilitate up-rating, |
| | Measurement and expertise services including technical expertise and experienced staff to help customers address their nuclear measurement needs in every step of the measurement process from planning to operation to wind-down, |
| | Imaging systems and cameras for all stages of the nuclear lifecycle, from construction through operation, to decommissioning and waste management, and |
| | Waste management systems that are used during the lifetime of the reactors and are essential, in particular, in any decontamination and decommissioning project. |
We believe the following dynamics support the sustainability of our existing business and will drive new sources of organic growth.
Predictable upgrade, replacement and retirement cycles. Our radiation detection, measurement, analysis and monitoring products and systems have predictable life spans, typically ranging from four to twenty-five years. Our complex monitoring systems typically require at least one comprehensive upgrade during their useful life to optimize their functionality. In addition, many of our products require replacement parts, components and service due to normal wear during their useful lives.
Aging installed base. The existing global installed base of nuclear reactors has a median age of 34 years. This aging installed base requires frequent product replacements and upgrades over an operating life cycle that generally ranges from 40 to 80 years. Furthermore, as reactors reach the end of their useful lives, the onset of a multi-year decommissioning process represents a further revenue opportunity in the reactor life cycle for our products.
Increased decontamination and decommissioning activity and stricter environmental regulation. The total number of NPP shutdowns under decontamination and decommissioning is expected to increase over the next decade, with largest amount of expected plant shutdowns potentially in the U.S. market. In Europe, the UK represents the highest share of expected shutdowns as the operating fleet ages and passes the license extension period.
Large installed base of orphaned products and systems. Most currently operating reactors were commissioned prior to 1990. Operators of many aging NPPs often must consider new suppliers to meet their detection needs as many of the suppliers of legacy radiation detection, measurement, analysis and monitoring systems no longer service the nuclear industry.
Dosimetry outsourcing. NPPs have historically managed the majority of their dosimetry service requirements internally. However, the cost benefits of outsourcing these services have become increasingly attractive to NPP operators as they focus on improving profitability and enhancing service.
New build opportunity. We expect the construction of new nuclear reactors worldwide to provide opportunities across our product and service offerings. The nuclear industry is experiencing robust growth in activity related to new reactor builds. As of May 2021, there were 55 reactors under construction and 425 planned or proposed, according to the WNA. This growth is occurring internationally and our global footprint positions us to capitalize on these opportunities. Since the early stages of reactor development generally represent a material share of our revenue opportunity over the life cycle of a reactor, we are positioned to benefit from increased global reactor construction. In addition, as new plants are added to the global nuclear fleet, we believe our recurring revenue opportunity associated with replacements, spares, software, services and system upgrades will continue to increase as we are well-positioned with customers due to our incumbent position.
239
Table of Contents
Defense
Focus on military personnel. Global militaries must contend with radiological threats and the difficulties of protecting soldiers and monitoring areas of enemy engagement. The combination of our active dosimeters and telemetry technology provides a differentiated solution that addresses the radiation detection needs of modern militaries.
Increased civil defense spending on radiation detection. Civil defense and homeland security organizations are focused on preventing the illicit transportation of radiological materials across borders. The commercial application of our radiation detection expertise positions us to benefit from government spending on detection technologies.
Enhanced event specific security. The visibility of high profile events and venues has increased their value as targets of terrorist activity. In response, security spending at events, such as the Olympic Games, has increased, as has the utilization of radiation detection technology, providing an expanding market opportunity for our products.
Our Competitive Strengths
We believe that the following competitive strengths will enable us to maintain our position and capitalize on growth opportunities in our end markets:
Trusted ionizing radiation detection and measurement provider. Our end markets, including the medical, defense and nuclear industries, are highly regulated and require compliance with strict product specifications. Our track record enables us to gain market share across our product and service offerings. We and our predecessor companies have served the radiation detection measurement, analysis and monitoring needs of our customers for over 60 years, having developed trusted, recognized brands supported by our tradition of technical excellence, product reliability and customer service. We believe we have a leadership position in 14 of the 17 market segments we serve. In addition, we have leveraged our ionizing detection expertise to develop new applications for our core historical markets and to expand into adjacent markets through acquisitions.
Broad and complementary product and service portfolio. We are one of the only companies that offers ionizing radiation detection and measurement products and services to satisfy customer requirements throughout the medical and industrial markets. Our comprehensive product line supports virtually all radiation detection and monitoring needs associated with these markets. As a result, we believe that we have consistently gained market share as some of our key customers rationalize their supply chain. Furthermore, our portfolio provides us with a natural opportunity to cross-sell our products and services to our customers. As a result, we have a diversified portfolio across end markets and geographies.
Large installed base driving recurring revenue. We possess longstanding customer relationships in all of our end markets. For example, we estimate our QA products are used by 95% of cancer treatment centers in the United States and 65% globally. This drives recurring revenue and opportunities for cross sales from our other activities. Our products were also installed at the vast majority of the addressable installed base of active nuclear power reactors globally, which have a median age of about 34 years. This installed base drives recurring revenue through replacement and service cycles associated with our offerings and the typical 40 to 80 year operating life cycle of an NPP. The length and quality of supplier relationships are important customer buying criteria due to high switching costs and the importance of proven product reliability. In addition, we maintain relationships with global military and government organizations that value operating longevity and technological expertise. For example, our products have been sold to 19 of the 28 NATO militaries as well as the U.S. Departments of Energy, State, Defense and Homeland Security. Our customers focus on personnel protection drives their recurring expenditures on service, recalibration and product upgrades in our defense end market. In the laboratories and research markets, we have developed relationships with certain customers over the past 50 years, gaining their loyalty based on product performance and customer services. Such relationships provide us with recurrent revenues when our customers upgrade and replace their existing installed base.
240
Table of Contents
Technical complexity creates high barriers to entry. Across our end markets, we design our products to meet demanding customer specifications, qualifications and regulatory requirements. In many circumstances, we design our products to be compatible with highly complex facilities and operate effectively in harsh environments. Replicating Mirions products is difficult given underlying technical specifications. In addition, customers generally work with their incumbent suppliers to service, maintain and replace equipment over product lifetime resulting in a natural barrier to entry.
Global footprint designed to meet local customer needs. Our global footprint, augmented by our established network of suppliers and distributors, enables us to be responsive to our customers and provide locally customized solutions. We operate facilities in 12 countries, accommodating the desire of certain of our customers to procure products and services from local providers. Sales to customers outside of the United States and Canada accounted for 56% of total revenue for fiscal year 2020. We believe that our established global infrastructure provides a scalable platform to meet the growing worldwide demand for our products and services.
Proven M&A strategy and track record of integrating acquisitions. Mirion has been built through successive mergers and acquisitions. Since 2016, we have acquired and integrated ten companies. Through these acquisitions, we have developed tools and experience across deal sourcing, modeling and integrating acquired companies. We have a business ecosystem in place to identify and act upon cost saving opportunities as well as the ability to leverage our scale platform to capture cross-selling opportunities. Historically, we have consistently exceeded our synergy targets and improved profitability of acquired businesses.
Seasoned management team complemented by highly skilled engineers. We are led by an experienced management team with a mix of private sector and government experience across different industries and functions. Our divisional presidents have an average tenure of over 15 years. Our senior management team is complemented by an engineering and research and development organization of 376 scientists, engineers and technicians as of March 31, 2021. A number of our employees are participants in international and U.S. standards setting organizations related to radiation detection in the nuclear, defense and medical end markets. Through these activities, we help define the setting of standards and preview changes that impact our products, customers and end markets.
Our Strategy
Our objective is to continue enhancing our position as a global provider of radiation detection, measurement, analysis and monitoring products and services for the global medical and industrial end markets. We intend to achieve this through the following strategies:
Exploit under-penetrated market opportunities. We believe that we can exploit historically under-penetrated segments of our end markets by leveraging our existing positions across our major product categories. For example, we have leveraged our technical expertise to develop and commercialize innovative products to increase sales in the U.S. dosimetry services market and in the radiotherapy quality assurance market, and we have expanded our radiation monitoring solutions offering by leading integrated offers with other key suppliers for some nuclear new build projects in Europe to increase our scope of supply and gain share in the nuclear market.
Expand addressable market. We believe that substantial opportunities exist for us to expand our addressable market by marketing our products and services to customers in new geographic regions; providing products and services to customers moving to an outsource model; entering markets where the government is privatizing services; introducing new applications for existing technologies and pursuing strategic acquisitions.
| | Geographic expansion. Although we sold products and services to customers in 120 countries in fiscal 2020, we believe we have additional opportunities in certain international markets. For example, in India, we intend to leverage our relationships with leading reactor design firms to capitalize on the |
241
Table of Contents
| opening of the nuclear end market to U.S. and European firms. Another such market is the European dosimetry services market. Through acquisitions, we have developed our presence in the Netherlands and Germany, and we plan to continue expanding into other European countries. Other markets for expansion include the Middle East, Eastern Europe and the former Soviet Union, where we intend to increase our presence by leveraging relationships with local partners. |
| | Customer outsourcing. We believe we will continue to capitalize on customer outsourcing within the nuclear end market. Within the United States, several NPP operators have recently outsourced their dosimetry services in order to reduce costs. We have been able to benefit from economies of scale as well as advantages in materials procurement and processing technology to provide enhanced dosimetry services to many of these NPPs at a lower cost. |
| | Service privatization. In regions outside the United States, dosimetry services have historically been provided by government agencies. However, privatization of dosimetry services is occurring in some regions, such as Europe. As illustrated by our acquisitions in the Netherlands and Germany, providers seek to reduce costs and benefit from enhanced service offerings. This provides us with an opportunity to leverage our expertise and North American service experience, where we have demonstrated a strong track record of success, to expand market share in other geographies. |
| | Expand into new end markets. We periodically review our adjacent markets and identify opportunities for expansion. For example, we have developed a new personal radiation detector, or PRD, called Accurad to expand our presence in the civil services markets such as the police and fire departments. We have also entered in the nuclear imaging and radiotherapy markets through the acquisitions of Capintec, Biodex and Sun Nuclear in fiscal 2020 and 2021. |
| | New applications for existing technologies. A portion of our development effort is focused on adapting existing technologies to alternative applications. For example, we have adapted the technology used for the medical and nuclear markets to develop the Mirion Battlefield Dosimeter which is currently being deployed by the U.S. Army and the U.S. Navy. |
Develop new products and services. We believe that significant near-term opportunities exist for us to develop new products and services by capitalizing on our understanding of our customers needs and requirements. Cross pollination of technologies between end markets also drives new growth opportunities. For example, we created a new product called evrCAM to meet the needs of the radiation oncology market by leveraging our core technology from decades of experience in radiation tolerant cameras for the nuclear power industry.
Continuously improve our cost structure and productivity. As we continue to grow our business, we have implemented a coordinated program of ongoing operating improvements, such as optimizing our manufacturing footprint, rationalizing excess costs and minimizing working capital requirements. We are continuously implementing Mirions business system principles to challenge our practices and improve our performance across all our businesses. For example, we have optimized and simplified our footprint by transferring the activities from our facilities in Hamburg, Germany, in Oulu, Finland and in Horseheads, NY to other Mirion sites. Our global procurement team also delivers value across the business from sourcing of key materials and services to supply chain design.
Pursue strategic acquisitions. We have successfully integrated acquisitions to augment our organic growth. We were formed by the merger of Global Dosimetry Solutions, or GDS, Imaging and Sensing Technologies, or IST, and Synodys in 2005. In 2016, we acquired Canberra Industries, which is our largest acquisition to date. Between October 2018 and December 2020, we acquired eight companies, with the objective of complementing our portfolio, reinforcing our supply chain and expanding into new markets such as nuclear imaging and radiotherapy. Since then, we have effectively integrated these businesses, creating a global platform of ionizing radiation detection and measurement solutions. We continuously monitor potential acquisitions and intend to further complement our organic growth with selective acquisitions that enhance our existing products and services, strengthen our position with existing customers and enable us to expand into new markets.
242
Table of Contents
As of December 2020, we believe our core addressable market represents approximately $4 billion of annual revenue and we have identified eight attractive adjacent markets related to ionizing radiation technology with a total market size of approximately $13 billion that we believe we can expand into. In combination with our estimated $4 billion core addressable market, this represents a total estimated addressable market of $17 billion.
Adjacent markets for expansion were identified based on a set of criteria measuring the attractiveness of the segments (market growth, market profitability, valuation multiples, current market size), their accessibility (executability, possibility to become the leader of the segment, proximity with our core business) and their relation to ionizing radiation technology. Identified adjacent markets with approximate market sizes as of December 2020 (based on our estimates in consultation with a global consulting firm) include:
| | Adjacent markets within radiation therapy include external beam-proton (proton therapy equipment for treatment of tumors; $0.7 billion market size) and external beam-advanced stereotactic (equipment for radiotherapy systems which target the tumor from multiple angles; $0.7 billion market size). |
| | Adjacent markets within nuclear medicine include planar scintigraphy (diagnosis equipment to image the distribution of radioactive material within a patients body; $0.3 billion market size) and radiopharmaceuticals (market surrounding drugs containing radioactive isotopes for imaging and therapeutics purposes; $5.1 billion market size). |
| | Adjacent markets within ionizing radiation sterilization include equipment market (equipment used for sterilization procedures; $0.7 billion market size) and service market (sterilization as a service; $1.4 billion market size). |
| | Additional adjacent markets include non-destructive testing equipment market (equipment to detect flaws using radiation; $1.2 billion market size), X-ray screening security market (airport & civil security; $2.5 billion market size) and adjacent sub-system supplier markets (equipment for radiation therapy external beam Linacs, nuclear medicine 3D imaging and dental imaging; $0.8 - $1.1 billion market size). |
Our Segments
Medical
Our Medical segment encompasses five major product categories focused on supporting applications in medical diagnostics, cancer treatment, practitioner safety and rehabilitation. Our products in these fields are focused on enhancing the effectiveness and safety of life-saving procedures.
| | Cancer Diagnostics and Therapeutics QA: we provide integrated solutions for independent quality management in the diagnosis and treatment of cancer. Our suite of patient, machine, and diagnostic QA solutions are relied on in the field to mitigate errors, reduce inefficiencies, validate technologies/techniques and elevate the quality of clinical care. Our products include arrays for machine and patient QA solutions, software platforms for centralized analysis and data storage, lasers to align Linacs to patient or QA devices, and phantoms (devices to simulate the imaging and radiation dose absorption characteristics of human tissue) for machine and patient QA. |
| | Nuclear Medicine and Medical Imaging: we provide solutions for patient dosing, imaging, diagnosis and radiopharma production and handling as well as specialized medical imaging tables and accessories that support imaging techniques and procedures. Our products include our range of dose calibrators, radiation shielding, phantoms for quality assurance, phantoms, thyroid uptake systems, lung scan ventilation systems, ultrasound tables, C-Arm tables and accessories. |
| | Medical Imaging: we provide specialized medical imaging tables and accessories that support imaging techniques and procedures, including ultrasound tables, C-Arm tables and accessories. |
| | Dosimetry Services: our product offering is an information service, which provides environmental radiation monitoring services, as well as an official dose of record to employers and occupationally |
243
Table of Contents
| exposed employees, enhancing the effectiveness and efficiency of radiation safety programs at practitioner sites. Key product lines include the innovative Instadose dosimetry platform, optically stimulated luminescence, or OSL, dosimeters, and our range of eye, finger, and extremity dosimeters that integrate with our Dose Central data platform. |
| | Rehabilitation: we provide neuromuscular assessment and rehabilitation technology solutions. Our products are used to manage and rehabilitate the physical and performance deficits that cause functional limitations. Our technology safely progresses a patient through the physical rehabilitation progress. Our rehabilitation products are used in patients throughout the continuum of life from injuries requiring sports medicine and orthopedics to interventions for our aging population such as fall prevention and all ages with neurologic conditions due to strokes, Parkinsons Disease, spinal cord and traumatic brain injury. Our products include isokinetic testing and rehabilitation systems, balance assessment and rehabilitation, specialized gait training treadmills, body weight support training systems and upper, lower and total body ergometers. |
Industrial
Our Industrial segment is focused on addressing critical radiation safety, measurement and analysis applications across defense, nuclear energy, laboratories and research and other industrial markets.
Reactor Safety and Control Systems: we provide radiation monitoring systems and reactor instrumentation and control systems that ensure the safe operation of nuclear reactors and other nuclear fuel cycle facilities. Product lines include, but are not limited to, a range of areas such as effluent release and operational process monitors, as well as in-core and ex-core detector systems, electrical penetrations, boron meters, and nuclear containment seals. Select product categories include:
| | Radiation Monitoring Systems: sensors, displays, control electronics and software used for barrier leak control, effluent release monitoring, operational process monitoring and post event monitoring in NPPs, nuclear fuel cycle industry, research reactors and laboratories, military reactors and installations. |
| | Reactor Instrumentation and Control Equipment and Systems: sensors, cables and electronics designed to monitor radiation and temperature within a reactor core and in surrounding areas. |
| | Neutron Flux Measurement Systems: sensors, displays, control electronics and software used to control the core of a reactor in NPPs, research reactors, and military reactors. |
Radiological Search, Measurement and Analysis Systems: we provide solutions to locate, measure and perform in-depth scientific analysis of radioactive sources for radiation safety, security, and scientific applications. Product portfolios include but are not limited to our laboratory and scientific analysis systems (gamma/alpha spectroscopy, alpha/beta counting, specialty detectors, spectroscopy software), radiation measurement and health physics instrumentation (contamination and clearance monitors, portable radiation measurement, electronic dosimetry, telemetry, waste measurement) and search and radiological security systems (Military CBRNE, or Chemical, Biological, Radiological, Nuclear and high-yield Explosives, security and search). We also provide a wide range of on-site managed and professional services to our end market customers. Select product categories include:
| | Dosimeters: active and passive dosimeters which monitor radiation dose rate and cumulative dose, along with readers, calibrators, telemetry, software and other accessories. |
| | Contamination and Clearance Monitors: stationary systems designed to detect radioactive contamination of people, waste, tools, laundry, vehicles and cargo. |
| | Detection & Identification Devices: hand-held and fixed devices to detect and locate ionizing radiation. |
| | Customized Research Detectors: highly customized detectors for scientific research, including nuclear physics research, space and synchrotron applications, and ruggedized detectors. |
244
Table of Contents
| | Environmental Monitoring Systems: sensors, displays, control electronics and software used for environmental monitoring in NPPs, nuclear fuel cycle industry, research reactors and laboratories, military reactors and installations. |
| | Radiochemistry: high precision instruments for detection and analysis of sample radioactivity, identification of radionuclide and quantification of activity used in laboratories, research, education, defense and NPPs. |
| | Imaging Systems: radiation-hardened imaging systems for nuclear fuel handling, control, monitoring and inspection; reactor vessel maintenance; underwater surveillance; tank and vessel inspection; and cameras for remotely operated vehicles. |
| | Waste measurement systems: systems to measure the radioactivity content of waste such as gamma neutron counting systems, non-destructive assay systems and neutron counting systems |
| | Services: we offer services to measure and analyze nuclear material more efficiently, calibration services, customer training programs, installation of instruments and software, technical support and repairs for Mirion products, as well as local operational support, technical support, and a wide range of consulting services |
Competition
The global markets for our products and services are competitive and continually evolving. Within each of our operating segments, we encounter a variety of competitors, ranging from small independent companies providing niche solutions to larger multi-national corporations providing a broader set of products and services to our targeted end markets. We believe that the principal bases upon which we compete in our target end markets include product quality and reliability, technical capability and product qualification, strength of customer relationships, customer service and price. In particular, customers in the defense and nuclear end markets tend to emphasize product quality and reliability, technical capability and strength of supplier relationships, while customers in the medical end markets, in particular for passive dosimetry products and services, tend to make purchasing decisions based on a combination of brand recognition, price, service and reliability.
We believe the primary competitors in each of our segments are as follows:
| | Medical: Landauer (Fortive), PTW, IBA, Standard Imaging, Comecer and LAP |
| | Industrial: Thermo Fisher Scientific, Ortek (Ametek), FLIR (Teledyne), Framatome, Ludlum, Fuji Electric, Caen System, Fluke (Fortive) and Berthold Technologies |
Research and Development
Our research and development efforts allow us to introduce new products to the marketplace, fulfill specific customer needs and continue to meet qualification requirements and other evolving regulatory standards. Our Medical and Industrial segments are committed to both technology research and product development to fulfill their strategic objectives and are supported by our engineering and research and development organization consisting of 376 scientists, technicians and engineers, representing approximately 14% of our total workforce, as of March 31, 2021. A number of these individuals participate in international standards setting organizations and committees. We engage in research and development activities at most of our facilities worldwide.
Our research and development expenses were $15.9 million, $14.0 million and $19.3 million for fiscal 2020, 2019 and 2018 and $21.2 million and $12.3 million for the nine months ended March 31, 2021 and 2020. We conduct these efforts through a mix of in-house research, collaboration with academia, customers and regulatory authorities as well as selected outsourcing through external vendors. The scope and extent of the outsourced portion of research and development activities vary by segment but typically, critical hardware design, software development and project management activities are conducted in-house while specialized services such as consulting services, algorithm design, thermal analysis, complex modeling and calculations and testing services are provided by third parties.
245
Table of Contents
Sales and Marketing
We sell our products and services through our direct sales organization and indirectly through our global network of independent, third-party sales representatives and distributors. Our internal sales team is organized by operating segment and end market to provide a higher level of service and understanding of our customers unique needs. We have 14 sales offices throughout North America, Europe and Asia, and as of March 31, 2021, our sales and marketing personnel consisted of 239 employees, which represents approximately 9% of our total workforce.
We derive a portion of our revenue from sales of our products and services through channel partners, such as independent sales representatives and distributors. In particular, our independent sales representatives are an important source of sales leads for us and augment our internal resources in remote geographies. We sell through distributors in situations in which our customers prefer to purchase from a local business entity or purchase in smaller volume.
Our marketing activities include participation in many tradeshows worldwide across our defense, medical and nuclear end markets. We advertise in technical journals, publish articles in leading industry periodicals and utilize direct mail campaigns.
Except when prevented by exceptional circumstances (for example, the COVID-19 crisis), we host our Mirion Connect Seminar annually, where customers participate in a variety of programs designed to exchange ideas and discuss occupational challenges. The event also brings together key channel partners and vendors to strengthen our sales and marketing network. Attendees gain insight into our product plans and participate in interactive sessions that give them the opportunity to better understand our current suite of products and services as well as provide feedback on our product roadmap.
Our Customers
Our principal customers include hospitals, clinics and urgent care facilities, dental offices, veterinary offices, radiation treatment facilities, OEMs for radiation therapy, laboratories, military organizations, government agencies, industrial companies, power and utility companies, reactor design firms and NPPs. We have long-standing relationships with our customers. For fiscal 2020, no customer accounted for greater than 10% of our consolidated revenue, our top ten customers together accounted for approximately 30% of our consolidated revenue, and our top five customers represented 21% of our consolidated revenue.
Manufacturing and Supply Chain
Given the diversity of our products, we employ numerous manufacturing techniques, including high-volume process manufacturing, discrete manufacturing, cellular manufacturing and hybrid approaches. Our production personnel engage in manufacturing, procurement and logistics activities. Our production activities are located in the United States, Canada, France, Germany, Belgium, Estonia, Finland and the United Kingdom. As of March 31, 2021, our production personnel consisted of 1,135 employees, which represents approximately 42% of our total workforce.
Our manufacturing activities are focused mainly on the production of the core value-add devices and components of our products, while non-core components and sub-assemblies are generally outsourced. This strategy enables us to protect important intellectual property and trade secrets while minimizing the time, cost and effort to produce commoditized components. Most of the time, the design, assembly and integration of the components are performed in-house, allowing our engineers to customize the products according to customer specifications. For highly engineered nuclear products, production volumes are typically low. For other product lines, such as, the DMC 3000 Electronic Dosimeter, the Mirion Battlefield Dosimeter, Accurad PRD and the Instadose dosimeter, production volumes tend to be higher. We apply rigorous quality control processes and calibrate
246
Table of Contents
radiation detection devices internally, leading to high quality standards and customization capabilities. Most of our production sites are certified to production quality standards such as those of ISO 9001, the U.S. Nuclear Regulatory Commission (10 C.F.R. 50 Appendix B) and the American Society of Engineers (ASME NQA-1).
The principal materials used in our manufacturing processes are commodities that are available from a variety of sources. The key metal materials used in our manufacturing processes include precious metals, tungsten, copper, aluminum, magnesium products, steel, stainless steel and various alloys, which are formed into parts such as detectors, sensors, metal housings and frames, and cable assemblies. The key non-metal materials used in our manufacturing processes include amorphous and crystalline scintillator materials, ceramics, epoxies, silicon and fused silica, polyethylene, polyurethane and injection molded plastic parts and components such as lenses, monitors, sensors, dosimeters, electronic boards, detectors and cables.
Properties
We maintain offices and manufacturing facilities at approximately 38 locations, in 12 countries. Our principal executive offices are located at 1218 Menlo Drive, Atlanta, Georgia. We are also a lessee under a number of operating leases for certain real properties, including our principal executive offices, and equipment. In addition to our executive offices, our principal properties include two facilities in Lamanon, France and our facility in Meriden, Connecticut. We lease our principal executive offices in Atlanta, Georgia, and our lease expires in 2031. We own our facilities in Lamanon, France and in Meriden, Connecticut. Management believes that the existing manufacturing facilities are adequate for our operations and that the facilities are maintained in good condition. We do not anticipate difficulty in renewing leases as they expire or in finding alternative facilities.
Intellectual Property
The success of our business depends, in part, on our ability to maintain and protect our proprietary technologies, information, processes and know-how. We rely on a combination of intellectual property rights, including trade secrets, patents, copyrights and trademarks, as well as contractual protections, to protect our proprietary products, methods, documentation and other technology.
As of June 30, 2021, we own approximately 76 issued U.S. utility patents, 95 issued foreign utility patents (including in Canada, the European Union, Russia, China and Japan), four pending U.S. utility non-provisional patent applications, four pending foreign utility patent applications (including in the European Union and France) and two pending Patent Cooperation Treaty, or PCT, patent applications. These issued patents are expected to expire between 2021 to 2038 and these pending applications, if issued, are expected to expire between 2039 to 2040, in each case without taking into account any possible patent term adjustment or extensions and assuming payment of all appropriate maintenance, renewal, annuity, or other governmental fees. Mirion does not expect the expiration of any of the patents that are scheduled to expire in 2021 to have a material impact on its business. These patents include one co-owned issued U.S. patent and three co-owned issued foreign patents. We also hold exclusive and non-exclusive licenses related to patents and other intellectual property of third parties. We also own trademark registrations or registration applications for Mirion in the United States and in certain foreign jurisdictions.
Medical Segment
As of June 30, 2021, we own approximately 37 issued U.S. utility patents, 26 issued foreign utility patents (including in the European Union, China, Japan and Canada), three pending U.S. non-provisional utility patent applications and one pending foreign utility patent application in the European Union that include claims directed to products in our medical segment, including our cancer diagnostics and therapeutics QA, occupational dosimetry, medical imaging and nuclear medicine equipment products. These issued patents are expected to expire between 2021 to 2038 and these pending applications, if issued, are expected to expire between 2039 to 2040, in each case without taking into account any possible patent term adjustment or extensions and assuming payment of all appropriate maintenance, renewal, annuity, or other governmental fees.
247
Table of Contents
Industrial Segment
As of June 30, 2021, we own approximately 40 issued U.S. utility patents, 69 issued foreign utility patents (including in the European Union, Canada, Russia and Japan), one pending U.S. non-provisional utility patent application, three pending foreign utility patent applications (including in the European Union and France) and two pending PCT patent applications that include claims directed to products in our industrial segment, including our alpha/beta counting instruments, contamination and clearance monitors, gamma spectroscopy software and detector systems, NDA and waste measurement systems, portable radiation measurement instruments, radiation monitoring systems and reactor instrumentation and controls products. These issued patents are expected to expire between 2021 to 2037 and these pending applications, if issued, are expected to expire between 2032 to 2040, in each case without taking into account any possible patent term adjustment or extensions and assuming payment of all appropriate maintenance, renewal, annuity, or other governmental fees. These patents include one co-owned issued U.S. patent and three co-owned issued foreign patents.
In many instances, we rely on trade secret protection and confidentiality agreements to safeguard our interests. Due to the long useful life of certain aspects of our technology, we believe that the patent registration process, which requires public disclosure of patented claims and inventions, could harm our competitive position. We differentiate our products and technologies primarily through our proprietary know-how, technology or data that are not covered by patents or patent applications, including technical processes, equipment designs, testing and other procedures. Our employees are generally required to assign to us all of the inventions, designs and technologies they develop during the course of employment with us, either through written agreements or by operation of law, depending on the jurisdiction. Where appropriate, we require third parties with whom we deal to enter into agreements with us that address issues of confidentiality and intellectual property. For a discussion of the risks and uncertainties affecting our business related to our protection of intellectual property and other proprietary information, please see Risk FactorsLegal and Regulatory Risks.
Environmental Matters
We are subject to a variety of environmental, health and safety and pollution-control laws and regulations in the jurisdictions in which we operate. We use, generate, discharge and dispose of hazardous substances, chemicals and wastes at some of our facilities in connection with our product development, testing and manufacturing activities. In addition, some of our facilities are located on properties with a history of use involving hazardous substances, chemicals and wastes and may be contaminated.
Under the Comprehensive Environmental Response, Compensation and Liability Act of 1980, or CERCLA (also known as the Superfund Law) and its state analogues, we may be subject to joint and several liability for environmental investigations and cleanups, including at properties that we currently or previously owned or operated, or at sites at which waste we generated was disposed, even if the contamination was not caused by us or was legal at the time it occurred. Although we have not incurred any material liabilities in connection with contamination, we may be required to make expenditures for environmental remediation in the future with respect to contamination at our or our predecessors former or current facilities or at third-party waste disposal sites under these laws. The Resource Conservation and Recovery Act of 1976 as amended by the Hazardous and Solid Waste Amendments of 1984, or RCRA, provides a comprehensive framework for the regulation of hazardous and solid waste which applies to our operations. RCRA prohibits improper hazardous waste disposal and imposes criminal and civil liability for failure to comply with its requirements. The Toxic Substances Control Act of 1976, or, TSCA provides a comprehensive framework for the management by the EPA of over 60,000 commercially produced chemical substances, some of which are used by our operations. The Clean Water Act regulates the discharge of pollutants into certain waters and may require us to apply for and obtain discharge permits, conduct sampling and monitoring and, under certain circumstances, reduce the quantity of pollutants in those discharges. The Occupational Safety and Health Act, or OSHA provides for the establishment of standards governing workplace safety and health requirements, including setting permissible exposure levels for hazardous
248
Table of Contents
chemicals. We must follow OSHA standards, including the preparation of material safety data sheets, hazardous response training and process safety management, as well as various record-keeping, disclosure and procedural requirements.
Our operations outside the United States are subject to similar, and sometimes more stringent, laws and regulations. For example, an EU directive relating to the restriction of hazardous substances in electrical and electronic equipment, or RoHS directive, and a directive relating to waste electrical and electronic equipment, or WEEE directive, have been implemented in EU member states. Among other things, the RoHS directive restricts the use of certain hazardous substances in the manufacture of electrical and electronic equipment and the WEEE directive requires producers of electrical goods to be responsible for the collection, recycling, treatment and disposal of these goods. China and South Korea have laws similar to the RoHS and WEEE directives. In addition, the EU has a regulation regarding the registration, authorization and restriction of chemical substances in industrial products, or REACH. REACH and other regulations requires us or our suppliers to substitute certain chemicals contained in our products with substances the EU considers less dangerous. See Risk FactorsLegal and Regulatory RisksWe could incur substantial costs as a result of violations of, or liabilities under, environmental laws.
Regulation
We are subject to a variety of laws and regulations, including but not limited to those of the United States, Canada, the EU, the EU member states and the Peoples Republic of China, that impose regulatory systems that govern many aspects of our operations. In addition, these jurisdictions impose trade controls requirements that restrict trade to comply with applicable export controls and economic sanctions laws and requirements, and legal requirements that are intended to curtail bribery and corruption. These laws and regulations apply by virtue of the nature of our industry, end markets and products, as well as the range of potential uses of our products, the origin of the technology incorporated into our products, and the jurisdictions in which we produce and sell our products.
The multi-jurisdictional legal and regulatory environments in which we operate are subject to extensive and changing laws and regulations administered by various national, regional and local governmental agencies both within and outside the United States.
We are a federal government contractor and, as such, we are subject to Executive Order 11246 and other relevant laws and regulations. As part of our compliance obligations, we implement on an annual basis an affirmative action plan and program which, in part, include our good faith efforts to achieve in our workforce full utilization of qualified women and minorities. In addition, we have in place an affirmative action plan with respect to disabled individuals, as well as Vietnam era, disabled or other veterans.
Some of the U.S. laws affecting our operations include, but are not limited to, the Atomic Energy Act, or AEA, the Energy Reorganization Act of 1974, or ERA, as well as the state laws governing radiation control in the states of New York, Georgia, California, Connecticut, Tennessee, New Jersey, Florida and Wisconsin, each as from time to time amended. We are also subject to a variety of U.S. federal and state employment and labor laws and regulations, including the Americans with Disabilities Act, the Federal Fair Labor Standards Act, the Worker Adjustment and Restructuring Notification Act, or WARN Act, which requires employers to give affected employees at least 60 days notice of a plant closing or mass layoff, and other regulations related to working conditions, wage-hour pay, overtime pay, employee benefits, anti-discrimination and termination of employment. We are also subject to the employment and labor laws and regulations of the foreign jurisdictions where many of our employees are located. The classified work that we currently perform at one of our U.S. facilities subjects us to the industrial security regulations of the Department of Defense and other federal agencies that are designed to safeguard against unauthorized access by foreigners and others to classified and other sensitive information.
In the United States, the AEA and ERA authorize the NRC, and state authorities where applicable, to regulate the receipt, possession, use and transfer of radioactive materials. The NRC, and state authorities where applicable,
249
Table of Contents
sets regulatory standards for worker protection and public exposure to radioactive materials or wastes to which we are required to adhere in our operations that use radioactive materials in research and development, product manufacture, testing and calibration.
Certain of our products require the use of radioactive sources. For certain of our products, these radioactive sources are often obtained by our customers directly from third-party providers, and for others, we directly incorporate these radioactive sources into our products. Certain of our reactor instrumentation and control equipment and systems for NPPs incorporate radioactive materials. In all such cases, licenses for radioactive sources and materials are provided by the appropriate regulatory authority in the relevant jurisdiction and such authorities may be at the state or national level. For example, at our sites in the United States that handle radioactive sources or materials, the appropriate licenses are issued by state-level authorities which are, respectively, the New York State Department of Health, Georgia Department of Natural Resources, California Department of Public Health, Connecticut Department of Energy & Environmental Protection, New Jersey Department of Environmental Protection, Tennessee Department of Environment and Conservation, Florida Department of Health and Wisconsin Department of Health Services. Similarly, licenses for radioactive sources and materials are maintained at each of our international sites where such licenses are required, including in Belgium, China, Canada, Estonia, Finland, Germany, France, Japan and the Netherlands.
While the specific process and criteria for receiving a license differ from jurisdiction to jurisdiction, it generally involves an application process in which we: identify a person or persons who have appropriate training and experience to be a health physics/radiation safety officer; specify the radioactive sources or materials sought to be licensed, their physical form (i.e., sealed or unsealed) and maximum possession limits on the amount of each type of radioactive element or compound sought under the license; specify their intended use (e.g., calibration, testing, quality assurance, manufacturing); and, set forth written policies and procedures to ensure that we have adequate measures in place to ensure health and safety. These policies and procedures typically must be designed to ensure worker, workplace, and public safety, including emergency plans; set forth the proper handling, control and security of radioactive sources or materials on site; detail any disposal or decommissioning considerations; and adequately train personnel at the site in proper access to, and handling of, radioactive sources or materials.
The particular license requirements in a given jurisdiction are normally tailored to the specific radioactive elements or compounds involved, their physical form, and possession limits. Once authorities complete their application review and any required follow-up, the authority issues the site a license which imposes specific on-going compliance obligations that typically include requirements for us to pay periodic licensing fees, submit periodic written compliance reports, and agree to periodic site inspections by regulators, which may be announced or unannounced. Once a site has an existing license, the process for expanding or reducing the licensing scope generally is simpler than applying for a new license.
We have numerous licenses in effect at our various facilities in the United States, Canada, Finland, Germany, France, China, Japan, the Netherlands, Belgium and Estonia and the expiration dates of individual licenses differ by their term and effective date. Typical license terms range from two to five years, with authorities in some jurisdictions (e.g., Finland and Bavaria, Germany) issuing licenses that are perpetual subject to our on-going license compliance. For radioactive materials licenses in the United States, preapproval is generally required from the NRC or Agreement State regulator before a direct or indirect transfer of a license, whether done through a sale or acquisition, restructuring, or other method. While specific regulations vary by jurisdiction, generally a license may be terminated by the regulatory authority immediately upon a finding of a substantial safety violation or other material violation of licensing requirements. For more minor violations, regulatory authorities typically provide the licensee with a written statement of deficiency or notice of violation stating required remediation steps, or requesting the licensee to identify corrective actions, and a demand for proof of remediation; depending on the severity of the violation, a re-inspection of the site may be performed by the authority to ensure adequate remedial steps have been completed.
In most cases, our various sites (including our predecessors) have held, maintained and (where required) renewed their licenses for a decade or more. In all cases, the licenses we require related to radioactive sources or materials
250
Table of Contents
are current and in force and, to the best of our knowledge, we are not aware of any basis to expect that any existing licenses subject to periodic renewals will not be renewed.
As a supplier of equipment and systems to the nuclear power industry, we are subject to regulations promulgated by the NRC that are applicable to vendors. Owners of nuclear power plants in the United States are licensed to build, operate, and maintain those plants by the NRC. Their license and applicable NRC regulations require that they qualify their suppliers and contractors to ensure that the suppliers and contractors comply with NRC regulations. The NRC has a robust inspection regime for commercial nuclear plants, which includes verification that, for example, design, procurement, maintenance, and radiation protection programs comply with NRC safety and quality assurance regulations and requirements. Inspections of nuclear materials licensees are conducted frequently, in areas such as personnel training, radiation protection, and security of nuclear materials. Parts of the NRCs inspection regimeincluding portions of 10 C.F.R. Part 21 on reporting of defects and noncompliance and Appendix B of 10 C.F.R. Part 50 related to Quality Assuranceare also directly applicable to contractors, suppliers, and other non-licensees. The NRC routinely conducts inspections at vendor sites on these matters and others. As a supplier to the nuclear power industry, we must demonstrate to our customers that we comply with NRC regulations related to quality assurance, reporting of defects and safety issues, security and control of personnel access and conduct. Section 170 of the AEA, which is known as the Price-Anderson Act, supports the nuclear services industry by offering broad nuclear liability and insurance coverage and indemnification to commercial NPP operators and their suppliers, as well as Department of Energy, or DOE, contractors, for liabilities arising out of nuclear incidents at power plants licensed by the NRC and at DOE nuclear facilities. The indemnification authority of the NRC and DOE under the Price-Anderson Act was extended through 2025 by the Energy Policy Act of 2005. Our nuclear power plant customers are covered by the nuclear liability insurance and indemnification provisions of the Price-Anderson Act. In addition, other jurisdictions have similar nuclear liability protection and indemnification regimes for nuclear facilities.
We deal with numerous U.S. and non-U.S. government agencies and entities, including the U.S. military, the armed forces of many NATO countries, the U.S. Department of Defense, the U.S. Department of State, the U.S. Department of Treasury, the U.S. NRC, the U.S. Department of Energy, the U.S. Department of Homeland Security and the corresponding governmental agencies and entities in the European Union and Canada. When working with these and other government agencies and entities, we must comply with, and are affected by, laws and regulations relating to the formation, administration and performance of contracts. These laws and regulations, among other things require certification and disclosure of all cost or pricing data in connection with various contract negotiations; impose acquisition regulations that define allowable and unallowable costs and otherwise govern our right to reimbursement under various cost-based U.S. government contracts; and restrict the use and dissemination of information classified for national security purposes and the exportation of certain products and technical data.
Export Controls
Our products and technologies are subject to export controls under the laws of the United States, Canada, the United Kingdom and the member states of the European Union. Depending on a number of factors, including the specific product or technology, the origin of that product or technology, the destination, the end-user and the end-use, exports of our products and technologies may require export licenses, permits or other authorizations from government export control authorities. Whether we will be able to conclude proposed transactions involving products or technologies that are subject to those export licensing requirements will depend on the relevant government agencys determination on whether the proposed transaction is consistent with the exporting countrys national security and foreign policy interests.
As examples of export control laws and regulations potentially applicable to our products and technologies, our products, when manufactured in or exported from the United States, are subject to export controls under the U.S. Department of Energys Part 810 regulations (10 C.F.R. Part 810) governing transfer of commercial nuclear technology and assistance, the U.S. Commerce Departments Export Administration Regulations, or EAR, the
251
Table of Contents
U.S. State Departments International Traffic in Arms Regulations, or ITAR, or the Nuclear Regulatory Commissions, or NRC, export licensing regulations in 10 C.F.R. Part 110 governing exports of nuclear materials and equipment. Canadian and EU export control regimes have separate, sometimes overlapping requirements, which must also be considered for a proper export compliance system.
We have implemented detailed export control compliance procedures, in the form of our Export Management and Control Program, EMCP, to identify those products, technologies and transactions for which export licenses, permits or other authorizations are required, and to assure that all transactions are handled in accordance with all applicable export control laws and regulations. Among other things, the Mirion EMCP includes (i) third party service provider screening of all parties against the various governments lists of prohibited, restricted and sanctioned parties; (ii) end-use reviews and certification procedures; (iii) monitoring regulatory announcements; and (iv) periodic reviews of applicable export control regulations in order to assure that the compliance procedures are up to date and properly maintained. See Risk FactorsLegal and Regulatory RisksLegal compliance with import and export controls, as well as with sanctions, in the United States and other countries, is complex, and compliance restrictions and expenses could materially and adversely impact our revenue and supply chain.
Economic Sanctions
Various United States laws and regulations implemented by the U.S. Treasury Departments Office of Foreign Assets Control, or OFAC, impose economic sanctions on certain countries, business entities and individuals. Those OFAC economic sanctions regulations: (i) impose comprehensive commercial and financial embargoes on transactions directly and indirectly with Cuba, Iran, North Korea, Syria or the Crimean Region, including any entity or person located in those jurisdictions; and (ii) include a very substantial list of persons and entities that have been determined to be closely affiliated with the government of an embargoed country, engaged in or supporting international terrorism, trafficking in narcotics, engaged in activities related to the proliferation of weapons of mass destruction, or otherwise acting in a manner contrary to United States foreign policy interests. United States persons (i.e., United States citizens, permanent residents and companies) are generally prohibited from engaging in any transaction which involves any property or any interest in property in which an embargoed country, a person in an embargoed country or a person on the OFAC list of sanctioned parties has an interest. The prohibitions on engaging in transactions with Cuba and Iran also extend to foreign subsidiaries of United States companies. Moreover, no United States person may approve, ratify, participate in, or otherwise facilitate any offshore transaction between a foreign company and any country, entity or person that is sanctioned under the OFAC economic sanctions regulations. The Department of Commerces Bureau of Industry and Security, or BIS, keeps an Entity List and other sanctions-related lists that are separate from the OFAC requirements.
Violations of United States export control regulations or the OFAC economic sanctions regulations are punishable by criminal and civil fines, imprisonment, loss of export privileges, debarment from United States Government contracts and, in extreme cases, listing on the OFAC list of sanctioned parties. See Risk FactorsLegal and Regulatory RisksLegal compliance with import and export controls, as well as with sanctions, in the United States and other countries, is complex, and compliance restrictions and expenses could materially and adversely impact our revenue and supply chain.
Anti-Corruption Laws
We are subject to anti-bribery and anti-corruption laws, including the United States Foreign Corrupt Practices Act, or FCPA, the United Kingdom Bribery Act, or UKBA, and anti-corruption laws enacted in various other countries which implement the Organization of Economic Cooperation and Developments, or OECDs, Convention on Combating Bribery of Foreign Officials in International Business. Those laws generally prohibit any person or company from making payments to any foreign official for the purpose of obtaining or retaining business or obtaining any other unfair or improper advantage.
252
Table of Contents
In particular, the FCPA prohibits any publicly traded company, or issuer, and any domestic concern from paying or giving, or promising or offering to pay or give, any money or any other thing of value directly or indirectly to a foreign official for the purpose of obtaining or retaining any business or obtaining any other unfair advantage. An issuer or domestic concern may be liable for penalties for violation of the FCPA if it make a payment, or provides any other thing of value, to a third party, such as a distributor, sales representative or other third party with knowledge that some or all of that money or thing of value will be paid or given to a foreign official for an improper purpose. In addition, the FCPA imposes upon issuers obligations to maintain complete and accurate books and records of account and to establish internal accounting controls, in order to prevent the diversion of corporate funds to the payment of bribes and other improper payments, and to prevent the establishment of off books accounts that might be used to fund improper payments to foreign officials.
Violations of the FCPA are punishable by criminal and civil fines and imprisonment and disgorgement of revenues derived from improper conduct. Any investigation or proceeding involving allegations of improper payments under the FCPA could have a material adverse effect on our business, financial condition, results of operations, standing with customers, particularly government customers, and/or business reputation. See Risk FactorsLegal and Regulatory RisksWe must comply with the FCPA and analogous non-U.S. anti-bribery statutes including the UKBA. Our or our sales representatives or distributors failure to comply with such laws could subject us to, among other things, penalties and legal expenses that could harm our reputation and materially and adversely affect our business, financial condition and results of operations.
Compliance Procedures
To address the compliance challenges presented by the foregoing laws and regulations, we have adopted and implemented compliance policies and detailed compliance procedures. Our commitment to compliance with anti-corruption laws and regulations is memorialized in the Mirion Code of Ethics and Conduct, which sets forth our overall compliance policies and informs all Mirion employees of their compliance responsibilities. Our export controls and economic sanctions compliance policies are set forth in our Export Management and Compliance Program, or EMCP, and implemented at each of our sites via local procedures. Our compliance programs are reinforced with (i) ethics and compliance training for all employees; (ii) due diligence reviews of all prospective distributors, sales representatives and other third party intermediaries; (iii) detailed anti-corruption compliance contractual covenants in third-party agreements; (iv) detailed recordkeeping procedures; and (v) auditing of third parties business practices as needed.
Medical Device Regulation
We are required to register for permits and/or licenses with, obtain approvals from and comply with operating standards of the U.S. Food and Drug Administration, or FDA, the NRC, the U.S. Department of Health and Human Services, or HHS, the European Medicines Agency, or EMA, the U.K. Medicines and Healthcare Products Regulatory Agency, or MHRA, and other foreign agencies, and accrediting bodies depending upon the type of operations we are conducting and the location of product distribution, manufacturing and sale.
Many of our products--for instance our nuclear medicine products for cardiology, oncology, endocrinology, diagnostic radiology and radiation therapy; imaging products in the form of positioning devices, ultrasound tables and MRI stretchers; and our energy measurement products, including radiation monitoring and measuring instrumentsin the medical end market are classified as medical devices and are subject to restrictions under domestic and foreign laws, rules, regulations, self-regulatory codes, circulars, and orders, including, but not limited to, the U.S. Food, Drug, and Cosmetic Act, or FDCA. We incur a number of costs associated with obtaining and maintaining the approval to market our products. Furthermore, the FDA conducts detailed inspections of and controls over our manufacturing, marketing, distribution, import and export, record keeping and storage and disposal practices, together with various post-marketing requirements.
Specifically, the FDCA requires these products, when sold in the United States, to be safe and effective for their intended uses and to comply with the regulations promulgated and enforced by the FDA. The FDA regulates the
253
Table of Contents
design, development, research, preclinical and clinical testing, introduction, manufacture, advertising, labeling, packaging, marketing, distribution, import and export, and record keeping for such products.
Medical devices can be marketed only for the indications for which they are cleared or approved. After a device has received 510(k) clearance (a pathway for the FDA to approve a new medical device for marketing) for a specific intended use, any change or modification that significantly affects its safety or effectiveness, such as a significant change in the design, materials, method of manufacture, or intended use, may require a new 510(k) clearance and payment of an FDA user fee.
Any medical devices we manufacture and distribute are subject to pervasive and continuing regulation by the FDA, state and certain other comparable foreign authorities. As a medical device manufacturer, our manufacturing facilities are subject to inspection on a routine basis by the FDA and other comparable foreign authorities, as well as audits by our notified body in the European Economic Area, or EEA, as described below. We are required to adhere to the Current Good Manufacturing Practices requirements, as set forth in the Quality Systems Regulation, which require manufacturers, including third-party manufacturers, to follow stringent design, testing, control, documentation and other quality assurance procedures during all phases of the design and manufacturing process.
We must also comply with post-market surveillance regulations, including adverse event reporting requirements, which require that we review and report to the FDA and other comparable foreign authorities any incident in which our products may have caused or contributed to a death or serious injury. Further, we are required to report any incident in which our product has malfunctioned if that malfunction would likely cause or contribute to a death or serious injury if it were to recur.
Labeling, advertising and promotional activities are subject to scrutiny by the FDA and other comparable foreign authorities and, in certain circumstances, by the Federal Trade Commission and other foreign counterparts. Medical devices approved or cleared by the FDA, foreign regulators, or our notified bodies may not be promoted for undocumented, unapproved or uncleared uses, otherwise known as off-label promotion. The FDA, other U.S. agencies and other comparable foreign authorities actively enforce the laws and regulations prohibiting the promotion of off-label uses.
The FDA can withdraw marketing authorization for a medical device product if compliance with regulatory standards is not maintained or if problems occur after the product reaches the market. Later discovery of previously unknown problems with a product may result in restrictions on the product or complete withdrawal of the product from the market. Because our operations include the manufacture and distribution of nuclear medical products, we are also subject to regulation by the NRC and the departments of health of each state in which we operate, which leaves us with a complex collection of requirements to navigate.
Market access, sales and marketing of medical devices in non-U.S. countries are subject to foreign regulatory requirements that vary widely from country to country. The time required to obtain approval for marketing a medical device in a foreign country could be longer or shorter than the time required by the FDA. Furthermore, the requirements are different in each country. For example in the EEA, a medical device must meet the Medical Devices Directives, or MDD, Essential Requirements or the Medical Devices Regulations, or MDR, General Safety and Performance Requirements, if certified from May 26, 2021. Before placing a medical device on the EEA market, the manufacturer must prepare a declaration of conformity, certifying that the device complies with the MDD/MDR, and must then affix the CE mark. The notified body typically audits and examines the devices technical documentation, and the quality system for the manufacture, design and final inspection of the relevant device before issuing a CE certificate. Following the issuance of this CE certificate, manufacturers may prepare the declaration of conformity and affix the CE mark to the devices covered by this CE certificate. Similar requirements apply in the UK. For access to the UK market, manufacturers must obtain a UKCA Certificate and affix a UKCA mark to their medical devices. However, the CE mark will be accepted in the UK until July 1, 2023.
254
Table of Contents
The standard by which conformity with applicable standards and directives is measured is dependent upon the type and class of the product, but normally involves a combination of self-assessment by the manufacturer and a third party assessment by a notified body. In the European Union, or EU, the third party assessment may consist of an audit of the manufacturers quality system (currently ISO 13485), provisions of the MDD and specific testing of the manufacturers device. Further, the MDR came into effect in the European Union on May 26, 2021, which requires us to obtain certification against the MDR to include a CE mark on new products, or make significant changes to existing products.
We are subject to additional regulations in other foreign countries, including, but not limited to, the United Kingdom and the EU to sell our products. We intend that either we or our distributors will receive any necessary approvals or clearance prior to marketing our products in those international markets.
We are subject to various healthcare related laws regulating fraud and abuse, research and development, pricing and sales and marketing practices, and the privacy and security of health information. In particular, the U.S. Federal Anti-Kickback Statute prohibits persons from knowingly and willfully soliciting, offering, receiving, or providing remuneration (including any kickback or bribe), directly or indirectly, in exchange for or to induce either the referral of an individual, or the furnishing or arranging for a good or service, for which payment may be made in whole or in part under a federal healthcare program, such as Medicare or Medicaid. A person or entity does not need to have actual knowledge of the statute or specific intent to violate it in order to have committed a violation. Similar laws and regulations apply in many foreign countries.
The Health Insurance Portability and Accountability Act of 1996, or HIPAA, prohibits knowingly and willfully (1) executing, or attempting to execute, a scheme to defraud any healthcare benefit program, including private payors, or (2) falsifying, concealing, or covering up a material fact or making any materially false, fictitious, or fraudulent statement in connection with the delivery of or payment for healthcare benefits, items, or services. In addition, HIPAA, as amended by the Health Information Technology for Economic and Clinical Health Act of 2009, also restricts the use and disclosure of patient identifiable health information, mandates the adoption of standards relating to the privacy and security of patient identifiable health information, and requires the reporting of certain security breaches with respect to such information. Similar to the U.S. Federal Anti-Kickback Statute, a person or entity does not need to have actual knowledge of the healthcare fraud statute implemented under HIPAA or specific intent to violate it in order to have committed a violation. Similar laws and regulations apply in many foreign countries.
The False Claims Act imposes liability on any person or entity that, among other things, knowingly presents, or causes to be presented, a false or fraudulent claim for payment by a federal healthcare program, knowingly makes, uses, or causes to be made or used, a false record or statement material to a false or fraudulent claim, or knowingly makes a false statement to avoid, decrease, or conceal an obligation to pay money to the U.S. federal government. The qui tam provisions of the False Claims Act allow a private individual to bring actions on behalf of the federal government alleging that the defendant has submitted a false claim to the federal government, and to share in any monetary recovery. In addition, the government may assert that a claim including items and services resulting from a violation of the U.S. Federal Anti-Kickback Statute constitutes a false or fraudulent claim for purposes of the civil False Claims Act. Similar laws and regulations apply in many foreign countries.
Federal consumer protection and unfair competition laws broadly regulate marketplace activities and activities that potentially harm consumers. Analogous U.S. state laws and regulations, such as state anti-kickback and false claims laws, also may apply to our business practices, including but not limited to, research, distribution, sales and marketing arrangements, and claims involving healthcare items or services reimbursed by any third-party payor, including private insurers. Further, there are state laws that require medical device manufacturers to comply with the voluntary compliance guidelines and the relevant compliance guidance promulgated by the U.S. federal government, or otherwise restrict payments that may be made to healthcare providers and other potential referral sources; state laws and regulations that require manufacturers to file reports relating to pricing and marketing information, which requires tracking gifts and other remuneration and items of value provided to
255
Table of Contents
healthcare professionals and entities; state and local laws requiring the registration of sales representatives; and state laws governing the privacy and security of health information in certain circumstances, many of which differ from each other in significant ways and often are not preempted by HIPAA. Similar laws and regulations apply in many non-U.S. countries.
Privacy and Information Security Laws
In the ordinary course of our business, we collect, store, use transmit and otherwise process certain types of data, including personal information, which subjects us to certain privacy and information security laws in the United States and internationally, including, for example and depending on the particular activity, the EU General Data Protection Regulation, or GDPR and the California Consumer Privacy Act of 2018, or CCPA, and other laws, rules and regulations designed to regulate the processing of personal information and for example reduce risks of identity theft. These laws impose obligations with respect to the collection, processing, storage, disposal, use, transfer, retention and disclosure of personal information. In addition, under certain of these laws, we must provide notice to individuals of our policies and practices for sharing personal information with third parties, provide advance notice of any changes to our policies and in some cases give individuals the right to prevent processing of their personal information and disclosure of it to third parties. Further, all 50 states in the United States have laws including obligations to provide notification of unauthorized acquisition of personal information to affected individuals, state officers and others. Some laws may also impose physical and electronic security requirements regarding the safeguarding of personal information. In order to comply with privacy and information security laws, we have confidentiality and information security standards and procedures in place for our business activities. Privacy and information security laws evolve regularly, and complying with these various laws, rules, regulations and standards, and with any new laws or regulations or changes to existing laws, could cause us to incur substantial costs that are likely to increase over time, requiring us to adjust our compliance program on an ongoing basis and presenting compliance challenges, change our business practices in a manner adverse to our business, divert resources from other initiatives and projects, and restrict the way products and services involving data are offered. See Risk FactorsLegal and Regulatory RisksAny actual or perceived failure to comply with evolving data privacy and data security laws and regulations in the jurisdictions where we operate, both inside and outside of the United States, could lead to government enforcement actions (which could include civil or criminal penalties), private litigation or adverse publicity and could materially and adversely affect our business.
Backlog and Deferred Contract Revenue
Total backlog represents committed but undelivered contracts and purchase orders at period end. Backlog excludes maintenance-related activity and agreements that do not represent firm purchase orders. Customer agreements that contain cancellation for convenience terms are generally not reflected in backlog until firm purchase orders are received. Backlog is not a complete measure of our future business due to these customer agreements. Backlog can fluctuate significantly due to the timing of large project awards. In addition, annual or multi-year contracts are subject to rescheduling and cancellation by customers due to the long-term nature of the contracts.
Deferred contract revenue represents prepayments from customers, including milestone or installment payments, on projects for which services have commenced, as well as unbilled amounts attributable to services rendered and products constructed associated with customer contracts for which revenue is not able to be recognized.
Information on backlog and deferred contract revenue follows (in millions):
| As of June 30, | As of March 31, | |||||||||||||||
| 2020 | 2019 | 2021 | 2020 | |||||||||||||
| Backlog |
$ | 601.4 | $ | 346.3 | $ | 766.5 | $ | 652.3 | ||||||||
| Deferred contract revenue |
39.5 | 40.8 | 40.7 | 31.3 | ||||||||||||
256
Table of Contents
Legal Proceedings
From time to time, we are involved in various routine legal proceedings. We cannot predict the outcome of these lawsuits, legal proceedings and claims with certainty. Nevertheless, we believe that the outcome of these proceedings, even if determined adversely, would not have a material adverse effect on our business, financial condition and results of operations.
Human Capital Resources
As of March 31, 2021, we employed 2,572 full-time and part-time employees. Approximately 59% of our employees are in operations and we use talent acquisition and retention practices, including but not limited to: college and university recruiting programs; job fairs; compensation benchmarking; employee engagement; communication through email, social media and other communication platforms; employee development and training programs including mentoring, new product training for our sales and services organizations; Navex global training for our staff and management level employees; and, quarterly check-ins between employees and their managers as key human capital measures and objectives.
257
Table of Contents
MANAGEMENT OF NEW MIRION FOLLOWING THE BUSINESS COMBINATION
Board of Directors and Management
The following sets forth certain information, as of the date of this proxy statement/prospectus, concerning the persons who are expected to serve as directors and executive officers of the Company following the closing of the Business Combination and assuming the election of the nominees at the Special Meeting as set forth in Director Election Proposal.
| Name |
Age |
Position |
||
| Thomas D. Logan | 60 |
Director Nominee, Founder and Chief Executive Officer | ||
| Brian Schopfer | 37 |
Chief Financial Officer | ||
| Michael Freed | 45 | Chief Operating Officer | ||
| Lawrence D. Kingsley | 58 | Director Nominee and Chairman | ||
| Jyothsna (Jo) Natauri | 44 | Director Nominee | ||
| Christopher Warren | 45 | Director Nominee | ||
| Steven Etzel | 58 | Director Nominee |
Upon the consummation of the Business Combination, we anticipate increasing the initial size of the Board of Directors from six directors to nine directors, each of whom will be voted upon by GSAHs stockholders at the Special Meeting. If all director nominees are elected and the Business Combination is consummated, the Board of Directors will initially consist of nine directors.
Thomas D. Logan currently serves, and has served, as Mirions founding Chairman and Chief Executive Officer since 2005, and he has served as a member of Mirons board of directors since 2005. Prior to joining Mirion, Mr. Logan served as Chief Executive Officer for Global Dosimetry Solutions, a radiation dosimetry provider, from 2004. Mr. Logan has more than 30 years of energy industry experience, as well as extensive experience within the contract manufacturing and consumer products industries. Mr. Logan received a M.B.A. and a B.S. from Cornell University. We believe Mr. Logans extensive history with Mirion, as well as his business expertise, qualify him to serve on our board of directors.
Brian Schopfer has served as our Chief Financial Officer and a member of Mirions board of directors since 2020. Mr. Schopfer joined Mirion in 2015 and previously served as Mirions Executive and Senior Vice President of Business Transformation. In February 2018, Mr. Schopfer left Mirion and joined Omnimax International, a building products company, where he served as Chief Financial Officer until March 2019. Mr. Shopfer rejoined Mirion in March 2019. Prior to joining Mirion, Mr. Schopfer served as Chief Financial Officer for HillPhoneix (part of the Dover Corporation), a commercial refrigeration manufacturer, from 2014 to 2015. Mr. Schopfer also served as the Director of Financial Planning and Analysis for the Dover Corporation, a global manufacturing company, from 2013 to 2014. Mr. Schopfer received a B.S. in Finance and Marketing from the University of Pittsburgh.
Michael Freed has served as our Chief Operating Officer since 2017 and a member of Mirions board of directors since 2016. He also served as the President of Mirions Health Physics division from 2016 to 2017. Prior to joining Mirion, Mr. Freed was employed as a Principal Consultant for Bain and Company, a global management consulting firm, from 2010 to 2016. Mr. Freed served as a Submarine Officer in the United States Navy from 1998 to 2008 where he was certified as a Naval Nuclear Engineer and completed two tours of duty on Los Angeles Class fast attack submarines. He also served as a special assistant to the Chief of Naval Operations in Washington, DC. Mr. Freed received his M.B.A. from the University of Virginia Darden School of Business and a B.A. in Economics from Northwestern University.
Lawrence D. Kingsley currently serves as the independent Non-Executive Board Chair of IDEXX Laboratories, Inc., a public company, since November 2019 and as an Advisory Director to Berkshire Partners LLC, an
258
Table of Contents
investment company, since May 2016. Mr. Kingsley also currently serves as a Director of Polaris Industries Inc., a public company, since January 2016 and as a Director of Rockwell Automation, Inc., a public company, since April 2013. Prior to joining IDEXX Laboratories, Inc., Mr. Kingsley served as Chairman of Pall Corporation from October 2013 to August 2015 and as President and Chief Executive Officer of Pall Corporation from October 2011 to August 2015 until Danaher Corporation, a public company, acquired Pall Corporation in August 2015. Before his experience at Pall Corporation, Mr. Kingsley served as the Chief Executive Officer and President of IDEX Corporation, a public company specializing in the development, design and manufacture of fluid and metering technologies, health and science technologies and fire, safety and other diversified products, from March 2005 to August 2011 and the Chief Operating Officer of IDEX Corporation from August 2004 to March 2005. Mr. Kingsley previously served as a Director of Pall Corporation from October 2011 to August 2015, Cooper Industries plc (formerly Cooper Industries Ltd.), a public company, from 2007 to 2012 and IDEX Corporation from 2005 to 2011. Mr. Kingsley served in various positions of increasing responsibility at Danaher Corporation, including Corporate Vice President and Group Executive from March 2004 to August 2004, President of Industrial Controls Group from April 2002 to July 2004 and President of Motion Group, Special Purpose Systems from January 2001 to March 2002. Mr. Kingsley also previously held management positions of increasing responsibility at Kollmorgen Corporation and Weidmuller Incorporated. Mr. Kingsley received an undergraduate degree in Industrial Engineering and Management from Clarkson University and an M.B.A. from the College of William and Mary. We believe that Mr. Kingsleys strong executive leadership and operational skills, in-depth knowledge of and experience in strategic planning, corporate development, and operations analysis and experience serving on other public company boards provide him with the qualifications and skills to serve on our Board of Directors.
Jyothsna (Jo) Natauri is a Partner of Goldman Sachs & Co. LLC and has served as the Global Head of Private Healthcare Investing within Goldman Sachs Asset Management since May 2018. Prior to assuming her current role, Ms. Natauri was an investment banker with Goldman Sachs for 12 years, where she led coverage of large cap companies in healthcare and other industries. She was named managing director in 2008 and partner in 2012. Ms. Natauri has served as a director on the board of Flywire Corporation since November 2020, and also serves on the boards of MyEyeDr, Sita Foundation and Safe Horizon. She previously served on the board of Avantor from November 2018 to May 2021. Ms. Natauri received a B.A. from the University of Virginia in Economics and Biology. We believe that Ms. Natauris experience of over 20 years in covering companies and executing transactions provides her with the qualifications and skills to serve on our Board of Directors.
Christopher Warren currently serves, and has served, as a partner at Charterhouse Capital Partners LLP since he joined in 2013. Prior to joining Charterhouse, Mr. Warren served as a partner at ECI Partners, a private equity group, from 2003 to 2013. He also served as Associate at BC Partners, an international investment firm, and as Consultant at COBA, a UK-based strategy consulting firm. Mr. Warren received a Master of Arts in Philosophy, Politics and Economics from Oxford University and an MBA from INSEAD. We believe that Mr. Warrens extensive business experience provides him with the qualifications and skills to serve on our Board of Directors.
Steven Etzel has served as Senior Vice President and Chief Financial Officer of Rockwell Automation, Inc., a company focused on industrial automation and information, from November 2020 to February 2021, and subsequently as Senior Vice President, Finance of Rockwell until his retirement in April 2021. Mr. Etzel joined Rockwell in 1989 and served in various positions, including Vice President and Treasurer from 2007 to 2020 and Vice President, Finance from October 2020 to November 2020. Mr. Etzel received his Bachelor of Science degree in Business Administration from Clarion University of Pennsylvania. We believe Mr. Etzels extensive financial and management experience, including financial reporting, internal controls, investor relations, financial planning and analysis, capital markets financing transactions, mergers and acquisitions and risk management provides him with the qualifications and skills to serve on our Board of Directors.
Family Relationships
There are no family relationships among any of the individuals who shall serve as directors or executive officers of New Mirion following the completion of the Business Combination.
259
Table of Contents
Board of Directors
The New Mirion Board will establish the authorized number of directors from time to time by resolution. The New Mirion Board will initially consist of nine members. Lawrence D. Kingsley will serve as Chairman of the New Mirion Board.
The New Mirion Board nominees were designated as follows pursuant to the Business Combination Agreement:
| | Thomas D. Logan, as Chief Executive Officer of New Mirion |
| | Lawrence D. Kingsley and Jo Natauri were designated by the Sponsor; |
| | Christopher Warren was designated by the Charterhouse Parties; |
| | Steven Etzel, , and were mutually designated by the Sponsor, the Charterhouse Parties and New Mirion. |
In addition, we are a party to a Director Nomination Agreement with certain entities affiliated with Charterhouse and a director nomination with the Sponsor that provide Charterhouse with the right to nominate one director to the New Mirion Board and the Sponsor to nominate two directors to the New Mirion Board, subject to certain fallaway provisions. See Certain Relationships and Related Persons TransactionsDirector Nomination Agreements for more information.
Each of New Mirions current directors will continue to serve until the election and qualification of his or her successor, or his or her earlier death, resignation or removal.
The primary responsibilities of the New Mirion Board will be to provide oversight, strategic guidance, counseling and direction to New Mirions management. The New Mirion Board will meet on a regular basis and additionally, as required.
Role of Board in Risk Oversight
The New Mirion Board will have extensive involvement in the oversight of risk management related to New Mirion and its business and will accomplish this oversight through the regular reporting to the New Mirion Board by the audit committee. The audit committee will represent the New Mirion Board by periodically reviewing New Mirions accounting, reporting and financial practices, including the integrity of its financial statements, the surveillance of administrative and financial controls and its compliance with legal and regulatory requirements. Through its regular meetings with management, including the finance, legal, internal audit and information technology functions, the audit committee will review and discuss all significant areas of New Mirions business and summarize for the New Mirions all areas of risk and the appropriate mitigating factors. In addition, the New Mirion Board will receive periodic detailed operating performance reviews from management.
Board Committees
The New Mirion Board has an audit committee, a compensation committee and a nominating and corporate governance committee, each of which will have the composition and responsibilities described below upon completion of the Business Combination. Members serve on these committees until their resignation or until otherwise determined by the New Mirion Board.
Audit Committee
The audit committee will consist of and , with serving as the chair of the committee. The New Mirion Board has determined that , and are independent as defined under applicable NYSE listing standards, including the standards specific to members of an audit committee.
260
Table of Contents
The New Mirion Board has determined that qualifies as an audit committee financial expert within the meaning of SEC regulations and meets the financial sophistication requirements of the NYSE listing rules. In making this determination, the New Mirion Board has considered s formal education and previous and current experience in financial and accounting roles. The independent registered public accounting firm and management periodically will meet privately with the audit committee.
The audit committee is responsible for, among other things:
| | appointing, compensating, retaining, evaluating, terminating and overseeing New Mirions independent registered public accounting firm; |
| | discussing with New Mirions independent registered public accounting firm their independence; |
| | reviewing with New Mirions independent registered public accounting firm the scope and results of their audit; |
| | approving all audit and permissible non-audit services to be performed by New Mirions independent registered public accounting firm; |
| | overseeing the financial reporting process and discussing with management and New Mirions independent registered public accounting firm the interim and annual financial statements that New Mirion files with the SEC; |
| | reviewing our policies on risk assessment and risk management; |
| | reviewing related person transactions; |
| | designing and implementing the internal audit function; |
| | overseeing our financial and accounting controls and compliance with legal and regulatory requirements; and |
| | establishing procedures for the confidential anonymous submission of concerns regarding questionable accounting, internal controls or auditing matters. |
Compensation Committee
The compensation committee will consist of and , with serving as the chair of the committee. and are non-employee directors, as defined in Rule 16b-3 promulgated under the Exchange Act. The New Mirion Board has determined that and are independent as defined under applicable NYSE listing standards, including the standards specific to members of a compensation committee.
The compensation committee is responsible for, among other things:
| | determining, or recommending to the New Mirion Board for determination, the compensation of New Mirions executive officers, including the chief executive officer; |
| | administering New Mirions equity compensation plans; |
| | overseeing New Mirions overall compensation policies and practices, compensation plans, and benefits programs; and |
| | appointing and overseeing any compensation consultants. |
We believe that the composition and functioning of the compensation committee meets the requirements for independence under applicable NYSE listing standards.
261
Table of Contents
Nominating and Corporate Governance Committee
The nominating and corporate governance committee will consist of and , with serving as the chair of the committee. The New Mirion Board has determined that each of these individuals is independent as defined under applicable SEC rules and NYSE listing standards.
The nominating and corporate governance committee is responsible for, among other things:
| | evaluating and making recommendations regarding the composition, organization and governance of the New Mirion Board and its committees; |
| | reviewing and making recommendations with regard to New Mirions corporate governance guidelines and compliance with laws and regulations; and |
| | overseeing an evaluation of the New Mirion Board and its committees. |
We believe that the composition and functioning of the nominating and corporate governance committee meets the requirements for independence under current NYSE listing standards.
The audit, compensation, and nominating and corporate governance committees will each operate under a written charter to be effective prior to the completion of the Business Combination that satisfies the applicable rules and regulations of NYSE and the SEC except where New Mirion intends to rely on the NYSE transition rules applicable to companies completing an initial listing.
New Mirion intends to post the charters of its audit, compensation and nominating and corporate governance committees, and any amendments thereto that may be adopted from time to time, on New Mirions website. Information on or that can be accessed through the New Mirions website is not part of this proxy statement/ prospectus. The New Mirion Board may from time to time establish other committees.
Independent New Mirion Board of Directors
NYSE rules generally require that independent directors must comprise a majority of a listed companys board of directors. Based upon information requested from and provided by each proposed director concerning his or her background, employment and affiliations, including family relationships, New Mirion has determined that , , and , representing of New Mirions proposed directors, will be independent as that term is defined under the applicable rules and regulations of the SEC and the listing requirements and rules of the NYSE.
Code of Business Conduct and Ethics
Prior to the completion of the Business Combination, New Mirion will adopt a code of business conduct and ethics that will apply to all of New Mirions employees, officers, and directors, including New Mirions chief executive officer, chief financial officer, and other executive and senior financial officers. Upon the completion of the Business Combination, the full text of New Mirions code of business conduct and ethics will be available on the investor relations page on New Mirions website. Information on or that can be accessed through New Mirions website is not part of this proxy statement/prospectus.
Compensation Committee Interlocks and Insider Participation
None of New Mirions executive officers currently serves, or has served during the last year, as a member of the New Mirion Board or compensation committee of any entity that has one or more executive officers serving as a member of the New Mirion Board.
262
Table of Contents
MIRIONS MANAGEMENTS DISCUSSION AND ANALYSIS OF FINANCIAL CONDITION AND RESULTS OF OPERATIONS
Stockholders of GSAH should read the following discussion and analysis of Mirion Technologies (TopCo), Ltd.s financial condition and results of operations together with the consolidated financial statements and related notes of Mirion Technologies (TopCo), Ltd. that are included elsewhere in this proxy statement/prospectus. This discussion contains forward-looking statements based upon current expectations that involve risks and uncertainties. Mirions actual results may differ materially from those anticipated in these forward-looking statements as a result of various factors, including those set forth under the section entitled Risk Factors or in other parts of this proxy statement/prospectus. Please also see the section entitled Cautionary Note Regarding Forward-Looking Statements.
Unless the context otherwise requires, all references in this section to Mirion, Mirion TopCo, the Company, we, us or our refer to Mirion Technologies (TopCo), Ltd. and its consolidated subsidiaries prior to the consummation of the Business Combination. References to New Mirion refer to GSAH after the consummation of the Business Combination.
Unless the context otherwise requires or unless otherwise specified, all dollar amounts in this section are in millions.
Overview
Mirion is a global provider of products, services, and software that allow our customers to safely leverage the power of ionizing radiation for the greater good of humanity through critical applications in the medical, nuclear and defense markets, as well as laboratories, scientific research, analysis, and exploration.
Mirion provides dosimetry solutions for monitoring the total amount of radiation medical staff members are exposed to over time, radiation therapy quality assurance solutions for calibrating and verifying imaging and treatment accuracy, and radionuclide therapy products for nuclear medicine applications such as shielding, product handling, medical imaging furniture, and rehabilitation products. Mirion provides robust, field-ready personal radiation detection and identification equipment for defense applications and radiation detection and analysis tools for power plants, labs, and research applications. Nuclear power plant product offerings are used for the full nuclear power plant lifecycle including core detectors, essential measurement devices for new build, maintenance, decontamination and decommission, and equipment for monitoring and control during fuel dismantling and remote environmental monitoring.
Mirion manages and reports results of operations in two business segments: Medical and Industrial.
| | For the nine months ended March 31, 2021, Mirions revenues were $431.6 million, of which 24.0% was generated in the Medical segment and 76.0% was generated in the Industrial segment, as compared with revenues for the nine months ended March 31, 2020 of $337.0 million with 13.2% and 86.8% generated in the Medical and Industrial segments, respectively. |
| | For the year ended June 30, 2020, Mirions revenues were $478.2 million, of which 13.1% was generated in the Medical segment and 86.9% was generated in the Industrial segment, as compared with revenues for the year ended June 30, 2019 of $440.1 million with 9.7% and 90.3% generated in the Medical and Industrial segments, respectively. |
| | Backlog (representing committed but undelivered contracts and purchase orders, including funded and unfunded government contracts) was $766.4 million and $601.4 million as of March 31, 2021, and June 30, 2020, respectively. |
263
Table of Contents
Key Factors Affecting Our Performance
We believe that the business and results of operations may be impacted in the future by various trends and conditions, including the following:
| | Medical end market trends Growth and operating results in our Medical segment are impacted by: |
| | Increased or changes to global regulatory standards; |
| | Increased focus on healthcare safety; |
| | Changes to healthcare reimbursement; |
| | Potential budget constraints in hospitals and other healthcare providers; |
| | Medical/lab dosimetry growth supported by growing and aging demographics, increased number of healthcare professionals, and penetration of radiation therapy/diagnostics; and |
| | Medical radiation therapy quality assurance (RT QA) growth driven by growing and aging population demographics, low penetration of RT QA technology in emerging markets, and increased adoption of advanced software and hardware solutions for improved outcomes and administrative and labor efficiencies. |
| | Business combinations A large driver of our historical growth has been the acquisition and integration of related businesses. Our ability to integrate, restructure, and leverage synergies of these businesses will impact our operating results over time. |
| | Environmental objectives of governments Growth and operating results in our Industrial segment are impacted by environmental policy decisions made by governments in the countries where we operate. Our nuclear power customers may benefit from decarbonization efforts given the relatively low carbon footprint of nuclear power to other existing energy sources. In addition, decisions by governments to build new power plants or decommission existing plants can positively and negatively impact our customer base. |
| | Government budgets While we believe that we are poised for growth from governmental customers in both of our segments, our revenues and cash flows from government customers are influenced, particularly in the short-term, by budgetary cycles. This impact can be either positive or negative. |
| | Nuclear new build projects A portion of our backlog is driven by contracts associated with the construction of new nuclear power plants. These contracts can be long-term in nature and provide us with a strong pipeline for the recognition of future revenues in our Industrial segment. |
| | Research and developments A portion of our operating expenses is associated with research and development activities associated with the design of new products. Given the specific design and application of certain of these products, there is some risk that these costs will not result in successful products in the market. Further, the timing of these products can move and be challenging to predict. |
| | COVID-19 COVID-19 may affect revenue growth in certain of our businesses, primarily those serving our medical end markets, and it is uncertain how materially COVID-19 will affect our global operations generally if these impacts were to persist or worsen over an extended period of time. The extent and duration of the impacts are uncertain and dependent in part on customers returning to work and economic activity ramping up. The impact of COVID-19 on our customers has affected our sales operations in certain ways, including increased customer disputes regarding orders, delayed customer notices to proceed with production, delayed payment from customers and, on rare occasions, customers have refused to pay for their orders entirely. Further, access to customer sites for sales was limited in some cases. |
Non-GAAP Financial Measures
We report our financial results in accordance with generally accepted accounting principles in the U.S. (GAAP). However, management believes certain non-GAAP financial measures provide investors and other
264
Table of Contents
users with additional meaningful information that should be considered when assessing our ongoing performance. Management also uses these non-GAAP financial measures in making financial, operating, and planning decisions, and in evaluating our performance. Non-GAAP financial measures should be viewed in addition to, and not as an alternative for, our GAAP results. The non-GAAP financial measures we present may differ from similarly captioned measures presented by other companies.
We use the non-GAAP financial measures Adjusted revenues, Adjusted net (loss) income, EBITDA, EBITA, Adjusted EBITDA, Free Cash Flow, and Adjusted Free Cash Flow. See the Quarterly Results of Operations and Cash flows sections below for definitions of our non-GAAP financial measures and reconciliation to their most directly comparable GAAP measures.
The following tables present a reconciliation of certain non-GAAP financial measures for the nine months ended March 31, 2021 and 2020, and the years ended June 30, 2020, 2019 and 2018.
| Nine Months Ended March 31, 2021 |
Nine Months Ended March 31, 2020 |
Year Ended June 30, 2020 |
Year Ended June 30, 2019 |
Year Ended June 30, 2018 |
||||||||||||||||||||||||||||||||||||
| ($ in millions) |
Revenues | Net Loss | Revenues | Net Loss | Revenues | Net Loss | Revenues | Net Loss | Revenues | Net Loss | ||||||||||||||||||||||||||||||
| Total GAAP |
$ | 431.6 | $ | (131.0 | ) | $ | 337.0 | $ | (94.6 | ) | $ | 478.2 | $ | (119.1 | ) | $ | 440.1 | $ | (122.0 | ) | $ | 444.1 | $ | (103.7 | ) | |||||||||||||||
| Revenue reduction from purchase accounting |
4.3 | 4.3 | | | 0.2 | 0.2 | | | 0.6 | 0.6 | ||||||||||||||||||||||||||||||
| Cost of revenues impact from inventory valuation purchase accounting |
5.2 | 1.1 | 1.6 | 0.1 | 0.9 | |||||||||||||||||||||||||||||||||||
| Foreign currency (gain) loss, net |
12.2 | (4.0 | ) | (0.6 | ) | (3.2 | ) | 5.3 | ||||||||||||||||||||||||||||||||
| Amortization of acquired intangibles |
44.3 | 38.2 | 50.6 | 53.0 | 59.8 | |||||||||||||||||||||||||||||||||||
| Non-operating expenses(1)(2)(3)(4)(5) |
27.4 | 13.7 | 20.1 | 14.7 | 32.4 | |||||||||||||||||||||||||||||||||||
| Tax impact of adjustments above |
(20.5 | ) | (12.3 | ) | (16.1 | ) | (19.9 | ) | (25.8 | ) | ||||||||||||||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||||||||
| Adjusted |
$ | 435.9 | $ | (58.1 | ) | $ | 337.0 | $ | (57.9 | ) | $ | 478.4 | $ | (63.3 | ) | $ | 440.1 | $ | (77.3 | ) | $ | 444.7 | $ | (30.5 | ) | |||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||||||||
| ($ in millions) |
Nine Months Ended March 31, 2021 |
Nine Months Ended March 31, 2020 |
Year Ended June 30, 2020 |
Year Ended June 30, 2019 |
Year Ended June 30, 2018 |
|||||||||||||||||
| Net loss |
$ | (131.0 | ) | $ | (94.6 | ) | $ | (119.1 | ) | $ | (122.0 | ) | $ | (103.7 | ) | |||||||
| Interest expense, net |
119.5 | 110.6 | 149.2 | 143.5 | 128.9 | |||||||||||||||||
| Income tax (benefit) provision |
6.2 | (4.8 | ) | (5.5 | ) | (4.2 | ) | (36.8 | ) | |||||||||||||
| Amortization |
44.3 | 38.2 | 50.6 | 53.0 | 59.8 | |||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
||||||||||||
| EBITA |
$ | 39.0 | $ | 49.4 | $ | 75.2 | $ | 70.3 | $ | 48.2 | ||||||||||||
| Depreciation |
13.9 | 13.4 | 17.9 | 16.5 | 17.2 | |||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
||||||||||||
| EBITDA |
$ | 52.9 | $ | 62.8 | $ | 93.1 | $ | 86.8 | $ | 65.4 | ||||||||||||
| Stock compensation expense |
| 0.1 | 0.2 | 0.1 | 0.2 | |||||||||||||||||
| Debt extinguishment |
| | | 12.8 | | |||||||||||||||||
| Foreign currency (gain) loss, net |
12.2 | (4.0 | ) | (0.6 | ) | (3.2 | ) | 5.3 | ||||||||||||||
| Revenue reduction from purchase accounting |
4.3 | | 0.2 | | 0.6 | |||||||||||||||||
| Cost of revenues impact from inventory valuation purchase accounting |
5.2 | 1.1 | 1.6 | 0.1 | 0.9 | |||||||||||||||||
| Non-operating expenses(1)(2)(3)(4)(5) |
27.4 | 13.7 | 20.1 | 14.7 | 32.4 | |||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
||||||||||||
| Adjusted EBITDA |
$ | 102.0 | $ | 73.7 | $ | 114.6 | $ | 111.3 | $ | 104.8 | ||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
||||||||||||
| (1) | Pre-tax non-operating expenses of $27.4 million for the nine-months ended March 31, 2021 includes $12.5 million restructuring and other costs to achieve operational synergies, $11.5 million of mergers and acquisition expenses, $3.0 million of costs to achieve information technology system integration and efficiency and $0.6 million of expenses related to debt refinancing. |
265
Table of Contents
| (2) | Pre-tax non-operating expenses of $13.7 million for the nine-months ended March 31, 2020 includes $8.8 million of mergers and acquisition expenses, $2.5 million costs to achieve operational synergies, $2.2 million of costs to achieve information technology system integration and efficiency and $0.2 million of expenses related to debt refinancing. |
| (3) | Pre-tax non-operating expenses of $20.1 million for the year ended June 30, 2020 includes $10.8 million of mergers and acquisition expenses, $3.8 million of costs to achieve operational synergies, $3.4 million of costs to achieve information technology system integration and efficiency, and $1.6 million of expenses related to debt refinancing. |
| (4) | Pre-tax non-operating expenses of $14.7 million for the year ended June 30, 2019 includes $6.5 million of mergers and acquisition expenses, $2.8 million of costs to achieve information technology system integration and efficiency, $2.8 million of costs to achieve operational synergies, and $0.5 million of expenses related to debt refinancing. |
| (5) | Pre-tax non-operating expenses of $32.4 million for the year ended June 30, 2018 includes $28.9 million of costs to achieve operational synergies, $1.6 million of costs to achieve information technology system integration and efficiency, and $0.4 million of mergers and acquisition expenses. |
Our Business Segments
We manage and report our business in two business segments: Medical and Industrial.
Medical includes products and services for radiation therapy and personal dosimetry. This segments principal offerings are:
| | Radiation Therapy Quality Assurance Solutions for calibrating and/or verifying imaging, treatment machine, patient treatment plan, and patient treatment accuracy (hardware and software); |
| | Dosimetry Solutions for monitoring the total amount of radiation medical staff members are exposed to over time; and |
| | Radionuclide Therapy Solutions, which includes products for nuclear medicine in radiation measurement, shielding, product handling, medical imaging furniture and rehabilitation. |
Industrial includes products and services for defense, nuclear energy, laboratories and research and other industrial markets. This segments principal offerings are:
| | Reactor Safety and Control Systems, which includes radiation monitoring systems and reactor instrumentation and control systems that ensure the safe operation of nuclear reactors and other nuclear fuel cycle facilities; and |
| | Radiological Search, Measurement and Analysis Systems, which includes solutions to locate, measure and perform in-depth scientific analysis of radioactive sources for radiation safety, security, and scientific applications |
Recent Developments
On December 18, 2020, we purchased 100% of the issued and outstanding shares of Sun Nuclear Corporation (SNC) for an aggregate of $258.1 million, net of cash acquired of $18.8 million. SNC is a global leader in radiation therapy quality assurance, delivering patient safety solutions for diagnostic imaging and radiation therapy providers in multiple countries around the world. The acquisition is included in our Medical segment and will advance the Companys strategy to further expand into the radiation therapy markets globally.
On September 1, 2020, the Company purchased 100% of the issued and outstanding shares of Biodex Medical Systems, Inc. (Biodex) for an aggregate of $26.9 million, net of cash acquired of $4.1 million. Biodex is a manufacturer and distributor of medical devices and related replacement parts for physical and nuclear medicine, as well as medical imaging applications located in the United States. The acquisition is included in our Medical segment and will advance the Companys strategy to further expand into the medical treatment markets globally.
The 2019 Credit Facility was amended to provide an additional $225.0 million in gross proceeds from the USD term loan in December 2020 to fund the SNC acquisition. The term loan has a seven-year term (expiring March
266
Table of Contents
2026), bears interest at the greater of LIBOR or 0%, plus 4.00% and has quarterly principal repayments of 0.25% of the original principal balance. The interest rate was 4.27% and 5.07% at March 31, 2021 and June 30, 2020, respectively. During the nine months ended March 31, 2021, Mirion borrowed a net $218.8 million of notes payable under the 2019 Credit Facility and a net $70.0 million of borrowings from related parties at 11.5% interest, offset by $35.0 million of net repayments of borrowings on the revolver term loan, $6.4 million net repayments under the 2019 Credit Facility, and $6.0 million net repayments of the NRG loan. See Note 8 to the unaudited interim condensed consolidated financial statements.
Business Combination
Mirion TopCo entered into a Business Combination Agreement (the Agreement), dated as of June 17, 2021, by and among Mirion TopCo, GSAH, CCP IX CCP IX LP No. 1, CCP IX LP No. 2, CCP IX Co-Investment LP and CCP IX Co-Investment No. 2 LP, each acting by their general partner, Charterhouse General Partners (IX) Limited, for the limited purpose set forth in the Agreement, each of the other persons set forth on Annex I to the Agreement and the other holders of existing shares of Mirion TopCo who become a party to the Agreement by executing a joinder agreement. Pursuant to the terms of the Agreement, the parties thereto will enter into a business combination transaction (the Business Combination) pursuant to which Mirion TopCo will combine with a subsidiary of GSAH. Upon completion of the Business Combination, GSAH will be renamed Mirion Technologies, Inc. and will be listed on the NYSE under the ticker symbol MIR. The Business Combination is expected to close in 2021 (the Closing Date).
The proposed Business Combination is expected to be consummated after the required approval by the stockholders of the Company and the satisfaction of certain other conditions described in the Agreement.
The Business Combination will be accounted for under ASC 805, Business Combinations. GSAH has been determined to be the accounting acquirer. Mirion constitutes a business in accordance with ASC 805 and the business combination constitutes a change in control. Accordingly, the business combination will be accounted for using the acquisition method. Under this method of accounting, Mirion will be treated as the acquired company for financial reporting purposes and the net assets of the post-business combination company will be stated at fair value, with goodwill or other intangible assets recorded.
Commitment Letter
In connection with the Agreement, Mirion Technologies (HoldingSub2) Ltd., a subsidiary of Mirion TopCo, entered into a commitment letter (the Commitment Letter) with Goldman Sachs Lending Partners LLC (GS Lending) and Citigroup Global Markets Inc. (Citi) pursuant to which GS Lending and Citi have committed to provide to Mirion Technologies, Inc., as the subsidiary borrower, and a parent entity of the Mirion TopCo business to be formed, a $830 million senior secured term loan B facility (the Term Loan) and a $90 million revolving facility (the Revolving Facility). The Term Loan will mature seven years after the Closing Date and will amortize in equal quarterly installments in an aggregate annual amount equal to 1% of the initial principal amount of the Term Loan. The Revolving Facility will mature five years after the Closing Date. See Liquidity and Capital Resources.
Public Company Costs
Subsequent to the Business Combination, we expect New Mirion will continue as an SEC-registered and NYSE-listed company. We expect to hire additional staff and implement new processes and procedures to address public company requirements. We also expect to incur substantial additional expenses for, among other things, directors and officers liability insurance, director fees, and additional internal and external costs for investor relations, accounting, audit, legal and other functions.
267
Table of Contents
Basis of Presentation
Financial information presented was derived from Mirions historical consolidated financial statements and accounting records, and they reflect the historical financial position, results of operations and cash flows of the business in conformity with U.S. GAAP. The Consolidated Financial Statements include the accounts of the Company and its wholly owned and majority-owned or controlled subsidiaries. For consolidated subsidiaries where our ownership is less than 100%, the portion of the net income or loss allocable to noncontrolling interests is reported as Income (Loss) attributable to noncontrolling interests in the Consolidated Statements of Operations. All intercompany accounts and transactions have been eliminated in consolidation. See the notes to the financial statements included in this proxy statement/prospectus for additional information.
Results of Operations
Nine months ended March 31, 2021 and March 31, 2020
| (Dollars in millions) |
Nine months ended March 31, 2021 |
Nine months ended March 31, 2020 |
$ Change | % Change | ||||||||||||
| Revenues |
$ | 431.6 | $ | 337.0 | $ | 94.6 | 28.1 | % | ||||||||
| Cost of revenues |
259.5 | 201.2 | 58.3 | 29.0 | % | |||||||||||
|
|
|
|
|
|||||||||||||
| Gross profit |
172.1 | 135.8 | 36.3 | 26.7 | % | |||||||||||
| Selling, general and administrative expenses |
144.4 | 116.4 | 28.0 | 24.1 | % | |||||||||||
| Research and development |
21.2 | 12.3 | 8.9 | 72.4 | % | |||||||||||
|
|
|
|
|
|||||||||||||
| Income from operations |
6.5 | 7.1 | (0.6 | ) | (8.5 | )% | ||||||||||
| Interest expense, net |
119.5 | 110.6 | 8.9 | 8.0 | % | |||||||||||
| Foreign currency (gain) loss, net |
12.3 | (4.0 | ) | 16.3 | (407.5 | )% | ||||||||||
| Other (income) expense, net |
(0.5 | ) | (0.1 | ) | (0.4 | ) | 400.0 | % | ||||||||
|
|
|
|
|
|||||||||||||
| Loss before provision (benefit) from income taxes |
(124.8 | ) | (99.4 | ) | (25.4 | ) | (25.6 | )% | ||||||||
| Income tax (benefit) expense |
6.2 | (4.8 | ) | 11.0 | (229.2 | )% | ||||||||||
|
|
|
|
|
|||||||||||||
| Net loss |
(131.0 | ) | (94.6 | ) | (36.4 | ) | 38.5 | % | ||||||||
| Loss attributable to noncontrolling interests |
(0.1 | ) | | (0.1 | ) | N/A | ||||||||||
|
|
|
|
|
|||||||||||||
| Net loss attributable to stockholders |
$ | (130.9 | ) | $ | (94.6 | ) | $ | (36.3 | ) | 38.4 | % | |||||
|
|
|
|
|
|||||||||||||
Overview
Revenues for the nine months ended March 31, 2021 were $431.6 million, resulting in an increase of $94.6 million, or 28.1%, from the same period in the prior year primarily driven by acquisitions in the Medical segment and organic growth in the Industrial segment. Cost of revenues of $259.5 million, also increased 29.0% compared to the same period in the prior year reflecting the increase in revenues and a $4.9 million increase in non-operational restructuring and other costs to achieve operational synergies. Gross profit increased by $36.3 million and as a percentage of revenue was consistent period over period for the Company, including a decrease in percentage of revenue in our Medical segment, offset by an increase in percentage of revenue in our Industrial segment. There was a net loss of $131.0 million for the nine months ended March 31, 2021 compared to a net loss of $94.6 million during the nine months ended March 31, 2020. The $36.4 million, or 38.5% increase, is the result of the increase in gross profit, offset by higher SG&A expenses of $28.0 million, primarily driven by acquisitions in the Medical segment and an $8.6 million increase in non-operational costs related to restructuring, mergers and acquisitions and costs to achieve synergies. Also contributing to the increase in net loss period over period was increased net interest expense of $8.9 million, the negative impact of foreign currency exchange of $16.3 million, and net increase in income tax expense of $11.0 million. The impact of purchase accounting related to the fair value adjustment of deferred revenue, reduced revenue in the nine months
268
Table of Contents
ended March 31, 2021 by $4.3 million. The impact of purchase accounting related to the fair value of inventory increased cost of revenues by $5.2 million for the nine months ended March 31, 2021.
Revenues
Revenues were $431.6 million for the nine months ended March 31, 2021, an increase of $94.6 million, or 28.1%, compared with $337.0 million for the nine months ended March 31, 2020. The majority of the increase was a result of the acquisitions in the Medical segment contributing $57.7 million (of which $27.4 million was generated by SNC, $21.4 million by Biodex, $8.6 million from AWST and $0.3 million from Dosimetrics). The Industrial segment revenues also increased $35.5 million resulting from increased product orders and release of new products and the positive impact from foreign currency exchange rate fluctuations of $11.0 million. The impact of purchase accounting related to the fair value adjustment of deferred revenue, reduced revenue in the nine months ended March 31, 2021 by $4.3 million.
By segment, revenues for the nine months ended March 31, 2021 were $103.6 million in the Medical segment and $328.0 million in the Industrial segment. Movements in revenues by segment are detailed in the Business Segments section below.
Cost of revenues
Cost of revenues was $259.5 million for the nine months ended March 31, 2021, an increase of $58.3 million, or 29.0% compared to the nine months ended March 31, 2020. Cost of revenues as a percentage of revenues was 60.1% for the nine months ended March 31, 2021, a 0.4% increase compared with 59.7% for the nine months ended March 31, 2020. The increase in cost of revenues was primarily due to acquisitions in our Medical segment contributing $35.9 million (of which $15.9 million was from SNC, $15.2 million from Biodex and $4.8 million from AWST), an increase in our Industrial segment cost of revenues of $19.9 million related to the increase in revenues, including the impacts from foreign currency exchange rate fluctuations of $8.5 million, and $4.9 million of restructuring costs and costs to achieve operational synergies. Cost of revenues includes a $5.2 million increase from purchase accounting related to the fair value of inventory for the nine months ended March 31, 2021.
Selling, general and administrative expenses
Selling, general and administrative (SG&A) expenses were $144.4 million for the nine months ended March 31, 2021, an increase of $28.0 million, or 24.1%, compared to the nine months ended March 31, 2020. SG&A expenses as a percentage of revenues were 33.5% for the nine months ended March 31, 2021, a 1.0 percentage point decrease compared with 34.5% for the nine months ended March 31, 2020. The primary drivers behind the increase in SG&A expenses were the impact of acquisitions in the Medical Segment ($22.4 million combined from AWST, Biodex, Dosimetrics, and SNC), $8.6 million of restructuring costs and costs to achieve operational synergies, mergers and acquisitions, increase in legacy business compensation-related expenses of $7.4 million and the impact from foreign currency exchange rate fluctuations of $3.7 million, partially offset by a decrease in amortization of $4.3 million, and a decrease in travel, entertainment and other operating expenses of $6.1 million.
Research and development
Research and development (R&D) expenses were $21.2 million for the nine months ended March 31, 2021, an increase of $8.9 million, or 72.4%, compared to the nine months ended March 31, 2020. The increase in R&D expense was primarily due to business combinations ($6.0 million combined from AWST, Biodex, Dosimetrics, and SNC), increased R&D activity of $1.7 million to develop new products in the Industrial segment and the impact from foreign currency exchange rate fluctuations of $0.4 million.
Income (loss) from operations
Income (loss) from operations for the nine months ended March 31, 2021 was $6.5 million, a decrease of $0.6 million, or 8.5%, when compared to income of $7.1 million for the nine months ended March 31, 2020. On
269
Table of Contents
a segment basis, income from operations was $6.4 million in the Medical segment, including an $8.4 million increase in purchase accounting impacts described above, and $51.6 million in Industrial segment. Corporate expenses were $51.5 million for the nine months ended March 31, 2021. See Business segments and Mirion corporate and other below for further details.
Interest expense, net
Interest expense, net, was $119.5 million for the nine months ended March 31, 2021 compared to $110.6 million for the nine months ended March 31, 2020. The $8.9 million, or 8.0%, is a non-cash increase in interest related to the shareholder notes which are described in Note 8 to the unaudited interim condensed consolidated financial statements.
Foreign currency (gain) loss, net
The Company recorded a loss of $12.3 million for the nine months ended March 31, 2021, compared to a gain of $4.0 million for the nine months ended March 31, 2020, from foreign currency exchange. The change in net foreign currency losses is due to appreciation in European and Canadian local currencies in relation to the U.S. dollar.
Income taxes
Income tax expense was $6.2 million for the nine months ended March 31, 2021 versus a benefit of $4.8 million in for the nine months ended March 31, 2020. The change is primarily due to the mix of earnings and jurisdictions during each respective period.
Business segments
The following provides detail for business segment results for the nine months ended March 31, 2021 and 2020. Segment income from operations includes revenues of the segment less expenses that are directly related to those revenues, but excludes certain charges to cost of revenues and selling, general and administrative expenses predominantly related to corporate costs, shared overhead and other costs related to restructuring activities and costs to achieve operational initiatives, which are included in Corporate and Other in the table below. Interest expense, loss on debt extinguishment, foreign currency loss (gain), net, and other expense (income), net are not allocated to segments.
For reconciliations of segment revenues and operating income to Mirions consolidated results, see Note 11Segment Information to the unaudited interim condensed consolidated financial statements.
Medical
| (Dollars in millions) |
Nine months ended March 31, 2021 |
Nine months ended March 31, 2020 |
$ Change |
% Change |
||||||||||||
| Revenues |
$ | 103.6 | $ | 44.5 | $ | 59.1 | 132.8 | % | ||||||||
| Income from operations |
$ | 6.4 | $ | 8.9 | $ | (2.5 | ) | (28.1 | %) | |||||||
| Income from operations as a % of revenues |
6.2 | % | 20.0 | % | ||||||||||||
Medical segment revenues were $103.6 million, for the nine months ended March 31, 2021, which is an increase of $59.1 million, or 132.8%, from the nine months ended March 31, 2020. Revenues increased primarily due to the impact of acquisitions contributing $57.7 million (of which $27.4 million was generated by SNC, $21.4 million by Biodex, $8.6 million from AWST and $0.3 million from Dosimetrics) and an increase of
270
Table of Contents
$1.4 million in our legacy business. Additionally, foreign currency exchange rates positively impacted Medical revenues by approximately $0.8 million. The impact of purchase accounting related to the fair value adjustment of deferred revenue, reduced revenue for the nine months ended March 31, 2021 by $4.3 million.
Income (loss) from operations, which excludes non-operational costs, for the nine months ended March 31, 2021 was $6.4 million, a decrease of $2.5 million compared with the nine months ended March 31, 2020. Income from operations as a percentage of revenues decreased approximately 13.8% due in part to lower margins of the acquisitions in the nine months ended March 31, 2021 contributing approximately 11% of the decrease, with the remainder attributable to the legacy business driven by an increase in lower margin product mix. Additionally, income from operations as a percentage of revenues was impacted by the $4.3 million reduction in revenue and $5.2 million increase in cost of revenues resulting from purchase accounting.
Industrial
| (Dollars in millions) |
Nine months ended March 31, 2021 |
Nine months ended March 31, 2020 |
$ Change |
% Change |
||||||||||||
| Revenues |
$ | 328.0 | $ | 292.5 | $ | 35.5 | 12.1 | % | ||||||||
| Income from operations |
$ | 51.6 | $ | 32.7 | $ | 18.9 | 57.8 | % | ||||||||
| Income from operations as a % of revenues |
15.7 | % | 11.2 | % | ||||||||||||
Industrial segment revenues were $328.0 million for the nine months ended March 31, 2021, an increase of $35.5 million, or 12.1% from the nine months ended March 31, 2020. Revenues increased in both product and service revenues, primarily due to new product offerings such as MBD-2 dosimeter and Aegis spectrometer. Foreign currency positively impacted revenues by approximately $12.2 million.
Income (loss) from operations, which excludes non-operational costs, was $51.6 million for the nine months ended March 31, 2021, an increase of $18.9 million compared with the nine months ended March 31, 2020. Income from operations as a percentage of revenues increased 4.5% primarily due to operating expense savings driven primarily by COVID-19 restrictions on employee travel and fixed overhead absorption.
Corporate and other
Corporate and other costs include costs associated with Mirions headquarters located in Georgia, as well as centralized global functions including Executive, Finance, Legal and Compliance, Human Resources, Technology, Strategy, and Marketing and other costs related to company-wide initiatives (e.g., merger and acquisition activities, restructuring and other initiatives). Corporate and other costs were $51.5 million and $34.5 million for the nine months ended March 31, 2021 and March 31, 2020, respectively. The $17.0 million increase in corporate and other expenses during the nine months ended March 31, 2021 versus the comparable period was predominantly expenses related to acquisitions, integrations, operational efficiency costs and strategic initiatives. Functional costs were flat period over period. For reconciliations of segment operating income and corporate and other costs to Mirions consolidated results, see Note 11Segment Information to the unaudited interim condensed consolidated financial statements.
271
Table of Contents
Year ended June 30, 2020 compared to year ended June 30, 2019
| (Dollars in millions) |
2020 | 2019 | $ Change |
% Change |
||||||||||||
| Revenues |
$ | 478.2 | $ | 440.1 | $ | 38.1 | 8.7 | % | ||||||||
| Cost of revenues |
281.2 | 251.9 | 29.3 | 11.6 | % | |||||||||||
|
|
|
|
|
|||||||||||||
| Gross profit |
197.0 | 188.2 | 8.8 | 4.7 | % | |||||||||||
| Selling, general and administrative expenses |
158.1 | 145.4 | 12.7 | 8.7 | % | |||||||||||
| Research and development |
15.9 | 14.0 | 1.9 | 13.6 | % | |||||||||||
|
|
|
|
|
|||||||||||||
| Income from operations |
23.0 | 28.8 | (5.8 | ) | (20.1 | )% | ||||||||||
| Interest expense, net |
149.2 | 143.5 | 5.7 | 4.0 | % | |||||||||||
| Loss on debt extinguishment |
| 12.8 | (12.8 | ) | (100.0 | )% | ||||||||||
| Foreign currency (gain) loss, net |
(0.6 | ) | (3.2 | ) | 2.6 | (81.3 | )% | |||||||||
| Other (income) expense, net |
(1.0 | ) | 1.9 | (2.9 | ) | (152.6 | )% | |||||||||
|
|
|
|
|
|||||||||||||
| Loss before provision from income taxes |
(124.6 | ) | (126.2 | ) | 1.6 | 1.3 | % | |||||||||
| Income tax (benefit) expense |
(5.5 | ) | (4.2 | ) | (1.3 | ) | 31.0 | % | ||||||||
|
|
|
|
|
|||||||||||||
| Net loss |
(119.1 | ) | (122.0 | ) | 2.9 | (2.4 | )% | |||||||||
| Income (loss) attributable to noncontrolling interests |
| | | N/A | ||||||||||||
|
|
|
|
|
|||||||||||||
| Net loss attributable to stockholders |
$ | (119.1 | ) | $ | (122.0 | ) | $ | 2.9 | (2.4 | )% | ||||||
|
|
|
|
|
|||||||||||||
Overview
Revenues for the year ended June 30, 2020 (FY 2020) were $478.2 million, an increase of 8.7% from the year ended June 30, 2019 (FY 2019) primarily driven by acquisitions in FY 2020 in both the Medical and Industrial segment. Cost of revenues increased $29.3 million, or 11.6% primarily reflecting the increase in revenues. Gross profit increased by $8.8 million and as a percentage of revenue for the Company was consistent period over period. There was a net loss of $119.1 million in FY 2020 compared to net loss of $122.0 million in FY 2019. The 2.4% decrease in net loss in FY 2020 is primarily the result of an increase in gross profit, offset by an increase in SG&A of $12.7 million, including $7.6 million of acquisition costs and costs to achieve operational synergies, decreased loss on debt extinguishment of $12.8 million and increase in other income of $2.9 million, increase in interest expense of $5.7 million, and the negative impact of foreign currency exchange of $2.6 million. The impact of purchase accounting related to the fair value of inventory increased our cost of revenues by $1.6 million for FY 2020.
Revenues
Revenues were $478.2 million for FY 2020, an increase of $38.1 million, or 8.7%, compared with $440.1 million for FY 2019. The increase in revenues was primarily due to the impact of FY 2020 acquisitions in both the Medical and Industrial segments ($31.7 million combined from Capintec, Selmic, Premium Analyse, and AWST) and higher volumes in legacy operations.
By segment, revenues were $62.6 million in the Medical segment and $415.6 million in the Industrial segment. Movements in revenues by segment are discussed in greater detail in the Business segment discussion below.
Cost of revenues
Cost of revenues was $281.2 million in FY 2020, an increase of $29.3 million, or 11.6% compared to FY 2019. Cost of revenues as a percentage of revenues was 58.8% for FY 2020, a 1.6% increase compared with 57.2% for FY 2019. The increase in cost of revenues was primarily due to the impact of business combinations ($22.9 million combined from Capintec, Selmic, Premium Analyse, and AWST) and unfavorable product sales mix (i.e., higher sales of products with lower margin versus products with higher margin during the period). The impact of purchase accounting related to the fair value of inventory increased our cost of revenues by $1.6 million in FY 2020.
272
Table of Contents
Selling, general and administrative expenses
SG&A expenses were $158.1 in FY 2020, an increase of $12.7 million, or 8.7%, compared to FY 2019. SG&A expenses as a percent of revenues was 33.1% in FY 2020, compared to 33.0% in FY 2019. The primary drivers behind the increase in SG&A expenses were expenses associated with business combinations ($7.1 million combined from Capintec, Selmic, Premium Analyse, and AWST), an increase of $7.6 million in costs to achieve operational synergies and mergers and acquisitions, increase in professional fees of $2.1 million, partially offset by a decrease in amortization expense of $4.3 million.
Research and development
R&D expenses were $15.9 million in FY 2020, an increase of $1.9 million, or 13.6%, compared to FY 2019. The increase in R&D expenses was primarily due to business combinations ($1.3 million combined from Capintec, Selmic, Premium Analyse, and AWST) and increased R&D activity in existing businesses to develop new products ($0.9 million).
Income (loss) from operations
Income from operations in FY 2020 was $23.0 million, a decrease of $5.8 million, or 20.1%, when compared to income from operations of $28.8 million in FY 2019. On a segment basis, income from operations was $13.9 million in the Medical segment and $59.6 million in the Industrial segment in FY 2020 compared to $10.2 million in Medical and $55.0 million in Industrial in FY 2019. Corporate expenses were $50.5 million in FY 2020 compared to $36.4 million in FY 2019. See Business segments and Mirion corporate and other below for further details.
Interest expense
Interest expense, net was $149.2 million in FY 2020 and $143.5 million in FY 2019. The $5.7 million, or 4.0%, increase in interest expense in FY 2020 was due primarily to increased non-cash interest of $11.9 million on related-party shareholder notes, partially offset by a decrease in interest expense related to our third-party debt due to lower interest rates on third-party debt. See Note 8Borrowings in the consolidated financial statements.
Loss on debt extinguishment
There was no loss on debt extinguishment in FY 2020, as compared to the FY 2019 loss on extinguishment of $12.8 million. In FY 2019, Mirion entered into a new credit agreement, resulting in the extinguishment of previous debt. No similar debt extinguishment occurred in FY 2020.
Foreign currency (gain) loss, net
The Company recorded a gain of $0.6 million in FY 2020, compared to a gain of $3.2 million from foreign currency exchange in FY 2019. Foreign currency gain decreased $2.6 million, or 81.3%, primarily due to less favorable exchange rates in FY 2020 between the U.S. dollar and currencies used in Mirions European operations.
Other (income) expenses, net
Other income was $1.0 million in FY 2020, compared to other expense of $1.9 million in FY 2019. The change in other (income) expenses, net from FY 2019 is primarily due to investment income received in FY 2020 compared to losses recorded on the disposal of property, plant, and equipment in FY 2019.
273
Table of Contents
Income taxes
Income tax benefit was $5.5 million in FY 2020 as compared to income tax benefit of $4.2 million in FY 2019, which increased $1.3 million due to the impact of the release of unrecognized tax benefits related to uncertain tax positions offset by increases in valuation allowances and mix of income between U.S. and foreign operations.
Business segments
The following is an analysis of business segment results for FY 2020 as compared with FY 2019. Segment income from operations is defined as revenues less cost of revenues, segment selling, general and administrative expenses, and research and development expenses. Costs not specifically allocated to segment operating include those discussed in further detail in the Corporate and other section below. Interest expense, loss on debt extinguishment, foreign currency gain, and other income / expense are not allocated to segments. For reconciliations of segment revenues and earnings to Mirions consolidated results, see Note 15Segment Information to the consolidated financial statements included elsewhere in this proxy statement/prospectus.
Medical
| (Dollars in millions) |
June 30, 2020 |
June 30, 2019 |
$ Change |
% Change |
||||||||||||
| Revenues |
$ | 62.6 | $ | 42.9 | $ | 19.7 | 45.9 | % | ||||||||
| Income from operations |
$ | 13.9 | $ | 10.2 | $ | 3.7 | 36.3 | % | ||||||||
| Income from operations as a % of revenues |
22.2 | % | 23.8 | % | ||||||||||||
Medical revenues were $62.6 million in FY 2020, an increase of $19.7 million or 45.9% from FY 2019 primarily due to the impact of business combinations ($17.7 million from Capintec and AWST in FY 2020 and $0.9 million from the full fiscal year impact of the NRG Dosimetry Services Group (NRG) FY 2019 acquisition).
Income from operations was $13.9 million in FY 2020, representing an increase in earnings of $3.7 million, or 36.3%, from FY 2019 primarily due to the impact of business combinations ($1.3 million from Capintec and AWST), higher gross profit from legacy operations of $1.5 million due to product mix, and lower amortization expense related to legacy operations of $0.7 million. Income from operations as a percentage of revenues declined 1.6% primarily due to the product mix impact of business combinations, as certain products had lower margins than Mirions legacy medical businesses.
Industrial
| Dollars in millions) |
June 30, 2020 |
June 30, 2019 |
$ Change |
% Change |
||||||||||||
| Revenues |
$ | 415.6 | $ | 397.2 | $ | 18.4 | 4.6 | % | ||||||||
| Income from operations |
$ | 59.6 | $ | 55.0 | $ | 4.6 | 8.4 | % | ||||||||
| Income from operations as a % of revenues |
14.3 | % | 13.8 | % | ||||||||||||
Industrial revenues were $415.6 million in FY 2020, an increase of $18.4 million, or 4.6% from FY 2019. Revenues increased primarily due to business combinations ($14.0 million from Selmic and Premium Analyse), new product offerings, and government year-end purchases driving increased revenues from certain customers.
Income from operations, which includes an inventory valuation impact of $1.3 million but excludes non-operational costs, was $59.6 million in FY 2020, an increase of $4.6 million, or 8.4%, compared with the prior year period, while income from operations as a percentage of revenues increased 0.5%. The $4.6 million increase in income from operations was primarily due to the impact of business combinations ($1.3 million from
274
Table of Contents
Selmic and Premium Analyse), lower amortization expense related to legacy operations of $3.4 million and reduced travel expenses of $1.3 million, offset by lower gross profit impact of approximately $1.4 million from legacy operations due to product mix.
Corporate and other
Corporate and other costs include costs associated with Mirions headquarters, as well as centralized global functions including Executive, Finance, Legal and Compliance, Human Resources, Technology, Strategy, and Marketing. Corporate and other costs were $50.5 million and $36.4 million in the 2020 and 2019 periods, respectively. The $14.1 million increase in corporate and other expenses in FY 2020 versus the comparable prior year period was primarily the result of $8.8 million increase in costs to achieve synergies, acquisition, integration and strategic initiatives, an increase of $2.0 in compensation and related costs and $1.8 million increase in professional fees. For reconciliations of segment operating income and corporate and other costs to Mirions consolidated results, see Note 15Segment Information to the consolidated financial statements.
Year ended June 30, 2019 compared to year ended June 30, 2018
| (Dollars in millions) |
2019 | 2018 | $ Change |
% Change |
||||||||||||
| Revenues |
$ | 440.1 | $ | 444.1 | $ | (4.0 | ) | (0.9 | )% | |||||||
| Cost of revenues |
251.9 | 275.7 | (23.8 | ) | (8.6 | )% | ||||||||||
|
|
|
|
|
|||||||||||||
| Gross profit |
188.2 | 168.4 | 19.8 | 11.8 | % | |||||||||||
| Selling, general and administrative expenses |
145.4 | 153.8 | (8.4 | ) | (5.5 | )% | ||||||||||
| Research and development |
14.0 | 19.3 | (5.3 | ) | (27.5 | )% | ||||||||||
|
|
|
|
|
|||||||||||||
| Income (loss) from operations |
28.8 | (4.7 | ) | 33.5 | (712.8 | )% | ||||||||||
| Interest expense, net |
143.5 | 128.9 | 14.6 | 11.3 | % | |||||||||||
| Loss on debt extinguishment |
12.8 | | 12.8 | N/A | ||||||||||||
| Foreign currency (gain) loss, net |
(3.2 | ) | 5.3 | (8.5 | ) | (160.4 | )% | |||||||||
| Other (income) expense, net |
1.9 | 1.6 | 0.3 | 18.8 | % | |||||||||||
|
|
|
|
|
|||||||||||||
| Loss before benefit from income taxes |
(126.2 | ) | (140.5 | ) | 14.3 | 10.2 | % | |||||||||
| Income tax expense (benefit) |
(4.2 | ) | (36.8 | ) | 32.6 | (88.6 | )% | |||||||||
|
|
|
|
|
|||||||||||||
| Net loss |
(122.0 | ) | (103.7 | ) | (18.3 | ) | 17.6 | % | ||||||||
| Loss attributable to noncontrolling interests |
| (0.3 | ) | 0.3 | (100.0 | )% | ||||||||||
| Net loss attributable to stockholders |
$ | (122.0 | ) | $ | (103.4 | ) | $ | (18.6 | ) | 18.0 | % | |||||
|
|
|
|
|
|||||||||||||
Overview
Revenues were $440.1 million for the year ended June 30, 2019 (FY 2019) and $444.1 million for the year ended June 30, 2018 (FY 2018). Cost of revenues decreased $23.8 million primarily due to lower margin product mix and a decrease in costs to achieve synergies. There was a net loss of $122.0 million in 2019 compared to a net loss of $103.7 million in 2018. The 17.6% increase in net loss in 2019 is primarily the result of increase in gross profit, a decrease in SG&A of $8.4 million driven by a decrease in costs to achieve operational synergies and mergers and acquisitions, decreased income tax benefit of $32.6 million, and a positive impact of fluctuation in foreign currency of approximately $8.5 million, partially offset by increased net interest expense of $14.6 million, and increased loss on debt extinguishment of $12.8 million.
Revenues
Revenues were $440.1 million in FY 2019, a decrease of $4.0 million, or 0.9%, compared with $444.1 million in FY 2018. Product revenues decreased $7.2 million primarily due to lower volumes and unfavorable impacts of
275
Table of Contents
foreign currency. Service revenues increased $3.2 million primarily due to business combinations ($1.9 million from NRG Dosimetry Services Group (NRG)) and $1.3 million from our legacy Medical segment business.
By segment, FY 2019 revenues were $42.9 million in Medical and $397.2 million in Industrial. Changes in revenues by segment are detailed in the Business Segments section below.
Cost of revenues
Cost of revenues were $251.9 million in FY 2019, a decrease of $23.8 million, or 8.6% compared to FY 2018. Cost of revenues as a percentage of revenues was 57.2% for FY 2019, a 4.9% decrease compared with 62.1% for FY 2018. The decrease in cost of revenues was primarily due to improved efficiencies in the production of certain dosimeter products, favorable product mix across other offerings and costs and to achieve operational synergies decreased $8.7 million year over year.
Selling, general and administrative expenses
SG&A expenses were $145.4 million in FY 2019, a decrease of $8.4 million, or 5.5%, compared to FY 2018. SG&A expenses as a percentage of revenues were 33.0% in FY 2019, a 1.6 percentage point decrease compared with 34.6% in FY 2018. The primary driver behind the decrease in SG&A expenses was reduced integration and synergy charges associated with the FY 2017 acquisition of Canberra.
Research and development
R&D expenses were $14.0 million in FY 2019, a decrease of $5.3 million, or 27.5%, compared to FY 2018. The decrease in R&D expenses was due to approximately $3.7 million of reduced investment in product redesigns and fewer new products released in FY 2019 compared to FY 2018 and lower compensation expense and professional fees of $1.4 million year over year.
Income (loss) from operations
Income from operations in FY 2019 was $28.8 million, an increase of $33.5 million when compared to loss from operations of $4.7 million in FY 2018. On a segment basis, income from operations was $10.2 million in the Medical segment and $55.0 million in Industrial in FY 2019 and $6.0 million in the Medical segment and $46.9 million in the Industrial segment in FY 2018. Corporate expenses were $36.4 million in FY 2019 compared to $57.6 million in FY 18. See Business segments and Mirion corporate and other below for further details.
Interest expense, net
Interest expense, net, was $143.5 million in FY 2019 compared to $128.9 million in FY 2018. The $14.6 million increase is primarily due to higher non-cash interest on our shareholder notes of $10.2 million and higher interest of $4.4 million on third-party debt related to increased borrowings in FY 2019 to finance business acquisition activity.
Loss on debt extinguishment
Loss on debt extinguishment was $12.8 million in FY 2019 as compared to no debt extinguishment in FY 2018. In FY 2019, Mirion entered into a new credit agreement, resulting in the extinguishment of previous debt. No similar extinguishment occurred in FY 2018.
Foreign currency gain (loss), net
The Company recorded a gain of $3.2 million in FY 2019, compared to a loss of $5.3 million from foreign currency exchange in FY 2018. This change is primarily due to more favorable foreign currency exchange rates between the U.S. dollar and the Euro.
276
Table of Contents
Other expenses, net
Other expenses, net, were $1.9 million in FY 2019, an increase of $0.3 million, or 18.8%, compared with FY 2018. The increase is primarily due to increased losses on disposals of property, plant, and equipment.
Income taxes
Income tax benefit was $4.2 million in FY 2019 versus a benefit of $36.8 million in FY 2018. The FY 2019 tax benefits are lower than FY 2018 primarily due the impacts of U.S. tax reform in FY 2018, which resulted in the recognition of deferred tax benefits for the revaluation of the U.S. net deferred tax liability and the reversal of liability for investment in foreign earnings as a result of the one-time transition tax net of foreign tax credits.
The 2017 Tax Act was enacted on December 22, 2017. Among other things, effective in the first taxable year after the enactment, the 2017 Tax Act reduces the U.S. federal corporate tax rate from 35% to 21% and exempts from U.S. federal income taxation dividends from certain foreign corporations to their U.S. stockholders. The 2017 Tax Act also requires U.S. companies to pay a one-time transition tax on earnings of certain foreign corporate subsidiaries that were previously deferred from U.S. taxation. Further, the 2017 Tax Act impacts certain deductions by limiting or eliminating them and it subjects certain foreign earnings to U.S. tax.
Business segments
The following provides detail for business segment results for the years ended June 30, 2019 and 2018. Segment income from operations is defined as revenues less cost of revenues, segment selling, general and administrative expenses, and research and development expenses. Costs not specifically allocated to segment operating income include those discussed in further detail in the Corporate and other section below. Interest expense, loss on debt extinguishment, foreign currency gain, and other income / expense are not allocated to segments. For reconciliations of segment revenues and earnings to Mirions consolidated results, see Note 15Segment Information to the consolidated financial statements.
Medical
| (Dollars in millions) |
June 30, 2019 |
June 30, 2018 |
$ Change |
% Change |
||||||||||||
| Revenues |
$ | 42.9 | $ | 37.8 | $ | 5.1 | 13.5 | % | ||||||||
| Income from operations |
$ | 10.2 | $ | 6.0 | $ | 4.2 | 70.0 | % | ||||||||
| Income from operations as a % of revenues |
23.8 | % | 15.9 | % | ||||||||||||
Medical revenues were $42.9 million in FY 2019, an increase of $5.1 million, or 13.5% from FY 2018. Revenues increased primarily due to the performance of the Instadose Dosimetry platform increasing $3.1 million and fiscal year business combinations ($1.9 million from NRG).
Income from operations in FY 2019 was $10.2 million, an increase of $4.2 million compared with FY 2018. Income from operations as a percentage of revenues improved 7.9% from increased revenues combined with favorable overhead and consumable costs primarily as a result of improved efficiencies in the production of certain dosimeter products.
Industrial
| (Dollars in millions) |
June 30, 2019 |
June 30, 2018 |
$ Change |
% Change |
||||||||||||
| Revenues |
$ | 397.2 | $ | 406.3 | $ | (9.1 | ) | (2.2 | )% | |||||||
| Income from operations |
$ | 55.0 | $ | 46.9 | $ | 8.1 | 17.3 | % | ||||||||
| Income from operations as a % of revenues |
13.8 | % | 11.5 | % | ||||||||||||
277
Table of Contents
Industrial revenues were $397.2 million in FY 2019, a decrease of $9.1 million, or 2.2% from FY 2018. This decrease reflects unfavorable impacts from lower recurring revenues from our nuclear power plant customers, closure of the Mirion Russia site and lower military detection and identification orders, as well as the impact of unfavorable impacts of foreign currency.
Income from operations was $55.0 million in FY 2019, an increase of $8.1 million compared with FY 2018. Income from operations as a percentage of revenues improved 2.2% from the completion of synergy and integration initiatives related to the FY 2017 acquisition of Canberra in FY 2018 (non-recurring in FY 2019) and favorable product mix.
Corporate and Other
Corporate and other costs include costs associated with Mirions headquarters, as well as centralized global functions including Executive, Finance, Legal and Compliance, Human Resources, Technology, Strategy, and Marketing. Corporate and other costs were $36.4 million and $57.6 million in FY 2019 and FY 2018, respectively. The $21.2 million decrease in corporate and other expenses in FY 2019 versus FY 2018 was primarily the result of decreased acquisition and integration costs and costs to achieve operational synergies in FY 2019. Functional costs were primarily flat year over year. For reconciliations of segment operating income and corporate and other costs to Mirions consolidated results, see Note 15Segment Information to the consolidated financial statements.
Quarterly Results of Operations
The following table sets forth selected unaudited quarterly financial data for our last eight completed fiscal quarters. The information for each of these quarters reflects all adjustments that are of a normal, recurring nature and that we consider necessary for a fair presentation of our operating results for such periods. The quarterly results of operations presented should be read in conjunction with our audited consolidated financial statements and notes thereto appearing elsewhere in this document and are not necessarily indicative of our operating results for any future period. Revenues for certain quarters are impacted by the capital spending patterns of government customers, which are influenced by budgetary considerations and driven by timing of fiscal year-ends.
| Three Months Ended | ||||||||||||||||||||||||||||||||
| ($ in millions) | March 31, 2021 |
December 31, 2020 |
September 30, 2020 |
June 30, 2020 |
March 31, 2020 |
December 31, 2019 |
September 30, 2019 |
June 30, 2019 |
||||||||||||||||||||||||
| Revenues |
$ | 166.2 | $ | 150.8 | $ | 114.6 | $ | 141.2 | $ | 109.8 | $ | 132.1 | $ | 95.1 | $ | 126.7 | ||||||||||||||||
| Adjusted revenues(1)(2) |
$ | 170.5 | $ | 150.8 | $ | 114.6 | $ | 141.4 | $ | 109.8 | $ | 132.1 | $ | 95.1 | $ | 126.7 | ||||||||||||||||
| Net (loss) income |
$ | (71.4 | ) | $ | (19.2 | ) | $ | (40.4 | ) | $ | (24.5 | ) | $ | (36.4 | ) | $ | (22.5 | ) | $ | (35.7 | ) | $ | (15.5 | ) | ||||||||
| Adjusted net (loss) income(1)(3) |
$ | (40.7 | ) | $ | 3.7 | $ | (20.9 | ) | $ | (5.4 | ) | $ | (24.7 | ) | $ | (7.1 | ) | $ | (26.1 | ) | $ | (2.3 | ) | |||||||||
| EBITA(1)(4) |
$ | 13.8 | $ | 16.4 | $ | 8.8 | $ | 25.9 | $ | 17.9 | $ | 20.4 | $ | 11.1 | $ | 30.2 | ||||||||||||||||
| EBITDA(1)(4) |
$ | 18.8 | $ | 21.0 | $ | 13.1 | $ | 30.4 | $ | 22.1 | $ | 24.6 | $ | 16.1 | $ | 34.4 | ||||||||||||||||
| Adjusted EBITDA(1)(4) |
$ | 39.8 | $ | 38.3 | $ | 24.1 | $ | 40.9 | $ | 25.0 | $ | 32.9 | $ | 15.9 | $ | 39.6 | ||||||||||||||||
| (1) | Adjusted revenues, Adjusted net (loss) income, EBITA, EBITDA and Adjusted EBITDA are supplemental measures of our performance that are not required by, or presented in accordance with, U.S. GAAP. Adjusted revenues, Adjusted net (loss) income, EBITA, EBITDA, and Adjusted EBITDA are included in this document because they are key metrics used by management to assess our financial performance. We believe that these measures are useful because they provide investors with information regarding our operating performance that is used by our management in its reporting and planning processes. These measures may not be comparable to similarly titled measures and disclosures reported by other companies. |
278
Table of Contents
| Adjusted revenues are defined as U.S. GAAP revenues adjusted to remove the impact of purchase accounting on the recognition of deferred revenue. We have acquired businesses whose net tangible assets include deferred revenue. In accordance with GAAP reporting requirements, we recorded adjustments reducing deferred revenue under arrangements predating the business combination to fair value. Therefore, our GAAP revenues after the date of acquisition will not reflect the full amount of revenues that would have been reported if the acquired deferred revenue was not written down to fair value. Therefore, Adjusted revenues reverses the impact of this deferred revenue write-down to provide another view of the revenue run-rate in a given period and providing meaningful information for comparative results in future periods. |
Adjusted net (loss) income is defined as U.S. GAAP net income adjusted for foreign currency gains and losses, amortization of acquired intangible assets, the impact of purchase accounting on the recognition of deferred revenue, certain non-operating expenses (certain purchase accounting impacts related to revenues and inventory, restructuring and costs to achieve operational synergies, merger and acquisition expenses and IT project implementation expenses), and income tax impacts of these adjustments.
EBITA is defined as income before net interest expenses (including loss on debt extinguishment), income tax (benefit) provision, and amortization. EBITDA is defined as income before net interest expense (including loss on debt extinguishment), income tax (benefit) provision, and depreciation and amortization. EBITA and EBITDA are not terms defined under U.S. GAAP and do not purport to be alternatives to net income as measures of operating performance or to cash flows from operating activities as measures of liquidity. Additionally, EBITA and EBITDA are not intended to be measures of free cash flow available for managements discretionary use as they do not consider certain cash requirements such as interest payments, tax payments and debt service requirements.
Adjusted EBITDA is defined as EBITDA excluding the items described in the table below. Adjusted EBITDA is used by management as a measure of operating performance. We believe that the inclusion of supplementary adjustments to EBITDA applied in presenting Adjusted EBITDA is appropriate to provide additional information to investors about our results of operations that management utilizes on an ongoing basis to assess our core operating performance.
EBITA, EBITDA and Adjusted EBITDA may not be comparable to similarly titled measures used by other companies. You should not consider our EBITA, EBITDA and Adjusted EBITDA as alternatives to operating income or net income, determined in accordance with U.S. GAAP.
| (2) | The following table reconciles Adjusted revenues to the most directly comparable U.S. GAAP financial performance measure, which is revenues: |
| Three Months Ended | ||||||||||||||||||||||||||||||||
| ($ in millions) | March 31, 2021 |
December 31, 2020 |
September 30, 2020 |
June 30, 2020 |
March 31, 2020 |
December 31, 2019 |
September 30, 2019 |
June 30, 2019 |
||||||||||||||||||||||||
| Revenues |
$ | 166.2 | $ | 150.8 | $ | 114.6 | $ | 141.2 | $ | 109.8 | $ | 132.1 | $ | 95.1 | $ | 126.7 | ||||||||||||||||
| Revenue reduction from purchase accounting |
4.3 | | | 0.2 | | | | | ||||||||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||||
| Adjusted revenues |
$ | 170.5 | $ | 150.8 | $ | 114.6 | $ | 141.4 | $ | 109.8 | $ | 132.1 | $ | 95.1 | $ | 126.7 | ||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||||
279
Table of Contents
| (3) | The following table reconciles Adjusted net (loss) income to the most directly comparable U.S. GAAP financial performance measure, which is net loss: |
| Three Months Ended | ||||||||||||||||||||||||||||||||
| ($ in millions) | March 31, 2021 |
December 31, 2020 |
September 30, 2020 |
June 30, 2020 |
March 31, 2020 |
December 31, 2019 |
September 30, 2019 |
June 30, 2019 |
||||||||||||||||||||||||
| Net loss |
$ | (71.4 | ) | $ | (19.2 | ) | $ | (40.4 | ) | $ | (24.5 | ) | $ | (36.4 | ) | $ | (22.5 | ) | $ | (35.7 | ) | $ | (15.5 | ) | ||||||||
| Revenue reduction from purchase accounting |
4.3 | | | 0.2 | | | | | ||||||||||||||||||||||||
| Cost of revenues impact from inventory valuation purchase accounting |
4.7 | 0.5 | | 0.5 | 0.5 | 0.4 | 0.2 | 0.1 | ||||||||||||||||||||||||
| Foreign currency (gain) loss, net |
(4.0 | ) | 8.2 | 8.1 | 3.4 | (2.0 | ) | 4.7 | (6.7 | ) | 2.6 | |||||||||||||||||||||
| Amortization of acquired intangibles |
18.6 | 13.5 | 12.2 | 12.4 | 12.7 | 12.7 | 12.8 | 13.1 | ||||||||||||||||||||||||
| Non-operating expenses |
16.1 | 8.5 | 2.9 | 6.4 | 4.3 | 3.2 | 6.2 | 2.4 | ||||||||||||||||||||||||
| Tax impact of adjustments above |
(9.0 | ) | (7.8 | ) | (3.7 | ) | (3.8 | ) | (3.8 | ) | (5.6 | ) | (2.9 | ) | (5.0 | ) | ||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||||
| Adjusted net (loss) income |
$ | (40.7 | ) | $ | 3.7 | $ | (20.9 | ) | $ | (5.4 | ) | $ | (24.7 | ) | $ | (7.1 | ) | $ | (26.1 | ) | $ | (2.3 | ) | |||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||||
| (4) | The following table reconciles EBITA, EBITDA and Adjusted EBITDA to the most directly comparable U.S. GAAP financial performance measure, which is net loss: |
| Three Months Ended | ||||||||||||||||||||||||||||||||
| ($ in millions) | March 31, 2021 |
December 31, 2020 |
September 30, 2020 |
June 30, 2020 |
March 31, 2020 |
December 31, 2019 |
September 30, 2019 |
June 30, 2019 |
||||||||||||||||||||||||
| Net loss |
$ | (71.4 | ) | $ | (19.2 | ) | $ | (40.4 | ) | $ | (24.5 | ) | $ | (36.4 | ) | $ | (22.5 | ) | $ | (35.7 | ) | $ | (15.5 | ) | ||||||||
| Interest expense, net |
43.0 | 38.5 | 38.0 | 38.7 | 39.2 | 35.9 | 35.5 | 36.8 | ||||||||||||||||||||||||
| Income tax (benefit) provision |
23.6 | (16.4 | ) | (1.0 | ) | (0.7 | ) | 2.4 | (5.7 | ) | (1.5 | ) | (4.2 | ) | ||||||||||||||||||
| Amortization |
18.6 | 13.5 | 12.2 | 12.4 | 12.7 | 12.7 | 12.8 | 13.1 | ||||||||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||||
| EBITA |
$ | 13.8 | $ | 16.4 | $ | 8.8 | $ | 25.9 | $ | 17.9 | $ | 20.4 | $ | 11.1 | $ | 30.2 | ||||||||||||||||
| Depreciation |
5.0 | 4.6 | 4.3 | 4.5 | 4.2 | 4.2 | 5.0 | 4.2 | ||||||||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||||
| EBITDA |
$ | 18.8 | $ | 21.0 | $ | 13.1 | $ | 30.4 | $ | 22.1 | $ | 24.6 | $ | 16.1 | $ | 34.4 | ||||||||||||||||
| Stock compensation expense |
(0.1 | ) | 0.1 | | | 0.1 | | 0.1 | (0.1 | ) | ||||||||||||||||||||||
| Debt extinguishment |
| | | | | | | 0.2 | ||||||||||||||||||||||||
| Foreign currency (gain) loss, net |
(4.0 | ) | 8.2 | 8.1 | 3.4 | (2.0 | ) | 4.7 | (6.7 | ) | 2.6 | |||||||||||||||||||||
280
Table of Contents
| Three Months Ended | ||||||||||||||||||||||||||||||||
| ($ in millions) | March 31, 2021 |
December 31, 2020 |
September 30, 2020 |
June 30, 2020 |
March 31, 2020 |
December 31, 2019 |
September 30, 2019 |
June 30, 2019 |
||||||||||||||||||||||||
| Revenue reduction from purchase accounting |
$ | 4.3 | $ | | $ | | $ | 0.2 | $ | | $ | | $ | | $ | | ||||||||||||||||
| Cost of revenues impact from inventory valuation purchase accounting |
4.7 | 0.5 | | 0.5 | 0.5 | 0.4 | 0.2 | 0.1 | ||||||||||||||||||||||||
| Non-operating expenses |
16.1 | 8.5 | 2.9 | 6.4 | 4.3 | 3.2 | 6.2 | 2.4 | ||||||||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||||
| Adjusted EBITDA |
$ | 39.8 | $ | 38.3 | $ | 24.1 | $ | 40.9 | $ | 25.0 | $ | 32.9 | $ | 15.9 | $ | 39.6 | ||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||||
Liquidity and Capital Resources
Overview of Liquidity
Mirions primary future cash needs relate to working capital, operating activities, capital spending, strategic investments, and debt service.
Mirion management believes that net cash provided by operating activities, augmented by long-term debt arrangements, will provide adequate liquidity for the next 12 months of independent operations, as well as the resources necessary to invest for growth in existing businesses and manage its capital structure on a short- and long-term basis. Access to capital and availability of financing on acceptable terms in the future will be affected by many factors, including Mirions credit rating, economic conditions, and the overall liquidity of capital markets. There can be no assurance of continued access to capital markets on acceptable terms.
At March 31, 2021, and June 30, 2020, Mirion had $86.8 million and $118.4 million, respectively, in cash and cash equivalents, which include amounts held by entities outside of the United States of approximately $49.2 million and $73.7 million, respectively, primarily in Europe and Canada. Non-U.S. cash is generally available for repatriation without legal restrictions, subject to certain taxes, mainly withholding taxes. Mirion is not asserting indefinite reinvestment of cash for certain non-U.S. subsidiaries due to the outstanding debt obligations in instances where alternative repatriation options other than dividends are not available. The 2019 Credit Facility provides for up to $90.0 million of revolving borrowings. As of March 31, 2021, Mirion had borrowing availability of $81.1 million under the 2019 Credit Facility after giving effect to $8.9 million of outstanding letters of credit.
Long-term debt obligations
There is a discussion in Note 8 of the unaudited condensed consolidated financial statements and Note 8 of the consolidated financial statements included elsewhere in this proxy statement/prospectus of the long-term debt arrangements issued by Mirion.
Debt Profile
Third Party Debt Before the Business Combination
In March 2019, Mirion Technologies (HoldingRep), Ltd., a wholly owned subsidiary of the Company, and its subsidiaries entered into a credit agreement with Morgan Stanley Senior Funding Inc., as administrative agent,
281
Table of Contents
certain other revolving lenders and a syndicate of institutional lenders (collectively, the Existing Credit Facility). The Existing Credit Facility originally provided for financing of a $450.0 million senior secured term loan facility and a 125 million senior secured term loan facility, as well as a $90.0 million revolving line of credit. The Existing Credit Facility was amended to provide an additional $34.0 million, $66.0 million and $225.0 million of senior secured term loans in July 2019, December 2019 and December 2020, respectively. As of March 31, 2021, the aggregate principal amount of the term loan outstanding under the Existing Credit Facility was $907.0 million.
The USD portion of the term loan has a 7-year term (maturing in March 2026), bears interest at the greater of Adjusted London Interbank Offered Rate (LIBOR) or 0%, plus 4.00%. The interest rate was 4.20% and 5.07% at March 31, 2021 and June 30, 2020, respectively. The Company repaid $5.3 million and $4.1 million for the nine-months ended March 31, 2021 and 2020, respectively. The Euro portion of the term loan also has a 7-year term (maturing in March 2026), bears interest at the greater of European Union interbank market (Euribor) or 0%, plus 4.25%. Annual principal payments on the first lien term loan facilities are due quarterly, and are (x) $1,947,108.17 per quarter with respect to the US Dollar denominated term loans and (y) 312,500 per quarter with respect to the Euro denominated term loans. As of March 31, 2021 and June 30, 2020, the interest rate was 4.25%. The Company repaid $1.1 million and $0.6 million during the nine-months ended March 31, 2021 and 2020, respectively. The revolving line of credit has a 5-year term and bears interest at the greater of LIBOR or 0%, plus 4.00%. The agreement requires the payment of a commitment fee of 0.50% per annum for unused revolving commitments. The revolving line of credit matures in March 2024, at which time all outstanding revolving facility loans and accrued and unpaid interest are due. Any outstanding letters of credit issued under the Existing Credit Agreement reduce the availability under the revolving line of credit. The outstanding balance under the revolving credit facility was $35.0 million as of June 30, 2020, with no outstanding balance as of March 31, 2021. Additionally, the Company has standby letters of credit issued under the Existing Credit Facility that reduced the availability under the revolving credit facility by $8.9 million and $9.0 million at March 31, 2021 and June 30, 2020, respectively, the amount available on the revolving credit facility was approximately $81.1 million and $46.0 million, for the same periods, respectively
The Existing Credit Facility is secured by a first priority lien on substantially all of the assets of MT HoldingRep and its borrower and guarantor subsidiaries organized in the United States and by certain assets of borrower or guarantor subsidiaries organized in Germany, United Kingdom, Canada, France, Belgium and Luxembourg, in each case, subject to customary exceptions and limitations. Loan fees recorded as debt discounts are amortized using the effective interest method. The Existing Credit Facility contains customary affirmative and negative covenants. The revolving facility also contains a financial covenant that requires MT HoldingRep and subsidiaries, under certain conditions, to maintain a consolidated first lien secured debt to consolidated EBITDA ratio (as defined in the Existing Credit Facility) of 7.70:1.00. The negative covenants, subject to certain thresholds and exceptions, generally limit the ability of MT HoldingRep and subsidiaries to, among other things, incur additional debt, create liens, make fundamental changes, make certain investments, pay dividends, purchase or retire equity interests, or prepay or retire certain debt. The covenants also contain limitations on the activities of MT HoldingRep as the passive holding company.
The Existing Credit Facility will be repaid in full upon the consummation of the Business Combination and replaced with the New Credit Facilities (as defined below). In addition to the Existing Credit Facility, at March 31, 2021, the Company had other outstanding third-party debt with an aggregate principal amount of $1.5 million, of which $0.3 million will be paid off at the Closing, the remainder of which will be unaffected by the closing of the Business Combination. At March 31, 2021, the Company also had $1,170.3 million in notes payable to related parties, all of which will be extinguished upon the closing of the Business Combination. See Note 8. Borrowings in the Companys unaudited interim condensed consolidated financial statements as of, and for the nine months ended, March 31, 2021, included elsewhere in this proxy statement / prospectus.
Debt After the Business Combination
In connection with the Business Combination Agreement, Mirion Technologies (HoldingSub2) Ltd. (MT HoldingSub2) entered into a Commitment Letter (the Debt Commitment Letter) with Goldman Sachs Lending
282
Table of Contents
Partners LLC (GS Lending) and Citigroup Global Markets Inc. (Citi) and certain other financial institutions that may become party thereto from time to time (GS Lending and Citi, together with those other financial institutions that may also become party to the Debt Commitment Letter, the financing sources), pursuant to which the financing sources have committed to provide to Mirion Technologies (US), Inc., as the subsidiary borrower, and a parent entity of the Mirion TopCo business to be formed (which will be an indirect subsidiary of Mirion TopCo), senior secured credit facilities to be used to, among other things (i) finance the transactions contemplated by the Business Combination Agreement, (ii) repay and terminate the existing indebtedness of Mirion (the Debt Refinancing) and (iii) pay all fees, premiums, expenses and other transaction costs incurred in connection with the foregoing. The aggregate commitment of the senior secured credit facilities consists of a $830 million dollar-denominated first lien term facility and a $90 million revolving credit facility (of which up to $50 million will be available for letters of credit) (collectively, the New Credit Facilities). Upon the consummation of the Business Combination, all or a portion of the New Credit Facilities will become the direct obligations of MT HoldingSub2 or one or more subsidiaries of MT HoldingSub2 and will be secured by substantially all of MT HoldingSub2s and the Guarantors assets (subject to customary exclusions and limitations), as further described under Description of New Mirion Indebtedness elsewhere in this proxy statement/prospectus.
Borrowings under the New Credit Facilities will accrue interest at a rate of LIBOR plus an applicable margin to be determined with respect to the term loans and 3.00% with respect to the revolving credit facility. The agreement requires the payment of a commitment fee of 0.50% per annum for unused revolving commitments. The first lien term facility will mature on the seventh anniversary of the date of the closing of the Business Combination. The revolving facility will mature on the fifth anniversary of the date of the closing of the Business Combination. The definitive documentation for the New Credit Facilities has not been finalized and, accordingly, the actual terms of the debt financing, including the interest rate, may differ from those described in this proxy statement/prospectus.
The obligations of the borrowers under the New Credit Facilities and certain of their (and their subsidiaries) respective obligations under hedging arrangements and cash management arrangements will be unconditionally guaranteed by MT HoldingSub2 and each existing and subsequently acquired or organized direct or indirect wholly-owned US subsidiary of the borrowers (collectively, the Guarantors), in each case, other than certain excluded subsidiaries and subject to other customary limitations to be set forth in the definitive documentation with respect to the debt financing. The New Credit Facilities will be secured by a security interest in substantially all of the assets of the Guarantors (and including a pledge of the equity interests of each borrower and of each Guarantor and of their respective direct, wholly-owned restricted subsidiaries), in each case subject to customary exceptions (including exceptions for certain adverse tax consequences).
The revolving facility will require MT HoldingSub2 to maintain a maximum first lien net leverage ratio (to be defined in the definitive documentation with respect to the debt financing) at a level equal to a single first lien net leverage ratio level calculated based on providing a 40% cushion to consolidated EBITDA for the most recent four fiscal quarter period ending prior to the date of the closing. This springing financial covenant will be tested on the last day of each fiscal quarter, commencing with the last day of the second full fiscal quarter after the closing date, but only if on such date the revolving loans (excluding, for the first two fiscal quarters following the initial potential testing date, the amount of revolving loans drawn at closing) are outstanding in an aggregate principal amount exceeding 40% of the total revolving commitments under the revolving facility.
The New Credit Facilities will also contain a number of customary negative covenants. Such covenants, among other things, will limit or restrict the ability of each of the borrowers, their restricted subsidiaries, and where applicable, MT HoldingSub2, to, among other things, incur additional indebtedness, incur liens on assets, sell assets and make certain dividends or other payments, make and acquisitions and other investments, subject to certain exceptions. See Description of New Mirion Indebtedness for more information about the negative covenants.
283
Table of Contents
Cash flows
Nine months Ended March 31, 2021 and March 31, 2020
| (Dollars in millions) |
March 31, 2021 |
March 31, 2020 |
$ Change |
% Change |
||||||||||||
| Net cash provided by operating activities |
$ | 30.7 | $ | 17.2 | $ | 13.5 | 78.5 | % | ||||||||
| Net cash used in investing activities |
$ | (307.4 | ) | $ | (65.0 | ) | $ | (242.4 | ) | 372.9 | % | |||||
| Net cash provided by financing activities |
$ | 241.6 | $ | 118.6 | $ | 123.0 | 103.7 | % | ||||||||
| Non-GAAP: | ||||||||||||||||
| (Dollars in millions) |
March 31, 2021 |
March 31, 2020 |
||||||||||||||
| Net cash provided by (used for) operating activities |
$ | 30.7 | $ | 17.2 | ||||||||||||
| Purchases of property, plant, and equipment and badges |
(17.3 | ) | (11.8 | ) | ||||||||||||
|
|
|
|
|
|||||||||||||
| Free cash flow(1) |
$ | 13.4 | $ | 5.4 | ||||||||||||
| Cash used for non-operating expenses |
16.5 | 12.7 | ||||||||||||||
|
|
|
|
|
|||||||||||||
| Adjusted free cash flow(1) |
$ | 29.9 | $ | 18.1 | ||||||||||||
|
|
|
|
|
|||||||||||||
| (1) | Free cash flow and Adjusted free cash flow are supplemental measures of our performance that are not required by, or presented in accordance with, U.S. GAAP. We believe that free cash flow and Adjusted free cash flow are important because they provide management with measurements of cash generated from operations that is available for payment obligations and investment opportunities, such as repaying debt and funding acquisitions. |
Free cash flow is defined as U.S. GAAP net cash provided by operating activities adjusted to include the impact of purchases of property, plant, and equipment and purchases of badges. Adjusted free cash flow is defined as free cash flow adjusted to include the impact of cash used to fund non-operating expenses (as previously defined). We believe that the inclusion of supplementary adjustments to free cash flow applied in presenting Adjusted free cash flow is appropriate to provide additional information to investors about our cash flows that management utilizes on an ongoing basis to assess our ability to generate cash for use in acquisitions and other investing and financing activities.
Free cash flow and Adjusted free cash flow may not be comparable to similarly titled measures used by other companies. You should not consider our Free cash flow or Adjusted free cash flow as alternatives to net cash provided by (used for) operating activities in accordance with U.S. GAAP.
Net Cash Provided by Operating Activities
Net cash provided by operating activities was $30.7 million during the nine months ended March 31, 2021, a $13.5 million, or 78.5%, increase compared to the nine months ended March 31, 2020 primarily due to cash outflow resulting from a decrease in net loss adjusted for non-cash items of approximately $20.9 million offset by cash inflow of $34.4 million from improved working capital.
Net Cash Used in Investing Activities
Net cash used in investing activities was $307.4 million during the nine months ended March 31, 2021 compared to net cash used in investing activities of $65.0 million in the nine months ended March 31, 2020. The $242.4 million, or 372.9%, increase is primarily the result of greater acquisition activity (an increase of $236.9 million) in addition to purchases of property, plant and equipment and badges. Capital expenditures were
284
Table of Contents
$17.3 million and $11.8 million in the nine months ended March 31, 2021 and the nine months ended March 31, 2020, respectively, related to property, plant, and equipment and badges.
Net Cash Provided by Financing Activities
Net cash provided by financing activities was $241.6 million in the nine months ended March 31, 2021 compared to $118.6 million of cash provided in the nine months ended March 31, 2020. During the nine months ended March 31, 2021, Mirion borrowed a net $218.8 million of notes payable under the 2019 Credit Facility and a net $70.0 million of borrowings from related parties, offset by $35.0 million of repayments of borrowings on the revolver term loan and $12.4 million of repayments of principal ($6.4 million under the 2019 Credit Facility and $6.0 million of the NRG loan). During the nine months ended March 31, 2020, Mirion borrowed a net $86.1 million of notes payable under the 2019 Credit Facility and a net $34.9 million of borrowings under the revolver, offset by $2.0 million of contingent consideration payments and $0.4 million of distributions to noncontrolling interests.
Year ended June 30, 2020 compared to Year ended June 30, 2019
| (Dollars in millions) |
2020 | 2019 | $ Change |
% Change |
||||||||||||
| Net cash provided by operating activities |
$ | 39.5 | $ | 14.7 | $ | 24.8 | 168.7 | % | ||||||||
| Net cash used in investing activities |
$ | (75.6 | ) | $ | (25.6 | ) | $ | (50.0 | ) | 195.3 | % | |||||
| Net cash provided by financing activities |
$ | 118.9 | $ | 15.0 | $ | 103.9 | 692.7 | % | ||||||||
| Non-GAAP: | ||||||||||||||||
| (Dollars in millions) |
2020 | 2019 | ||||||||||||||
| Net cash provided by operating activities |
$ | 39.5 | $ | 14.7 | ||||||||||||
| Purchases of property, plant, equipment and badges |
(19.9 | ) | (16.5 | ) | ||||||||||||
|
|
|
|
|
|||||||||||||
| Free cash flow |
$ | 19.6 | $ | (1.8 | ) | |||||||||||
| Cash used for non-operating expenses |
16.4 | 10.3 | ||||||||||||||
|
|
|
|
|
|||||||||||||
| Adjusted free cash flow |
$ | 36.0 | $ | 8.5 | ||||||||||||
|
|
|
|
|
|||||||||||||
Net Cash Provided by Operating Activities
Net cash from operating activities was $39.5 million in FY 2020, a $24.8 million, or 168.7%, increase compared to FY 2019 due primarily to primarily due to cash outflow resulting from a decrease in net loss adjusted for non-cash items of approximately $3.2 million offset by cash inflow of $28.0 million from improved working capital.
Net Cash Used in Investing Activities
Net cash used in investing activities was $75.6 million in FY 2020 compared to net cash used in investing activities of $25.6 million in FY 2019. The $50.0 million, or 195.3%, increase is primarily the result of greater acquisition activity (an increase of $46.6 million) in addition to purchases of property, plant and equipment and badges. Capital expenditures were $19.9 million and $16.5 million in FY 2020 and FY 2019, respectively, related to property, plant, and equipment.
Net Cash Provided by Financing Activities
Net cash provided by financing activities was $118.9 million in FY 2020 compared to net cash provided by financing activities of $15.0 million in FY 2019. During FY 2020, Mirion borrowed $98.8 million of notes
285
Table of Contents
payable (primarily under the 2019 Credit Facility) and $80.0 million of borrowings under the revolver, offset by $13.4 million principal repayments of notes payable, $45.0 million repayments of borrowings on the revolver term loan, $2.0 million of contingent consideration payments, and $0.4 million of distributions to noncontrolling interests. During FY 2019, net cash provided by financing activities was driven primarily by net borrowings due to the refinancing of debt and issuance of notes payable under the 2019 Credit Facility of $28.5 million offset by repayments of $13.0 million for the revolver.
Year ended June 30, 2019 compared to Year ended June 30, 2018
| (Dollars in millions) |
2019 | 2018 | $ Change |
% Change |
||||||||||||
| Net cash provided by operating activities |
$ | 14.7 | $ | 15.6 | $ | (0.9 | ) | (5.8 | )% | |||||||
| Net cash used in investing activities |
$ | (25.6 | ) | $ | (12.6 | ) | $ | (13.0 | ) | 103.2 | % | |||||
| Net cash provided by (used in) financing activities |
$ | 15.0 | $ | (5.5 | ) | $ | 20.5 | (372.7 | )% | |||||||
| Non-GAAP: |
||||||||||||||||
| (Dollars in millions) |
2019 | 2018 | ||||||||||||||
| Net cash provided by operating activities |
$ | 14.7 | $ | 15.6 | ||||||||||||
| Purchases of property, plant, equipment and badges |
(16.5 | ) | (12.6 | ) | ||||||||||||
|
|
|
|
|
|||||||||||||
| Free cash flow |
$ | (1.8 | ) | $ | 3.0 | |||||||||||
| Cash used for non-operating expenses |
10.3 | 30.7 | ||||||||||||||
|
|
|
|
|
|||||||||||||
| Adjusted free cash flow |
$ | 8.5 | $ | 33.7 | ||||||||||||
|
|
|
|
|
|||||||||||||
Net Cash Provided by Operating Activities
Net cash provided by operating activities of $14.7 million in FY 2019, a decrease of $0.9 million, or 5.8%, compared to FY 2018 due to net loss adjusted for non-cash items of approximately $24.6 million cash inflow offset by a cash outflow of $25.5 million from changes in working capital.
Net Cash Used for Investing Activities
Net cash used for investing activities was $25.6 million in FY 2019 compared to $12.6 million utilized by investing activities in FY 2018, which represented an increase of $13.0 million, or 103.2%. The increase is primarily the result of greater acquisition activity (an increase of $9.1 million) in addition to purchases of property, plant and equipment and badges. Capital expenditures were $16.5 million and $12.6 million in FY 2019 and FY 2018, respectively, related to property, plant, and equipment.
Net Cash Provided by (Used in) Financing Activities
Net cash provided by financing activities was $15.0 million in FY 2019 compared to net cash used in financing activities of $5.5 million in FY 2018. During FY 2019, net cash provided by financing activities was driven primarily by net borrowings due to the refinancing of debt and issuance of notes payable under the 2019 credit facility of $28.5 million offset by repayments of $13.0 million for the revolver. During FY 2018, net cash used in financing activities was driven primarily by principal repayments of $4.9 million.
Off-Balance Sheet Arrangements
Mirion does not have any off-balance sheet arrangements for any of the periods presented.
286
Table of Contents
Critical Accounting Policies and Estimates
Mirions consolidated financial statements are prepared in accordance with U.S. GAAP. The preparation of these financial statements requires management to make estimates and assumptions that affect the reported amounts of assets, liabilities, revenues, costs and expenses and related disclosures. Such estimates are based on historical experience and on various other factors that management believes are reasonable under the circumstances, the results of which form the basis for making judgments about the carrying value of assets and liabilities that are not readily apparent from other sources. Actual results may differ from these judgments and estimates under different assumptions or conditions and any such differences may be material. Mirion believes that the accounting policies discussed below are critical to understanding historical and future performance, as these policies relate to the more significant areas involving managements judgments and estimates.
Business combinations
We account for business acquisitions in accordance with the Financial Accounting Standards Board (FASB) Accounting Standards Codification (ASC) 805, Business Combinations. This standard requires the acquiring entity in a business combination to recognize all the assets acquired and liabilities assumed in the transaction and establishes the acquisition date fair value as the measurement objective for all assets acquired and liabilities assumed in a business combination. Certain provisions of this standard prescribe, among other things, the determination of acquisition date fair value of consideration paid in a business combination (including contingent consideration) and the exclusion of transaction and acquisition-related restructuring costs from acquisition accounting.
The determination of the fair value of assets acquired and liabilities assumed involves assessments of factors such as the expected future cash flows associated with individual assets and liabilities and appropriate discount rates at the closing date of the acquisition. For non-observable market values, the Company determines fair value using acceptable valuation principles (e.g., multiple excess earnings, relief from royalty and cost methods).
Goodwill
Goodwill represents the excess of the purchase price paid over the estimated fair value of the net assets acquired and liabilities assumed in the acquisition of a business.
Goodwill has an indefinite useful life, and is not amortized, but instead tested for impairment annually during the fiscal year fourth quarter or more often if events or changes in circumstances indicate that the carrying amount may exceed fair value as set forth in ASC 350, Intangibles Goodwill and Other. The Company tests for goodwill impairment at the reporting unit level, which is an operating segment or one level below an operating segment. The amount of goodwill acquired in a business combination that is assigned to one or more reporting units as of the acquisition date is the excess of the purchase price of the acquired businesses (or portion thereof) included in the reporting unit, over the fair value assigned to the individual assets acquired or liabilities assumed from a market participant perspective. Goodwill is assigned to the reporting unit(s) expected to benefit from the synergies of the combination even though other assets or liabilities of the acquired entity may not be assigned to that reporting unit.
ASC 350 allows an optional qualitative assessment as part of annual impairment testing, prior to a quantitative assessment test, to determine whether it is more likely than not that the fair value of a reporting unit exceeds its carrying amount. In performing the quantitative test, which compares the fair value of a reporting unit with its carrying amount, we determine the fair value of each reporting unit by estimating the present value of expected future cash flows, discounted by the applicable discount rate, and peer company multiples. If the carrying value exceeds the fair value, the Company recognizes an impairment loss in the amount equal to the excess, not to exceed the total amount of goodwill allocated to that reporting unit.
Based upon our review and analysis, each reporting unit had significant headroom and no impairments were deemed to have occurred during any of the years presented.
287
Table of Contents
Intangible Assets
Intangible assets relate to the value associated with our developed technology, customer relationships, backlog, trade names and non-compete agreements at the time of acquisition through business combinations. Definite lived intangible assets are amortized over their estimated useful lives, ranging from 1 to 16 years.
Revenue Recognition
Prior to July 1, 2019, the Company recognized revenue based on ASC 605, Revenue Recognition, when there was persuasive evidence of an arrangement, product delivery had occurred or services had been provided, the sales price was fixed or determinable and collectability was reasonably assured. Beginning July 1, 2019, the Company recognizes revenue based on ASC 606, Revenue from Contracts with Customers as performance obligations are satisfied by transferring control of promised goods or service to customers in an amount that reflects the consideration to which the Company expects to be entitled in exchange for those goods or services. See Recently Adopted Accounting Guidance below for a discussion of the change in revenue recognition accounting that became effective on July 1, 2019.
ASC 606
Performance Obligations
The Company identifies a performance obligation for each promise in a contract to transfer a distinct good or service to the customer. As part of its assessment, the Company considers all goods and/or services promised in the contract, regardless of whether they are explicitly stated or implied by customary business practices. The Companys contracts may contain either a single performance obligation, including the promise to transfer individual goods or services that are not separately distinct within the context of the respective contracts, or multiple performance obligations. For contracts that contain multiple performance obligations, the Company allocates the consideration to which it expects to be entitled to each performance obligation based on relative standalone selling prices and recognizes the related revenue when or as control of each individual performance obligation is transferred to customers. Service revenues (warranty contracts, post contract support, and subscription-based services) are recognized over time as the customers receive and consume benefits of such services simultaneously.
The Company exercises judgment in determining the timing of revenue by analyzing the point in time or the period over which the customer has the ability to direct the use of and obtain substantially all of the remaining benefits of the performance obligation. Typically, over-time revenue recognition is based on the utilization of an input measure used to measure progress, such as costs incurred to date relative to total estimated costs. Changes in total estimated costs are recognized using the cumulative catch-up method of accounting which recognize the cumulative effect of the changes on current and prior periods in the current period. Accordingly, the effect of the changes on future periods of contract performance is recognized as if the revised estimate had been the original estimate. A significant change in an estimate on one or more contracts could have a material effect on the Companys consolidated financial position, results from operations, or cash flows. However, there were no significant changes in estimated contract costs for the year ended June 30, 2020.
If a performance obligation does not qualify for over-time revenue recognition, revenue is then recognized at the point-in-time in which control of the distinct good or service is transferred to the customer, typically based upon the terms of delivery.
Certain of the Companys products are sold through distributors and third-party sales representatives under standard agreements whereby distributors purchase products from the Company and resell them to customers. These agreements give distributors the right to sell the Companys products within certain territories and establish minimum order requirements. These arrangements do not provide stock rotation or price protection rights and do not contain extended payment terms. Rights of return are limited to repair or replacement of
288
Table of Contents
delivered products that are defective or fail to meet the Companys published specifications. Provisions for these warranty costs are recognized in the same period that the related revenue is recorded.
The remaining performance obligation for open contracts as of June 30, 2020 include assembly, delivery, installation and training. Payment terms for shipments to end-users are generally net 30 days. Payment terms for distributor shipments may range from 30 to 90 days. Service arrangements can call for payments in advance of performing the work (e.g., extended warranty and service contracts), upon completion of contract milestones (e.g., custom development manufacturing), or a combination of each.
ASC 605
Prior to July 1, 2019, the Company recognized revenue from sales contracts when there was persuasive evidence of an arrangement, product delivery had occurred or services had been provided, the sales price was fixed or determinable and collectability was reasonably assured. For sales contracts that contain customer-specific acceptance provisions, revenue and the related costs were deferred until the customer had indicated successful completion of site acceptance tests or the Company had otherwise determined that all customer-specific acceptance criteria had been met. Where the Company performed detailed factory acceptance testing on completed products, which, in some instances, was sufficiently extensive and reliable to demonstrate that its products meet the customer-specified objective acceptance criteria set forth in the related sales arrangements. In such instances, the Company recognized revenue based on delivery terms and prior to the receipt of notification of formal acceptance from the customer.
The Company combined a group of contracts as one project if they are closely related and were in substance, part of a single project with an overall project margin. The Company segmented a contract into several projects when they were of different business substance, for example, with different business negotiation, solutions, implementation plans, and margins.
The Company evaluated each deliverable in an arrangement to determine whether they represented separate units of accounting. A deliverable constituted a separate unit of accounting when it had stand-alone value, and for an arrangement that included a general right of return relative to the delivered products or services, when delivery or performance of the undelivered product or service was considered probable and is substantially controlled by the Company. The Company considers a deliverable to have stand-alone value if the product or service is sold separately by the Company or another vendor or could be resold by the customer on a stand-alone basis at an amount that would substantially recover the original purchase price. Further, the revenue arrangements generally do not include a general right of return relative to the delivered products. Where the aforementioned criteria for a separate unit of accounting are not met, the deliverable is combined with the undelivered element(s) and treated as a single unit of accounting for the purposes of allocation of the arrangement consideration and revenue recognition.
When a sales arrangement contains multiple units of accounting, the Company allocates the total arrangement consideration to each separable element of an arrangement based upon the relative selling price of each element. Allocation of the consideration is determined at arrangement inception based on each units relative selling price, which is determined based on a selling price hierarchy. The relative selling price for a deliverable is based on its vendor-specific objective evidence (VSOE) if available, third-party evidence (TPE) if VSOE is not available or estimated selling price if neither VSOE nor TPE is available. The Company then recognizes revenue on each deliverable in accordance with its policies for product and service revenue recognition. The Company is not typically able to determine VSOE or TPE and therefore uses estimated selling prices to allocate revenue between the elements of the arrangement. The Company establishes its best estimate of the selling price considering multiple factors, including, but not limited to, pricing practices in different geographies and through different sales channels, costs and margin objectives, competitive pricing strategies and general market conditions.
289
Table of Contents
The Company limits the amount of revenue recognition for delivered elements to the amount that is not contingent on the future delivery of products or services or future performance obligations or subject to customer-specific cancellation rights.
For all arrangements, amounts billed to a customer related to shipping and handling are classified as revenue while all costs incurred by the Company for shipping and handling are classified as cost of revenue. Provisions and allowances for discounts to customers, estimated sales returns, service cancellations, and other adjustments are provided for in the same period that the related revenue is recorded.
Certain of the Companys products are sold through distributors and third-party sales representatives under standard agreements whereby distributors purchase products from the Company and resell them to customers. These agreements give distributors the right to sell the Companys products within certain territories and establish minimum order requirements. These arrangements do not provide stock rotation or price protection rights and do not contain extended payment terms. Rights of return are limited to repair or replacement of delivered products that are defective or fail to meet the Companys published specifications. Provisions for these warranty costs are recognized in the same period that the related revenue is recorded.
Revenue from certain fixed-price contracts that involve customization of equipment to customer specifications is recorded using a percentage-of-completion method measured on the cost-to-cost basis. Contract costs include all direct materials and labor costs, as well as indirect costs related to contract performance. Changes in job performance, job conditions, and estimated profitability result in revisions to costs and revenue and are recognized in the period in which the revisions are determined. Provisions for estimated losses on uncompleted contracts are made in the period in which such losses are first determined. Revenue earned in excess of billings on contracts in progress is classified in the consolidated balance sheet as a current asset and included in costs in excess of billings on uncompleted contracts. Amounts billed in excess of revenue earned are classified as a current liability and included in deferred contract revenue.
Revenue derived from passive dosimetry and analytical services is of a subscription nature, with passive dosimetry and analytical services provided to customers on an agreed-upon recurring monthly, quarterly, or annual basis. Services are provided to the customer through passive dosimeter badges that the Company supplies to customer personnel. Depending on the type of badge utilized, either customers return the used badges to the Company for analysis, or they obtain the analysis directly through a self-service web portal. The Company believes that badge production, badge wearing, badge analysis and report preparation are all integral to the benefit that the Company provides to its customers and, therefore, the service period is defined as the period over which all of these services are provided. Revenue is recognized on a straight-line basis over the service period as the service is continuous, and no other discernible pattern of recognition is evident. Many customers pay for these measuring and monitoring services in advance. The amounts are recorded as deferred contract revenue in the consolidated balance sheets and represent customer deposits invoiced in advance for services to be rendered over the service period, net of a reserve for estimated cancellations.
Pertinent to ASC 606 and 605
The Company sells its products and services mainly to large, private, and governmental organizations in the Americas, Europe, the Middle East, and Asia Pacific regions. The Company performs ongoing evaluations of its customers financial condition and limits the amount of credit extended when deemed necessary. The Company generally does not require its customers to provide collateral or other security to support accounts receivable.
Accounting for Income Taxes
The Company accounts for income taxes and the related accounts under the asset and liability method. Deferred tax assets and liabilities are recognized for the expected tax consequences of temporary differences between the tax bases of assets and liabilities and their reported amounts. Valuation allowances are recorded to reduce
290
Table of Contents
deferred tax assets to the amount that will more likely than not be realized. The Company classifies all deferred tax assets and liabilities, and any related valuation allowance, as non-current in the consolidated balance sheet.
The Company accounts for uncertainty in income taxes using a two-step approach to recognizing and measuring uncertain tax positions. The first step is to evaluate the tax position for recognition by determining if the weight of available evidence indicates that it is more likely than not that the position will be sustained on audit, including resolution of related appeals or litigation processes, if any. The second step is to measure the tax benefit as the largest amount that is more than 50% likely of being realized upon settlement. The Company classifies the liability for unrecognized tax benefits as current in the balance sheet, to the extent that the Company anticipates payment or receipt of cash within one year. Interest and penalties related to uncertain tax positions are recognized in the provision for income taxes.
Foreign Currency Translation
Local currency is the functional currency for substantially all of the Companys foreign operations. Assets and liabilities of foreign operations are translated into U.S. dollars using the exchange rates in effect at the balance sheet reporting date, while income and expenses are translated at the average monthly exchange rates during the period. We record gains and losses from the translation of financial statements in foreign currencies into U.S. dollars in other comprehensive income. The income tax effect of currency translation adjustments related to foreign subsidiaries that are not considered indefinitely reinvested is recorded as a component of deferred taxes with an offset to other comprehensive income. We record gains and losses from changes in exchange rates on transactions denominated in currencies other than each reporting locations functional currency in the consolidated statements of operations for each period.
Litigation
The Company is subject to various legal proceedings, claims, litigation, investigations, and contingencies arising out of the ordinary course of business. While the ultimate results of such suits or other proceedings against us cannot be predicted with certainty, we believe the resolution of these matters will not have a material effect on our results of operations, financial condition, or cash flows. If we believe the likelihood of an adverse legal outcome is probable and the amount is reasonably estimable, we accrue a liability in accordance with accounting guidance for contingencies. We consult with legal counsel on matters related to litigation and seek input both within and outside the Company.
Quantitative and Qualitative Disclosures about Market Risk
Market risk
The market risk inherent in financial statements represents the potential loss in fair value, earnings or cash flows arising from adverse changes in foreign currency exchange rates, commodity prices or interest rates. Mirion may use derivative financial instruments like interest rate swaps to manage exposure to market risks. Mirion does not use derivative financial instruments for trading purposes.
Foreign currency exchange rate risk
In the normal course of business, Mirion is exposed to changes in foreign currency exchange rates due to its worldwide presence and business profile. Foreign currency exposures relate to transactions denominated in currencies that differ from the function currencies of our subsidiaries.
We derived approximately 61% of our revenues during FY 2020 from outside the United States through international operations, some of which were transacted in U.S. dollars. In addition, certain of our domestic operations have sales to foreign customers. Although we are impacted by the exchange rates of several
291
Table of Contents
currencies, our largest exposures are generally to the Euro, Canadian dollar, British Pound, and Japanese Yen. In conducting our foreign operations, we also make inter-company sales denominated in different currencies. These activities expose us to the effect of changes in foreign currency exchange rates. Flows of foreign currencies into and out of our operations are generally stable, regularly occurring and are recorded at fair market value in our financial statements.
During FY 2020 and FY 2019, the effect of a hypothetical 10% change in foreign currencies that we have exposure to compared to the U.S. dollar would have impacted our revenues by approximately $31.6 million and $30.0 million, respectively.
During FY 2020 and FY 2019, the effect of a hypothetical 1% change in exchange rates would have impacted accumulated other comprehensive income by approximately $5.1 million and $6.1 million, respectively. This impact does not consider the effects of a stronger or weaker dollar on our ability to compete for export business or the overall economic activity that could exist in such an environment. Changes in foreign exchange rates could impact the price and the demand for our products such as a strengthening dollar causes exports to become more expensive to foreign customers and businesses that must pay for them in other currencies.
Interest rates
We are exposed to changes in interest rates primarily as a result of our long-term debt. We may from time to time use interest rate swap agreements to manage the interest rate characteristics of a portion of our outstanding debt. However, as of March 31, 2021, we did not have any active interest rate swap agreements. In March 2020, we executed an interest rate cap agreement effective September 30, 2020 through March 31, 2022 for a 2% LIBOR interest rate cap on $542 million notional value. There is no value for this agreement as of March 31, 2021. Based on the amounts and mix of our floating rate debt at March 31, 2021, if market interest rates increase an average of 100 basis points, our year-to-date interest expense would increase by approximately $5.9 million. We determined these amounts by considering the impact of the hypothetical interest rates on our borrowing costs. This analysis does not consider the effects of changes in the level of overall economic activity that could exist in such an environment.
292
Table of Contents
DESCRIPTION OF NEW MIRION INDEBTEDNESS
Debt Financing
Debt Commitment Letter
In connection with the Business Combination Agreement, MT HoldingSub2 entered into a Commitment Letter pursuant to which GS Lending and Citi (together with those other financial institutions that may become party to the Debt Commitment Letter) have committed to provide to Mirion Technologies, Inc., as the subsidiary borrower, and a parent entity of the Mirion TopCo business to be formed (which will be an indirect subsidiary of Mirion TopCo), senior secured credit facilities to be used to, among other things, (i) fund the Business Combination, (ii) repay and terminate the existing indebtedness of Mirion Technologies, Inc. and Mirion Technologies (Luxembourg) S.A R.L (the debt refinancing) and (iii) pay all fees, premiums, expenses and other transaction costs incurred in connection with the foregoing. The aggregate commitment of the senior secured credit facilities consists of a $830 million dollar-denominated first lien term facility and a $90 million revolving facility.
The obligations of the lenders to provide debt financing under the Debt Commitment Letter are subject to a number of customary conditions, including (1) the execution and delivery of definitive documentation consistent with the terms of the Debt Commitment Letter; (2) the simultaneous or substantially concurrent completion of the Business Combination in accordance in all material respects with the Business Combination Agreement (without giving effect to any amendment, waiver, consent or other modification thereof that is materially adverse to the interests of the lenders (in their capacities as such) unless it is approved by the lenders); (3) since the date of the Business Combination Agreement, there shall not having occurred a Material Adverse Effect (as defined in the Business Combination Agreement); (4) the completion of the refinancing of the Existing Credit Facility, prior to, or substantially concurrently with, the initial funding of the debt financing; (5) delivery of certain audited, unaudited and pro forma financial statements; (6) payment of all applicable fees and expenses; (7) the receipt of documentation and other information about the borrowers and guarantors thereto required by regulatory authorities under applicable know your customer, beneficial ownership and anti-money laundering rules and regulations (including the PATRIOT Act); (8) the execution and delivery of guarantees by guarantors and the taking of certain actions necessary to establish and perfect a security interest in specified items of collateral; (9) the accuracy in all material respects of specified representations and warranties in the loan documents under which the debt financing will be provided and of certain representations and warranties in the Business Combination Agreement and (10) the delivery of certain customary closing documents.
The obligations of the lenders to provide the debt financing under the Debt Commitment Letter will terminate at the earliest of (1) the closing date of the New Credit Facilities; (2) April 7, 2022; (3) the termination of the Business Combination Agreement without the consummation of the Business Combination having occurred; and (4) the consummation of the Business Combination without the use of the debt financing.
As of the last practicable date before the printing of this proxy statement/prospectus, the Debt Commitment Letter remains in effect. The documentation governing the debt financing contemplated by the Debt Commitment Letter has not been finalized and, accordingly, the actual terms of the debt financing may differ from those described in this proxy statement/prospectus.
The New Credit Facilities
The first lien term facility and revolving facility will accrue interest at a rate of LIBOR plus an applicable margin to be determined with respect to the term loans and 3.00% with respect to the revolving credit facility. The revolving credit facility requires the payment of a commitment fee of 0.50% per annum for unused revolving commitments. The term loan facility will mature on the seventh anniversary of the closing date. The revolving facility will mature on the fifth anniversary of the date of the closing. The definitive documentation for the New Credit Facilities has not been finalized and, accordingly, the actual terms of the debt financing, including the interest rate, may differ from those described in this proxy statement/prospectus
293
Table of Contents
The revolving facility will include borrowing capacity available for letters of credit of up to $50 million. Any issuance of letters of credit will reduce the amount available under the revolving facility.
The obligations of the borrowers under the New Credit Facility and certain of their (and their subsidiaries) respective obligations under hedging arrangements and cash management arrangements will be unconditionally guaranteed by MT HoldingSub2 and each existing and subsequently acquired or organized direct or indirect wholly-owned US restricted subsidiary of the borrowers (collectively, the Guarantors), in each case, other than certain excluded subsidiaries and subject to other customary limitations to be set forth in the definitive documentation with respect to the debt financing. The debt financing will be secured by a security interest in substantially all of the assets of the borrowers and the Guarantors (and including a pledge of the equity interests of each borrower and of each Guarantor and of their respective direct, wholly-owned restricted subsidiaries), in each case subject to customary exceptions (including exceptions for certain adverse tax consequences).
The revolving facility will require MT HoldingSub2 to maintain a maximum first lien net leverage ratio (to be defined in the definitive documentation with respect to the debt financing) at a level equal to a single first lien net leverage ratio level calculated based on providing a 40% cushion to consolidated EBITDA for the most recent four fiscal quarter period ending prior to the date of the closing. This springing financial covenant will be tested on the last day of each fiscal quarter, commencing with the last day of the second full fiscal quarter after the closing date, but only if on such date the revolving loans (excluding, for the first two fiscal quarters following the initial potential testing date, the amount of revolving loans drawn at closing) are outstanding in an aggregate principal amount exceeding 40% of the total revolving commitments under the revolving facility.
The senior secured credit facilities will also contain a number of customary negative covenants. Such covenants, among other things, will limit or restrict the ability of each of the borrowers, their restricted subsidiaries, and where applicable, the direct parent holding company of the borrowers, to, among other things:
| | incur additional indebtedness and make guarantees; |
| | incur liens on assets; |
| | engage in mergers or consolidations or fundamental changes; |
| | sell assets; |
| | pay dividends and distributions or repurchase capital stock; |
| | make investments, loans and advances, including acquisitions; |
| | enter into certain agreements that would restrict the ability to grant liens; |
| | repay certain junior indebtedness; and |
| | in the case of MT HoldingSub2, engage in activities other than passively holding the equity interests in the borrowers. |
The aforementioned restrictions will be subject to certain exceptions and thresholds including (i) the ability to incur additional indebtedness and liens, make investments, dividends and distributions, asset sales and prepayments of junior indebtedness subject, in each case, to compliance with certain financial metrics, thresholds and certain other conditions and (ii) a number of other traditional exceptions that grant the borrowers continued flexibility to operate and develop their businesses. The senior secured credit facilities will also contain certain customary representations and warranties, affirmative covenants and events of default.
The documentation governing the debt financing has not been finalized and, accordingly, the actual terms of the debt financing may differ from that described in this proxy statement/prospectus.
294
Table of Contents
This section discusses the material components of the executive compensation program for Mirion executive officers who would be Mirions named executive officers if Mirion was subject to the reporting requirements under the Securities Exchange Act of 1934. We expect that at least some of these executive officers will be named executive officers of the combined business after the closing of the Business Combination. In 2021, Mirions named executive officers and their positions were as follows:
| | Thomas Logan, Chairman and Chief Executive Officer; |
| | Michael Freed, Chief Operating Officer; and |
| | Brian Schopfer, Chief Financial Officer. |
This discussion may contain forward-looking statements that are based on Mirions current plans, considerations, expectations, and determinations regarding future compensation programs. Actual compensation programs that New Mirion adopts following the completion of the Business Combination may differ materially from the currently planned programs summarized in this discussion.
The Company is, and after the Business Combination, New Mirion will be, an emerging growth company and therefore is subject to reduced disclosure obligations regarding executive compensation and exemptions from the requirements of holding a nonbinding advisory vote on executive compensation and stockholder approval of any golden parachute payments not previously approved.
Summary Compensation Table
The following table presents all of the compensation awarded to or earned by or paid to Mirions named executive officers for the year ended June 30, 2021.
| Name and |
Year | Salary ($)(1) |
Bonus ($) |
Stock Awards(2) ($) |
Option Awards ($) |
Non-Equity Incentive Plan Compensation(3) ($) |
All Other Compensation ($)(4) |
Total ($) |
||||||||||||||||||||||||
| Thomas Logan Chairman and Chief Executive Officer |
2021 | 639,262 | | 19,240,000 | | | 72,025 | 19,951,287 | ||||||||||||||||||||||||
| Michael Freed Chief Operating Officer |
2021 | 398,851 | | | | | 12,167 | 411,018 | ||||||||||||||||||||||||
| Brian Schopfer Chief Financial Officer |
2021 | 355,227 | | 4,208,750 | | | 15,964 | 4,579,941 | ||||||||||||||||||||||||
| (1) | Amounts reflect the base salary in effect for each named executive officer during fiscal 2021. For additional information, see Base Salaries and 2021 Bonuses below. |
| (2) | Reflects the grant date value of a one-time grant of profits interests in the Sponsor, which was approved and granted by the Sponsor in recognition of Messrs. Logan and Schopfers efforts in connection with the Business Combination. As discussed below under Profits Interests, the Sponsor granted Messrs. Logan and Schopfer the award of profits interests on June 16, 2021 in connection with the signing of the Business Combination Agreement. The profits interests award provides for service and performance-vesting, with the award only vesting upon the achievement of specified share price conditions. The grant date fair value of the profits interests is based upon a valuation model using Monte Carlo simulations in accordance with FASB Accounting Standards Codification (ASC) Topic 718. |
295
Table of Contents
| (3) | Amounts payable pursuant to the Companys annual performance bonus program for the 2021 fiscal year have not yet been determined, but are expected to be determined in September 2021 and will be set forth in a subsequent filing. For additional information on these payments, see 2021 Bonuses below. |
| (4) | Amounts reflect: (i) for Mr. Logan, a $21,312 cash payment in respect of accrued vacation days, a $14,400 automobile allowance, a company contribution of $12,495 to Mr. Logans account under Mirions 401(k) plan, a $5,000 reimbursement for financial planning services, $3,500 to cover the costs of an annual physical examination, $13,135 in stipends paid to Mr. Logan for time spent flying his personal aircraft to business events plus corresponding reimbursement for fuel costs associated with such flights, $1,200 for continued automobile maintenance and $983 in Company-paid long-term care insurance premiums; (ii) for Mr. Freed, a company contribution of $11,454 to Mr. Freeds account under Mirions 401(k) plan and $713 in Company-paid long-term care insurance premiums; and (iii) for Mr. Schopfer, company contributions of $10,848 to Mr. Schopfers account under Mirions 401(k) plan, $2,500 to cover the costs of an annual physical examination, a $1,500 reimbursement for financial planning services, $500 to Mr. Schopfers Mirion-sponsored health savings account and $616 in Company-paid long-term care insurance premiums. |
Elements of Mirions Executive Compensation Program
For the year ended June 30, 2021, the compensation for each named executive officer generally consisted of a base salary, performance-based bonus and standard employee benefits. These elements (and the amounts of compensation and benefits under each element) were selected because Mirion believes they are necessary to help attract and retain executive talent which is fundamental to its success. Below is more detailed summary of the current executive compensation program as it relates to Mirions named executive officers.
Base Salaries
The named executive officers receive a base salary to compensate them for services rendered to Mirion. The base salary payable to each named executive officer is intended to provide a fixed component of compensation reflecting the executives skill set, experience, role, and responsibilities. Each named executive officers initial base salary was provided in his employment agreement, and the base salaries of Mirion executives are reviewed and, if appropriate, adjusted on an annual basis.
The actual base salaries paid to each named executive officer for Mirions fiscal year 2021 are set forth above in the Summary Compensation Table in the column entitled Salary.
2021 Bonuses
Mirion maintains a cash-based annual bonus program for its executives, including the named executive officers, in which such executives are eligible to receive bonuses based on performance goals. Such awards are designed to incentivize the named executive officers with a variable level of compensation that is based on performance measures established by Mirions remuneration committee (and, for officers other than Mr. Logan, by Mr. Logan) that are tied to pre-defined business and personal goals and objectives.
In Mirions fiscal year 2021, Messrs. Logan, Freed and Schopfer were eligible to earn annual cash bonuses targeted at 80%, 50% and 50%, respectively, of their base salaries. Each named executive officer was eligible to earn his bonus based on the attainment of business unit and personal goals and objectives, set and approved by Mr. Logan and by the Remuneration Committee of Mirion (Mr. Logans target bonus was set and approved by the Remuneration Committee).
Profits Interests
On June 16, 2021 and in connection with the Business Combination, the Sponsor agreed to issue 3,200,000 membership interests to Thomas Logan and 700,000 membership interests to Brian Schopfer (collectively, the Profits Interests), pursuant to which Messrs. Logan and Schopfer will have an indirect interest in the founder
296
Table of Contents
shares held by the Sponsor. The Profits Interests are subject to service and performance vesting conditions, including the occurrence of the Closing, and do not fully vest until all of the applicable conditions are satisfied, including the achievement of specified share price conditions. The grant of the Profits Interests is intended to be a one-time grant by the Sponsor in recognition of Messrs. Logan and Schopfers efforts in connection with the Business Combination. In addition, the Profits Interests are subject to certain forfeiture conditions. The vesting terms and other terms and conditions of the Profits Interests are described more fully in the section entitled Certain Relationships and Related Person TransactionsMirions Related Person TransactionsProfits Interests.
Equity Compensation
None of the named executive officers, nor any other service providers of the Company, currently hold any equity or equity-based incentive awards relating to shares of the Companys stock.
2021 Omnibus Incentive Plan
New Mirion has adopted and is seeking stockholder approval of the 2021 Omnibus Incentive Plan (the Incentive Plan). We expect that, if stockholders approve the Incentive Plan and the Incentive Plan becomes effective, awards will be made following the closing of the Business Combination.
The aggregate number of shares of Class A common stock of New Mirion reserved for issuance pursuant to awards under the Incentive Plan is equal to 10% of the Class A common stock outstanding, including shares authorized under the Incentive Plan. The total number of shares of Class A common stock available for issuance under the Incentive Plan shall be increased on the first day of each fiscal year following the date on which the Incentive Plan is adopted in an amount equal to the least of (i) three percent (3%) of the outstanding shares of Class A common stock on the last day of the immediately preceding fiscal year, (ii) five percent (5%) of the shares of Class A common stock immediately following the closing of the Business Combination and (iii) such number of shares of Class A common stock as determined by the Committee (as defined and designated under the terms of the Incentive Plan) in its discretion. Any employee, director or consultant of New Mirion or any of its subsidiaries or affiliates is eligible to receive an award under the Incentive Plan, to the extent that an offer of such award is permitted by applicable law, stock market or exchange rules, and regulations or accounting or tax rules and regulations.
The Incentive Plan provides for the grant of stock options (including incentive stock options and non-qualified stock options), stock appreciation rights, restricted stock, restricted stock units, performance-based awards, other stock-based awards, or any combination thereof. No determination has been made as to the types or amounts of awards that will be granted to specific individuals under the Incentive Plan. Each award will be set forth in a separate grant notice or agreement and will indicate the type and terms and conditions of the award. Please see the section entitled Proposal No.6 Summary of The Incentive Plan Approval of the Incentive Plan Including the Authorization of the Initial Share Reserve Under the Incentive Plan for a summary of the material terms of the Incentive Plan.
Exit Bonuses
Each of the named executive officers is a party to a letter with Mirion Technologies (Global) Ltd., pursuant to which they are entitled to cash bonuses (the Exit Bonuses) in the event of an Exit (as such term is defined below), subject to the applicable named executive officer remaining actively employed with Mirion in good standing through the date of such Exit. The amount of each named executive officers Exit Bonus will be calculated (i) for Messrs. Logan and Freed, as the product of (x) the number of Class A ordinary shares of Mirion that were subscribed for or acquired by Mr. Logan or Mr. Freed, as applicable, at a price per share equal to $9.65 and that Mr. Logan or Mr. Freed, as applicable, holds immediately prior to the consummation of the applicable Exit event, multiplied by (y) $8.65 and (ii) for Mr. Schopfer, as the product of (x) the number of Class A ordinary shares of Mirion that were subscribed for or acquired by Mr. Schopfer at a price per share equal to $9.99 and that Mr. Schopfer holds immediately prior to the consummation of the applicable Exit event, multiplied by (y) $8.99. For purposes of the Exit Bonuses, Exit means the transfer of shares (whether through a single transaction or a
297
Table of Contents
series of transactions) as a result of which any person, or persons connected (as defined in Section 252 of the U.K. Companies Act) or acting in concert (as defined in the City Code on Takeovers and Mergers) with such person, holds more than 50% of the Class A and Class B ordinary shares of Mirion. The consummation of the Business Combination will constitute an Exit, and the Exit Bonuses will vest and become immediately payable in connection therewith.
Other Elements of Compensation
Retirement Plans
Mirion maintains a 401(k) retirement savings plan for its employees in the United States, including the named executive officers, who satisfy certain eligibility requirements. The named executive officers are eligible to participate in the 401(k) plan on the same terms as other full-time employees, including matching contributions which at the beginning of fiscal year 2021 were equal to 100% of a participating employees contribution up to the first 1% of the employees eligible compensation and 50% of the employees contribution up to the next 5% of the employees eligible compensation. In 2021, the Company amended its matching contribution under the 401(k) plan such that matching contributions are currently equal to 100% of a participating employees contribution up to the first 2% of the employees eligible compensation and 50% of the employees contribution up to the next 4% of the employees eligible compensation.
Employee Benefits and Perquisites
All of Mirions full-time employees in the United States, including the named executive officers, are eligible to participate in health and welfare plans, including medical, dental and vision benefits, medical and dependent care flexible spending accounts, short-term and long-term disability insurance and life insurance.
Additionally, each of the named executive officers is entitled to company-paid premiums for long-term care insurance. Pursuant to his employment agreement, Mr. Logan is entitled to reimbursement for certain incurred air travel and automobile expenses, as described further below. In addition to the benefits set forth in his employment agreement, Mr. Logan also receives a $100 per month allowance for automobile maintenance and, in the event that Mr. Logan flies his personal aircraft to or from business-related events, the Company pays Mr. Logan a stipend of $100 per flying hour and reimburses Mr. Logan for the cost of fuel associated with such trips. Messrs. Logan and Freed are each also entitled to reimbursement for the costs of an annual physical examination and financial planning services. Mr. Schopfer is entitled to an annual allowance for costs relating to certain personal financial or tax advisory services and reimbursement for the cost of an annual physical examination, in each case, pursuant to provisions in his employment agreement, as described further below.
Executive Compensation Arrangements
Executive Employment Agreements
Logan Employment Agreement
On August 15, 2006, Mirion entered into an employment agreement with Mr. Logan, which was subsequently amended on December 22, 2008, January 1, 2009, June 16, 2010, January 1, 2011, July 1, 2011 and June 16, 2021 (as amended, the Logan Employment Agreement), providing for his employment as Chief Executive Officer of Mirion. The Logan Employment Agreement provides that Mr. Logan is entitled to an annual base salary, which at the beginning of fiscal year 2021 was $629,350 (and which was subsequently increased to $642,566 on October 1, 2020) in connection with the Companys ordinary course annual merit increase process) and eligibility for an annual incentive bonus based on Mirions achievement of targets and milestones as determined by Mirions board of directors. Mr. Logan is also entitled to reimbursement for the following expenses: (i) air travel for first or business class commercial airline travel to and/or from his home to Orange County, California no more than twice per week, including the reasonable costs of ground transportation and parking associated with such air travel, with such costs not to exceed $350 per round trip (provided that, if Mr. Logan elects to make such trips with his personal aircraft, he will be reimbursed for the costs associated with such travel up to $500 per round trip) and (ii) a $1,200 monthly automobile allowance.
298
Table of Contents
The Logan Employment Agreement also provides that if, in the event that any compensatory payments or benefits to which Mr. Logan is or becomes entitled in connection with a change in ownership or effective control (under Section 280G(b)(2) of the Code) of Mirion (the Transaction Payments) become subject to the excise tax imposed by Section 4999 of the Code, then Mirion will pay to Mr. Logan (i) an additional amount (the Gross-Up Payment) such that the net amount retained by Mr. Logan, after deduction for (A) any such excise tax on the Transaction Payments, other than any excise tax imposed in respect of the portion of the Profits Interest granted to Mr. Logan as discussed above under the section entitled Certain Relationships and Related Person TransactionsGSAH, and (B) any federal, state or local income or payroll taxes on the Gross-Up Payment (but before deduction for any federal, state or local income or payroll taxes on the applicable Transaction Payments), is equal to the aggregate value of such Transaction Payments and (ii) an amount equal to the product of any deductions disallowed for federal, state or local income tax purposes because of the inclusion of the Gross-Up Payment in Mr. Logans adjusted gross income multiplied by the highest applicable marginal rate of federal, state or local income taxation, respectively, for the calendar year in which the Gross-Up Payment is made to Mr. Logan.
Pursuant to the Logan Employment Agreement, upon the termination of Mr. Logans employment by Mirion without Cause or by Mr. Logan for Good Reason, subject to his execution and non-revocation of a general release of claims against Mirion, Mr. Logan will be entitled, in addition to any accrued amounts, to (i) continuation of his annual base salary for twelve (12) months following the date of the termination of Mr. Logans employment with Mirion (the Logan Severance Period), (ii) a pro rata portion of Mr. Logans annual incentive bonus for the fiscal year in which the termination of his employment with Mirion occurs, payable at the same time as such payment would otherwise have been made to Mr. Logan had his employment with Mirion not been terminated, and (iii) continuation of any health benefits provided by Mirion to Mr. Logan and his dependents for the Logan Severance Period. The Logan Employment Agreement also provides that in the event of the termination of Mr. Logans employment with Mirion as a result of his death or permanent disability, Mr. Logan or his estate, as applicable, will be entitled, in addition to any accrued amounts, to a pro-rata annual incentive bonus and Mr. Logan and/or his dependents will be entitled to continued health benefits for 12 months.
Cause is defined in the Logan Employment Agreement generally as Mr. Logans (i) commission of or engagement in an act of fraud, embezzlement, sexual harassment, dishonesty or theft in connection with Mr. Logans duties for Mirion or any of its subsidiaries, (ii) material breach of or default under the Logan Employment Agreement or Mr. Logans non-disclosure agreement with Mirion or any similar agreement with Mirion or any of its subsidiaries (which such breach or default, if reasonably capable of being cured, is not cured within two business days after written notice thereof is received by Mr. Logan, or, if reasonably capable of being cured but not within two business days, if Mr. Logan has not commenced cure in good faith within such two business day period and completed such cure as promptly as reasonably practicable thereafter), (iii) conviction of, or plea of nolo contendere with respect to, a felony, or (iv) engagement in an act of gross negligence or willful failure to perform his duties or responsibilities, including the failure to follow in any material respect a direction or written policy of the board of directors of Mirion (which such breach or default, if reasonably capable of cure, is not cured within five business days after written notice thereof or, if reasonably capable of cure but not within five business days, if Mr. Logan has not commenced cure in good faith within such five business day period and completed such cure as promptly as reasonably practicable thereafter). Good Reason is defined in the Logan Employment Agreement generally as any of the following, without Mr. Logans consent: (i) a reduction in Mr. Logans base salary, a material reduction or discontinuation of any material incentive compensation or expense reimbursement plan or the taking of any action with the purpose of materially adversely affecting Mr. Logans participation in benefits under any fringe benefit provided to Mr. Logan (other than with respect to such actions taken by Mirion (other than a reduction in Mr. Logans base salary) as part of an overall plan by Mirion and made applicable to the same extent to all employees of Mirion), (ii) a diminution in Mr. Logans title or position or a significant diminution in Mr. Logans authorities, duties or responsibilities with respect to Mirion, (iii) the requirement by Mirion that Mr. Logan be based in an office which is more than twenty-five (25) miles from Mirions headquarters at Bishop Ranch 8, 3000 Executive Parkway Suite 518, San Ramon, CA or be required to relocate, or (iv) any failure by Mirion to comply with any material provision of the Logan Employment Agreement, any stock option agreement or any other material agreement between Mr. Logan
299
Table of Contents
and Mirion. If Mr. Logan provides written notice of termination of his employment for Good Reason for any of the circumstances described above, Mirion will have the opportunity to cure such circumstances within fifteen (15) days of receipt of such notice. If Mr. Logan does not deliver to Mirion a notice of termination of his employment within ninety (90) days after Mr. Logan has knowledge that an event constituting Good Reason has occurred, such event will no longer constitute Good Reason.
In addition, pursuant to a Confidentiality and Intellectual Property Agreement attached as an exhibit to the Logan Employment Agreement, Mr. Logan is subject to a perpetual obligation not to disclose the confidential information of Mirion.
Freed Employment Agreement
On July 16, 2016, Mirion entered into an employment agreement with Mr. Freed (the Freed Employment Agreement). The Freed Employment Agreement provides that Mr. Freed is entitled to an annual base salary, which was $392,666 at the beginning of fiscal year 2021 and which was increased to $400,912 on October 1, 2020 in connection with the Companys ordinary course annual merit increase process, and eligibility for an annual performance bonus based on personal and corporate performance goals as determined by Mirions board of directors.
Pursuant to the Freed Employment Agreement, upon the termination of Mr. Freeds employment by Mirion without Cause or by Mr. Freed for Good Reason, subject to his execution and non-revocation of a general release of claims against Mirion, Mr. Freed will be entitled, in addition to any accrued amounts, to (i) continuation of his annual base salary for twelve (12) months following the date of the termination of Mr. Freeds employment with Mirion (such twelve (12)-month period, the Freed Severance Period), (ii) a pro rata portion of Mr. Freeds annual incentive bonus for the fiscal year in which the termination of his employment with Mirion occurs, payable at the same time as such payment would otherwise have been made to Mr. Freed had his employment with Mirion not been terminated, and (iii) continued payment by Mirion, for the Freed Severance Period or, if earlier, until the date on which Mr. Freed commences employment with and becomes eligible for health care benefits from a new employer, of the premiums associated with group health continuation coverage premiums for Mr. Freed and his dependents under COBRA. The Freed Employment Agreement also provides that in the event of the termination of Mr. Freeds employment with Mirion as a result of his death or permanent disability, Mr. Freed or his estate, as applicable, will be entitled, in addition to any accrued amounts, to a pro-rata annual incentive bonus.
For purposes of the Freed Employment Agreement, Cause is defined in a manner that is substantially similar to the definition of such term in the Logan Employment Agreement. Under the Freed Employment Agreement, Good Reason is defined generally as any of the following, without Mr. Freeds consent: (i) a material reduction in Mr. Freeds base salary, (ii) a material diminution in Mr. Freeds authorities, duties or responsibilities with respect to Mirion, (iii) the requirement by Mirion that Mr. Freed be based in an office which increases his commute by more than twenty-five (25) miles in relation to Mr. Freeds then-principal place of employment, or (iv) any material breach by Mirion of any material provision of the Freed Employment Agreement. If Mr. Freed provides written notice of termination of his employment for Good Reason for any of the circumstances described above, Mirion will have the opportunity to cure such circumstances within thirty (30) days of receipt of such notice. If Mr. Freed does not deliver to Mirion a notice of termination of his employment within thirty (30) days after Mr. Freed has knowledge that an event constituting Good Reason has occurred, such event will no longer constitute Good Reason.
In addition, pursuant to a Confidentiality, Non-Interference and Intellectual Property Agreement attached as an exhibit to the Freed Employment Agreement, Mr. Freed is subject to (i) non-competition restrictions with respect to certain competitors of Mirion in certain geographical locations for a period of 12 months following his
termination of employment with Mirion for any reason, (ii) non-solicitation restrictions (with respect to certain employees and customers of Mirion) for a period of 12 months following his termination of employment with Mirion for any reason and (iii) a perpetual obligation not to disclose the confidential information of Mirion.
300
Table of Contents
Schopfer Employment Agreement
On May 1, 2020, Mirion entered into the Third Amended and Restated Employment Agreement with Mr. Schopfer (the Schopfer Employment Agreement), which terminated Mr. Schopfers original employment agreement with Mirion dated March 19, 2019, as amended on May 16, 2019 and January 23, 2020, providing for his employment as Chief Financial Officer of Mirion. The Schopfer Employment Agreement provides that Mr. Schopfer is entitled to an annual base salary of $330,000 (and which was later increased to $336,930 on October 1, 2020 and then to $385,000 on January 25, 2021), and eligibility for an annual performance bonus based on personal and corporate performance goals as determined by Mirions board of directors. Mr. Schopfer is also entitled to an annual allowance of $5,000 to cover costs for any personal financial or tax advisory services retained in connection with any matter arising as a result of Mr. Schopfer holding shares of, or any other investment in, Mirion and reimbursement for the cost of an annual physical examination.
Pursuant to the Schopfer Employment Agreement, upon the termination of Mr. Schopfers employment by Mirion without Cause or by Mr. Schopfer for Good Reason, subject to his execution and non-revocation of a general release of claims against Mirion, Mr. Schopfer will be entitled, in addition to any accrued amounts, to (i) continuation of his annual base salary for twelve (12) months following the date of the termination of Mr. Schopfers employment with Mirion (such twelve (12)-month period, the Schopfer Severance Period), (ii) a pro rata portion of Mr. Schopfers annual incentive bonus for the fiscal year in which the termination of his employment with Mirion occurs, payable at the same time as such payment would otherwise have been made to Mr. Schopfer had his employment with Mirion not been terminated, and (iii) continued payment by Mirion, for the Schopfer Severance Period or, if earlier, until the date on which Mr. Schopfer commences employment with and becomes eligible for health care benefits from a new employer, of the premiums associated with group health continuation coverage premiums for Mr. Schopfer and his dependents under COBRA. In addition, in the event of Mr. Schopfers termination of employment without Cause, the balance of a $131,000 loan that was extended by Mirion to Mr. Schopfer on September 24, 2019 to repay a sign-on bonus owed by Mr. Schopfer to his previous employer will be forgiven in full. The Schopfer Employment Agreement also provides that in the event of the termination of Mr. Schopfers employment with Mirion as a result of his death or permanent disability, Mr. Schopfer or his estate, as applicable, will be entitled, in addition to any accrued amounts, to a pro-rata annual incentive bonus. The Schopfer Employment Agreement also provides for a loan from Mirion in a principal amount of $168,833.09 to enable Mr. Schopfer to acquire shares of Topco pursuant to the terms of a loan agreement dated June 2, 2020 as discussed further in the section entitled Certain Relationships and Related Persons TransactionsExecutive Loans.
For purposes of the Schopfer Employment Agreement, Cause is defined in a manner that is substantially similar to the definition of such term in the Logan Employment Agreement, and Good Reason is defined in a manner that is substantially similar to the definition of such term in the Freed Employment Agreement.
In addition, pursuant to a Confidentiality, Non-Interference and Intellectual Property Agreement attached as an exhibit to the Schopfer Employment Agreement, Mr. Schopfer is subject to (i) a covenant restricting him from interfering with the business of the Company by soliciting, diverting or enticing away any officer, employee or consultant of the Company or any of its subsidiaries to accept employment with a third party for a period of 12 months following his termination of employment with Mirion for any reason, (ii) a covenant restricting him in perpetuity from using the confidential information of the Company to solicit, divert or entice away (A) any actual or prospective customer of the Company or any of its subsidiaries to become a customer of any third party that is
engaged in any business or operations that were also engaged in by the Company during Mr. Schopfers employment with the Company or (B) any customer or supplier to cease doing business with the Company or any of its subsidiaries and (iii) a perpetual obligation not to disclose the confidential information of Mirion.
301
Table of Contents
DIRECTOR COMPENSATION
The following table sets forth a summary of the compensation we paid to Lawrence Kingsley for fiscal year 2021. Other than as set forth in the table, Mirion did not pay any compensation to, make any equity awards or non-equity awards to, or pay any other compensation to any of the other member of Mirions board of directors during Mirions fiscal year 2021.
| Name |
Fees Earned or Paid in Cash |
Stock Awards ($) | Total ($) |
|||||||||
| Lawrence D. Kingsley |
| 32,466,000 | (1) | 32,466,000 | ||||||||
| (1) | Reflects the grant date value of a one-time grant of profits interests in the Sponsor, which was approved and granted by the Sponsor in recognition of Mr. Kingsleys efforts in connection with the Business Combination. As discussed above under Executive CompensationProfits Interests, the Sponsor granted Mr. Kingsley the award of profits interests on June 16, 2021 in connection with the signing of the Business Combination Agreement. The profits interests award provides for service and performance-vesting, with the award only vesting upon the achievement of specified share price conditions. The grant date fair value of the profits interests is based upon a valuation model using Monte Carlo simulations in accordance with FASB Accounting Standards Codification (ASC) Topic 718. |
Following the Business Combination, we intend to adopt the initial terms of a non-employee director compensation policy. Our board of directors is still in the process of considering the non-employee director compensation policy.
302
Table of Contents
DESCRIPTION OF NEW MIRION SECURITIES
In connection with the Business Combination, GSAH will adopt a new certificate of incorporation and bylaws. The following summary of certain provisions of New Mirions securities does not purport to be complete and is subject to the New Mirion Charter, the New Mirion Bylaws and the provisions of applicable law. Copies of the New Mirion Charter and the New Mirion Bylaws are attached to this proxy statement/prospectus as Annex B and Annex C, respectively. You are encouraged to read the applicable provisions of Delaware law, the New Mirion Charter and the New Mirion Bylaws in their entirety for a complete description of the rights and preferences of New Mirion securities following the Business Combination.
General
Immediately following the completion of the Business Combination, New Mirions authorized capital stock will consist of shares of capital stock, par value $0.0001 per share, of which:
| | 2,000,000,000 shares are designated as Class A common stock; |
| | 100,000,000 shares are designated as Class B common stock; and |
| | 100,000,000 shares are designated as preferred stock. |
New Mirions Board will be authorized, without stockholder approval, except as required by the listing standards of the NYSE, to issue additional shares of capital stock.
As of March 31, 2021, GSAH had 75,000,000 shares of Class A common stock, 18,750,000 shares of Class B common stock issued and outstanding and no shares of preferred stock outstanding. GSAH also had issued and outstanding, as of March 31, 2021, 27,250,000 warrants, consisting of 18,750,000 public warrants and 8,500,000 private placement warrants, and no GSAH units outstanding. As of March 31, 2021 Mirion had approximately 11 holders of record of A Ordinary Shares and approximately 20 holders of record of B Ordinary Shares. After giving effect to the Business Combination, New Mirion will have approximately 171,000,000 shares of New Mirion Class A common stock outstanding (assuming no redemptions) and approximately 9,000,000 shares of New Mirion Class B common stock outstanding (assuming no redemptions and excluding the founder shares).
Common Stock
Class A Common Stock
Holders of shares of New Mirion Class A common stock are entitled to one vote for each share held of record on all matters on which stockholders are entitled to vote generally, including the election or removal of directors. The holders of New Mirion Class A common stock do not have cumulative voting rights in the election of directors.
Holders of shares of New Mirion Class A common stock are entitled to receive dividends when and if declared by the New Mirion Board out of funds legally available therefor, subject to any statutory or contractual restrictions on the payment of dividends and to any restrictions on the payment of dividends imposed by the terms of any outstanding preferred stock.
Upon New Mirions liquidation, dissolution or winding up and after payment in full of all amounts required to be paid to creditors and to the holders of preferred stock having liquidation preferences, if any, the holders of shares of New Mirion Class A common stock will be entitled to receive pro rata New Mirions remaining assets available for distribution.
Class B Common Stock
Holders of shares of New Mirion Class B common stock are entitled to one vote for each share held of record on all matters on which stockholders are entitled to vote generally, including the election or removal of directors. If
303
Table of Contents
at any time the ratio at which shares of IntermediateCo Class B common stock are redeemable or exchangeable for shares of New Mirions Class A common stock changes from one-for-one as described under Certain Relationships and Related Person TransactionsIntermediateCo Charter, the number of votes to which New Mirion Class B common stockholders are entitled will be adjusted accordingly. The holders of New Mirions Class B common stock do not have cumulative voting rights in the election of directors.
Except for transfers to us pursuant to the IntermediateCo Charter or to certain permitted transferees set forth in the New Mirion Charter, the shares of New Mirion Class B common stock and corresponding shares of IntermediateCo Class B common stock may not be sold, transferred or otherwise disposed of.
Holders of shares of New Mirion Class B common stock are not entitled to economic interests in New Mirion or to receive dividends or to receive a distribution upon a liquidation or winding up of Mirion Technologies, Inc. However, if IntermediateCo makes distributions to us other than solely with respect to the New Mirion Class A common stock, the holders of IntermediateCo Class B common stock will be entitled to receive distributions pro rata in accordance with the percentages of their respective shares of IntermediateCo Class B common stock. See Certain Relationships and Related Person TransactionsIntermediateCo Charter.
Voting Rights
Except as otherwise required in the New Mirion Charter or by applicable law, the holders of New Mirion common stock will vote together as a single class on all matters on which stockholders generally are entitled to vote.
The holders of the outstanding shares of New Mirion Class A common stock shall be entitled to vote separately upon any amendment to the New Mirion Charter (including by merger, consolidation, reorganization or similar event) that would alter or change the powers, preferences or special rights of the New Mirion Class A common stock in a manner that is materially and disproportionately adverse as compared to any alteration or change to the New Mirion Class B common stock.
The holders of the outstanding shares of New Mirion Class B common stock shall be entitled to vote separately upon any amendment to the New Mirion Charter (including by merger, consolidation, reorganization or similar event) that would alter or change the powers, preferences or special rights of the New Mirion Class B common stock in a manner that is materially and disproportionately adverse as compared to any alteration or change to the New Mirion Class A common stock; subject to certain exceptions set forth in the New Mirion Charter.
Redemption and Exchange
Under the IntermediateCo Charter, the holders of IntermediateCo Class B common stock will have the right, from and after the completion of the Business Combination (subject to the terms of the IntermediateCo Charter), to require IntermediateCo to redeem all or a portion of their shares of IntermediateCo Class B common stock for, at New Mirions election, (1) newly issued shares of New Mirion Class A common stock on a one-for-one basis or (2) a cash payment equal to the arithmetic average of the closing stock prices for a share of New Mirion Class A common stock for each of three (3) consecutive full trading days ending on and including the last full trading day immediately prior to the date of redemption (subject to customary adjustments, including for stock splits, stock dividends and reclassifications) in accordance with the terms of the IntermediateCo Charter. Additionally, in the event of a redemption request by a holder of IntermediateCo Class B common stock, we may, at our election, effect a direct exchange of cash or New Mirion Class A common stock for IntermediateCo Class B common stock in lieu of such a redemption. Shares of New Mirion Class B common stock will be canceled on a one-for-one basis if we, following a redemption request of a holder of IntermediateCo Class B common stock, redeem or exchange IntermediateCo Class B common stock of such holder of IntermediateCo Class B common stock pursuant to the terms of the IntermediateCo Charter. See Certain Relationships and Related Person TransactionsIntermediateCo Charter.
304
Table of Contents
Valid Issuance
All shares of New Mirion common stock that will be outstanding at the closing of the Business Combination will be fully paid and non-assessable. The New Mirion common stock will not be subject to calls or assessments by New Mirion. The rights, powers and privileges of New Mirions common stock will be subject to those of the holders of any shares of New Mirions preferred stock or any other series or class of stock New Mirion may authorize and issue in the future.
Preferred Stock
Pursuant to the New Mirion Charter, the New Mirion Board will have the authority, without further action by the stockholders, to issue from time to time shares of preferred stock in one or more series. The New Mirion Board may designate the rights, preferences, privileges and restrictions of the New Mirion preferred stock, including dividend rights, conversion rights, voting rights, redemption rights, liquidation preference, sinking fund terms, and the number of shares constituting any series or the designation of any series. There will be no shares of New Mirion preferred stock outstanding immediately upon consummation of the Business Combination.
The issuance of New Mirion preferred stock could have the effect of restricting dividends on the New Mirion common stock, diluting the voting power of the New Mirion common stock, impairing the liquidation rights of the New Mirion common stock or delaying, deterring, or preventing a change in control. Such issuance could have the effect of decreasing the market price of the New Mirion common stock. There are currently no plans to issue any shares of New Mirion preferred stock.
Warrants
As of March 31, 2021, GSAH had issued GSAH warrants to purchase 27,250,000 shares of GSAH Class A common stock, of which GSAH warrants to purchase 8,500,000 shares of GSAH Class A common stock are held by the Sponsor. Each GSAH warrant that is outstanding will be automatically converted into a warrant to acquire New Mirion Class A common stock on substantially the same terms and conditions as specified in the GSAH warrants but with references to GSAH Class A common stock replaced with references to New Mirion Class A common stock.
Authorized but Unissued Capital Stock
Delaware law does not require stockholder approval for any issuance of authorized shares. However, the listing requirements of the NYSE, which would apply so long as shares of New Mirion Class A common stock remain listed on the NYSE, require stockholder approval of certain issuances equal to or exceeding 20% of the then outstanding voting power or the then outstanding number of shares of Class A common stock (we believe the position of the NYSE is that the calculation in this latter case treats as outstanding shares of New Mirion Class A common stock issuable upon redemption or exchange of outstanding shares of IntermediateCo Class B common stock not held by New Mirion). These additional shares of New Mirion Class A common stock may be used for a variety of corporate purposes, including future public offerings, to raise additional capital or to facilitate acquisitions.
Transfer Restrictions
Holders of Mirion securities receiving shares of New Mirion common stock pursuant to the Business Combination will be subject to certain transfer restrictions set forth in the New Mirion Registration Rights Agreement. See Certain Relationships and Related Person TransactionsNew Mirion Registration Rights Agreement.
Exclusive Forum
The New Mirion Charter provides that the sole and exclusive forum for (1) any derivative action or proceeding brought on New Mirions behalf, (2) any action asserting a claim of breach of a fiduciary duty owed by any of
305
Table of Contents
New Mirions current or former directors, officers or other employees to New Mirion or its stockholders, (3) any action asserting a claim against New Mirion or any current or former director, officer or other employee of New Mirion arising out of or pursuant to any provision of the DGCL, the New Mirion Charter or the New Mirion Bylaws; (4) any action to interpret, apply, enforce, or determine the validity of the New Mirion Charter or the New Mirion Bylaws, (5) any claim or cause of action as to which the DGCL confers jurisdiction on the Court of Chancery of the State of Delaware, and (6) any other action asserting a claim that is governed by the internal affairs doctrine shall be a state or federal court located within the State of Delaware, in all cases subject to the court having jurisdiction over indispensable parties named as defendants. However, this exclusive forum provision would not apply to suits brought to enforce a duty or liability created by the Securities Act, or the Exchange Act or any claim for which the federal district courts of the United States have exclusive jurisdiction.
In addition, the New Mirion Charter provides that, unless New Mirion consents in writing to the selection of an alternative forum, the federal district courts of the United States will be the exclusive forum for resolving any complaint asserting a cause of action arising under the Securities Act. However, this exclusive forum provision would not apply to suits brought to enforce a duty or liability created by the Exchange Act or any claim for which the federal district courts of the United States have exclusive jurisdiction.
Any person or entity purchasing or otherwise acquiring any interest in New Mirion capital stock shall be deemed to have notice of and consented to these provisions and will not be deemed to have waived New Mirions compliance with the federal securities laws and the regulations promulgated thereunder. Although the GSAH Board believes these provisions benefit New Mirion by providing increased consistency in the application of Delaware law or federal law for the specified types of actions and proceedings, these provisions may have the effect of discouraging lawsuits against New Mirion or its directors and officers.
Limitations on Liability and Indemnification of Officers and Directors
The New Mirion Charter provides that New Mirion will indemnify New Mirions directors to the fullest extent authorized or permitted by applicable law. New Mirion expects to enter into agreements to indemnify New Mirions directors, executive officers and other employees as determined by the New Mirion Board. Under the New Mirion Bylaws, New Mirion is required to indemnify each of New Mirions directors and officers if the basis of the indemnitees involvement was by reason of the fact that the indemnitee is or was a director or officer of New Mirion or was serving at New Mirions request as a director, officer, employee or agent for another entity. New Mirion must indemnify New Mirions officers and directors against all expenses, judgments, fines and amounts paid in settlement actually and reasonably incurred by the indemnitee in connection with such action, suit or proceeding if the indemnitee acted in good faith and in a manner the indemnitee reasonably believed to be in or not opposed to the best interests of New Mirion, and, with respect to any criminal action or proceeding, had no reasonable cause to believe the indemnitees conduct was unlawful. The New Mirion Charter also requires New Mirion to advance expenses incurred by a director or officer in connection with such action, suit or proceeding to the maximum extent permitted under Delaware law. Any claims for indemnification by New Mirions directors and officers may reduce New Mirions available funds to satisfy successful third-party claims against New Mirion and may reduce the amount of money available to New Mirion.
Corporate Opportunities
Delaware law permits corporations to adopt provisions renouncing any interest or expectancy in certain opportunities that are presented to the corporation or its officers, directors or stockholders. The New Mirion Charter will, to the extent permitted by Delaware law, renounce any interest or expectancy that New Mirion has in, or right to be offered an opportunity to participate in, specified business opportunities that are from time to time presented to a member of the New Mirion Board who is not an employee, or any partner, member, director, stockholder, employee or agent of such member. Notwithstanding the foregoing, the New Mirion Charter to be in effect upon the Closing will not renounce any interest in a business opportunity that is expressly offered to a director solely in his or her capacity as a director of New Mirion.
306
Table of Contents
Anti-takeover Effects of the New Mirion Charter and the New Mirion Bylaws
The New Mirion Charter and New Mirion Bylaws contain provisions that could have the effect of delaying, deferring or discouraging another party from acquiring control of New Mirion. These provisions and certain provisions of Delaware law, which are summarized below, could discourage takeovers, coercive or otherwise. These provisions are also designed, in part, to encourage persons seeking to acquire control of New Mirion to negotiate first with the New Mirion Board. The GSAH Board believes that the benefits of increased protection of the potential ability to negotiate with an unfriendly or unsolicited acquirer outweigh the disadvantages of discouraging a proposal to acquire New Mirion.
Issuance of Undesignated Preferred Stock
As discussed above in the section titled Preferred Stock, the New Mirion Board will have the ability to designate and issue preferred stock with voting or other rights or preferences that could deter hostile takeovers or delay changes in New Mirions control or management.
Limits on Ability of Stockholders to Act by Written Consent or Call a Special Meeting
The New Mirion Charter provides that New Mirion stockholders may not act by written consent This limit on the ability of stockholders to act by written consent may lengthen the amount of time required to take stockholder actions. As a result, the holders of a majority of New Mirion common stock would not be able to amend the New Mirion Charter or New Mirion Bylaws or remove directors without holding a meeting of stockholders called in accordance with the New Mirion Bylaws.
In addition, the New Mirion Charter provides that special meetings of the stockholders may be called only by the chairman of the New Mirion Board of, the chief executive officer or the New Mirion Board acting pursuant to a resolution adopted by a majority of the New Mirion Board. A stockholder may not call a special meeting, which may delay the ability of New Mirion stockholders to force consideration of a proposal or for holders controlling a majority of New Mirion capital stock to take any action, including the removal of directors.
Advance Requirements for Advance Notification of Stockholder Nominations and Proposals
The New Mirion Bylaws will establish advance notice procedures with respect to stockholder proposals and the nomination of candidates for election as directors, other than nominations made by or at the direction of the New Mirion Board or a committee thereof. These advance notice procedures may have the effect of precluding the conduct of certain business at a meeting if the proper procedures are not followed and may also discourage or deter a potential acquirer from conducting a solicitation of proxies to elect its own slate of directors or otherwise attempt to obtain control of New Mirion.
Election and Removal of Directors
The New Mirion Charter and New Mirion Bylaws contain provisions that establish specific procedures for appointing and removing members of the New Mirion Board. Under the New Mirion Charter and New Mirion Bylaws, vacancies and newly created directorships on the New Mirion Board may be filled only by a majority of the directors then serving on the New Mirion Board. We will also be subject to certain director nomination agreements at the closing of the Business Combination which will require us to nominate certain directors for election to the New Mirion Board. See Certain Relationships and Related Person TransactionsDirector Nomination Agreements.
No Cumulative Voting
The DGCL provides that stockholders are not entitled to the right to cumulate votes in the election of directors unless the New Mirion Charter provides otherwise. The New Mirion Charter does not expressly provide for
307
Table of Contents
cumulative voting. Without cumulative voting, a minority stockholder may not be able to gain as many seats on the New Mirion Board as the stockholder would be able to gain if cumulative voting were permitted. The absence of cumulative voting makes it more difficult for a minority stockholder to gain a seat on the New Mirion Board to influence New Mirion Boards decision regarding a takeover.
Amendment of New Mirion Charter and New Mirion Bylaws
The affirmative vote of holders of not less than 66 2/3% of the total voting power of all outstanding securities of New Mirion generally entitled to vote in the election of directors, voting together as a single class will be required to amend, alter, change or repeal specified provisions of the New Mirion Charter, including those relating to the terms of the New Mirion common stock, actions by written consent of stockholders, calling of special meetings of stockholders, election and removal of directors, certain indemnification and corporate opportunity matters, and the required vote to amend the New Mirion Charter and New Mirion Bylaws. The New Mirion Bylaws may only be amended by the New Mirion Board or the affirmative vote of holders of not less than 66 2/3% of the total voting power of all outstanding securities of the Corporation generally entitled to vote in the election of directors, voting together as a single class. This requirement of a super-majority vote to approve amendments to the New Mirion Charter and New Mirion Bylaws could enable a minority of our stockholders to exercise veto power over any such amendments.
Delaware Anti-Takeover Statute
New Mirion will be subject to the provisions of Section 203 of the DGCL regulating corporate takeovers. In general, Section 203 prohibits a publicly held Delaware corporation from engaging, under certain circumstances, in a business combination with an interested stockholder for a period of three years following the date the person became an interested stockholder unless:
| | prior to the date of the transaction, the New Mirion Board approved either the business combination or the transaction that resulted in the stockholder becoming an interested stockholder; |
| | upon completion of the transaction that resulted in the stockholder becoming an interested stockholder, the interested stockholder owned at least 85% of the voting stock of the corporation outstanding at the time the transaction commenced, excluding for purposes of determining the voting stock outstanding, but not the outstanding voting stock owned by the interested stockholder, (1) shares owned by persons who are directors and also officers and (2) shares owned by employee stock plans in which employee participants do not have the right to determine confidentially whether shares held subject to the plan will be tendered in a tender or exchange offer; or |
| | at or subsequent to the date of the transaction, the business combination is approved by New Mirion Board and authorized at an annual or special meeting of stockholders, and not by written consent, by the affirmative vote of at least two-thirds of the outstanding voting stock that is not owned by the interested stockholder. |
Generally, a business combination includes a merger, asset or stock sale, or other transaction resulting in a financial benefit to the interested stockholder. An interested stockholder is a person who, together with affiliates and associates, owns or, within three years prior to the determination of interested stockholder status, did own 15% or more of a corporations outstanding voting stock. The GSAH Board expects the existence of this provision to have an anti-takeover effect with respect to transactions the New Mirion Board does not approve in advance.
The provisions of Delaware law and the provisions of the New Mirion Charter and New Mirion Bylaws could have the effect of discouraging others from attempting hostile takeovers and as a consequence, they might also inhibit temporary fluctuations in the market price of New Mirion Class A common stock that often result from actual or rumored hostile takeover attempts. These provisions might also have the effect of preventing changes in
308
Table of Contents
New Mirions management. It is also possible that these provisions could make it more difficult to accomplish transactions that stockholders might otherwise deem to be in their best interests
Listing
New Mirion will apply to have its Class A common stock approved for listing on The New York Stock Exchange under the symbol MIR.
Transfer Agent
The transfer agent for New Mirion common stock will be Continental Stock Transfer & Trust Company.
309
Table of Contents
COMPARISON OF NEW MIRION STOCKHOLDER RIGHTS
General
GSAH is incorporated under the laws of the State of Delaware and the rights of GSAH Stockholders are governed by the laws of the State of Delaware, including the DGCL, and the GSAH Certificate of Incorporation and the GSAH Bylaws. Following the Business Combination, GSAH will be renamed Mirion Technologies, Inc., but will remain incorporated under the laws of the State of Delaware. Thus, following the Business Combination, the rights of GSAH Stockholders who become New Mirion stockholders in the Business Combination will continue to be governed by Delaware law but will no longer be governed by the GSAH Certificate of Incorporation and the GSAH Bylaws and instead will be governed by the New Mirion Charter and the New Mirion Bylaws.
Comparison of Stockholders Rights
Set forth below is a comparison of material differences between the rights of GSAH Stockholders under the GSAH Certificate of Incorporation and the GSAH Bylaws (left column), and the rights of New Mirions stockholders under forms of the New Mirion Charter and the New Mirion Bylaws (right column). The summary set forth below is not intended to be complete or to provide a comprehensive discussion of each set of governing documents. This summary is qualified in its entirety by reference to the full text of the GSAH Certificate of Incorporation and GSAH Bylaws and forms of the New Mirion Charter and the New Mirion Bylaws, which are attached as Annex B and Annex C, respectively, as well as the relevant provisions of the DGCL.
| GSAH |
New Mirion |
|
| Authorized Capital Stock |
||
| GSAH is currently authorized to issue 555,000,000 shares of capital stock, each with a par value of $0.0001 per share, consisting of (a) 550,000,000 shares of common stock, including 500,000,000 shares of GSAH Class A common stock and 50,000,000 shares of GSAH Class B common stock and (b) 5,000,000 shares of preferred stock.
As of March 31, 2021, there were 75,000,000 shares of GSAH Class A common stock and 18,750,000 shares of GSAH Class B common stock outstanding. |
New Mirion will be authorized to issue 2,200,000,000 shares of capital stock, each with a par value of $0.0001 per share, consisting of (a) 2,100,000,000 shares of common stock, including 2,000,000,000 shares of New Mirion Class A common stock and 100,000,000 shares of New Mirion Class B common stock and (b) 100,000,000 shares of preferred stock.
After giving effect to the Business Combination and following the assumptions set forth under Summary of the Proxy Statement/Prospectus, New Mirion will have approximately 171,000,000 shares of New Mirion Class A common stock outstanding (assuming no redemptions) and approximately 9,000,000 shares of New Mirion Class B common stock outstanding (assuming no redemptions and excluding the founder shares). Following the consummation of the Business Combination, New Mirion is not expected to have any preferred stock outstanding. |
|
| The Conversion |
||
| Shares of GSAH Class B common stock will automatically convert into GSAH Class A common stock on the Closing of the Business Combination on a one-for-one basis, subject to adjustment.
In the case that additional shares of GSAH Class A common stock, or equity-linked securities convertible or exercisable for shares of GSAH Class A common |
There are no conversion rights relating to the New Mirion common stock. | |
310
Table of Contents
| GSAH |
New Mirion |
|
| stock, are issued or deemed issued in excess of the amounts offered in the initial public offering and related to the Closing of the Business Combination, the ratio at which the GSAH Class B common stock will convert into GSAH Class A common stock at a ratio for which: (i) the numerator will equal to the sum of (A) 25% of all shares of GSAH Class A common stock issued or issuable by GSAH, related or in connection with the consummation of the Business Combination (net the number of shares redeemed in connection with the transaction and excluding securities issued or issuable to any seller in the Business Combination) plus (B) the number of shares of GSAH Class B common stock issued and outstanding prior to the closing of the Business Combination; and (ii) the denominator will be the number of shares of GSAH Class B common stock issued and outstanding prior to the closing of the Business Combination.
Holders of a majority of GSAH Class B common stock may waive the one-for-one basis conversion ratio by written consent or agreeing separately as a single class; provided, that the conversion ratio for GSAH Class B common stock cannot be less than one-for-one. |
||
| Rights of Preferred Stock |
||
| The GSAH Board is authorized to issue one or more series of preferred stock and to establish from time to time the number of shares to be included in each such series and to fix the voting rights, designations, powers, preferences and relative, participating, optional, special and other rights of each such series, and any qualifications, limitations and restrictions thereof. | The New Mirion Board is authorized to issue one or more series of preferred stock and to establish from time to time the number of shares to be included in each such series and to fix the voting rights, designations, powers, preferences and relative, participating, optional, special and other rights of each such series, and any qualifications, limitations and restrictions thereof. | |
| Number and Qualification of Directors |
||
| Subject to any rights of holders of preferred stock to elect one or more directors, the number of directors is fixed exclusively by the GSAH Board pursuant to a resolution adopted by a majority of the GSAH Board. | Subject to any rights of holders of preferred stock to elect one or more directors, the number of directors is fixed exclusively by the New Mirion Board pursuant to a resolution adopted by a majority of the New Mirion Board. | |
| Election of Directors |
||
| At each annual meeting, the GSAH stockholders entitled to vote on such matters shall elect each director to a term ending on the date of the annual meeting of the stockholders or until the election and qualification of their respective successors in office, subject to their earlier death, resignation or removal.
Prior to the closing of the Business Combination, the holders of GSAH Class B common stock have the exclusive right to elect and remove any director, and |
At each annual meeting, the New Mirion stockholders entitled to vote on such matters shall elect each director to a term ending on the date of the annual meeting of the stockholders or until the election and qualification of their respective successors in office, subject to their earlier death, resignation or removal.
Subject to any rights of holders of preferred stock, the election of directors shall be determined by a |
|
311
Table of Contents
| GSAH |
New Mirion |
|
| the holders of GSAH Class A common stock have no right to elect or remove any director. This provision can only be amended by a resolution passed by holders of at least a majority of the then-outstanding GSAH Class B common stock.
Subject to any rights of holders of preferred stock and the GSAH Class B common stock prior to the Business Combination, the election of directors shall be determined by a plurality of the votes cast by the stockholders present in person or represented by proxy at the meeting and entitled to vote on the election of directors. |
plurality of the votes cast by the stockholders present in person or represented by proxy at the meeting and entitled to vote on the election of directors. | |
| Removal of Directors |
||
| The holders of GSAH Class B common stock have the exclusive right to remove any GSAH director of GSAH from office, but only for cause and only by an affirmative vote of the holders of a majority of the total voting power of then-outstanding GSAH Class B common stock entitled to vote in the election of directors, voting together as a single class. This provision can only be amended by a resolution passed by a majority of holders of at least ninety percent (90%) of the outstanding common stock entitled to vote thereon. | Subject to any rights of holders of preferred stock, the directors of New Mirion may be removed from office at any time, with or without cause, by the affirmative vote of the holders of a majority of the total voting power of the then-outstanding securities of New Mirion entitled to vote in the election of directors, voting together as a single class. | |
| Vacancies on the Board of Directors |
||
| Subject to any rights of holders of preferred stock and the GSAH Class B common stock prior to the Business Combination, newly created directorships resulting from an increase in the number of directors and any vacancies on the GSAH Board resulting from death, resignation, retirement, disqualification, removal or other cause may be filled solely and exclusively by a majority vote of the remaining directors then in office, even if less than a quorum, or by a sole remaining director. | Newly created directorships resulting from an increase in the number of directors and any vacancies on the New Mirion Board resulting from death, resignation, retirement, disqualification, removal or other cause may be filled solely and exclusively by a majority vote of the remaining directors then in office, even if less than a quorum, or by a sole remaining director. | |
| Voting |
||
| Except as required by law or the GSAH Certificate of Incorporation, holders of GSAH Class A common stock and GSAH Class B common stock are entitled to one vote on all matters submitted to a vote of the stockholders, voting together as a single class.
However, prior to the Business Combination, only holders of GSAH Class B common stock will have the right to elect and remove any director.
Except as required by law or the GSAH Certificate of Incorporation, holders of GSAH Class A common stock and GSAH Class B common stock are not entitled to vote on any amendment to the GSAH |
Holders of New Mirion Class A common stock and Class B common stock are entitled to one vote on all matters submitted to a vote of the stockholders, including the election of directors, voting together as a single class.
The holders of the outstanding shares of New Mirion Class A common stock shall be entitled to vote separately upon any amendment to the New Mirion Charter (including by merger, consolidation, reorganization or similar event) that would alter or change the powers, preferences or special rights of the New Mirion Class A common stock in a manner |
|
312
Table of Contents
| GSAH |
New Mirion |
|
| Certificate of Incorporation that relates solely to terms of one or more outstanding series of preferred stock, or other series of common stock, if the holders of such affected series of preferred stock or common stock are entitled, either separately or together with the holders of one or more other such series, to vote thereon pursuant to the GSAH Certificate of Incorporation or pursuant to the DGCL. | that is materially and disproportionately adverse as compared to any alteration or change to the New Mirion Class B common stock.
The holders of the outstanding shares of New Mirion Class B common stock shall be entitled to vote separately upon any amendment to the New Mirion Charter (including by merger, consolidation, reorganization or similar event) that would alter or change the powers, preferences or special rights of the New Mirion Class B common stock in a manner that is materially and disproportionately adverse as compared to any alteration or change to the New Mirion Class A common stock; subject to certain exceptions set forth in the New Mirion Charter.
Subject to the rights of any holders of preferred stock, the number of authorized shares of New Mirion common stock or preferred stock may be increased or decreased (but not below the number of shares thereof then outstanding) by the affirmative vote of the holders of capital stock representing a majority of the voting power of all the then-outstanding shares of capital stock entitled to vote irrespective of Section 242(b)(2) of the DGCL.
Except as required by law or the New Mirion Charter, holders of New Mirion common stock are not entitled to vote on any amendment to the New Mirion Charter that relates solely to terms of one or more outstanding series of preferred stock if the holders of such affected series of preferred stock are entitled, either separately or together with the holders of one or more other such series, to vote thereon pursuant to the New Mirion Charter or pursuant to the DGCL. |
|
| Cumulative Voting |
||
| The DGCL allows for cumulative voting only if provided for in the certificate of incorporation. The GSAH Certificate of Incorporation does not authorize cumulative voting. | The DGCL allows for cumulative voting only if provided for in the certificate of incorporation. The New Mirion Charter does not authorize cumulative voting. | |
| Supermajority Voting Provisions |
||
| The affirmative vote of GSAH Stockholders holding at least sixty-five percent (65%) of the voting power of then-outstanding shares of capital stock is required to amend or repeal Article IX of the GSAH Certificate of Incorporation concerning business combination requirements.
The affirmative vote of GSAH Stockholders holding at least ninety percent (90%) of the voting power of then-outstanding shares of capital stock is required to amend |
The affirmative vote of holders of not less than 66 2/3% of the total voting power of all outstanding securities of New Mirion generally entitled to vote in the election of directors, voting together as a single class will be required to amend, alter, change or repeal specified provisions of the New Mirion Charter, including those relating to the terms of the New Mirion common stock, actions by written consent of stockholders, calling of special meetings of stockholders, election and removal of directors, | |
313
Table of Contents
| GSAH |
New Mirion |
|
| or repeal the exclusive right of GSAH Class B common stockholders to elect, remove and replace any director prior to the Closing of the Business Combination. | certain indemnification and corporate opportunity matters, and the required vote to amend the New Mirion Charter and New Mirion Bylaws. The New Mirion Bylaws may only be amended by the New Mirion Board or the affirmative vote of holders of not less than 66 2/3% of the total voting power of all outstanding securities of New Mirion generally entitled to vote in the election of directors, voting together as a single class. | |
| Special Meeting of the Board of Directors |
||
| The GSAH Bylaws provide that special meetings of the GSAH Board may be called by the chairman of the board, the president or the secretary of GSAH, and must be called upon the written request of at least a majority of directors then in office or the sole director, as the case may be. Any and all business that may be transacted at a regular meeting of the GSAH Board may be transacted at a special meeting. A special meeting may be held at any time without notice if all the directors are present or if those not present waive notice of the meeting. | The New Mirion Bylaws provide that special meetings of the New Mirion Board may be called by the chairperson of the board, the chief executive officer or on the written request of a majority of directors then in office. Any and all business that may be transacted at a regular meeting of the New Mirion Board may be transacted at a special meeting. | |
| Stockholder Action by Written Consent |
||
| Except as may otherwise be provided for or fixed pursuant to the GSAH Certificate of Incorporation relating to the rights of any holders of preferred stock, any action required or permitted to be taken by GSAH stockholders must be effected by a duly called annual or special meeting of such stockholders and may not be effected by written consent of the stockholders other than with respect to GSAH Class B common stock. |
Except as may otherwise be provided for or fixed pursuant to the New Mirion Charter relating to the rights of any holders of preferred stock, any action by New Mirion stockholders required or permitted to be made by the stockholders must be effected by a duly called annual or special meeting of such stockholders, and may not be effected by written consent without a meeting. | |
| Any action required or permitted to be taken at any meeting of the holders of GSAH Class B common stock may be taken without a meeting, without prior notice and without a vote. The written consents must set forth the action taken and must be signed by the holders of the outstanding GSAH Class B common stock having not less than the minimum number of votes necessary to authorize or take such action at a meeting at which all shares of GSAH Class B common stock were present and voted. | ||
| Amendment to Certificate of Incorporation |
||
| Except as otherwise required by the GSAH Certificate of Incorporation, any amendment to the GSAH Certificate of Incorporation requires (i) the approval of the GSAH Board, (ii) the approval of a majority of the voting power of the outstanding shares of GSAH stock entitled to vote upon the proposed amendment, voting together as a single class and (iii) the approval of the | Any amendment to the New Mirion Charter requires (i) the approval of the New Mirion Board, and (ii) the approval of a majority of the voting power of the outstanding securities of New Mirion entitled to vote upon the proposed amendment, voting together as a single class, subject to the additional provisions described above under Voting and Supermajority Voting Provisions. | |
314
Table of Contents
| GSAH |
New Mirion |
|
| holders of a majority of the outstanding stock of each class entitled to vote thereon as a class, if any.
As long as any shares of GSAH Class B common stock remain outstanding, approval by majority of the then-outstanding shares of GSAH Class B common stock, voting separately a class, is required to amend, alter or repeal any provision of the GSAH Certificate of Incorporation, which would alter or change the powers, preferences or relative, participating, optional or other or special rights of the GSAH Class B common stock. |
||
| Amendment of the Bylaws |
||
| The GSAH Bylaws may be adopted, amended, altered or repealed by a majority of the GSAH Board or by the affirmative vote of the holders of at least two-thirds (66 2/3%) of the total voting power of all outstanding securities generally entitled to vote in the election of directors, voting together as a single class. | The New Mirion Bylaws may be adopted, amended, altered or repealed by a majority of the New Mirion Board or by the affirmative vote of the holders of at least two-thirds (66 2/3%) of the total voting power of all outstanding securities generally entitled to vote in the election of directors, voting together as a single class. | |
| Quorum |
||
| Board of Directors. A majority of the GSAH Board constitutes a quorum.
Stockholders. The presence, in person or by proxy, at a stockholders meeting of the holders of shares of outstanding capital stock representing a majority of the voting power of all outstanding shares of capital stock constitutes a quorum, except that when specified business is to be voted on by a class or series of stock voting as a class, the holders of shares representing a majority of the voting power of the outstanding shares of such class or series will constitute a quorum. |
Board of Directors. A majority of the New Mirion Board constitutes a quorum.
Stockholders. The presence, in person or by proxy, at a stockholders meeting of the holders of shares of outstanding capital stock representing a majority of the voting power of all outstanding shares of capital stock constitutes a quorum, except that when specified business is to be voted on by a class or series of stock voting as a class, the holders of shares representing a majority of the voting power of the outstanding shares of such class or series will constitute a quorum. |
|
| Doctrine of Corporate Opportunity |
||
| The GSAH Certificate of Incorporation provides that the doctrine of corporate opportunity will not apply with respect to GSAH or any of its directors or officers in circumstances where the application of any such doctrine to a corporate opportunity would conflict with any fiduciary duties or contractual obligations they may have. In addition, prior to the consummation of the Business Combination, the doctrine of corporate opportunity will not apply to any other corporate opportunity with respect to any of GSAHs directors or officers unless such corporate opportunity is offered to such person solely in his or her capacity as a GSAH director or officer and such opportunity is one GSAH is legally and contractually permitted to undertake and would otherwise be reasonable for GSAH to pursue and the director or officer is permitted to refer that | The New Mirion Charter provides that the doctrine of corporate opportunity will not apply with respect to any member of the New Mirion Board who is not an employee of New Mirion or its subsidiaries, or any employee or agent of such member, other than someone who is an employee of New Mirion or its subsidiaries (collectively, the Covered Persons) except with respect to those business opportunity matters, potential transactions, interests or other matters that a Covered Person obtains expressly and solely in connection with the individuals service as a member of the New Mirion Board. | |
315
Table of Contents
| GSAH |
New Mirion |
|
| opportunity to GSAH without violating any legal obligation | ||
| Special Stockholder Meetings |
||
| Subject to the rights of any holders of preferred stock, special meetings of GSAH stockholders, for any purpose or purposes, may be called only by the (i) chairman of the GSAH Board, (ii) the chief executive officer (or any co-chief executive officer, if applicable) or (iii) the GSAH Board pursuant to a resolution adopted by a majority of the GSAH Board. | Subject to the rights of any holders of preferred stock, special meetings of New Mirion stockholders, for any purpose or purposes, may be called only by (i) the New Mirion Board pursuant to a resolution adopted by a majority of the New Mirion Board, (ii) the chairperson of the New Mirion Board or (iii) the chief executive officer. | |
| Notice of Stockholder Meetings |
||
| Written notice of each stockholders meeting, in a manner permitted in Section 232 of the DGCL, stating the place, if any, date, and time of the meeting, and the means of remote communication, if any, by which stockholders and proxy holders may be deemed to be present in person and vote at such meeting and the record date for determining the stockholders entitled to vote at the meeting (if such date is different from the record date for determining stockholders entitled to notice of the meeting) must be delivered not less than ten (10) nor more than sixty (60) days before the date of the meeting unless otherwise required by law.
Special Meetings. Written notice must also state the purpose for calling the meeting. The business transacted at such meeting is limited to the matters stated in the notice. |
Written notice of each stockholders meeting, in a manner provided in Section 232 of the DGCL, stating the place, if any, date, and time of the meeting, and the means of remote communication, if any, by which stockholders and proxy holders may be deemed to be present in person and vote at such meeting and the record date for determining the stockholders entitled to vote at the meeting (if such date is different from the record date for determining stockholders entitled to notice of the meeting) must be delivered not less than ten (10) nor more than sixty (60) days before the date of the meeting unless otherwise required by law.
Special Meetings. Written notice must also state the purpose for calling the meeting. The business transacted at such meeting is limited to the matters stated in the notice. |
|
| Stockholder Proposals (Other than Nomination of Persons for Election as Directors) |
||
| No business may be transacted at an annual meeting of stockholders, other than business that is either (i) specified in GSAHs notice of meeting (or any supplement thereto), (ii) otherwise properly brought before the annual meeting by or at the direction of the GSAH Board or (iii) otherwise properly brought before the annual meeting by any GSAH stockholder who is entitled to vote at the meeting and who complies with the notice procedures described below.
The GSAH stockholder must (i) give timely notice thereof in proper written form to the secretary of GSAH and (ii) the business must be a proper matter for stockholder action. To be timely, a stockholders notice must be received at GSAHs principal executive offices not later than the close of business on the ninetieth (90th) day nor earlier than the opening of business on the one-hundred twentieth (120th) day prior to the first anniversary of the preceding years annual meeting. However, if the annual meeting is |
No business may be conducted at an annual meeting of New Mirion stockholders, other than business that is either (i) specified in New Mirion notice of meeting, (ii) otherwise properly brought before the annual meeting by or at the direction of the New Mirion Board or (iii) otherwise properly brought before the annual meeting by any New Mirion stockholder who is entitled to vote at the meeting and who complies with the notice procedures described below.
The New Mirion stockholder must (i) give timely notice thereof in proper written form to the secretary of New Mirion and (ii) the business must be a proper matter for stockholder action. To be timely, a stockholders notice must be received at the New Mirions principal executive offices not sooner than the close of business on the one-hundred twentieth (120th) day nor later than the close of business on the one-hundred fiftieth (150th) day prior to the first |
|
316
Table of Contents
| GSAH |
New Mirion |
|
| more than thirty (30) days before or more than sixty (60) days after such anniversary date, the notice must be delivered no earlier than the close of business on the one-hundred twentieth (120th) day prior to such annual meeting and no later than the close of than the close of business on the later of the seventieth (90th) day prior to such annual meeting or the tenth (10th) day following the day on which public announcement of the date of the annual meeting is first made or sent by GSAH.
Additionally, the stockholder notice must set forth the information required by the advance notice provisions of the GSAH Bylaws. |
anniversary of the preceding years annual meeting. However, if the date of the annual meeting is more than thirty (30) days before or more than seventy (70) days after such anniversary date, the notice must be delivered no earlier than the close of business on the one-hundred twentieth (120th) day prior to such annual meeting and no later than the close of business on the later of the seventieth (70th) day prior to such annual meeting or the tenth (10th) day following the day on which public announcement of the date of the annual meeting is first made or sent by New Mirion.
Additionally, the stockholder notice must set forth the information required by advance notice provisions of the New Mirion Bylaws. |
|
| Stockholder Nominations of Persons for Election as Directors |
||
| Subject to any rights of holders of preferred stock to elect directors and the number of directors to be elected at the annual or special meeting, nominations of persons for election to the GSAH Board at any annual meeting of stockholders, or at any special meeting of stockholders called for the purpose of electing directors as set forth in GSAHs notice of such special meeting, may be made (i) by or at the direction of the GSAH Board or (ii) by any GSAH stockholder of record entitled to vote in the election of directors who comply with the notice procedures described below. |
Subject to any rights of holders of preferred stock to elect directors and the number of directors to be elected at the annual or special meeting, nominations of persons for election to the New Mirion Board at any annual meeting of stockholders, or at any special meeting of stockholders called for the purpose of electing directors as set forth in New Mirions notice of such special meeting, may be made (i) by or at the direction of the New Mirion Board or (ii) by any New Mirion stockholder of record entitled to vote in the election of directors who complies with the notice procedures described below. |
|
| To be timely, a stockholders notice must be received at GSAHs principal executive offices not later than the close of business on the ninetieth (90th) day nor earlier than the opening of business on the one-hundred twentieth (120th) day prior to the first anniversary of the preceding years annual meeting. However, if the annual meeting is more than thirty (30) days before or more than sixty (60) days after such anniversary date, the notice must be delivered no earlier than the close of business on the one-hundred twentieth (120th) day prior to such annual meeting and no later than the close of than the close of business on the later of the seventieth (90th) day prior to such annual meeting or the tenth (10th) day following the day on which public announcement of the date of the annual meeting is first made or sent by GSAH. | To be timely, a stockholders notice must be received at New Mirions principal executive offices not sooner than the close of business on the one-hundred twentieth (120th) day nor later than the close of business on the one-hundred fiftieth (150th) day prior to the first anniversary of the preceding years annual meeting. However, if the date of the annual meeting is more than thirty (30) days before or more than seventy (70) days after such anniversary date, the notice must be delivered no earlier than the close of business on the one-hundred twentieth (120th) day prior to such annual meeting and no later than the close of business on the later of the seventieth (70th) day prior to such annual meeting or the tenth (10th) day following the day on which public announcement of the date of the annual meeting is first made or sent by New Mirion. | |
| Limitation of Liability of Directors |
||
| The GSAH Certificate of Incorporation provides that no director will be personally liable, except to the extent an exemption from liability or limitation is not permitted under the DGCL. | The New Mirion Charter provides that no director will be personally liable, except to the extent an exemption from liability or limitation is not permitted under the DGCL. | |
317
Table of Contents
| GSAH |
New Mirion |
|
| Indemnification of Directors and Officers |
||
| Each of the GSAH Certificate of Incorporation and GSAH Bylaws provide that GSAH will indemnify its directors and officers to the fullest extent permitted by applicable law, if such officer or director is made a party or threatened to be made a party to any threatened, pending or completed action, suit or proceeding, whether civil, criminal, administrative or investigative, as a result of his or her involvement as a director or officer of GSAH or service at the request of GSAH as a director, officer, employee or agent for another entity.
GSAH will pay the expenses (including attorneys fees) incurred by an indemnitee in defending or otherwise participating in any proceeding in advance of its final disposition, provided that such person will repay any such advance if it is ultimately determined that such person is not entitled to indemnification by GSAH. GSAH has the burden of proving that the indemnitee is not entitled to the requested indemnification or to advancement of expenses under the GSAH Bylaws or applicable law
Except for proceedings to enforce rights to indemnification and advancement of expenses, GSAH will only indemnify and advance expenses for indemnitee-initiated proceedings if such proceeding was authorized by the GSAH Board. |
The New Mirion Charter provides that New Mirion will indemnify its directors and officers to the fullest extent permitted by applicable law, if such officer or director is made a party or threatened to be made a party to any threatened, pending or completed action, suit or proceeding, whether civil, criminal, administrative or investigative, as a result of his or her involvement as a director or officer of New Mirion or service at the request of New Mirion as a director, officer, employee or agent for another entity.
New Mirion will pay the expenses (including attorneys fees) incurred by an indemnitee in defending or otherwise participating in any proceeding in advance of its final disposition, provided that such person will repay any such advance if it is ultimately determined that such person is not entitled to indemnification by New Mirion. New Mirion has the burden of proving that the indemnitee is not entitled to the requested indemnification or to advancement of expenses under the New Mirion Charter or applicable law.
Except for proceedings to enforce rights to indemnification and advancement of expenses, New Mirion will only indemnify and advance expenses for indemnitee-initiated proceedings if such proceeding was authorized by the New Mirion Board. |
|
| The repeal or amendment to the GSAH Certificate of Incorporations indemnification provisions by a majority vote of the outstanding shares of GSAH stock or by changes in law will be prospective only, unless such change provides for broader indemnification rights on a retroactive basis. | The repeal or amendment to the New Mirion Charters indemnification provisions by a majority vote of the outstanding shares of New Mirion common stock or by changes in law will be prospective only, unless such change provides for broader indemnification rights on a retroactive basis. | |
| Dividends |
||
| Subject to applicable law and preferences that may be applicable to any outstanding preferred stock, holders of shares of GSAH Class A and GSAH Class B common stock are entitled to receive dividends and other distributions (payable in cash, property or capital stock of GSAH) declared by the GSAH Board out of assets or funds legally available. | Subject to applicable law and preferences that may be applicable to any outstanding preferred stock, holders of shares of New Mirion Class A and New Mirion Class B common stock are entitled to receive dividends (payable in cash, property or capital stock of New Mirion) declared by the New Mirion Board out of any assets or funds legally available. | |
| Liquidation |
||
| In the event of GSAHs liquidation, dissolution or winding up, holders of GSAH Class A and GSAH Class B common stock will be entitled to share ratably (on an as converted basis with respect to the GSAH Class B common stock) in the net assets legally available for distribution after the payment of all | In the event of New Mirions liquidation, dissolution or winding up, holders of New Mirion Class A and New Mirion Class B common stock will be entitled to share ratably in the net assets legally available for distribution after the payment of all New Mirions debts and other liabilities and the satisfaction of any | |
318
Table of Contents
| GSAH |
New Mirion |
|
| GSAHs debts and other liabilities and the satisfaction of any liquidation preferences granted to the holders of any then outstanding shares of preferred stock. | liquidation preference granted to the holders of any then outstanding shares of preferred stock. | |
| Preemptive Rights |
||
| There are no preemptive rights relating to GSAH Class A or GSAH Class B common stock. | There are no preemptive rights relating to New Mirion Class A or New Mirion Class B common stock. | |
| Anti-Takeover Provisions and Other Stockholder Protections |
||
| The GSAH Certificate of Incorporation includes certain anti-takeover and other stockholder protections such as a dual-class stock structure, prohibition on stockholder action by written consent and blank check preferred stock.
Section 203 of the DGCL prohibit a Delaware corporation from engaging in a business combination with an interested stockholder (i.e., a stockholder owning 15% or more of GSAH voting stock) for three years following the time that the interested stockholder becomes such, subject to certain exceptions. |
The New Mirion Charter includes certain anti-takeover and other stockholder protections such as a prohibition on stockholder action by written consent and blank check preferred stock. For additional information about New Mirions anti-takeover provisions and other stockholder protections, see Description of New Mirion Securities.
Section 203 of the DGCL prohibit a Delaware corporation from engaging in a business combination with an interested stockholder (i.e., a stockholder owning 15% or more of New Mirion voting stock) for three years following the time that the interested stockholder becomes such, subject to certain exceptions. |
|
| Inspection of Books and Records |
||
| Inspection. Under the DGCL, any stockholder or beneficial owner has the right, upon written demand under oath stating the proper purpose thereof, either in person or by attorney or other agent, to inspect and make copies and extracts from GSAHs stock ledger, list of stockholders and its other books and records for a proper purpose during the usual hours for business.
Voting List. GSAH must prepare, at least ten (10) days before every meeting of the stockholders, a complete list of the stockholders entitled to vote at the meeting. The list is open to the examination of any stockholder, for any purpose germane to the meeting at least ten (10) days prior to the meeting either (i) on a reasonably accessible electronic network or (ii) during ordinary business hours at GSAHs principal place of business. If the meeting is to be held at a place, then a list of stockholders entitled to vote at the meeting will be produced and kept at the time and place of the meeting during the whole time thereof and may be examined by any stockholder who is present. If a meeting of stockholders is to be held solely by means of remote communication, the list shall be open to the examination of any stockholder during the whole time of the meeting on a reasonably accessible electronic |
Inspection. Under the DGCL, any stockholder or beneficial owner has the right, upon written demand under oath stating the proper purpose thereof, either in person or by attorney or other agent, to inspect and make copies and extracts from New Mirions stock ledger, list of stockholders and its other books and records for a proper purpose during the usual hours for business.
Voting List. New Mirion must prepare, at least ten (10) days before every meeting of the stockholders, a complete list of the stockholders entitled to vote at the meeting. The list is open to the examination of any stockholder, for any purpose germane to the meeting at least ten (10) days prior to the meeting either (i) on a reasonably accessible electronic network or (ii) during ordinary business hours at New Mirions principal place of business. If the meeting is to be held at a place, then a list of stockholders entitled to vote at the meeting will be produced and kept at the time and place of the meeting during the whole time thereof and may be examined by any stockholder who is present. If a meeting of stockholders is to be held solely by means of remote communication, the list shall be open to the examination of any stockholder during the whole time of the meeting on a reasonably accessible |
|
319
Table of Contents
| GSAH |
New Mirion |
|
| network, and the information required to access such list shall be provided with the notice of meeting. | electronic network, and the information required to access such list shall be provided with the notice of meeting. | |
| Choice of Forum |
||
| The GSAH Certificate of Incorporation generally designates the Court of Chancery of the State of Delaware (the Court of Chancery) as the sole and exclusive forum for (i) any derivative action or proceeding brought on behalf of GSAH, (ii) any action asserting a claim of breach of a fiduciary duty owed by any director, officer, employee or agent of GSAH to GSAH or GSAH Stockholders, or any claim for aiding and abetting any such alleged breach, (iii) any action asserting a claim against GSAH, its directors, officers or employees arising pursuant to any provision of the DGCL, the GSAH Certificate of Incorporation or the GSAH Bylaws or (iv) any action asserting a claim against GSAH, its directors, officers or employees governed by the internal affairs doctrine. |
The New Mirion Charter generally designates, unless New Mirion otherwise consents in writing, the Court of Chancery as the sole and exclusive forum for (i) any derivative action or proceeding brought on New Mirions behalf, (ii) any action asserting a claim of breach of a fiduciary duty owed by any of New Mirions current or former directors, officers or other employees to New Mirion or its stockholders, (iii) any action asserting a claim against New Mirion or any current or former director, officer or other employee of New Mirion arising out of or pursuant to any provision of the DGCL, the New Mirion Charter or the New Mirion Bylaws; (iv) any action to interpret, apply, enforce, or determine the validity of the New Mirion Charter or the New Mirion Bylaws, (v) any claim or cause of action as to which the DGCL confers jurisdiction on the Court of Chancery of the State of Delaware, and (vi) any other action asserting a claim that is governed by the internal affairs doctrine. |
|
| This provision applies unless GSAH otherwise consents in writing or the action is one (A) as to which the Court of Chancery determines there is an indispensable party not subject to the Court of Chancerys jurisdiction and the indispensable party does not consent, (B) which another court or courts has exclusive jurisdiction over the action, including suits brought to enforce any liability or duty created by the Exchange Act or (C) that arises under the federal securities laws, including the Securities Act, as to which the Court of Chancery and the U.S. federal district court for the District of Delaware shall have concurrent jurisdiction.
If an action is brought outside of Delaware, the stockholder bringing the suit will be deemed to have consented to service of process on such stockholders counsel. |
This provision applies unless New Mirion otherwise consents in writing or the action is one (A) as to which the Court of Chancery determines there is an indispensable party not subject to the Court of Chancerys jurisdiction and the indispensable party does not consent or (B) which another court or courts has exclusive jurisdiction over the action, including suits brought to enforce any liability or duty created by the Exchange Act.
The New Mirion Charter also provides that, unless Mirion Consents in writing to the selection of alternative forum, to the fullest extent permitted by law, the federal district courts of the United States of America shall be the exclusive forum for the resolution of any complaint asserting a cause of action arising under the Securities Act, provided this provision does not apply to claims or causes of action brought to enforce a duty or liability created by the Exchange Act, or any other claim for which the federal courts have exclusive jurisdiction. Although the New Mirion Charter provides that the U.S. federal district courts will be the exclusive forum for resolving any complaint asserting a cause of action arising under the Securities Act, it is possible that a court could rule that such provisions are inapplicable for a particular claim or action or that such provisions are unenforceable. |
|
320
Table of Contents
SECURITIES ACT RESTRICTIONS ON RESALE OF SECURITIES
Pursuant to Rule 144 under the Securities Act (Rule 144), a person who has beneficially owned restricted shares of our common stock or our warrants for at least six months would be entitled to sell their securities provided that (1) such person is not deemed to have been an affiliate of us at the time of, or at any time during the three months preceding, a sale and (2) we are subject to the Exchange Act periodic reporting requirements for at least three months before the sale and have filed all required reports under Section 13 or 15(d) of the Exchange Act during the 12 months (or such shorter period as we were required to file reports) preceding the sale.
Persons who have beneficially owned restricted shares of our common stock or our warrants for at least six months but who are affiliates of us at the time of, or at any time during the three months preceding, a sale, would be subject to additional restrictions, by which such person would be entitled to sell within any three-month period only a number of securities that does not exceed the greater of:
| | 1% of the total number of shares of our common stock then outstanding; or |
| | the average weekly reported trading volume of our common stock during the four calendar weeks preceding the filing of a notice on Form 144 with respect to the sale. |
Sales by our affiliates under Rule 144 are also limited by manner of sale provisions and notice requirements and to the availability of current public information about us.
Restrictions on the Use of Rule 144 by Shell Companies or Former Shell Companies
Rule 144 is generally not available for the resale of securities initially issued by shell companies or issuers that have been at any time previously a shell company. However, Rule 144 also includes an important exception to this prohibition if the following conditions are met:
| | the issuer of the securities that was formerly a shell company has ceased to be a shell company; |
| | the issuer of the securities is subject to the reporting requirements of Section 13 or 15(d) of the Exchange Act; |
| | the issuer of the securities has filed all Exchange Act reports and material required to be filed, as applicable, during the preceding 12 months (or such shorter period that the issuer was required to file such reports and materials), other than Form 8-K reports; and |
| | at least one year has elapsed from the time that the issuer filed current Form 10 type information with the SEC reflecting its status as an entity that is not a shell company. |
As a result, the Sponsor will be able to sell its founder shares and private placement warrants, as applicable, pursuant to Rule 144 without registration one year after we have completed our initial business combination.
We anticipate that following the consummation of the Business Combination, we will no longer be a shell company, and so, once the conditions set forth in the exceptions listed above are satisfied, Rule 144 will become available for the resale of the above noted restricted securities as long as we continue to remain current in our Exchange Act reporting.
As of the date of this proxy statement/prospectus, we had 93,750,000 shares of our common stock outstanding. Of these shares, 75,000,000 shares sold in our IPO are freely tradable without restriction or further registration under the Securities Act, except for any shares purchased by one of our affiliates within the meaning of Rule 144 under the Securities Act. All of the 18,750,000 founder shares are restricted securities under Rule 144, in that they were issued in private transactions not involving a public offering. If the business combination is approved, the shares of our common stock we issue to the Mirion Stockholder pursuant to the Business Combination Agreement will be restricted securities for purposes of Rule 144.
321
Table of Contents
As of the date of this proxy statement/prospectus, there are approximately warrants to purchase 27,250,000 shares of GSAH Class A common stock outstanding, consisting of approximately 18,750,000 million public warrants originally sold as part of the units issued in the IPO and approximately 8,500,000 million private placement warrants that were sold by the Company to the Sponsor in a private placement prior to the IPO. Each warrant is exercisable for one share of GSAH Class A common stock, in accordance with the terms of the warrant agreement governing the warrants. The public warrants are freely tradable. In addition, we have agreed that as soon as practicable, but in no event later than 15 business days after the closing of our initial business combination, we will use our reasonable best efforts to file with the SEC, and within 60 business days following our initial business combination to have declared effective, a registration statement covering the issuance of the shares of GSAH Class A common stock issuable upon exercise of the warrants and to maintain a current prospectus relating to those shares of GSAH Class A common stock until the warrants expire or are redeemed; provided, that if GSAH Class A common stock is at the time of any exercise of a warrant not listed on a national securities exchange such that it satisfies the definition of a covered security under Section 18(b)(1) of the Securities Act, we may, at our option, require holders of public warrants who exercise their warrants to do so on a cashless basis in accordance with Section 3(a)(9) of the Securities Act and, in the event we so elect, we will not be required to file or maintain in effect a registration statement.
Registration Rights
See Proposal No.1 Approval of the Business CombinationRelated AgreementsAmended and Restated Registration Rights Agreement.
Listing of Securities
We intend to apply to continue the listing of GSAH Class A common stock and warrants on the NYSE under the symbols MIR and MIR.WS, respectively, upon the closing of the Business Combination, though such securities may not be listed, for instance if there is not a sufficient number of round lot holders. At the closing of the Business Combination, each unit will separate into its components consisting of one share of GSAH Class A common stock and one-fourth of one warrant.
322
Table of Contents
BENEFICIAL OWNERSHIP OF SECURITIES
The following table sets forth information regarding (1) the beneficial ownership of GSAH common stock as of March 31, 2021 (pre-Business Combination) and (2) the expected beneficial ownership of shares of New Mirion common stock immediately following consummation of the Business Combination (assuming a no redemption scenario and assuming a maximum redemption scenario as described below) by:
| | each person who is known to be the beneficial owner of more than 5% of GSAH common stock |
| | each person who is expected to be the beneficial owner of more than 5% of shares of New Mirion common stock post-Business Combination; |
| | GSAHs current executive officer and each of GSAHs current directors; |
| | each person who will become an executive officer or director of New Mirion post-Business Combination; and |
| | all executive officers and directors of GSAH as a group pre-Business Combination, and all executive officers and directors of New Mirion post-Business Combination. |
Beneficial ownership is determined according to the rules of the SEC, which generally provide that a person has beneficial ownership of a security if he, she or it possesses sole or shared voting or investment power over that security, including options and warrants that are currently exercisable or exercisable within 60 days (both before and after the Business Combination).
The beneficial ownership of GSAH common stock pre-Business Combination is based on 93,750,000 shares of common stock outstanding as of March 31, 2021, which includes an aggregate of 18,750,000 shares of GSAH Class B common stock outstanding as of such date.
The expected beneficial ownership of shares of our common stock post-Business Combination is based on our shares of common stock outstanding as of March 31, 2021 (except for those shares of common stock held by the Sellers, which are based on the shares of common stock outstanding as of October 31, 2021 due to the impact of the PIK Notes held by the Sellers as discussed below) and assumes two redemption scenarios as follows:
| | Assuming no redemptions: This presentation assumes that no shares of GSAH Class A common stock are redeemed. The no redemption scenario results in 213,798,777 shares of GSAH Class A common stock (including 18,750,000 founder shares subject to vesting upon certain Founder Share Vesting Events or forfeiture after five years if not vested) and 8,951,209 shares of GSAH Class B common stock for a total of 222,749,986 shares of GSAH common stock issued and outstanding following the Business Combination. |
| | Assuming maximum redemptions: This presentation assumes that the maximum number of shares of GSAH Class A common stock are redeemed such that the remaining funds held in the trust account after the payment of the redeeming shares pro-rata allocation along with the proceeds from the PIPE Investment and are sufficient to fund the Minimum Cash Condition (not less than $1,310 million available for use as cash consideration to the Sellers and to be retained on the balance sheet of the Combined Company). Based on the amount of $750.1 million in the trust account as of March 31, 2021, inclusive of accrued dividends, and considering the anticipated gross proceeds of approximately $900.0 million from the PIPE Investment, the aggregate commitment of $830 million from a first lien term facility pursuant to the Debt Commitment Letter and approximately $125.0 million from the Backstop Party, approximately 36,610,000 shares of GSAH Class A common stock may be redeemed and still enable us to have sufficient cash to satisfy the cash closing conditions in the Business Combination Agreement, including the debt refinancing of the Mirion Existing Credit Facility. The maximum redemption scenario results in 189,698,777 shares of GSAH Class A common stock (including 18,750,000 founder shares subject to vesting upon certain Founder Share Vesting Events or forfeiture after five years if not vested) and 8,951,209 shares of GSAH Class B common stock for a total of 198,649,986 shares of GSAH common stock issued and outstanding following the business combination. |
323
Table of Contents
Before the Closing of the Business Combination, the Sellers will have the option to elect to have their equity consideration exchanged for either shares of New Mirion Class A common stock or Paired Interests. New Mirion will own 100% of the voting shares (Class A) of IntermediateCo and greater than 80% of the non-voting Class B shares. We have assumed that of the existing Mirion stockholders, only Mirion management will elect to receive shares of New Mirion Class B common stock. For more information on the no redemption and maximum redemption scenarios, see Unauditd Pro Forma Condensed Combined Financial Information.
In addition, the consideration to be paid to the Mirion Sellers in connection with the Business Combination is allocated first in respect of the PIK Notes and second in respect of the Existing Mirion Shares. The PIK Notes accrue payment-in-kind (PIK) interest daily at a rate of 11.5% annually and are contemplated to be sold at the Closing for a price equal to the full outstanding principal amount together with all accrued but unpaid interest up to but excluding the Closing Date. See Certain Relationships and Related Persons TransactionsMirions Related Person TransactionsShareholder Notes. Accordingly, as the amount of accrued and unpaid interest increases, the holders of the PIK Notes will receive more consideration as compared to the holders of the Existing Mirion Shares. This has the effect over time of increasing the amount of shares of GSAH Class A Common Stock to be issued to the Mirion Sellers (excluding members of Mirion management) and reducing the number of shares of GSAH Class B common stock that will be issued to Mirion management (there is no incremental dilution to Public Stockholders). For purposes of the beneficial ownership described below, we have assumed an amount of principal and interest of the PIK Notes as if the Closing Date was October 31, 2021, but for all other purposes have assumed the Closing Date is March 31, 2021.
For example, in the no redemption scenario and assuming an amount of PIK Notes principal and interest to be outstanding as of October 31, 2021, the Charterhouse Parties would beneficially own 24.8 million shares of New Mirion Class A common stock, or 12% of the outstanding shares of New Mirion common stock; and all New Mirion directors and executive officers as a group (11 individuals) would beneficially own 5.8 million shares of New Mirion Class B common stock, or 3% of the outstanding shares of New Mirion common stock. In the no redemption scenario and assuming an amount of PIK Notes principal and interest to be outstanding as of September 30, 2021, the Charterhouse Parties would beneficially own 24.6 million shares of New Mirion Class A common stock, or 12% of the outstanding shares of New Mirion common stock; and all New Mirion directors and executive officers as a group would beneficially own 6.0 million shares of New Mirion Class B common stock, or 3% of the outstanding shares of New Mirion common stock. In addition, in the no redemption scenario and assuming an amount of PIK Notes principal and interest to be outstanding as of November 30, 2021, the Charterhouse Parties would beneficially own 25.0 million shares of New Mirion Class A common stock, or 12% of the outstanding shares of New Mirion common stock; and all New Mirion directors and executive officers as a group would hold 5.6 million shares of New Mirion Class B common stock, or 2.5% of the outstanding shares of New Mirion common stock.
324
Table of Contents
Unless otherwise indicated, GSAH believes that all persons named in the table below have sole voting and investment power with respect to the voting securities beneficially owned by them.
| Before the Business Combination | After the Business Combination | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| GSAH | Mirion TopCo | Assuming No Redemptions | Assuming Maximum Redemptions | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Name and Address |
Number of Shares of Class A Common Stock |
Approximate % of Class A Common Stock |
Number of Shares of Class B Common Stock |
Approximate % of Class B Common Stock |
Approximate % of Common Stock |
Number of Ordinary Shares |
Approximate % of Ordinary Shares |
Number of Shares of Class A Common Stock |
Approximate % of Class A Common Stock |
Number of Shares of Class B Common Stock |
Approximate % of Class B Common Stock |
Approximate % of Common Stock |
Number of Shares of Class A Common Stock |
Approximate % of Class A Common Stock |
Number of Shares of Class B Common Stock |
Approximate % of Class B Common Stock |
Approximate % of Common Stock |
|||||||||||||||||||||||||||||||||||||||||||||||||||
| Directors and Executive Officers Pre-Business Combination |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Raanan A. Agus |
| | | | | | | | | | | | | | | | | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Tom Knott |
| | | | | | | | | | | | | | | | | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Senator William Frist |
| | | | | | | | | | | | | | | | | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Steven S. Reinemund |
| | | | | | | | | | | | | | | | | |||||||||||||||||||||||||||||||||||||||||||||||||||
| David Robinson |
| | | | | | | | | | | | | | | | | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Martha Sullivan |
| | | | | | | | | | | | | | | | | |||||||||||||||||||||||||||||||||||||||||||||||||||
| All GSAH directors and executive officers as a group (six individuals) |
| | | | | | | | | | | | | | | |||||||||||||||||||||||||||||||||||||||||||||||||||||
| 5% Holders of GSAH (Other than Directors and Executive Officers) |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| GS Sponsor II LLC (our sponsor)(3)(4) |
| | 17,425,000 | (4) | 92.90 | % | 18.60 | % | | | 25,925,000 | 11.7 | % | 11.2 | % | 25,925,000 | 13.1 | % | 12.5 | % | ||||||||||||||||||||||||||||||||||||||||||||||||
| GSAM Holdings LLC(3)(4) |
| | 18,750,000 | (4) | 100 | % | 20 | % | | | 47,250,000 | 21.3 | % | 20.4 | % | 59,750,l000 | 30.1 | % | 28.8 | % | ||||||||||||||||||||||||||||||||||||||||||||||||
325
Table of Contents
| Before the Business Combination | After the Business Combination | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| GSAH | Mirion TopCo | Assuming No Redemptions | Assuming Maximum Redemptions | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Name and Address |
Number of Shares of Class A Common Stock |
Approximate % of Class A Common Stock |
Number of Shares of Class B Common Stock |
Approximate % of Class B Common Stock |
Approximate % of Common Stock |
Number of Ordinary Shares |
Approximate % of Ordinary Shares |
Number of Shares of Class A Common Stock |
Approximate % of Class A Common Stock |
Number of Shares of Class B Common Stock |
Approximate % of Class B Common Stock |
Approximate % of Common Stock |
Number of Shares of Class A Common Stock |
Approximate % of Class A Common Stock |
Number of Shares of Class B Common Stock |
Approximate % of Class B Common Stock |
Approximate % of Common Stock |
|||||||||||||||||||||||||||||||||||||||||||||||||||
| Alyeska Investment Group, |
3,978,203 | 5.30 | % | | | | | 11,478,203 | 5.4 | % | 5.0 | % | 11,478,203 | 6.1 | % | 5.8 | % | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Millennium Management LLC(6) |
5,469,279 | 7.30 | % | | | | | 5,469,279 | 2.6 | % | 2.4 | % | 5,469,279 | 2.9 | % | 2.8 | % | |||||||||||||||||||||||||||||||||||||||||||||||||||
| 5% Holders of New Mirion (Other than Directors and Executive Officers) |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Charterhouse Parties(7) |
| | | | | 4,445,212 | 65.0 | % | 24,818,173 | 11.6 | % | | | 11.1 | % | 24,818,173 | 13.1 | % | | | 12.5 | % | ||||||||||||||||||||||||||||||||||||||||||||||
| Directors and Executive Officers Post-Business Combination |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Thomas D. Logan(8) |
| | | | | 666,898 | 9.8 | % | | | 4,100,829 | 45.8 | 1.8 | % | | | 4,100,829 | 45.8 | % | 2.1 | % | |||||||||||||||||||||||||||||||||||||||||||||||
| Larry Kingsley |
| | | | | | | | | | | | | | | | | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Brian Schopfer |
| | | | | 123,792 | 1.8 | % | | | 761,210 | 8.5 | % | 0.3 | % | | | 761,210 | 8.5 | % | 0.4 | % | ||||||||||||||||||||||||||||||||||||||||||||||
| Michael Freed |
| | | | | 152,318 | 2.2 | % | | | 936,620 | 10.5 | % | 0.4 | % | | | 936,620 | 10.5 | % | 0.5 | % | ||||||||||||||||||||||||||||||||||||||||||||||
| Chris Warren |
| | | | | | | | | | | | | | | | | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Jo Natauri |
| | | | | | | | | | | | | | | | | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Steve Etzel |
| | | | | | | | | | | | | | | | | |||||||||||||||||||||||||||||||||||||||||||||||||||
| All New Mirion directors and executive officers as a group (11 individuals) |
| | | | | 943,008 | 13.8 | % | | | 5,798,659 | 64.8 | % | 2.6 | % | | | 5,798,659 | 64.8 | % | 2.9 | % | ||||||||||||||||||||||||||||||||||||||||||||||
326
Table of Contents
| (1) | Unless otherwise noted, the business address of each of the following entities or individuals pre-Business Combination is 200 West Street, New York, New York 10282 and post-Business Combination is 1218 Menlo Drive, Atlanta, Georgia 30318. |
| (2) | The calculations assume the number of Founder Shares held by such reporting person(s) are converted into shares of Class A common on a one-for-one basis and no other Founder Shares are then converted. The founder shares are subject to performance vesting. Holders of the founder shares are entitled to vote such founder shares and receive dividends and other distributions with respect to such founder shares prior to vesting, but such dividends and other distributions with respect to unvested founder shares will be set aside by the Company and shall only be paid to the holders of the founder shares upon the vesting of such founder shares. The founder shares will be forfeited to the Company for no consideration if they fail to vest within five years of the closing of the Business Combination. |
| (3) | GSAM Holdings LLC is the managing member of GS Sponsor II LLC. GSAM Holdings LLC is a wholly owned subsidiary of The Goldman Sachs Group, Inc. In addition to the shares held by our sponsor, GS Acquisition Holdings II Employee Participation LLC (Participation LLC), which is managed by a subsidiary of GSAM Holdings LLC, directly owns 1,325,000 founder shares. Each of GSAM Holdings LLC and The Goldman Sachs Group, Inc. may be deemed to beneficially own the shares held by GS Sponsor II LLC and Participation LLC by virtue of their direct and indirect ownership, as applicable, over GS Sponsor II LLC and Participation LLC. Each of GSAM Holdings LLC and The Goldman Sachs Group, Inc. disclaims beneficial ownership of any such shares except to the extent of their respective pecuniary interest therein. |
| (4) | Interests shown before the Business Combination for GSAM Sponsor and GSAM Holdings consist solely of founder shares. Such shares will automatically convert into shares of Class A common stock at the time of our Initial Business Combination, or earlier at the option of the holder, on a one-for-one basis, subject to adjustment. Interests shown following the Business Combination for GSAM Sponsor consist of (i) founder shares and (ii) 8,500,000 shares of GSAH Class A Common Stock underlying the private placement warrants. Interests shown following the Business Combination for GSAM Holdings consist of (i) founder shares, (ii) 8,500,000 shares of GSAH Class A Common Stock underlying the private placement warrants, (iii) 20,000,000 shares of GSAH Class A Common Stock to be issued to GSAM Holdings in connection with the PIPE Investment (subject to its right to syndicate such subscription) and (iv) in the maximum redemption scenario, 12,500,000 shares of GSAH Class A Common Stock in connection with the Backstop Agreement. |
| (5) | According to a Schedule 13G filed with the SEC on February 16, 2021 by Alyeska Investment Group, L.P., Alyeska Fund GP, LLC and Anand Parekh, each of Alyeska Investment Group, L.P., Alyeska Fund GP, LLC and Anand Parekh share voting and dispositive power with regard to 3,978,203 shares of Class A common stock of the Company as of December 31, 2020. The business address for each is 77 West Wacker Drive, 7th Floor, Chicago, IL 60601. Interests shown after the Business Combination include 7,500,000 shares of GSAH Class A Common Stock to be issued to Alyeska and its affiliated entities in connection with the PIPE Investment. |
| (6) | According to Amendment No. 1 to a Schedule 13G filed with the SEC on February 8, 2021 by Integrated Core Strategies (US) LLC (Integrated Core Strategies), Riverview Group LLC (Riverview Group), ICS Opportunities, Ltd. (ICS Opportunities), Millennium International Management LP (Millennium International Management), Millennium Management LLC (Millennium Management), Millennium Group Management LLC (Millennium Group Management) and Israel A. Englander (Mr. Englander), the parties reported that, as of December 31, 2020, Integrated Core Strategies was the beneficial owner of 3,119,279 shares of Class A common stock of the Company, that Riverview Group was the beneficial owner of 2,000,000 shares of Class A common stock of the Company and that ICS Opportunities was the beneficial owner of 350,000 shares of Class A common stock of the Company. Millennium International Management LP (Millennium International Management) is the investment manager to ICS Opportunities and may be deemed to have shared voting control and investment discretion over securities owned by ICS Opportunities. Millennium Management LLC (Millennium Management) is the general partner of the managing member of Integrated Core Strategies and Riverview Group and may be deemed to have shared voting control and investment discretion over securities owned by Integrated Core Strategies and Riverview Group. Millennium Management is also the general partner |
327
Table of Contents
| of the 100% owner of ICS Opportunities and may also be deemed to have shared voting control and investment discretion over securities owned by ICS Opportunities. Millennium Group Management LLC (Millennium Group Management) is the managing member of Millennium Management and may also be deemed to have shared voting control and investment discretion over securities owned by Integrated Core Strategies and Riverview Group. Millennium Group Management is also the general partner of Millennium International Management and may also be deemed to have shared voting control and investment discretion over securities owned by ICS Opportunities. The managing member of Millennium Group Management is a trust of which Israel A. Englander currently serves as the sole voting trustee. Therefore, Mr. Englander may also be deemed to have shared voting control and investment discretion over securities owned by Integrated Core Strategies, Riverview Group and ICS Opportunities. The address for the reporting persons principal business office is 666 Fifth Avenue, New York, New York 10103. |
| (7) | Represents 2,377,011 B Ordinary Shares held by CCP IX LP No. 1, 1,981,040 B Ordinary Shares held by CCP IX LP No. 2, 65,370 B Ordinary Shares held by CCP IX Co-investment LP and 21,791 B Ordinary Shares held by CCP IX Co-investment No. 2 LP (together, CCP IX). Charterhouse General Partners (IX) Ltd (CGP IX) is the general partner of each of the limited partnerships comprising CCP IX. Charterhouse Capital Partners LLP (CCP) acts as the investment adviser to CGP IX. CCPs advice with respect to investment decisions requires the approval of its Investment Committee comprised of 10 members, including the approval of CCPs Managing Partner, which is currently Lionel Giacomotto. However, it is CGP IX which ultimately makes all investment decisions. As a result, CGP IX may be deemed to have beneficial ownership of the securities held by the limited partnerships comprising CCP IX. CGP IX is managed by a five member board of directors. Each of the CGP IX board members disclaims beneficial ownership of the securities beneficially owned by each of the limited partnerships comprising CCP IX, except to the extent of their pecuniary interest therein, if any. The address for each of the foregoing persons principal business office is 6th Floor, Belgrave House, 76 Buckingham Palace Road, London, SW1W 9TQ. |
| (8) | Mr. Logans Mirion TopCo shares consist of (i) 245,666 A Ordinary Shares held by Mr. Logan; (ii) 3,031 B Ordinary Shares held by Mr. Logan; (iii) 139,400 A Ordinary Shares held by the J.P. Morgan Trust Company of Delaware in its capacity as Trustee of the Mary Hancock Logan GST Exempt Trust; (iv) 139,400 A Ordinary Shares held by the J.P. Morgan Trust Company of Delaware in its capacity as Trustee of the Alison Paige Logan GST Exempt Trust; and (v) 139,401 A Ordinary Shares held by the J.P. Morgan Trust Company of Delaware in its capacity as Trustee of the Thomas Darrell Logan, Jr. GST Exempt Trust. In both the no redemption and maximum redemption scenarios, Mr. Logans New Mirion shares consist of (i) 1,529,265 shares of Class B common stock held by Mr. Logan; (ii) 857,186 shares of Class B common stock held by the J.P. Morgan Trust Company of Delaware in its capacity as Trustee of the Mary Hancock Logan GST Exempt Trust; (iii) 857,186 shares of Class B common stock held by the J.P. Morgan Trust Company of Delaware in its capacity as Trustee of the Alison Paige Logan GST Exempt Trust; and (iv) 857,192 shares of Class B common stock held by the J.P. Morgan Trust Company of Delaware in its capacity as Trustee of the Thomas Darrell Logan, Jr. GST Exempt Trust. The J.P. Morgan Trust Company of Delaware in its capacity as Trustee of the foregoing trust entities has sole voting and dispositive power over the shares held by such trust entities. |
328
Table of Contents
CERTAIN RELATIONSHIPS AND RELATED PERSONS TRANSACTIONS
GSAH and Mirion describe below transactions and series of similar transactions, during GSAHs and Mirions last three fiscal years or that are currently proposed, to which GSAH or Mirion were a party or will be a party, in which:
| | the amounts involved exceeded or will exceed $120,000; and |
| | any of GSAHs or Mirions directors, executive officers or beneficial holders beneficial holders of more than 5% of any class of GSAHs or Mirions capital stock had or will have a direct or indirect material interest. |
GSAHs Related Person Transactions
Founder Shares
In July 2018, the Sponsor purchased an aggregate of 575 founder shares for an aggregate purchase price of $5,000. In April 2020 and June 2020, GSAH conducted stock splits, resulting in the Sponsor holding 20,125,000 founder shares, resulting in an effective purchase price per founder share of approximately $0.0003. The number of founder shares issued in the stock split was determined based on the expectation that the founder shares would represent 20% of the outstanding shares of common stock upon the completion of the IPO. Prior to the initial investment in GSAH of $5,000 by the Sponsor, GSAH had no assets, tangible or intangible. Following the partial exercise of the option to purchase additional shares, 1,375,000 founder shares were forfeited by the Sponsor on August 13, 2020, at no cost in order to maintain the number of founder shares equal to 20% of the outstanding shares of common stock, upon the completion of the initial public offering.
Private Placement Warrants
In connection with the completion of our initial public offering, the Sponsor purchased an aggregate of 8,500,000 private placement warrants, each exercisable to purchase one share of the GSAHs Class A common stock for $11.50 per share, at a price of $2.00 per private placement warrant, generating proceeds, before expenses, of $17,000,000. Subject to certain limited exceptions, the private placement warrants (including the Class A common stock issuable upon exercise of the private placement warrants) will not be transferable, assignable or salable until 30 days after the completion of our initial business combination and they will not be redeemable by us so long as they are held by the Sponsor or its permitted transferees. If GSAH does not complete an initial business combination by July 2, 2022, the proceeds of the sale of the private placement warrants will be used to fund the redemption of our public shares, subject to the requirements of applicable law, and the private placement warrants will expire worthless. Effective March 30, 2021, the Sponsor agreed not to transfer its Private Placement Warrants.
Related Party Notes
On April 17, 2020, an affiliate of the Sponsor agreed to loan GSAH an aggregate amount of up to $300,000 to be used to pay a portion of the expenses related to the initial public offering pursuant to a promissory note (the Note). The Note was non-interest bearing, unsecured and payable on the earlier of December 31, 2020 and the closing of the initial public offering. On May 28, 2020, GSAH borrowed $300,000 under the Note. On July 2, 2020, the full $300,000 balance of the Note was repaid.
On November 12, 2020, the Sponsor agreed to loan GSAH up to an aggregate of $2,000,000 pursuant to the working capital note (the Working Capital Note). Any amounts borrowed under the Working Capital Note are non-interest bearing, unsecured and are due at the earlier of the date GSAH is required to complete its Initial Business Combination pursuant to its amended and restated certificate of incorporation, as amended from time to time, and the closing of the Initial Business Combination. On March 12, 2021, GSAH borrowed $1,500,000 under the Working Capital Note.
329
Table of Contents
Sponsor Commitment
On March 11, 2019, the Sponsor provided GSAH with a commitment pursuant to which the Sponsor agreed that, if funds are needed by GSAH through June 12, 2020 to pay ordinary course expenses, the Sponsor would provide GSAH with liquidity of up to an aggregate of $2.0 million. The Sponsor will not receive any additional interest in GSAH in exchange for any such contribution and any liquidity provided under the commitment will be in the form of a contribution with respect to the Sponsors founder shares. This commitment was not exercised.
Administrative Services Agreement
GSAH entered into an agreement to pay an affiliate of the Sponsor a total of $10,000 per month for office space, utilities, administrative and support services. Upon the earlier of the completion of the initial business combination and our liquidation, this agreement will terminate and GSAH will cease paying these monthly fees.
Registration Rights
The holders of the founder shares, private placement warrants and warrants that may be issued upon conversion of working capital loans (and any shares of common stock issuable upon the exercise of the private placement warrants or warrants issued upon conversion of the working capital loans and upon conversion of the founder shares) are currently entitled to registration rights pursuant to the Existing Registration Rights Agreement. In connection with the Business Combination, the Existing Registration Rights Agreement will be amended and restated.
At the Closing, GSAH will enter into the Amended and Restated Registration Rights Agreement with the GS Holders and the Sellers, pursuant to which such parties will be entitled to registration rights in respect of certain shares of the Companys Class A common stock and certain other equity securities of GSAH that are held by the RRA Parties from time to time. For additional information, see Proposal No. 1Approval of the Business CombinationRelated AgreementsAmended and Restated Registration Rights Agreement.
Subscription Agreements
Concurrently with the execution of the Business Combination Agreement, GSAH entered into a Subscription Agreement with GSAM Holdings, pursuant to, and on the terms and subject to the conditions of which, GSAM subscribed for 20,000,000 PIPE Shares of the Companys Class A common stock for an aggregate purchase price equal to $200,000,000, subject to GSAMs rights to syndicate prior to the Closing. The PIPE Investment will be consummated substantially concurrently with Closing. The PIPE Investment will be consummated substantially concurrently with the closing of the Business Combination. For additional information, see Proposal No. 1Approval of the Business CombinationRelated AgreementsSubscription Agreements.
A&R Sponsor Agreement
In connection with the execution of the Business Combination Agreement, GSAH amended and restated that certain letter agreement, dated June 29, 2020, by and among GSAH, the Sponsor, GSAM Holdings, GS Employee Participation (collectively, the Insiders), pursuant to which, among other things, the Insiders agreed (i) to vote any shares of GSAHs securities in favor of the Business Combination and other Business Combination proposals, (ii) not to redeem any shares of the Companys Class A common stock or the Companys Class B common stock, in connection with the optional stockholder redemption, and (iii) certain transfer restrictions. For additional information, see Proposal No. 1Approval of the Business CombinationRelated AgreementsAmended and Restated Sponsor Agreement.
Backstop Agreement
In connection with the execution of the Business Combination Agreement, GSAM Holdings and GSAH have entered into the Backstop Agreement pursuant to which GSAM Holdings has committed to purchase from GSAH
330
Table of Contents
up to 12,500,000 shares of GSAHs Class A common stock at a price per share equal to $10.00 immediately prior to (and contingent upon) the Closing, solely to the extent necessary to fund any valid redemptions by the Companys stockholders that results in the Cash Shortfall being greater than zero dollars, contingent upon the terms and subject to the conditions set forth in the Backstop Agreement. For additional information, see Proposal No. 1Approval of the Business CombinationRelated AgreementsBackstop Agreement.
Option Agreement
In connection with the execution of the Business Combination Agreement, at the Closing, GSAM Holdings and certain of the Sellers will enter into the Option Agreement pursuant to which such Sellers will agree to, at the option of GSAM Holdings and subject to the conditions set forth in the Option Agreement, sell to GSAM Holdings up to 12,500,000 shares of GSAHs Class A common stock to be received by such Sellers pursuant to the Business Combination Agreement at the Closing at a price per share equal to $10.00 in cash, on the terms and subject to the conditions set forth in the Option Agreement. For additional information, see Proposal No. 1Approval of the Business CombinationRelated AgreementsOption Agreement.
GS Director Nomination Agreement
Upon the Closing, New Mirion and the Sponsor will also enter into a director nomination agreement, or the GS Director Nomination Agreement, that provides the Sponsor with a right to representation on the New Mirion Board. The GS Director Nomination Agreement will grant the Sponsor the ongoing right (but not the obligation) to appoint or nominate to the Board of Directors two (2) individuals, or the GS Sponsor Directors, to serve as director of New Mirion. As of the Closing, the Sponsor will designate [] and [] as the initial GS Sponsor Directors. The GS Director Nomination Agreement will terminate automatically when the Sponsor and GS Employee Participation, collectively with their respective affiliates, hold less than 50% of the founder shares held by them at the Closing, or upon the mutual written agreement of the parties.
Related Party Payments
Goldman Sachs & Co. LLC (Goldman Sachs), an affiliate of us and the Sponsor (Thomas R. Knott, Chief Executive Officer, Chief Financial Officer and Secretary and a director of GSAH, is a Managing Director of Goldman Sachs and Raanan A. Agus, one of our directors, is Participating Managing Directors of Goldman Sachs), acted as financial advisor to GSAH in connection with, and participated in certain of the negotiations leading to, the transaction contemplated by the Merger Agreement.
In connection with the Business Combination, an aggregate amount of approximately $33 million in deferred underwriting discount, advisory fees and placement agent fees, will be payable to Goldman Sachs & Co. LLC contingent upon completion of the Business Combination. Goldman Sachs & Co. LLC has provided certain financial advisory and/or underwriting services to the Company from time to time for which the Investment Banking Division of Goldman Sachs has received, and may receive, compensation, including having acted as sole bookrunner with respect to the initial public offering. During the two year period ended June 17, 2021, Goldman Sachs has not recognized any compensation for financial advisory and/or underwriting services provided by its Investment Banking Division to GSAH other than the upfront underwriting fees in connection with the Initial Public Offering.
In addition, Goldman Sachs (together with its affiliates) is a full service financial institution engaged in various activities, which may include sales and trading, commercial and investment banking, advisory, investment management, investment research, principal investing, hedging, market making, brokerage and other financial and non-financial activities and services. From time to time, Goldman Sachs and its affiliates have provided various investment banking and other commercial dealings unrelated to the Business Combination or the PIPE to Mirion, Charterhouse and their affiliates, and GSAH and its affiliates, and has received customary compensation in connection therewith. In addition, Goldman Sachs and its affiliates may provide investment banking and other
331
Table of Contents
commercial dealings to the Company, Mirion, Charterhouse and their respective affiliates in the future, for which they would expect to receive customary compensation.
In addition, in the ordinary course of its business activities, Goldman Sachs and its affiliates, officers, directors and employees may make or hold a broad array of investments and actively trade debt and equity securities (or related derivative securities) and financial instruments (including bank loans) for their own account and for the accounts of their customers. Such investments and securities activities may involve securities and/or instruments of the Company or Mirion, or their respective affiliates. Goldman Sachs and its affiliates may also make investment recommendations and/or publish or express independent research views in respect of such securities or financial instruments and may hold, or recommend to clients that they acquire, long and/or short positions in such securities and instruments.
Goldman Sachs Private Credit Funds, affiliates of GSAH, are a current lender to Mirion, holding $137.6mm of the USD Term Loan and 122.8mm of the EUR Term Loan under the Mirion Credit Agreement. GSAH intends to use a portion of the proceeds from the Business Combination, including the PIPE Investment, to repay the outstanding Mirion Credit Agreement and, as a result, such affiliates would receive their pro rata portion of such proceeds.
Related Party Policy
GSAH has adopted a code of ethics that requires us to avoid, wherever possible, all conflicts of interests, except under guidelines or resolutions approved by our Board (or the appropriate committee of our Board) or as disclosed in our public filings with the SEC. Under our code of ethics, conflict of interest situations will include any financial transaction, arrangement or relationship (including any indebtedness or guarantee of indebtedness) involving the company.
In addition, our audit committee is responsible for reviewing and approving related party transactions to the extent that GSAH enters into such transactions. An affirmative vote of a majority of the members of the audit committee present at a meeting at which a quorum is present will be required in order to approve a related party transaction. A majority of the members of the entire audit committee will constitute a quorum. Without a meeting, the unanimous written consent of all of the members of the audit committee will be required to approve a related party transaction. Our audit committee will review on a quarterly basis all payments that were made by GSAH to the Sponsor, officers or directors, or our or any of their affiliates.
These procedures are intended to determine whether any such related party transaction impairs the independence of a director or presents a conflict of interest on the part of a director, employee or officer.
To further minimize conflicts of interest, GSAH has agreed not to consummate an initial business combination with an entity that is affiliated with any of the Sponsor, officer or directors unless GSAH, or a committee of independent and disinterested directors, have obtained an opinion from an independent investment banking firm which is a member of the Financial Industry Regulatory Authority or an independent accounting firm that our initial business combination is fair to our company from a financial point of view. There will be no finders fees, reimbursements or cash payments made by us to the Sponsor, officer or directors or our or any of their respective affiliates, for services rendered to us prior to or in connection with the completion of our initial business combination, other than the following payments, none of which will be made from the proceeds of the initial public offering and the sale of the private placement warrants held in the trust account prior to the completion of our initial business combination:
| | payment to an affiliate of the Sponsor of a total of $10,000 per month, for up to 24 months, for office space, administrative and support services; |
| | reimbursement for any out-of-pocket expenses related to identifying, investigating and completing an initial business combination; |
332
Table of Contents
| | payment to Goldman Sachs of its underwriting commission, fees for any financial advisory, placement agency or other similar investment banking services provided by it to our company, and reimbursement of Goldman Sachs for any out-of-pocket expenses incurred by Goldman Sachs in connection with the performance of such services; and |
| | repayment of loans which may be made by the Sponsor, an affiliate of the Sponsor or our officer and directors to finance transaction costs in connection with an intended initial business combination, the terms of which have not been determined nor have any written agreements been executed with respect thereto. |
These payments may be funded using the net proceeds of the initial public offering and the sale of the private placement warrants not held in the trust account or, upon completion of the initial business combination, from any amounts remaining from the proceeds of the trust account released to us in connection therewith or from the PIPE Investment.
Mirions Related Person Transactions
IntermediateCo Charter
In connection with the Business Combination, we will form Mirion IntermediateCo, Inc., a new Delaware corporation, as a direct subsidiary (IntermediateCo). IntermediateCo will own, directly or indirectly, all of our operating subsidiaries. As the holder of 100% of the voting securities of IntermediateCo, we will have control over all of the affairs and decision making of IntermediateCo. As such, through our officers and directors, we will be responsible for all operational and administrative decisions of IntermediateCo and the day-to-day management of IntermediateCo. We will fund any dividends to our stockholders by causing IntermediateCo to make distributions to us and the holders of IntermediateCo Class B common stock (including us) on a ratable basis.
Under the IntermediateCo Charter, the holders of IntermediateCo Class B common stock will have the right, from and after the Closing (subject to the terms of the New Mirion Charter), to require IntermediateCo to redeem all or a portion of their IntermediateCo Class B common stock for, at our election, (1) newly issued shares of New Mirion Class A common stock on a one-for-one basis or (2) a cash payment equal to the product of the number of shares of IntermediateCo Class B common stock subject to redemption and the arithmetic average of the closing stock prices for a share of New Mirion Class A common stock for each of three (3) consecutive full trading days ending on and including the last full trading day immediately prior to the date of redemption (subject to customary adjustments, including for stock splits, stock dividends and reclassifications). If we decide to make a cash payment, the holder of IntermediateCo Class B common stock has the option to rescind its redemption request within a specified time period. The IntermediateCo Charter requires that we contribute, as applicable, cash or shares of our Class A common stock to IntermediateCo in exchange for an amount of newly-issued shares of IntermediateCo Class A common stock equal to the number of shares of IntermediateCo Class B common stock redeemed from the holders of IntermediateCo Class B common stock. IntermediateCo will then distribute the cash or shares of our Class A common stock to such holder of IntermediateCo Class B common stock to complete the redemption. In the event of a redemption request by a holder of IntermediateCo Class B common stock, we may, at our option, effect a direct exchange of cash or our Class A common stock for IntermediateCo Class B common stock in lieu of such a redemption. Shares of our Class B common stock will be canceled on a one-for-one basis if we, following a redemption request of a holder of IntermediateCo Class B common stock, redeem or exchange such holders IntermediateCo Class B common stock pursuant to the terms of the IntermediateCo Charter.
If at any time we issue a share of our Class A common stock or any other equity security with economic rights, the net proceeds received by us with respect to such share, if any, shall be concurrently contributed to IntermediateCo and IntermediateCo shall issue to us one share of IntermediateCo Class A common stock (or a corresponding other equity security of IntermediateCo), unless such share was issued by us solely to fund the
333
Table of Contents
purchase of a share of IntermediateCo Class B common stock from a holder of IntermediateCo Class B common stock (upon an election by us to exchange such IntermediateCo Class B common stock in lieu of redemption following a redemption request by such holder of IntermediateCo Class B common stock), in which case such net proceeds shall instead be transferred to the selling holder of IntermediateCo Class B common stock as consideration for such purchase, and IntermediateCo will not issue an additional share of Class A common stock to us. Similarly, (i) IntermediateCo may not issue any additional shares of its Class A common stock or Class B common stock to us or any of our subsidiaries unless substantially simultaneously therewith we or any of our subsidiaries issue or sell an equal number of shares of our Class A common stock, (ii) IntermediateCo may not issue any additional shares of its Class B common stock to any person other than us or any of our subsidiaries unless substantially simultaneously therewith we issue or sell an equal number of shares of our Class B common stock to such person and (iii) IntermediateCo may not issue any other equity securities to us or any of our subsidiaries unless substantially simultaneously therewith, we or such subsidiary issues or sells, to another person, an equal number of shares of a new class or series of equity securities of us or such subsidiary with substantially the same rights to dividends and distributions (including distributions upon liquidation) and other economic rights as those of such equity securities of IntermediateCo. Conversely, if at any time any shares of our Class A common stock are redeemed, purchased or otherwise acquired by us or any of our subsidiaries, IntermediateCo will substantially simultaneously therewith redeem, purchase or otherwise acquire an equal number of shares of its common stock held by us or our subsidiaries, upon the same terms and for the same price per security, as the shares of our Class A common stock are redeemed, purchased or otherwise acquired. In addition, IntermediateCo will not effect any subdivision (by any unit split, unit distribution, reclassification, reorganization, recapitalization or otherwise) or combination (by reverse unit split, reclassification, reorganization, recapitalization or otherwise) of its common stock unless it is accompanied by a substantively identical subdivision or combination, as applicable, of each class of our common stock, and we will not effect any subdivision or combination of any class of our common stock unless it is accompanied by a substantively identical subdivision or combination, as applicable, of the IntermediateCo common stock.
The IntermediateCo Charter will provide that, in the event that a tender offer, share exchange offer, issuer bid, take-over bid, recapitalization or similar transaction with respect to our Class A common stock is proposed by us or to us and our stockholders and approved by our board of directors or is otherwise consented to or approved by our board of directors, the holders of paired interests comprised of shares of IntermediateCo Class B common stock and shares of our Class B common stock will be permitted to participate in such offer by delivery of a notice of redemption or exchange that is effective immediately prior to the consummation of such offer. In the case of any such offer proposed by us, we are obligated to use our commercially reasonable efforts to enable and permit the holders of such paired interests to participate in such offer to the same extent or on an economically equivalent basis as the holders of shares of our Class A common stock without discrimination. In addition, we are obligated to use our reasonable efforts to ensure that the holders of such paired interests may participate in each such offer without being required to redeem or exchange IntermediateCo Class B common stock.
The IntermediateCo Charter will provide that, except for transfers to us as provided above or to certain permitted transferees, the IntermediateCo Class B common stock may not be sold, transferred or otherwise disposed of.
Subject to certain exceptions, IntermediateCo will indemnify all of its directors and officers and other related parties, against all losses or expenses arising from claims or other legal proceedings in which such person (in its capacity as such) may be involved or become subject to in connection with IntermediateCos business or affairs or the IntermediateCo Charter or any related document.
Director Nomination Agreements
Upon the Closing, New Mirion and the Charterhouse Parties, will enter into a director nomination agreement, or the Charterhouse Director Nomination Agreement, that provides the Charterhouse Parties with a right to representation on the New Mirion Board. The Charterhouse Director Nomination Agreement will grant the Charterhouse Parties the ongoing right (but not the obligation) to appoint or nominate to the Board of Directors
334
Table of Contents
one (1) individual, or the Charterhouse Director, to serve as director of New Mirion. The Charterhouse Parties have designated Chris Warren as the initial Charterhouse Director. The Charterhouse Director Nomination Agreement will terminate automatically when the Charterhouse Parties, collectively with its respective affiliates, holds less than 5% of the then outstanding common stock of New Mirion, or upon the mutual written agreement of the parties.
Upon the Closing, New Mirion and the Sponsor will also enter into a director nomination agreement, or the GS Director Nomination Agreement, that provides the Sponsor with a right to representation on the New Mirion Board. The GS Director Nomination Agreement will grant the Sponsor the ongoing right (but not the obligation) to appoint or nominate to the Board of Directors two (2) individuals, or the GS Sponsor Directors, to serve as director of New Mirion. As of the Closing, GS Sponsor will designate Larry Kingsley and Jo Natauri as the initial GS Sponsor Directors. The GS Director Nomination Agreement will terminate automatically when the Sponsor and GS Employee Participation, collectively with their respective affiliates, hold less than 50% of the founder shares held by them at the Closing, or upon the mutual written agreement of the parties.
Shareholder Notes
UKTopco has issued certain shareholder notes, or the Shareholder Notes, to certain of its shareholders and members of Mirion management. All outstanding principal is due on March 31, 2026. The Shareholder Notes accrue payment-in-kind (PIK) interest daily at a rate of 11.5% annually with such annual amount added to the outstanding principal amount on December 31 of each year in arrears. The Shareholder Notes can be prepaid without penalty at UKTopcos option and are subordinate in right of payment to any indebtedness of UKTopco subsidiaries to banks or to other financial institutions (either currently existing or to occur in the future). Certain of the Shareholder Notes have been admitted to trading on The International Stock Exchange, or TISE. The Shareholder Notes may be redeemed, in whole or in part, at the option of UKTopco by paying to the noteholder the full outstanding principal amount together with accrued but unpaid interest up to but excluding the redemption date. All of the Shareholder Notes are contemplated to be repaid in connection with the closing of the Business Combination for the redemption price.
At March 31, 2021 and June 30, 2020, there were $976.9 million in Shareholder Notes issued and outstanding. At June 30, 2019, there were $877.3 million in Shareholder Notes issued and outstanding.
Management Notes
UKTopco has also issued certain notes, or the Management Notes, to Thomas D. Logan, Mirions Chairman and Chief Executive Officer. The terms of the Management Notes are substantially similar to the Shareholder Notes, except that 5.75% interest is payable in cash on December 31 of each year. The Management Notes have been admitted to trading on the TISE. At June 30, 2020 and 2019, there were $976.9 million and $877.3 million in Management Notes issued and outstanding. All of the Management Notes are contemplated to be repaid in connection with the closing of the Business Combination for the redemption price.
Investment Agreement and Co-Investment Agreement
In November 2014, Mirion entered into an investment agreement, or the Investment Agreement, with certain holders of our Management Notes, Shareholder Notes, A Ordinary Shares and B Ordinary Shares, including certain members of the Mirion management team, or the Managers, certain investors affiliated with the Charterhouse Holders, or the Investors, and certain of Mirions subsidiaries. The Managers and the Investors are collectively referred to as the Ordinary Shareholders. The Investment Agreement provides the Ordinary Shareholders with certain registration rights, including the right to demand that Mirion file a registration statement and certain piggyback rights with respect to including their shares part of a registration statement that Mirion would otherwise file. The Investment Agreement also provides the Investors with certain information rights. The Investment Agreement also provides our Ordinary Shareholders with a right of first refusal with regard to certain issuances of our equity securities, which will not apply to, and will terminate upon, the
335
Table of Contents
consummation of the Business Combination. Finally, a majority of the Investors may from time to time appoint to, and remove from, the board of directors three non-executive directors; however, this board designation right will terminate upon the consummation of the Business Combination.
In June 2016, we entered into an amended and restated co-investment agreement, or Co-Investment Agreement, with the Managers, the Investors, additional investors, or Co-Investors, and certain of our subsidiaries, including Mirion Technologies, Inc., in order to finance the acquisition of Canberra Industries, Inc. and Canberra France. The terms of the Co-Investment Agreement are substantially similar to those of the Investment Agreement, however, there is no board designation right under the Co-Investment Agreement.
We anticipate that the Investment Agreement and the Co-Investment Agreement will be terminated prior to the Closing of the Business Combination.
Director and Executive Officer Compensation
Please see Executive Compensation for information regarding the compensation of Mirions directors and executive officers.
Employee Agreements
Mirion has entered into employment agreements with Mirions executive officers. For more information regarding these agreements, see Excecutive Compensation.
Indemnification Agreements and Directors and Officers Liability Insurance
Mirion previously entered into indemnification agreements with Mirions current directors and certain of Mirions executive officers. Mirion previously obtained an insurance policy that insures certain of Mirions directors and officers against certain liabilities. Upon the consummation of the Business Combination, New Mirion will enter into new indemnification agreements each of New Mirions directors and certain of New Mirions executive officers. New Mirion will also obtain a new insurance policy that will insure each of its directors and certain executive officers against certain liabilities.
Amended and Restated Registration Rights Agreement
At the Closing, New Mirion will enter into the Amended and Restated Registration Rights Agreement, or the RRA, with the Sponsor, GS Employee Participation, GSAM Holdings, or collectively, the GS Holders, the Charterhouse Holders and all of the other pre-Business Combination shareholders of Mirion, or collectively, with each other person who has executed and delivered a joinder thereto, the RRA Parties, pursuant to which the RRA Parties will be entitled to registration rights in respect of New Mirions Class A common stock held by the RRA Parties, or issuable upon redemption of shares of IntermediateCo Class B common stock or upon exercise of warrants to purchase shares of New Mirion Class A common stock held by them, in each case at the closing of the Business Combination (these securities are collectively referred to as the Registrable Securities). In addition, the Charterhouse Holders will be entitled to registration rights on any outstanding shares of New Mirion common stock acquired by them following the Closing to the extent such securities are restricted securities or control securities within the meaning of Rule 144 under the Securities Act. See Proposal No.1 Approval of the Business CombinationRelated AgreementsAmended and Restated Registration Rights Agreement.
General Lock-Up Restrictions
Pursuant to the RRA, after the completion of the Business Combination, holders of any shares of New Mirion common stock or paired interests received by such holder as consideration pursuant to the Business Combination Agreement (such holders, the Target Shareholders, and such common stock or paired interests, the Lock-Up Securities) will be subject to certain transfer restrictions (the Lock-Up Restrictions). Pursuant to the Lock-Up Restrictions, Target Shareholders may not (a) sell, assign, offer to sell, contract or agree to sell, hypothecate, pledge, grant any option to purchase or otherwise dispose of or agree to dispose of, directly or indirectly, or
336
Table of Contents
establish or increase a put equivalent position or liquidation with respect to or decrease a call equivalent position within the meaning of Section 16 of the Exchange Act with respect to, any security, (b) enter into any hedging, swap or other arrangement that transfers to another, in whole or in part, any of the economic consequences of ownership of any security, whether any such transaction is to be settled by delivery of such securities, in cash or otherwise, or (c) publicly announce any intention to effect any transaction specified in clause (a) or (b) (each of clauses (a), (b) and (c), a Transfer), until the 180th day after the Closing Date (the Lock-Up Period).
Notwithstanding the foregoing, the Lock-Up Securities may be transferred during the Lock-Up Period:
| (i) | by will, other testamentary document or intestacy; |
| (ii) | as a bona fide gift or gifts, including to charitable organizations or for bona fide estate planning purposes; |
| (iii) | to any trust for the direct or indirect benefit of the Target Shareholder or the immediate family of the Target Shareholder, or if the Target Shareholder is a trust, to a trustor or beneficiary of the trust or to the estate of a beneficiary of such trust; |
| (iv) | to a partnership, limited liability company or other entity of which such Target Shareholder and the immediate family of such Target Shareholder are the legal and beneficial owner of all of the outstanding equity securities or similar interests; |
| (v) | if the Target Shareholder is a corporation, partnership, limited liability company, trust or other business entity, (A) to another corporation, partnership, limited liability company, trust or other business entity that is an affiliate (as defined in Rule 405 promulgated under the Securities Act) of such Target Shareholder, or to any investment fund or other entity controlling, controlled by, managing or managed by or under common control with such Target Shareholder or affiliates of such Target Shareholder (including, for the avoidance of doubt, where such Target Shareholder is a partnership, to its general partner or a successor partnership or fund, or any other funds managed by such partnership), or (B) as part of a distribution to members or shareholders of such Target Shareholder; |
| (vi) | to a nominee or custodian of any person or entity to whom a Transfer would be permissible under clauses (i) through (v) above; |
| (vii) | in the case of an individual, by operation of law, such as pursuant to a qualified domestic order, divorce settlement, divorce decree, separation agreement or related court order; |
| (viii) | with the prior written consent of the Board (subject to the determination of the Board in its sole discretion at any time); provided such consent must be approved by each of the Charterhouse Director (unless waived by the Charterhouse Holders) and the GS Directors (unless waived by the GS Sponsor Member); |
| (ix) | from an employee or a director of, or a service provider to, the New Mirion or any of its subsidiaries upon the death, disability or termination of employment or services, in each case, of such person; |
| (x) | pursuant to a bona fide third-party tender offer, merger, consolidation or other similar transaction that is approved by the Board and made to all holders of shares of the Mirions capital stock involving a Change of Control (as defined below) (including negotiating and entering into an agreement providing for any such transaction), provided that in the event that such tender offer, merger, consolidation or other similar transaction is not completed, the Target Shareholders Lock-Up Securities shall remain subject to the Lock-Up Restrictions; and |
| (xi) | to the GS Equity Investor pursuant to the Option Agreement (as defined in the Business Combination Agreement) |
provided that (x) in the case of any Transfer of Lock-Up Securities pursuant to clauses (i) through (vi), (1) such Transfer shall not involve a disposition for value; (2) the Lock-Up Securities shall remain subject to the Lock-Up
337
Table of Contents
Restrictions and the transferee shall sign a joinder to the Amended and Restated Registration Rights Agreement before such Transfer is effective; (3) any required public report or filing (including filings under Section 16(a) of the Exchange Act), shall disclose the nature of such Transfer and that the Lock-Up Securities remain subject to the Lock-Up Restrictions; and (4) there shall be no voluntary public disclosure or other announcement of such Transfer; and (y) a Target Shareholder may enter into a trading plan established in accordance with Rule 10b5-1 under the Exchange Act during the Lock-Up Period so long as no Transfers are effected under such trading plan prior to the expiration of the Lock-Up Period and no voluntary public disclosure or announcement of such plan is made.
Charterhouse Demand Lock-Up Restrictions
Pursuant to the RRA, during the Lock-Up Period and during the Charterhouse Demand Period (together, the Charterhouse Demand Lock-Up Period), (a) the GS Founder Share Members will not be permitted to Transfer any shares of New Mirion common stock (other than to their Permitted Transferees) or request a Demand Registration and (b) the GS Equity Investor and its Affiliates may not Transfer any PIPE Shares, Backstop Shares or Option Shares (other than any such shares distributed to the GS Equity Investors Permitted Transferees) or request a Demand Registration (in each case of clauses (a) and (b), whether as part of a Shelf Registration, an unregistered transaction or otherwise). Provided, however, that the Charterhouse Demand Lock-Up Period shall be extended for any day during which the Registration Statement is not effective or sales pursuant to the Registration Statement are suspended.
Profits Interests
On June 16, 2021 and in connection with the Business Combination, the Sponsor issued 3,200,000 membership interests to Thomas Logan, 700,000 membership interests to Brian Schopfer and 4,200,000 membership interests to Lawrence Kingsley (collectively, the Profits Interests). The Profits Interests are intended to be treated as profits interests for U.S. income tax purposes, pursuant to which Messrs. Logan, Schopfer and Kingsley (the Award Holders) will have an indirect interest in the founder shares held by the Sponsor. The Profits Interests are subject to service and performance vesting conditions as described in this section, including the occurrence of the Closing, and do not fully vest until all of the applicable conditions are satisfied. In addition, the Profits Interests are subject to certain forfeiture conditions.
Fifty percent (50%) of the Profits Interests granted to each of Messrs. Logan and Schopfer service-vest on each of the second and third anniversaries of the Closing, and fifty percent (50%) of the Profits Interests granted to Mr. Kingsley service-vest on each of the first and second anniversaries of the Closing. All of the Profits Interests immediately service vest upon a change in control of GSAH, Mirion or any of their respective subsidiaries. If Messrs. Logan or Schopfers service (i) is terminated without cause or (ii) voluntarily ceases for good reason or his service terminates due to death or disability, 1/3, 2/3 and 100% respectively of the Profits Interests will become service-vested if the termination occurs before the respective first, second or third anniversaries of the Closing, and if Mr. Kingsleys service terminates due to death or disability, 1/3 and 100% of the Profits Interests will become service-vested if the termination occurs before the respective first or second anniversary of the Closing. If Messrs. Logan or Schopfer voluntarily ceases to provide services without good reason after the second anniversaries of the Closing. In addition, if a change in control of GSAH, Mirion or any of their respective subsidiaries occurs within six months immediately following such termination of employment, then the all of the outstanding Profits Interests will service-vest as of immediately prior to the change of control. and if Mr. Kingsley voluntarily ceases to provide services after the first anniversary of the Closing, an additional of each such Award Holders Profits Interests will service-vest in respect of each full quarter the Award Holder provided services since the most recent service-vesting date.
Twenty-five (25%) and seventy-five (75%) of Profits Interests granted to each of Messrs. Logan and Schopfer become vested with respect to the performance vesting condition on the first trading day after the Closing for which the volume weighted average price of GSAH Class A common stock is $14.00 or $16.00, respectively, for
338
Table of Contents
at least 20 of 30 consecutive trading days, if such date occurs on or before the fifth anniversary of the Closing, and seventy-five (75%) and twenty-five (25%) of Profits Interests granted to Mr. Kingsley performance-vest on the first trading day after the Closing for which the volume weighted average price of GSAH Class A common stock is $12.00 or $14.00, respectively, for at least 20 of 30 consecutive trading days, if such date occurs on or before the fifth anniversary of the Closing. In addition, if a change in control of GSAH, Mirion or any of their respective subsidiaries occurs at any time on or prior to the fifth anniversary of the Closing and the per share value received is at least equal to the specified price then the performance vesting conditions of the applicable Profits Interests will be deemed satisfied.
The Profits Interests granted to an Award Holder are forfeited entirely if (i) the Award Holders services terminate for cause, (ii) Messrs. Logan or Schopfer voluntarily ceases to provide services without good reason prior to the second anniversary of the Closing or Mr. Kingsley voluntarily ceases to provide services prior to the first anniversary of the Closing, (iii) an Award Holder voluntarily ceases to provide services where grounds for a termination for cause exist, (iv) the Business Combination does not close prior to November 30, 2021 (or such later date as mutually agreed by Mirion and GSAH) or (v) an Award Holder materially breaches a restrictive covenant agreement. In addition, 320,000, 70,000 and 420,000 of the Profits Interests issued to each of Messrs. Logan, Schopfer and Kingsley, respectively, are forfeited from the total number of Profits Interests granted to each Award Holder that fully vest (if any) if Mirion fails to implement the compliance work plan and remedy any such failures.
Executive Loans
Mirion has extended loans pursuant to individualized loan agreements, each dated as of June 2, 2020, with each of (i) Thomas Logan, with a principal amount of $528,005.98, (ii) Michael Freed, with a principal amount of $529,021.51 and (iii) Brian Schopfer, with an aggregate principal amount of $474,003.98 (collectively, the Executive Loans), each of which were intended to enable the applicable executive to acquire Class A ordinary shares of Mirion (the terms and circumstances of Mr. Schopfers Executive Loan are described further below). Each of the Executive Loans carries interest at a rate of 0.58% per annum. The Executive Loans will become repayable, together with any accrued but unpaid interest thereon, 10 business days prior to the Closing, or, if earlier, on the date that the applicable executive ceases employment with Mirion for any reason (or receives notice of termination of his employment with Mirion) or experiences a Borrower Insolvency Event (as such term is defined in the Executive Loan agreements). The Executive Loans may be prepaid, in whole or in part and together with any accrued but unpaid interest thereon, at any time without penalty. The Executive Loan agreements contemplate repayment by the executives by applying (i) 50% of any after-tax amount of the applicable executives annual cash bonus in respect of any given year and (ii) any cash proceeds received by the executive directly attributable to his transfer of any Class A ordinary shares of Mirion, which such amounts each executive has expressly agreed may be deducted by Mirion to discharge the applicable Executive Loan.
Mr. Schopfers Executive Loan agreement provides for two separate tranches of loans. The first tranche amends and restates the terms applicable to a loan that Mirion originally extended to Mr. Schopfer on May 16, 2019, which, as of June 2, 2020, had $173,047.84, comprising principal and accrued but unpaid interest, outstanding thereunder. The second tranche of Mr. Schopfers Executive Loan consists of a principal amount of $168,313.50, applied as consideration for Mr. Schopfers acquisition of 110,000 shares of Mirion Class A common stock Mirion Topco and $55,681.96 principal amount of loan notes, plus interest.
Mirion also extended a loan to Mr. Schopfer on September 24, 2019 to enable Mr. Schopfer to repay a sign-on bonus granted to him by a previous employer that became repayable when Mr. Schopfer resigned his employment with such former employer to commence employment with Mirion (the Schopfer Sign-On Bonus Loan), the terms of which were amended and restated pursuant to an amended loan agreement with Mirion executed on June 2, 2020. On June 2, 2020, $132,642.64 of the Schopfer Sign-On Bonus Loan, comprising principal and accrued but unpaid interest, was outstanding. The terms of the Schopfer Sign-On Bonus Loan are substantially similar to those of Mr. Schopfers Executive Loan, including that the Schopfer Sign-On Bonus Loan
339
Table of Contents
carries interest at a rate of 0.58% per annum and will become repayable 10 business days prior to the anticipated date of the Closing.
Exit Bonuses
Please see Executive CompensationExit Bonuses for information regarding cash bonuses that may become payable to Mirions executive officers upon the Closing.
Related Party Policy
Mirion as a private company does not have a formal written related party transaction policy. The post-business combination company will implement policies and procedures with respect to the approval of related party transactions in connection with the closing of the Business Combination.
GSAHs publicly-traded Class A common stock, units and warrants are currently listed on the NYSE under the symbols GSAH, GSAH.U and GSAH WS, respectively. We intend to apply to continue the listing of GSAH Class A common stock and warrants on the NYSE under the symbols MIR and MIR.WS, respectively, upon the closing of the Business Combination, though such securities may not be listed, for instance if there is not a sufficient number of round lot holders. At the closing of the Business Combination, each unit will separate into its components consisting of one share of GSAH Class A common stock and one-fourth of one warrant.
On June 16, 2021, the trading date before the public announcement of the Business Combination, GSAHs public units, Class A common stock and warrants closed at $10.43, $10.00 and $1.81, respectively. As of August 6, 2021, the most recent practicable date prior to the date of this proxy statement/prospectus, the most recent closing price for each unit, common stock and redeemable warrant was $10.45, $9.96 and $1.88, respectively.
Holders of the units, public shares and public warrants should obtain current market quotations for their securities. The market price of our securities could vary at any time before the Business Combination.
Historical market price information regarding Mirion is not provided because there is no public market for Mirions securities. For information regarding Mirions capital resources and liquidity, see Mirion Managements Discussion and Analysis of Financial Condition and Results of OperationsCapital Resources and Liquidity.
LEGAL MATTERS
Weil, Gotshal & Manges LLP, New York, New York, will pass upon the validity of the securities of Mirion Technologies, Inc. offered by this proxy statement/prospectus and certain other legal matters related to this proxy statement/prospectus.
EXPERTS
The financial statements of Mirion Technologies (TopCo), Ltd. and subsidiaries as of June 30, 2020 and 2019, and for each of the three years in the period ended June 30, 2020, included in this proxy statement/prospectus and the related financial statement schedules included elsewhere in the proxy statement/prospectus, have been audited by Deloitte & Touche LLP, an independent registered public accounting firm, as stated in their report
340
Table of Contents
appearing herein and elsewhere in the proxy statement/prospectus (which report expresses an unqualified opinion on the financial statements and financial statement schedules and includes an explanatory paragraph referring to significant transactions with related parties). Such financial statements and financial statement schedules have been so included in reliance upon the report of such firm given upon their authority as experts in accounting and auditing.
The financial statements of Sun Nuclear Corporation as of December 18, 2020 and for the period from January 1, 2020 to December 18, 2020, included in this proxy statement/prospectus have been audited by Deloitte & Touche LLP, independent auditors, as stated in their report appearing herein, and are included in reliance upon the report of such firm given upon their authority as experts in accounting and auditing.
The financial statements of GS Acquisition Holdings Corp II (the Company) as of December 31, 2020 and 2019 and for each of the two years in the period ended December 31, 2020 and for the period from May 31, 2018 (date of inception) to December 31, 2018 included in this proxy statement/prospectus have been so included in reliance on the report (which contains an explanatory paragraph relating to the Companys restatement of its financial statements as described in Note 2 and the disclosures related to warrant liabilities in Notes 2, 3, 4 and 7 to the financial statements) of Pricewaterhouse Coopers LLP, and independent registered accounting firm, given on the authority of said firm as experts in auditing and accounting.
Neither our stockholders nor our warrant holders have appraisal rights in connection with the Business Combination under the DGCL.
DELIVERY OF DOCUMENTS TO STOCKHOLDERS
Pursuant to the rules of the SEC, we and services that we employ to deliver communications to our stockholders are permitted to deliver to two or more stockholders sharing the same address a single copy of each of our annual report to stockholders and our proxy statement/prospectus. Upon written or oral request, we will deliver a separate copy of the annual report to stockholders or proxy statement/prospectus to any stockholder at a shared address to which a single copy of each document was delivered and who wishes to receive separate copies of such documents. GSAH stockholders receiving multiple copies of such documents may likewise request that we deliver single copies of such documents in the future. GSAH stockholders receiving multiple copies of such documents may request that we deliver single copies of such documents in the future. GSAH stockholders may notify us of their requests by calling or writing us at our principal executive offices at 200 West Street, New York, NY 10282 or (212) 902-1000.
341
Table of Contents
The transfer agent for our common stock is Continental Stock Transfer & Trust Company, N.A. and our warrant agent for our warrants is Continental Stock Transfer & Trust Company, N.A.
SUBMISSION OF STOCKHOLDER PROPOSALS
Our Board is aware of no other matter that may be brought before the Special Meeting. Under Delaware law, only business that is specified in the notice of Special Meeting to stockholders may be transacted at the Special Meeting.
For any proposal to be considered for inclusion in our proxy statement/prospectus and form of proxy for submission to the stockholders at our 2022 annual meeting of stockholders, it must be submitted in writing and comply with the requirements of Rule 14a-8 of the Exchange Act and our bylaws. Such proposals must be received by us at our executive offices a reasonable time before we begin to print and mail our 2022 annual meeting proxy materials in order to be considered for inclusion in our proxy materials for the 2022 annual meeting.
In addition, our bylaws provide notice procedures for stockholders to nominate a person as a director and to propose business to be considered by stockholders at a meeting. To be timely, a stockholders notice must be delivered to us at the principal executive offices of the Company not later than the close of business on the 90th nor earlier than the opening of business on the 120th day before the anniversary date of the immediately preceding annual meeting of stockholders; provided, however, that in the event that the annual meeting is called for a date that is not within 45 days before or after such anniversary date, notice by the stockholder to be timely must be so received no earlier than the opening of business on the 120th day before the meeting and not later than the later of (x) the close of business on the 90th day before the meeting or (y) the close of business on the 10th day following the day on which public announcement of the date of the annual meeting was first made by the Company. Nominations and proposals also must satisfy other requirements set forth in the bylaws. The chairman of our Board may refuse to acknowledge the introduction of any stockholder proposal not made in compliance with the foregoing procedures.
342
Table of Contents
WHERE YOU CAN FIND MORE INFORMATION
We file reports, proxy statement/prospectus and other information with the SEC as required by the Exchange Act. You can read the Companys SEC filings, including this proxy statement/prospectus, over the Internet at the SECs website at http://www.sec.gov. Those filings are also available free of charge to the public on, or accessible through, GSAHs corporate website under the heading SEC Filings, at http:// www.gsacquisition.com. GSAHs website and the information contained on, or that can be accessed through, the website is not deemed to be incorporated by reference in, and is not considered part of, this proxy statement/prospectus.
If you would like additional copies of this proxy statement/prospectus or if you have questions about the Business Combination or the proposals to be presented at the Special Meeting, you should contact the Company at the following address and telephone number:
GS Acquisition Holdings Corp II
200 West Street
New York, NY 10282
(212) 902-1000
You may also obtain these documents by requesting them in writing or by telephone from the Companys proxy solicitation agent at the following address and telephone number:
Innisfree M&A Incorporated
501 Madison Avenue, 20th Floor
New York, NY 10022
Shareholders may call toll free: (877) 456-3463
Banks and Brokers may call collect: (212) 750-5833
If you are a stockholder of the Company and would like to request documents, please do so by [●], 2021 (five business days prior to the date of the Special Meeting), in order to receive them before the Special Meeting. If you request any documents from us, we will mail them to you by first class mail, or another equally prompt means.
All information contained in this proxy statement/prospectus relating to the GSAH has been supplied by GSAH, and all such information relating to Mirion has been supplied by Mirion. Information provided by either GSAH or Mirion does not constitute any representation, estimate or projection of any other party.
This document is a proxy statement/prospectus of the Company for the Special Meeting. We have not authorized anyone to give any information or make any representation about the Business Combination, GSAH or Mirion that is different from, or in addition to, that contained in this proxy statement/prospectus. Therefore, if anyone does give you information of this sort, you should not rely on it. The information contained in this proxy statement/prospectus speaks only as of the date of this proxy statement/prospectus, unless the information specifically indicates that another date applies.
343
Table of Contents
GS ACQUISITION HOLDINGS CORP II
INDEX TO FINANCIAL STATEMENTS
| Page | ||||
| Index to Financial Statements |
||||
| F-6 | ||||
| F-7 | ||||
| F-8 | ||||
| F-9 | ||||
| F-10 | ||||
| Index to Financial Statements |
||||
| F-23 | ||||
| F-24 | ||||
| Unaudited Condensed Statements of Changes in Stockholders Equity |
F-25 | |||
| F-26 | ||||
| F-27 | ||||
F-1
Table of Contents
MIRION TECHNOLOGIES (TOPCO), LTD.
INDEX TO FINANCIAL STATEMENTS
| Page | ||||
| Consolidated Financial Statements (Audited) as of June 30, 2020 and 2019 and for the years ended June 30, 2020, 2019 and 2018 |
||||
| F-39 | ||||
| F-40 | ||||
| F-41 | ||||
| F-42 | ||||
| F-43 | ||||
| F-44 | ||||
| Unaudited Interim Condensed Consolidated Financial Statements as of March 31, 2021 and June 30, 2020 and for the nine months ended March 31, 2021 and 2020 |
||||
| F-88 | ||||
| Unaudited Interim Condensed Consolidated Statements of Operations |
F-89 | |||
| Unaudited Interim Condensed Consolidated Statements of Comprehensive Loss |
F-90 | |||
| Unaudited Interim Condensed Consolidated Statements of Stockholders Deficit |
F-91 | |||
| Unaudited Interim Condensed Consolidated Statements of Cash Flows |
F-92 | |||
| Notes to Unaudited Interim Condensed Consolidated Financial Statements |
F-93 | |||
F-2
Table of Contents
SUN NUCLEAR CORPORATION
INDEX TO FINANCIAL STATEMENTS
| Page | ||||
| Consolidated Financial Statements (Audited) as of December 18, 2020 and for the period from January 1, 2020 through December 18, 2020 |
||||
| F-112 | ||||
| F-113 | ||||
| F-114 | ||||
| Consolidated Statement of Cash Flows for the Period from January 1, 2020 through December 18, 2020 |
F-115 | |||
| F-116 | ||||
F-3
Table of Contents
GS Acquisition Holdings Corp II
Audited Financial Statements
| Report of Independent Registered Public Accounting Firm | F-5 | |||
| F-6 | ||||
| F-7 | ||||
| F-8 | ||||
| F-9 | ||||
| F-10 | ||||
F-4
Table of Contents
Report of Independent Registered Public Accounting Firm
To the Board of Directors and Stockholders of GS Acquisition Holdings Corp II
Opinion on the Financial Statements
We have audited the accompanying balance sheets of GS Acquisition Holdings Corp II (the Company) as of December 31, 2020 and 2019, and the related statements of operations, of changes in stockholders equity and of cash flows for each of the two years in the period ended December 31, 2020 and for the period from May 31, 2018 (date of inception) to December 31, 2018, including the related notes (collectively referred to as the financial statements). In our opinion, the financial statements present fairly, in all material respects, the financial position of the Company as of December 31, 2020 and 2019, and the results of its operations and its cash flows for each of the two years in the period ended December 31, 2020 and for the period from May 31, 2018 (date of inception) to December 31, 2018 in conformity with accounting principles generally accepted in the United States of America.
Restatement of Previously Issued Financial Statements
As discussed in Note 2 to the financial statements, the Company has restated its 2020 financial statements to correct errors.
Basis for Opinion
These financial statements are the responsibility of the Companys management. Our responsibility is to express an opinion on the Companys financial statements based on our audits. We are a public accounting firm registered with the Public Company Accounting Oversight Board (United States) (PCAOB) and are required to be independent with respect to the Company in accordance with the U.S. federal securities laws and the applicable rules and regulations of the Securities and Exchange Commission and the PCAOB.
We conducted our audits of these financial statements in accordance with the standards of the PCAOB. Those standards require that we plan and perform the audits to obtain reasonable assurance about whether the financial statements are free of material misstatement, whether due to error or fraud.
Our audits included performing procedures to assess the risks of material misstatement of the financial statements, whether due to error or fraud, and performing procedures that respond to those risks. Such procedures included examining, on a test basis, evidence regarding the amounts and disclosures in the financial statements. Our audits also included evaluating the accounting principles used and significant estimates made by management, as well as evaluating the overall presentation of the financial statements. We believe that our audits provide a reasonable basis for our opinion.
/s/ PricewaterhouseCoopers LLP
New York, New York
March 31, 2021, except for the effects of the restatement discussed in Note 2 and the disclosures related to warrant liabilities in Notes 2, 3, 4 and 7 to the financial statements, as to which the date is May 17, 2021
We have served as the Companys auditor since 2020.
F-5
Table of Contents
GS Acquisition Holdings Corp II
| December 31, 2020 (As Restated) |
December 31, 2019 |
|||||||
| ASSETS |
||||||||
| Current assets: |
||||||||
| Cash |
$ | 383,246 | $ | 5,000 | ||||
| Prepaid expenses |
599,170 | | ||||||
|
|
|
|
|
|||||
| Total current assets |
982,416 | 5,000 | ||||||
|
|
|
|
|
|||||
| Deferred tax asset |
265,954 | | ||||||
| Cash and cash equivalents held in Trust Account |
750,063,158 | | ||||||
| Accrued dividends receivable held in Trust Account |
3,883 | | ||||||
|
|
|
|
|
|||||
| Total assets |
$ | 751,315,411 | $ | 5,000 | ||||
|
|
|
|
|
|||||
| LIABILITIES AND STOCKHOLDERS EQUITY |
||||||||
| Current liabilities: |
||||||||
| Accounts payable |
$ | 965,370 | $ | 636 | ||||
| Accrued offering costs |
375,000 | | ||||||
| Income tax payable |
57 | | ||||||
| Warrant liability |
71,676,615 | | ||||||
|
|
|
|
|
|||||
| Total current liabilities |
73,017,042 | 636 | ||||||
| Deferred underwriting discount |
26,250,000 | | ||||||
|
|
|
|
|
|||||
| Total liabilities |
99,267,042 | 636 | ||||||
|
|
|
|
|
|||||
| Commitments and contingencies |
||||||||
| Class A common stock subject to possible redemption; 75,000,000 and -0- shares at redemption value at December 31, 2020 and December 31, 2019, respectively |
750,000,000 | | ||||||
| Stockholders equity: |
||||||||
| Preferred stock, $0.0001 par value, 5,000,000 shares authorized, none issued and outstanding at December 31, 2020 and December 31, 2019, respectively |
| | ||||||
| Class A common stock, $0.0001 par value, 500,000,000 shares authorized at December 31, 2020 and December 31, 2019, respectively |
| | ||||||
| Class B common stock, $0.0001 par value, 50,000,000 shares authorized, 18,750,000 and 20,125,000 issued and outstanding at December 31, 2020 and December 31, 2019, respectively |
1,874 | 2,012 | ||||||
| Additional paid-in capital |
| 2,988 | ||||||
| Accumulated deficit |
(97,953,505 | ) | (636 | ) | ||||
|
|
|
|
|
|||||
| Total stockholders equity/(deficit) |
(97,951,631 | ) | 4,364 | |||||
|
|
|
|
|
|||||
| Total liabilities and stockholders equity |
$ | 751,315,411 | $ | 5,000 | ||||
|
|
|
|
|
|||||
See accompanying notes to financial statements
F-6
Table of Contents
GS Acquisition Holdings Corp II
|
For the period from May 31, 2018 (date of inception) to December 31, 2018 |
||||||||||||
| For the Year Ended December 31, |
||||||||||||
| 2020 (As Restated) |
2019 | |||||||||||
| Dividend income |
$ | 67,041 | $ | | $ | | ||||||
| General and administrative expenses |
(2,449,094 | ) | (341 | ) | (295 | ) | ||||||
| Change in fair value of warrant liability |
(43,139,251 | ) | | | ||||||||
|
|
|
|
|
|
|
|||||||
| Loss before income taxes |
(45,521,304 | ) | (341 | ) | (295 | ) | ||||||
| Income tax benefit/(expense) |
265,897 | | | |||||||||
|
|
|
|
|
|
|
|||||||
| Net loss |
$ | (45,255,407 | ) | $ | (341 | ) | $ | (295 | ) | |||
|
|
|
|
|
|
|
|||||||
| Weighted average number of shares outstanding of Class A common stock |
37,397,260 | | | |||||||||
|
|
|
|
|
|
|
|||||||
| Basic and diluted net loss per share, Class A |
$ | (0.79 | ) | $ | | $ | | |||||
|
|
|
|
|
|
|
|||||||
| Weighted average number of shares outstanding of Class B common stock |
19,597,603 | 20,125,000 | 20,125,000 | |||||||||
|
|
|
|
|
|
|
|||||||
| Basic and diluted net loss per share, Class B |
$ | (0.79 | ) | $ | (0.00 | ) | $ | (0.00 | ) | |||
|
|
|
|
|
|
|
|||||||
See accompanying notes to financial statements
F-7
Table of Contents
GS Acquisition Holdings Corp II
STATEMENTS OF CHANGES IN STOCKHOLDERS EQUITY
| For the period from May 31, 2018 (date of inception) to December 31, 2018 |
Class A Common Stock | Class B Common Stock |
Additional Paid-in Capital |
Accumulated Deficit |
Stockholders Equity/(Deficit) |
|||||||||||||||||||||||
| Shares | Amount | Shares | Amount | |||||||||||||||||||||||||
| Sale of common shares to GS Sponsor II LLC at $0.0002 per share |
| $ | | 20,125,000 | $ | 2,012 | $ | 2,988 | $ | | $ | 5,000 | ||||||||||||||||
| Net loss |
| | | | | (295 | ) | (295 | ) | |||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||
| Balance, December 31, 2018 |
$ | | 20,125,000 | $ | 2,012 | $ | 2,988 | $ | (295 | ) | $ | 4,705 | ||||||||||||||||
| Net loss |
| | | | | (341 | ) | (341 | ) | |||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||
| Balance, December 31, 2019 |
| $ | | 20,125,000 | $ | 2,012 | $ | 2,988 | $ | (636 | ) | $ | 4,364 | |||||||||||||||
| Excess of cash received over fair value of private placement warrants |
| | | | 8,049,674 | | 8,049,674 | |||||||||||||||||||||
| Forfeiture of Founder Shares pursuant to partial exercise of underwritersover-allotment option |
| | (1,375,000 | ) | (138 | ) | 138 | | | |||||||||||||||||||
| Accretion for Class A common stock to redemption amount |
| | | | (8,052,800 | ) | (52,697,462 | ) | (60,750,262 | ) | ||||||||||||||||||
| Net loss |
| | | | | (45,255,407 | ) | (45,255,407 | ) | |||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||
| Balance, December 31, 2020 (As Restated) |
| $ | | 18,750,000 | $ | 1,874 | $ | | $ | (97,953,505 | ) | $ | (97,951,631 | ) | ||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||
See accompanying notes to financial statements
F-8
Table of Contents
GS Acquisition Holdings Corp II
| For the Year Ended December 31, | ||||||||||||
| 2020 (As Restated) |
2019 | 2018 | ||||||||||
| Cash flows from operating activities: |
||||||||||||
| Net loss |
$ | (45,255,407 | ) | $ | (341 | ) | $ | (295 | ) | |||
| Adjustments to reconcile net loss to net cash used in operating activities: |
||||||||||||
| Change in fair value of warrant liability |
43,139,251 | | | |||||||||
| Issuance costs related to warrant liability |
1,075,021 | | | |||||||||
| Change in operating assets and liabilities: |
||||||||||||
| Increase in dividend receivable |
(3,883 | ) | | | ||||||||
| Increase in prepaid expenses |
(599,170 | ) | | | ||||||||
| Increase in deferred tax assets |
(265,954 | ) | | | ||||||||
| Increase in accounts payable |
964,734 | 341 | 295 | |||||||||
| Increase in income tax payable |
57 | | | |||||||||
|
|
|
|
|
|
|
|||||||
| Net cash used for operating activities |
(945,351 | ) | | | ||||||||
|
|
|
|
|
|
|
|||||||
| Cash flows from financing activities: |
||||||||||||
| Proceeds from sale of Class B common stock to GS Sponsor II LLC |
| | 5,000 | |||||||||
| Proceeds from sale of Class A common stock to public |
750,000,000 | | | |||||||||
| Proceeds from sale of Private Placement Warrants |
17,000,000 | | | |||||||||
| Payment of underwriting discounts |
(15,000,000 | ) | | | ||||||||
| Payment of offering costs |
(613,245 | ) | | | ||||||||
| Proceeds from promissory note |
300,000 | | | |||||||||
| Repayment of promissory note |
(300,000 | ) | | | ||||||||
|
|
|
|
|
|
|
|||||||
| Net cash provided by financing activities |
751,386,755 | | 5,000 | |||||||||
|
|
|
|
|
|
|
|||||||
| Increase in cash and restricted cash |
750,441,404 | | 5,000 | |||||||||
| Cash and restricted cash and cash equivalents at beginning of year |
5,000 | 5,000 | | |||||||||
|
|
|
|
|
|
|
|||||||
| Cash and restricted cash and cash equivalents at end of year |
$ | 750,446,404 | $ | 5,000 | $ | 5,000 | ||||||
|
|
|
|
|
|
|
|||||||
| Supplemental disclosure of non-cash financing activities |
||||||||||||
| Accrued offering costs |
$ | 375,000 | $ | | $ | | ||||||
| Deferred underwriting discount |
$ | 26,250,000 | $ | | $ | | ||||||
See accompanying notes to financial statements
F-9
Table of Contents
GS ACQUISITION HOLDINGS CORP II
Note 1Description of Organization and Business Operations
Organization and General
GS Acquisition Holdings Corp II (the Company) was incorporated as a Delaware corporation on May 31, 2018. The Company was formed for the purpose of effecting a merger, capital stock exchange, asset acquisition, stock purchase, reorganization or similar business combination with one or more businesses (the Initial Business Combination). The Company is an emerging growth company, as defined in Section 2(a) of the Securities Act of 1933, as amended (the Securities Act), as modified by the Jumpstart Our Business Startups Act of 2012 (the JOBS Act).
All activity for the period from May 31, 2018 (Inception) through December 31, 2020 relates to the Companys formation and its initial public offering (the Public Offering) described below and identifying and evaluating prospective acquisition targets for an Initial Business Combination. The Company will not generate any operating revenues until after completion of its Initial Business Combination, at the earliest. The Company will generate non-operating income in the form of interest or dividend income on cash and cash equivalents from the proceeds derived from the Public Offering (as defined below in Note 4) and the Private Placement (as defined below in Note 5). The Company has selected December 31st as its fiscal year end.
Sponsor and Financing
The Companys sponsor is GS Sponsor II LLC, a Delaware limited liability company (the Sponsor).
The registration statement for the Companys Public Offering was declared effective by the United States Securities and Exchange Commission (the SEC) on June 29, 2020. On June 30, 2020, the underwriters partially exercised their option to purchase additional Units (as defined below in Note 4). The Companys Public Offering of 75,000,000 Units, including 5,000,000 Units pursuant to the underwriters partial exercise of such option, closed on July 2, 2020 (as described in Note 4). Upon the closing of the Public Offering and the Private Placement, $750,000,000 was placed in a U.S. based trust account (the Trust Account). The Company intends to finance its Initial Business Combination with the net proceeds from the Public Offering (as defined below in Note 4) and the sale of the Private Placement Warrants (as defined below in Note 5).
The Trust Account
The proceeds held in the Trust Account are invested in a money market fund registered under the Investment Company Act of 1940 as amended (the Investment Company Act) and meeting certain conditions under Rule 2a-7.
Except with respect to dividends earned on the funds held in the Trust Account that may be released to the Company to pay its taxes, the proceeds from the Public Offering and the Private Placement will not be released from the Trust Account until the earliest of: (i) the completion of the Initial Business Combination; (ii) the redemption of any public shares properly submitted in connection with a stockholder vote to amend the Companys amended and restated certificate of incorporation (A) to modify the substance or timing of the Companys obligation to allow redemptions in connection with the Initial Business Combination or to redeem 100% of its public shares if it does not complete the Initial Business Combination within 24 months from the closing of the Public Offering or (B) with respect to any other provision relating to stockholders rights or pre-Initial Business Combination activity; and (iii) the redemption of all of the Companys public shares if the Company has not completed the Initial Business Combination within 24 months from the closing of the Public Offering, subject to applicable law. The proceeds deposited in the Trust Account could become subject to the claims of the Companys creditors, if any, which could have priority over the claims of the Companys public stockholders.
F-10
Table of Contents
The balance in the Trust Account as of December 31, 2020 was $750,067,041, including $3,883 of accrued dividends.
Initial Business Combination
The Companys management has broad discretion with respect to the specific application of the net proceeds of the Public Offering, although substantially all of the net proceeds of the Public Offering and the Private Placement are intended to be generally applied toward consummating an Initial Business Combination. The Initial Business Combination must occur with one or more operating businesses or assets with a fair market value equal to at least 80% of the net assets held in the Trust Account (excluding the amount of any deferred underwriting discount). There is no assurance that the Company will be able to successfully effect an Initial Business Combination.
The Company, after signing a definitive agreement for an Initial Business Combination, will provide its public stockholders with the opportunity to redeem all or a portion of their shares upon the completion of the Initial Business Combination, either (i) in connection with a stockholder meeting called to approve the business combination or (ii) by means of a tender offer. However, in no event will the Company redeem its public shares in an amount that would cause its net tangible assets, after payment of deferred underwriting commissions, to be less than $5,000,001 following such redemptions. In such case, the Company would not proceed with the redemption of its public shares and the related Initial Business Combination, and instead may search for an alternate Initial Business Combination.
If the Company holds a stockholder vote or there is a tender offer for shares in connection with an Initial Business Combination, a public stockholder will have the right to redeem its shares for an amount in cash equal to its pro rata share of the aggregate amount then on deposit in the Trust Account, calculated as of two business days prior to the consummation of the Initial Business Combination, including interest but less taxes payable. As a result, such shares of Class A common stock are recorded at redemption amount and classified as temporary equity upon the completion of the Public Offering, in accordance with the Accounting Standards Codification (ASC) 480, Distinguishing Liabilities from Equity.
Pursuant to the Companys amended and restated certificate of incorporation, if the Company is unable to complete the Initial Business Combination within 24 months from the closing of the Public Offering, the Company will (i) cease all operations except for the purpose of winding up, (ii) as promptly as reasonably possible but no more than ten business days thereafter redeem the public shares, at a per-share price, payable in cash, equal to the aggregate amount then on deposit in the Trust Account, including interest (which interest shall be net of taxes payable, and less up to $100,000 of interest to pay dissolution expenses), divided by the number of then outstanding public shares, which redemption will completely extinguish public stockholders rights as stockholders (including the right to receive further liquidating distributions, if any), and (iii) as promptly as reasonably possible following such redemption, subject to the approval of the Companys remaining stockholders and the Companys board of directors, dissolve and liquidate, subject in each case to the Companys obligations under Delaware law to provide for claims of creditors and the requirements of other applicable law.
The Sponsor, Employee Participation LLC (as defined below in Note 5) and the Companys officers and directors have entered into a letter agreement with the Company, pursuant to which they have waived their rights to liquidating distributions from the Trust Account with respect to any Founder Shares (as defined below in Note 5) held by them if the Company fails to complete the Initial Business Combination within 24 months of the closing of the Public Offering, or during any extended time that the Company has to consummate an Initial Business Combination beyond 24 months as a result of a stockholder vote to amend its amended and restated certificate of incorporation. However, if the Sponsor, Employee Participation LLC or any of the Companys directors or officers hold any shares of Class A common stock in or after the Public Offering, they will be entitled to liquidating distributions from the Trust Account with respect to such shares if the Company fails to complete the Initial Business Combination within the prescribed time period.
F-11
Table of Contents
In the event of a liquidation, dissolution or winding up of the Company after an Initial Business Combination, the Companys stockholders are entitled to share ratably in all assets remaining available for distribution to them after payment of liabilities and after provision is made for each class of stock, if any, having preference over the common stock. The Companys stockholders have no preemptive or other subscription rights. There are no sinking fund provisions applicable to the common stock, except that the Company will provide its stockholders with the opportunity to redeem their public shares for cash equal to their pro rata share of the aggregate amount then on deposit in the Trust Account, under the circumstances, and, subject to the limitations, described herein.
Note 2Restatement of Previously Issued Financial Statements
In April 2021 the Company re-evaluated its accounting for its Public Warrants (as defined below in Note 4) and Private Placement Warrants (as defined below in Note 5) issued in connection with the Companys initial public offering (collectively, the Warrants) and determined that they should be treated as derivative liabilities pursuant to ASC 815 (ASC 815), Derivative and Hedging, rather than as components of stockholders equity as the Company previously treated the Warrants.
The restated classification and reported values of the Warrants as accounted for under ASC 815 are included in the financial statements herein. In the process of re-evaluating its financial statements the Company also restated its financial statements to classify all Class A common stock as temporary equity and to record accretion on the shares of Class A common stock. The Company had previously classified 3,133,926 shares of its Class A common stock as permanent equity. Since the Company classified the Public Warrants as derivative liabilities, offering costs totaling $1,075,021 that were previously allocated to the reported amount of the Public Warrants are now reflected as an expense in the statement of operations.
Impact of the Restatement
The impact of the restatement on the balance sheets, statements of operations and statements of cash flows for the Non-Reliance Periods is presented below.
| As of December 31, 2020 | ||||||||||||
| As Previously Reported |
Restatement Adjustment |
As Restated | ||||||||||
| BALANCE SHEET |
||||||||||||
| Warrant liability |
$ | | $ | 71,676,615 | $ | 71,676,615 | ||||||
| Total liabilities |
27,590,427 | 71,676,615 | 99,267,042 | |||||||||
| Class A common stock subject to possible redemption |
718,724,982 | 31,275,018 | 750,000,000 | |||||||||
| Class A common stock - $0.0001 par value |
313 | (313 | ) | | ||||||||
| Additional paid-in capital |
6,039,586 | (6,039,586 | ) | | ||||||||
| Accumulated deficit |
(1,041,771 | ) | (96,911,734 | ) | (97,953,505 | ) | ||||||
| Total stockholders equity/(deficit) |
5,000,002 | (102,951,633 | ) | (97,951,631 | ) | |||||||
| For the Year-Ended December 31, 2020 | ||||||||||||
| As Previously Reported |
Restatement Adjustment |
As Restated | ||||||||||
| STATEMENT OF OPERATIONS |
||||||||||||
| General and administrative expenses |
$ | (1,374,073 | ) | $ | (1,075,021 | ) | $ | (2,449,094 | ) | |||
| Change in fair value of warrant liability |
| (43,139,251 | ) | (43,139,251 | ) | |||||||
| Net loss |
(1,041,135 | ) | (44,214,272 | ) | (45,255,407 | ) | ||||||
| Basic and diluted net loss per share, Class A |
$ | (0.02 | ) | $ | (0.77 | ) | $ | (0.79 | ) | |||
| Basic and diluted net loss per share, Class B |
$ | (0.02 | ) | $ | (0.77 | ) | $ | (0.79 | ) | |||
F-12
Table of Contents
| For the Year-Ended December 31, 2020 | ||||||||||||
| As Previously Reported |
Restatement Adjustment |
As Restated | ||||||||||
| STATEMENT OF CASH FLOWS |
||||||||||||
| Net loss |
$ | (1,041,135 | ) | $ | (44,214,272 | ) | $ | (45,255,407 | ) | |||
| Change in fair value of warrant liability |
| 43,139,251 | 43,139,251 | |||||||||
| Issuance costs related to warrant liability |
| 1,075,021 | 1,075,021 | |||||||||
| As of July 2, 2020 | ||||||||||||
| As Previously Reported |
Restatement Adjustment |
As Restated | ||||||||||
| BALANCE SHEET |
||||||||||||
| Warrant liability |
$ | | $ | 28,537,364 | $ | 28,537,364 | ||||||
| Total liabilities |
26,965,589 | 28,537,364 | 55,502,953 | |||||||||
| Class A common stock subject to possible redemption |
719,775,570 | 30,224,430 | 750,000,000 | |||||||||
| Class A common stock - $0.0001 par value |
302 | (302 | ) | | ||||||||
| Additional paid-in capital |
5,044,723 | (5,044,723 | ) | | ||||||||
| Accumulated deficit |
(47,035 | ) | (53,716,769 | ) | (53,763,804 | ) | ||||||
| Total stockholders equity/(deficit) |
5,000,002 | (58,761,794 | ) | (53,761,792 | ) | |||||||
|
As of September 30, 2020 (UNAUDITED) |
||||||||||||
| As Previously Reported |
Restatement Adjustment |
As Restated | ||||||||||
| BALANCE SHEET |
||||||||||||
| Warrant liability |
$ | | $ | 59,344,241 | $ | 59,344,241 | ||||||
| Total liabilities |
27,002,221 | 59,344,241 | 86,346,462 | |||||||||
| Class A common stock subject to possible redemption |
719,471,496 | 30,528,504 | 750,000,000 | |||||||||
| Class A common stock - $0.0001 par value |
306 | (306 | ) | | ||||||||
| Additional paid-in capital |
5,293,078 | (5,293,078 | ) | | ||||||||
| Accumulated deficit |
(295,255 | ) | (84,579,361 | ) | (84,874,616 | ) | ||||||
| Total stockholders equity/(deficit) |
5,000,003 | (89,872,745 | ) | (84,872,742 | ) | |||||||
| Nine months ended September 30, 2020 (UNAUDITED) |
||||||||||||
| As Previously Reported |
Restatement Adjustment |
As Restated | ||||||||||
| STATEMENT OF OPERATIONS |
||||||||||||
| General and administrative expenses |
$ | (428,397 | ) | $ | (1,075,021 | ) | $ | (1,503,418 | ) | |||
| Change in fair value of warrant liability |
| (30,806,877 | ) | (30,806,877 | ) | |||||||
| Net loss |
(294,620 | ) | (31,881,898 | ) | (32,176,518 | ) | ||||||
| Basic and diluted net loss per share, Class A |
$ | (0.01 | ) | $ | (0.71 | ) | $ | (0.72 | ) | |||
| Basic and diluted net loss per share, Class B |
$ | (0.01 | ) | $ | (0.71 | ) | $ | (0.72 | ) | |||
| Nine months ended September 30, 2020 (UNAUDITED) |
||||||||||||
| As Previously Reported |
Restatement Adjustment |
As Restated | ||||||||||
| STATEMENT OF CASH FLOWS |
||||||||||||
| Net loss |
$ | (294,620 | ) | $ | (31,881,898 | ) | $ | (32,176,518 | ) | |||
| Change in fair value of warrant liability |
| 30,806,877 | 30,806,877 | |||||||||
| Issuance costs related to warrant liability |
| 1,075,021 | 1,075,021 | |||||||||
F-13
Table of Contents
Note 3Summary of Significant Accounting Policies
Basis of Presentation
The accompanying financial statements have been prepared in accordance with accounting principles generally accepted in the United States of America and pursuant to the accounting and disclosure rules and regulations of the SEC.
Emerging Growth Company
Section 102(b)(1) of the JOBS Act exempts emerging growth companies from being required to comply with new or revised financial accounting standards until private companies (that is, those that have not had a Securities Act registration statement declared effective or do not have a class of securities registered under the Exchange Act) are required to comply with the new or revised financial accounting standards. The JOBS Act provides that a company can elect to opt out of the extended transition period and comply with the requirements that apply to non-emerging growth companies but any such an election to opt out is irrevocable. The Company has elected not to opt out of such extended transition period which means that when a standard is issued or revised and it has different application dates for public or private companies, the Company, as an emerging growth company, can adopt the new or revised standard at the time private companies adopt the new or revised standard.
This may make comparison of the Companys financial statements with another public company which is neither an emerging growth company nor an emerging growth company which has opted out of using the extended transition period difficult or impossible because of the potential differences in accounting standards used.
Net Loss Per Common Share
Net loss per share of common stock is computed by dividing net loss by the weighted average number of common shares outstanding during the period. The Company applies the two-class method in calculating earnings per share. Accretion associated with the redeemable shares of Class A common stock is excluded from EPS as the redemption value approximates fair value.
At December 31, 2020, the Company had outstanding warrants to purchase of up to 27,250,000 shares of Class A common stock. The weighted average of these shares was excluded from the calculation of diluted net loss per share of common stock since the exercise of the Warrants is contingent upon the occurrence of future events. As of December 31, 2020, the Company did not have any dilutive securities or other contracts that could, potentially, be exercised or converted into shares of common stock and then share in the earnings of the Company. As a result, diluted net loss per share of common stock is the same as basic net loss per share of common stock for the period.
Cash and Cash Equivalents
Cash and cash equivalents include cash on hand and on deposit at banking institutions as well as all highly liquid short-term investments with original maturities of ninety (90) days or less. As of December 31, 2020, the Company held deposits of $383,246 at custodian account and $750,063,158 in Goldman Sachs Financial Square Treasury Instruments Fund, a money market fund managed by an affiliate of the Sponsor. Money market funds are characterized as Level I investments within the fair value hierarchy under ASC 820 (as defined below). The cash held in the money market account is considered restricted. Dividend income from money market funds is recognized on an accrual basis.
Redeemable Shares of Class A Common Stock
As discussed in Note 1, all of the 75,000,000 shares of Class A common stock sold as parts of the Units in the Public Offering contain a redemption feature. In accordance with the Accounting Standards Codification 480-10-S99-3A Classification and Measurement of Redeemable Securities, redemption provisions not solely
F-14
Table of Contents
within the control of the Company require the security to be classified outside of permanent equity. Ordinary liquidation events, which involve the redemption and liquidation of all of the entitys equity instruments, are excluded from the provisions of ASC 480. The Company had previously classified 3,133,926 shares of Class A common stock as permanent equity. As part of the restatement of the Companys financial statements, the Company has classified all of the shares of Class A common stock as redeemable. Immediately upon the closing of the Public Offering, the Company recognized the accretion from initial book value to redemption amount value. The change in the carrying value of redeemable shares of Class A common stock resulted in charges against additional paid-in capital and accumulated deficit.
As of December 31, 2020, the shares of Class A common stock reflected on the balance sheet are reconciled in the following table:
| As of December 31, 2020 |
||||
| Gross proceeds |
$ | 750,000,000 | ||
| Less: |
||||
| Proceeds allocated to public warrants |
$ | (19,587,038 | ) | |
| Class A shares issuance costs |
$ | (41,163,224 | ) | |
| Plus: |
||||
| Accretion of carrying value to redemption value |
$ | 60,750,262 | ||
|
|
|
|||
| Class A common stock subject to possible redemption |
$ | 750,000,000 | ||
|
|
|
|||
Concentration of Credit Risk
Financial instruments that potentially subject the Company to concentrations of credit risk consist of cash accounts in a financial institution, which, at times, may exceed the Federal Depository Insurance Coverage of $250,000. The Company has not experienced losses on these accounts and management believes the Company is not exposed to significant risks on such accounts.
Financial Instruments
The fair value of the Companys assets and liabilities, which qualify as financial instruments under the Accounting Standards Codification 820 (ASC 820), Fair Value Measurements and Disclosures, approximates the carrying amounts represented in the balance sheets, primarily due to their short term nature.
Use of Estimates
The preparation of financial statements in conformity with GAAP requires the Companys management to make estimates and assumptions that affect the reported amounts of assets and liabilities and disclosure of contingent assets and liabilities at the date of the financial statements and the reported amounts of expenses during the reporting period. One of the more significant accounting estimates included in these financial statements is the determination of the fair value of the warrant liability. Such estimates may be subject to change as more current information becomes available, and accordingly the actual results could differ significantly from those estimates.
Offering Costs
The Company complies with the requirements of the Accounting Standards Codification 340-10-S99-1 and SEC Staff Accounting Bulletin Topic 5AExpenses of Offering. The Company incurred offering costs in connection with its Public Offering of $988,245. These costs, together with the upfront underwriter discount and deferred underwriter discount, of $41,250,000 were allocated to the shares of Class A common stock and Public Warrants upon the closing of the Public Offering (as defined in Note 4). See Note 2 for further information about accounting for offering costs.
F-15
Table of Contents
Warrant Liability
The Company accounts for the Warrants in accordance with the guidance contained in ASC 815 under which the Warrants do not meet the criteria for equity treatment and must be recorded as derivative liabilities. Accordingly, the Company classifies the Warrants as liabilities at their fair value and adjusts the Warrants to fair value at each reporting period. This liability is subject to re-measurement at each balance sheet date until the Warrants are exercised or expire, and any change in fair value is recognized in the Companys statement of operations. The fair value of the Public Warrants was initially measured using a Monte Carlo simulation. The Public Warrants have subsequently been measured based on the listed market price. The fair value of the Private Warrants has been estimated using a Black-Scholes-Merton model since the initial measurement date. (see Note 7)
Income Taxes
The Company is taxed as a corporation for U.S. federal income tax purposes. As a corporation, for tax purposes, the Company is subject to U.S. federal and various state and local income taxes on its earnings.
Prior to July 2020, the Company was included with The Goldman Sachs Group Inc. and subsidiaries (the Group Inc.) in the consolidated corporate federal income tax return as well as consolidated/ combined state and local tax returns. The Company computed its tax liability on a modified separate company basis and will settle such liability with the Group Inc. pursuant to a tax sharing arrangement.
To the extent the Company generates tax benefits from losses during such time that it is consolidated with the Group Inc., the amounts will be reimbursed by the Group Inc. pursuant to the tax sharing arrangement. The Companys state and local tax liabilities are allocated to reflect its share of the consolidated/combined state and local income tax liability.
Following changes in ownership starting July 2020, the Company deconsolidated from the Group Inc. for tax purposes and the tax sharing arrangement with the Group Inc. was terminated. Beginning July 2020, the Company will file separate corporate federal and state and local income tax returns. To the extent the Company generates tax losses after it ceases being consolidated with the Group Inc., tax benefits from losses will be accrued if it is more likely than not the losses may be carried forward and utilized against future expected profits.
Income taxes are provided for using the assets and liabilities method under which deferred tax assets and liabilities are recognized for temporary differences between the financial reporting and tax bases of assets and liabilities.
Deferred Income Taxes
The Company follows the asset and liability method of accounting for income taxes under Accounting Standards Codification 740, Income Taxes. Deferred tax assets and liabilities are recognized for the estimated future tax consequences attributable to differences between the financial statements carrying amounts of existing assets and liabilities and their respective tax bases. Deferred tax assets and liabilities are measured using enacted tax rates expected to apply to taxable income in the years in which those temporary differences are expected to be recovered or settled. The effect on deferred tax assets and liabilities of a change in tax rates is recognized in income in the period that included the enactment date. Valuation allowances are established, when necessary, to reduce deferred tax assets to the amount expected to be realized.
Unrecognized Tax Benefits
The Company recognizes tax positions in the financial statements only when it is more likely than not that the position will be sustained on examination by the relevant taxing authority based on the technical merits of the position. A position that meets this standard is measured at the largest amount of benefit that will more likely
F-16
Table of Contents
than not be realized on settlement. A liability is established for differences between positions taken in a tax return and amounts recognized in the financial statements. There were no unrecognized tax benefits as of December 31, 2020. The Company recognizes accrued interest and penalties related to unrecognized tax benefits as income tax expense. No amounts were accrued for interest expense and penalties related to income tax matters as of December 31, 2020, 2019 and 2018. The Company is subject to income tax examinations by major taxing authorities since Inception.
Recent Accounting Pronouncements
Management does not believe that any recently issued, but not yet effective, accounting pronouncements, if currently adopted, would have a material effect on the Companys financial statements.
Note 4Public Offering
Upon the closing of the Public Offering, the Company sold 75,000,000 units at an offering price of $10.00 per unit (the Units) including 5,000,000 Units as a result of the underwriters partial exercise of their option to purchase additional Units. The Sponsor purchased an aggregate of 8,500,000 Private Placement Warrants (as defined below) at a price of $2.00 per Private Placement Warrant in a private placement that closed simultaneously with the closing of the Public Offering.
Each Unit consists of one share of the Companys Class A common stock, $0.0001 par value, and one-fourth of one redeemable warrant, with each whole warrant exercisable for one share of Class A common stock (each, a Public Warrant and, collectively, the Public Warrants). One Public Warrant entitles the holder thereof to purchase one whole share of Class A common stock at a price of $11.50 per share, subject to adjustment. No fractional shares will be issued upon exercise of the Public Warrants and only whole Public Warrants will trade. Each Public Warrant will become exercisable on the later of 30 days after the completion of the Initial Business Combination and 12 months from the closing of the Public Offering and will expire at 5:00 p.m., New York City time, five years after the completion of the Initial Business Combination or earlier upon redemption or liquidation. Once the Public Warrants become exercisable, the Company may redeem the outstanding Public Warrants in whole and not in part at a price of $0.01 per Public Warrant upon a minimum of 30 days prior written notice of redemption, if and only if the last reported sale price of the Companys Class A common stock equals or exceeds $18.00 per share (as adjusted) for any 20 trading days within a 30-trading day period ending on the third trading day prior to the date on which the Company sends the notice of redemption to the Public Warrant holders. Additionally, commencing 90 days after the Public Warrants become exercisable, the Company may redeem the outstanding Public Warrants in whole and not in part at a price of $0.10 per Public Warrant upon a minimum of 30 days prior written notice of redemption provided that holders will be able to exercise their Public Warrants on a cashless basis prior to redemption and receive that number of shares of Class A common stock to be determined by reference to a table included in the warrant agreement, based on the redemption date and the fair market value of Class A common stock, if and only if the last reported sale price of the Companys Class A common stock equals or exceeds $10.00 per share (as adjusted) on the trading day prior to the date on which the Company sends the notice of redemption to the Public Warrant holders.
The Company paid an underwriting commission of 2.0% of the gross proceeds of the Public Offering, (or $15,000,000), to the underwriters at the closing of the Public Offering, with an additional fee (the Deferred Underwriting Discount) of 3.5% of the gross proceeds of the Public Offering (or $26,250,000) payable upon the Companys completion of the Initial Business Combination. The Deferred Underwriting Discount will become payable to the underwriters from the amounts held in the Trust Account solely in the event the Company completes the Initial Business Combination. The Deferred Underwriting Discount has been recorded as a deferred liability on the balance sheet at December 31, 2020 as management has deemed the consummation of an Initial Business Combination to be probable.
The Public Warrants issued as part of the Units are accounted for as liabilities as they contain terms and features that do not qualify for equity classification under ASC 815. The fair value of the Public Warrants at issuance date
F-17
Table of Contents
was a liability of $19,587,038. At December 31, 2020, the fair value was $48,000,000. The change in fair value of $28,412,962 is reflected in change in fair value of warrant liability in the statement of operations.
All of the 75,000,000 shares of Class A common stock sold as part of the Units in the Public Offering contain a redemption feature which allows for the redemption of such public shares in connection with the Companys liquidation, if there is a stockholder vote or tender offer in connection with the Business Combination and in connection with certain amendments to the Companys amended and restated certificate of incorporation. In accordance with ASC 480, redemption provisions not solely within the control of the Company require common stock subject to redemption to be classified outside of permanent equity. Given that the Class A common stock was issued with other freestanding instruments (i.e., Public Warrants), the initial carrying value of Class A common stock classified as temporary equity is based on allocated proceeds in accordance with Accounting Standards Codification 470-20 Debt with Conversion and Other Options.
Note 5Related Party Transactions
Founder Shares
In July 2018, the Sponsor purchased 575 shares of Class B common stock (the Founder Shares) for an aggregate price of $5,000. On April 17, 2020, the Company conducted a 1:5000 stock split, resulting in the Sponsor holding 2,875,000 Founder Shares. Subsequently, on June 11, 2020, the Company conducted a 1:7 stock split, resulting in the Sponsor holding 20,125,000 Founder Shares, as well as increased the authorized shares of Class B common stock to 50,000,000. The financial statements reflect the changes of these splits retroactively for all periods presented. On June 29, 2020, the Sponsor transferred 1,325,000 of its Founder Shares to GS Acquisition Holdings II Employee Participation LLC (Employee Participation LLC), an affiliate of the Sponsor. The 20,125,000 Founder Shares included an aggregate of up to 2,625,000 shares that were subject to forfeiture if the underwriters option to purchase additional shares was not exercised in full by the underwriters to maintain the number of Founder Shares equal to 20% of the outstanding shares upon completion of the Public Offering. Following the partial exercise of the option to purchase additional shares, 1,375,000 Founder Shares were forfeited on August 13, 2020, at no cost in order to maintain the number of Founder Shares equal to 20% of the outstanding shares of common stock, upon the completion of the Public Offering. As used herein, unless the context otherwise requires, Founder Shares shall be deemed to include the shares of Class A common stock issuable upon conversion thereof. The Founder Shares are identical to the Class A common stock included in the Units sold in the Public Offering, except that: prior to the Initial Business Combination only holders of the Founder Shares have the right to vote on the election of the Companys directors and holders of a majority of the outstanding shares of Class B common stock may remove members of the Companys board of directors for any reason; the Founder Shares automatically convert into shares of Class A common stock at the time of the Initial Business Combination, or earlier at the option of the holder, on a one-for-one basis, subject to adjustment pursuant to certain anti-dilution rights; and are subject to certain transfer restrictions, as described in more detail below, and the holders of the Founder Shares, as described in more detail below, have agreed to certain restrictions and will have certain registration rights with respect thereto.
The Companys initial stockholders, officers and directors have agreed not to transfer, assign or sell any Founder Shares held by them until the earlier to occur of: (i) one year after the completion of the Initial Business Combination, (ii) the last sale price of Class A common stock equals or exceeds $12.00 per share (as adjusted for stock splits, stock dividends, reorganizations, recapitalizations and the like) for any 20 trading days within any 30-trading day period commencing at least 150 days after the Initial Business Combination, and (iii) the date following the completion of the Initial Business Combination on which the Company completes a liquidation, merger, stock exchange, reorganization or other similar transaction that results in all of the public stockholders having the right to exchange their shares of common stock for cash, securities or other property.
The Sponsor has purchased an aggregate of 8,500,000 private placement warrants at a price of $2.00 per whole warrant ($17,000,000 in the aggregate) in a private placement (the Private Placement) that closed concurrently
F-18
Table of Contents
with the closing of the Public Offering (the Private Placement Warrants). Each Private Placement Warrant is exercisable for one whole share of Class A common stock at a price of $11.50 per share, subject to adjustment in certain circumstances, including upon the occurrence of certain reorganization events. A portion of proceeds from the sale of the Private Placement Warrants were added to the proceeds from the Public Offering deposited in the Trust Account such that at the closing of the Public Offering, $750,000,000 was held in the Trust Account. If the Initial Business Combination is not completed within 24 months from the closing of the Public Offering, the proceeds from the sale of the Private Placement Warrants held in the Trust Account will be used to fund the redemption of the public shares (subject to the requirements of applicable law) and the Private Placement Warrants will expire worthless. The Private Placement Warrants will be non-redeemable and exercisable on a cashless basis so long as they are held by the Sponsor or its permitted transferees.
The Sponsor and the Companys officers and directors have agreed, subject to limited exceptions, not to transfer, assign or sell any of their Private Placement Warrants until 30 days after the completion of the Initial Business Combination.
Registration Rights
The holders of Founder Shares and Private Placement Warrants are, and holders of warrants that may be issued upon conversion of working capital loans, if any, will be, entitled to registration rights to require the Company to register the resale of any of its securities held by them (in the case of the Founder Shares, only after conversion of such shares to shares of Class A common stock) pursuant to a registration rights agreement dated June 29, 2020. These holders are also entitled to certain piggyback registration rights. The Company will bear the expenses incurred in connection with the filing of any such registration statements.
Related Party Sponsor Note
On April 17, 2020, an affiliate of the Sponsor agreed to loan the Company an aggregate amount of up to $300,000 to be used to pay a portion of the expenses related to the Public Offering pursuant to a promissory note (the Note). The Note was non-interest bearing, unsecured and payable on the earlier of December 31, 2020 and the closing of the Public Offering. On May 28, 2020 the Company borrowed $300,000 under the Note. On July 2, 2020, the full $300,000 balance of the Note was repaid.
On November 12, 2020, the Sponsor agreed to loan the Company up to an aggregate of $2,000,000 pursuant to the working capital note (the Working Capital Note). Any amounts borrowed under the Working Capital Note are non-interest bearing, unsecured and are due at the earlier of the date the Company is required to complete its Initial Business Combination pursuant to its amended and restated certificate of incorporation, as amended from time to time, and the closing of the Initial Business Combination. As of December 31, 2020, the Company has not drawn funds under the Working Capital Note.
Administrative Support Agreement
The Company has entered into an agreement to pay an affiliate of the Sponsor a total of $10,000 per month for office space, administrative and support services. Upon the earlier of the completion of the Initial Business Combination and the Companys liquidation, the Company will cease paying these monthly fees. For the year ended December 31, 2020, the Company incurred expenses of $60,000 under this agreement.
Note 6Stockholders Equity
Common Stock
The authorized common stock of the Company includes up to 500,000,000 shares of Class A common stock and 50,000,000 shares of Class B common stock. If the Company enters into an Initial Business Combination, it may
F-19
Table of Contents
(depending on the terms of such an Initial Business Combination) be required to increase the number of shares of Class A common stock which the Company is authorized to issue at the same time as the Companys stockholders vote on the Initial Business Combination to the extent the Company seeks stockholder approval in connection with the Initial Business Combination. Holders of the Companys common stock are entitled to one vote for each share of common stock; provided that only holders of the Class B common stock have the right to vote on the election of the Companys directors prior to the Initial Business Combination. At December 31, 2020, there were 75,000,000 shares of Class A common stock issued and outstanding, of which 75,000,000 shares were subject to possible redemption and are classified outside of permanent equity at the balance sheet, and 18,750,000 shares of Class B common stock issued and outstanding. In connection with issuance of shares of Class A common stock, the Company issued 18,750,000 Public Warrants. The Company has determined that the Public Warrants are accounted for separately from shares of Class A common stock.
Preferred Stock
The Company is authorized to issue 5,000,000 shares of preferred stock with such designations, voting and other rights and preferences as may be determined from time to time by the Companys board of directors. At December 31, 2020, there were no shares of preferred stock issued or outstanding.
Note 7Fair Value Measurements
The fair value of a financial instrument is the amount that would be received to sell an asset or paid to transfer a liability in an orderly transaction between market participants at the measurement date (i.e., the exit price).
The fair value hierarchy under ASC 820 prioritizes the inputs to valuation techniques used to measure fair value. The hierarchy gives the highest priority to unadjusted quoted prices in active markets for identical assets or liabilities (Level 1 measurements) and the lowest priority to unobservable inputs (Level 3 measurements). The three levels of the fair value hierarchy are as follows:
Basis for Fair Value Measurement
| Level 1: | Unadjusted quoted prices in active markets that are accessible at the measurement date for identical, unrestricted assets or liabilities; | |
| Level 2: | Quoted prices in markets that are not active or financial instruments for which significant inputs to models are observable (including but not limited to quoted prices for similar securities, interest rates, foreign exchange rates, volatility and credit risk), either directly or indirectly; | |
| Level 3: | Prices or valuations that require significant unobservable inputs (including the Managements assumptions in determining fair value measurement). | |
The following table presents information about the Companys assets and liabilities that are measured at fair value on a recurring basis at December 31, 2020 by level within the fair value hierarchy:
| Description |
December 31, 2020 |
Quoted Prices in Active Markets (Level 1) |
Significant Other Observable Inputs (Level 2) |
Significant Other Unobservable Inputs (Level 3) |
||||||||||||
| Assets: |
||||||||||||||||
| Money market fund held in Trust Account |
$ | 750,063,158 | $ | 750,063,158 | $ | | $ | | ||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Liabilities: |
||||||||||||||||
| Warrant LiabilityPublic Warrants |
$ | 48,000,000 | $ | 48,000,000 | $ | | $ | | ||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Warrant LiabilityPrivate Placement Warrants |
$ | 23,676,615 | $ | | $ | | $ | 23,676,615 | ||||||||
|
|
|
|
|
|
|
|
|
|||||||||
F-20
Table of Contents
Transfers to/from Levels 1, 2, and 3 are recognized at the end of the reporting period. The estimated fair value of the Public Warrants transferred from a Level 3 measurement as of September 30, 2020 as a result of the separate listing and trading of the Public Warrants as of August 20, 2020.
The fair value of the Public Warrants was initially measured using a Monte Carlo simulation. The Public Warrants have subsequently been measured based on the listed market price. The fair value of the Private Warrants has been estimated using a Black-Scholes-Merton model since initial measurement date. For the period from the closing of the Public Offering through December 31, 2020, the Company recognized a charge in the statement of operations resulting from an increase in the fair value of warrant liabilities of approximately $43.1 million presented as change in fair value of derivative warrant liability.
The estimated fair value of the Private Placement Warrants, and the Public Warrants prior to being separately listed and traded, was determined using Level 3 inputs. Inherent in a Black-Scholes-Merton model and Monte Carlo simulation are assumptions related to expected stock-price volatility, expected life, risk-free interest rate and dividend yield. The Company estimates the volatility of its Class A common stock warrants based on implied volatility from the Companys traded warrants and from historical volatility of select peer companies Class A common stock that matches the expected remaining life of the Warrants. The risk-free interest rate is based on the U.S. Treasury zero-coupon yield curve on the grant date for a maturity similar to the expected remaining life of the Warrants. The expected life of the Warrants is assumed to be equivalent to their remaining contractual term. The dividend rate is based on the historical rate, which the Company anticipates remaining at zero.
The following table provides quantitative information regarding Level 3 fair value measurements inputs:
| As of July 2, 2020 | As of December 31, 2020 |
|||||||||||
| Public Warrants |
Private Warrants |
Private Warrants |
||||||||||
| Stock price |
$ | 10.54 | $ | 10.54 | $ | 10.90 | ||||||
| Strike Price |
$ | 11.50 | $ | 11.50 | $ | 11.50 | ||||||
| Term (in years) |
6 | 6 | 5.75 | |||||||||
| Volitility |
10 - 20 | % | 10 - 18 | % | 28.30 | % | ||||||
| Risk-free rate |
0.40 | % | 0.40 | % | 0.47 | % | ||||||
| Dividend yield |
0.00 | % | 0.00 | % | 0.00 | % | ||||||
The change in the fair value of the Warrants measured with Level 3 inputs for the period from July 2, 2020 (Initial Measurement) through December 31, 2020 is summarized as follows:
| Issuance of Public and Private Warrants with Level 3 measurements |
$ | 28,537,364 | ||
| Change in fair value of warrant liability measured with Level 3 inputs |
14,726,289 | |||
| Transfer of Public Warrants to Level 1 measurements |
(19,587,038 | ) | ||
|
|
|
|||
| Warrants liability at December 31, 2020 measured utilizing Level 3 inputs |
$ | 23,676,615 | ||
|
|
|
Note 8Subsequent Events
On March 12, 2021, the Company borrowed $1,500,000 under the Working Capital Note. Effective March 30, 2021, the Sponsor agreed not to transfer its Private Placement Warrants.
F-21
Table of Contents
GS Acquisition Holdings Corp II
Unaudited Condensed Financial Statements
| F-23 | ||||
| F-24 | ||||
| F-25 | ||||
| F-26 | ||||
| F-27 | ||||
F-22
Table of Contents
PART IFINANCIAL INFORMATION
GS Acquisition Holdings Corp II
UNAUDITED CONDENSED BALANCE SHEETS
|
March 31, 2021 |
December 31, 2020 (As Restated) |
|||||||
| ASSETS |
||||||||
| Current assets: |
||||||||
| Cash |
$ | 1,740,897 | $ | 383,246 | ||||
| Prepaid expenses |
504,684 | 599,170 | ||||||
|
|
|
|
|
|||||
| Total current assets |
2,245,581 | 982,416 | ||||||
|
|
|
|
|
|||||
| Deferred tax asset |
403,767 | 265,954 | ||||||
| Cash and cash equivalents held in Trust Account |
750,074,432 | 750,063,158 | ||||||
| Accrued dividends receivable held in Trust Account |
3,883 | 3,883 | ||||||
|
|
|
|
|
|||||
| Total assets |
$ | 752,727,663 | $ | 751,315,411 | ||||
|
|
|
|
|
|||||
| LIABILITIES AND STOCKHOLDERS EQUITY |
||||||||
| Current liabilities: |
||||||||
| Accounts payable |
$ | 1,396,003 | $ | 965,370 | ||||
| Accrued offering costs |
375,000 | 375,000 | ||||||
| Income tax payable |
114 | 57 | ||||||
| Working capital note (see Note 4) |
1,500,000 | | ||||||
| Warrant liability |
61,475,828 | 71,676,615 | ||||||
|
|
|
|
|
|||||
| Total current liabilities |
64,746,945 | 73,017,042 | ||||||
| Deferred underwriting discount |
26,250,000 | 26,250,000 | ||||||
|
|
|
|
|
|||||
| Total liabilities |
90,996,945 | 99,267,042 | ||||||
|
|
|
|
|
|||||
| Commitments and contingencies |
||||||||
| Class A common stock subject to possible redemption; 75,000,000 shares at March 31, 2021 and December 31, 2020, respectively |
750,000,000 | 750,000,000 | ||||||
| Stockholders equity: |
||||||||
| Preferred stock, $0.0001 par value, 5,000,000 shares authorized, none issued and outstanding at March 31, 2021 and December 31, 2020, respectively |
| | ||||||
| Class A common stock, $0.0001 par value, 500,000,000 shares authorized at March 31, 2021 and December 31, 2020, respectively |
| | ||||||
| Class B common stock, $0.0001 par value, 50,000,000 shares authorized, 18,750,000 issued and
outstanding at March 31, 2021 and |
1,874 | 1,874 | ||||||
| Additional paid-in capital |
| | ||||||
| Accumulated deficit |
(88,271,156 | ) | (97,953,505 | ) | ||||
|
|
|
|
|
|||||
| Total stockholders equity/(deficit) |
(88,269,282 | ) | (97,951,631 | ) | ||||
|
|
|
|
|
|||||
| Total liabilities and stockholders equity |
$ | 752,727,663 | $ | 751,315,411 | ||||
|
|
|
|
|
|||||
See accompanying notes to financial statements
F-23
Table of Contents
GS Acquisition Holdings Corp II
UNAUDITED CONDENSED STATEMENTS OF OPERATIONS
| Three months ended March 31, | ||||||||
| 2021 | 2020 | |||||||
| Dividend income |
$ | 11,273 | $ | | ||||
| General and administrative expenses |
(667,467 | ) | | |||||
| Change in fair value of warrant liability |
10,200,787 | | ||||||
|
|
|
|
|
|||||
| Income (loss) before income taxes |
9,544,593 | | ||||||
| Income tax benefit/(expense) |
137,756 | | ||||||
|
|
|
|
|
|||||
| Net income |
$ | 9,682,349 | $ | | ||||
|
|
|
|
|
|||||
| Weighted average number of shares outstanding of Class A common stock |
75,000,000 | | ||||||
|
|
|
|
|
|||||
| Basic and diluted net income per share, Class A |
$ | 0.10 | $ | | ||||
|
|
|
|
|
|||||
| Weighted average number of shares outstanding of Class B common stock |
18,750,000 | 20,125,000 | ||||||
|
|
|
|
|
|||||
| Basic and diluted net income per share, Class B |
$ | 0.10 | $ | (0.00 | ) | |||
|
|
|
|
|
|||||
See accompanying notes to financial statements
F-24
Table of Contents
GS Acquisition Holdings Corp II
UNAUDITED CONDENSED STATEMENTS OF CHANGES IN STOCKHOLDERS EQUITY
| For the three months ended March 31, 2020 | ||||||||||||||||||||||||||||
| Class A Common Stock |
Class B Common Stock |
Additional Paid-in Capital |
Accumulated Deficit |
Stockholders Equity |
||||||||||||||||||||||||
| Shares | Amount | Shares | Amount | |||||||||||||||||||||||||
| Balance, December 31, 2019 |
| $ | | 20,125,000 | $ | 2,012 | $ | 2,988 | $ | (636 | ) | $ | 4,364 | |||||||||||||||
| Net loss |
| | | | | | | |||||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||
| Balance, March 31, 2020 |
| $ | | 20,125,000 | $ | 2,012 | $ | 2,988 | $ | (636 | ) | $ | 4,364 | |||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||
| For the three months ended March 31, 2021 | ||||||||||||||||||||||||||||
| Class A Common Stock |
Class B Common Stock |
Additional Paid-in Capital |
Accumulated Deficit |
Stockholders Equity |
||||||||||||||||||||||||
| Shares | Amount | Shares | Amount | |||||||||||||||||||||||||
| Balance, December 31, 2020 (As restated) |
| $ | | 18,750,000 | $ | 1,874 | $ | | $ | (97,953,505 | ) | $ | (97,951,631 | ) | ||||||||||||||
| Net income |
| | | | | 9,682,349 | 9,682,349 | |||||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||
| Balance, March 31, 2021 |
| $ | | 18,750,000 | $ | 1,874 | $ | | $ | (88,271,156 | ) | $ | (88,269,282 | ) | ||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||
See accompanying notes to financial statements
F-25
Table of Contents
GS Acquisition Holdings Corp II
UNAUDITED CONDENSED STATEMENTS OF CASH FLOWS
| Three months ended March 31, | ||||||||
| 2021 | 2020 | |||||||
| Cash flows from operating activities: |
||||||||
| Net income |
$ | 9,682,349 | $ | | ||||
| Adjustments to reconcile net income (loss) to net cash used in operating activities: |
||||||||
| Change in fair value of warrant liability |
(10,200,787 | ) | | |||||
| Change in operating assets and liabilities: |
||||||||
| Decrease in prepaid expenses |
94,486 | | ||||||
| Increase in deferred tax assets |
(137,813 | ) | | |||||
| Increase in accounts payable |
430,633 | | ||||||
| Increase in income tax payable |
57 | | ||||||
|
|
|
|
|
|||||
| Net cash used for operating activities |
(131,075 | ) | | |||||
|
|
|
|
|
|||||
| Cash flows from financing activities: |
||||||||
| Proceeds from working capital note |
1,500,000 | | ||||||
|
|
|
|
|
|||||
| Net cash provided by financing activities |
1,500,000 | | ||||||
|
|
|
|
|
|||||
| Increase in cash and restricted cash |
1,368,925 | | ||||||
| Cash and restricted cash and cash equivalents at beginning of period |
750,446,404 | 5,000 | ||||||
|
|
|
|
|
|||||
| Cash and restricted cash and cash equivalents at end of period |
$ | 751,815,329 | $ | 5,000 | ||||
|
|
|
|
|
|||||
| Supplemental disclosure of non-cash financing activities |
||||||||
| Accrued offering costs |
$ | | $ | 81,857 | ||||
See accompanying notes to financial statements
F-26
Table of Contents
NOTES TO CONDENSED FINANCIAL STATEMENTS
(UNAUDITED)
Note 1Description of Organization and Business Operations
Organization and General
GS Acquisition Holdings Corp II (the Company) was incorporated as a Delaware corporation on May 31, 2018. The Company was formed for the purpose of effecting a merger, capital stock exchange, asset acquisition, stock purchase, reorganization or similar business combination with one or more businesses (the Initial Business Combination). The Company is an emerging growth company, as defined in Section 2(a) of the Securities Act of 1933, as amended (the Securities Act), as modified by the Jumpstart Our Business Startups Act of 2012 (the JOBS Act).
All activity for the period from May 31, 2018 (inception) through March 31, 2021 relates to the Companys formation and its initial public offering (the Public Offering) described below and identifying and evaluating prospective acquisition targets for an Initial Business Combination. The Company will not generate any operating revenues until after completion of its Initial Business Combination, at the earliest. The Company will generate non-operating income in the form of interest or dividend income on cash and cash equivalents from the proceeds derived from the Public Offering and the Private Placement (as defined below in Note 3). The Company has selected December 31st as its fiscal year end.
Sponsor and Financing
The Companys sponsor is GS Sponsor II LLC, a Delaware limited liability company (the Sponsor).
The registration statement for the Companys Public Offering was declared effective by the United States Securities and Exchange Commission (the SEC) on June 29, 2020. On June 30, 2020, the underwriters partially exercised their option to purchase additional Units (as defined below in Note 3). The Companys Public Offering of 75,000,000 Units, including 5,000,000 Units pursuant to the underwriters partial exercise of such option, closed on July 2, 2020 (as described in Note 3). Upon the closing of the Public Offering and the Private Placement, $750,000,000 was placed in a U.S. based trust account (the Trust Account) (discussed below). The Company intends to finance its Initial Business Combination with the net proceeds from the Public Offering and the sale of the Private Placement Warrants (as defined below in Note 3).
The Trust Account
The proceeds held in the Trust Account are invested in a money market fund registered under the Investment Company Act of 1940, as amended (the Investment Company Act) and meeting certain conditions under Rule 2a-7.
Except with respect to dividends earned on the funds held in the Trust Account that may be released to the Company to pay its taxes, the proceeds from the Public Offering and the Private Placement will not be released from the Trust Account until the earliest of: (i) the completion of the Initial Business Combination; (ii) the redemption of any public shares properly submitted in connection with a stockholder vote to amend the Companys amended and restated certificate of incorporation (A) to modify the substance or timing of the Companys obligation to allow redemptions in connection with the Initial Business Combination or to redeem 100% of its public shares if it does not complete the Initial Business Combination within 24 months from the closing of the Public Offering or (B) with respect to any other provision relating to stockholders rights or pre-Initial Business Combination activity; and (iii) the redemption of all of the Companys public shares if the Company has not completed the Initial Business Combination within 24 months from the closing of the Public Offering, subject to applicable law. The proceeds deposited in the Trust Account could become subject to the claims of the Companys creditors, if any, which could have priority over the claims of the Companys public stockholders.
F-27
Table of Contents
The balance in the Trust Account as of March 31, 2021 was $750,078,315, including $3,883 of accrued dividends.
Initial Business Combination
The Companys management has broad discretion with respect to the specific application of the net proceeds of the Public Offering, although substantially all of the net proceeds of the Public Offering and the Private Placement are intended to be generally applied toward consummating an Initial Business Combination. The Initial Business Combination must occur with one or more operating businesses or assets with a fair market value equal to at least 80% of the net assets held in the Trust Account (excluding the amount of any deferred underwriting discount). There is no assurance that the Company will be able to successfully effect an Initial Business Combination.
The Company, after signing a definitive agreement for an Initial Business Combination, will provide its public stockholders with the opportunity to redeem all or a portion of their shares upon the completion of the Initial Business Combination, either (i) in connection with a stockholder meeting called to approve the business combination or (ii) by means of a tender offer. However, in no event will the Company redeem its public shares in an amount that would cause its net tangible assets, after payment of deferred underwriting commissions, to be less than $5,000,001 following such redemptions. In such case, the Company would not proceed with the redemption of its public shares and the related Initial Business Combination, and instead may search for an alternate Initial Business Combination.
If the Company holds a stockholder vote or there is a tender offer for shares in connection with an Initial Business Combination, a public stockholder will have the right to redeem its shares for an amount in cash equal to its pro rata share of the aggregate amount then on deposit in the Trust Account, calculated as of two business days prior to the consummation of the Initial Business Combination, including interest but less taxes payable. As a result, such shares of Class A common stock are recorded at redemption amount and classified as temporary equity upon the completion of the Public Offering, in accordance with the Financial Accounting Standards Board (FASB) Accounting Standards Codification (ASC) 480, Distinguishing Liabilities from Equity.
Pursuant to the Companys amended and restated certificate of incorporation, if the Company is unable to complete the Initial Business Combination within 24 months from the closing of the Public Offering, the Company will (i) cease all operations except for the purpose of winding up, (ii) as promptly as reasonably possible but no more than ten business days thereafter redeem the public shares, at a per-share price, payable in cash, equal to the aggregate amount then on deposit in the Trust Account, including interest (which interest shall be net of taxes payable, and less up to $100,000 of interest to pay dissolution expenses), divided by the number of then outstanding public shares, which redemption will completely extinguish public stockholders rights as stockholders (including the right to receive further liquidating distributions, if any), and (iii) as promptly as reasonably possible following such redemption, subject to the approval of the Companys remaining stockholders and the Companys board of directors, dissolve and liquidate, subject in each case to the Companys obligations under Delaware law to provide for claims of creditors and the requirements of other applicable law.
The Sponsor, Employee Participation LLC (as defined below in Note 4) and the Companys officers and directors have entered into a letter agreement with the Company, pursuant to which they have waived their rights to liquidating distributions from the Trust Account with respect to any Founder Shares (as defined below in Note 4) held by them if the Company fails to complete the Initial Business Combination within 24 months of the closing of the Public Offering or during any extended time that the Company has to consummate an Initial Business Combination beyond 24 months as a result of a stockholder vote to amend its amended and restated certificate of incorporation. However, if the Sponsor, Employee Participation LLC or any of the Companys directors or officers hold any shares of Class A common stock in or after the Public Offering, they will be entitled to liquidating distributions from the Trust Account with respect to such shares if the Company fails to complete the Initial Business Combination within the prescribed time period.
F-28
Table of Contents
In the event of a liquidation, dissolution or winding up of the Company after an Initial Business Combination, the Companys stockholders are entitled to share ratably in all assets remaining available for distribution to them after payment of liabilities and after provision is made for each class of stock, if any, having preference over the common stock. The Companys stockholders have no preemptive or other subscription rights. There are no sinking fund provisions applicable to the common stock, except that the Company will provide its stockholders with the opportunity to redeem their public shares for cash equal to their pro rata share of the aggregate amount then on deposit in the Trust Account, under the circumstances, and, subject to the limitations, described herein.
Note 2Summary of Significant Accounting Policies
Basis of Presentation
The Companys unaudited condensed financial statements have been prepared in accordance with accounting principles generally accepted in the United States of America (U.S. GAAP) and the rules and regulations of the SEC for interim financial information and the instructions to Form 10-Q. Certain disclosures included in the annual financial statements have been condensed or omitted from these financial statements as they are not required for interim financial statements under U.S. GAAP and the rules of the SEC. These unaudited condensed financial statements reflect all adjustments that are, in the opinion of management, necessary for a fair statement of the results for the interim periods presented. These adjustments are of a normal, recurring nature. Interim period operating results may not be indicative of the operating results for a full year.
The accompanying unaudited condensed financial statements should be read in conjunction with the Companys audited financial statements and notes thereto included in the Companys restated Annual Report on Form 10-K/A for the year ended December 31, 2020.
Emerging Growth Company
Section 102(b)(1) of the JOBS Act exempts emerging growth companies from being required to comply with new or revised financial accounting standards until private companies (that is, those that have not had a Securities Act registration statement declared effective or do not have a class of securities registered under the Exchange Act) are required to comply with the new or revised financial accounting standards. The JOBS Act provides that a company can elect to opt out of the extended transition period and comply with the requirements that apply to non-emerging growth companies but any such an election to opt out is irrevocable. The Company has elected not to opt out of such extended transition period which means that when a standard is issued or revised and it has different application dates for public or private companies, the Company, as an emerging growth company, can adopt the new or revised standard at the time private companies adopt the new or revised standard.
This may make comparison of the Companys financial statements with another public company which is neither an emerging growth company nor an emerging growth company which has opted out of using the extended transition period difficult or impossible because of the potential differences in accounting standards used.
Cash and Cash Equivalents
Cash and cash equivalents include cash on hand and on deposit at banking institutions as well as all highly liquid short-term investments with original maturities of ninety (90) days or less. As of March 31, 2021, the Company held deposits of $1,740,897 in a custodian account and $750,074,432 in Goldman Sachs Financial Square Treasury Instruments Fund, a money market fund managed by an affiliate of the Sponsor. Money market funds are characterized as Level I investments within the fair value hierarchy under ASC 820 (as defined below). The cash held in the money market account is considered restricted. Dividend income from money market funds is recognized on an accrual basis.
F-29
Table of Contents
Redeemable Shares of Class A Common Stock
As discussed in Note 1, all of the 75,000,000 shares of Class A common stock sold as parts of the Units in the Public Offering contain a redemption feature. In accordance with the Accounting Standards Codification 480-10-S99-3AClassification and Measurement of Redeemable Securities, redemption provisions not solely within the control of the Company require the security to be classified outside of permanent equity. Ordinary liquidation events, which involve the redemption and liquidation of all of the entitys equity instruments, are excluded from the provisions of ASC 480. The Company classifies all shares of Class A common stock as redeemable.
Net Income Per Common Share
Net income per share of common stock is computed by dividing net income by the weighted average number of common shares outstanding during the period. The Company applies the two-class method in calculating earnings per share. Accretion associated with the redeemable shares of Class A common stock is excluded from earnings per share as the redemption value approximates fair value.
As of March 31, 2021, the Company had outstanding warrants to purchase of up to 27,250,000 shares of Class A common stock. The weighted average of these shares was excluded from the calculation of diluted net income per share of common stock since the exercise of the warrants is contingent upon the occurrence of future events. As of March 31, 2021, the Company did not have any dilutive securities or other contracts that could, potentially, be exercised or converted into shares of common stock and then share in the earnings of the Company. As a result, diluted net income per share of common stock is the same as basic net income per share of common stock for the period.
Concentration of Credit Risk
Financial instruments that potentially subject the Company to concentrations of credit risk consist of cash accounts in a financial institution, which, at times, may exceed the Federal Depository Insurance Coverage of $250,000. The Company has not experienced losses on these accounts and management believes the Company is not exposed to significant risks on such accounts.
Financial Instruments
The fair value of the Companys assets and liabilities, which qualify as financial instruments under the Accounting Standards Codification 820 (ASC 820), Fair Value Measurements and Disclosures, approximates the carrying amounts represented in the balance sheets, primarily due to their short term nature.
Use of Estimates
The preparation of financial statements in conformity with GAAP requires the Companys management to make estimates and assumptions that affect the reported amounts of assets and liabilities and disclosure of contingent assets and liabilities at the date of the financial statements and the reported amounts of expenses during the reporting period. One of the more significant accounting estimates included in these financial statements is the determination of the fair value of the warrant liability. Such estimates may be subject to change as more current information becomes available and accordingly, the actual results could differ significantly from those estimates.
Warrant Liability
The Company accounts for the warrants in accordance with the guidance contained in Accounting Standards Codification 815 (ASC 815), Derivatives and Hedging, under which the warrants do not meet the criteria for
F-30
Table of Contents
equity treatment and must be recorded as derivative liabilities. Accordingly, the Company classifies the warrants as liabilities at their fair value and adjusts the warrants to fair value at each reporting period. This liability is subject to re-measurement at each balance sheet date until the warrants are exercised or expire, and any change in fair value is recognized in the Companys statement of operations. The fair value of the Private Placement Warrants (as defined in Note 4) has been estimated using a Black-Scholes-Merton model and the fair value of the Public Warrants (as defined in Note 3) issued in connection with the Public Offering has been measured based on the listed market price of such Public Warrants (see Note 6).
Income Taxes
The Company is taxed as a corporation for U.S. federal income tax purposes. As a corporation, for tax purposes, the Company is subject to U.S. federal and various state and local income taxes on its earnings. Prior to July 2020, the Company was included with The Goldman Sachs Group Inc. and subsidiaries (the Group Inc.) in the consolidated corporate federal income tax return as well as consolidated/combined state and local tax returns. The Company computed its tax liability on a modified separate company basis and will settle such liability with the Group Inc. pursuant to a tax sharing arrangement.
To the extent the Company generates tax benefits from losses during such time that it is consolidated with the Group Inc., the amounts will be reimbursed by the Group Inc., pursuant to the tax sharing arrangement. The Companys state and local tax liabilities are allocated to reflect its share of the consolidated/combined state and local income tax liability.
Following changes in ownership starting July 2020, the Company deconsolidated from the Group Inc. for tax purposes and the tax sharing arrangement with the Group Inc. was terminated. Beginning July 2020, the Company will file separate corporate federal and state and local income tax returns. To the extent the Company generates tax losses after it ceases being consolidated with the Group Inc., tax benefits from losses will be accrued if it is more likely than not the losses may be carried forward and utilized against future expected profits.
Income taxes are provided for using the assets and liabilities method under which deferred tax assets and liabilities are recognized for temporary differences between the financial reporting and tax bases of assets and liabilities.
Deferred Income Taxes
The Company follows the asset and liability method of accounting for income taxes under Accounting Standards Codification 740, Income Taxes. Deferred tax assets and liabilities are recognized for the estimated future tax consequences attributable to differences between the financial statements carrying amounts of existing assets and liabilities and their respective tax bases. Deferred tax assets and liabilities are measured using enacted tax rates expected to apply to taxable income in the years in which those temporary differences are expected to be recovered or settled. The effect on deferred tax assets and liabilities of a change in tax rates is recognized in income in the period that included the enactment date. Valuation allowances are established, when necessary, to reduce deferred tax assets to the amount expected to be realized.
Unrecognized Tax Benefits
The Company recognizes tax positions in the financial statements only when it is more likely than not that the position will be sustained on examination by the relevant taxing authority based on the technical merits of the position. A position that meets this standard is measured at the largest amount of benefit that will more likely than not be realized on settlement. A liability is established for differences between positions taken in a tax return and amounts recognized in the financial statements. There were no unrecognized tax benefits as of March 31, 2021. The Company recognizes accrued interest and penalties related to unrecognized tax benefits as income tax expense. No amounts were accrued for interest expense and penalties related to income tax matters as of March 31, 2021 and December 31, 2020. The Company is subject to income tax examinations by major taxing authorities since inception.
F-31
Table of Contents
Recent Accounting Pronouncements
Management does not believe that any recently issued, but not yet effective, accounting pronouncements, if currently adopted, would have a material effect on the Companys financial statements.
Note 3Public Offering
Upon the closing of the Public Offering, the Company sold 75,000,000 units at an offering price of $10.00 per unit (the Units) including 5,000,000 Units as a result of the underwriters partial exercise of their option to purchase additional Units. The Sponsor purchased an aggregate of 8,500,000 Private Placement Warrants (as defined below) at a price of $2.00 per Private Placement Warrant in a private placement that closed simultaneously with the closing of the Public Offering.
Each Unit consists of one share of the Companys Class A common stock, $0.0001 par value, and one-fourth of one redeemable warrant, with each whole warrant exercisable for one share of Class A common stock (each, a Public Warrant and, collectively, the Public Warrants). One Public Warrant entitles the holder thereof to purchase one whole share of Class A common stock at a price of $11.50 per share, subject to adjustment. No fractional shares will be issued upon exercise of the Public Warrants and only whole Public Warrants will trade. Each Public Warrant will become exercisable on the later of 30 days after the completion of the Initial Business Combination and 12 months from the closing of the Public Offering and will expire at 5:00 p.m., New York City time, five years after the completion of the Initial Business Combination or earlier upon redemption or liquidation. Once the Public Warrants become exercisable, the Company may redeem the outstanding Public Warrants in whole and not in part at a price of $0.01 per Public Warrant upon a minimum of 30 days prior written notice of redemption, if and only if the last reported sale price of the Companys Class A common stock equals or exceeds $18.00 per share (as adjusted) for any 20 trading days within a 30-trading day period ending on the third trading day prior to the date on which the Company sends the notice of redemption to the Public Warrant holders. Additionally, commencing 90 days after the Public Warrants become exercisable, the Company may redeem the outstanding Public Warrants in whole and not in part at a price of $0.10 per Public Warrant upon a minimum of 30 days prior written notice of redemption provided that holders will be able to exercise their Public Warrants on a cashless basis prior to redemption and receive that number of shares of Class A common stock to be determined by reference to a table included in the warrant agreement, based on the redemption date and the fair market value of Class A common stock, if and only if the last reported sale price of the Companys Class A common stock equals or exceeds $10.00 per share (as adjusted) on the trading day prior to the date on which the Company sends the notice of redemption to the Public Warrant holders.
The Company paid an underwriting commission of 2.0% of the gross proceeds of the Public Offering (or $15,000,000) to the underwriters at the closing of the Public Offering, with an additional fee (the Deferred Underwriting Discount) of 3.5% of the gross proceeds of the Public Offering (or $26,250,000) payable upon the Companys completion of the Initial Business Combination. The Deferred Underwriting Discount will become payable to the underwriters from the amounts held in the Trust Account solely in the event the Company completes the Initial Business Combination. The Deferred Underwriting Discount has been recorded as a deferred liability on the balance sheet as of March 31, 2021 as management has deemed the consummation of an Initial Business Combination to be probable.
The Public Warrants issued as part of the Units are accounted for as liabilities as they contain terms and features that do not qualify for equity classification under ASC 815. The fair value of the Public Warrants at December 31, 2020 was a liability of $48,000,000. At March 31, 2021, the fair value was $41,250,000. The change in fair value of $6,750,000 is reflected in change in fair value of warrant liability.
All of the 75,000,000 shares of Class A common stock sold as part of the Units in the Public Offering contain a redemption feature which allows for the redemption of such public shares in connection with the Companys liquidation, if there is a stockholder vote or tender offer in connection with the Business
F-32
Table of Contents
Combination and in connection with certain amendments to the Companys amended and restated certificate of incorporation. In accordance with ASC 480, redemption provisions not solely within the control of the Company require common stock subject to redemption to be classified outside of permanent equity. Given that the Class A common stock was issued with other freestanding instruments (i.e., Public Warrants), the initial carrying value of Class A common stock clasified as temporary equity is based on allocated proceeds in accordance with Accounting Standards Codification 470-20, Debt with Conversion and Other Options.
Note 4Related Party Transactions
Founder Shares
In July 2018, the Sponsor purchased 575 shares of Class B common stock (the Founder Shares) for an aggregate price of $5,000. On April 17, 2020, the Company conducted a 1:5000 stock split, resulting in the Sponsor holding 2,875,000 Founder Shares. Subsequently, on June 11, 2020, the Company conducted a 1:7 stock split, resulting in the Sponsor holding 20,125,000 Founder Shares, as well as increased the authorized shares of Class B common stock to 50,000,000. The unaudited condensed financial statements reflect the changes of these splits retroactively for all periods presented. On June 29, 2020, the Sponsor transferred 1,325,000 of its Founder Shares to GS Acquisition Holdings II Employee Participation LLC (Employee Participation LLC), an affiliate of the Sponsor. The 20,125,000 Founder Shares included an aggregate of up to 2,625,000 shares that were subject to forfeiture if the underwriters option to purchase additional shares was not exercised in full by the underwriters to maintain the number of Founder Shares equal to 20% of the outstanding shares upon completion of the Public Offering. Following the partial exercise of the option to purchase additional shares, 1,375,000 Founder Shares were forfeited on August 13, 2020, at no cost in order to maintain the number of Founder Shares equal to 20% of the outstanding shares of common stock, upon the completion of the Public Offering. As used herein, unless the context otherwise requires, Founder Shares shall be deemed to include the shares of Class A common stock issuable upon conversion thereof. The Founder Shares are identical to the Class A common stock included in the Units sold in the Public Offering, except that: prior to the Initial Business Combination only holders of the Founder Shares have the right to vote on the election of the Companys directors and holders of a majority of the outstanding shares of Class B common stock may remove members of the Companys board of directors for any reason; the Founder Shares automatically convert into shares of Class A common stock at the time of the Initial Business Combination, or earlier at the option of the holder, on a one-for-one basis, subject to adjustment pursuant to certain anti-dilution rights; and are subject to certain transfer restrictions, as described in more detail below, and the holders of the Founder Shares, as described in more detail below, have agreed to certain restrictions and will have certain registration rights with respect thereto.
The Companys initial stockholders, officers and directors have agreed not to transfer, assign or sell any Founder Shares held by them until the earlier to occur of: (i) one year after the completion of the Initial Business Combination, (ii) the last sale price of Class A common stock equals or exceeds $12.00 per share (as adjusted for stock splits, stock dividends, reorganizations, recapitalizations and the like) for any 20 trading days within any 30-trading day period commencing at least 150 days after the Initial Business Combination, and (iii) the date following the completion of the Initial Business Combination on which the Company completes a liquidation, merger, stock exchange, reorganization or other similar transaction that results in all of the public stockholders having the right to exchange their shares of common stock for cash, securities or other property.
The Sponsor has purchased an aggregate of 8,500,000 private placement warrants at a price of $2.00 per whole warrant ($17,000,000 in the aggregate) in a private placement (the Private Placement) that closed concurrently with the closing of the Public Offering (the Private Placement Warrants). Each Private Placement Warrant is exercisable for one whole share of Class A common stock at a price of $11.50 per share, subject to adjustment in certain circumstances, including upon the occurrence of certain reorganization events. A portion of proceeds from the sale of the Private Placement Warrants were added to the proceeds from the Public Offering deposited in the Trust Account such that at the closing of the Public Offering, $750,000,000 was held in the Trust Account. If the Initial Business Combination is not completed within 24 months from the closing of the Public
F-33
Table of Contents
Offering, the proceeds from the sale of the Private Placement Warrants held in the Trust Account will be used to fund the redemption of the public shares (subject to the requirements of applicable law) and the Private Placement Warrants will expire worthless. The Private Placement Warrants will be non-redeemable and exercisable on a cashless basis so long as they are held by the Sponsor or its permitted transferees. Effective March 30, 2021, the Sponsor agreed not to transfer its Private Placement Warrants.
The Sponsor and the Companys officers and directors have agreed, subject to limited exceptions, not to transfer, assign or sell any of their Private Placement Warrants until 30 days after the completion of the Initial Business Combination.
Registration Rights
The holders of Founder Shares and Private Placement Warrants are, and holders of warrants that may be issued upon conversion of working capital loans, if any, will be, entitled to registration rights to require the Company to register the resale of any of its securities held by them (in the case of the Founder Shares, only after conversion of such shares to shares of Class A common stock) pursuant to a registration rights agreement dated June 29, 2020. These holders are also entitled to certain piggyback registration rights. The Company will bear the expenses incurred in connection with the filing of any such registration statements.
Related Party Sponsor Note
On April 17, 2020, an affiliate of the Sponsor agreed to loan the Company an aggregate amount of up to $300,000 to be used to pay a portion of the expenses related to the Public Offering pursuant to a promissory note (the Note). The Note was non-interest bearing, unsecured and payable on the earlier of December 31, 2020 and the closing of the Public Offering. On May 28, 2020 the Company borrowed $300,000 under the Note. On July 2, 2020, the full $300,000 balance of the Note was repaid to an affiliate of the Sponsor.
On November 12, 2020, the Sponsor agreed to loan the Company up to an aggregate of $2,000,000 pursuant to the working capital note (the Working Capital Note). Any amounts borrowed under the Working Capital Note are non-interest bearing, unsecured and are due at the earlier of the date the Company is required to complete its Initial Business Combination pursuant to its amended and restated certificate of incorporation, as amended from time to time, and the closing of the Initial Business Combination. On March 12, 2021, the Company borrowed $1,500,000 under the Working Capital Note.
Administrative Support Agreement
The Company has entered into an agreement to pay an affiliate of the Sponsor a total of $10,000 per month for office space, administrative and support services. Upon the earlier of the completion of the Initial Business Combination and the Companys liquidation, the Company will cease paying these monthly fees. For the three months ended March 31, 2021, the Company incurred expenses of $30,000 under this agreement.
Note 5Stockholders Equity
Common Stock
The authorized common stock of the Company includes up to 500,000,000 shares of Class A common stock and 50,000,000 shares of Class B common stock. If the Company enters into an Initial Business Combination, it may (depending on the terms of such an Initial Business Combination) be required to increase the number of shares of Class A common stock which the Company is authorized to issue at the same time as the Companys stockholders vote on the Initial Business Combination to the extent the Company seeks stockholder approval in connection with the Initial Business Combination. Holders of the Companys common stock are entitled to one vote for each share of common stock; provided that only holders of the Class B common stock have the right to
F-34
Table of Contents
vote on the election of the Companys directors prior to the Initial Business Combination. At March 31, 2021, there were 75,000,000 shares of Class A common stock issued and outstanding, of which 75,000,000 shares were subject to possible redemption and are classified outside of permanent equity at the balance sheet, and 18,750,000 shares of Class B common stock issued and outstanding. In connection with issuance of shares of Class A common stock, the Company issued 18,750,000 Public Warrants. The Company has determined that the Public Warrants are accounted for separately from shares of Class A common stock.
Preferred Stock
The Company is authorized to issue 5,000,000 shares of preferred stock with such designations, voting and other rights and preferences as may be determined from time to time by the Companys board of directors. At March 31, 2021, there were no shares of preferred stock issued or outstanding.
Note 6Fair Value Measurements
The fair value of a financial instrument is the amount that would be received to sell an asset or paid to transfer a liability in an orderly transaction between market participants at the measurement date (i.e., the exit price).
The fair value hierarchy under ASC 820 prioritizes the inputs to valuation techniques used to measure fair value. The hierarchy gives the highest priority to unadjusted quoted prices in active markets for identical assets or liabilities (Level 1 measurements) and the lowest priority to unobservable inputs (Level 3 measurements). The three levels of the fair value hierarchy are as follows:
Basis for Fair Value Measurement
| Level 1: | Unadjusted quoted prices in active markets that are accessible at the measurement date for identical, unrestricted assets or liabilities; | |
| Level 2: | Quoted prices in markets that are not active or financial instruments for which significant inputs to models are observable (including but not limited to quoted prices for similar securities, interest rates, foreign exchange rates, volatility and credit risk), either directly or indirectly; | |
| Level 3: | Prices or valuations that require significant unobservable inputs (including the Managements assumptions in determining fair value measurement). | |
The following table presents information about the Companys assets and liabilities that are measured at fair value on a recurring basis at March 31, 2021 and December 31, 2021 by level within the fair value hierarchy:
| March 31, 2021 | Quoted Prices in Active Markets (Level 1) |
Significant Other Observable Inputs (Level 2) |
Significant Other Unobservable Inputs (Level 3) |
|||||||||||||
| Assets: |
||||||||||||||||
| Money market funds held in Trust Account |
$ | 750,074,432 | $ | 750,074,432 | $ | | $ | | ||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Liabilities: |
||||||||||||||||
| Warrant LiabilityPublic Warrants |
$ | 41,250,000 | $ | 41,250,000 | $ | | $ | | ||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Warrant LiabilityPrivate Placement Warrants |
$ | 20,225,828 | $ | | $ | | $ | 20,225,828 | ||||||||
|
|
|
|
|
|
|
|
|
|||||||||
F-35
Table of Contents
| Description | December 31, 2020 | Quoted Prices in Active Markets (Level 1) |
Significant Other Observable Inputs (Level 2) |
Significant Other Unobservable Inputs (Level 3) |
||||||||||||
| Assets: |
||||||||||||||||
| Money market funds held in Trust Account |
$ | 750,063,158 | $ | 750,063,158 | $ | | $ | | ||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Liabilities: |
||||||||||||||||
| Warrant LiabilityPublic Warrants |
$ | 48,000,000 | $ | 48,000,000 | $ | | $ | | ||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Warrant LiabilityPrivate Placement Warrants |
$ | 23,676,615 | $ | | $ | | $ | 23,676,615 | ||||||||
|
|
|
|
|
|
|
|
|
|||||||||
For the period ended March 31, 2021, the fair value of Public Warrants issued in connection with the Public Offering have been measured based on the listed market price of such Public Warrants, a Level 1 measurement. For the period ended March 31, 2021, the Company recognized a gain in the statement of operations resulting from a decrease in the fair value of the warrant liability of $10,200,787 presented as change in fair value of warrant liability.
The estimated fair value of the Private Placement Warrants was determined using a Black-Scholes-Merton model with Level 3 inputs. Inherent in a Black-Scholes-Merton model are assumptions related to expected life (term), expected stock price, volatility, risk-free interest rate and dividend yield. The Company estimates the volatility of its Class A common stock warrants based on implied volatility from the Companys traded warrants and from historical volatility of select peer companies Class A common stock that matches the expected remaining life of the warrants. The risk-free interest rate is based on the U.S. Treasury zero-coupon yield curve on the grant date for a maturity similar to the expected remaining life of the warrants. The expected life of the warrants is assumed to be equivalent to their remaining contractual term. The dividend rate is based on the historical rate, which the Company anticipates remaining at zero.
The following table provides quantitative information regarding Level 3 fair value measurements inputs:
|
As of March 31, 2021 |
As of December 31, 2020 |
|||||||
| Stock price |
$ | 10.43 | $ | 10.90 | ||||
| Strike Price |
$ | 11.50 | $ | 11.50 | ||||
| Term (in years) |
5.63 | 5.75 | ||||||
| Volitility |
28.00 | % | 28.30 | % | ||||
| Risk-free interest rate |
1.07 | % | 0.47 | % | ||||
| Dividend yield |
0.00 | % | 0.00 | % | ||||
| Fair value |
$ | 2.38 | $ | 2.79 | ||||
The change in the fair value of the warrants measured with Level 3 inputs for the three months ended March 31, 2021 is summarized as follows:
| Value of warrant liability measured with Level 3 inputs at December 31, 2020 |
$ | 23,676,615 | ||
| Change in fair value of warrant liability measured with Level 3 inputs |
(3,450,787 | ) | ||
| Transfer in/out |
| |||
|
|
|
|||
| Value of warrant liability measured with Level 3 inputs at March 31, 2021 |
$ | 20,225,828 | ||
|
|
|
Note 7Subsequent Events
Management has performed an evaluation of subsequent events through the date of issuance of the financial statements, noting no items which require adjustment or disclosure.
F-36
Table of Contents
Mirion Technologies (TopCo), Ltd.
Consolidated Financial Statements
As of June 30, 2020 and 2019 and for the years ended June 30, 2020, 2019, and 2018
Contents
| F-38 | ||||
| Consolidated Financial Statements |
||||
| F-39 | ||||
| F-40 | ||||
| F-41 | ||||
| F-42 | ||||
| F-43 | ||||
| F-44 | ||||
| F-86 | ||||
F-37
Table of Contents
REPORT OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM
To the Board of Directors and Stockholders of Mirion Technologies (TopCo), Ltd.
Opinion on the Financial Statements
We have audited the accompanying consolidated balance sheets of Mirion Technologies (TopCo), Ltd. and its subsidiaries (the Company) as of June 30, 2020 and 2019, the related consolidated statements of operations, comprehensive loss, stockholders equity, and cash flows for each of the three years in the period ended June 30, 2020 and the related notes and the financial statement schedules (collectively referred to as the financial statements). In our opinion, the financial statements present fairly, in all material respects, the financial position of the Company as of June 30, 2020 and 2019, and the results of its operations and its cash flows for each of the three years in the period ended June 30, 2020, in conformity with accounting principles generally accepted in the United States of America.
Basis for Opinion
These financial statements are the responsibility of the Companys management. Our responsibility is to express an opinion on the Companys financial statements based on our audits. We are a public accounting firm registered with the Public Company Accounting Oversight Board (United States) (PCAOB) and are required to be independent with respect to the Company in accordance with the U.S. federal securities laws and the applicable rules and regulations of the Securities and Exchange Commission and the PCAOB.
We conducted our audits in accordance with the standards of the PCAOB and in accordance with auditing standards generally accepted in the United States of America. Those standards require that we plan and perform the audit to obtain reasonable assurance about whether the financial statements are free of material misstatement, whether due to error or fraud. The Company is not required to have, nor were we engaged to perform, an audit of its internal control over financial reporting. As part of our audits, we are required to obtain an understanding of internal control over financial reporting but not for the purpose of expressing an opinion on the effectiveness of the Companys internal control over financial reporting. Accordingly, we express no such opinion.
Our audits included performing procedures to assess the risks of material misstatement of the financial statements, whether due to error or fraud, and performing procedures that respond to those risks. Such procedures included examining, on a test basis, evidence regarding the amounts and disclosures in the financial statements. Our audits also included evaluating the accounting principles used and significant estimates made by management, as well as evaluating the overall presentation of the financial statements. We believe that our audits provide a reasonable basis for our opinion.
Emphasis of Matter
As discussed in Note 8 to the consolidated financial statements, the Company has entered into significant transactions with related parties. Accordingly, the consolidated financial statements may not be indicative of the financial conditions or results of operations that would have been achieved had the Company operated without such related party affiliations. Our opinion is not modified with respect to this matter.
/s/ Deloitte & Touche LLP
San Jose, California
June 29, 2021
We have served as the Companys auditor since 2015.
F-38
Table of Contents
Mirion Technologies (TopCo), Ltd.
Consolidated Balance Sheets
(In millions, except share data)
| June 30, 2020 | June 30, 2019 | |||||||
| ASSETS |
||||||||
| Current assets: |
||||||||
| Cash and cash equivalents |
$ | 118.4 | $ | 35.8 | ||||
| Restricted cash |
1.1 | 1.4 | ||||||
| Accounts receivable, net of allowance for doubtful accounts |
97.3 | 99.1 | ||||||
| Costs in excess of billings on uncompleted contracts |
59.5 | 56.9 | ||||||
| Inventories |
90.2 | 89.3 | ||||||
| Deferred cost of revenue |
6.5 | 3.0 | ||||||
| Prepaid expenses and other currents assets |
16.7 | 15.1 | ||||||
|
|
|
|
|
|||||
| Total current assets |
389.7 | 300.6 | ||||||
| Property, plant, and equipment, net |
75.2 | 65.8 | ||||||
| Goodwill |
522.6 | 511.6 | ||||||
| Intangible assets, net |
248.3 | 267.6 | ||||||
| Restricted cash |
0.5 | 0.4 | ||||||
| Other assets |
7.5 | 10.0 | ||||||
|
|
|
|
|
|||||
| Total assets |
$ | 1,243.8 | $ | 1,156.0 | ||||
|
|
|
|
|
|||||
| LIABILITIES AND STOCKHOLDERS DEFICIT |
||||||||
| Current liabilities: |
||||||||
| Accounts payable |
$ | 38.7 | $ | 39.8 | ||||
| Deferred contract revenue |
39.6 | 40.8 | ||||||
| Notes payable to third-parties, current |
41.1 | 10.5 | ||||||
| Accrued expenses and other current liabilities |
64.1 | 52.2 | ||||||
|
|
|
|
|
|||||
| Total current liabilities |
183.5 | 143.3 | ||||||
| Notes payable to related parties, non-current |
987.1 | 885.2 | ||||||
| Notes payable to third-parties, non-current |
669.8 | 579.6 | ||||||
| Interest accrued on notes payable to related parties |
56.4 | 50.2 | ||||||
| Deferred income taxes and other liabilities |
63.5 | 85.4 | ||||||
|
|
|
|
|
|||||
| Total liabilities |
1,960.3 | 1,743.7 | ||||||
| Commitments and contingencies (Note 9) |
||||||||
| Stockholders deficit: |
||||||||
| A Ordinary shares, $0.01 nominal value, 3,000,000 shares authorized, 1,483,795 issued and outstanding at both June 30, 2020 and June 30, 2019 |
| | ||||||
| B Ordinary shares, $0.01 nominal value, 7,000,000 shares authorized, 5,353,970 issued and outstanding at both June 30, 2020 and June 30, 2019 |
0.1 | 0.1 | ||||||
| Additional paid-in capital |
9.5 | 9.3 | ||||||
| Receivable from Employees for purchase of Common Stock |
(2.7 | ) | (2.5 | ) | ||||
| Accumulated deficit |
(729.7 | ) | (610.6 | ) | ||||
| Accumulated other comprehensive income |
4.1 | 13.4 | ||||||
|
|
|
|
|
|||||
| Mirion Technologies (TopCo), Ltd. stockholders deficit |
(718.7 | ) | (590.3 | ) | ||||
| Noncontrolling interests |
2.2 | 2.6 | ||||||
|
|
|
|
|
|||||
| Total stockholders deficit |
(716.5 | ) | (587.7 | ) | ||||
|
|
|
|
|
|||||
| Total liabilities and stockholders deficit |
$ | 1,243.8 | $ | 1,156.0 | ||||
|
|
|
|
|
|||||
The accompanying notes are an integral part of these consolidated financial statements.
F-39
Table of Contents
Mirion Technologies (TopCo), Ltd.
Consolidated Statements of Operations
(In millions, except per share data)
| Years Ended June 30, |
||||||||||||
| 2020 | 2019 | 2018 | ||||||||||
| Revenues: |
||||||||||||
| Product |
$ | 353.0 | $ | 325.7 | $ | 332.9 | ||||||
| Service |
125.2 | 114.4 | 111.2 | |||||||||
|
|
|
|
|
|
|
|||||||
| Total revenues |
478.2 | 440.1 | 444.1 | |||||||||
| Cost of revenues: |
||||||||||||
| Product |
216.8 | 190.7 | 212.8 | |||||||||
| Service |
64.4 | 61.2 | 62.9 | |||||||||
|
|
|
|
|
|
|
|||||||
| Total cost of revenues |
281.2 | 251.9 | 275.7 | |||||||||
|
|
|
|
|
|
|
|||||||
| Gross profit |
197.0 | 188.2 | 168.4 | |||||||||
| Operating expenses: |
||||||||||||
| Selling, general and administrative |
158.1 | 145.4 | 153.8 | |||||||||
| Research and development |
15.9 | 14.0 | 19.3 | |||||||||
|
|
|
|
|
|
|
|||||||
| Total operating expenses |
174.0 | 159.4 | 173.1 | |||||||||
|
|
|
|
|
|
|
|||||||
| Income (loss) from operations |
23.0 | 28.8 | (4.7 | ) | ||||||||
| Other expense (income): |
||||||||||||
| Third party interest expense |
41.5 | 47.7 | 43.3 | |||||||||
| Related party interest expense |
107.7 | 95.8 | 85.6 | |||||||||
| Loss on debt extinguishment |
| 12.8 | | |||||||||
| Foreign currency (gain) loss, net |
(0.6 | ) | (3.2 | ) | 5.3 | |||||||
| Other (income) expense, net |
(1.0 | ) | 1.9 | 1.6 | ||||||||
|
|
|
|
|
|
|
|||||||
| Loss before benefit from income taxes |
(124.6 | ) | (126.2 | ) | (140.5 | ) | ||||||
| Benefit from income taxes |
(5.5 | ) | (4.2 | ) | (36.8 | ) | ||||||
|
|
|
|
|
|
|
|||||||
| Net loss |
(119.1 | ) | (122.0 | ) | (103.7 | ) | ||||||
| Income (loss) attributable to noncontrolling interests |
| | (0.3 | ) | ||||||||
|
|
|
|
|
|
|
|||||||
| Net loss attributable to Mirion Technologies (TopCo), Ltd. stockholders |
$ | (119.1 | ) | $ | (122.0 | ) | $ | (103.4 | ) | |||
|
|
|
|
|
|
|
|||||||
| Net loss per common share attributable to Mirion Technologies (TopCo) Ltd. stockholdersbasic and diluted |
$ | (18.45 | ) | $ | (19.36 | ) | $ | (17.44 | ) | |||
|
|
|
|
|
|
|
|||||||
| Weighted average common shares outstandingbasic and diluted |
6.453 | 6.300 | 5.929 | |||||||||
|
|
|
|
|
|
|
|||||||
The accompanying notes are an integral part of these consolidated financial statements.
F-40
Table of Contents
Mirion Technologies (TopCo), Ltd.
Consolidated Statements of Comprehensive Loss
(In millions)
| Years Ended June 30, | ||||||||||||
| 2020 | 2019 | 2018 | ||||||||||
| Net loss |
$ | (119.1 | ) | $ | (122.0 | ) | $ | (103.7 | ) | |||
| Other comprehensive loss, net of tax: |
||||||||||||
| Foreign currency translation, net of tax |
(9.3 | ) | (15.1 | ) | 12.0 | |||||||
| Unrecognized actuarial gain (loss) and prior service benefit, net of tax |
| (1.5 | ) | (0.1 | ) | |||||||
|
|
|
|
|
|
|
|||||||
| Other comprehensive income (loss), net of tax |
(9.3 | ) | (16.6 | ) | 11.9 | |||||||
|
|
|
|
|
|
|
|||||||
| Comprehensive loss |
(128.4 | ) | (138.6 | ) | (91.8 | ) | ||||||
| Less: Comprehensive income (loss) attributable to noncontrolling interest |
| | (0.3 | ) | ||||||||
|
|
|
|
|
|
|
|||||||
| Comprehensive loss attributable to Mirion Technologies (TopCo), Ltd. stockholders |
$ | (128.4 | ) | $ | (138.6 | ) | $ | (91.5 | ) | |||
|
|
|
|
|
|
|
|||||||
The accompanying notes are an integral part of these consolidated financial statements.
F-41
Table of Contents
Mirion Technologies (TopCo), Ltd.
Consolidated Statements of Stockholders Deficit
(In millions, except share amounts)
| A Ordinary Shares |
A Ordinary Amount |
B Ordinary Shares |
B Ordinary Amount |
Additional Paid-In Capital |
Receivable from Employees for purchase of Common Stock |
Accumulated Deficit |
Accumulated Other Comprehensive Income (Loss) |
Noncontrolling Interests |
Total Stockholders Deficit |
|||||||||||||||||||||||||||||||
| Balance July 1, 2017 |
1,483,795 | $ | | 5,353,970 | $ | 0.1 | $ | 7.1 | | $ | (385.2 | ) | $ | 18.1 | $ | 3.5 | $ | (356.4 | ) | |||||||||||||||||||||
| Dividend |
| | | | | | | | (0.2 | ) | (0.2 | ) | ||||||||||||||||||||||||||||
| Distribution to noncontrolling interests |
| | | | | | | | (0.4 | ) | (0.4 | ) | ||||||||||||||||||||||||||||
| Share-based compensation expense |
| | | | 0.2 | | | | | 0.2 | ||||||||||||||||||||||||||||||
| Receivable from Employees |
| | | | | (0.2 | ) | | | | (0.2 | ) | ||||||||||||||||||||||||||||
| Net loss |
| | | | | | (103.4 | ) | | (0.3 | ) | (103.7 | ) | |||||||||||||||||||||||||||
| Other comprehensive income |
| | | | | | | 11.9 | | 11.9 | ||||||||||||||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||||||||
| Balance June 30, 2018 |
1,483,795 | | 5,353,970 | 0.1 | 7.3 | (0.2 | ) | (488.6 | ) | 30.0 | 2.6 | (448.8 | ) | |||||||||||||||||||||||||||
| Contribution from noncontrolling interests |
| | | | | | | | 0.1 | 0.1 | ||||||||||||||||||||||||||||||
| Distribution to noncontrolling interests |
| | | | | | | | (0.1 | ) | (0.1 | ) | ||||||||||||||||||||||||||||
| Share-based compensation expense |
| | | | 0.1 | | | | | 0.1 | ||||||||||||||||||||||||||||||
| Receivable from Employees |
| | | | 1.9 | (2.3 | ) | | | | (0.4 | ) | ||||||||||||||||||||||||||||
| Net loss |
| | | | | | (122.0 | ) | | | (122.0 | ) | ||||||||||||||||||||||||||||
| Other comprehensive loss |
| | | | | | | (16.6 | ) | | (16.6 | ) | ||||||||||||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||||||||
| Balance June 30, 2019 |
1,483,795 | | 5,353,970 | 0.1 | 9.3 | (2.5 | ) | (610.6 | ) | 13.4 | 2.6 | (587.7 | ) | |||||||||||||||||||||||||||
| Distribution to noncontrolling interests |
| | | | | | | | (0.4 | ) | (0.4 | ) | ||||||||||||||||||||||||||||
| Share-based compensation expense |
| | | | 0.2 | | | | | 0.2 | ||||||||||||||||||||||||||||||
| Receivable from Employees |
| | | | | (0.2 | ) | | | | (0.2 | ) | ||||||||||||||||||||||||||||
| Net loss |
| | | | | | (119.1 | ) | | | (119.1 | ) | ||||||||||||||||||||||||||||
| Other comprehensive loss |
| | | | | | | (9.3 | ) | | (9.3 | ) | ||||||||||||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||||||||
| Balance June 30, 2020 |
1,483,795 | $ | | 5,353,970 | $ | 0.1 | $ | 9.5 | $ | (2.7 | ) | $ | (729.7 | ) | $ | 4.1 | $ | 2.2 | $ | (716.5 | ) | |||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||||||||
The accompanying notes are an integral part of these consolidated financial statements.
F-42
Table of Contents
Mirion Technologies (TopCo), Ltd.
Consolidated Statements of Cash Flows
(In millions)
| Years Ended | ||||||||||||
| June 30, | ||||||||||||
| 2020 | 2019 | 2018 | ||||||||||
| OPERATING ACTIVITIES: |
||||||||||||
| Net loss |
$ | (119.1 | ) | $ | (122.0 | ) | $ | (103.7 | ) | |||
| Adjustments to reconcile net loss to net cash provided by operating activities: |
||||||||||||
| Accrual of in-kind interest on notes payable to related parties |
107.7 | 95.6 | 85.3 | |||||||||
| Depreciation and amortization expense |
68.4 | 69.5 | 77.0 | |||||||||
| Share-based compensation expense |
0.2 | 0.1 | 0.2 | |||||||||
| Loss on debt extinguishment |
| 12.8 | | |||||||||
| Amortization of debt issuance costs |
2.6 | 3.6 | 4.1 | |||||||||
| Provision for doubtful accounts |
0.6 | 0.5 | (0.9 | ) | ||||||||
| Inventory obsolescence write down |
1.9 | | 3.7 | |||||||||
| Change in deferred income taxes |
(15.5 | ) | (16.1 | ) | (48.5 | ) | ||||||
| Loss on disposal of property, plant and equipment |
0.4 | 1.2 | 2.6 | |||||||||
| Loss (gain) on foreign currency transactions |
(1.7 | ) | 2.7 | 2.5 | ||||||||
| Other |
(0.9 | ) | (0.1 | ) | 0.9 | |||||||
| Changes in operating assets and liabilities: |
||||||||||||
| Accounts receivable |
3.8 | 10.6 | 10.8 | |||||||||
| Costs in excess of billings on uncompleted contracts |
(2.9 | ) | (8.1 | ) | (22.1 | ) | ||||||
| Inventories |
4.3 | (7.8 | ) | (6.0 | ) | |||||||
| Deferred cost of revenue |
(3.5 | ) | (0.2 | ) | 0.7 | |||||||
| Prepaid expenses and other current assets |
(1.6 | ) | 1.4 | 0.7 | ||||||||
| Accounts payable |
(2.5 | ) | (2.7 | ) | 4.4 | |||||||
| Accrued expenses and other current liabilities |
7.3 | (13.1 | ) | 2.4 | ||||||||
| Deferred contract revenue |
(1.7 | ) | (8.4 | ) | 4.6 | |||||||
| Other assets |
0.2 | 0.5 | | |||||||||
| Other liabilities |
(8.5 | ) | (5.3 | ) | (3.1 | ) | ||||||
|
|
|
|
|
|
|
|||||||
| Net cash provided by operating activities |
39.5 | 14.7 | 15.6 | |||||||||
|
|
|
|
|
|
|
|||||||
| INVESTING ACTIVITIES: |
||||||||||||
| Acquisitions of businesses, net of cash and cash equivalents acquired |
(55.7 | ) | (9.1 | ) | | |||||||
| Purchases of property, plant, and equipment and badges |
(19.9 | ) | (16.5 | ) | (12.6 | ) | ||||||
|
|
|
|
|
|
|
|||||||
| Net cash used in investing activities |
(75.6 | ) | (25.6 | ) | (12.6 | ) | ||||||
|
|
|
|
|
|
|
|||||||
| FINANCING ACTIVITIES: |
||||||||||||
| Borrowings from notes payable to third-parties, net of discount and issuance costs |
98.8 | 596.8 | | |||||||||
| Principal repayments |
(13.4 | ) | (560.2 | ) | (4.9 | ) | ||||||
| Deferred finance costs |
| (8.1 | ) | | ||||||||
| Borrowing on revolving term loan |
80.0 | | | |||||||||
| Payment on revolving term loan |
(45.0 | ) | (13.0 | ) | | |||||||
| Payment of contingent considerations |
(2.0 | ) | | | ||||||||
| Contribution from noncontrolling interests |
| 0.1 | | |||||||||
| Distributions to noncontrolling interests |
(0.4 | ) | (0.1 | ) | (0.4 | ) | ||||||
| Dividends to noncontrolling interests Other financing |
|
0.9 |
|
|
(0.5 |
) |
|
(0.2 |
)
|
|||
|
|
|
|
|
|
|
|||||||
| Net cash provided by (used in) financing activities |
118.9 | 15.0 | (5.5 | ) | ||||||||
|
|
|
|
|
|
|
|||||||
| Effect of exchange rate changes on cash, cash equivalents, and restricted cash |
(0.4 | ) | (2.4 | ) | 0.4 | |||||||
|
|
|
|
|
|
|
|||||||
| Net increase (decrease) in cash, cash equivalents, and restricted cash |
82.4 | 1.7 | (2.1 | ) | ||||||||
| Cash, cash equivalents, and restricted cash at beginning of year |
37.6 | 35.9 | 38.0 | |||||||||
|
|
|
|
|
|
|
|||||||
| Cash, cash equivalents, and restricted cash at end of year |
$ | 120.0 | $ | 37.6 | $ | 35.9 | ||||||
|
|
|
|
|
|
|
|||||||
The accompanying notes are an integral part of these consolidated financial statements.
F-43
Table of Contents
Mirion Technologies (TopCo), Ltd.
Notes to Consolidated Financial Statements
1. Nature of Business and Summary of Significant Accounting Policies
Nature of Business
Mirion Technologies (TopCo), Ltd. (Mirion TopCo or the Company or us) is a global provider of radiation detection, measurement, analysis, and monitoring products and services to the medical, nuclear, and defense end markets. We provide products and services through our two operating and reportable segments; (i) Medical, and (ii) Industrial. The medical segment provides radiation oncology quality assurance, delivering patient safety solutions for diagnostic imaging and radiation therapy centers around the world, dosimetry solutions for monitoring the total amount of radiation medical staff members are exposed to over time, radiation therapy quality assurance solutions for calibrating and verifying imaging and treatment accuracy, and radionuclide therapy products for nuclear medicine applications such as shielding, product handling, medical imaging furniture, and rehabilitation products. The industrial segment provides robust, field ready personal radiation detection and identification equipment for defense applications and radiation detection and analysis tools for power plants, labs, and research applications. Nuclear power plant product offerings are used for the full nuclear power plant lifecycle including core detectors and essential measurement devices for new build, maintenance, decontamination and decommission equipment for monitoring and control during fuel dismantling and remote environmental monitoring.
The Company is headquartered in Atlanta, Georgia and has operations in the United States, Canada, the United Kingdom, France, Germany, Finland, China, Belgium, Netherlands, Estonia, and Japan.
Basis of Presentation and Principles of Consolidation
The accompanying Consolidated Financial Statements and Notes to Consolidated Financial Statements have been prepared in accordance with accounting principles generally accepted in the United States (US GAAP). The Consolidated Financial Statements include the accounts of the Company and its wholly owned and majority-owned or controlled subsidiaries. For consolidated subsidiaries where our ownership is less than 100%, the portion of the net income or loss allocable to noncontrolling interests is reported as Income (Loss) attributable to noncontrolling interests in the Consolidated Statements of Operations. All intercompany accounts and transactions have been eliminated in consolidation.
Use of Estimates
Management estimates and judgments are an integral part of financial statements prepared in accordance with accounting principles generally accepted in the United States of America (GAAP). We believe that the critical accounting policies listed below address the more significant estimates required of Management when preparing our consolidated financial statements in accordance with GAAP. We consider an accounting estimate critical if changes in the estimate may have a material impact on our financial condition or results of operations. We believe that the accounting estimates employed are appropriate and resulting balances are reasonable; however, actual results could differ from the original estimates, requiring adjustment to these balances in future periods. The accounting policies that reflect our more significant estimates, judgments and assumptions and which we believe are the most critical to aid in fully understanding and evaluating our reported financial results include, but are not limited to: goodwill and intangible assets; standalone selling prices for revenue arrangements with multiple elements and estimated progress toward completion for certain revenue contracts; uncertain tax positions and tax valuation allowances.
Cash and Cash Equivalents
The Company considers all cash on deposit and money market accounts purchased with original maturities of three months or less to be cash and cash equivalents. Cash equivalents primarily consist of amounts held in interest-bearing money market accounts that are readily convertible to cash.
F-44
Table of Contents
The Company maintains cash in bank deposit accounts that, at times, may exceed the insured limits of the local country, which may lead to a concentration of credit risk. Substantially all of the Companys cash and cash equivalent balances were deposited with financial institutions which management has determined to be high credit quality institutions. The Company has not experienced any losses in such accounts.
Restricted Cash
The Company maintains restricted cash and cash equivalent accounts with various financial institutions to support performance bonds with irrevocable letters of credit for contractual obligations to certain customers. As of June 30, 2020 and 2019, combined current and non-current restricted cash on the consolidated balance sheets was $1.6 million, and $1.8 million, respectively.
Accounts Receivable and Allowance for Doubtful Accounts
Accounts receivable are recorded at the invoiced amount and do not bear interest. The allowance for doubtful accounts is based on the Companys assessment of the collectability of customer accounts. The Company regularly reviews the allowance by considering factors such as historical experience, credit quality, the age of the accounts receivable balances and current economic conditions that may affect a customers ability to pay. The allowance for doubtful accounts was $1.9 million and $1.7 million as of June 30, 2020 and 2019, respectively.
Inventories
Inventories are stated at the lower of cost or net realizable value. Cost is computed using actual costs or standard costs that approximate actual cost, determined on a first-in, first-out basis. A portion of the inventory relates to evaluation units located at customer locations to facilitate customer tests prior to purchasing. Inventories also include completed products and in-process customer projects for which the related revenue has been deferred pending delivery, completion of services or determination that all customer-specific acceptance criteria have been met. Inventory in excess of expected future demand or obsolete inventory is written down to its estimated realizable value based on future demand forecasts and historical demand trends.
Deferred Cost of Revenue
Deferred cost of revenue consists of the direct costs associated with production for identified projects for which the revenue has been deferred in accordance with the Companys revenue recognition policies. Deferred costs are recognized as cost of revenues in the same period that the related revenues are recognized.
Property, Plant, and Equipment
Property, plant, and equipment are carried at cost, net of accumulated depreciation and amortization. Property, plant and equipment acquired through the acquisition of a business are recorded at their estimated fair value at the date of acquisition.
Depreciation is computed when an asset is placed into service using the straight-line method over the estimated useful life of the asset. The Company capitalizes costs incurred in the acquisition and development of software for internal use, including the costs of software, materials, consultants, and payroll-related costs of employees incurred in developing internal-use computer software. Development costs related to internal-use software are amortized using the straight-line method over the shorter of the software license or the estimated useful life of the software. Leasehold improvements are amortized using the straight-line method over the shorter of the related lease term or the estimated useful life of the improvements. Repair and maintenance costs are expensed as incurred.
Estimated useful lives are periodically reviewed and, when appropriate, changes to estimates are made prospectively. When certain events or changes in operating conditions occur, asset lives may be adjusted, and an
F-45
Table of Contents
impairment assessment may be performed on the recoverability of the carrying amounts. Refer to Note 5 for disclosure of estimated useful lives.
When property, plant equipment is retired or otherwise disposed of, the cost and related accumulated depreciation are removed from the balance sheet. Any difference between the net asset value and the proceeds on sale are charged or credited to income.
Other Current Assets
Other current assets are primarily comprised of various prepaid assets, short-term marketable securities, and income tax receivables.
Business Combinations
We account for business acquisitions in accordance with the Financial Accounting Standards Board (FASB) Accounting Standards Codification (ASC) 805, Business Combinations. This standard requires the acquiring entity in a business combination to recognize all the assets acquired and liabilities assumed in the transaction and establishes the acquisition date fair value as the measurement objective for all assets acquired and liabilities assumed in a business combination. Certain provisions of this standard prescribe, among other things, the determination of acquisition date fair value of consideration paid in a business combination (including contingent consideration) and the exclusion of transaction and acquisition-related restructuring costs from acquisition accounting.
The determination of the fair value of assets acquired and liabilities assumed involves assessments of factors such as the expected future cash flows associated with individual assets and liabilities and appropriate discount rates at the closing date of the acquisition. For non-observable market values, the Company determines fair value using acceptable valuation principles (e.g., multiple excess earnings, relief from royalty and cost methods).
Results of operations for acquired companies are included in our consolidated results of operations from the date of acquisition.
Goodwill
Goodwill represents the excess of the purchase price paid over the estimated fair value of the net assets acquired and liabilities assumed in the acquisition of a business.
Goodwill has an indefinite useful life, and is not amortized, but instead tested for impairment annually during the fiscal year fourth quarter or more often if events or changes in circumstances indicate that the carrying amount may exceed fair value as set forth in ASC 350, IntangiblesGoodwill and Other. The Company tests for goodwill impairment at the reporting unit level, which is an operating segment or one level below an operating segment. The amount of goodwill acquired in a business combination that is assigned to one or more reporting units as of the acquisition date is the excess of the purchase price of the acquired businesses (or portion thereof) included in the reporting unit, over the fair value assigned to the individual assets acquired or liabilities assumed from a market participant perspective. Goodwill is assigned to the reporting unit(s) expected to benefit from the synergies of the combination even though other assets or liabilities of the acquired entity may not be assigned to that reporting unit.
ASC 350 allows an optional qualitative assessment as part of annual impairment testing, prior to a quantitative assessment test, to determine whether it is more likely than not that the fair value of a reporting unit exceeds its carrying amount. In performing the quantitative test, which compares the fair value of a reporting unit with its carrying amount, we determine the fair value of each reporting unit by estimating the present value of expected future cash flows, discounted by the applicable discount rate, and peer company multiples. If the carrying value exceeds the fair value, the Company recognizes an impairment loss in the amount equal to the excess, not to exceed the total amount of goodwill allocated to that reporting unit.
F-46
Table of Contents
Based upon our review and analysis, no impairments were deemed to have occurred during any of the years presented. Refer to Note 7 for further detail.
Intangible Assets
Intangible assets relate to the value associated with our developed technology, customer relationships, backlog, and trade names at the time of acquisition through business combinations.
The Company determined the fair value of intangible assets acquired through an income approach, using the excess earnings method for customer relationships and backlog. Under the excess earnings method, an intangible assets fair value is equal to the present value of the incremental after-tax cash flows attributable solely to the intangible asset over its remaining useful life. The relief from royalty method was used to determine the fair value of developed technology and tradename. The valuation models were based on estimates of future operating projections of the acquired business and rights to sell products as well as judgments on the discount rates used and other variables. We determined the forecasts based on a number of factors, including our best estimate of near-term net sales expectations and long-term projections, which include review of internal and independent market analyses. The discount rate used was representative of the weighted average cost of capital.
The customer relationships definite lived intangible assets are amortized using the double declining balance method with estimated useful lives ranging from 6 to 17 years, while all other definite lived intangible assets are amortized on a straight-line basis over their estimated useful lives, ranging from 1 to 7 years. The Company regularly evaluates the amortization period assigned to each intangible asset to ensure that there have not been any events or circumstances that warrant revised estimates of useful lives.
Impairment of Long-Lived Assets
The Company reviews long-lived assets and definite-lived intangible assets for impairment whenever events or changes in circumstances indicate that the carrying amount of an asset may not be recoverable. If an evaluation of recoverability is required, the estimated undiscounted future cash flows associated with the asset group are compared to the asset groups carrying amount to determine if a write-down is required. If the undiscounted cash flows are less than the carrying amount, an impairment loss is recorded to the extent that the carrying amount exceeds the fair value. No impairment was recorded during fiscal years ended June 30, 2020, 2019 and 2018.
Facility and Equipment Decommissioning Liabilities
The Company has asset retirement obligations (ARO) consisting primarily of equipment and facility decommissioning costs. The estimated fair value of these ARO liabilities is recognized in the period in which the liability is generated and a corresponding increase to the carrying valueof the related asset is recorded and depreciated over the useful life of the asset. The Companys estimates of its ultimate AROs could change because of changes in regulations, the extent of environmental remediation required, the means of reclamation, cost estimates, exit or disposal activities or time period estimates.
ARO liabilities totaled $4.0 million at both June 30, 2020 and 2019 and were included in deferred income taxes and other liabilities on the consolidated balance sheets. Accretion expense related to these liabilities was not material for the fiscal years ended June 30, 2020, 2019 and 2018, respectively.
Product Warranty
The Company offers warrants against material defects for most of its products for a specified time period, usually twelve to twenty-four months from delivery or acceptance. When the related revenues are recognized, the Company provides for the estimated future costs of warranty obligations in cost of revenues. The accrued warranty costs represent the Companys best estimate at the time of sale of the total costs that will be incurred to repair or replace product parts that fail while still under warranty.
F-47
Table of Contents
The amount of the accrued estimated warranty cost obligations for established products is based on historical experience as to product failures adjusted for current information on repair costs. For new products, estimates include the historical experience of similar products, as well as a reasonable allowance for warranty expenses associated with the new products. On a quarterly basis, the Company reviews the accrued warranty costs and updates the historical warranty cost trends, if required.
Revenue Recognition
Prior to July 1, 2019, the Company recognized revenue based on ASC 605, when there was persuasive evidence of an arrangement, product delivery had occurred or services had been provided, the sales price was fixed or determinable and collectability was reasonably assured. Beginning on July 1, 2019, the Company recognizes revenue based on ASC 606 as performance obligations are satisfied by transferring control of promised goods or service to customers in an amount that reflects the consideration to which the company expects to be entitled in exchange for those goods or services. See Recently Adopted Accounting Guidance below for a discussion of the change in revenue recognition accounting that became effective on July 1, 2019.
ASC 606
The Company recognizes revenue from arrangements that include performance obligations to design, engineer, manufacture, deliver, and install products. The Company identifies a performance obligation for each promise in a contract to transfer a distinct good or service to the customer. As part of its assessment, the Company considers all goods and/or services promised in the contract, regardless of whether they are explicitly stated or implied by customary business practices. The Companys contracts may contain either a single performance obligation, including the promise to transfer individual goods or services that are not separately distinct within the context of the respective contracts, or multiple performance obligations. For contracts that contain multiple performance obligations, the Company allocates the consideration to which it expects to be entitled to each performance obligation based on relative standalone selling prices and recognizes the related revenue when or as control of each individual performance obligation is transferred to customers. The Company does not assess whether promised goods or services are performance obligations if they are immaterial in the context of the contract with the customer. The Company combines multiple contracts entered into at or around the same time with a customer if the contracts are negotiated as a package with a single commercial objective, the consideration paid under the contracts depends on the price or performance of the other contract, or if the goods or services promised in the contracts are a single performance obligation. Service revenues (service-type warranty, post contract support, installation, and subscription-based services) are recognized over time as the customers receive and consume benefits of such services simultaneously. Assurance-type warranties guarantee that a product complies with agreed-upon specifications and accordingly are not separate performance obligations. A provision for these warranties is recognized in the period during which the associated revenue is recognized. In most cases, installation services represent a separate performance obligation recognized over time if the customer benefits from the service. When the product and installation service are determined to be a combined performance obligation, revenue is recognized over time as the installation is performed and included in product revenue in the consolidated statement of operations.
Variable consideration such as rebates, sales discounts and sales returns are estimated and treated as a reduction of revenue in the same period the related revenue is recognized. These are estimated based on contractual terms, historical practices, and current trends, and are adjusted as new information becomes available. Revenues exclude any taxes that the Company collects from customers and remits to tax authorities. Amounts billed to customer for shipping and handling are included in revenue, while the related shipping and handling costs are reflected in cost of products in the period in which revenue is recognized. The Company has elected a practical expedient under ASC 606 that allows for shipping and handling activities that occur after the customer has obtained control of a good to be accounted for as a fulfillment cost. The Company does not adjust the promised amount of consideration for the effects of a significant financing component, if, at contract inception, the Company expects the period between the time when the Company transfers a promised good or service to the customer and the time when the customer pays for that good or service will be one year or less.
F-48
Table of Contents
The Company exercises judgment in determining the timing of revenue by analyzing the point in time or the period over which the customer has the ability to direct the use of and obtain substantially all of the remaining benefits of the performance obligation. Typically, over-time revenue recognition is based on the utilization of an input measure used to measure progress, such as costs incurred to date relative to total estimated costs. Changes in total estimated costs are recognized using the cumulative catch-up method of accounting which recognize the cumulative effect of the changes on current and prior periods in the current period. Accordingly, the effect of the changes on future periods of contract performance is recognized as if the revised estimate had been the original estimate. Provisions for estimated losses on uncompleted contracts are made in the period in which such losses are first determined. A significant change in an estimate on one or more contract could have a material effect on the Companys consolidated financial position, results from operations, or cash flows. However, there were no significant changes in estimated contract costs for the year ended June 30, 2020 and 2019.
If a performance obligation does not qualify for over-time revenue recognition, revenue is then recognized at the point-in-time in which control of the distinct good or service is transferred to the customer, typically based upon the terms of delivery.
Certain of the Companys products are sold through distributors and third-party sales representatives under standard agreements whereby distributors purchase products from the Company and resell them to customers. These agreements give distributors the right to sell the Companys products within certain territories and establish minimum order requirements. These arrangements do not provide stock rotation or price protection rights and do not contain extended payment terms. Rights of return are limited to repair or replacement of delivered products that are defective or fail to meet the Companys published specifications. Provisions for these warranty costs are recognized in the same period that the related revenue is recorded similar to other assurance-type warranties.
Revenue derived from passive dosimetry and analytical services is of a subscription nature and is provided to customers on an agreed-upon recurring monthly, quarterly or annual basis. Services are provided to the customer via passive dosimeter badges that the Company supplies to customer personnel. Depending on the type of badge utilized, either customers return the used badges to the Company for analysis, or they obtain the analysis directly via a self-service web portal. The Company believes that badge production, badge wearing, badge analysis and report preparation are not individually distinct and therefore a single performance obligation recognized over time. Revenue is recognized ratably over the service period as the service is continuous, and no other discernible pattern of recognition is evident. Many customers pay for these measuring and monitoring services in advance. The amounts are recorded as deferred contract revenue in the consolidated balance sheets and represent customer deposits invoiced in advance for services to be rendered over the service period, net of a reserve for estimated cancellations.
Payment terms for shipments to end-users are generally net 30 days. Payment terms for distributor shipments may range from 30 to 90 days. Service arrangements commonly call for payments in advance of performing the work (e.g. extended warranty and service contracts), upon completion of contract milestones (e.g. custom development manufacturing), or a combination of each.
The Companys costs to obtain contracts are typically comprised of sales commissions. A majority of these costs relate to revenue that is recognized over a period that is less than one year and as such, the Company has elected a practical expedient under ASC 606 to expense these costs as incurred.
Remaining Performance Obligations
The remaining performance obligations for all open contract as of June 30, 2020 include assembly, delivery, installation, and trainings. The aggregate amount of the transaction price allocated to the remaining performance obligations for all open customer contract as of June 30, 2020 was approximately $601.4 million. The Company expects to recognize approximately 44%, 17% and 14% of the remaining performance obligations as of June 30, 2020 as revenue during the fiscal years 2021, 2022 and 2023, respectively.
F-49
Table of Contents
Disaggregation of Revenues
A disaggregation of the Companys revenues by segment, geographic region and timing of revenue recognition is provided in Note 15.
ASC 605
Prior to July 1, 2019, the Company recognized revenue from sales contracts when there was persuasive evidence of an arrangement, product delivery had occurred or services had been provided, the sales price was fixed or determinable and collectability was reasonably assured. For sales contracts that contain customer-specific acceptance provisions, revenue and the related costs were deferred until the customer had indicated successful completion of site acceptance tests or the Company had otherwise determined that all customer-specific acceptance criteria had been met. Where the Company performed detailed factory acceptance testing on completed products, which, in some instances, was sufficiently extensive and reliable to demonstrate that its products meet the customer-specified objective acceptance criteria set forth in the related sales arrangements. In such instances, the Company recognized revenue based on delivery terms and prior to the receipt of notification of formal acceptance from the customer.
The Company combined a group of contracts as one project if they are closely related and were in substance, part of a single project with an overall project margin. The Company segmented a contract into several projects when they were of different business substance, for example, with different business negotiation, solutions, implementation plans, and margins.
The Company evaluated each deliverable in an arrangement to determine whether they represented separate units of accounting. A deliverable constituted a separate unit of accounting when it had stand-alone value, and for an arrangement that included a general right of return relative to the delivered products or services, when delivery or performance of the undelivered product or service was considered probable and is substantially controlled by the Company. The Company considers a deliverable to have stand-alone value if the product or service is sold separately by the Company or another vendor or could be resold by the customer on a stand-alone basis at an amount that would substantially recover the original purchase price. Further, the revenue arrangements generally do not include a general right of return relative to the delivered products. Where the aforementioned criteria for a separate unit of accounting are not met, the deliverable is combined with the undelivered element(s) and treated as a single unit of accounting for the purposes of allocation of the arrangement consideration and revenue recognition.
When a sales arrangement contains multiple units of accounting, the Company allocates the total arrangement consideration to each separable element of an arrangement based upon the relative selling price of each element. Allocation of the consideration is determined at arrangement inception based on each units relative selling price, which is determined based on a selling price hierarchy. The relative selling price for a deliverable is based on its vendor-specific objective evidence (VSOE) if available, third-party evidence (TPE) if VSOE is not available, or estimated selling price if neither VSOE nor TPE is available. The Company then recognizes revenue on each deliverable in accordance with its policies for product and service revenue recognition. The Company is not typically able to determine VSOE or TPE and therefore uses estimated selling prices to allocate revenue between the elements of the arrangement. The Company establishes its best estimate of the selling price considering multiple factors, including, but not limited to, pricing practices in different geographies and through different sales channels, costs and margin objectives, competitive pricing strategies and general market conditions.
The Company limits the amount of revenue recognition for delivered elements to the amount that is not contingent on the future delivery of products or services or future performance obligations or subject to customer-specific cancellation rights.
For all arrangements, amounts billed to a customer related to shipping and handling are classified as revenue while all costs incurred by the Company for shipping and handling are classified as cost of revenue. Provisions
F-50
Table of Contents
and allowances for discounts to customers, estimated sales returns, service cancellations, and other adjustments are provided for in the same period that the related revenue is recorded.
Certain of the Companys products are sold through distributors and third-party sales representatives under standard agreements whereby distributors purchase products from the Company and resell them to customers. These agreements give distributors the right to sell the Companys products within certain territories and establish minimum order requirements. These arrangements do not provide stock rotation or price protection rights and do not contain extended payment terms. Rights of return are limited to repair or replacement of delivered products that are defective or fail to meet the Companys published specifications. Provisions for these warranty costs are recognized in the same period that the related revenue is recorded.
Revenue from certain fixed-price contracts that involve customization of equipment to customer specifications is recorded using a percentage-of-completion method measured on the cost-to-cost basis. Contract costs include all direct materials and labor costs, as well as indirect costs related to contract performance. Changes in job performance, job conditions, and estimated profitability result in revisions to costs and revenue and are recognized in the period in which the revisions are determined. Provisions for estimated losses on uncompleted contracts are made in the period in which such losses are first determined. Revenue earned in excess of billings on contracts in progress is classified in the consolidated balance sheet as a current asset and included in costs in excess of billings on uncompleted contracts. Amounts billed in excess of revenue earned are classified as a current liability and included in deferred contract revenue.
Revenue derived from passive dosimetry and analytical services is of a subscription nature and is provided to customers on an agreed-upon recurring monthly, quarterly or annual basis. Services are provided to the customer via passive dosimeter badges that the Company supplies to customer personnel. Depending on the type of badge utilized, either customers return the used badges to the Company for analysis, or they obtain the analysis directly via a self-service web portal. The Company believes that badge production, badge wearing, badge analysis and report preparation are all integral to the benefit that the Company provides to its customers and, therefore, the service period is defined as the period over which all of these services are provided. Revenue is recognized on a straight-line basis over the service period as the service is continuous, and no other discernible pattern of recognition is evident. Many customers pay for these measuring and monitoring services in advance. The amounts are recorded as deferred contract revenue in the consolidated balance sheets and represent customer deposits invoiced in advance for services to be rendered over the service period, net of a reserve for estimated cancellations.
Pertinent to ASC 606 and 605
The Company sells its products and services mainly to large, private and governmental organizations in the Americas, Europe, the Middle East and Asia Pacific regions. The Company performs ongoing evaluations of its customers financial condition and limits the amount of credit extended when deemed necessary. The Company generally does not require its customers to provide collateral or other security to support accounts receivable.
One customer represented 9% of consolidated revenue for fiscal years ended June 30, 2020 and 2019 and 11% of consolidated revenue for the fiscal year ended June 30, 2018.
Contract Balances
Revenue earned in excess of billings on contracts in progress (contract assets) are classified in the consolidated balance sheet as a current asset and included in costs in excess of billings on uncompleted contracts. Amounts billed in excess of revenue earned (contract liabilities) are included in deferred contract revenue. For more information, see Note 3.
F-51
Table of Contents
Selling, General, and Administrative
The Companys selling, general and administrative expenses consist of direct and indirect costs related to sales and corporate personnel, facilities, professional services, amortization of intangible assets, share-based compensation, and other operating activities.
Advertising Costs
Advertising costs, which the Company expenses when incurred, were approximately $0.9 million, $0.8 million, and $0.6 million for the years ended June 30, 2020, 2019, and 2018, respectively.
Research and Development
Research and development expenses include costs of developing new products and processes, as well as non-project specific design and engineering costs. Research and development costs are expensed as incurred. Development costs related to software incorporated in the Companys products are not material.
Derivative Activities
The Company uses certain derivative financial instruments to help manage its risk or exposure to changes in interest rates in relation to variable rate debt and foreign currency exchange rate fluctuations. The Company records these derivatives at fair value in the balance sheet as either an asset or a liability and any changes in fair value are recognized in earnings as incurred.
Prior to July 1, 2017, the Company entered into an interest rate swap cap agreement and an interest rate swap agreement. The interest rate swap had an initial notional value of $223.4 million that declined through expiration in March 2018. There was no notional amount at June 30, 2018. The interest rate cap had a notional value of 47.3 million, a cap of 1.5% and also expired in March 2018.
Prior to July 1, 2017, the Company entered into an interest rate swap cap agreement, which had an initial notional value of $135.0 million, a cap of 2.0% and expired September 2019. In addition, in fiscal 2018, the Company executed an interest rate swap agreement that has a fixed notional value of $205.0 million and expired in March 2020. There was no notional amount at June 30, 2020.
As of June 30, 2019, the fair value of these interest rate agreements was ($0.5) million, which was recorded in other liabilities in the consolidated balance sheet. The Company recorded an aggregate net income (loss) of $0.8 million, and ($0.8) million in interest expense in the consolidated statements of operations for the fiscal year ended June 30, 2020 and 2019, respectively, related to these interest rate agreements. The amount recorded in 2018 was not material.
Share-Based Compensation Awards
The Company accounts for share-based compensation related to restricted stock awards granted by Mirion TopCo to certain employees by recognizing the grant date fair value of the awards over the requisite service period, which is equal to the vesting period. Mirion TopCo has an option to buy back the unvested awards upon termination of employment at the lesser of the original issuance price paid by employees or the fair value of the shares on the buy-back date. The Company estimates the value of the restricted stock awards by using the Black-Scholes option valuation model, which requires the use of certain subjective assumptions. Significant assumptions include managements estimates of the estimated stock price volatility, the expected life of the awards and related employee forfeiture rates. For more information, see Note 13.
Accounting for Income Taxes
The Company accounts for income taxes and the related accounts under the asset and liability method. Deferred tax assets and liabilities are recognized for the expected tax consequences of temporary differences between the
F-52
Table of Contents
tax bases of assets and liabilities and their reported amounts. Valuation allowances are recorded to reduce deferred tax assets to the amount that will more likely than not be realized. The Company classifies all deferred tax assets and liabilities, and any related valuation allowance, as non-current in the consolidated balance sheet.
The Company accounts for uncertainty in income taxes using a two-step approach to recognizing and measuring uncertain tax positions. The first step is to evaluate the tax position for recognition by determining if the weight of available evidence indicates that it is more likely than not that the position will be sustained on audit, including resolution of related appeals or litigation processes, if any. The second step is to measure the tax benefit as the largest amount that is more than 50% likely of being realized upon settlement. The Company classifies the liability for unrecognized tax benefits as current in the balance sheet, to the extent that the Company anticipates payment or receipt of cash within one year. Interest and penalties related to uncertain tax positions are recognized in the provision for income taxes.
Defined Benefit Pension Plans and Other Employee Benefits
The Company has defined benefit pension plans that cover certain of its employees in France, Japan, Netherlands, and Germany. The Company also has a postretirement plan that provides for the reimbursement of a portion of medical and life insurance premiums for certain retirees and eligible dependents in the United States. Plan liabilities are revalued annually based on assumptions relating to the discount rates used to measure future obligations and expenses, salary-scale inflation rates, mortality and other assumptions. The selection of assumptions is based on historical trends and known economic and market conditions at the time of valuation; however, actual results may differ from the Companys estimates.
Fair Value Measurements
The Company applies fair value accounting to all financial assets and liabilities that are recognized or disclosed at fair value in the consolidated financial statements on a recurring basis. The fair value of the Companys cash and cash equivalents, restricted cash, accounts receivable, and other current assets and liabilities approximates their carrying amounts due to the relatively short maturity of these items. The fair value of third-party notes payable approximates the carrying value because the interest rates are variable and reflect market rates.
Fair Value of Financial Instruments
The Company categorizes assets and liabilities recorded at fair value in the consolidated balance sheets based upon the level of judgment associated with inputs used to measure their fair value. It is not practicable due to cost and effort for the Company to estimate the fair value of notes issued to related parties primarily due to the nature of their terms relative to the entitys capital structure.
Assets and liabilities carried at fair value are valued and disclosed in one of the following three levels of the valuation hierarchy:
Level 1Inputs are unadjusted quoted prices in active markets for identical assets or liabilities.
Level 2Inputs are quoted prices in active markets for similar assets or liabilities or inputs that can be corroborated by observable market data for substantially the full term of the assets or liabilities.
Level 3Inputs are unobservable and require significant management judgment or estimation.
F-53
Table of Contents
The following table summarizes the financial assets and liabilities of the Company that are measured at fair value on a recurring basis (in millions):
| Fair Value Measurements at June 30, 2020 | ||||||||||||
| Level 1 | Level 2 | Level 3 | ||||||||||
| Assets |
||||||||||||
| Discretionary retirement plan (Note 12) |
$ | 2.4 | $ | 1.1 | $ | | ||||||
| Liabilities |
||||||||||||
| Discretionary retirement plan (Note 12) |
2.4 | 1.1 | | |||||||||
| Fair Value Measurements at June 30, 2019 | ||||||||||||
| Level 1 | Level 2 | Level 3 | ||||||||||
| Assets |
||||||||||||
| Discretionary retirement plan (Note 12) |
$ | 2.9 | $ | | $ | | ||||||
| Liabilities |
||||||||||||
| Discretionary retirement plan (Note 12) |
2.9 | | | |||||||||
| Interest rate swap and cap |
| 0.1 | | |||||||||
The interest rate swap and cap contracts were measured at fair value using a pricing model with significant inputs derived from observable market data.
Foreign Currency Translation
Local currency is the functional currency for substantially all of the Companys foreign operations. Assets and liabilities of foreign operations are translated into U.S. dollars using the exchange rates in effect at the balance sheet reporting date, while income and expenses are translated at the average monthly exchange rates during the period. We record gains and losses from the translation of financial statements in foreign currencies into U.S. dollars in other comprehensive income. The income tax effect of currency translation adjustments related to foreign subsidiaries that are not considered indefinitely reinvested is recorded as a component of deferred taxes with an offset to other comprehensive income. We record gains and losses from changes in exchange rates on transactions denominated in currencies other than each reporting locations functional currency in the consolidated statements of operations for each period.
Concentrations of Risk
Financial instruments that are potentially subject to concentration of credit risk consist primarily of cash and cash equivalents and accounts receivable. The Company maintains cash in bank deposit accounts that, at times, may exceed the insured limits of the local country. The Company has not experienced any losses in such accounts.
The Company sells its products and services mainly to large, private and governmental organizations in the Americas, Europe, the Middle East and Asia Pacific regions. The Company performs ongoing evaluations of its customers financial condition and limits the amount of credit extended when deemed necessary. The Company generally does not require its customers to provide collateral or other security to support accounts receivable. As of June 30, 2020 and 2019, there was no customer that accounted for more than 10% of the accounts receivable balance.
Loss Per Share
Net loss per share of common stock is computed using the two-class method required for multiple classes of common stock and participating securities based upon their respective rights to receive dividends as if all income for the period has been distributed. The rights, including the liquidation, dividend rights, sharing of losses, and voting rights of the Ordinary Class A and Class B common stock are identical. As the rights of both classes of
F-54
Table of Contents
shares are identical, the undistributed earnings are allocated on a proportionate basis and the resulting net loss per share attributed to common stockholders is therefore the same for Ordinary Class A and Class B common stock on an individual or combined basis.
The Companys participating securities include the Companys nonvested Ordinary Class A shares, as the holders are entitled to non-forfeitable dividend rights in the event a dividend is paid on common stock. The holders of nonvested Ordinary Class A shares do not have a contractual obligation to share in losses.
Basic loss per share is computed by dividing loss available to common stockholders by the weighted average number of common shares outstanding, adjusted for the outstanding nonvested shares. Diluted loss per share is computed by giving effect to all potentially dilutive securities outstanding for the period using the treasury stock method or the if-converted method based on the nature of such securities. For periods in which the Company reports net losses, diluted net loss per common share attributable to common stockholders is the same as basic net loss per common share attributable to common stockholders, because potentially dilutive common shares are not assumed to have been issued if their effect is anti-dilutive.
Recent Accounting Pronouncements
Accounting Guidance Issued But Not Yet Adopted
In March 2020, the FASB issued ASU 2020-04 Reference Rate Reform (Topic 848): Facilitation of the Effects of Reference Rate Reform on Financial Reporting. ASU 2020-04 provides temporary optional expedients and exceptions for applying GAAP guidance on contract modifications and hedge accounting to ease the financial reporting burdens of the expected market transition from the LIBOR and other interbank offered rates to alternative reference rates, such as the Secured Overnight Financing Rate. For all entities, ASU 2020-04 can be adopted after its issuance date through December 31, 2022. The Company is currently evaluating the impact of this ASU.
In August 2018, the FASB issued ASU 2018-13, Fair Value Measurement (Topic 820): Disclosure FrameworkChanges to the Disclosure Requirements for Fair Value Measurement (ASU 2018-13). ASU 2018-13 eliminates, adds and modifies certain disclosure requirements for fair value measurements as part of the FASBs disclosure framework project. For all entities, ASU 2018-13 is effective for annual and interim reporting periods beginning after December 15, 2019. Certain amendments must be applied prospectively while others are to be applied on a retrospective basis to all periods presented. The Company expects to implement the provisions of ASU 2018-13 for annual periods beginning July 1, 2020. The Company does not expect this will have a material impact on the consolidated financial statements and related disclosures.
In August 2018, the FASB issued ASU 2018-15, IntangiblesGoodwill and OtherInternal Use Software (Subtopic 350-40): Customers Accounting for Implementation Costs Incurred in a Cloud Computing Arrangement that is a Service Contract, which amend the accounting for implementation, setup and other upfront costs for entities that are a customer in a hosting arrangement that is a service contract. ASU 2018-15 was effective for public business entities for fiscal years beginning after December 15, 2019, and interim periods within those fiscal years. For all other entities, including emerging growth companies, the amendments are effective for fiscal years beginning after December 15, 2020, and interim periods within fiscal years beginning after December 15, 2021. Early adoption is permitted, including adoption in any interim period for which financial statements have not been issued. Reporting entities can choose to adopt the new guidance prospectively to eligible costs incurred on or after the date the guidance is first applied or retrospectively. Application of the amendments can be applied either retrospectively or prospectively to all implementation costs incurred after the date of adoption. The Company expects to implement the provisions of ASU 2018-15 for annual periods beginning July 1, 2021. The Company is currently evaluating the impact ASU 2018-15 will have on its consolidated financial statements.
In June 2016, the FASB issued ASU 2016-13, Financial InstrumentsCredit Losses (Topic 326): Measurement of Credit Losses on Financial Instruments, which replaced the incurred loss impairment methodology in current
F-55
Table of Contents
GAAP with a methodology that reflects expected credit losses and requires consideration of a broader range of reasonable and supportable information to inform credit loss estimates. For trade and other receivables, loans, and other financial instruments, we will be required to use a forward-looking expected loss model rather than the incurred loss model for recognizing credit losses which reflects losses that are probable. Credit losses relating to available-for-sale debt securities will also be recorded through an allowance for credit losses rather than as a reduction in the amortized cost basis of the securities. The effective date of ASU 2016-13 varies depending on the type of entity. Public business entities that meet the definition of a Securities and Exchange Commission (SEC) filer, excluding eligible smaller reporting companies (SRCs) as defined by the SEC, should adopt the amendments in ASU 2016-13 for fiscal years beginning after December 15, 2019. For all other entities, the amendments in ASU 2016-13 are effective for fiscal years beginning after December 15, 2022. Early adoption will continue to be permitted. Application of the amendments is through a cumulative-effect adjustment to retained earnings as of the effective date. The company expects to implement provisions of ASU 2016-13 for annual periods beginning July 1, 2023. We are currently evaluating the impact of this standard on our consolidated financial statements.
In February 2016, the FASB issued ASU 2016-02, Leases (Topic 842), which is a new accounting and reporting guidance for leasing arrangements. The new standard requires organizations that lease assets to recognize assets and liabilities on the balance sheet related to the rights and obligations created by those leases with terms of more than 12 months, regardless of whether they are classified as financing or operating leases. Consistent with current guidance, the recognition, measurement, and presentation of expenses and cash flows arising from a lease primarily will depend on its classification as a finance or operating lease. The guidance also requires new disclosures to help financial statement users better understand the amount, timing, and uncertainty of cash flows arising from leases. For public companies, the new standard was effective for annual and interim periods in fiscal years beginning after December 15, 2018. On June 3, 2020, the FASB issued guidance (ASU 2020-05) that deferred the effective date of the leases standards (ASC 842) for non-public business entities that had not yet issued financial statements adopting the standards. For these entities, including emerging growth companies, ASU 2016-02 is effective for fiscal years beginning after December 15, 2021, and interim periods within fiscal years beginning after December 15, 2022. Early application continues to be allowed. The Company expects to implement the provisions of ASU 2016-02 for annual periods beginning July 1, 2021. The Company is currently evaluating the impact of the new pronouncement on its consolidated financial statements and disclosures, however upon adoption, the Company will recognize a lease liability measured as the present value of remaining lease payments as well as a right of use asset measured as the amount of the lease liability adjusted for certain assets and liabilities such as deferred lease obligations related to operating leases previously recognized in the balance sheet. The Company will elect the modified retrospective transition method and under such method no recast of prior-period financial statements will be presented. We will elect the transition package of practical expedients, which among other things, allows for the carryforward of historical lease classifications, and we will make an accounting policy election to not apply the recognition requirements to leases with a term of less than twelve months. We will also elect to account for lease and non-lease components as a combined single lease component. The Companys existing leases consist primarily of offices, equipment, and vehicles and the related future commitments under lease obligations are summarized in Note 9.
Recently Adopted Accounting Guidance
In May 2014, the FASB issued ASU 2014-19, Revenue from Contract with Customers (ASC 606). On July 1, 2019, the Company adopted ASC 606 using modified retrospective method for all contracts. Revenue for periods prior to July 1, 2019 has not been adjusted and continues to be reported under ASC Topic 605, Revenue Recognition (ASC 605). The impact of adoption of ASC 606 was not material to the Companys consolidated financial statements other than the newly required disclosures and therefore the periods reported under ASC 606 and ASC 605 are considered comparable in all material respects.
In January 2016, the FASB issued ASU 2016-01, Financial Instruments (Subtopic 825-10): Recognition and Measurement of Financial Assets and Financials Liabilities, which amended certain aspects of recognition,
F-56
Table of Contents
measurement, presentation, and disclosure of financial instruments. Most prominent among the amendments is the requirement for changes in the fair value of our equity investments, with certain exceptions, to be recognized through net income rather than OCI. The application of the amendments resulted in a cumulative-effect adjustment to our consolidated balance sheets as of the effective date. The Company adopted this guidance as of July 1, 2019 and it did not have a material impact on our consolidated financial statements.
In October 2016, the FASB issued ASU 2016-16, Income Taxes (Topic 740): Intra-Entity Transfers of Assets Other Than Inventory (ASU 2016-16), which changes the timing of when certain intercompany transactions are recognized within the provision for income taxes. The Company adopted this guidance, on a modified retrospective basis, as of July 1, 2019 and it did not have a material impact on our consolidated financial statements.
In November 2016, the FASB issued ASU 2016-18, Statement of Cash Flows (Topic 230): Restricted Cash, which requires amounts generally described as restricted cash and restricted cash equivalents to be included with cash and cash equivalents when reconciling the beginning of period and end of period total amounts shown on the statement of cash flows. The Company adopted this guidance as of July 1, 2019 using a retrospective transition method to each period presented. For more information, see Note 11.
In January 2017, the FASB issued ASU 2017-04, Simplifying the Test for Goodwill Impairment. The standard simplifies the accounting for goodwill impairment by removing Step 2 of the goodwill impairment test, which requires a hypothetical purchase price allocation. Early adoption is permitted for annual goodwill impairment testing dates on or after January 1, 2017. The guidance should be applied on a prospective basis. The Company adopted this guidance as of July 1, 2018 and it did not have a material impact on our consolidated financial statements.
In March 2017, the FASB issued ASU 2017-07, Improving the Presentation of Net Periodic Pension Cost and Net Periodic Postretirement Benefit Cost. ASU 2017-07 changes how employers that sponsor defined benefit pension and/or other postretirement benefit plans present the cost of the benefits in the income statement. The Company adopted this guidance as of July 1, 2019 retrospectively and it did not have a material impact on our consolidated financial statements.
In August 2016, the FASB issued ASU 2016-15, Statement of Cash Flows (Topic 230): Classification of Certain Cash Receipts and Cash Payments, which clarifies how entities should classify certain cash receipts and cash payments in the statement of cash flows and amends certain disclosure requirements. The Company adopted this guidance as of July 1, 2019 retrospectively and it did not have a material impact on our consolidated financial statements.
In February 2018, the FASB issued ASU 2018-02 (ASC Topic 220), Income StatementReporting Comprehensive Income: Reclassification of Certain Tax Effects from Accumulated Other Comprehensive Income. This ASU was issued following the enactment of the US TCJA of 2017 and permits entities to elect a reclassification from accumulated other comprehensive income to retained earnings for stranded tax effects resulting from the Tax Act. For all entities, ASU 2018-02 is effective for annual and interim reporting periods beginning after December 15, 2018. The Company adopted this guidance as of July 1, 2019 and has elected not to reclassify the income tax effects of the TCJA from OCI to retained earnings.
In December 2019, as part of its initiative to reduce complexity in the accounting standards, the Financial Accounting Standards Board issued ASU 2019-12, Income Taxes (Topic 740): Simplifying the Accounting for Income Taxes (ASU 2019-12), which eliminates certain exceptions related to the approach for intra-period tax allocation, the methodology for calculating income taxes in an interim period and the recognition of deferred tax liabilities for outside basis differences. ASU 2019-12 also clarifies and simplifies other aspects of the accounting for income taxes. The standard is effective for fiscal years, and interim periods within those fiscal years, beginning after December 15, 2021, with early adoption permitted. The Company early adopted ASU 2019-12 on
F-57
Table of Contents
July 1, 2019 prospectively, and this adoption had no material impact on the Companys financial position or results of operations.
2. Acquisitions
The Company continually evaluates potential acquisitions that strategically fit with the Companys existing portfolio and as a result, the Company completed a number of acquisitions during the years ended June 30, 2020, 2019 and 2018.
All acquisitions are accounted for under the acquisition method of accounting, and the related assets acquired and liabilities assumed are recorded at fair value. The Company makes an initial allocation of the purchase price at the date of acquisition based upon its understanding of the fair value of the acquired assets and assumed liabilities. The Company obtains the information used for the purchase price allocation during due diligence and through other sources. In the months after closing, as the Company obtains additional information about the acquired assets and liabilities, including through tangible and intangible asset appraisals, and learns more about the newly acquired business, it is able to refine the estimates of fair value and more accurately allocate the purchase price. The fair values of acquired intangibles are determined based on estimates and assumptions that are deemed reasonable by the Company. Significant assumptions include the discount rates and certain assumptions that form the basis of the forecasted results of the acquired business including revenue, earnings before interest, taxes, depreciation and amortization (EBITDA), and growth rates. These assumptions are forward looking and could be affected by future economic and market conditions. The Company engages third-party valuation specialists who review the Companys critical assumptions and calculations of the fair value of acquired intangible assets in connection with material acquisitions. Only facts and circumstances that existed as of the acquisition date are considered for subsequent adjustment. The Company will make appropriate adjustments to the purchase price allocation prior to completion of the measurement period, as required.
The purchases of these acquired businesses resulted in the recognition of goodwill in the Companys Consolidated Financial Statements, which is calculated as the excess of the consideration transferred over the net assets recognized and represents the future economic benefits arising from the other assets acquired that could not be individually identified and separately recognized. The goodwill is not amortized and is not deductible for income tax purposes. This goodwill recorded includes the following:
| | The expected synergies and other benefits that we believe will result from combining the operations of the acquired business with the operations of Mirion; |
| | Any intangible assets that did not qualify for separate recognition, as well as future, yet unidentified projects and products; |
| | The value of the existing business as an assembled collection of net assets versus if the Company had acquired all of the net assets separately. |
F-58
Table of Contents
The following briefly describes the Companys acquisition activity for the three years ended June 30, 2020.
| Year Ended |
Company |
Description of the Business |
Description of the |
|||
| 2020 | AWST | AWST is a provider of calibration and measurement technologies for radiation medicine applications. | On March 31, 2020, the Company acquired 100% of the equity interest for approximately 24.5 million (or $26.9 million) of purchase consideration. | |||
| 2020 | Selmic | Selmic is an electronic component manufacturer of sensors, modules, and devices serving in automotive, transportation, medical, security, defense, and telecom industries. | On October 31, 2019, the Company acquired 100% of the equity interest for approximately 9.1 million (or $10.2 million) of purchase consideration. | |||
| 2020 | Premium Analyse | Premium Analyse is a provider in the radioactive gas detection market and measurement of tritium. | On July 19, 2019, the Company acquired 100% of the equity interest for approximately 7.9 million ($8.9 million) of purchase consideration. | |||
| 2020 | Capintec | Capintec is a provider of calibration and measurement technologies for nuclear medicine applications. Capintec provides solutions for applications in nuclear medicine, nuclear cardiology, oncology, endocrinology, diagnostic radiology, and radiation therapy. | On July 9, 2019, the Company acquired 100% of the equity interest for approximately $14.5 million of purchase consideration. | |||
| 2019 | NRG Dosimetry Services Group | NRG Dosimetry Services Group is a provider of dosimetry services in the Netherlands. | On October 31, 2018, the Company acquired 100% of the equity interest for approximately 7.8 million (or $9.1 million) of purchase consideration. | |||
F-59
Table of Contents
The following summarizes the fair value of assets acquired and liabilities assumed for the acquisitions during the year ended June 30, 2020 (in millions):
| Company Name | ||||||||||||||||
| Capintec | Premium Analyse |
Selmic | AWST | |||||||||||||
| Date of acquisition |
|
July 9, 2019 |
|
|
July 19, 2019 |
|
|
October 31, 2019 |
|
|
March 31, 2020 |
|
||||
| Segment |
Medical | Industrial | Industrial | Medical | ||||||||||||
| Goodwill |
$ | 6.0 | $ | 4.3 | $ | 2.7 | $ | 4.1 | ||||||||
| Customer relationships (1) |
2.4 | 2.1 | 2.2 | 20.9 | ||||||||||||
| Developed technology (2) |
2.5 | 1.7 | | | ||||||||||||
| Tradename (3) |
0.1 | 0.1 | 0.2 | | ||||||||||||
| Backlog (4) |
0.3 | | 0.1 | | ||||||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Amortizable intangible assets |
$ | 5.3 | $ | 3.9 | $ | 2.5 | $ | 20.9 | ||||||||
| Cash |
0.6 | 1.2 | 0.4 | | ||||||||||||
| Accounts receivable |
2.0 | 0.5 | 1.0 | | ||||||||||||
| Inventory |
2.7 | 1.4 | 4.2 | 0.6 | ||||||||||||
| Property and equipment |
0.8 | 0.2 | 2.1 | 3.8 | ||||||||||||
| Other assets |
0.6 | 0.1 | | 0.3 | ||||||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Total tangible assets acquired |
$ | 6.7 | $ | 3.4 | $ | 7.7 | $ | 4.7 | ||||||||
| Accounts payable |
(1.0 | ) | (0.2 | ) | (1.0 | ) | | |||||||||
| Accrued expenses and other current liabilities |
(1.1 | ) | (0.1 | ) | (0.5 | ) | (0.4 | ) | ||||||||
| Pension obligations |
| (0.1 | ) | | | |||||||||||
| Deferred tax liability |
(0.5 | ) | (1.1 | ) | (0.7 | ) | | |||||||||
| Deferred revenue |
| | | (0.6 | ) | |||||||||||
| Financing obligations |
| | | (1.8 | ) | |||||||||||
| Other liabilities |
(0.3 | ) | | (0.1 | ) | | ||||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Total liabilities assumed |
$ | (2.9 | ) | $ | (1.5 | ) | $ | (2.3 | ) | $ | (2.8 | ) | ||||
|
|
|
|
|
|
|
|
|
|||||||||
| Net tangible assets acquired |
$ | 3.8 | $ | 1.9 | $ | 5.4 | $ | 1.9 | ||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Purchase consideration |
15.1 | 10.1 | 10.6 | 26.9 | ||||||||||||
| Less: cash acquired |
(0.6 | ) | (1.2 | ) | (0.4 | ) | | |||||||||
|
|
|
|
|
|
|
|
|
|||||||||
| Purchase consideration, net of cash acquired |
$ | 14.5 | $ | 8.9 | $ | 10.2 | $ | 26.9 | ||||||||
| Acquiree revenue post acquisition for twelve-months ended June 30, 2020 |
$ | 15.2 | $ | 3.0 | $ | 11.0 | $ | 2.5 | ||||||||
| Acquiree income (loss) from operations post acquisition for twelve-months ended June 30, 2020 |
$ | 0.7 | $ | | $ | (0.1 | ) | $ | (0.4 | ) | ||||||
| (1) | The useful life for customer relationships ranges from 7-17 years. |
| (2) | The useful life for developed technology is 7 years. |
| (3) | The useful life for tradenames is 5 years. |
| (4) | The useful life for backlog is 1 year. |
Transaction costs related to the above acquisitions were not material for the year ended June 30, 2020.
F-60
Table of Contents
The following summarizes the fair value of assets acquired and liabilities assumed for the NRG acquisition during the year ended June 30, 2019 (in millions):
| Date of acquisition | October 31, 2018 |
|||
| Segment Impacted |
Medical | |||
| Goodwill |
$ | 4.2 | ||
| Customer relationships (1) |
4.7 | |||
| Cash |
0.3 | |||
| Accounts receivable |
0.8 | |||
| Unbilled receivables |
0.3 | |||
| Property and equipment |
0.6 | |||
|
|
|
|||
| Total tangible assets acquired |
$ | 2.0 | ||
| Accounts payable |
(0.1 | ) | ||
| Accrued expenses and other current liabilities |
(0.1 | ) | ||
| Other liabilities |
(0.1 | ) | ||
| Deferred tax liability |
(1.2 | ) | ||
|
|
|
|||
| Total liabilities assumed |
$ | (1.5 | ) | |
|
|
|
|||
| Net tangible assets acquired |
$ | 0.5 | ||
|
|
|
|||
| Purchase consideration |
9.4 | |||
| Less: cash acquired |
(0.3 | ) | ||
|
|
|
|||
| Purchase consideration, net of cash acquired |
$ | 9.1 | ||
| (1) | The useful life for customer relationships is 15 years. |
The Company made no acquisitions during the year ended June 30, 2018.
Unaudited Pro Forma Financial Information
The following unaudited pro forma financial information presents the Companys results of operations for the twelve-months ended June 30, 2020 and 2019 to illustrate the estimated effects of the acquisition of Capintec and AWST as if it had occurred on July 1, 2018. The pro forma financial information is presented for comparative purposes only and is not necessarily indicative of the Companys operating results that may have actually occurred had the acquisition of Capintec and AWST been completed on July 1, 2018. The unaudited pro forma financial information does not reflect the expected realization of any anticipated cost savings, operating efficiencies, or other synergies that may have been associated with the acquisition.
| (amounts in millions) | Twelve months ended June 30, |
|||||||
| 2020 | 2019 | |||||||
| Total revenues |
$ | 486.9 | $ | 464.3 | ||||
| Net loss |
(118.1 | ) | (123.1 | ) | ||||
| Net loss before noncontrolling interests attributable to Mirion |
(118.1 | ) | (123.1 | ) | ||||
The unaudited pro forma financial information reflects pro forma adjustments to present the combined pro forma results of the operations as if the acquisition had occurred on July 1, 2018 to give effect to certain events the Company believes to be directly attributable to the acquisitions. These pro forma adjustments primarily include:
| | A net increase in cost of revenues, depreciation and amortization expense that would have been recognized due to acquired inventory, property, plant and equipment and intangible assets; |
| | An increase to interest expense to reflect the additional borrowings of the Company in conjunction with the acquisition; |
F-61
Table of Contents
| | A reduction in expenses for the twelve months ended June 30, 2020 and a corresponding increase in the twelve months ended June 30, 2019 for acquisition-related transaction costs directly attributable to the acquisition; |
| | A reduction in revenues due to the elimination of deferred revenues assigned no value at the acquisition date; |
| | An increase in income tax expense using the U.S. statutory rate of 25% to reflect a change in tax status had the Capintec and AWST results of operations been included in the Companys consolidated tax return; |
| | The related income tax effects of the adjustments noted above. |
3. Contracts in Progress
Costs and billings on uncompleted construction-type contracts consist of the following (in millions):
| June 30, 2020 | June 30, 2019 | |||||||
| Costs incurred on contracts (from inception to completion) |
$ | 152.4 | $ | 125.7 | ||||
| Estimated earnings |
108.9 | 99.7 | ||||||
|
|
|
|
|
|||||
| Contracts in progress |
261.3 | 225.4 | ||||||
| Less billings to date |
(209.8 | ) | (178.6 | ) | ||||
|
|
|
|
|
|||||
| $ | 51.5 | $ | 46.8 | |||||
|
|
|
|
|
|||||
| Included in the accompanying balance sheets under the following captions: |
||||||||
| Costs and estimated earnings in excess of billings on uncompleted contracts |
$ | 59.5 | $ | 56.9 | ||||
| Billings in excess of costs and estimated earnings on uncompleted contracts (1) |
(8.0 | ) | (10.1 | ) | ||||
|
|
|
|
|
|||||
| $ | 51.5 | $ | 46.8 | |||||
|
|
|
|
|
|||||
| (1) | Included in deferred contract revenue within the consolidated balance sheets. |
Substantially all of the contract liabilities balance as of June 30, 2019 was recognized as revenue during the year ended June 30, 2020.
4. Inventories
The components of inventories consist of the following (in millions):
| June 30, 2020 | June 30, 2019 | |||||||
| Raw materials |
$ | 40.6 | $ | 37.2 | ||||
| Work in progress |
16.1 | 17.9 | ||||||
| Finished goods |
33.5 | 34.2 | ||||||
|
|
|
|
|
|||||
| $ | 90.2 | $ | 89.3 | |||||
|
|
|
|
|
|||||
F-62
Table of Contents
5. Property, Plant, and Equipment, Net
Property and equipment, net consist of the following (in millions):
| Depreciable Lives |
June 30, 2020 |
June 30, 2019 |
||||||||||
| Land, buildings, and leasehold improvements |
3-39 years | $ | 43.9 | $ | 40.3 | |||||||
| Machinery and equipment |
5-15 years | 38.9 | 30.6 | |||||||||
| Badges |
3-5 years | 29.4 | 23.8 | |||||||||
| Furniture, fixtures, computer equipment and other |
3-10 years | 27.6 | 21.9 | |||||||||
| Construction in progress |
| 7.3 | 6.1 | |||||||||
|
|
|
|
|
|||||||||
| 147.1 | 122.7 | |||||||||||
| Less: accumulated depreciation and amortization |
(71.9 | ) | (56.9 | ) | ||||||||
|
|
|
|
|
|||||||||
| $ | 75.2 | $ | 65.8 | |||||||||
|
|
|
|
|
|||||||||
Total depreciation expense included in cost of revenue was $12.7 million, $11.1 million, and $11.1 million for the fiscal years ended June 30, 2020, 2019, and 2018, respectively. Total depreciation expense included in operating expenses was $5.2 million, $5.4 million, and $6.1 million for the fiscal years ended June 30, 2020, 2019, and 2018, respectively. Construction in progress includes capitalized internal use software costs totaling $1.2 million and $2.9 million as of June 30, 2020 and 2019, respectively.
6. Accrued Expenses and Other Current Liabilities, and Deferred Income Taxes and Other Long-Term Liabilities
Accrued expenses and other current liabilities consist of the following (in millions):
| June 30, 2020 | June 30, 2019 | |||||||
| Compensation and related benefit costs |
$ | 30.3 | $ | 28.4 | ||||
| Customer deposits |
3.1 | 5.3 | ||||||
| Accrued commissions |
3.7 | 4.4 | ||||||
| Accrued warranty costs |
5.5 | 4.2 | ||||||
| Non-income taxes payable |
4.9 | 2.8 | ||||||
| Pension and other post-retirement obligations |
0.3 | 0.3 | ||||||
| Income taxes payable |
9.2 | 0.2 | ||||||
| Other accrued expenses |
7.1 | 6.6 | ||||||
|
|
|
|
|
|||||
| $ | 64.1 | $ | 52.2 | |||||
|
|
|
|
|
|||||
Deferred income taxes and other long-term liabilities consist of the following (in millions):
| June 30, 2020 | June 30, 2019 | |||||||
| Deferred income taxes |
$ | 33.1 | $ | 52.4 | ||||
| Pension and other post-retirement obligations, non-current |
12.4 | 11.9 | ||||||
| Other long-term liabilities |
18.0 | 21.1 | ||||||
|
|
|
|
|
|||||
| $ | 63.5 | $ | 85.4 | |||||
|
|
|
|
|
|||||
7. Goodwill and Intangible Assets
Goodwill
Goodwill is calculated as the excess of consideration transferred over the net assets recognized for acquired businesses and represents future economic benefits arising from the other assets acquired that could not be
F-63
Table of Contents
individually identified and separately recognized. The Company assesses goodwill for impairment at the reporting unit level annually during the fiscal fourth quarter and upon the occurrence of a triggering event or change in circumstance that would more likely than not reduce the fair value of a reporting unit below its carrying amount.
Goodwill is assigned to reporting units at the date the goodwill is initially recorded and is reallocated as necessary based on the composition of reporting units over time. During the year ended June 30, 2019, the Company reassessed its reporting units as a result of a change in managerial and divisional structure. The Spectroscopy, Characterization, and Health Physics reporting units within the Industrial reportable segment were combined into two Detection and Measurement (DMD) reporting units based on geography: North America and the Europe / Asia markets. Goodwill was then allocated to DMD North America and DMD Europe and Asia based on each reporting units relative fair value at the date of the reorganization. The Company performed a Step 1 goodwill impairment test at the reporting unit level both immediately before and after the change in reporting units. As a result of the impairment tests, the Company determined the fair value of the reporting units exceeded their carrying values both before and after the change in reporting units.
No goodwill impairment was recognized for the years ended June 30, 2020, 2019 and 2018.
The following table shows changes in the carrying amount of goodwill by reportable segment for the years ended June 30, 2020 and 2019 (in millions):
| Medical | Industrial | Consolidated | ||||||||||
| BalanceJune 30, 2018 |
$ | 92.6 | $ | 424.5 | $ | 517.1 | ||||||
| Acquisition of NRG Dosimetry Services Group |
4.2 | | 4.2 | |||||||||
| Other |
| (0.6 | ) | (0.6 | ) | |||||||
| Translation adjustment |
(0.1 | ) | (9.0 | ) | (9.1 | ) | ||||||
|
|
|
|
|
|
|
|||||||
| BalanceJune 30, 2019 |
$ | 96.7 | $ | 414.9 | $ | 511.6 | ||||||
| Acquisition of Capintec |
6.0 | | 6.0 | |||||||||
| Acquisition of Premium Analyse |
| 4.3 | 4.3 | |||||||||
| Acquisition of Selmic |
| 2.7 | 2.7 | |||||||||
| Acquisition of AWST |
4.1 | | 4.1 | |||||||||
| Translation adjustment |
| (6.1 | ) | (6.1 | ) | |||||||
|
|
|
|
|
|
|
|||||||
| BalanceJune 30, 2020 |
$ | 106.8 | $ | 415.8 | $ | 522.6 | ||||||
|
|
|
|
|
|
|
|||||||
We do not expect goodwill to be deductible for income tax purposes.
Intangible Assets
Intangible assets consist of our developed technology, customer relationships, backlog, trade names, and non-compete agreements at the time of acquisition through business combinations. The customer relationships definite lived intangible assets are amortized using the double declining balance method with estimated useful lives ranging from 6 to 17 years, while all other definite lived intangible assets are amortized on a straight-line basis over their estimated useful lives, ranging from 1 to 7 years.
F-64
Table of Contents
Many of our intangible assets are not deductible for income tax purposes. A summary of intangible assets and related accumulated amortization (in millions) is below:
| June 30, 2020 | ||||||||||||||||
| Original Average Life in Years |
Gross Carrying Amount |
Accumulated Amortization |
Net Book Value |
|||||||||||||
| Customer relationships |
6-17 | $ | 358.5 | $ | (170.5 | ) | $ | 188.0 | ||||||||
| Developed technology |
3-16 | 130.0 | (81.0 | ) | 49.0 | |||||||||||
| Trade names |
5-9 | 32.9 | (22.4 | ) | 10.5 | |||||||||||
| Backlog and other |
1-9 | 22.8 | (22.0 | ) | 0.8 | |||||||||||
|
|
|
|
|
|
|
|||||||||||
| Total |
$ | 544.2 | $ | (295.9 | ) | $ | 248.3 | |||||||||
|
|
|
|
|
|
|
|||||||||||
| June 30, 2019 | ||||||||||||||||
| Original Average Life in Years |
Gross Carrying Amount |
Accumulated Amortization |
Net Book Value |
|||||||||||||
| Customer relationships |
6-15 | $ | 332.4 | $ | (144.9 | ) | $ | 187.5 | ||||||||
| Developed technology |
3-16 | 126.8 | (63.4 | ) | 63.4 | |||||||||||
| Trade names |
5-9 | 32.6 | (17.4 | ) | 15.2 | |||||||||||
| Backlog and other |
1-9 | 25.1 | (23.6 | ) | 1.5 | |||||||||||
|
|
|
|
|
|
|
|||||||||||
| Total |
$ | 516.9 | $ | (249.3 | ) | $ | 267.6 | |||||||||
|
|
|
|
|
|
|
|||||||||||
Aggregate amortization expense for intangible assets included in cost of revenue was $17.9 million, $18.4 million, and $20.9 million for the fiscal years ended June 30, 2020, 2019 and 2018 respectively. Aggregate amortization expense for intangible assets included in operating expenses was $32.7 million, $34.5 million, and $39.0 million for the fiscal years ended June 30, 2020, 2019, and 2018, respectively. Future annual amortization expense at current exchange rates is as follows (in millions):
| Fiscal year ending June 30: |
||||
| 2021 |
$ | 47.6 | ||
| 2022 |
39.3 | |||
| 2023 |
26.3 | |||
| 2024 |
23.2 | |||
| 2025 |
21.5 | |||
| 2026 and thereafter |
90.4 | |||
|
|
|
|||
| Total |
$ | 248.3 | ||
|
|
|
8. Borrowings
Third-party notes payable consist of the following (in millions):
| June 30, 2020 |
June 30, 2019 |
|||||||
| 2019 Credit Facilityfirst lien term loan |
$ | 682.1 | $ | 590.6 | ||||
| NRG Loan |
5.8 | 7.4 | ||||||
| Canadian Financial Institution |
1.2 | 1.3 | ||||||
| JLG Note Payable |
0.2 | 0.2 | ||||||
| French Financial Institutions |
| 5.1 | ||||||
| Overdraft facilities |
| 0.1 | ||||||
| Draw on revolving line of credit |
35.0 | | ||||||
|
|
|
|
|
|||||
| Total third-party borrowings |
724.3 | 604.7 | ||||||
| Less: notes payable to third-parties, current |
(41.1 | ) | (10.5 | ) | ||||
| Less: deferred financing costs |
(13.4 | ) | (14.6 | ) | ||||
|
|
|
|
|
|||||
| Notes payable to third-parties, non-current |
$ | 669.8 | $ | 579.6 | ||||
|
|
|
|
|
|||||
F-65
Table of Contents
As of June 30, 2020 and June 30, 2019, the fair market value of the Companys 2019 Credit Facilityfirst lien term loan was $662.5 million and $ 592.1 million, respectively. The fair market value for the credit facility were estimated using primarily level 2 inputs, including borrowing rates available to the Company at the respective period ends. The fair market value for the Companys remaining third-party debt approximates the respective carrying amounts as of June 30, 2020 and 2019.
2019 Credit Facility
In March 2019, Mirion Technologies (HoldingRep), Ltd., a wholly owned subsidiary of the Company, and subsidiaries entered into a credit agreement with an intermediary and a syndicate of institutional lenders (2019 Credit Facility). The 2019 Credit Facility provided for financing of a $450.0 million senior secured term loan facility and a 125 million term loan facility, as well as a $90.0 million revolving line of credit. In fiscal 2020, the 2019 Credit Facility was amended to provide an additional $34.0 million and $66.0 million in gross proceeds from the USD term loan in July 2019 and December 2019, respectively.
USD term loanThe term loan has a seven-year term (expiring March 2026), bears interest at the greater of Adjusted London Interbank Offered Rate (LIBOR) or 0%, plus 4.00% and has quarterly principal repayments of 0.25% of the original principal balance. The interest rate was 5.07% and 6.32% at June 30, 2020 and 2019, respectively. The Company repaid $5.5 million and $1.1 million for the fiscal year ended June 30, 2020 and 2019, respectively, yielding an outstanding balance of approximately $543.5 million and $448.9 million as of June 30, 2020 and 2019, respectively.
Euro term loanThe Euro portion of the term loan has a seven-year term (expiring March 2026), bears interest at the greater of European union interbank market (Euribor) or 0%, plus 4.25% and has quarterly principal repayments of 0.25% of the original principal balance. As of June 30, 2020, and 2019, the interest rate was 4.25%. The Company repaid $1.4 million and $0.4 million for the fiscal year ended June 30, 2020 and 2019, respectively, yielding an outstanding balance of approximately 123.4 million (approximately $138.6 million) and 124.7 million (approximately $141.7 million) as of June 30, 2020 and 2019, respectively.
Revolving Line of CreditThe revolving line of credit arrangement has a five-year term and bears interest at the greater of LIBOR or 0%, plus 4.00%. The agreement requires the payment of a commitment fee of 0.50% per annum for unused commitments. The revolving line of credit matures on March 2024, at which time all outstanding revolving facility loans and accrued and unpaid interest are due. Any outstanding letters of credit reduce the availability of the revolving line of credit. The outstanding balance under the arrangement was $35.0 million and $0 as of June 30, 2020 and 2019, respectively. Additionally, the Company has standby letters of credit issued under its Credit Facility that reduce the availability under the revolver of $9.0 million and $9.8 million at June 30, 2020 and 2019, respectively, the amount available on the revolver was approximately $46.0 million and $80.2 million, for the same periods, respectively.
Deferred Financing Costs
In connection with the issuance of the 2019 Credit Facility, we incurred debt issuance costs of $16.3 million on date of issuance, and an additional $1.2 million of costs for incremental proceeds in fiscal year 2020. In conjunction with the issuance of 2019 Credit Facility, we concluded there was an extinguishment of a previous debt. We wrote off the remaining unamortized original issue discounts (OID) and debt issuance costs of $12.8 million in March 2019. In accordance with accounting for debt issuance costs, we recognize and present deferred finance costs associated with non-revolving debt and financing obligations as a reduction from the face amount of related indebtedness in our consolidated balance sheet.
In connection with the issuance of the 2019 Credit Facility revolving line of credit, we incurred debt issuance costs of $0.9 million. We wrote off the remaining unamortized debt issuance costs of $0.2 million of a previous revolving credit agreement in March 2019. We recognize and present debt issuance costs associated with revolving debt arrangements as an asset and include the deferred finance costs within other assets on our consolidated balance sheet. We amortize all debt issuance costs over the life of the related indebtedness.
F-66
Table of Contents
During fiscal 2020 and 2019, we incurred approximately $2.6 million and $2.8 million, respectively, of amortization expense of the deferred finance costs, in addition to the write off of $12.8 million included in Loss on debt extinguishment in fiscal year end 2019.
Both Credit Facilities are secured by a first priority lien on substantially all of Mirion HoldingRep and subsidiaries assets in the United States, certain assets of guarantor subsidiaries in Germany, United Kingdom, Canada, France, Belgium and Luxembourg and two-thirds of assets in non-guarantors and other countries. Loan fees recorded as debt discounts are amortized using the effective interest method. The Credit Facility contains customary restrictive covenants, as well as financial covenants that require Mirion HoldingRep and subsidiaries to maintain a certain total level of debt to income ratio and interest coverage ratio, each as defined in the Credit Facility, as well as non-financial affirmative and negative covenants. The negative covenants, subject to certain exceptions, generally limit the ability of Mirion HoldingRep and subsidiaries to incur additional debt, create liens, make fundamental changes, make certain investments, pay dividends, purchase or retire equity interests, or prepay or retire certain debt. Mirion HoldingRep and subsidiaries were in compliance with all debt covenants at both June 30, 2020 and 2019.
NRG LoanIn conjunction with the acquisition of NRG (see Note 2), the Company entered into a loan agreement for 7.2 million ($7.4 million) at the date of the acquisition. This agreement expires in December 2023. The loan bears interest which is Euribor of 3 months, plus 2.0%, and mandatory costs if any.
French Financial InstitutionsIn September 2018, the Company entered into two term loans aggregating to a 4.5 million ($5.1 million) with French financial institutions that mature in July and October 2019. The notes bear interest at greater of Euribor or 0%, plus 1.0% and 1.5%, respectively. As June 30, 2020, both term loans have been paid off in full.
Canadian Financial InstitutionIn May 2019, the Company entered into a credit agreement for C$1.7M ($1.3 million) with a Canadian financial institution that matures in April 2039. The note bears annual interest at 4.15%. The credit agreement is secured by the facility acquired using the funds obtained.
JLG Note PayableIn May 2019, the Company entered into a note payable for $0.2 million with an individual that has left the organization, which is due upon a change in control of Mirion Technologies (Global), Ltd, a wholly owned subsidiary of the Company. The note bears annual interest at 6.00%.
Overdraft Facilities
The Company has overdraft facilities with certain German and French financial institutions. As of June 30, 2020 and 2019, the amounts outstanding under these arrangements were $0.0 million and $0.1 million, respectively.
Performance Bonds and Other Credit Facilities
The Company has entered into various line of credit arrangements with local banks in France and Germany. These arrangements provide for the issuance of documentary and standby letters of credit of up to 47.3 million ($53.1 million) and 25.1 million ($28.5 million) as of June 30, 2020 and 2019, respectively, subject to certain local restrictions. As of June 30, 2020, and 2019, 21.2 million ($23.7 million) and 6.9 million ($7.9 million), respectively, of the lines had been utilized to guarantee documentary and standby letters of credit, with interest rates ranging from 0.5% to 2.00%. In addition, the Company posts performance bonds with irrevocable letters of credit to support certain contractual obligations to customers for equipment delivery. These letters of credit are supported by restricted cash accounts, which totaled $1.7 million as of both June 30, 2020 and 2019.
F-67
Table of Contents
At June 30, 2020, contractual principal payments of total third-party borrowings are as follows (in millions):
| Fiscal year ending June 30: |
||||
| 2021 |
$ | 43.5 | ||
| 2022 |
8.4 | |||
| 2023 |
8.3 | |||
| 2024 |
7.4 | |||
| 2025 |
6.6 | |||
| Thereafter |
650.1 | |||
|
|
|
|||
| Gross Payments |
724.3 | |||
| Unamortized debt issuance costs |
(13.4 | ) | ||
|
|
|
|||
| Total third-party borrowings, net of debt issuance costs |
$ | 710.9 | ||
|
|
|
Notes payable to related parties consists of the following (in millions):
| June 30, 2020 |
June 30, 2019 |
|||||||
| Shareholder Notes |
$ | 983.7 | $ | 882.0 | ||||
| Management Notes |
3.4 | 3.2 | ||||||
|
|
|
|
|
|||||
| Notes payable to related parties |
$ | 987.1 | $ | 885.2 | ||||
|
|
|
|
|
|||||
The estimated fair value of the Companys related party debt was approximately $987.1 million and $885.2 million as of June 30, 2020 and 2019, respectively. The fair value of this instrument approximates book value due to the reasonable possibility of redemption prior to the term date.
Shareholder and Management NotesMirion Technologies (HoldingSub1), Ltd., is authorized to issue $900 million (plus accrued paid in-kind interest) of notes to shareholders (Shareholder Notes) and up to $5.0 million (plus paid in-kind cash and interest) of notes to certain members of management (Management Notes). The notes rank pari passu between each other and other unsecured obligations of the Company. The notes can be prepaid without penalty at the Companys option and are subordinate in right of payment to any indebtedness of the Company to banks or to other financial institutions (either currently existing or to occur in the future). Certain of the Shareholder and Management Notes have been admitted to trading and are on the official listing of The International Stock Exchange (TISE).
During fiscal 2020 and 2019, an additional $99.6 million and $274.9 million in Shareholder Notes were admitted to trading and are on the official listing of TISE, respectively. At June 30, 2020 and 2019, there were $976.9 million and $877.3 million in Shareholder Notes issued and outstanding, respectively, as listed on TISE. Of the amount available for trading, $613.9 million relates to principal balance and $363.0 million relates to accrued interest as of June 30, 2020. There was no trading activity related to Shareholder and Management Notes during fiscal 2020, and at June 30, 2020 and 2019 there were $3.4 million and $3.0 million in Management Notes issued and outstanding, respectively, as listed on TISE.
The notes bear simple annual interest at 11.5%. For the Shareholder Notes, the interest is added to the principal outstanding on December 31 of each year until 2025 and is referred to as Shareholder Funding Bonds on TISE. For the Management Notes, half of the interest is added to the principal outstanding on December 31 of each year until 2025 and is referred to as Management Funding Bonds on TISE, while the remaining half is payable in cash annually. The listing on the Exchange for Shareholder and Management Funding Bonds is an optional election and certain shareholders have elected to opt-out of listing their Shareholder Funding Bonds. All other shareholders and management have elected to list their funding bonds on the Exchange. The notes are due when the Company completes a public offering, a winding-up, a sale, or on March 30, 2026, whichever occurs first. The redemption price is equal to the outstanding principal plus all accrued and unpaid interest then outstanding.
F-68
Table of Contents
At June 30, 2020 and 2019, interest of $56.3 million and $50.1 million was accrued on the Shareholder Notes principal outstanding, respectively, and $0.1 million and $0.2 million was accrued on the Management Notes principal outstanding, respectively. As of December 31, 2019 and 2018, accrued interest of $101.3 million and $89.5 million, respectively, was added to the principal of the Shareholder Notes; and $0.2 million and $0.4 million, respectively, was added to the principal of the Management Notes.
9. Commitments and Contingencies
Leases
In the normal course of business, the Company enters into contractual arrangements with third-parties for non-cancellable operating lease agreements for its offices, equipment and vehicles. Future minimum lease payments for these operating leases as of June 30, 2020, are as follows (in millions):
| Fiscal year ending June 30: |
||||
| 2021 |
$ | 7.2 | ||
| 2022 |
6.7 | |||
| 2023 |
5.1 | |||
| 2024 |
3.9 | |||
| 2025 |
3.6 | |||
| 2026 and thereafter |
9.0 | |||
|
|
|
|||
| Total |
$ | 35.5 | ||
|
|
|
Total rent expense for the fiscal years ended June 30, 2020, 2019 and 2018 was $5.8 million, $4.6 million and $5.7 million, respectively.
Unconditional Purchase Obligations
The Company has entered into certain long-term unconditional purchase obligations with suppliers. These agreements are non-cancellable and specify terms, including fixed or minimum quantities to be purchased, fixed or variable price provisions, and the approximate timing of payment. As of June 30, 2020, unconditional purchase obligations were $11.0 million and $0.9 million for the fiscal years ending June 30, 2021 and 2022, respectively.
Litigation
The Company is subject to various legal proceedings, claims, litigation, investigations and contingencies arising out of the ordinary course of business. While the ultimate results of such suits or other proceedings against us cannot be predicted with certainty, we believe the resolution of these matters will not have a material effect on our results of operations, financial condition, or cash flows. If we believe the likelihood of an adverse legal outcome is probable and the amount is reasonably estimable, we accrue a liability in accordance with accounting guidance for contingencies. We consult with legal counsel on matters related to litigation and seek input both within and outside the Company.
F-69
Table of Contents
10. Income Taxes
The domestic and foreign components of (loss) before provision for income taxes and the provision for income taxes were as follows (in millions):
| Years Ended June 30, |
||||||||||||
| 2020 | 2019 | 2018 | ||||||||||
| United Kingdom |
$ | (118.2 | ) | $ | (96.3 | ) | $ | (84.5 | ) | |||
| United States |
(24.5 | ) | (42.0 | ) | (53.6 | ) | ||||||
| Other foreign |
18.1 | 12.1 | (2.4 | ) | ||||||||
|
|
|
|
|
|
|
|||||||
| Net loss before benefit from income taxes |
$ | (124.6 | ) | $ | (126.2 | ) | $ | (140.5 | ) | |||
|
|
|
|
|
|
|
|||||||
| Income tax provision (benefit): |
||||||||||||
| Current: |
||||||||||||
| United Kingdom |
0.6 | 1.1 | (0.3 | ) | ||||||||
| United States |
(6.2 | ) | 1.9 | (0.8 | ) | |||||||
| Other foreign |
16.1 | 9.6 | 13.7 | |||||||||
|
|
|
|
|
|
|
|||||||
| Total current provision |
$ | 10.5 | $ | 12.6 | $ | 12.6 | ||||||
|
|
|
|
|
|
|
|||||||
| Deferred: |
||||||||||||
| United Kingdom |
(0.4 | ) | (0.3 | ) | (0.4 | ) | ||||||
| United States |
1.3 | (7.2 | ) | (38.5 | ) | |||||||
| Other foreign |
(16.9 | ) | (9.3 | ) | (10.5 | ) | ||||||
|
|
|
|
|
|
|
|||||||
| Total deferred benefit |
(16.0 | ) | (16.8 | ) | (49.4 | ) | ||||||
|
|
|
|
|
|
|
|||||||
| Total benefit from income taxes |
$ | (5.5 | ) | $ | (4.2 | ) | $ | (36.8 | ) | |||
|
|
|
|
|
|
|
|||||||
The provision (benefit) for income taxes differs from the amount computed by applying the U.K. statutory income tax rate to loss before provision for income taxes as follows:
|
Years Ended June 30, |
||||||||||||
| 2020 | 2019 | 2018 | ||||||||||
| Income tax at U.K. statutory rate |
19 | % | 19 | % | 19 | % | ||||||
| Subpart F & GILTI |
(2 | %) | (4 | %) | (2 | %) | ||||||
| Foreign taxes, including U.S. |
1 | % | 3 | % | 2 | % | ||||||
| U.S. federal rate change due to tax reform |
| | 3 | % | ||||||||
| Undistributed Foreign Earnings and US Transition Tax |
| | 5 | % | ||||||||
| Change in valuation allowance |
(8 | %) | 1 | % | 11 | % | ||||||
| Unrecognized tax benefits |
11 | % | | | ||||||||
| Nondeductible interest expense |
(17 | %) | (14 | %) | (11 | )% | ||||||
| Other |
| (2 | %) | (1 | %) | |||||||
|
|
|
|
|
|
|
|||||||
| Total effective income tax rate |
4 | % | 3 | % | 26 | % | ||||||
|
|
|
|
|
|
|
|||||||
On March 27, 2020, President Trump signed into U.S. federal law the Coronavirus Aid, Relief and Economic Security Act (CARES Act), which provides emergency assistance and health care for individuals, families, and businesses affected by the COVID-19 pandemic. Namely, the Adjusted Taxable Income (ATI) limit calculation for Section 163(j) interest limitation purposes was increased from 30% to 50%. This temporary change in tax law is in effect for tax years beginning during 2019 and 2020, and reduced U.S. taxable income by $2.2 million for June 30, 2020 with no impact to the effective tax rate. The other relief provisions will not have a material impact on the Companys consolidated financial statements.
F-70
Table of Contents
On June 29, 2020, the California governor signed Assembly Bill 85 (AB 85) into law. AB 85 limits the use of business incentive tax credits and suspends the use of California net operating losses for 2020, 2021 and 2022 for companies with taxable income of $1 million or more. As this law change will not impact Mirion until the year end June 30, 2021, the Company is still assessing the impact of AB 85.
On December 22, 2017, the U.S. government enacted comprehensive tax legislation, commonly referred to as the Tax Cuts and Jobs Act (the Tax Legislation). The Tax Legislation significantly revised U.S. corporate income tax law by, among other things, lowering U.S. corporate income tax rates from 35% to 21%, implementing a territorial tax system, and imposing a one-time transition tax on deemed repatriated earnings of non-U.S. subsidiaries.
The change in tax rate resulted in the recognition of a $9.7 million deferred tax benefit for the revaluation of U.S. net deferred tax liability and the change in territorial tax system resulted in a $23.2 million deferred tax benefit for the year ended June 30, 2018, as a result of reversing a liability for investment in foreign earnings as a result of the one-time transition tax net of foreign tax credits.
The Tax Legislations new international rules, including Global Intangible Low-Taxed Income (GILTI), Foreign Derived Intangible Income, and Base Erosion Anti-Abuse Tax are effective beginning in fiscal year 2019. We have included tax expense of $1.2 million for the years ended June 30, 2020 and 2019 for the effect of the Tax Legislation.
The FASB Staff Q&A, Topic 740, No. 5, Accounting for Global Intangible Low-Taxed Income, states that an entity can make an accounting policy election to either recognize deferred taxes for the temporary basis difference expected to reverse as GILTI in future years or provide for the tax expense related to GILTI in the year the tax is incurred. We have elected to recognize GILTI tax expense as a current period cost.
Mirion has recorded a deferred tax liability of $0.7 million, $0.2 million, and $23 million related to undistributed earnings of certain foreign subsidiaries as of fiscal year 2020, fiscal year 2019 and fiscal year 2018 respectively. The impact in FY19 was due to the Tax Legislation as discussed above. Taxes have not been provided on approximately $220M of certain earnings and profits of U.S. controlled foreign corporations which are permanently reinvested and approximately $47M of undistributed foreign US GAAP retained earnings of other non-US foreign subsidiaries held outside the US group. The determination of any incremental tax liability associated with these earnings is not practicable due to the complexity of local country withholding rules, interactions with tax treaties, foreign exchange consideration and the diversity of tax treatment on actual distributions.
F-71
Table of Contents
The components of the Companys net deferred tax assets and liabilities consist of the following (in millions):
|
Years Ended June 30, |
||||||||
| 2020 | 2019 | |||||||
| Deferred tax assets: |
||||||||
| Net operating loss carryforwards |
$ | 29.2 | $ | 21.5 | ||||
| Federal and state credit carryforwards |
16.4 | 13.9 | ||||||
| Depreciation and amortization |
2.6 | 3.5 | ||||||
| Section 163(j) disallowed interest |
4.9 | 6.6 | ||||||
| Other reserves and accrued expenses |
9.0 | 10.1 | ||||||
| Other assets |
4.7 | 4.8 | ||||||
|
|
|
|
|
|||||
| Total deferred tax assets |
66.8 | 60.4 | ||||||
| Less: valuation allowance |
(29.0 | ) | (18.7 | ) | ||||
|
|
|
|
|
|||||
| Net deferred tax assets |
37.8 | 41.7 | ||||||
|
|
|
|
|
|||||
| Deferred tax liabilities: |
||||||||
| Purchased technologies and other intangibles |
(58.2 | ) | (73.9 | ) | ||||
| Deferred revenue |
(0.8 | ) | (3.2 | ) | ||||
| Depreciation |
(3.0 | ) | (4.1 | ) | ||||
| Other liabilities |
(4.1 | ) | (5.0 | ) | ||||
|
|
|
|
|
|||||
| Total deferred tax liabilities |
(66.1 | ) | (86.2 | ) | ||||
|
|
|
|
|
|||||
| Net deferred tax liabilities |
$ | (28.3 | ) | $ | (44.5 | ) | ||
|
|
|
|
|
|||||
Management regularly evaluates the recoverability of deferred tax assets and recognizes the tax benefit only if reassessment demonstrates that they are more likely than not realizable. At such time, if it is determined that it is more likely than not that the deferred tax assets are realizable, the valuation allowance will be adjusted. In assessing the need for a valuation allowance, Management considers all available evidence, both positive and negative, including: reversals of existing temporary differences; historical levels of income; expectations and risks associated with estimates of future taxable income; and any ongoing tax planning strategies. At June 30, 2020, the Company evaluated the realizability of the deferred tax assets and concluded that a valuation allowance of $29.0 million mostly relating to U.S. federal and state net operating losses and foreign tax credit carryovers should be recorded. At June 30, 2019 the valuation allowance was $18.7 million mostly relating to U.S. foreign tax credits that are more likely than not to expire before utilization. The Company increased the valuation allowance by $10.3 million and $0.6 million for the periods ended June 30, 2020 and June 30, 2019, respectively.
| Years Ended June 30, |
||||||||
| 2020 | 2019 | |||||||
| Valuation allowance balancebeginning of fiscal year |
$ | 18.7 | $ | 18.1 | ||||
| Increases resulting from business combinations |
0.3 | | ||||||
| Other Increases |
10.0 | 1.3 | ||||||
| Other Decreases |
| (0.7 | ) | |||||
|
|
|
|
|
|||||
| Valuation allowance balanceend of fiscal year |
$ | 29.0 | $ | 18.7 | ||||
|
|
|
|
|
|||||
As of June 30, 2020, the Company had U.S. federal, U.S. state and non-U.S. net operating loss carryforwards of $69.0 million, $64.5 million, and $57.0 million, respectively. The U.S. federal net operating losses will expire in fiscal year 2037 through fiscal year 2038. The U.S. state net operating losses will continue to expire in fiscal year 2021 through fiscal year 2039. Materially, the foreign net operating losses have an indefinite carryover period. As of June 30, 2020, the Company had U.S. and foreign tax credit carryforwards of $14.1 million and
F-72
Table of Contents
$2.2 million, respectively, available to offset future U.S. federal and foreign income taxes payable. U.S. and foreign tax credit carryforwards will expire in fiscal year 2021 through fiscal year 2028.
The Companys ability to utilize U.S. net operating loss and tax credit carryforwards in the future may be subject to substantial restriction in the event of past or future ownership changes as defined in Section 382 of the Internal Revenue Code and similar state tax laws. In the event the Company should experience an ownership change, as defined, utilization of its net operating loss carryforwards and tax credits could be limited.
As of June 30, 2020, the Company had $0.8 million of unrecognized tax benefits related to uncertain tax positions, $0.7 million of which would affect its effective tax rate if recognized. Management does not believe the unrecognized tax benefits as of June 30, 2020, will materially change in the next 12 months. As of June 30, 2019, the Company had $13.9 million of unrecognized tax benefits related to uncertain tax positions, $12.1 million of which would affect its effective tax rate if recognized. A reconciliation of the beginning and ending amount of unrecognized tax benefits is as follows (in millions):
| Years Ended June, 30 |
||||||||
| 2020 | 2019 | |||||||
| Balance, beginning of year |
$ | 13.9 | $ | 14.0 | ||||
| Lapse of applicable statute of limitations |
(13.1 | ) | (0.1 | ) | ||||
|
|
|
|
|
|||||
| Balance, end of year |
$ | 0.8 | $ | 13.9 | ||||
|
|
|
|
|
|||||
The Company has recorded $0.8 million and $6.0 million of unrecognized tax benefits as noncurrent income taxes payable as of June 30, 2020 and June 30, 2019, respectively, and $7.8 million of unrecognized tax benefits as a reduction of net deferred tax assets included in other liabilities in the accompanying consolidated balance sheets at June 30, 2019.
The Company recognizes interest and penalties related to uncertain tax positions as a component of income tax expense. Accrued interest and penalties as of June 30, 2020 and June 30, 2019, were approximately $0.3 million and $2.2 million, respectively. The ultimate amount and timing of any future cash settlements cannot be predicted with reasonable certainty.
The Company conducts business globally and, as a result, one or more of its subsidiaries files income tax returns in the U.K. jurisdiction, U.S. federal and various state and other foreign jurisdictions. In the normal course of business, the Company is subject to examination by taxing authorities throughout the world, including such major jurisdictions as the United Kingdom, France, Germany, Canada, and the United States. With the exception of a few insignificant jurisdictions, the Company is no longer subject to U.S. federal or non-U.S. income tax examinations for years prior to fiscal 2013. The Company is no longer subject to U.S. state and local income tax examinations for years prior to fiscal 2004.
In many cases, the Companys uncertain tax positions are related to tax years that remain subject to examination by tax authorities. The following describes open tax years by major tax jurisdictions as of June 30, 2020:
| Years Open | ||||
| Jurisdiction: |
||||
| Canada |
2013 2020 | |||
| France |
2017 2020 | |||
| Germany |
2014 2020 | |||
| United Kingdom |
2017 2020 | |||
| United StatesFederal |
2014 2020 | |||
| United StatesState |
2004 2020 | |||
F-73
Table of Contents
11. Supplemental Disclosures to Consolidated Statements of Cash Flows
Supplemental cash flow information and schedules of noncash investing and financing activities (in millions):
| Years Ended June 30, |
||||||||||||
| 2020 | 2019 | 2018 | ||||||||||
| Cash Paid For: |
||||||||||||
| Cash paid for interest |
$ | 39.2 | $ | 39.2 | $ | 40.0 | ||||||
| Cash paid for income taxes |
10.6 | 11.3 | 11.6 | |||||||||
| Non-Cash Investing and Financing Activities: |
||||||||||||
| Property, plant, and equipment purchases in accounts payable |
2.0 | 2.7 | 1.9 | |||||||||
| Acquisition purchases in accrued expense and other liabilities |
2.8 | | | |||||||||
| Accounts payable converted to note payable to third-parties |
| 0.2 | | |||||||||
The following table provides a reconciliation of cash, cash equivalents and restricted cash reported within the consolidated balances sheets that sum to the total of the same such amounts shown in the consolidated statements of cash flow.
| Years Ended June 30, |
||||||||||||
| 2020 | 2019 | 2018 | ||||||||||
| Cash and cash equivalents |
$ | 118.4 | $ | 35.8 | $ | 33.1 | ||||||
| Restricted cashcurrent |
1.1 | 1.4 | 2.0 | |||||||||
| Restricted cashnoncurrent |
0.5 | 0.4 | 0.8 | |||||||||
|
|
|
|
|
|
|
|||||||
| Total cash, cash equivalents, and restricted cash shown in the statements of cash flow |
$ | 120.0 | $ | 37.6 | $ | 35.9 | ||||||
|
|
|
|
|
|
|
|||||||
Amounts included in restricted cash represent funds with various financial institutions to support performance bonds with irrevocable letters of credit for contractual obligations to certain customers.
12. Employee Benefit Plans
Defined Benefit Pension Plans
The Company maintains contributory and noncontributory defined benefit plans for certain employees in France, Japan and Germany. Plan benefits are generally based on each employees years of service and final salary. The unfunded benefit obligation recognized in the consolidated balance sheets related to these plans was $11.9 million and $11.6 million at June 30, 2020 and 2019, respectively. Benefits expense related to these plans was $1.2 million, $1.1 million and $0.5 million for the years ended June 30, 2020, 2019 and 2018, respectively. The amount recognized in accumulated other comprehensive loss related to these plans was $2.8 million, $3.0 million and $0.7 million at June 30, 2020, 2019 and 2018, respectively. The estimated future benefit payments over the next ten years are $5.7 million. The estimated gains and losses, net, that will be amortized from accumulated other comprehensive income into benefits expense over the next fiscal year are not significant.
Other Postretirement Benefit Plans
The Company maintains a postretirement benefit plan for certain eligible employees in the United States. Under the provisions of the plan, certain retired employees will secure their own health insurance coverage, and the Company will reimburse the retired employee an amount specified in the plan. The unfunded benefit obligation recognized in the consolidated balance sheets related to this plan was $0.7 million at both June 30, 2020 and June 30, 2019. Benefits expense related to these plans was negligible for the fiscal years ended June 30, 2020, 2019 and 2018. The Company also offers a discretionary retirement plan to certain eligible employees whereby they may defer a portion of their compensation until retirement.
F-74
Table of Contents
Defined Contribution Plans
The Company maintains 401(k) savings plans and other voluntary defined contribution retirement plans for other eligible employees. Under each plan, eligible employees may make voluntary contributions, while the Company makes contributions as defined by each plan agreement. Employee contributions in each plan are fully vested and Company contributions vest based on years of service in accordance with the provisions of each plan agreement. Total benefits expense for all defined contribution retirement plans was $3.1 million, $1.7 million, and $2.3 million for the fiscal years ended June 30, 2020, 2019 and 2018, respectively.
13. Share-Based Compensation
The Companys board of directors issued ordinary A shares of Mirion TopCo common stock for the fair value at the time of issuance. In total, the Company has issued 1,483,795 units as of June 30, 2020. The shares were issued subject to certain vesting conditions, restrictions on transfer and repurchase rights (further collectively referred to as Shares) by Mirion TopCo, other employees of the Company or by investors in Mirion TopCo. Under the service vesting conditions, the Shares, vest over four years, with one-quarter vesting after one year of service, and the remainder vesting in equal installments over the subsequent thirty-six months.
Vesting of all Shares is subject to acceleration in the event of certain change of control transactions. The Shares have voting rights and participate in dividends and distributions, if declared; however, the holders of the Shares forfeit their voting rights upon termination of employment regardless of vesting status.
The unvested Shares are subject to repurchase at a price equal to the lesser of (i) the fair value at the issuance date or (ii) the fair value of the Shares as determined on the repurchase date. The Company has determined that this repurchase right is a forfeiture provision and accounted for the Shares issued to management as a share-based compensation arrangement, with a requisite service period of 4 years. The fair value of the Shares were estimated using the Black-Scholes option-pricing model, with the following assumptions:
|
Years Ended June 30, |
||||||||||||
| 2020 | 2019 | 2018 | ||||||||||
| Dividend yield |
0.0 | % | 0.0 | % | 0.0 | % | ||||||
| Risk-free interest rate |
0.2 | % | 2.7 | % | 1.4 | % | ||||||
| Expected volatility |
55.7 | % | 25.1 | % | 92.7 | % | ||||||
| Expected term (in years) |
3 | 2 | 4 | |||||||||
| Fair Value |
$ | 0.37 | $ | 0.16 | $ | 0.57 | ||||||
| 1 | The risk-free rate is based on the US Treasury yields in effect at the time of grant corresponding with the expected term. |
| 2 | Expected volatility is based on historical volatilities from a group of comparable entities for a time period similar to that of the expected term and the expected term. |
| 3 | Expected term is based on probability and expected timing of market events. |
During the years ended June 30, 2020 and 2019, no options existed or were granted.
F-75
Table of Contents
A summary of restricted stock activity within the Companys equity plans and changes for the years ended June 30, 2020 and 2019, is as follows:
| Restricted Stock Awards |
Shares (in millions) |
Weighted Average Grant- Date Fair Value |
Total Fair Value (in millions) |
|||||||||
| Nonvested at July 1, 2018 |
0.6 | $ | 0.57 | $ | 0.3 | |||||||
| Granted |
0.2 | 0.16 | 0.1 | |||||||||
| Vested |
(0.2 | ) | 0.57 | (0.1 | ) | |||||||
| Repurchased |
(0.2 | ) | 0.57 | (0.1 | ) | |||||||
|
|
|
|
|
|
|
|||||||
| Nonvested at June 30, 2019 |
0.4 | $ | 0.39 | $ | 0.2 | |||||||
| Granted |
0.2 | 0.37 | 0.1 | |||||||||
| Vested |
(0.1 | ) | 0.27 | | ||||||||
| Repurchased |
(0.1 | ) | 0.57 | (0.1 | ) | |||||||
|
|
|
|
|
|
|
|||||||
| Nonvested at June 30, 2020 |
0.4 | $ | 0.41 | $ | 0.1 | |||||||
As a result of management turnover, the Company repurchased and reissued 144,219 and 218,428 shares to other members of the management team during fiscal year ended June 30, 2020 and 2019, respectively. Any forfeited shares of restricted common stock were treated as a cancellation with remaining unrecognized expense for the unvested awards recognized on the date of cancellation. The Company did not reverse previously recognized compensation expenses as a result of these cancellations.
Total share-based compensation expense in our consolidated statement of operations and comprehensive loss for fiscal 2020, 2019 and 2018 was $0.2 million, $0.1 million and $0.2 million, respectively. The total value of the shares is amortized as compensation expense ratably over the vesting period of each individual tranche, beginning at the grant date. As of June 30, 2020, there was $0.3 million of unrecognized compensation expense related to the Shares that is expected to be recognized over the remaining service period.
14. Related-Party Transactions
Charterhouse Capital Partners LLP (CCP)
The Company has entered into agreements with its primary investor, CCP, which obligate the Company to pay quarterly management fees of $0.1 million per year. In return, CCP provides various investment banking services relating to financing arrangements, mergers and acquisitions and other services. During the fiscal years ended June 30, 2020 and 2019, the Company paid CCP and an aggregate of $0.3 million and $0.2 million for professional fees and expense reimbursements in each respective fiscal year. During the fiscal year ended June 30, 2018, the Company reimbursed CCP $0.2 million for travel, professional fees and other expense.
Receivable from Employees for purchase of Common Stock
In conjunction with the Share-Based Compensation, as discussed in Note 13, the Company has made loans to certain members of the management team, to acquire the Shares at fair value, which are paid back to the Company over the requisite service period. As of June 30, 2020 and 2019, the outstanding balance approximated $2.7 million and $2.5 million, respectively, and are classified within stockholders equity on the Companys consolidated balance sheet as they represent a receivable in payment of shares. Payments made by the related employees will be recorded as an increase to stockholders equity.
See Note 8 for notes payable to related parties.
15. Segment Information
The Company manages its operations through two operating and reportable segments: Medical and Industrial. These segments align the Companys products and service offerings with customer use in medical and industrial
F-76
Table of Contents
markets and are consistent with how the Companys Chief Executive Officer, its Chief Operating Decision Maker (CODM), reviews and evaluates the Companys operations. The CODM allocates resources and evaluates the financial performance of each operating segment. The Companys segments are strategic businesses that are managed separately because each one develops, manufactures and markets distinct products and services. Prior period information herein has been conformed to the current reportable segment structure.
Description of Segments
The Medical segment provides radiation oncology quality assurance, delivering patient safety solutions for diagnostic imaging and radiation therapy centers around the world, dosimetry solutions for monitoring the total amount of radiation medical staff members are exposed to over time, radiation therapy quality assurance solutions for calibrating and verifying imaging and treatment accuracy, and radionuclide therapy products for nuclear medicine applications such as shielding, product handling, medical imaging furniture, and rehabilitation products.
The Industrial segment provides robust, field ready personal radiation detection and identification equipment for defense applications and radiation detection and analysis tools for power plants, labs, and research applications. Nuclear power plant product offerings are used for the full nuclear power plant lifecycle including core detectors and essential measurement devices for new build, maintenance, decontamination and decommission equipment for monitoring and control during fuel dismantling and remote environmental monitoring.
F-77
Table of Contents
The following table summarizes select operating results for each reportable segment. The CODM evaluates operating results and allocates capital resources among segments, in part, based on segment income from operations, which includes revenues of the segment less expenses that are directly related to those revenues, including purchase accounting impacts to revenue and cost of revenues, but excluding certain charges to cost of revenues and selling, general and administrative expenses predominantly related to corporate costs, shared overhead and other non operational costs related to restructuring activities and costs to achieve operational initiatives, which are included in Corporate and Other in the table below. Interest expense, loss on debt extinguishment, foreign currency loss (gain), net, and other expense (income), net are not allocated to segments.
| For Years Ended | ||||||||||||
| (In millions) | June 30, 2020 |
June 30, 2019 |
June 30, 2018 |
|||||||||
| Revenue |
||||||||||||
| Medical |
62.6 | 42.9 | 37.8 | |||||||||
| Industrial |
415.6 | 397.2 | 406.3 | |||||||||
|
|
|
|
|
|
|
|||||||
| Consolidated revenue |
$ | 478.2 | $ | 440.1 | $ | 444.1 | ||||||
| Segment Income from Operations |
||||||||||||
| Medical |
13.9 | 10.2 | 6.0 | |||||||||
| Industrial |
59.6 | 55.0 | 46.9 | |||||||||
|
|
|
|
|
|
|
|||||||
| Total Segment Income from Operations |
$ | 73.5 | $ | 65.2 | $ | 52.9 | ||||||
| Corporate and other |
(50.5 | ) | (36.4 | ) | (57.6 | ) | ||||||
|
|
|
|
|
|
|
|||||||
| Consolidated Income from Operations |
$ | 23.0 | $ | 28.8 | $ | (4.7 | ) | |||||
| Capital Expenditures |
||||||||||||
| Medical |
10.1 | 8.0 | 5.4 | |||||||||
| Industrial |
11.4 | 10.4 | 9.2 | |||||||||
|
|
|
|
|
|
|
|||||||
| Total operating and reportable segments |
21.5 | 18.4 | 14.6 | |||||||||
| Corporate and other |
0.4 | 0.8 | (0.1 | ) | ||||||||
|
|
|
|
|
|
|
|||||||
| Total Capital Expenditures |
$ | 21.9 | $ | 19.2 | $ | 14.5 | ||||||
| Depreciation and Amortization |
||||||||||||
| Medical |
15.8 | 15.4 | 15.6 | |||||||||
| Industrial |
52.2 | 53.7 | 60.8 | |||||||||
|
|
|
|
|
|
|
|||||||
| Total operating and reportable segments |
68.0 | 69.1 | 76.4 | |||||||||
| Corporate and other |
0.4 | 0.4 | 0.6 | |||||||||
|
|
|
|
|
|
|
|||||||
| Total Depreciation and Amortization |
$ | 68.4 | $ | 69.5 | $ | 77.0 | ||||||
The Companys assets by reportable segment were not included, as this information is not reviewed by, nor otherwise provided to, the chief operating decision maker to make operating decisions or allocate resources.
F-78
Table of Contents
The following details revenue and property, plant, and equipment by geographic region. Revenues generated from external customers are attributed to geographic regions through sales from site locations.
| Revenues for Year Ended | ||||||||||||
| (In millions) | June 30, 2020 |
June 30, 2019 |
June 30, 2018 |
|||||||||
| North America |
||||||||||||
| Medical |
57.5 | 41.0 | 37.8 | |||||||||
| Industrial |
193.3 | 188.3 | 179.1 | |||||||||
|
|
|
|
|
|
|
|||||||
| Total North America |
250.8 | 229.3 | 216.9 | |||||||||
| Europe |
||||||||||||
| Medical |
5.2 | 1.9 | | |||||||||
| Industrial |
206.2 | 194.9 | 214.2 | |||||||||
|
|
|
|
|
|
|
|||||||
| Total Europe |
211.4 | 196.8 | 214.2 | |||||||||
| Asia Pacific |
||||||||||||
| Medical |
| | | |||||||||
| Industrial |
16.0 | 14.0 | 13.0 | |||||||||
|
|
|
|
|
|
|
|||||||
| Total Asia Pacific |
16.0 | 14.0 | 13.0 | |||||||||
|
|
|
|
|
|
|
|||||||
| Total |
$ | 478.2 | $ | 440.1 | $ | 444.1 | ||||||
|
|
|
|
|
|
|
|||||||
Revenues generated in the United States were $215.5 million, $198.3 million, and $185.5 million for the years ended June 30, 2020, 2019, and 2018, respectively, while revenue in France were $134.5 million, $128.0 million, and $134.9 million, respectively.
| Property, Plant, and Equipment, net |
||||||||
| (In millions) | As of June 30, 2020 |
As of June 30, 2019 |
||||||
| North America |
$ | 36.5 | $ | 31.3 | ||||
| Europe |
38.6 | 34.4 | ||||||
| Asia Pacific |
0.1 | 0.1 | ||||||
|
|
|
|
|
|||||
| Total |
$ | 75.2 | $ | 65.8 | ||||
|
|
|
|
|
|||||
The following details revenue by timing of recognition:
| Revenues for Year Ended | ||||||||||||
| (In millions) | June 30, 2020 |
June 30, 2019 |
June 30, 2018 |
|||||||||
| Point in time |
$ | 337.3 | $ | 331.1 | $ | 328.3 | ||||||
| Over time |
140.9 | 109.0 | 115.8 | |||||||||
|
|
|
|
|
|
|
|||||||
| Total revenues |
$ | 478.2 | $ | 440.1 | $ | 444.1 | ||||||
|
|
|
|
|
|
|
|||||||
F-79
Table of Contents
16. Loss Per Share
A reconciliation of the numerator and denominator used in the calculation of basic and diluted loss per common share is as follows:
| Year Ended June 30, |
||||||||||||
| In millions, except per share amounts | 2020 | 2019 | 2018 | |||||||||
| Net loss attributable to Mirion Technologies (TopCo), Ltd. stockholders |
$ | (119.1 | ) | $ | (122.0 | ) | $ | (103.4 | ) | |||
| Weighted average common shares outstandingbasic and diluted |
6.453 | 6.300 | 5.929 | |||||||||
|
|
|
|
|
|
|
|||||||
| Loss per share attributable to Mirion Technologies (TopCo), Ltd. stockholdersbasic and diluted |
$ | (18.45 | ) | $ | (19.36 | ) | $ | (17.44 | ) | |||
|
|
|
|
|
|
|
|||||||
| Anti-dilutive employee share-based awards, excluded |
0.4 | 0.5 | 0.9 | |||||||||
Potentially dilutive unvested stock awards using the treasury stock method or the if-converted method, as applicable, are included when calculating diluted net income earnings (loss) per share attributable to Mirion Technologies (TopCo), Ltd. when their effect is dilutive. Because the Company incurred a net loss for the fiscal years ending June 30, 2020, 2019 and 2018 respectively, none of the potentially dilutive common shares were included in the diluted share calculations for those periods as they would have been anti-dilutive.
17. Parent Company Financial Information
Mirion Technologies (TopCo), Ltd. has no material assets or standalone operations other than its ownership in its consolidated subsidiaries. There are restrictions under credit agreements governing the 2019 Credit Facility, described in Note 8, on the Companys ability to obtain funds from any of its subsidiaries through dividends. Accordingly, the following condensed financial information is presented on a Parent-only basis in which Mirion Technologies (TopCo), Ltd.s investments in its consolidated subsidiaries are presented under the equity method of accounting.
F-80
Table of Contents
MIRION TECHNOLOGIES (TOPCO), LTD.
(PARENT COMPANY ONLY)
CONDENSED BALANCE SHEET
(in millions)
| Years Ended June 30, |
||||||||
| 2020 | 2019 | |||||||
| Assets: |
||||||||
| Other assets |
$ | 0.1 | $ | 0.1 | ||||
|
|
|
|
|
|||||
| Total Assets |
$ | 0.1 | $ | 0.1 | ||||
|
|
|
|
|
|||||
| Liabilities and Stockholders Equity: |
||||||||
| Investment in subsidiary |
716.5 | 587.7 | ||||||
| Deferred income taxes and other liabilities |
$ | 0.1 | $ | 0.1 | ||||
|
|
|
|
|
|||||
| Total Liabilities |
$ | 716.6 | $ | 587.8 | ||||
| Total Stockholders Equity |
(716.5 | ) | (587.7 | ) | ||||
|
|
|
|
|
|||||
| Total Liabilities and Stockholders Equity |
$ | 0.1 | $ | 0.1 | ||||
|
|
|
|
|
|||||
MIRION TECHNOLOGIES (TOPCO), LTD.
(PARENT COMPANY ONLY)
CONDENSED STATEMENT OF OPERATIONS AND COMPREHENSIVE LOSS INCOME
(in millions)
| Years Ended June 30, |
||||||||||||
| 2020 | 2019 | 2018 | ||||||||||
| Equity in net loss of subsidiaries |
$ | (119.1 | ) | $ | (122.0 | ) | $ | (103.4 | ) | |||
|
|
|
|
|
|
|
|||||||
| Net loss |
(119.1 | ) | (122.0 | ) | (103.4 | ) | ||||||
| Foreign currency translation, net of tax |
(9.3 | ) | (15.1 | ) | 12.0 | |||||||
| Unrecognized actuarial gain (loss) and prior service benefit, net of tax |
| (1.5 | ) | (0.1 | ) | |||||||
|
|
|
|
|
|
|
|||||||
| Other comprehensive loss (income), net of tax |
(9.3 | ) | (16.6 | ) | 11.9 | |||||||
|
|
|
|
|
|
|
|||||||
| Comprehensive loss |
$ | (128.4 | ) | $ | (138.6 | ) | $ | (91.5 | ) | |||
|
|
|
|
|
|
|
|||||||
| Loss per sharebasic and diluted |
$ | (18.45 | ) | $ | (19.36 | ) | $ | (17.44 | ) | |||
|
|
|
|
|
|
|
|||||||
| Weighted average number of shares outstandingbasic and diluted |
6.453 | 6.300 | 5.929 | |||||||||
|
|
|
|
|
|
|
|||||||
A statement of cash flows has not been presented as Mirion Technologies (TopCo). Ltd. parent company did not have any cash as of, or at any point in time during the years ended June 30, 2020, 2019 or 2018.
F-81
Table of Contents
Note to Condensed Financial Statements of Registrant (Parent Company Only)
Basis of Presentation
These condensed parent company-only financial statements have been prepared in accordance with Rule 12-04, Schedule I of Regulation S-X, as the restricted net assets of the subsidiaries of Mirion TopCo. (as defined in Rule 4-08(e)(3) of Regulation S-X) exceed the specified threshold amount of the consolidated net assets of the Company. Because we have a consolidated accumulated deficit, the 25% threshold described in Rule 4-08 does not apply and any restrictions of net assets at our subsidiaries trigger the requirement to present parent company-only financial information. The ability of Mirion Topcos operating subsidiaries to pay dividends may be restricted due to the terms of the subsidiaries outstanding term loan and revolving credit facility borrowings as described in Note 8 to the audited consolidated financial statements.
These condensed parent company-only financial statements have been prepared using the same accounting principles and policies described in the notes to the consolidated financial statements, with the only exception being that the parent company accounts for its subsidiaries using the equity method. These condensed parent company-only financial statements should be read in conjunction with the consolidated financial statements and related notes thereto included elsewhere in this prospectus.
18. Subsequent Events
Management has evaluated subsequent events through June 29, 2021, the date the financial statements were issued.
Biodex Acquisition
On September 1, 2020, the Company purchased 100% of the issued and outstanding shares of Biodex Medical Systems, Inc. (Biodex), a manufacturer and distributor of medical devices and related replacement parts for physical and nuclear medicine, as well as medical imaging applications located in the United States. The acquisition of Biodex will advance the Companys strategy to further expand into the medical treatment markets globally and will be reported within the Companys Medical segment.
The acquisition of Biodex was accounted for under the acquisition method of accounting, and the related assets acquired and liabilities assumed are recorded at fair value. The Company makes an initial allocation of the purchase price at the date of acquisition based upon its understanding of the fair value of the acquired assets and assumed liabilities. Determining the fair value of assets and liabilities assumed is obtained from information and knowledge gathered through the due diligence process and other sources. Fair values are preliminary and subject to refinement for up to one year after the closing date of the acquisition as additional information regarding the closing date fair values becomes available.
F-82
Table of Contents
The Company is still finalizing the fair value estimates, but the following table presents the preliminary estimates of the fair value of the assets acquired and liabilities assumed for the material Biodex acquisition on September 1, 2020 (in millions):
| Biodex | ||||
| Date of acquisition |
September 1, 2020 | |||
| Segment |
Medical | |||
| Goodwill |
$ | 10.4 | ||
| Customer relationships (1) |
2.3 | |||
|
Tradenames (2) |
1.4 | |||
| Non-Compete Agreements (3) |
0.3 | |||
| Developed Technology (4) |
2.6 | |||
|
|
|
|||
| Amortizable intangible assets |
$ | 6.6 | ||
| Cash |
4.1 | |||
| Accounts receivable |
4.0 | |||
| Inventory |
6.4 | |||
| Property, Plant and Equipment |
1.0 | |||
| Other current and non-current assets |
1.5 | |||
| Current liabilities |
(2.8 | ) | ||
| Deferred revenue |
(0.2 | ) | ||
|
|
|
|||
| Net tangible assets acquired |
$ | 14.0 | ||
|
|
|
|||
| Purchase consideration |
31.0 | |||
| Less: cash acquired |
(4.1 | ) | ||
|
|
|
|||
| Purchase consideration, net of cash acquired |
$ | 26.9 | ||
Sun Nuclear Acquisition
On December 18, 2020, the Company purchased 100% of the issued and outstanding shares of Sun Nuclear Corporation (SNC), global leader in radiation oncology quality assurance, delivering patient safety solutions for diagnostic imaging and radiation therapy centers around the world. The acquisition of SNC will advance the Companys strategy to further expand into the radiation treatment markets globally and will be reported within the Companys Medical segment.
The acquisition of SNC was accounted for under the acquisition method of accounting, and the related assets acquired and liabilities assumed are recorded at fair value. The Company makes an initial allocation of the purchase price at the date of acquisition based upon its understanding of the fair value of the acquired assets and assumed liabilities. Determining the fair value of assets and liabilities assumed is obtained from information and knowledge gathered through the due diligence process and other sources. Fair values are preliminary and subject to refinement for up to one year after the closing date of the acquisition as additional information regarding the closing date fair values becomes available.
F-83
Table of Contents
The Company is still finalizing the fair value estimates, but the following table presents the preliminary estimates of the fair value of the assets acquired and liabilities assumed for the material SNC acquisition on December 18, 2020 (in millions):
| SNC | ||||
| Date of acquisition |
December 18, 2020 | |||
| Segment |
Medical | |||
| Goodwill |
$ | 130.1 | ||
| Customer relationships (1) |
59.5 | |||
|
Tradenames (2) |
12.0 | |||
| Non-Compete Agreements (3) |
7.5 | |||
| Developed Technology (4) |
46.5 | |||
|
|
|
|||
| Amortizable intangible assets |
$ | 125.5 | ||
| Cash |
18.8 | |||
| Accounts receivable |
24.0 | |||
| Inventory |
13.9 | |||
| Property, Plant and Equipment |
5.9 | |||
| Other current and non-current assets |
5.7 | |||
| Current liabilities |
(9.3 | ) | ||
| Deferred revenue |
(6.5 | ) | ||
| Other long-term liabilities |
(31.2 | ) | ||
|
|
|
|||
| Net tangible assets acquired |
$ | 21.3 | ||
|
|
|
|||
| Purchase consideration |
276.9 | |||
| Less: cash acquired |
(18.8 | ) | ||
|
|
|
|||
| Purchase consideration, net of cash acquired |
$ | 258.1 | ||
Unaudited Pro Forma Financial Information
The following unaudited pro forma financial information presents the Companys results of operations for the twelve-months ended June 30, 2020 and 2019 to illustrate the estimated effects of the acquisitions of Biodex and SNC as if they had occurred on July 1, 2018. The pro forma financial information is presented for comparative purposes only and is not necessarily indicative of the Companys operating results that may have actually occurred had the acquisitions of Biodex and SNC been completed on July 1, 2018. The unaudited pro forma financial information does not reflect the expected realization of any anticipated cost savings, operating efficiencies, or other synergies that may have been associated with the acquisition.
| (amounts in millions) | Twelve months ended June 30, |
|||||||
| 2020 | 2019 | |||||||
| Total revenues |
$ | 613.5 | $ | 554.6 | ||||
| Net loss |
(134.8 | ) | (163.5 | ) | ||||
| Net loss attributable to Mirion Technologies (TopCo), Ltd stockholders |
(134.8 | ) | (163.5 | ) | ||||
The unaudited pro forma financial information reflects pro forma adjustments to present the combined pro forma results of the operations as if the acquisition had occurred on July 1, 2018 to give effect to certain events the Company believes to be directly attributable to the acquisitions. These pro forma adjustments primarily include:
| | A net increase in cost of revenues, depreciation and amortization expense that would have been recognized due to acquired inventory, property, plant and equipment and intangible assets; |
F-84
Table of Contents
| | An increase to interest expense to reflect the additional borrowings of the Company in conjunction with the acquisition; |
| | A reduction in expenses for the twelve months ended June 30, 2020 and a corresponding increase in the twelve months ended June 30, 2019 for acquisition-related transaction costs directly attributable to the acquisition; |
| | A reduction in revenues due to the elimination of deferred revenues assigned no value at the acquisition date; |
| | An increase in income tax expense using the U.S. statutory rate of 25% to reflect a change in tax status had the SNC and Biodex results of operations been included in the Companys consolidated tax return; and |
| | The related income tax effects of the adjustments noted above. |
Business Combination
On June 17, 2021, Mirion entered into a Business Combination Agreement with GS Acquisition Holdings Corp II (GSAH) a special purpose acquisition company. In accordance with the terms and subject to the conditions of the Business Combination Agreement, the consideration to be paid to Mirions stockholders and holders of Shareholder Notes in connection with the Business Combination is $1.7 billion, of which $1.3 billion will be paid in cash and the remainder will consist of shares of GSAH common stock. The balance of the 2019 Credit Facility as of the closing date will be paid in full through cash consideration or the new financing per the Commitment Letter described below. The transaction is expected to close in the second half of calendar year 2021, subject to certain closing conditions, including regulatory approvals, and approval of GSAHs stockholders.
Commitment Letter
In connection with the transaction, Mirion Technologies (HoldingSub2) Ltd., a subsidiary of Mirion TopCo, entered into a commitment letter (the Commitment Letter) with Goldman Sachs Lending Partners LLC (GS Lending) and Citigroup Global Markets Inc. (Citi) pursuant to which GS Lending and Citi have committed to provide to Mirion Technologies, Inc., as the subsidiary borrower, and a parent entity of the Mirion TopCo business to be formed, a $830 million senior secured term loan B facility (the Term Loan) and a $90 million revolving facility (the Revolving Facility). The Term Loan will mature seven years after the closing date and will amortize in equal quarterly installments in an aggregate annual amount equal to 1% of the initial principal amount of the Term Loan. The Revolving Facility will mature five years after the closing date.
F-85
Table of Contents
Valuation and Qualifying Accounts
(In millions)
| Description |
Balance at Beginning of Period |
Charged to Costs and Expenses |
Deductions (a) | Other (b) | Balance at End of Period |
|||||||||||||||
| Year Ended June 30, 2020 |
||||||||||||||||||||
| Allowance for doubtful accounts |
$ | 1.7 | $ | 0.9 | $ | (0.7 | ) | $ | | $ | 1.9 | |||||||||
| Product warranty |
4.2 | 2.9 | (1.6 | ) | | 5.5 | ||||||||||||||
| Year Ended June 30, 2019 |
||||||||||||||||||||
| Allowance for doubtful accounts |
$ | 1.9 | $ | 0.2 | $ | (0.4 | ) | $ | | $ | 1.7 | |||||||||
| Product warranty |
5.4 | 1.5 | (2.6 | ) | (0.1 | ) | 4.2 | |||||||||||||
| Year Ended June 30, 2018 |
||||||||||||||||||||
| Allowance for doubtful accounts |
$ | 3.2 | $ | 0.2 | $ | (1.5 | ) | $ | | $ | 1.9 | |||||||||
| Product warranty |
5.1 | 1.3 | (1.1 | ) | 0.1 | 5.4 | ||||||||||||||
| (a) | Charges to the accounts included in this column are for the purposes for which the reserves were created |
| (b) | Amounts included in this column relate to foreign currency translation and tax valuation increases from business combinations |
F-86
Table of Contents
Mirion Technologies (TopCo), Ltd.
Unaudited Interim Condensed Consolidated Financial Statements
As of March 31, 2021 and June 30, 2020 and
for the nine months ended March 31, 2021 and 2020
Contents
| Unaudited Interim Condensed Consolidated Financial Statements |
||||
| F-88 | ||||
| Unaudited Interim Condensed Consolidated Statements of Operations |
F-89 | |||
| Unaudited Interim Condensed Consolidated Statements of Comprehensive Loss |
F-90 | |||
| Unaudited Interim Condensed Consolidated Statements of Stockholders Deficit |
F-91 | |||
| Unaudited Interim Condensed Consolidated Statements of Cash Flows |
F-92 | |||
| Notes to Unaudited Interim Condensed Consolidated Financial Statements |
F-93 | |||
F-87
Table of Contents
Mirion Technologies (TopCo), Ltd.
Unaudited Interim Condensed Consolidated Balance Sheets
(In millions, except share data)
|
March 31, 2021 |
June 30, 2020 |
|||||||
| ASSETS |
||||||||
| Current assets: |
||||||||
| Cash and cash equivalents |
$ | 86.8 | $ | 118.4 | ||||
| Restricted cash |
0.8 | 1.1 | ||||||
| Accounts receivable, net of allowance for doubtful accounts of $5.5 and $1.9, respectively |
127.1 | 97.3 | ||||||
| Costs in excess of billings on uncompleted contracts |
59.7 | 59.5 | ||||||
| Inventories |
118.1 | 90.2 | ||||||
| Deferred cost of revenue |
6.5 | 6.5 | ||||||
| Prepaid expenses and other currents assets |
24.9 | 16.7 | ||||||
|
|
|
|
|
|||||
| Total current assets |
423.9 | 389.7 | ||||||
| Property, plant, and equipment, net |
89.0 | 75.2 | ||||||
| Goodwill |
677.5 | 522.6 | ||||||
| Intangible assets, net |
343.3 | 248.3 | ||||||
| Restricted cash |
0.1 | 0.5 | ||||||
| Other assets |
10.8 | 7.5 | ||||||
|
|
|
|
|
|||||
| Total assets |
$ | 1,544.6 | $ | 1,243.8 | ||||
|
|
|
|
|
|||||
| LIABILITIES AND STOCKHOLDERS DEFICIT |
||||||||
| Current liabilities: |
||||||||
| Accounts payable |
$ | 55.5 | $ | 38.7 | ||||
| Deferred contract revenue |
49.9 | 39.6 | ||||||
| Notes payable to third-parties, current |
5.5 | 41.1 | ||||||
| Accrued expenses and other current liabilities |
91.7 | 64.1 | ||||||
|
|
|
|
|
|||||
| Total current liabilities |
202.6 | 183.5 | ||||||
| Notes payable to related parties, non-current |
1,170.3 | 987.1 | ||||||
| Notes payable to third-parties, non-current |
885.5 | 669.8 | ||||||
| Interest accrued on notes payable to related parties |
32.2 | 56.4 | ||||||
| Deferred income taxes and other liabilities |
72.8 | 63.5 | ||||||
|
|
|
|
|
|||||
| Total liabilities |
2,363.4 | 1,960.3 | ||||||
| Commitments and contingencies (Note 9) |
||||||||
| Stockholders deficit: |
||||||||
| A Ordinary shares, $0.01 nominal value, 3,000,000 shares authorized, 1,483,795 issued and outstanding at both March 31, 2021 and June 30, 2020 |
| | ||||||
| B Ordinary shares, $0.01 nominal value, 7,000,000 shares authorized, 5,353,970 issued and outstanding at both March 31, 2021 and June 30, 2020 |
0.1 | 0.1 | ||||||
| Additional paid-in capital |
9.5 | 9.5 | ||||||
| Receivable from Employees for purchase of Common Stock |
(2.5 | ) | (2.7 | ) | ||||
| Accumulated deficit |
(860.6 | ) | (729.7 | ) | ||||
| Accumulated other comprehensive income |
32.6 | 4.1 | ||||||
|
|
|
|
|
|||||
| Mirion Technologies (TopCo), Ltd. stockholders deficit |
(820.9 | ) | (718.7 | ) | ||||
| Noncontrolling interests |
2.1 | 2.2 | ||||||
|
|
|
|
|
|||||
| Total stockholders deficit |
(818.8 | ) | (716.5 | ) | ||||
|
|
|
|
|
|||||
| Total liabilities and stockholders deficit |
$ | 1,544.6 | $ | 1,243.8 | ||||
|
|
|
|
|
|||||
The accompanying notes are an integral part of these unaudited interim condensed consolidated financial statements.
F-88
Table of Contents
Mirion Technologies (TopCo), Ltd.
Unaudited Interim Condensed Consolidated Statements of Operations
(In millions, except per share data)
| Nine months ended March 31, |
||||||||
| 2021 | 2020 | |||||||
| Revenues: |
||||||||
| Product |
$ | 318.3 | $ | 246.5 | ||||
| Service |
113.3 | 90.5 | ||||||
|
|
|
|
|
|||||
| Total revenues |
431.6 | 337.0 | ||||||
| Cost of revenues: |
||||||||
| Product |
200.5 | 152.8 | ||||||
| Service |
59.0 | 48.4 | ||||||
|
|
|
|
|
|||||
| Total cost of revenues |
259.5 | 201.2 | ||||||
|
|
|
|
|
|||||
| Gross profit |
172.1 | 135.8 | ||||||
| Operating expenses: |
||||||||
| Selling, general and administrative |
144.4 | 116.4 | ||||||
| Research and development |
21.2 | 12.3 | ||||||
|
|
|
|
|
|||||
| Total operating expenses |
165.6 | 128.7 | ||||||
|
|
|
|
|
|||||
| Income from operations |
6.5 | 7.1 | ||||||
| Other expense (income): |
||||||||
| Third party interest expense |
30.0 | 31.2 | ||||||
| Related party interest expense |
89.5 | 79.4 | ||||||
| Foreign currency (gain) loss, net |
12.3 | (4.0 | ) | |||||
| Other (income) expense, net |
(0.5 | ) | (0.1 | ) | ||||
|
|
|
|
|
|||||
| Loss before provision (benefit) from income taxes |
(124.8 | ) | (99.4 | ) | ||||
| Provision (benefit) from income taxes |
6.2 | (4.8 | ) | |||||
|
|
|
|
|
|||||
| Net loss |
(131.0 | ) | (94.6 | ) | ||||
| Loss attributable to noncontrolling interests |
(0.1 | ) | | |||||
|
|
|
|
|
|||||
| Net loss attributable to Mirion Technologies (TopCo), Ltd. stockholders |
$ | (130.9 | ) | $ | (94.6 | ) | ||
| Loss per common share attributable to Mirion Technologies (TopCo), Ltd stockholdersbasic and diluted |
$ | (20.06 | ) | $ | (14.70 | ) | ||
| Weighted average common shares outstandingbasic and diluted |
6.525 | 6.435 | ||||||
|
|
|
|
|
|||||
The accompanying notes are an integral part of these unaudited interim condensed consolidated financial statements.
F-89
Table of Contents
Mirion Technologies (TopCo), Ltd.
Unaudited Interim Condensed Consolidated Statements of Comprehensive Loss
(In millions)
| Nine months ended March 31, |
||||||||
| 2021 | 2020 | |||||||
| Net loss |
$ | (131.0 | ) | $ | (94.6 | ) | ||
| Other comprehensive loss, net of tax: |
||||||||
| Foreign currency translation, net of tax of $0 and $0, respectively |
28.2 | (20.7 | ) | |||||
| Unrecognized actuarial gain (loss) and prior service benefit, net of tax of $(0.1) and $0, respectively |
0.3 | 0.7 | ||||||
|
|
|
|
|
|||||
| Other comprehensive income (loss), net of tax |
28.5 | (20.0 | ) | |||||
|
|
|
|
|
|||||
| Comprehensive loss |
(102.5 | ) | (114.6 | ) | ||||
| Less: Comprehensive income (loss) attributable to noncontrolling interest |
(0.1 | ) | | |||||
|
|
|
|
|
|||||
| Comprehensive loss attributable to Mirion Technologies (TopCo), Ltd. stockholders |
$ | (102.4 | ) | $ | (114.6 | ) | ||
|
|
|
|
|
|||||
The accompanying notes are an integral part of these unaudited interim condensed consolidated financial statements.
F-90
Table of Contents
Mirion Technologies (TopCo), Ltd.
Unaudited Interim Condensed Consolidated Statements of Stockholders Deficit
(In millions, except share amounts)
| Nine months ended March 31, 2021 | ||||||||||||||||||||||||||||||||||||||||
| A Ordinary Shares |
A Ordinary Amount |
B Ordinary Shares |
B Ordinary Amount |
Additional Paid-In Capital |
Receivable from Employees for purchase of Common Stock |
Accumulated Deficit |
Accumulated Other Comprehensive Income (Loss) |
Noncontrolling Interests |
Total Stockholders Deficit |
|||||||||||||||||||||||||||||||
| Balance June 30, 2020 |
1,483,795 | $ | | 5,353,970 | $ | 0.1 | $ | 9.5 | $ | (2.7 | ) | $ | (729.7 | ) | $ | 4.1 | $ | 2.2 | $ | (716.5 | ) | |||||||||||||||||||
| Receivable from Employees |
| | | | | 0.2 | | | | 0.2 | ||||||||||||||||||||||||||||||
| Net loss |
| | | | | | (130.9 | ) | | (0.1 | ) | (131.0 | ) | |||||||||||||||||||||||||||
| Other comprehensive loss |
| | | | | | | 28.5 | | 28.5 | ||||||||||||||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||||||||
| Balance March 31, 2021 |
1,483,795 | $ | | 5,353,970 | $ | 0.1 | $ | 9.5 | $ | (2.5 | ) | $ | (860.6 | ) | $ | 32.6 | $ | 2.1 | $ | (818.8 | ) | |||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||||||||
| Nine months ended March 31, 2020 | ||||||||||||||||||||||||||||||||||||||||
| A Ordinary Shares |
A Ordinary Amount |
B Ordinary Shares |
B Ordinary Amount |
Additional Paid-In Capital |
Receivable from Employees for purchase of Common Stock |
Accumulated Deficit |
Accumulated Other Comprehensive Income (Loss) |
Noncontrolling Interests |
Total Stockholders Deficit |
|||||||||||||||||||||||||||||||
| Balance June 30, 2019 |
1,483,795 | $ | | 5,353,970 | $ | 0.1 | $ | 9.3 | $ | (2.5 | ) | $ | (610.6 | ) | $ | 13.4 | $ | 2.6 | $ | (587.7 | ) | |||||||||||||||||||
| Distribution to noncontrolling interests |
| | | | | | | | (0.4 | ) | (0.4 | ) | ||||||||||||||||||||||||||||
| Share-based compensation expense |
| | | | 0.1 | | | | | 0.1 | ||||||||||||||||||||||||||||||
| Net loss |
| | | | | | (94.6 | ) | | (94.6 | ) | |||||||||||||||||||||||||||||
| Other comprehensive loss |
| | | | | | | (20.0 | ) | | (20.0 | ) | ||||||||||||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||||||||
| Balance March 31, 2020 |
1,483,795 | $ | | 5,353,970 | $ | 0.1 | $ | 9.4 | $ | (2.5 | ) | $ | (705.2 | ) | $ | (6.6 | ) | $ | 2.2 | $ | (702.6 | ) | ||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||||||||
The accompanying notes are an integral part of these unaudited consolidated financial statements.
F-91
Table of Contents
Mirion Technologies (TopCo), Ltd.
Unaudited Interim Condensed Consolidated Statements of Cash Flows
(In millions)
|
Nine months ended March 31, |
||||||||
| 2021 | 2020 | |||||||
| OPERATING ACTIVITIES: |
||||||||
| Net loss |
$ | (131.0 | ) | $ | (94.6 | ) | ||
| Adjustments to reconcile net loss to net cash provided by operating activities: |
||||||||
| Accrual of in-kind interest on notes payable to related parties |
88.7 | 79.3 | ||||||
| Depreciation and amortization expense |
58.2 | 51.6 | ||||||
| Share-based compensation expense |
| 0.1 | ||||||
| Amortization of debt issuance costs |
2.3 | 2.3 | ||||||
| Provision for doubtful accounts |
1.1 | 0.1 | ||||||
| Inventory obsolescence write down |
0.5 | 0.9 | ||||||
| Change in deferred income taxes |
(17.6 | ) | 4.9 | |||||
| Loss (Gain) on disposal of property, plant and equipment |
(0.3 | ) | | |||||
| Loss (Gain) on foreign currency transactions |
12.3 | (4.5 | ) | |||||
| Other |
1.5 | (3.5 | ) | |||||
| Changes in operating assets and liabilities: |
||||||||
| Accounts receivable |
2.1 | 15.8 | ||||||
| Costs in excess of billings on uncompleted contracts |
(3.9 | ) | 1.1 | |||||
| Inventories |
(4.2 | ) | (4.2 | ) | ||||
| Deferred cost of revenue |
0.3 | (2.4 | ) | |||||
| Prepaid expenses and other current assets |
(7.4 | ) | (1.1 | ) | ||||
| Accounts payable |
12.0 | (8.8 | ) | |||||
| Accrued expenses and other current liabilities |
5.4 | (15.5 | ) | |||||
| Deferred contract revenue |
3.5 | 2.4 | ||||||
| Other assets |
2.9 | (0.4 | ) | |||||
| Other liabilities |
4.3 | (6.3 | ) | |||||
|
|
|
|
|
|||||
| Net cash provided by operating activities |
30.7 | 17.2 | ||||||
|
|
|
|
|
|||||
| INVESTING ACTIVITIES: |
||||||||
| Acquisitions of businesses, net of cash and cash equivalents acquired |
(290.1 | ) | (53.2 | ) | ||||
| Purchases of property, plant, and equipment and badges |
(17.3 | ) | (11.8 | ) | ||||
|
|
|
|
|
|||||
| Net cash used in investing activities |
(307.4 | ) | (65.0 | ) | ||||
|
|
|
|
|
|||||
| FINANCING ACTIVITIES: |
||||||||
| Borrowings from notes payable to third-parties, net of discount and issuance costs |
218.8 | 97.5 | ||||||
| Principal repayments |
(12.4 | ) | (11.4 | ) | ||||
| Borrowings from notes payablerelated parties |
70.0 | | ||||||
| Borrowing on revolving term loan |
| 34.9 | ||||||
| Payment on revolving term loan |
(35.0 | ) | | |||||
| Payment of contingent consideration |
| (2.0 | ) | |||||
| Distributions to noncontrolling interests |
| (0.4 | ) | |||||
| Other Financing |
0.2 | | ||||||
|
|
|
|
|
|||||
| Net cash provided by financing activities |
241.6 | 118.6 | ||||||
| Effect of exchange rate changes on cash, cash equivalents, and restricted cash |
2.8 | (0.7 | ) | |||||
|
|
|
|
|
|||||
| Net increase (decrease) in cash, cash equivalents, and restricted cash |
(32.3 | ) | 70.1 | |||||
| Cash, cash equivalents, and restricted cash at beginning of year |
120.0 | 37.6 | ||||||
|
|
|
|
|
|||||
| Cash, cash equivalents, and restricted cash at end of year |
$ | 87.7 | $ | 107.7 | ||||
|
|
|
|
|
|||||
The accompanying notes are an integral part of these unaudited consolidated financial statements.
F-92
Table of Contents
Mirion Technologies (TopCo), Ltd.
Notes to Unaudited Interim Condensed Consolidated Financial Statements
1. Nature of Business and Summary of Significant Accounting Policies
Nature of Business
Mirion Technologies (TopCo), Ltd. (Mirion TopCo or the Company or us) is a global provider of radiation detection, measurement, analysis, and monitoring products and services to the nuclear, defense, and medical end markets. We provide products and services through our two operating and reportable segments; (i) Medical, and (ii) Industrial. The medical segment radiation oncology quality assurance, delivering patient safety solutions for diagnostic imaging and radiation therapy centers around the world, dosimetry solutions for monitoring the total amount of radiation medical staff members are exposed to over time, radiation therapy quality assurance solutions for calibrating and verifying imaging and treatment accuracy, and radionuclide therapy products for nuclear medicine applications such as shielding, product handling, medical imaging furniture, and rehabilitation products. The industrial segment provides robust, field ready personal radiation detection and identification equipment for defense applications and radiation detection and analysis tools for power plants, labs, and research applications. Nuclear power plant product offerings are used for the full nuclear power plant lifecycle including core detectors and essential measurement devices for new build, maintenance, decontamination and decommission equipment for monitoring and control during fuel dismantling and remote environmental monitoring.
The Company is headquartered in Atlanta, Georgia and has operations in the United States, Canada, the United Kingdom, France, Germany, Finland, China, Belgium, Netherlands, Estonia, and Japan.
Basis of Presentation and Principles of Consolidation
The unaudited interim condensed consolidated financial statements were prepared in conformity with accounting principles generally accepted in the United States of America (U.S. GAAP) and the rules and regulations of the U.S. Securities and Exchange Commission (the SEC) for interim financial information; therefore, as permitted under these rules, certain footnotes and other financial information included in audited financial statements were condensed or omitted. The interim financial statements presented include all adjustments, which are of a normal and recurring nature, necessary for a fair presentation of the results for such periods. Results for these interim periods are not necessarily indicative of full year results.
The unaudited interim condensed consolidated financial statements should be read in conjunction with the audited financial statements of Mirion Technologies as of and for the year ended June 30, 2020 and the notes related thereto. The unaudited interim condensed consolidated financial statements include the accounts of the Company and its wholly owned subsidiaries. All intercompany balances and transactions have been eliminated in consolidation.
The Company experiences seasonality with respect to the fiscal year ends of certain customers. Generally, the Companys highest revenues have occurred in the fourth quarter of each year.
Related-Party Transactions
Charterhouse Capital Partners LLP (CCP)
The Company has entered into agreements with its primary investor, CCP, which obligate the Company to pay quarterly management fees of $0.1 million in total per year. In return, CCP provides various investment banking services relating to financing arrangements, mergers and acquisitions and other services. There were no material
F-93
Table of Contents
Notes to Unaudited Interim Condensed Consolidated Financial Statements
fees or expense reimbursements made to CCP during the nine-month periods ended March 31, 2021 and March 31, 2020.
See Note 8 for notes payable to related parties.
Restructuring Costs
The Company periodically initiates restructuring activities in connection with certain acquisitions. The costs associated with restructuring actions are recorded in accordance with ASC 420, Exit or Disposal Cost Obligations, and include property, plant and equipment costs, severance and employee costs, professional fees, and other costs. Liabilities for costs associated with an exit or disposal activity are recognized in the period in which the liability is incurred. Restructuring liabilities are included in accrued expenses and other current liabilities in the Companys Consolidated Balance Sheet. Charges associated with restructuring actions are recorded in Cost of Revenue and Selling, General, and Administrative Expenses in the Companys Consolidated Statement of Operations and are accounted for separate from acquisition costs.
Recent Accounting Pronouncements
Accounting Guidance Issued But Not Yet Adopted
In August 2018, the FASB issued ASU 2018-15, Intangibles Goodwill and Other Internal Use Software (Subtopic 350-40): Customers Accounting for Implementation Costs Incurred in a Cloud Computing Arrangement that is a Service Contract, which amend the accounting for implementation, setup and other upfront costs for entities that are a customer in a hosting arrangement that is a service contract. ASU 2018-15 was effective for public business entities for fiscal years beginning after December 15, 2019, and interim periods within those fiscal years. For all other entities, including emerging growth companies, the amendments are effective for fiscal years beginning after December 15, 2020, and interim periods within fiscal years beginning after December 15, 2021. Early adoption is permitted, including adoption in any interim period for which financial statements have not been issued. Reporting entities can choose to adopt the new guidance prospectively to eligible costs incurred on or after the date the guidance is first applied or retrospectively. Application of the amendments can be applied either retrospectively or prospectively to all implementation costs incurred after the date of adoption. The Company expects to implement the provisions of ASU 2018-15 for annual periods beginning July 1, 2021. The Company is currently evaluating the impact ASU 2018-15 will have on its consolidated financial statements.
In June 2016, the FASB issued ASU 2016-13, Financial Instruments Credit Losses (Topic 326): Measurement of Credit Losses on Financial Instruments, which replaced the incurred loss impairment methodology in current GAAP with a methodology that reflects expected credit losses and requires consideration of a broader range of reasonable and supportable information to inform credit loss estimates. For trade and other receivables, loans, and other financial instruments, we will be required to use a forward-looking expected loss model rather than the incurred loss model for recognizing credit losses which reflects losses that are probable. Credit losses relating to available-for-sale debt securities will also be recorded through an allowance for credit losses rather than as a reduction in the amortized cost basis of the securities. The effective date of Update 2016-13 varies depending on the type of entity. Public business entities that meet the definition of a Securities and Exchange Commission (SEC) filer, excluding eligible smaller reporting companies (SRCs) as defined by the SEC, should adopt the amendments in Update 2016-13 during 2020. All other entities should adopt the amendments in Update 2016-13 during 2023. Early adoption will continue to be permitted. Application of the amendments is through a cumulative-effect adjustment to retained earnings as of the effective date. The company expects to implement provisions of ASU 2016-13 for annual periods beginning July 1, 2023. We are currently evaluating the impact of this standard on our consolidated financial statements.
F-94
Table of Contents
Notes to Unaudited Interim Condensed Consolidated Financial Statements
In February 2016, the FASB issued ASU 2016-02, Leases (Topic 842), which is a new accounting and reporting guidance for leasing arrangements. The new standard requires organizations that lease assets to recognize assets and liabilities on the balance sheet related to the rights and obligations created by those leases with terms of more than 12 months, regardless of whether they are classified as financing or operating leases. Consistent with current guidance, the recognition, measurement, and presentation of expenses and cash flows arising from a lease primarily will depend on its classification as a finance or operating lease. The guidance also requires new disclosures to help financial statement users better understand the amount, timing, and uncertainty of cash flows arising from leases. For public companies, the new standard was effective for annual and interim periods in fiscal years beginning after December 15, 2018. On June 3, 2020, the FASB issued guidance (ASU 2020-05) that deferred the effective date of the leases standards (ASC 842) for non-public business entities that had not yet issued financial statements adopting the standards. For these entities, including emerging growth companies, ASU 2016-02 is effective for fiscal years beginning after December 15, 2021, and interim periods within fiscal years beginning after December 15, 2022. Early application continues to be allowed. The Company expects to implement the provisions of ASU 2016-02 for annual periods beginning July 1, 2021. The Company is currently evaluating the impact of the new pronouncement on its consolidated financial statements and disclosures, however upon adoption, the Company will recognize a lease liability measured as the present value of remaining lease payments as well as a right of use asset measured as the amount of the lease liability adjusted for certain assets and liabilities such as deferred lease obligations related to operating leases previously recognized in the balance sheet. The Company will elect the modified retrospective transition method and under such method no recast of prior-period financial statements will be presented. We will elect the transition package of practical expedients, which among other things, allows for the carryforward of historical lease classifications, and we will make an accounting policy election to not apply the recognition requirements to leases with a term of less than twelve months. We will also elect to account for lease and nonlease components as a combined single lease component. The Companys existing leases consist primarily of offices, equipment, and vehicles and the related future commitments under lease obligations are summarized in Note 9.
In March 2020, the FASB issued ASU 2020-04 Reference Rate Reform (Topic 848): Facilitation of the Effects of Reference Rate Reform on Financial Reporting. ASU 2020-04 provides temporary optional expedients and exceptions for applying GAAP guidance on contract modifications and hedge accounting to ease the financial reporting burdens of the expected market transition from the LIBOR and other interbank offered rates to alternative reference rates, such as the Secured Overnight Financing Rate. For all entities, ASU 2020-04 can be adopted after its issuance date through December 31, 2022. The Company is currently evaluating the impact of this ASU.
Recently Adopted Accounting Guidance
In August 2018, the FASB issued ASU 2018-13, Fair Value Measurement (Topic 820): Disclosure Framework Changes to the Disclosure Requirements for Fair Value Measurement (ASU 2018-13). ASU 2018-13 eliminates, adds and modifies certain disclosure requirements for fair value measurements as part of the FASBs disclosure framework project. For all entities, ASU 2018-13 is effective for annual and interim reporting periods beginning after December 15, 2019. Certain amendments must be applied prospectively while others are to be applied on a retrospective basis to all periods presented. The Company adopted this guidance as of July 1, 2020 and it did not have a material impact on our consolidated financial statements or related disclosures.
2. Acquisitions
The Company continually evaluates potential acquisitions that strategically fit with the Companys existing portfolio and as a result, the Company completed three acquisitions during the nine months ended March 31, 2021.
F-95
Table of Contents
Notes to Unaudited Interim Condensed Consolidated Financial Statements
All acquisitions are accounted for under the acquisition method of accounting, and the related assets acquired, and liabilities assumed are recorded at fair value. The Company makes an initial allocation of the purchase price at the date of acquisition based upon its understanding of the fair value of the acquired assets and assumed liabilities. The Company obtains the information used for the purchase price allocation during due diligence and through other sources. In the months after closing, as the Company obtains additional information about the acquired assets and liabilities, including through tangible and intangible asset appraisals, and learns more about the newly acquired business, it is able to refine the estimates of fair value and more accurately allocate the purchase price. The fair values of acquired intangibles are determined based on estimates and assumptions that are deemed reasonable by the Company. Significant assumptions include the discount rates and certain assumptions that form the basis of the forecasted results of the acquired business including revenue, earnings before interest, taxes, depreciation and amortization (EBITDA), and growth rates. These assumptions are forward looking and could be affected by future economic and market conditions. The Company engages third-party valuation specialists who review the Companys critical assumptions and calculations of the fair value of acquired intangible assets in connection with material acquisitions. Only facts and circumstances that existed as of the acquisition date are considered for subsequent adjustment. The Company will make appropriate adjustments to the purchase price allocation prior to completion of the one-year measurement period, as required.
The purchases of these acquired businesses resulted in the recognition of goodwill in the Companys consolidated financial statements, which is calculated as the excess of the consideration transferred over the net assets recognized and represents the future economic benefits arising from the other assets acquired that could not be individually identified and separately recognized. The goodwill is not amortized and is not deductible for income tax purposes. This goodwill recorded includes the following:
| | The expected synergies and other benefits that we believe will result from combining the operations of the acquired business with the operations of Mirion; |
| | Any intangible assets that did not qualify for separate recognition, as well as future, yet unidentified projects and products; |
| | The value of the existing business as an assembled collection of net assets versus if the Company had acquired all of the net assets separately. |
The following briefly describes the Companys acquisition activity for the nine-month period ended March 31, 2021.
| Nine Months March 31, |
Company Name |
Description of the Business |
Description of the Acquisition |
|||
| 2021 |
Sun Nuclear | Sun Nuclear Corporation (SNC) is a provider in radiation oncology quality assurance, delivering patient safety solutions for diagnostic imaging and radiation therapy centers around the world. | On December 18, 2020, the Company acquired 100% of the equity interest for approximately $258.1 million of purchase consideration, net of cash acquired. | |||
| 2021 |
Dosimetrics | Dosimetrics is a provider in the development and production of OSL personal radiation dosimeters and dosimetry solutions, including readers, erasers, software, accessories, and automation systems. | On December 1, 2020, the Company acquired 100% of the equity interest for approximately $3.0 million of purchase consideration, net of cash acquired. |
F-96
Table of Contents
Notes to Unaudited Interim Condensed Consolidated Financial Statements
| Nine Months March 31, |
Company Name |
Description of the Business |
Description of the Acquisition |
|||
| 2021 |
Biodex | Biodex is a manufacturer and distributor of medical devices and related replacement parts for physical and nuclear medicine, as well as medical imaging applications located in the United States. | On September 1, 2020, the Company acquired 100% of the equity interest for approximately $26.9 million of purchase consideration, net of cash acquired. |
The Company is still finalizing the fair value estimates, but the following table presents the preliminary estimates of the fair value of the assets acquired and liabilities assumed for the material Biodex and SNC acquisitions during the nine-month period ended March 31, 2021 (in millions):
| Biodex | SNC | |||||||
| Date of acquisition | September 1, 2020 |
December 18, 2020 |
||||||
| Segment | Medical | Medical | ||||||
| Goodwill |
$ | 10.4 | $ | 130.1 | ||||
| Customer relationships (1) |
2.3 | 59.5 | ||||||
| Tradenames (2) |
1.4 | 12.0 | ||||||
| Non-Compete Agreements (3) |
0.3 | 7.5 | ||||||
| Developed Technology (4) |
2.6 | 46.5 | ||||||
|
|
|
|
|
|||||
| Amortizable intangible assets |
$ | 6.6 | $ | 125.5 | ||||
| Cash |
4.1 | 18.8 | ||||||
| Accounts receivable |
4.0 | 24.0 | ||||||
| Inventory |
6.4 | 13.9 | ||||||
| Property, Plant and Equipment |
1.0 | 5.9 | ||||||
| Other current and non-current assets |
1.5 | 5.7 | ||||||
| Current liabilities |
(2.8 | ) | (9.3 | ) | ||||
| Deferred revenue |
(0.2 | ) | (6.5 | ) | ||||
| Other long-term liabilities |
| (31.2 | ) | |||||
|
|
|
|
|
|||||
| Net tangible assets acquired |
$ | 14.0 | $ | 21.3 | ||||
|
|
|
|
|
|||||
| Purchase consideration (5) |
31.0 | 276.9 | ||||||
| Less: cash acquired |
(4.1 | ) | (18.8 | ) | ||||
|
|
|
|
|
|||||
| Purchase consideration, net of cash acquired |
$ | 26.9 | $ | 258.1 | ||||
| Acquiree revenue post acquisition through the period ended March 31, 2021 |
$ | 21.4 | $ | 28.9 | ||||
| Acquiree income (loss) from operations post acquisition through the period ended March 31, 2021 |
$ | (0.2 | ) | $ | (6.6 | ) | ||
| (1) | The useful life for customer relationships ranges from 10 - 11 years. |
| (2) | The useful life for tradenames ranges from 7 years. |
| (3) | The useful life for non-compete agreements is 2 - 3 years. |
| (4) | The useful life for developed technology ranges from 7 - 10 years. |
| (5) | Biodex purchase consideration consisted of cash. SNC purchase consideration consisted of $261.9 million cash and $15 million of deferred consideration paid in February 2021. |
In connection with the acquisition of Sun Nuclear, the Company incurred approximately $1.2 million of transaction expenses for the nine-months ended March 31, 2021.
F-97
Table of Contents
Notes to Unaudited Interim Condensed Consolidated Financial Statements
Pro Forma Financial Information
The following unaudited pro forma financial information presents the Companys results of operations for the nine months ended March 31, 2021 and 2020 to illustrate the estimated effects of the acquisition of Biodex and SNC as if it had occurred on July 1, 2019. The pro forma financial information is presented for comparative purposes only and is not necessarily indicative of the Companys operating results that may have actually occurred had the acquisition of Biodex and SNC been completed on July 1, 2019. The unaudited pro forma financial information does not reflect the expected realization of any anticipated cost savings, operating efficiencies, or other synergies that may have been associated with the acquisition.
| (amounts in millions) | Nine-months ended March 31, |
|||||||
| 2021 | 2020 | |||||||
| Total revenues |
$ | 495.1 | $ | 432.1 | ||||
| Net loss |
(133.8 | ) | (125.3 | ) | ||||
| Net loss attributable to Mirion Technologies (TopCo), Ltd stockholders |
(133.7 | ) | (125.3 | ) | ||||
The unaudited pro forma financial information reflects pro forma adjustments to present the combined pro forma results of the operations as if the acquisition had occurred on July 1, 2019 to give effect to certain events the Company believes to be directly attributable to the acquisitions. These pro forma adjustments primarily include:
| | A net increase in cost of revenues, depreciation and amortization expense that would have been recognized due to acquired inventory, property, plant and equipment and intangible assets; |
| | An increase to interest expense to reflect the additional borrowings of the Company in conjunction with the acquisition; |
| | A reduction in expenses for the nine-months ended March 31, 2021 and a corresponding increase in the nine-months ended March 30, 2020 for acquisition-related transaction costs directly attributable to the acquisition; |
| | A reduction in operating revenues due to the elimination of deferred revenues assigned no value at the acquisition date; |
| | An increase in income tax expense using the U.S. statutory rate of 25% to reflect a change in tax status had the SNC and Biodex results of operations been included in the Companys consolidated tax return. |
| | The related income tax effects of the adjustments noted above. |
For the nine-months ended March 31, 2020, pro forma adjustments directly attributable to the acquisitions include (i) the purchase accounting effect of inventories acquired of $5.2 million, (ii) transaction costs of $4.8 million and (iii) the reduction in revenues of $11.1 million due to the elimination of deferred revenues assigned no value at the acquisition date.
F-98
Table of Contents
Notes to Unaudited Interim Condensed Consolidated Financial Statements
3. Contracts in Progress
Costs and billings on uncompleted construction-type contracts consist of the following (in millions):
| March 31, 2021 |
June 30, 2020 |
|||||||
| Costs incurred on contracts (from inception to completion) |
$ | 173.1 | $ | 152.5 | ||||
| Estimated earnings |
123.2 | 108.9 | ||||||
|
|
|
|
|
|||||
| Contracts in progress |
296.3 | 261.4 | ||||||
| Less: billings to date |
(239.3 | ) | (209.8 | ) | ||||
| Less: write-offs |
(2.7 | ) | | |||||
|
|
|
|
|
|||||
| $ | 54.3 | $ | 51.6 | |||||
|
|
|
|
|
|||||
|
Included in the accompanying balance sheets under the following captions:
|
||||||||
| March 31, 2021 |
June 30, 2020 |
|||||||
| Costs and estimated earnings in excess of billings on uncompleted contractscurrent |
$ | 59.7 | $ | 59.6 | ||||
| Costs and estimated earnings in excess of billings on uncompleted contractsnoncurrent (1) |
5.2 | | ||||||
| Billings in excess of costs and estimated earnings on uncompleted contractscurrent (2) |
(9.2 | ) | (8.0 | ) | ||||
| Billings in excess of costs and estimated earnings on uncompleted contractsnoncurrent (3) |
(1.4 | ) | | |||||
|
|
|
|
|
|||||
| $ | 54.3 | $ | 51.6 | |||||
|
|
|
|
|
|||||
| (1) | Included in other assets within the consolidated balance sheets. |
| (2) | Included in deferred contract revenue current within the consolidated balance sheets. |
| (3) | Included in other liabilities within the consolidated balance sheets. |
Substantially all of the contract liabilities balance as of June 30, 2020 was subsequently recognized as revenue during the nine-month period ended March 31, 2021.
Remaining Performance Obligations
The remaining performance obligations for all open contract as of March 31, 2021 include assembly, delivery, installation, and trainings. The aggregate amount of the transaction price allocated to the remaining performance obligations for all open customer contracts as of March 31, 2021 was approximately $766.4 million. The Company expects to recognize approximately 22% of the remaining performance obligations as of March 31, 2021 as revenue during the remaining 3 months for fiscal year 2021 and 29% and 15% for the fiscal years 2022 and 2023, respectively.
4. Inventories
The components of inventories consist of the following (in millions):
| March 31, 2021 |
June 30, 2020 |
|||||||
| Raw materials |
$ | 57.4 | $ | 40.6 | ||||
| Work in progress |
18.5 | 16.1 | ||||||
| Finished goods |
42.2 | 33.5 | ||||||
|
|
|
|
|
|||||
| $ | 118.1 | $ | 90.2 | |||||
|
|
|
|
|
|||||
F-99
Table of Contents
Notes to Unaudited Interim Condensed Consolidated Financial Statements
5. Property, Plant, and Equipment, Net
Property and equipment, net consist of the following (in millions):
| Depreciable Lives |
March 31, 2021 |
June 30, 2020 |
||||||||||
| Land, buildings, and leasehold improvements |
3-39 years | $ | 46.2 | $ | 43.9 | |||||||
| Machinery and equipment |
5-15 years | 45.8 | 38.9 | |||||||||
| Badges |
3-5 years | 36.5 | 29.4 | |||||||||
| Furniture, fixtures, computer equipment and other |
3-10 years | 31.7 | 27.6 | |||||||||
| Construction in progress |
| 12.7 | 7.3 | |||||||||
|
|
|
|
|
|||||||||
| 172.9 | 147.1 | |||||||||||
| Less: accumulated depreciation and amortization |
(83.9 | ) | (71.9 | ) | ||||||||
|
|
|
|
|
|||||||||
| $ | 89.0 | $ | 75.2 | |||||||||
|
|
|
|
|
|||||||||
Total depreciation expense included in cost of revenue was $9.1 million and $9.5 million for the nine-months ended March 31, 2021 and 2020, respectively. Total depreciation expense included in operating expenses was $4.8 million and $3.9 million for the nine-months ended March 31, 2021 and 2020, respectively. Construction in progress includes capitalized internal use software costs totaling $2.0 million and $1.2 million as of March 31, 2021 and June 30, 2020, respectively.
6. Accrued Expenses and Other Current Liabilities, and Deferred Income Taxes and Other Long-Term Liabilities
Accrued expenses and other current liabilities consist of the following (in millions):
|
March 31, 2021 |
June 30, 2020 |
|||||||
| Compensation and related benefit costs |
$ | 35.7 | $ | 30.3 | ||||
| Customer deposits |
8.7 | 3.1 | ||||||
| Accrued commissions |
0.9 | 3.7 | ||||||
| Accrued warranty costs |
6.4 | 5.5 | ||||||
| Non-income taxes payable |
6.5 | 4.9 | ||||||
| Pension and other post-retirement obligations |
0.4 | 0.3 | ||||||
| Income taxes payable |
21.1 | 9.2 | ||||||
| Other accrued expenses |
12.0 | 7.1 | ||||||
|
|
|
|
|
|||||
| $ | 91.7 | $ | 64.1 | |||||
|
|
|
|
|
|||||
Deferred income taxes and other long-term liabilities consist of the following (in millions):
| March 31, 2021 |
June 30, 2020 |
|||||||
| Deferred income taxes |
$ | 38.5 | $ | 33.1 | ||||
| Pension and other post-retirement obligations, non-current |
13.0 | 12.4 | ||||||
| Other long-term liabilities |
19.9 | 18.0 | ||||||
| Billings in excess of costs on uncompleted contractsnoncurrent |
1.4 | | ||||||
|
|
|
|
|
|||||
| $ | 72.8 | $ | 63.5 | |||||
|
|
|
|
|
|||||
F-100
Table of Contents
Notes to Unaudited Interim Condensed Consolidated Financial Statements
7. Goodwill and Intangible Assets
Goodwill
Goodwill is calculated as the excess of consideration transferred over the net assets recognized for acquired businesses and represents future economic benefits arising from the other assets acquired that could not be individually identified and separately recognized. The Company assesses goodwill for impairment at the reporting unit level annually during the fiscal fourth quarter and upon the occurrence of a triggering event or change in circumstance that would more likely than not reduce the fair value of a reporting unit below its carrying amount.
No goodwill impairment was recognized for the nine-months ended March 31, 2021 and 2020.
The following table shows changes in the carrying amount of goodwill by reportable segment for the nine-month period ended March 31, 2021 (in millions):
| Medical | Industrial | Consolidated | ||||||||||
| BalanceJune 30, 2020 |
$ | 106.8 | $ | 415.8 | $ | 522.6 | ||||||
| Acquisition of Sun Nuclear |
130.1 | | 130.1 | |||||||||
| Acquisition of Biodex |
10.4 | | 10.4 | |||||||||
| Acquisition of Dosimetrics |
1.6 | | 1.6 | |||||||||
| Translation adjustment |
0.1 | 12.7 | 12.8 | |||||||||
|
|
|
|
|
|
|
|||||||
| Balance March 31, 2021 |
$ | 249.0 | $ | 428.5 | $ | 677.5 | ||||||
|
|
|
|
|
|
|
|||||||
We do not expect goodwill to be deductible for income tax purposes.
Intangible Assets
Intangible assets consist of our developed technology, customer relationships, backlog, trade names, and non-compete agreements at the time of acquisition through business combinations. The customer relationships definite lived intangible assets are amortized using the double declining balance method with estimated useful lives ranging from 6 to 17 years, while all other definite lived intangible assets are amortized on a straight-line basis over their estimated useful lives, ranging from 1 to 7 years.
Many of our intangible assets are not deductible for income tax purposes. A summary of intangible assets and related accumulated amortization (in millions) is below:
| March 31, 2021 | ||||||||||||||||
| Original Average Life in Years |
Gross Carrying Amount |
Accumulated Amortization |
Net Book Value |
|||||||||||||
| Customer relationships |
6-17 | $ | 418.8 | $ | (196.3 | ) | $ | 222.5 | ||||||||
| Developed technology |
3-16 | 183.7 | (98.1 | ) | 85.6 | |||||||||||
| Trade names |
5-9 | 47.1 | (27.3 | ) | 19.8 | |||||||||||
| Backlog and other |
1-9 | 40.5 | (25.1 | ) | 15.4 | |||||||||||
|
|
|
|
|
|
|
|||||||||||
| Total |
$ | 690.1 | $ | (346.8 | ) | $ | 343.3 | |||||||||
|
|
|
|
|
|
|
|||||||||||
F-101
Table of Contents
Notes to Unaudited Interim Condensed Consolidated Financial Statements
| June 30, 2020 | ||||||||||||||||
| Original Average Life in Years |
Gross Carrying Amount |
Accumulated Amortization |
Net Book Value |
|||||||||||||
| Customer relationships |
6-17 | $ | 358.5 | $ | (170.5 | ) | $ | 188.0 | ||||||||
| Developed technology |
3-16 | 130.0 | (81.0 | ) | 49.0 | |||||||||||
| Trade names |
5-9 | 32.9 | (22.4 | ) | 10.5 | |||||||||||
| Backlog and other |
1-9 | 22.8 | (22.0 | ) | 0.8 | |||||||||||
|
|
|
|
|
|
|
|||||||||||
| Total |
$ | 544.2 | $ | (295.9 | ) | $ | 248.3 | |||||||||
|
|
|
|
|
|
|
|||||||||||
Aggregate amortization expense for intangible assets included in cost of revenue was $16.6 million and $13.6 million for the nine-months ended March 31, 2021 and 2020 respectively. Aggregate amortization expense for intangible assets included in operating expenses was $27.7 million and $24.6 million for the nine-months ended March 31, 2021 and 2020 respectively.
As of March 31, 2021, future annual amortization expense at current exchange rates is as follows (in millions):
| Fiscal year ending June 30: |
||||
| Remainder of 2021 |
$ | 18.5 | ||
| 2022 |
63.6 | |||
| 2023 |
47.4 | |||
| 2024 |
39.6 | |||
| 2025 |
35.5 | |||
| 2026 and thereafter |
138.7 | |||
|
|
|
|||
| Total |
$ | 343.3 | ||
|
|
|
8. Borrowings
Third-party notes payable consist of the following (in millions):
| March 31, 2021 |
June 30, 2020 |
|||||||
| 2019 Credit Facilityfirst lien term loan |
$ | 907.0 | $ | 682.1 | ||||
| NRG Loan |
| 5.8 | ||||||
| Canadian Financial Institution |
1.2 | 1.2 | ||||||
| JLG Note Payable |
0.3 | 0.2 | ||||||
| Draw on revolving line of credit |
| 35.0 | ||||||
|
|
|
|
|
|||||
| Total third-party borrowings |
908.5 | 724.3 | ||||||
| Less: notes payable to third-parties, current |
(5.5 | ) | (41.1 | ) | ||||
| Less: deferred financing costs |
(17.5 | ) | (13.4 | ) | ||||
|
|
|
|
|
|||||
| Notes payable to third-parties, non-current |
$ | 885.5 | $ | 669.8 | ||||
|
|
|
|
|
|||||
As of March 31, 2021 and June 30, 2020, the fair market value of the Companys 2019 Credit Facilityfirst lien term loan was $906.9 million and $662.5 million, respectively. The fair market value for the credit facility were estimated using primarily level 2 inputs, including borrowing rates available to the Company at the respective period ends. The fair market value for the Companys remaining third-party debt approximates the respective carrying amounts as of March 31, 2021 and June 30, 2020.
2019 Credit Facility
In March 2019, Mirion Technologies (HoldingRep), Ltd., a wholly owned subsidiary of the Company, and subsidiaries entered into a credit agreement with an intermediary and a syndicate of institutional lenders (2019
F-102
Table of Contents
Notes to Unaudited Interim Condensed Consolidated Financial Statements
Credit Facility). The 2019 Credit Facility provided for financing of a $450.0 million senior secured term loan facility and a 125 million term loan facility, as well as a $90.0 million revolving line of credit. The 2019 Credit Facility was amended to provide an additional $225.0 million, $34.0 million and $66.0 million in gross proceeds from the USD term loan in December 2020, July 2019 and December 2019, respectively.
USD term loanThe term loan has a seven-year term (expiring March 2026), bears interest at the greater of Adjusted London Interbank Offered Rate (LIBOR) or 0%, plus 4.00% and has quarterly principal repayments of 0.25% of the original principal balance. The interest rate was 4.27% and 5.07% at March 31, 2021 and June 30, 2020, respectively. The Company repaid $5.3 million and $4.1 million for the nine-months ended March 31, 2021 and 2020, respectively.
Euro term loanThe Euro portion of the term loan has a seven-year term (expiring March 2026), bears interest at the greater of European union interbank market (Euribor) or 0%, plus 4.25% and has quarterly principal repayments of 0.25% of the original principal balance. As of March 31, 2021 and June 30, 2020, the interest rate was 4.25%. The Company repaid $1.1 million and $0.6 million during the nine-months ended March 31, 2021 and 2020, respectively.
Revolving Line of CreditThe revolving line of credit arrangement has a five-year term and bears interest at the greater of LIBOR or 0%, plus 4.00%. The agreement requires the payment of a commitment fee of 0.50% per annum for unused commitments. The revolving line of credit matures in March 2024, at which time all outstanding revolving facility loans and accrued and unpaid interest are due. Any outstanding letters of credit reduce the availability of the revolving line of credit. The outstanding balance under the arrangement was $35.0 million as of June 30, 2020, with no outstanding balance as of March 31, 2021. Additionally, the Company has standby letters of credit issued under its Credit Facility that reduce the availability under the revolver of $8.9 million and $9.0 million at March 31, 2021 and June 30, 2020, respectively, the amount available on the revolver was approximately $81.1 million and $46.0 million, for the same periods, respectively.
Deferred Financing Costs
In connection with the issuance of the 2019 Credit Facility, we incurred debt issuance costs of $16.3 million on date of issuance, with an additional $6.2 million and $1.2 million of costs for incremental proceeds in fiscal year 2021 and 2020, respectively. In accordance with accounting for debt issuance costs, we recognize, and present deferred finance costs associated with non-revolving debt and financing obligations as a reduction from the face amount of related indebtedness in our consolidated balance sheet.
In connection with the issuance of the 2019 Credit Facility revolving line of credit, we incurred debt issuance costs of $0.9 million. We recognize and present debt issuance costs associated with revolving debt arrangements as an asset and include the deferred finance costs within other assets on our consolidated balance sheet. We amortize all debt issuance costs over the life of the related indebtedness.
During the nine-months ended March 31, 2021 and 2020, we incurred approximately $2.3 million and $2.1 million, respectively, of amortization expense of the deferred finance costs.
Both Credit Facilities are secured by a first priority lien on substantially all of Mirion HoldingRep and subsidiaries assets in the United States, certain assets of guarantor subsidiaries in Germany, United Kingdom, Canada, France, Belgium and Luxembourg and two-thirds of assets in non-guarantors and other countries. Loan fees recorded as debt discounts are amortized using the effective interest method. The Credit Facility contains customary restrictive covenants, as well as financial covenants that require Mirion HoldingRep and subsidiaries to maintain a certain total level of debt-to-income ratio and interest coverage ratio, each as defined in the Credit Facility, as well as non-financial affirmative and negative covenants. The negative covenants, subject to certain exceptions, generally limit the ability of Mirion HoldingRep and subsidiaries to incur additional debt, create liens, make fundamental changes, make certain investments, pay dividends, purchase or retire equity interests, or
F-103
Table of Contents
Notes to Unaudited Interim Condensed Consolidated Financial Statements
prepay or retire certain debt. Mirion HoldingRep and subsidiaries are in compliance with all debt covenants at both March 31, 2021 and June 30, 2020.
NRG LoanIn conjunction with the acquisition of NRG, the Company entered into a loan agreement for 7.2 million ($7.4 million) at the date of the acquisition. This agreement expires in December 2023. The loan bears interest which is Euribor of 3 months, plus 2.0%, and mandatory costs if any. This agreement was paid off in the period ended March 31, 2021.
Canadian Financial InstitutionIn May 2019, the Company entered into a credit agreement for C$1.7M ($1.3 million) with a Canadian financial institution that matures in April 2039. The note bears annual interest at 4.15%. The credit agreement is secured by the facility acquired using the funds obtained.
JLG Note PayableIn May 2019, the Company entered into a note payable for $0.2 million with an individual that has left the organization, which is due upon a change in control of Mirion Technologies (Global), Ltd, a wholly owned subsidiary of the Company. The note bears annual interest at 6.00%.
Performance Bonds and Other Credit Facilities
The Company has entered into various line of credit arrangements with local banks in France and Germany. These arrangements provide for the issuance of documentary and standby letters of credit of up to 54.3 million ($63.6 million) and 47.3 million ($53.1 million) as of March 31, 2021 and June 30, 2020, respectively, subject to certain local restrictions. As of March 31, 2021 and June 30, 2020, 23.9 million ($28.1 million) and 21.2 million ($23.7 million), respectively, of the lines had been utilized to guarantee documentary and standby letters of credit, with interest rates ranging from 0.5% to 2.00%. In addition, the Company posts performance bonds with irrevocable letters of credit to support certain contractual obligations to customers for equipment delivery. These letters of credit are supported by restricted cash accounts, which totaled $1.0 million and $1.7 million as of March 31, 2021 and June 30, 2020.
At March 31, 2021 contractual principal payments of total third-party borrowings are as follows (in millions):
| For the year ending June 30: |
||||
| Remainder of 2021 |
$ | 2.3 | ||
| 2022 |
10.3 | |||
| 2023 |
9.0 | |||
| 2024 |
8.9 | |||
| 2025 |
8.8 | |||
| Thereafter |
869.2 | |||
|
|
|
|||
| Gross Payments |
908.5 | |||
| Unamortized debt issuance costs |
(17.5 | ) | ||
|
|
|
|||
| Total third-party borrowings, net of debt issuance costs |
$ | 891.0 | ||
|
|
|
Notes payable to related parties consists of the following (in millions):
| March 31, 2021 |
June 30, 2020 |
|||||||
| Shareholder Notes |
$ | 1,166.7 | $ | 983.7 | ||||
| Management Notes |
3.6 | 3.4 | ||||||
|
|
|
|
|
|||||
| Notes payable to related parties |
$ | 1,170.3 | $ | 987.1 | ||||
|
|
|
|
|
|||||
The estimated fair value of the Companys related party debt was approximately $1,170.3 million and $987.1 million as of March 31, 2021 and June 30, 2020, respectively. The fair value of this instrument approximates book value due to the reasonable possibility of redemption prior to the term date.
F-104
Table of Contents
Notes to Unaudited Interim Condensed Consolidated Financial Statements
Shareholder and Management Notes Mirion Technologies (HoldingSub1), Ltd., is authorized to issue $900 million (plus accrued paid in-kind interest) of notes to shareholders (Shareholder Notes) and up to $5.0 million (plus paid in-kind cash and interest) of notes to certain members of management (Management Notes). The notes rank pari passu between each other and other unsecured obligations of the Company. The notes can be prepaid without penalty at the Companys option and are subordinate in right of payment to any indebtedness of the Company to banks or to other financial institutions (either currently existing or to occur in the future). Certain of the Shareholder and Management Notes have been admitted to trading and are on the official listing of The International Stock Exchange (TISE).
For the fiscal year ended June 30, 2020, an additional $99.6 million in Shareholder Notes were admitted to trading and are on the official listing of TISE. No new Shareholder Notes were admitted to trading during the nine months ended March 31, 2021, however, subsequent to the period there was an issuance of an additional $70.0 million in Shareholder Notes admitted to trading in April 2021. At March 31, 2021 and June 30, 2020, there were $976.9 million in Shareholder Notes issued and outstanding, respectively, as listed on TISE. Of the amount available for trading, $613.9 million relates to principal balance and $363.0 million relates to accrued interest as of March 31, 2021. There was no trading activity related to Shareholder and Management Notes during the nine months ended March 31, 2021. As of March 31, 2021 and June 30, 2020 there were $3.4 million in Management Notes issued and outstanding, respectively, as listed on TISE.
The notes bear simple annual interest at 11.5%. For the Shareholder Notes, the interest is added to the principal outstanding on December 31 of each year until 2025 and is referred to as Shareholder Funding Bonds on TISE. For the Management Notes, half of the interest is added to the principal outstanding on December 31 of each year until 2025 and is referred to as Management Funding Bonds on TISE, while the remaining half is payable in cash annually. The listing on the Exchange for Shareholder and Management Funding Bonds is an optional election and certain shareholders have elected to opt-out of listing their Shareholder Funding Bonds. All other shareholders and management have elected to list their funding bonds on the Exchange. The notes are due when the Company completes a public offering, a winding-up, a sale, or on March 30, 2026, whichever occurs first. The redemption price is equal to the outstanding principal plus all accrued and unpaid interest then outstanding.
At March 31, 2021 and June 30, 2020, interest of $32.1 million and $56.3 million was accrued on the Shareholder Notes principal outstanding, respectively, and $0.1 million and $0.1 million was accrued on the Management Notes principal outstanding, respectively. As of December 31, 2020 and 2019, accrued interest of $113.6 million and $101.3 million, respectively, was added to the principal of the Shareholder Notes; $0.2 million was added to the principal of the Management Notes.
9. Commitments and Contingencies
Leases
In the normal course of business, the Company enters into contractual arrangements with third parties for non-cancellable operating lease agreements for its offices, equipment and vehicles. Future minimum lease payments for these operating leases as of March 31, 2021, are as follows (in millions):
| For the year ending June 30: |
||||
| Remainder of 2021 |
$ | 5.2 | ||
| 2022 |
10.1 | |||
| 2023 |
8.5 | |||
| 2024 |
7.3 | |||
| 2025 |
6.1 | |||
| 2026 and thereafter |
21.1 | |||
|
|
|
|||
| Total |
$ | 58.3 | ||
|
|
|
F-105
Table of Contents
Notes to Unaudited Interim Condensed Consolidated Financial Statements
Total rent expense for the nine months ended March 31, 2021 and 2020 was $7.0 million and $4.1 million, respectively.
Unconditional Purchase Obligations
The Company has entered into certain long-term unconditional purchase obligations with suppliers. These agreements are non-cancellable and specify terms, including fixed or minimum quantities to be purchased, fixed or variable price provisions, and the approximate timing of payment. As of March 31, 2020, unconditional purchase obligations were $8.1 million and $10.6 million for the fiscal years ending June 30, 2021 and 2022, respectively.
Litigation
The Company is subject to various legal proceedings, claims, litigation, investigations and contingencies arising out of the ordinary course of business. If we believe the likelihood of an adverse legal outcome is probable and the amount is reasonably estimable, we accrue a liability in accordance with accounting guidance for contingencies. We consult with legal counsel on matters related to litigation and seek input both within and outside the Company. We are not currently involved in any litigation that we believe could have a material effect on our financial condition or results of operations
10. Income Taxes
The effective income tax rate for the nine-months ended March 31, 2021 and March 31, 2020 was (5.0%) and 4.8%, respectively. The difference in effective income tax rates and tax expense is primarily due to changes in the mix of earnings and jurisdictions. The Companys effective income tax rates for the periods differ from the U.K. statutory rate of 19% primarily as a result of valuation allowances on certain UK losses.
11. Segment Information
The Company manages its operations through two operating and reportable segments: Medical and Industrial. These segments align the Companys products and service offerings with customer use in industrial and medical markets and are consistent with how the Companys Chief Executive Officer, its Chief Operating Decision Maker (CODM), reviews and evaluates the Companys operations. The CODM allocates resources and evaluates the financial performance of each operating segment. The Companys segments are strategic businesses that are managed separately because each one develops, manufactures and markets distinct products and services. Prior period information herein has been conformed to the current reportable segment structure.
Description of Segments
The medical segment provides radiation oncology quality assurance, delivering patient safety solutions for diagnostic imaging and radiation therapy centers around the world, dosimetry solutions for monitoring the total amount of radiation medical staff members are exposed to over time, radiation therapy quality assurance solutions for calibrating and verifying imaging and treatment accuracy, and radionuclide therapy products for nuclear medicine applications such as shielding, product handling, medical imaging furniture, and rehabilitation products.
The industrial segment provides robust, field ready personal radiation detection and identification equipment for defense applications and radiation detection and analysis tools for power plants, labs, and research applications. Nuclear power plant product offerings are used for the full nuclear power plant lifecycle including core detectors and essential measurement devices for new build, maintenance, decontamination and decommission equipment for monitoring and control during fuel dismantling and remote environmental monitoring.
The following table summarizes select operating results for each reportable segment.
F-106
Table of Contents
Notes to Unaudited Interim Condensed Consolidated Financial Statements
The CODM evaluates operating results and allocates capital resources among segments, in part, based on segment income from operations, which includes revenues of the segment less expenses that are directly related to those revenues, including purchase accounting impacts to revenue and cost of revenues, but before certain charges to cost of revenues and selling, general and administrative expenses predominantly related to corporate costs, shared overhead and other non operational costs related to restructuring activities and costs to achieve operational initiatives, which are included in Corporate and Other in the table below. Interest expense, loss on debt extinguishment, foreign currency loss (gain), net, and other expense (income), net are not allocated to segments.
| For the nine months ended March 31, |
||||||||
| (In millions) | 2021 | 2020 | ||||||
| Revenue |
||||||||
| Medical |
$ | 103.6 | $ | 44.5 | ||||
| Industrial |
328.0 | 292.5 | ||||||
|
|
|
|
|
|||||
| Consolidated revenue |
$ | 431.6 | $ | 337.0 | ||||
| Segment Income from Operations |
||||||||
| Medical |
6.4 | 8.9 | ||||||
| Industrial |
51.6 | 32.7 | ||||||
|
|
|
|
|
|||||
| Total segment Income from Operations |
$ | 58.0 | $ | 41.6 | ||||
| Corporate & Other |
(51.5 | ) | (34.5 | ) | ||||
|
|
|
|
|
|||||
| Consolidated Income from Operations |
$ | 6.5 | $ | 7.1 | ||||
|
|
|
|
|
|||||
The Companys total assets are not presented for each reportable segment as they are not reviewed by, nor otherwise provided to, the chief operating decision maker to make operating decisions or allocate resources.
The following details revenue by geographic region. Revenues from external customers are attributed to geographic regions through sales from site locations.
| Revenues | ||||||||
| For the nine months ended March 31, |
||||||||
| (In millions) | 2021 | 2020 | ||||||
| North America |
||||||||
| Medical |
$ | 91.9 | $ | 42.4 | ||||
| Industrial |
138.3 | 134.6 | ||||||
|
|
|
|
|
|||||
| Total North America |
230.2 | 177.0 | ||||||
| Europe |
||||||||
| Medical |
11.7 | 2.1 | ||||||
| Industrial |
176.5 | 143.4 | ||||||
|
|
|
|
|
|||||
| Total Europe |
188.2 | 145.5 | ||||||
| Asia Pacific |
||||||||
| Medical |
| | ||||||
| Industrial |
13.2 | 14.5 | ||||||
|
|
|
|
|
|||||
| Total Asia Pacific |
13.2 | 14.5 | ||||||
|
|
|
|
|
|||||
| Consolidated Total |
$ | 431.6 | $ | 337.0 | ||||
|
|
|
|
|
|||||
Revenues in the United States were $205.6 million and $152.9 million for the nine months ended March 31, 2021 and 2020, respectively, while revenues in France were $115.0 million and $91.0 million, respectively.
F-107
Table of Contents
Notes to Unaudited Interim Condensed Consolidated Financial Statements
The following details revenue by timing of recognition (in millions):
| Revenues | ||||||||
| For the nine months ended March 31, |
||||||||
| 2021 | 2020 | |||||||
| Point in time |
$ | 299.7 | $ | 242.9 | ||||
| Over time |
131.9 | 94.1 | ||||||
|
|
|
|
|
|||||
| Total revenues |
$ | 431.6 | $ | 337.0 | ||||
|
|
|
|
|
|||||
12. Loss Per Share
A reconciliation of the numerator and denominator used in the calculation of basic and diluted loss per common share is as follows:
| For the nine months March 31, |
||||||||
| In millions, except per share amounts | 2021 | 2020 | ||||||
| Net loss attributable to Mirion Technologies (TopCo), Ltd. stockholders |
$ | (130.9 | ) | $ | (94.6 | ) | ||
| Weighted average common shares outstandingbasic and diluted |
6.525 | 6.435 | ||||||
|
|
|
|
|
|||||
| Loss per share attributable to Mirion Technologies (TopCo), Ltd. stockholdersbasic and diluted |
$ | (20.06 | ) | $ | (14.70 | ) | ||
|
|
|
|
|
|||||
| Anti-dilutive employee share-based awards, excluded |
0.3 | 0.4 | ||||||
Potentially dilutive unvested stock awards using the treasury stock method or the if-converted method, as applicable, are included when calculating diluted net income earnings (loss) per share attributable to Mirion Technologies (TopCo), Ltd. when their effect is dilutive. Because the Company incurred a net loss for the period ending nine months ended March 31, 2021 and 2020, respectively, none of the potentially dilutive common shares were included in the diluted share calculations for those periods as they would have been anti-dilutive.
13. Restructuring
The Company incurs costs associated with restructuring initiatives intended to improve operating performance, profitability, and working capital levels. Actions associated with these initiatives may include improving productivity, workforce reductions, and the consolidation of facilities.
As of March 31, 2021, the Company has identified restructuring actions will result in additional charges of approximately $9.6 million, primarily in fiscal years 2022 and 2023.
The Companys restructuring expense for the nine months ended March 31, 2021 is comprised of the following:
| For the nine months ended March 31, 2021 |
||||||||||||
| (in millions) | Cost of revenue |
Selling, general and administrative |
Total | |||||||||
| Severance and employee costs |
$ | 2.1 | $ | 0.8 | $ | 2.9 | ||||||
| Other (1) |
| 0.7 | 0.7 | |||||||||
|
|
|
|
|
|
|
|||||||
| Total |
$ | 2.1 | $ | 1.5 | $ | 3.6 | ||||||
|
|
|
|
|
|
|
|||||||
| (1) | Includes facilities, outside services, and IT costs. |
F-108
Table of Contents
Notes to Unaudited Interim Condensed Consolidated Financial Statements
The Company does not allocate restructuring charges to segment income, instead these costs are included in Corporate & Other. Restructuring activity and expenses for the nine months ended March 31, 2020 were not material.
The following table summarizes the changes in the Companys accrued restructuring balance, which are included in Accrued expenses and other current liabilities in the accompanying balance sheet (in millions).
| Balance at June 30, 2020 |
$ | | ||
| Restructuring charges |
3.6 | |||
| Payments |
(1.6 | ) | ||
| Adjustments |
| |||
|
|
|
|||
| Balance at March 31, 2021 |
$ | 2.0 | ||
|
|
|
14. Subsequent Events
Management has evaluated subsequent events through June 29, 2021, the date the financials were issued.
Business Combination
On June 17, 2021, Mirion entered into a Business Combination Agreement with GS Acquisition Holdings Corp II (GSAH) a special purpose acquisition company. In accordance with the terms and subject to the conditions of the Business Combination Agreement, the consideration to be paid to Mirions stockholders and holders of Shareholder Notes in connection with the Business Combination is $1.7 billion, of which $1.3 billion will be paid in cash and the remainder will consist of shares of GSAH common stock. The balance of the 2019 Credit Facility as of the closing date will be paid in full through cash consideration or the new financing per the Commitment Letter described below. The transaction is expected to close in the second half of calendar year 2021, subject to certain closing conditions, including regulatory approvals, and approval of GSAHs stockholders.
Commitment Letter
In connection with the transaction, Mirion Technologies (HoldingSub2) Ltd., a subsidiary of Mirion TopCo, entered into a commitment letter (the Commitment Letter) with Goldman Sachs Lending Partners LLC (GS Lending) and Citigroup Global Markets Inc. (Citi) pursuant to which GS Lending and Citi have committed to provide to Mirion Technologies, Inc., as the subsidiary borrower, and a parent entity of the Mirion TopCo business to be formed, a $830 million senior secured term loan B facility (the Term Loan) and a $90 million revolving facility (the Revolving Facility). The Term Loan will mature seven years after the closing date and will amortize in equal quarterly installments in an aggregate annual amount equal to 1% of the initial principal amount of the Term Loan. The Revolving Facility will mature five years after the closing date.
F-109
Table of Contents
SUN NUCLEAR CORPORATION
INDEX TO CONSOLIDATED FINANCIAL STATEMENTS
| Contents |
Page | |||
| F-111 | ||||
| F-112 | ||||
| F-113 | ||||
| F-114 | ||||
| Consolidated Statement of Cash Flows for the Period from January 1, 2020 through December 18, 2020 |
F-115 | |||
| F-116 | ||||
F-110
Table of Contents
To the stockholders and the Board of Directors of Mirion Technologies, Inc.
We have audited the accompanying consolidated financial statements of Sun Nuclear Corporation and its subsidiaries (the Company), which comprise the balance sheet as of December 18, 2020, and the related statements of Operations and Comprehensive Income, Change in Stockholders Equity, and Cash Flows for the period from January 1, 2020 to December 18, 2020, and the related notes to the consolidated financial statements.
Managements Responsibility for the Consolidated Financial Statements
Management is responsible for the preparation and fair presentation of these consolidated financial statements in accordance with accounting principles generally accepted in the United States of America; this includes the design, implementation, and maintenance of internal control relevant to the preparation and fair presentation of consolidated financial statements that are free from material misstatement, whether due to fraud or error.
Auditors Responsibility
Our responsibility is to express an opinion on these consolidated financial statements based on our audit. We conducted our audit in accordance with auditing standards generally accepted in the United States of America. Those standards require that we plan and perform the audit to obtain reasonable assurance about whether the consolidated financial statements are free from material misstatement.
An audit involves performing procedures to obtain audit evidence about the amounts and disclosures in the consolidated financial statements. The procedures selected depend on the auditors judgment, including the assessment of the risks of material misstatement of the consolidated financial statements, whether due to fraud or error. In making those risk assessments, the auditor considers internal control relevant to the Companys preparation and fair presentation of the consolidated financial statements in order to design audit procedures that are appropriate in the circumstances, but not for the purpose of expressing an opinion on the effectiveness of the Companys internal control. Accordingly, we express no such opinion. An audit also includes evaluating the appropriateness of accounting policies used and the reasonableness of significant accounting estimates made by management, as well as evaluating the overall presentation of the consolidated financial statements.
We believe that the audit evidence we have obtained is sufficient and appropriate to provide a basis for our audit opinion.
Opinion
In our opinion, the consolidated financial statements referred to above present fairly, in all material respects, the financial position of Sun Nuclear Corporation as of December 18, 2020, and the results of its operations and its cash flows for the period from January 1, 2020 to December 18, 2020 in accordance with accounting principles generally accepted in the United States of America.
/s/ Deloitte & Touche LLP
Atlanta, Georgia
April 30, 2021
F-111
Table of Contents
Sun Nuclear Corporation and Subsidiaries
Consolidated Statement of Operations and Comprehensive Income
(Dollars in thousands, except share data)
|
Period from January 1, 2020 through December 18, 2020 |
||||
| Net revenue: |
||||
| Products |
$ | 75,706 | ||
| Services |
22,434 | |||
|
|
|
|||
| Total net revenue |
98,140 | |||
|
|
|
|||
| Cost of revenue: |
||||
| Cost of products |
21,871 | |||
| Cost of services |
11,028 | |||
|
|
|
|||
| Total cost of revenue |
32,899 | |||
|
|
|
|||
| Gross profit |
65,241 | |||
| Operating expenses: |
||||
| Research and development |
14,676 | |||
| Selling, general and administrative |
33,863 | |||
|
|
|
|||
| Total operating expenses |
48,539 | |||
|
|
|
|||
| Income from operations |
16,702 | |||
| Interest expense |
57 | |||
| Foreign current transaction gain |
(668 | ) | ||
| Other expenses |
243 | |||
|
|
|
|||
| Net income |
$ | 17,070 | ||
|
|
|
|||
| Other comprehensive income: |
||||
| Foreign currency translation adjustment |
94 | |||
|
|
|
|||
| Total other comprehensive income |
94 | |||
|
|
|
|||
| Total comprehensive income |
$ | 17,164 | ||
|
|
|
|||
The accompanying notes are an integral part of these consolidated financial statements.
F-112
Table of Contents
Sun Nuclear Corporation and Subsidiaries
Consolidated Balance Sheet
(Dollars in thousands, except share data)
|
As of December 18, 2020 |
||||
| ASSETS |
||||
| Current assets: |
||||
| Cash and cash equivalents |
$ | 24,292 | ||
| Accounts receivable, net of allowance for doubtful accounts of $2,892 |
23,856 | |||
| Inventory |
8,820 | |||
| Prepaid expenses and other current assets |
2,267 | |||
|
|
|
|||
| Total current assets |
59,235 | |||
|
|
|
|||
| Property and equipment, net of accumulated depreciation of $12,692 |
16,333 | |||
| Accounts receivable, non-current |
1,547 | |||
| Equity Investments |
2,423 | |||
| Goodwill |
2,317 | |||
| Intangible assets, net of accumulated amortization of $2,300 |
369 | |||
| Other non-current assets |
943 | |||
|
|
|
|||
| Total non-current assets |
23,932 | |||
|
|
|
|||
| Total assets |
$ | 83,167 | ||
|
|
|
|||
| LIABILITIES AND STOCKHOLDERS EQUITY |
||||
| Current liabilities: |
||||
| Accounts payable |
$ | 1,967 | ||
| Accrued compensation |
8,522 | |||
| Deferred revenue, current |
20,354 | |||
| Accrued expenses and other current liabilities |
4,792 | |||
|
|
|
|||
| Total current liabilities |
35,635 | |||
|
|
|
|||
| Non-current liabilities: |
||||
| Deferred revenue, excluding current portion |
995 | |||
| Notes payable |
6,556 | |||
|
|
|
|||
| Total non-current liabilities |
7,551 | |||
|
|
|
|||
| Commitments and contingencies (Note 14) |
||||
| Stockholders equity |
||||
| Common stock, $0 par value; 1,100,000 shares authorized, 465,634 shares issued and 220,032 shares outstanding |
436 | |||
| Treasury stock at cost, 245,602 shares |
(4,561 | ) | ||
| Additional paid-in capital |
2,485 | |||
| Accumulated other comprehensive income |
99 | |||
| Retained earnings |
41,522 | |||
|
|
|
|||
| Total stockholders equity |
39,981 | |||
|
|
|
|||
| Total liabilities and stockholders equity |
$ | 83,167 | ||
|
|
|
|||
The accompanying notes are an integral part of these consolidated financial statements.
F-113
Table of Contents
Sun Nuclear Corporation and Subsidiaries
Consolidated Statement of Changes in Stockholders Equity
(Dollars in thousands, except share data)
|
Period from January 1, 2020 through December 18, 2020 |
||||||||||||||||
| Common Stock | Treasury Stock | |||||||||||||||||||||||||||||||
| Number of Shares |
Common Stock |
Number of Shares |
Treasury Stock |
Additional Paid-In Capital |
Retained Earnings |
Accumulated Other Comprehensive Income |
Total | |||||||||||||||||||||||||
| Balance as of December 31, 2019 |
220,032 | $ | 436 | 245,602 | $ | (4,561 | ) | $ | 2,485 | $ | 48,191 | $ | 5 | $ | 46,556 | |||||||||||||||||
| Net Income |
17,070 | 17,070 | ||||||||||||||||||||||||||||||
| Other comprehensive income |
94 | 94 | ||||||||||||||||||||||||||||||
| Dividends paid |
(22,018 | ) | (22,018 | ) | ||||||||||||||||||||||||||||
| Asset distribution to shareholders related to SunRADON LLC |
(1,721 | ) | (1,721 | ) | ||||||||||||||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||||
| Balance as of December 18, 2020 |
220,032 | $ | 436 | 245,602 | $ | (4,561 | ) | $ | 2,485 | $ | 41,522 | $ | 99 | $ | 39,981 | |||||||||||||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|||||||||||||||||
The accompanying notes are an integral part of these consolidated financial statements.
F-114
Table of Contents
Sun Nuclear Corporation and Subsidiaries
Consolidated Statement of Cash Flows
(Dollars in thousands, except share data)
|
Period from January 1, 2020 through December 18, 2020 |
||||
| Cash flow from operating activities |
|
|||
| Net income |
$ | 17,070 | ||
| Adjustments to reconcile net income to net cash provided by operating activities: |
||||
| Depreciation |
1,574 | |||
| Amortization |
361 | |||
| Provision for allowance for doubtful accounts |
852 | |||
| Loss on asset dispositions |
95 | |||
| Changes in operating assets and liabilities: |
||||
| Accounts receivable |
3,670 | |||
| Inventory |
272 | |||
| Prepaid expenses and other current assets |
(523 | ) | ||
| Other non-current assets |
(528 | ) | ||
| Accounts payable |
(536 | ) | ||
| Accrued expenses |
1,446 | |||
| Accrued compensation |
944 | |||
| Deferred revenue |
(1,706 | ) | ||
| Other |
257 | |||
|
|
|
|||
| Net cash provided by operating activities |
23,248 | |||
|
|
|
|||
| Cash flows from investing activities |
||||
| Expenditures for property and equipment |
(1,660 | ) | ||
|
|
|
|||
| Net cash used in investing activities |
(1,660 | ) | ||
|
|
|
|||
| Cash flows from financing activities |
||||
| Dividends paid |
(22,018 | ) | ||
| Proceeds from debt |
6,407 | |||
| Cash distributions to shareholders related to SunRADON LLC |
(876 | ) | ||
| Payment of debt |
(81 | ) | ||
|
|
|
|||
| Net cash used in financing activities |
(16,568 | ) | ||
|
|
|
|||
| Effect of currency exchange rate changes on cash and cash equivalents |
35 | |||
|
|
|
|||
| Net increase in cash and cash equivalents |
5,055 | |||
| Cash and cash equivalents, beginning of period |
19,237 | |||
|
|
|
|||
| Cash and cash equivalents, end of period |
$ | 24,292 | ||
|
|
|
|||
| Supplemental non-cash investing and financing activities: |
||||
| Non-cash distribution to shareholders related to SunRADON LLC |
845 | |||
The accompanying notes are an integral part of these consolidated financial statements.
F-115
Table of Contents
Sun Nuclear Corporation and Subsidiaries
Notes to the Consolidated Financial Statements
Note 1: DESCRIPTION OF BUSINESS
Sun Nuclear Corporation (SNC or the Company) is a global leader in radiation oncology quality assurance (QA), delivering patient safety solutions for diagnostic imaging and radiation therapy centers around the world. Headquartered in Melbourne, FL, the Company manufactures and provides radiation measurement and quality assurance medical instrumentation and services. On December 18, 2020, the Company was acquired by Mirion Technologies, Inc.
SNC International, Inc., one of the Companys wholly-owned subsidiaries, is an Interest Charge Domestic International Sales Corporation (IC-DISC) and serves as a vehicle for tax savings on U.S. exports. The Companys wholly-owned subsidiaries also include Gammex, Inc. and Gammex RMI GmbH, with operations based out of Wisconsin and Germany, respectively. Gammex, Inc. designs and manufactures accessories and testing devices for the diagnostic imaging and radiation therapy markets. The equipment is used on linear accelerators, Computerized Tomography (CT) and Magnetic Resonance Imaging (MRI) scanners, X-ray, mammography and fluoroscopy machines and is distributed worldwide. Gammex RMI GmbH provides for sales and distribution of the Companys products and services.
SunRADON LLC (SunRadon or the SunRadon Business) was created as a wholly-owned subsidiary of the Company during 2019. The subsidiary is based in Melbourne, Florida and focuses on serving professionals, homeowners, and businesses worldwide, with the goal of improving indoor air quality, by focusing on Radon levels. On August 31, 2020, the Company distributed 100% of its interest in SunRadon to the Companys existing shareholders.
Note 2: SUMMARY OF SIGNIFICANT ACCOUNTING POLICIES
Basis of Accounting
The consolidated financial statements, which are prepared in conformity with generally accepted accounting principles in the United States (GAAP), include our wholly owned and controlled subsidiaries and affiliates. All significant intercompany transactions and balances have been eliminated in consolidation.
Principles of Consolidation
The consolidated financial statements include the accounts of the Company and all subsidiaries in which the Company has a controlling financial interest. The balance sheets of SNC International, Inc., Gammex, Inc. and Gammex RMI GmbH as of December 18, 2020 and the results of operations for the period from January 1, 2020 through December 18, 2020 are included in the consolidated financial statements, which have been prepared in accordance with ASC 810, Consolidation.
The Company has assessed certain affiliates, SNC North, LLC, 425 Pineda Court, LLC, 330 Pineda Court, LLC and 7600 Discovery Drive, LLC (the Affiliates), and determined that the Affiliates represent a variable interest entity (VIE) in accordance with ASC 810, Consolidation, of which the Company is the primary beneficiary. Therefore, the Affiliates have been included in the consolidated financial statements as of December 18, 2020 and for the period from January 1, 2020 through December 18, 2020. See Note 11 Variable Interest Entities for further details.
Distribution of SunRadon
In accordance with ASC 805-50, Business Combinations - Related Issues, the distribution of SunRadon has been accounted for as a transaction among entities under common control since the Companys shareholders controlled the Company and SunRadon prior to and after the distribution.
F-116
Table of Contents
Sun Nuclear Corporation and Subsidiaries
Notes to the Consolidated Financial Statements
Since the distribution of SunRadon constituted a transfer of net assets under common control, the Company did not record any gain or loss on the transaction. SunRadon was deconsolidated from the Companys financial statements and was no longer a wholly owned subsidiary of the Company as of December 18, 2020. However, operating results of SunRadon for the period from January 1, 2020 through its distribution on August 31, 2020 were included in the Companys consolidated statement of operations and comprehensive income, including net revenue of $2.6 million and net income of $0.5 million.
Use of Estimates
The preparation of financial statements in conformity with GAAP requires management to make estimates and assumptions that affect the reported amounts of assets and liabilities and disclosure of contingent assets and liabilities at the date of the financial statements and reported amounts of revenues and expenses during the reporting period. Significant estimates and judgments made by management in preparing these financial statements include but not limited to revenue recognition, collectability of accounts receivable, inventory valuations, expected future cash flows used to evaluate the recoverability of long-lived assets and the useful lives of depreciable assets.
Various assumptions and other factors underlie the determination of these significant estimates. Management evaluates its estimates and assumptions on an ongoing basis using historical experience and known facts and circumstances, including the current economic environment. The global impact of the COVID-19 pandemic may also affect the accounting estimates, which may materially change from period to period due to changing market factors. The Company regularly evaluates these significant factors and makes adjustments where facts and circumstances dictate.
Revenue Recognition
The Companys revenues are derived primarily from the sale of diagnostic imaging and radiation therapy hardware and software products and services, which include post-contract customer support (PCS), installation services, training and extended warranty and maintenance services.
The Company accounts for a contract with a customer when the parties to the contract have approved the contract and are committed to perform, the rights of the parties are identified, the contract has commercial substance, the payment terms are identified and collectability of the contract consideration is probable. Customers include end-customers and distributors through agreements whereby distributors purchase products on behalf of the end-customer.
The majority of the Companys revenue arrangements consist of multiple performance obligations including hardware, software, and services. For bundled arrangements, the Company accounts for individual products and services separately if they are distinct and separately identifiable from other performance obligations in the bundled package. The total contract consideration is allocated between separate products and services in a bundle based on their individual stand-alone selling price (SSP).
The Companys revenues are measured based on the consideration specified in the contract with each customer, net of any discounts and taxes collected from customers that are remitted to government authorities. The Companys products are generally sold with limited rights of return, and the Company typically does not provide credits, rebates, or incentives, which may be required to be accounted for as variable consideration when estimating the amount of revenue to be recognized.
Payment terms and conditions vary by contract type, although the terms are generally commensurate with a significant milestone, such as contract signing, shipment, delivery, acceptance or service commencement. In
F-117
Table of Contents
Sun Nuclear Corporation and Subsidiaries
Notes to the Consolidated Financial Statements
instances where the timing of revenue recognition differs from the timing of invoicing, the Company has determined its contracts do not include a significant financing component. The primary purpose of the Companys invoicing terms is to provide customers with simplified and predictable ways of purchasing the Companys products and services, rather than to receive financing from the Companys customers, such as invoicing at the beginning of a contract term with revenue recognized ratably over the contract period for a service contract. The Company applies the practical expedient to not adjust for a significant financing component if the difference between when the payment is received and revenue to be recognized is expected, at contract inception, to be less than one year or less.
From time to time, the Companys contracts are modified to account for additional performance obligations. The Companys contract modifications are generally accounted for prospectively.
The following is a description of the revenue recognition for the principal activities from which the Company generates revenues.
Hardware Products and Installation
Hardware products may include software that the hardware is dependent on and highly interrelated with and cannot operate without. The Company typically has a standard base configuration for its hardware products, but there are typically multiple options and configuration choices. Revenues from the sale of hardware are recognized at a point in time when the Company transfers control to the customer, which occurs upon shipment.
The customer may purchase installation services related to the hardware. Hardware product installation includes mounting and connecting to the customers radiation oncology and alignment equipment, calibrating and validating configuration. In addition, a number of testing protocols are completed to confirm the equipment is performing to the contracted specifications. The Company recognizes revenues for hardware installation when control and benefit transfer to the customer, which occurs over time as the installation service is completed. Hardware installation is included in service revenues when identified as a separate performance obligation.
Software Products and Installation
Software products relate primarily to the Companys SunCHECK software platform that integrates, automates, and standardizes radiation therapy quality management for both the hardware (SunCHECK Machine) and patients (SunCHECK Patient). Software installation includes downloading the software to the customers servers or new servers purchased from the Company, configuration of the software with the customers machines, custom beam modeling if applicable, device calibration support and verification of connectivity. The Company recognizes revenues for on-premise software and software installation when control and benefit transfer to the customer, which occurs over time as the software and installation service is performed.
Service
Service revenues include revenues from initial and extended software maintenance and support agreements, extended hardware warranty agreements, hardware installation, training, and repairs. Revenues from hardware and software support agreements represent a stand-ready performance obligation satisfied over time ratably over the term of the agreement as the customer benefits from the service throughout the service period. Installation and training revenues are recognized in the period the installation and training are performed.
Warranties
The Companys sale of hardware includes a standard assurance-type warranty. The Company uses the cost accrual method to account for assurance-type warranties. The Companys sale of software products includes a
F-118
Table of Contents
Sun Nuclear Corporation and Subsidiaries
Notes to the Consolidated Financial Statements
standard one-year warranty that provides services in addition to an assurance-type warranty such as bug fixes, customer support, and when and if available software upgrades. These service-type warranty features are recorded as a separate performance obligation and recognized ratably over the one-year warranty period.
Contract Balances
The timing of revenue recognition, billings and cash collections result in trade receivables, unbilled receivables and deferred revenues on the consolidated balance sheet. If revenue recognition occurs prior to billing, an unbilled receivable is recorded which may represent a contract asset. Contract liabilities include billings in excess of revenues recognized, such as those resulting from customer advances and deposits and deferred revenue on service contracts. Service contracts are usually billed at the beginning of the contract period or at annual intervals during the contract which typically results in deferred revenue which represents a contract liability. The contract assets and liabilities are reported on a contract-by-contract basis at the end of the reporting period.
Costs Incurred to Obtain Contracts
The Company capitalizes and amortizes the incremental costs of obtaining a contract, primarily related to sales commissions, if the Company expects the benefit of those costs to be longer than one year. The Company applies a practical expedient to expense costs as incurred for costs to obtain a contract when the amortization period would have been one year or less. The capitalized sales commissions are amortized over the contract term. The pattern of amortization is commensurate with the pattern of transfer of control of the performance obligations to the customer.
As of December 18, 2020, the balance of capitalized costs to obtain a contract was $1.4 million. The Company has classified the capitalized costs to obtain a contract as a component of prepaid expenses and other current assets and other assets with respect to the current and non-current portions of capitalized costs, respectively, on the consolidated balance sheet. The Company did not have any impairment losses on contract assets for the period presented. Amortization expenses related to these costs total $0.3 million for the period from January 1, 2020 through December 18, 2020. Such costs are included as a component of Selling, general and administrative expenses on the consolidated statement of operations and comprehensive income.
Shipping and handling costs for product shipments to customers are included in Cost of products. When control of the goods transfers prior to the completion of our obligation to ship the products to our customers, we have elected the practical expedient to account for the shipping services as a fulfillment cost. Shipping and handling costs for inventory purchases are capitalized in inventory and expensed in Cost of products.
Cash and Cash Equivalents
For purposes of the consolidated balance sheet and statement of cash flows, the Company considers all highly liquid investments with a maturity of three months or less to be cash equivalents.
Accounts Receivable, net
Accounts receivable is uncollateralized customer obligations due under normal trade terms typically requiring payment within 30 to 90 days from the invoice date. No interest is typically charged on unpaid accounts receivable with invoice dates over the payment term. Accounts receivable are stated at amortized cost. Credit is extended based on evaluation of the customers financial condition, and generally, collateral is not required.
The carrying amounts of accounts receivable are reduced by an allowance for doubtful accounts. The allowance for doubtful accounts is based on a combination of historical data, cash payment trends, specific customer issues,
F-119
Table of Contents
Sun Nuclear Corporation and Subsidiaries
Notes to the Consolidated Financial Statements
write-off trends, general economic conditions, and other factors. These factors are continuously monitored by management to arrive at the estimate for the amount of accounts receivable that may be ultimately uncollectible. In circumstances where the Company is aware of a specific customers inability to meet its financial obligations, the Company records a specific allowance for doubtful accounts against amounts due in order to reduce the net recognized receivable to the amount it reasonably believes will be collected.
Balances that are still outstanding after management has used reasonable collection efforts are written off through a charge to the allowance for doubtful accounts and a credit to accounts receivable.
Inventory
Inventory consists of spare parts for repairing equipment, which has been sold to customers, materials used in manufacturing, work-in-process and finished goods. Inventories are stated at the lower of cost or market and market is determined based on net realizable value. Inventories are valued using the first-in, first-out method. The Company periodically reviews the age and turnover of its inventory to determine whether any inventory has become obsolete or has declined in value and incurs a charge to operations for known and anticipated inventory obsolescence.
Prepaid Expenses
Certain payments to vendors reflect costs applicable to future accounting periods and are recorded as prepaid expenses. These amounts are recognized as expenses in the period in which the Company receives those benefits.
Property and Equipment, net
Property and equipment are stated at cost less accumulated depreciation and amortization. Depreciation and amortization expense for financial reporting purposes is computed on an accelerated and straight-line basis over the estimated useful lives of the assets. Estimated useful lives for various asset classes range from 3 to 39 years. Land is not subject to depreciation. Normal maintenance and repairs are expensed as incurred, and renovations and major improvements are capitalized.
Investments
The Company follows ASC Topic 321 Investments - Equity Securities (ASC 321), which requires all applicable investments in equity securities with readily determinable fair values to be valued at such. For those that do not have readily determinable fair values and over which the Company does not have significant influence, a measurement alternative can be elected to measure such investments at cost, less any impairment, plus or minus changes resulting from observable price changes in orderly transactions for identical or similar investments of the same issuer, if any. The Company performs a qualitative assessment of whether the investment is impaired at each reporting date. If a qualitative assessment indicates that the investment is impaired, the Company estimates the investments fair value in accordance with the principles of ASC Topic 820 Fair Value Measurements and Disclosures (ASC 820). If the fair value is less than the investments carrying value, the entity recognizes an impairment loss in net income equal to the difference between the carrying value and fair value of the investment.
Goodwill and Intangible Assets
Goodwill arises from the purchase price for acquired businesses exceeding the fair value of tangible and intangible assets acquired less assumed liabilities and noncontrolling interests and is stated at cost. Intangible assets, which consist of patents, trademarks and licenses, are stated at cost and are amortized using the straight-line method over five years.
F-120
Table of Contents
Sun Nuclear Corporation and Subsidiaries
Notes to the Consolidated Financial Statements
Impairment of Goodwill and Other Long-Lived Assets
Management assesses goodwill for impairment at least annually at the end of the reporting period or more frequently as triggering events occur that indicate that it is more likely than not that an impairment exists. The company is permitted to first assess qualitative factors to determine whether the quantitative goodwill impairment test is necessary. If the qualitative assessment results in a determination that the fair value of a reporting unit is more likely than not less than its carrying amount, the company performs a quantitative goodwill impairment test. However, if the assessment leads to a determination that it is more likely than not that the fair value of a reporting unit is greater than its carrying amount, then no further assessments are required. A quantitative assessment for the determination of impairment is made by comparing the carrying amount of the reporting unit with its fair value, which is generally calculated using a combination of market, comparable transaction and discounted cash flow approaches. We completed the required annual testing of goodwill for impairment and determined there was no impairment for the period from January 1, 2020 through December 18, 2020.
Impairment losses are recorded on long-lived assets, including finite-lived intangible assets, used in operations when indicators of impairment are present and the quoted market price, if available or the estimated fair value of those assets are less than the assets carrying value and are not recoverable. Determination of recoverability is based on an estimate of undiscounted future cash flows resulting from the use of the asset and its eventual disposition. In the event that such cash flows are not expected to be sufficient to recover the carrying amount of the assets, the assets are written down to their fair values. Impairment losses are charged to the consolidated statements of income and comprehensive income for the difference between the fair value and carrying value of the asset. The Company did not recognize any impairment charges for long-lived assets for the period from January 1, 2020 through December 18, 2020.
Warranty Reserve
The Company warrants most of its products for a specific period of time, usually 12 or 24 months from installation, against material defects. In addition, the Company often includes additional support services and extended warranties and recognizes these services as a separate performance obligation along with its standard warranty cost accrual. The accrued warranty costs represent the best estimate at the time of sale of the total costs that the Company will incur to repair or replace product parts that fail while still under warranty. The amount of the accrued estimated warranty costs obligation for established products is primarily based on historical experience as to product failures adjusted for current information on repair costs. For new products, estimates include the historical experience of similar products, as well as reasonable allowance for warranty expenses associated with new products.
Fair Value of Financial Instruments
ASC 825-10, Disclosure about Fair Value of Financial Instruments, requires disclosure of an estimate of fair value of certain financial instruments. The Companys significant financial instruments are cash, accounts receivable, inventory, investments, other short-term assets and liabilities and non-current accounts receivable. For these financial instruments except investments, carrying values approximate fair value. For investments, management has elected to use the measurement alternative in ASC 321.
Concentration of Credit Risk
The Company maintains cash balances at financial institutions that are insured by the Federal Deposit Insurance Corporation (FDIC). All deposit accounts are insured up to $250,000 by the FDIC in aggregate per financial
F-121
Table of Contents
Sun Nuclear Corporation and Subsidiaries
Notes to the Consolidated Financial Statements
institution. The amount of credit exposure in excess of federally insured limits as of December 18, 2020 was $17.1 million. Amounts held by the Company or its subsidiaries in foreign financial institutions are not insured by the FDIC. The amount of credit exposure for accounts held in foreign financial institutions as of December 18, 2020 was $1.7 million.
As of December 18, 2020 and for the period from January 1, 2020 through December 18, 2020, no single customer represented 10% or more of the Companys accounts receivable or revenue. For the period from January 1, 2020 through December 18, 2020, two vendors collectively accounted for 29% of the purchase of raw materials, supplies and tooling.
Advertising
The Company expenses advertising costs as they are incurred. Advertising expenses for the period from January 1, 2020 through December 18, 2020 were $0.2 million, which are included as a component of Selling, general and administrative expenses on the consolidated statement of operations and comprehensive income.
Software Development Costs
The costs incurred for the development of computer software to be sold, leased, or otherwise marketed are capitalized in accordance with ASC 985-20, Costs of Software to be Sold, Leased or Marketed, when technological feasibility has been established. Technological feasibility generally occurs when all planning, design, coding, and testing activities are completed that are necessary to establish that the product can be produced to meet its design specifications, including functions, features, and technical performance requirements. The establishment of technological feasibility is an ongoing assessment of judgment by management with respect to certain external factors, including, but not limited to, anticipated future revenues, estimated economic life and changes in technology. Software development costs incurred prior to the establishment of technological feasibility are expensed as incurred. Software development costs incurred subsequent to the establishment of technological feasibility through the period of general market availability of the products are capitalized, including direct labor and related expenses for software development for new products and enhancements to existing software products. Maintenance and training costs are expensed as incurred.
Amortization of capitalized software development costs begins when the product is available for general release. Amortization expense should be the greater of the amount calculated using the ratio of current gross revenue over the total current/anticipated gross revenues of the software or the straight-line method over the estimated useful lives of the software, which is typically three years. Unamortized capitalized software development costs determined to be in excess of the net realizable value of the product are expensed immediately.
Capitalized software costs are subject to an ongoing assessment of recoverability based on anticipated future revenues and changes in software technologies at least annually or more frequently when events or circumstances make it more likely than not that impairment may have occurred. In the event of impairment, unamortized capitalized software costs are compared to the net realizable value of the related product and the carrying value of the related assets are written down to the net realizable value to the extent the unamortized capitalized costs exceed such value. The net realizable value is the estimated future gross revenues from the related product reduced by the estimated future costs of completing and disposing of such product, including the costs of providing related maintenance and customer support.
For the period from January 1, 2020 through December 18, 2020, qualifying software costs that were capitalized totaled $0.6 million and amortization expenses associated with capitalized software costs totaled $0.2 million, which were included in Cost of product on the consolidated statement of operations and comprehensive income.
F-122
Table of Contents
Sun Nuclear Corporation and Subsidiaries
Notes to the Consolidated Financial Statements
Research and Development Costs
Costs related to the research, design and development of hardware products are expensed as incurred. These costs include direct compensation and benefits for research and development personnel, costs for material used in research and development activities, professional services and allocated portions of facilities and other corporate costs.
Income Tax Status
The Company, with the consent of its shareholders, elected under the Internal Revenue Code to be an S corporation. The shareholders of an S corporation, in lieu of corporate income taxes are taxed on their proportionate share of the Companys taxable income. Therefore, no provision or liability for federal or state income taxes has been included in the consolidated financial statements.
Tax positions are recognized only if it is more likely than not that the tax position would be sustained in a tax examination, with a tax examination being presumed to occur. The amount recognized is the largest amount that is greater than 50% likely of being realized on examination. For tax positions not meeting the more likely than not test, no tax position is recorded. The Company is subject to routine audits by taxing jurisdictions; however, there are currently no audits for any tax periods in progress.
Foreign Currency
Financial position and reported results of operations of the Companys non-U.S. dollar functional international subsidiary Gammex RMI GmbH are measured using Euros (the local currency) as the functional currency. Assets and liabilities of these operations are translated at the exchange rate in effect at the end of the reporting period. The translation adjustments related to assets and liabilities that arise from the use of differing exchange rates from period to period are included in Accumulated other comprehensive income in Stockholders equity section of the consolidated balance sheet. Accounts on the consolidated statement of operations and comprehensive income are translated at average rates of exchange prevailing during the reporting period. Gains and losses resulting from foreign currency monetary transactions are reported in Foreign currency transaction gain on the consolidated statement of operations and comprehensive income.
Recently Issued Accounting Pronouncements
In February 2016, the FASB issued Accounting Standards Update 2016-02 Leases, and subsequently issued additional ASUs amending this ASU which amends various aspects of existing guidance for leases. This guidance requires an entity to recognize assets and liabilities arising from a lease for both financing and operating leases, along with additional qualitative and quantitative disclosures. The effective date of the new standard for private companies has been extended to fiscal years beginning after December 15, 2021, with early adoption permitted. The Company will adopt this guidance in 2022 but has not determined which transition method will be utilized. The Company is still assessing the potential impact of this pronouncement to its consolidated financial statements.
In June 2016, the FASB issued Accounting Standards Update 2016-13, Financial InstrumentsCredit Losses. This ASU introduced a new accounting model, the Current Expected Credit Losses model (CECL), which requires earlier recognition of credit losses and additional disclosures related to credit risk. The CECL model utilizes a lifetime expected credit loss measurement objective for the recognition of credit losses for loans and other receivables at the time the financial asset is originated or acquired. The expected credit losses are adjusted each period for changes in expected lifetime credit losses. This model replaces the multiple existing impairment
F-123
Table of Contents
Sun Nuclear Corporation and Subsidiaries
Notes to the Consolidated Financial Statements
models in current GAAP, which generally require that a loss be incurred before it is recognized. The new standard will also apply to financial assets arising from revenue transactions such as contract assets and accounts receivables and is effective for private companies for fiscal years beginning after December 15, 2022 with early adoption permitted. The Company is currently evaluating the impact of the guidance on its consolidated financial statements.
In August 2018, the FASB issued ASU 2018-15, Intangibles - Goodwill and Other - Internal-Use Software (Subtopic 350-40): Customers Accounting for Implementation Costs Incurred in a Cloud Computing Arrangement That Is a Service Contract. This ASU requires a customer in a cloud computing arrangement that is a service contract to follow the internal-use software guidance to determine which implementation costs to defer and recognize as an asset. The ASU is effective for annual periods beginning after December 15, 2020, and interim periods in annual periods beginning after December 15, 2021. The guidance allows a prospective or retrospective application approach. Early adoption is permitted. The Company will adopt this guidance in 2021 but has not determined which transition method will be utilized. While the Company continues to assess all effects of the adoption, it currently believes the guidance will result in more capitalization of software costs as the Company continues to develop its Software as a Service (SaaS) platform.
Note 3: REVENUE
Contract Balances
The timing of revenue recognition, billings and cash collections result in trade receivables, unbilled receivables and deferred revenues on the consolidated balance sheet. If revenue recognition occurs prior to billing, an unbilled receivable is recorded which may represent a contract asset. Contract liabilities include billings in excess of revenues recognized, such as those resulting from customer advances and deposits and deferred revenue on service contracts. Contract liabilities are classified as current or noncurrent based on the periods over which remaining performance obligations are expected to be transferred to customers. Noncurrent contract liabilities are included within deferred revenue, excluding current portion on the consolidated balance sheet. Contract assets and liabilities are presented on a net basis in the consolidated balance sheet if they arise from different performance obligations in the same contract. The Company did not recognize a contract asset separate from contract liabilities as of December 18, 2020. Contract liability balances as of December 18, 2020 are as follows (in thousands):
| Deferred revenues - current |
$ | 20,354 | ||
| Deferred revenues - non-current |
995 | |||
|
|
|
|||
| Total deferred revenues |
$ | 21,349 | ||
|
|
|
During the period from January 1, 2020 through December 18, 2020, the Company recognized revenues of $20.4 million, which was included in the deferred revenues balance as of December 31, 2019.
Remaining Performance Obligations
Remaining performance obligations represent deferred revenue from open contracts for which performance has already started and the transaction price from executed non-cancelable contracts for which performance has not yet started. The remaining performance obligations primarily relate to hardware and software maintenance, warranty and support agreements, installation and training services,
F-124
Table of Contents
Sun Nuclear Corporation and Subsidiaries
Notes to the Consolidated Financial Statements
The following table represents the Companys remaining performance obligations as of December 18, 2020, and the estimated revenue expected to be recognized in the future related to these remaining performance obligations (in thousands):
| Fiscal years of revenue recognition | ||||||||||||||||
| 2021(1) | 2022 | 2023 | Thereafter | |||||||||||||
| Remaining performance obligations |
$ | 19,688 | $ | 3,736 | $ | 2,319 | $ | 3,330 | ||||||||
| (1) | Fiscal year 2021 includes the revenue recognition of $0.6 million from the remaining performance obligations from December 19, 2020 through December 31, 2020. |
Disaggregation of Revenue
The Company disaggregates its revenues from contracts by United States versus International, as the Company believes this best depicts how the nature, amount, timing and uncertainty of revenues and cash flows are affected by economic factors.
The following table summarizes the Companys total revenues by United States and International for the period from January 1, 2020 through December 18, 2020 (in thousands):
| Period from January 1, 2020 through December 18, 2020 | ||||
| United States |
$ | 56,921 | ||
| International |
41,219 | |||
|
|
|
|||
| Total Revenues |
$ | 98,140 | ||
|
|
|
|||
Note 4: ACCOUNTS RECEIVABLE
The following table summarizes the Companys accounts receivable as of December 18, 2020 (in thousands):
| Accounts receivable, gross |
$ | 28,295 | ||
| Allowance for doubtful accounts |
(2,892 | ) | ||
|
|
|
|||
| Accounts receivable, net |
25,403 | |||
|
|
|
|||
| Short-term |
23,856 | |||
|
|
|
|||
| Long-term(1) |
$ | 1,547 | ||
|
|
|
| (1) | Included in Accounts receivable, noncurrent on the consolidated balance sheet. |
The allowance for doubtful accounts is based on a number of factors, including an evaluation of customer credit worthiness, the age of the outstanding receivable, economic trends and historical experience. Provisions for doubtful accounts are recorded in selling, general and administrative expenses in the accompanying consolidated statements of income and comprehensive income.
Note 5: INVENTORY
Inventory consists of the following as of December 18, 2020 (in thousands):
| Parts and materials |
$ | 5,344 | ||
|
Work-in-process |
686 | |||
| Finished goods |
2,790 | |||
|
|
|
|||
| Total inventory |
$ | 8,820 | ||
|
|
|
F-125
Table of Contents
Sun Nuclear Corporation and Subsidiaries
Notes to the Consolidated Financial Statements
Note 6: PROPERTY AND EQUIPMENT, NET
Property and equipment consists of the following as of December 18, 2020 (in thousands):
| Estimated Useful Lives (In Years) |
December 18, 2020 |
|||||||
| Non-depreciable assets: |
||||||||
| Land |
N/A | $ | 1,785 | |||||
| Depreciable assets: |
||||||||
| Machinery and equipment |
3-15 | 9,738 | ||||||
| Furniture and fixtures |
5 | 2,804 | ||||||
| Building |
39 | 13,714 | ||||||
| Capitalized software |
3 | 948 | ||||||
| Vehicles |
5 | 36 | ||||||
| Less accumulated depreciation and amortization |
(12,692 | ) | ||||||
|
|
|
|
|
|||||
| Total property and equipment, net |
$ | 16,333 | ||||||
|
|
|
|
|
|||||
Depreciation and amortization expense related to property and equipment, net for the period from January 1, 2020 through December 18, 2020 was $1.6 million.
Note 7: GOODWILL AND INTANGIBLE ASSETS
There were no changes to the carrying value of goodwill for the period from January 1, 2020 through December 18, 2020, and no impairment charges have been recorded in prior periods.
Intangible assets consist of the following as of December 18, 2020 (in thousands):
| Licenses and license rights |
Patents and trademarks |
Total | ||||||||||
| Intangible asset |
$ | 2,616 | $ | 53 | $ | 2,669 | ||||||
| Less accumulated amortization |
(2,247 | ) | (53 | ) | (2,300 | ) | ||||||
|
|
|
|
|
|
|
|||||||
| Total intangible asset, net |
$ | 369 | $ | | $ | 369 | ||||||
|
|
|
|
|
|
|
|||||||
Amortization expense for the period from January 1, 2020 through December 18, 2020 was $0.4 million.
Estimated amortization expense for each of the next five years, is as follows (in thousands):
| Year ending December 31, |
||||
| 2021 |
$ | 281 | ||
| 2022 |
31 | |||
| 2023 |
31 | |||
| 2024 |
26 | |||
| 2025 |
| |||
|
|
|
|||
| Total estimated amortization |
$ | 369 | ||
|
|
|
|||
Note 8: INVESTMENTS
On August 31, 2017, the Company converted a promissory note from Oncora Medical, Inc. (Oncora), an unrelated third-party, in the amount of $0.5 million plus the related accrued interest of $0.03 million to 125,543
F-126
Table of Contents
Sun Nuclear Corporation and Subsidiaries
Notes to the Consolidated Financial Statements
shares of preferred stock of Oncora. The Company purchased an additional 44,143 shares of the preferred stock of Oncora on September 6, 2017 for $0.5 million. During 2018, the Company purchased an additional 44,130 shares of preferred stock of Oncora on June 27, 2018 for $0.5 million. During 2019, the Company made two additional payments to Oncora Medical, Inc. in the amounts of $0.1 million on February 27, 2019 and $0.4 million on April 16, 2019. These payments were made under a Simple Agreement for Future Equity (SAFE) which offers the rights to certain shares of the Capital Stock at a discounted rate of 80%. Sun Nuclear Corp. received $0.4 million on September 9, 2019 from Oncora Medical, Inc. cancelling the SAFE payments. As of December 18, 2020, the Companys ownership represents 9% of Oncoras stock, calculated on a fully diluted basis.
The Company has elected to account for the investment using the measurement alternative in ASC 321 and monitors the investment for impairment and any observable price changes, including recent purchases or sales of Oncoras preferred stock. Management determined there was no impairment for the period from January 1, 2020 through December 18, 2020.
Note 9: ACCRUED EXPENSES AND OTHER CURRENT LIABILITIES
Accrued expenses and other current liabilities consists of the following as of December 18, 2020 (in thousands):
| Accrued sales taxes payable |
$ | 1,553 | ||
| Warranty reserve |
452 | |||
| Royalty payable |
150 | |||
| Professional services |
114 | |||
| Other accrued expenses |
2,523 | |||
|
|
|
|||
| Total |
$ | 4,792 | ||
|
|
|
Note 10: PAYCHECK PROTECTION PROGRAM LOANS
On May 3, 2020, the Company applied to Wells Fargo Bank, National Association (the Lender) under the Small Business Administration (SBA) Paycheck Protection Program (PPP) of the Coronavirus Aid, Relief and Economic Security Act of 2020 (the CARES Act) for a loan of $5.3 million for Sun Nuclear Corporation (the Sun Nuclear PPP Loan). On May 15, 2020, the Company applied for an additional PPP loan of $1.2 million for Gammex, Inc. (the Gammex PPP Loan). Under the CARES Act and PPP, a borrower may apply for and be granted forgiveness for all or a part of its PPP loan, including accrued interest. The amount of loan proceeds eligible for forgiveness is based on a formula that takes into account a number of factors, including the amount of loan proceeds used by the borrower during the twenty-four-week period after the loan origination for certain purposes including payroll costs, rent payments on certain leases, and certain qualified utility payments.
On November 18, 2020, the Company submitted an application for forgiveness of the Sun Nuclear PPP Loan to the SBA for the entire amount of principal and interest due on the loan. As of December 18, 2020, the application for the Sun Nuclear PPP Loan is still pending approval from the SBA and the Company plans to start the forgiveness application process for the Gammex PPP Loan. In the event of a loan forgiveness, the beneficiary will be the prior owners of the Company. Therefore, as of December 18, 2020, the Company had $5.2 million and $1.2 million outstanding under the Sun Nuclear PPP Loan and Gammex PPP Loan, respectively, which are included in Notes payable on the consolidated balance sheet.
F-127
Table of Contents
Sun Nuclear Corporation and Subsidiaries
Notes to the Consolidated Financial Statements
Note 11: VARIABLE INTEREST ENTITIES
Accounting guidance requires that a variable interest entity, according to the provisions of ASC Topic 810, Consolidation, must be consolidated by the primary beneficiary. The primary beneficiary is the party that has both the power to direct activities of a VIE that most significantly impact the entitys economic performance and the obligation to absorb losses of the entity or the right to receive benefits from the entity that could potentially be significant to the VIE. We perform ongoing qualitative analysis to determine if we are the primary beneficiary of a VIE.
In 2015, the Company entered into four building lease agreements with the Affiliates that are owned by shareholders of the Company. The non-cancellable leases allow us to use manufacturing warehouse and office space for a term of up to 40 years. Under the lease agreements, the Company absorbs all variability created by the buildings through maintenance terms and provides capital for all expenditures related to material leasehold improvements, and prior approval from the Affiliates is not needed for such improvements. The Company determined that these common control lease arrangements represent VIEs under ASC 810. Accordingly, the Company is the primary beneficiary of the Affiliates and consolidates the Affiliates.
As of December 18, 2020 and for the period from January 1, 2020 through December 18, 2020, intercompany balances and activities between the Company and the Affiliates have been eliminated. The following assets and liabilities have been recorded in the consolidated balance sheet because the company is the primary beneficiary of the VIEs (in thousands):
| As of December 18, 2020 | ||||
| Cash and cash equivalents |
$ | 254 | ||
| Property and equipment, net |
5,460 | |||
|
|
|
|||
| Total assets |
5,714 | |||
|
|
|
|||
| Accrued expenses and other current liabilities |
$ | 109 | ||
| Notes payable |
146 | |||
|
|
|
|||
| Total liabilities |
255 | |||
|
|
|
|||
| Net assets |
$ | 5,459 | ||
|
|
|
|||
The assets of the Affiliates may only be used to settle obligations of the Affiliates and may not be used for other consolidated entities. The liabilities of the Affiliates are non-recourse to the general credit of the Companys other consolidated entities.
Note 12: ROYALTY AGREEMENTS
The Company is obligated for royalty payments to unrelated companies for exclusive use of certain patents owned by the unrelated entities. Payments under these agreements are based upon the net sales of all products to which these patents relate. Royalties are paid quarterly and are due 30 days after the end of the quarter. Royalties due as of December 18, 2020 were $0.2 million, which is recorded in royalty payable. Royalty expenses, which are included in Cost of products on the consolidated statement of operations and comprehensive income, were $0.4 million for the period from January 1, 2020 through December 18, 2020.
Note 13: BENEFIT PLANS
Beginning on January 1, 2008, the parent company added a 401(k) feature with safe harbor matching contributions to its already existing profit-sharing plan (the Plan). Employees who are at least 21 years of age
F-128
Table of Contents
Sun Nuclear Corporation and Subsidiaries
Notes to the Consolidated Financial Statements
with at least one year of service are eligible for the Plan. The Company recorded an accrued liability of $0.1 million as a safe harbor matching contribution as of December 18, 2020.
Contributions to the profit-sharing plan are at the discretion of the Board of Directors. The Company made no profit-sharing contributions to the Plan for the period from January 1, 2020 through December 18, 2020.
The subsidiary company, Gammex, Inc., maintains a profit-sharing plan covering substantially all employees. The Plan provides for additional discretionary contributions by the Company and voluntary contributions by the participants, through salary reduction, up to specified limits. For the period from January 1, 2020 through December 18, 2020, Gammex did not make any matching or discretionary contributions.
Note 14: COMMITMENTS AND CONTINGENCIES
Litigation
The Company is subject to various legal proceedings, claims, litigation, investigations and contingencies arising out of the ordinary course of business. If the potential loss from any claim, asserted or unasserted, or legal proceeding is considered probable and the amount can be reasonably estimated or a range of loss can be determined, the Company accrues a liability in accordance with accounting guidance for contingencies. These accruals represent managements best estimate of probable loss. Disclosure is also provided when it is reasonably possible that a loss will be incurred or when it is reasonably possible that the amount of a loss will exceed the recorded provision. We consult with legal counsel on matters related to litigation and seek input both within and outside the Company. The Company believes the outcome of pending legal proceedings, individually and in the aggregate, will not have a material impact on the Companys consolidated financial statements.
Product Warranty
The Company calculates an estimate for the warranty liability based on historical trend information. The following table reflects the changes in the Companys accrued product warranty (in thousands):
| Period from January 1, 2020 through December 18, 2020 | ||||
| Accrued product warranty, at beginning of period |
$ | 396 | ||
| Accruals for warranties issued during the year |
156 | |||
| Settlement of warranty claims |
(100 | ) | ||
|
|
|
|||
| Accrued product warranty, at end of period |
$ | 452 | ||
|
|
|
|||
The accrued product warranty is included in Accrued expenses and other current liabilities on the consolidated balance sheet.
F-129
Table of Contents
Sun Nuclear Corporation and Subsidiaries
Notes to the Consolidated Financial Statements
Note 15: SUBSEQUENT EVENTS
The Company evaluated subsequent events for recognition or disclosure through April 30, 2021, the date the consolidated financial statements were available to be issued. There was no subsequent event that would require disclosure to or adjustment to the financial statements.
F-130
Table of Contents
Table of Contents
TABLE OF CONTENTS
| Page | ||||||
| ARTICLE 1 |
|
|||||
| Definitions |
|
|||||
| Section 1.01. |
Definitions | A-3 | ||||
| Section 1.02. |
Other Definitional and Interpretative Provisions | A-17 | ||||
| Section 1.03. |
Equitable Adjustments | A-18 | ||||
| ARTICLE 2 |
|
|||||
| Business Combination |
|
|||||
| Section 2.01. |
Company Articles Amendment | A-18 | ||||
| Section 2.02. |
Election Procedures | A-18 | ||||
| Section 2.03. |
Closing | A-20 | ||||
| Section 2.04. |
Closing Deliverables | A-20 | ||||
| Section 2.05. |
Closing Transactions | A-22 | ||||
| Section 2.06. |
SPAC Closing Statement | A-23 | ||||
| Section 2.07. |
Company Closing Statement | A-23 | ||||
| Section 2.08. |
Exchange Procedures | A-24 | ||||
| ARTICLE 3 |
|
|||||
| Representations and Warranties of Sellers |
|
|||||
| Section 3.01. |
Corporate Existence and Power | A-25 | ||||
| Section 3.02. |
Seller Authorization | A-25 | ||||
| Section 3.03. |
Governmental Authorization | A-25 | ||||
| Section 3.04. |
Noncontravention | A-25 | ||||
| Section 3.05. |
Ownership and Transfer of Existing Company Shares | A-25 | ||||
| Section 3.06. |
Information Supplied | A-26 | ||||
| Section 3.07. |
Finders Fees | A-26 | ||||
| Section 3.08. |
No Other Representations | A-26 | ||||
| ARTICLE 4 |
|
|||||
| Representations and Warranties of the Company |
|
|||||
| Section 4.01. |
Existence and Power | A-26 | ||||
| Section 4.02. |
Authorization | A-26 | ||||
| Section 4.03. |
Governmental Authorization | A-27 | ||||
| Section 4.04. |
Noncontravention | A-27 | ||||
| Section 4.05. |
Capitalization | A-27 | ||||
| Section 4.06. |
Subsidiaries | A-28 | ||||
| Section 4.07. |
Information Supplied | A-28 | ||||
| Section 4.08. |
Financial Statements | A-29 | ||||
| Section 4.09. |
Absence of Certain Changes | A-29 | ||||
| Section 4.10. |
No Undisclosed Material Liabilities | A-30 | ||||
| Section 4.11. |
Material Contracts | A-30 | ||||
| Section 4.12. |
Litigation | A-31 | ||||
| Section 4.13. |
Compliance with Laws and Court Orders | A-32 | ||||
| Section 4.14. |
Real Property | A-32 | ||||
| Section 4.15. |
Intellectual Property | A-33 | ||||
| Section 4.16. |
Insurance Coverage | A-35 | ||||
| Section 4.17. |
Employees | A-35 | ||||
| Section 4.18. |
Employee Benefit Plans | A-37 | ||||
i
Table of Contents
| Section 4.19. |
Taxes | A-39 | ||||
| Section 4.20. |
Environmental Matters | A-41 | ||||
| Section 4.21. |
Affiliate Transactions | A-42 | ||||
| Section 4.22. |
Finders Fees | A-42 | ||||
| Section 4.23. |
Anti-Bribery; Anti-Corruption | A-42 | ||||
| Section 4.24. |
International Trade; Sanctions | A-43 | ||||
| Section 4.25. |
Data Privacy | A-43 | ||||
| Section 4.26. |
Customers and Suppliers | A-44 | ||||
| Section 4.27. |
Product Liabilities and Recalls | A-44 | ||||
| Section 4.28. |
No Other Representations | A-45 | ||||
| Section 4.29. |
PPP Loans | A-45 | ||||
| Section 4.30. |
Debt Financing | A-45 | ||||
| Section 4.31. |
Exclusivity of Representations and Warranties | A-46 | ||||
| ARTICLE 5 | ||||||
| Representations and Warranties of the SPAC | ||||||
| Section 5.01. |
Corporate Existence and Power | A-46 | ||||
| Section 5.02. |
Authorization | A-46 | ||||
| Section 5.03. |
Governmental Authorization | A-47 | ||||
| Section 5.04. |
Noncontravention | A-47 | ||||
| Section 5.05. |
Capital Structure | A-47 | ||||
| Section 5.06. |
SEC Documents; Controls | A-48 | ||||
| Section 5.07. |
Listing | A-49 | ||||
| Section 5.08. |
The Registration Statement and the Proxy Statement | A-49 | ||||
| Section 5.09. |
Absence of Certain Changes | A-49 | ||||
| Section 5.10. |
Litigation | A-50 | ||||
| Section 5.11. |
Compliance with Applicable Law | A-50 | ||||
| Section 5.12. |
Taxes | A-50 | ||||
| Section 5.13. |
Employees and Employee Benefits Plans | A-51 | ||||
| Section 5.14. |
Affiliate Transactions | A-51 | ||||
| Section 5.15. |
Properties | A-52 | ||||
| Section 5.16. |
Contracts | A-52 | ||||
| Section 5.17. |
Finders Fees | A-52 | ||||
| Section 5.18. |
Financial Ability | A-52 | ||||
| Section 5.19. |
PIPE Financing; Backstop Agreement | A-52 | ||||
| Section 5.20. |
Trust Account | A-53 | ||||
| Section 5.21. |
No Other Representations | A-54 | ||||
| Section 5.22. |
Exclusivity of Representations and Warranties | A-54 | ||||
| ARTICLE 6 | ||||||
| Covenants of the Company | ||||||
| Section 6.01. |
Conduct of Business | A-55 | ||||
| Section 6.02. |
Inspection | A-58 | ||||
| Section 6.03. |
No Claim Against the Trust Account |
A-58 | ||||
| Section 6.04. |
Pre-Closing Actions by the Company |
A-58 | ||||
| Section 6.05. |
Drag Along |
A-59 | ||||
| Section 6.06. |
Termination of Affiliate Transactions |
A-60 | ||||
| Section 6.07. |
Title Insurance Cooperation |
A-60 | ||||
| Section 6.08. |
No SPAC Securities Transactions |
A-60 | ||||
| Section 6.09. |
Repayment of Employee Loans |
A-60 | ||||
| Section 6.10. |
Debt Financing |
A-60 | ||||
| Section 6.11. |
PPP Loans |
A-62 | ||||
ii
Table of Contents
| ARTICLE 7 | ||||||
| Covenants of the SPAC | ||||||
| Section 7.01. |
Conduct of Business During the Interim Period | A-62 | ||||
| Section 7.02. |
SPAC Board of Directors | A-63 | ||||
| Section 7.03. |
Governing Documents |
A-63 | ||||
| Section 7.04. |
Registration Rights Agreement |
A-63 | ||||
| Section 7.05. |
NYSE Listing |
A-63 | ||||
| Section 7.06. |
SPAC Public Filings |
A-63 | ||||
| Section 7.07. |
PIPE Subscription Agreements |
A-63 | ||||
| Section 7.08. |
Backstop Agreement |
A-64 | ||||
| Section 7.09. |
Trust Account |
A-64 | ||||
| Section 7.10. |
Financing Cooperation; Alternative Financing |
A-64 | ||||
| ARTICLE 8 | ||||||
| Joint Covenants | ||||||
| Section 8.01. |
Best Efforts; Further Assurances | A-64 | ||||
| Section 8.02. |
Certain Filings | A-65 | ||||
| Section 8.03. |
Indemnification and Insurance |
A-65 | ||||
| Section 8.04. |
Registration Statement; Proxy Statement; SPAC Special Meeting |
A-66 | ||||
| Section 8.05. |
Form 8-K Filings |
A-68 | ||||
| Section 8.06. |
Public Announcements |
A-69 | ||||
| Section 8.07. |
Notification of Certain Matters |
A-69 | ||||
| Section 8.08. |
Exclusivity |
A-69 | ||||
| Section 8.09. |
Alternative Financing |
A-70 | ||||
| ARTICLE 9 | ||||||
| Tax Matters | ||||||
| Section 9.01. |
Transfer Taxes | A-70 | ||||
| Section 9.02. |
Tax Returns | A-71 | ||||
| Section 9.03. |
Tax Contest |
A-71 | ||||
| Section 9.04. |
Cooperation |
A-71 | ||||
| Section 9.05. |
Straddle Periods |
A-71 | ||||
| Section 9.06. |
Post-Closing Actions |
A-71 | ||||
| Section 9.07. |
Election |
A-72 | ||||
| Section 9.08. |
Withholding |
A-72 | ||||
| Section 9.09. |
Partnership Audits |
A-72 | ||||
| ARTICLE 10 | ||||||
| Employee Benefits | ||||||
| Section 10.01. |
Equity Incentive Plan | A-72 | ||||
| Section 10.02. |
[Reserved.] | A-72 | ||||
| Section 10.03. |
280G Approval |
A-72 | ||||
| ARTICLE 11 | ||||||
| Conditions to Closing | ||||||
| Section 11.01. |
Conditions to Obligations of All Parties | A-73 | ||||
| Section 11.02. |
Additional Conditions to Obligation of the SPAC | A-73 | ||||
| Section 11.03. |
Additional Conditions to Obligation of the Company and the Sellers |
A-74 | ||||
iii
Table of Contents
| ARTICLE 12 | ||||||
| Termination | ||||||
| Section 12.01. |
Grounds for Termination | A-75 | ||||
| Section 12.02. |
Effect of Termination | A-76 | ||||
| ARTICLE 13 | ||||||
| Miscellaneous | ||||||
| Section 13.01. |
Non-Survival of Representations, Warranties and Covenants | A-76 | ||||
| Section 13.02. |
Non-Recourse | A-77 | ||||
| Section 13.03. |
Notices |
A-77 | ||||
| Section 13.04. |
Amendments and Waivers |
A-78 | ||||
| Section 13.05. |
Expenses |
A-78 | ||||
| Section 13.06. |
Successors and Assigns |
A-78 | ||||
| Section 13.07. |
Governing Law |
A-79 | ||||
| Section 13.08. |
Jurisdiction |
A-79 | ||||
| Section 13.09. |
WAIVER OF JURY TRIAL |
A-79 | ||||
| Section 13.10. |
Counterparts; Effectiveness; Third Party Beneficiaries |
A-79 | ||||
| Section 13.11. |
Entire Agreement |
A-79 | ||||
| Section 13.12. |
Severability |
A-80 | ||||
| Section 13.13. |
Disclosure Schedules |
A-80 | ||||
| Section 13.14. |
Specific Performance |
A-80 | ||||
| Section 13.15. |
Debt Financing Sources |
A-80 | ||||
ANNEXES
Annex I Supporting Company Holders
Annex II Management Sellers
EXHIBITS
Exhibit A Form of New SPAC Certificate of Incorporation
Exhibit B Form of New SPAC Bylaws
Exhibit C Form of Intermediate TopCo Certificate of Incorporation
Exhibit D Form of Intermediate TopCo Bylaws
Exhibit E Form of Amended & Restated Registration Rights Agreement
Exhibit F Equity Incentive Plan
Exhibit G Election Agreement
Exhibit H Joinder Agreement
Exhibit I Pre-Closing Step Plan
Exhibit J Closing Step Plan
Exhibit K Drag Along Notice
Exhibit L Backstop Agreement
Exhibit M Option Agreement
Exhibit N Charterhouse Director Designation Agreement
Exhibit O SPAC Sponsor Director Designation Agreement
Exhibit P Company Articles Amendment
iv
Table of Contents
BUSINESS COMBINATION AGREEMENT
This Business Combination Agreement (this Agreement), dated as of June 17, 2021, by and among GS Acquisition Holdings Corp II, a Delaware corporation (the SPAC), Mirion Technologies (TopCo), Ltd., a Jersey private company limited by shares (the Company), for the limited purpose set forth herein, CCP IX LP No. 1, CCP IX LP No. 2, CCP IX Co-Investment LP and CCP IX Co-Investment No. 2 LP (collectively, the Charterhouse Parties), each acting by their general partner, Charterhouse General Partners (IX) Limited, for the limited purpose set forth herein, each of the other Persons set forth on Annex I hereto (together with the Charterhouse Parties, the Supporting Company Holders) and, for the limited purpose set forth herein, the other holders of Existing Company Shares from time to time becoming a party hereto by executing a Joinder Agreement in the form of Exhibit H hereto (each, a Joining Seller and collectively, the Joining Sellers and, together with each Supporting Company Holder, each, a Seller, and collectively, the Sellers, and together with the SPAC and the Company, the Parties).
RECITALS
WHEREAS, the SPAC is a blank check company incorporated in Delaware and formed to acquire one or more operating businesses through a Business Combination;
WHEREAS, on the terms and subject to the conditions of this Agreement and in accordance with Applicable Law, the Parties intend to enter into a Business Combination pursuant to and in accordance with this Agreement and which shall be accomplished in accordance with the Closing Step Plan attached hereto as Exhibit J (the Closing Step Plan);
WHEREAS, prior to the Closing, the Company intends to effect the transactions set forth in the Pre-Closing Step Plan attached hereto as Exhibit I (the Pre-Closing Step Plan);
WHEREAS, as of the date hereof, the Supporting Company Holders collectively own the issued and outstanding Existing Company Shares and outstanding Loan Notes set forth on Annex I hereto and desire to sell such Existing Company Shares and Loan Notes to the SPAC, and the SPAC desires to purchase such Existing Company Shares and Loan Notes from such Supporting Company Holders, upon the terms and subject to the conditions hereinafter set forth;
WHEREAS, each of the Joining Sellers who have not duly executed and delivered a signature page to this Agreement as of the date hereof, shall, in accordance with such Persons drag-along obligations as set forth in the Existing Company Articles, as amended by the Company Articles Amendment, duly execute and deliver to SPAC a Joinder Agreement in the form of Exhibit H hereto pursuant to which such Person shall become bound by the terms and conditions of this Agreement as a Seller;
WHEREAS, the respective boards of directors or equivalent governing bodies of each of the SPAC and the Company have unanimously approved and declared advisable the transactions contemplated by this Agreement upon the terms and subject to the conditions of this Agreement and in accordance with the DGCL and the Companies (Jersey) Law 1991, as applicable;
WHEREAS, prior to the Closing and immediately prior to the closing of the PIPE Financing, the SPAC shall, subject to obtaining the approval by the Requisite Existing SPAC Stockholders of the Transaction Proposals, file a certificate of incorporation (the New SPAC Certificate of Incorporation) with the Secretary of State of the State of Delaware substantially in the form attached hereto as Exhibit A and will adopt bylaws (the New SPAC Bylaws) substantially in the form attached hereto as Exhibit B, which provide, among other things, that (i) the SPAC will have two classes of common stock, New SPAC Class A Common Shares and New SPAC Class B Common Shares and (ii) the Board of Directors of the SPAC will be composed of up to nine (9) directors;
A-1
Table of Contents
WHEREAS, in connection with the Closing, and in accordance with the Closing Step Plan, the Company will be merged (the Up-C Merger) with and into a Jersey private company limited by shares (the Jersey Merger Sub) to be formed following the execution of this Agreement and wholly owned by a Delaware corporation to be formed following the date of this Agreement and wholly owned by the SPAC (Intermediate TopCo), and which shall have adopted and filed with the Secretary of State of the State of Delaware a certificate of incorporation (the Intermediate TopCo Certificate of Incorporation) substantially in the form attached hereto as Exhibit C and will have adopted bylaws (the Intermediate TopCo Bylaws) substantially in the form attached hereto as Exhibit D, which provide, among other things, that Intermediate TopCo will have two classes of common stock, New Company Class A Common Stock and New Company Class B Common Stock, pursuant to which merger (i) the separate existence of Jersey Merger Sub shall cease and the Company shall be the surviving corporation in the Up-C Merger, (ii) the Company shall become a wholly owned subsidiary of Intermediate TopCo, and (iii) the outstanding shares of the Company at the time of the Up-C Merger shall be converted into, (x) in the case of the shares of the Company owned by the SPAC following the previous steps taken as part of the Closing Step Plan, shares of New Company Class A Common Stock, and (y) in the case of shares of the Company held by Sellers that have made or have been deemed to have made a Unit Election for Shares, Units consisting of shares of New Company Class B Common Stock and New SPAC Class B Common Shares;
WHEREAS, as a condition to and as an inducement to the Companys willingness to enter into this Agreement, concurrently with the execution and delivery of this Agreement, the Sponsor (as defined below) and GS Acquisition Holdings II Employee Participation LLC have entered into a Sponsor Support Agreement (the Sponsor Support Agreement) pursuant to which each has agreed to (i) waive any adjustment to the conversion ratio set forth in the Existing SPAC Certificate of Incorporation resulting from the transaction contemplated by the PIPE Financing, and (ii) subject 18,750,000 shares in the aggregate of Existing SPAC Common Stock held by them to certain performance-based vesting conditions as set forth in the Sponsor Support Agreement, which vesting conditions shall become effective concurrently with the Closing;
WHEREAS, concurrently with the execution and delivery of this Agreement, Mirion Technologies (HoldingSub2) Ltd., a limited liability company incorporated in England and Wales with a company number 09299632 (the DCL Beneficiary), has entered into and delivered to the SPAC a Debt Commitment Letter providing for the Debt Financing;
WHEREAS, concurrently with the execution and delivery of this Agreement, the PIPE Investors (as defined below) and the SPAC have entered into subscription agreements (the PIPE Subscription Agreements) pursuant to which the PIPE Investors have agreed to subscribe for and purchase from the SPAC an aggregate of 90,000,000 New SPAC Class A Common Shares at a price per share equal to at least $10.00 immediately prior to (and contingent upon) the Closing (the PIPE Financing, and such aggregate amount, the PIPE Financing Amount);
WHEREAS, concurrently with the Closing, the SPAC will cause the Registration Rights Agreement, dated June 29, 2020, by and among the SPAC, the Sponsor, the Charterhouse Parties and the other parties listed on the signature pages thereto, to be amended and restated in the form of the Amended and Restated Registration Rights Agreement attached as Exhibit E hereto (the Registration Rights Agreement);
WHEREAS, concurrently with the execution and delivery of this Agreement, GSAM Holdings LLC, a Delaware limited liability company (the Backstop Party), and the SPAC have entered into a Backstop Agreement attached as Exhibit L hereto (the Backstop Agreement) pursuant to which Backstop Party has committed to purchase from the SPAC up to 12,500,000 New SPAC Class A Common Shares at a price per share equal to $10.00 immediately prior to (and contingent upon) the Closing, contingent upon the terms and subject to the conditions set forth therein;
WHEREAS, at the Closing, Backstop Party and the Sellers that have made a Cash Election for Shares shall enter into an Option Agreement in the form attached as Exhibit M hereto (the Option Agreement) pursuant to
A-2
Table of Contents
which such Sellers shall agree to, at the option of Backstop Party and subject to the conditions set forth therein, sell to Backstop Party up to 12,500,000 New SPAC Class A Common Shares to be received by such Sellers at the Closing at a price per share equal to $10.00 in cash, on the terms and subject to the conditions set forth therein (the Call Option);
WHEREAS, concurrently with the Closing, the SPAC will enter into the Director Designation Agreement with the Charterhouse Parties, in the form attached as Exhibit N hereto (the Charterhouse Director Designation Agreement);
WHEREAS, concurrently with the Closing, the SPAC will enter into the Director Designation Agreement with the Sponsor, in the form attached as Exhibit O hereto (the SPAC Sponsor Director Designation Agreement); and
NOW, THEREFORE, in consideration of the foregoing and the respective representations, warranties, covenants and agreements set forth in this Agreement, and intending to be legally bound hereby, the Parties hereto agree as follows:
ARTICLE 1
DEFINITIONS
Section 1.01. Definitions. (a) As used herein, the following terms shall have the following meanings:
A Ordinary Shares means the A Ordinary Shares of $0.01 each in the capital of the Company, having the rights set out in the Existing Company Articles.
Action means any claim, action, audit, litigation, suit, assessment, arbitration, mediation or inquiry, or any other proceeding or investigation (whether at law or in equity), in each case that is by or before any Governmental Authority.
Affiliate means, with respect to any Person, any other Person directly or indirectly controlling, controlled by or under common control with such Person; provided that (i) neither the Company nor any Subsidiary shall be considered an Affiliate of any Seller; (ii) no portfolio company of the Charterhouse Parties or any other Seller or any of their respective Affiliates shall be considered an Affiliate of any Sellers; and (iii) in no event shall the SPAC be considered an Affiliate of The Goldman Sachs Group, Inc. or of any investment fund affiliated with The Goldman Sachs Group, Inc., nor shall any portfolio company of any investment fund affiliated with The Goldman Sachs Group, Inc. be considered to be an Affiliate of the SPAC. For purposes of this definition, control (including with correlative meanings, the terms controlling, controlled by and under common control with), as applied to any Person, means the possession, directly or indirectly, of the power to direct or cause the direction of the management and policies of such Person, whether through the ownership of voting securities, by contract or otherwise.
Affiliated Group means a group of Persons that elects, is required to, or otherwise files a Tax Return or pays a Tax as an affiliated, consolidated, combined, unitary or other group recognized by applicable Tax Law.
Applicable Law means, with respect to any Person, any law (statutory, common or otherwise), constitution, treaty, convention, ordinance, code, rule, regulation, order, legislation, injunction, judgment, decree, ruling, directive, determination or other similar requirement of a Governmental Authority that is binding upon or applicable to such Person.
Available Closing Cash means, as of the Closing, (i) the amount of funds contained in the Trust Account (after reduction for the aggregate amount of payments required to be made in connection with the SPAC
A-3
Table of Contents
Stockholder Redemption), plus (ii) the PIPE Financing Amount, plus (iii) the Debt Financing Proceeds, plus (iv) the Closing Balance Sheet Cash, plus (v) the Backstop Amount Proceeds (if any), less (vi) the Debt Payoff Amount, less (vii) Transaction Expenses (other than Seller-Borne Transaction Expenses), less (viii) the Primary Capital.
Backstop Amount means up to $125,000,000 in proceeds from the sale by the SPAC to the Backstop Party of up to 12,500,000 New SPAC Class A Common Shares at a price per share equal to $10.00 immediately prior to (and contingent upon) the Closing, contingent upon the terms of and subject to the conditions set forth in the Backstop Agreement.
Backstop Amount Proceeds means the actual amount in proceeds from the sale by the SPAC to the Backstop Party of New SPAC Class A Common Shares at a price per share equal to $10.00 immediately prior to (and contingent upon) the Closing, pursuant to the Backstop Agreement.
B Ordinary Shares means the B Ordinary Shares of $0.01 each in the capital of the Company, having the rights set out in the Existing Company Articles.
Borrowed Indebtedness shall mean, as of the applicable date of determination, the aggregate principal amount of outstanding Indebtedness of the Company and its Subsidiaries under the Credit Agreement.
Business Combination has the meaning ascribed to such term in the Existing SPAC Certificate of Incorporation.
Business Day means a day, other than Saturday, Sunday or other day on which commercial banks in New York, New York, the City of London or Jersey are authorized or required by Applicable Law to close.
Called Shareholders has the meaning given to it in the Existing Company Articles.
Cash Consideration means $1,310,000,000; provided, however, if the Minimum Cash Condition is not satisfied, and the Company and the Charterhouse Parties elect to waive the Minimum Cash Condition within fifteen (15) Business Days of receipt by the Company and the Charterhouse Parties of a written notice from the SPAC that the Minimum Cash Condition has not been satisfied and is incapable of being satisfied, Cash Consideration shall mean $1,310,000,000 less the Cash Shortfall.
Cash Shortfall means the amount by which $1,310,000,000 exceeds the Available Closing Cash.
CARES Act means the Coronavirus Aid, Relief, and Economic Security Act of 2020 (as may be amended or modified).
Closing Balance Sheet Cash means cash and cash equivalents of the Company and its subsidiaries on a consolidated basis as of the Closing Date, as estimated in good faith by management two (2) Business Days prior to the Closing Date and set forth on the Company Closing Statement.
Code means the United States Internal Revenue Code of 1986, as amended.
Confidentiality Agreement means that certain Amended and Restated Confidentiality Agreement, dated as of April 13, 2021, by and between Mirion Technologies, Inc. and the SPAC.
Contract means any contract, subcontract, agreement, indenture, note, bond, loan or credit agreement, instrument, installment obligation, lease, mortgage, deed of trust, license, sublicense, commitment, power of attorney, guaranty or other legally binding commitment, arrangement, understanding or obligation, whether written or oral, in each case, as amended and supplemented from time to time and including all schedules, annexes and exhibits thereto.
A-4
Table of Contents
COVID-19 means SARS-CoV-2 or COVID-19, and any evolutions thereof or any other epidemics, pandemics or disease outbreaks.
COVID-19 Measures means any quarantine, shelter in place, stay at home, workforce reduction, social distancing, shut down, closure, sequester or any other Applicable Law, order, Action, directive, guidelines or recommendations by any Governmental Authority in connection with or in response to COVID-19.
Credit Agreement means that certain Credit Agreement, dated as of March 8, 2019, by and among Mirion Technologies (HoldingRep), Ltd., Mirion technologies (Global), Ltd., Mirion Technologies (USA), LLC, Mirion Technologies (Luxembourg) S.A.R.L., Mirion Technologies, Inc., the other borrowers party thereto, the several Lenders party thereto from time to time, Morgan Stanley Senior Funding, Inc., as the Administrative Agent and Collateral Agent, Morgan Stanley Senior Funding, Inc. and Goldman Sachs Bank USA, as Joint Lead Arrangers and Global Bookrunners, and JPMorgan Chase Bank, N.A. and HSBC Securities (USA) Inc., as Bookrunners, as amended on July 8, 2019, December 16, 2019 and December 18, 2020.
Customs & International Trade Authorizations shall mean any and all import and export licenses, license exceptions, consents, orders, authorizations, notices, waivers, notification requirements, registrations and approvals required pursuant to the Customs & International Trade Laws for the lawful export, deemed export, re-export, deemed re-export transfer or import of goods, software, technology, technical data and services.
Customs & International Trade Laws shall mean the applicable import, customs and trade, export and anti-boycott laws of any jurisdiction in which the Company or any of its Subsidiaries is incorporated or does business, including, but not limited to: (i) the laws, regulations, and programs administered or enforced by U.S. Customs and Border Protection, U.S. Immigration and Customs Enforcement, the U.S. Department of Commerce (International Trade Administration), the U.S. International Trade Commission, the U.S. Department of Commerce (Bureau of Industry and Security), the U.S. Department of State (Directorate of Defense Trade Controls) and their predecessor agencies; (ii) the Tariff Act of 1930, as amended (iii) the Export Administration Act of 1979, as amended; (iv) the Export Control Reform Act of 2018; (v) the Export Administration Regulations, including related restrictions with regard to transactions involving Persons on the U.S. Department of Commerce Denied Persons List, Unverified List or Entity List; (vi) the Arms Export Control Act, as amended; (vii) the International Traffic in Arms Regulations, including related restrictions with regard to transactions involving Persons on the Debarred List; (viii) the Foreign Trade Regulations pursuant to 15 C.F.R. Part 30; (ix) the anti-boycott laws and regulations administered by the U.S. Department of Commerce; and (x) the anti-boycott laws and regulations administered by the U.S. Department of the Treasury.
Debt Financing Proceeds means the amounts delivered pursuant to the Debt Financing.
Debt Financing Sources means the agents, arrangers, lenders and other entities that have committed to provide the Debt Financing, and the parties to the Debt Commitment Letter, joinder agreements, credit agreements or indentures related to the Debt Financing, together with their respective Affiliates and their and their respective Affiliates current or future general or limited partners, stockholders, managers, members, agents, officers, directors, employees, advisors, partners, members, managers, controlling persons and representatives and their respective successors and assigns.
DGCL means the Delaware General Corporation Law.
Drag Along Notice has the meaning given to it in the Existing Company Articles and, for the purposes of this Agreement, shall mean such a notice given with respect to the Transactions attached hereto as Exhibit K.
Employee Plan means any employee benefit plan, as defined in Section 3(3) of ERISA and any other employment, severance or similar contract, plan or policy and each other arrangement providing for compensation, bonuses, profit-sharing, equity-related rights or other forms of incentive or deferred
A-5
Table of Contents
compensation, vacation benefits, insurance (including any self-insured arrangements), health or medical benefits, employee assistance, disability or sick leave benefits, severance benefits, termination protection, change in control, retention, and post-employment or retirement benefits (including compensation, pension, health, medical or life insurance benefits), whether or not subject to ERISA, which the Company or any of its Subsidiaries sponsors or maintains for the benefit of its current or former employees, individuals who provide services and are compensated as individual independent contractors or directors, or with respect to which the Company or any Subsidiary has any direct or indirect current or contingent liability. For the avoidance of doubt, Employee Plans shall not include (i) any plan that is mandated by a Governmental Authority or by Applicable Law that is sponsored or maintained by a Governmental Authority (a Governmental Plan) and (ii) any multiemployer plan, as defined in Section 3(37) of ERISA.
Environmental Laws means any and all Applicable Law, in each case as in effect on or before the Closing Date, that relate to the protection of the environment, natural resources or human health and safety as it relates to the exposure to hazardous materials, substances or wastes, or the handling, use, presence, disposal, release or threatened release of any hazardous materials, substances or wastes (including any by- product of an industrial process and anything that is discarded, disposed of, spoiled, abandoned, unwanted or surplus irrespective of whether it is capable of being recovered or recycled or has any value); or radiation, wetlands, pollution, contamination or any injury or threat of injury to persons or property or the environment and shall include Applicable Law regulating Hazardous Materials in products manufactured or sold and associated labeling or packaging content requirements or restrictions relating to environmental attributes or as respects product takeback or end-of-life requirements.
Enforceability Exceptions means applicable bankruptcy, insolvency, fraudulent conveyance, reorganization, moratorium and similar Applicable Law affecting or relating to creditors rights generally and subject, as to enforceability, to general principles of equity, whether such enforceability is considered in a proceeding in equity or at law.
ERISA means the Employee Retirement Income Security Act of 1974, as amended, and the rules and regulations promulgated thereunder.
ERISA Affiliate means any other entity which, together with the Company, would be treated as a single employer under Section 414 of the Code.
Exchange Act means the Securities Exchange Act of 1934, as amended, and the rules and regulations promulgated thereunder.
Executive Loans means, collectively, the agreements between the Company and certain of its officers set forth on Section 1.01(a)(i) of the Company Disclosure Schedule.
Existing Company Articles means the Companys Memorandum and Articles of Association, as in effect on the date hereof, and following the effectiveness of the Company Articles Amendment, as so amended and restated.
Existing Company Shares means, collectively, the A Ordinary Shares and the B Ordinary Shares.
Existing Investment Agreements means (i) the Investment Agreement relating to the Company dated November 18, 2014 as amended by a deed of amendment dated November 17, 2016 and (ii) the amended and restated Co-Investment Agreement relating to the Company dated June 17, 2016 as amended by a deed of amendment dated March 3, 2017.
Existing SPAC Bylaws means the Bylaws of the SPAC.
A-6
Table of Contents
Existing SPAC Certificate of Incorporation means the Amended and Restated Certificate of Incorporation of the SPAC, filed with the Secretary of State of the State of Delaware on June 29, 2020, as amended and in effect on the date hereof.
Existing SPAC Common Stock means, collectively, the shares of Class A common stock of the SPAC, par value $0.0001 per share, and the shares of Class B common stock of the SPAC, par value $0.0001 per share, in each case as set forth in the Existing SPAC Certificate of Incorporation.
Existing SPAC Investors means the holders, as of the date of this Agreement, of shares of Class A common stock, par value $0.001 per share, of the SPAC.
Existing SPAC Stockholders means all holders, as of the date of this Agreement, of Existing SPAC Common Stock (including, for the avoidance of doubt, the Existing SPAC Investors).
Exit Bonuses means, collectively, the bonus amounts due to certain individuals upon and subject to the Closing as set forth on Section 2.04(e) of the Company Disclosure Schedule.
Flow-Through Tax Return means any Tax Return of any entity for which (for purposes of any Tax) income of such entity flows through such entity to such entitys member(s) (or other beneficial owner(s) for Tax purposes).
Foreign Employee Plan means an Employee Plan, irrespective of whether governed by ERISA or by any other national Applicable Law, including, for the avoidance of doubt, all comparable employment, severance or similar contracts, plans or policies and other arrangements of Subsidiaries not incorporated in the United States of America or Canada.
Fraud means actual and intentional common law fraud committed by a Party hereto. Under no circumstances shall fraud include any equitable fraud, constructive fraud, negligent misrepresentation, unfair dealings or any other fraud or torts based on recklessness or negligence.
GAAP means United States generally accepted accounting principles in effect from time to time.
Governmental Authority means any supra-national, transnational, or domestic or foreign federal, state, municipal, provincial, regional or local, governmental, administrative or regulatory authority, commission, department, bureau, court, arbitral body, tribunal, agency, instrumentality or official, including any political subdivision thereof, including, for the avoidance of doubt, any Regulatory Consent Authority.
HSR Act means the Hart-Scott-Rodino Antitrust Improvements Act of 1976, as amended, and the rules and regulations promulgated thereunder.
Hazardous Materials means any material, substance or waste classified, characterized, regulated or otherwise defined as hazardous, toxic, radioactive, or a pollutant, contaminant, or words of similar meaning or regulatory effect by, under or pursuant to any Environmental Laws, and shall include petroleum, petroleum by-products, asbestos or asbestos-containing material, polychlorinated biphenyls, per- and polyfluoroalkyl substances flammable or explosive substances, or pesticides.
Indebtedness shall mean all of the following: (a) any indebtedness for borrowed money including the Borrowed Indebtedness and any premiums, fees and expenses related to the paydown of any Borrowed Indebtedness concurrently with the Closing; (b) any obligations evidenced by bonds, debentures, notes or other similar instruments; (c) any obligations to pay the deferred purchase price of property, stock or services including any earn-out payments; (d) any obligations as lessee under capitalized leases; (e) any obligations, contingent or otherwise, under acceptance, letters of credit or similar facilities to the extent drawn; (f) any guaranty of any of
A-7
Table of Contents
the foregoing; (g) any accrued interest, fees and charges in respect of any of the foregoing; and (h) any prepayment premiums and penalties actually due and payable, and any other fees, expenses, indemnities and other amounts actually payable as a result of the prepayment or discharge of any of the foregoing.
Intellectual Property Rights means all rights, title and interest in or relating to intellectual property throughout the world, whether registered or unregistered, including: (i) all patents and patent applications, provisional patent applications or similar filings and any and all substitutions, divisions, continuations, continuations-in-part, divisions, reissues, renewals, extensions, reexaminations, patents of addition, supplementary protection certificates, utility models, inventors certificates, or the like and any foreign equivalents of the foregoing (including certificates of invention and any applications therefor) (collectively, Patents); (ii) all copyrights and copyrightable subject matter, whether registered or unregistered, including any of the foregoing that protect original works of authorship fixed in any tangible medium of expression, including all forms and types of computer Software) (collectively, Copyrights); (iii) all trademarks, servicemarks, trade names, business marks, service names, logos, brand names and corporate names together with the goodwill associated with any of the foregoing (collectively, Trademarks); (iv) all Internet domain names and social media accounts; (v) trade secrets, know-how and other confidential information (collectively, Trade Secrets); (vi) inventions, discoveries, ideas, creations, procedures, processes, methods, techniques, formulae, algorithms, specifications, designs, models, schematics, recordings, graphs, drawings, reports, analyses and improvements; (vii) all intellectual property rights in and to Software and Technology and (viii) rights to publicity, moral rights and all other similar rights throughout the world, however denominated.
IP License means (i) any grant (or covenant not to assert) by the Company or any of its Subsidiaries to another Person of or regarding any right relating to or under the Owned Intellectual Property Rights (Outbound IP License); and (ii) any grant (or covenant not to assert) by another Person to the Company or any of its Subsidiaries of or regarding any right relating to or under any third Persons Intellectual Property Rights (Inbound IP License).
IPO means the initial public offering of the SPAC pursuant to the Prospectus.
IRS means the Internal Revenue Service of the United States.
IT Systems means all computer systems, Software, servers, network equipment and other computer hardware, leased, licensed owned or purported to be owned by the Company or any of its Subsidiaries and used in the conduct of the Companys or any of its Subsidiaries businesses.
Key Employee means any Service Provider whose annual base salary (or, in the case of individual independent contractors, annualized aggregate fees) exceeds $275,000 (or local equivalent).
knowledge or any other similar knowledge qualification in this Agreement means the actual knowledge of, in the case of the Company, any of Michael Freed, Emmanuelle Lee, Thomas Logan or Brian Schopfer; and in the case of the SPAC, Thomas Knott.
Leased Real Property means all real property leased, subleased, or licensed by, the Company or its Subsidiaries as a tenant, sublessee or licensee, or as lessor, sublessor or licensor.
Letter of Transmittal means a letter of transmittal in a form reasonably agreeable by the Company and the SPAC.
Lien means any mortgage, lien, deed of trust, pledge, hypothecation, easement, license, option, right of first refusal, charge, security interest, condition, restriction, title defect, encroachment or encumbrance of any kind.
A-8
Table of Contents
Loan Note Equity Consideration means, if any, the aggregate Loan Notes Unit Consideration and Loan Notes SPAC Stock Consideration, based upon the election of the holder of such Loan Note made in accordance with Section 2.02.
Loan Notes means, collectively, the Management Loan Notes and the Shareholder Loan Notes.
Management Loan Notes means, collectively, those certain loan notes due 2026 issued to a certain Seller by Mirion Technologies (HoldingSub1), Ltd. pursuant to that certain Second Amended and Restated Loan Note Instrument dated September 10, 2018.
Management Sellers means the Sellers identified on Annex II.
Material Adverse Effect means any state of facts, development, effect, change, circumstance, event or occurrence, that, individually or in the aggregate, has had, or would reasonably be expected to have, a material adverse effect on the business, assets, results of operations or financial condition of the Company and the Subsidiaries, taken as a whole; provided, however, that in no event would any of the following (or the effect of any of the following), alone or in combination, be deemed to constitute a Material Adverse Effect or be taken into account in determining whether a Material Adverse Effect has occurred or would reasonably be expected to occur: (i) any change in Applicable Law or GAAP or any interpretation or enforcement thereof, (ii) any change in interest rates or economic, political, business, financial, commodity, currency or market conditions generally, (iii) any national or international political or social conditions in countries in which, or in the proximate geographic region in which, the Company or any of its Subsidiaries operates, including the engagement by the United States or such other countries in hostilities or the escalation thereof, whether or not pursuant to the declaration of a national emergency or war, or the occurrence or the escalation of any military or terrorist attack upon the United States or such other country, or any territories, possessions, or diplomatic or consular offices of the United States or such other countries or upon any military installation, equipment or personnel of the United States or such other country, (iv) any earthquake, hurricane, tsunami, tornado, flood, mudslide, wild fire or other natural disaster, act of God or other force majeure event, (v) any change generally affecting any of the industries or markets in which the Company or any of its Subsidiaries operates, (vi) the announcement or execution of this Agreement, or the pendency, performance or consummation of the transactions contemplated hereby, including the impact thereof on relationships, contractual or otherwise, with customers, suppliers, distributors, partners, licensors, providers or employees, (vii) the taking of any action required by the terms of this Agreement or with the prior written consent of the SPAC, (viii) any failure of the Company and its Subsidiaries, taken as a whole, to meet any projections, forecasts or budgets (provided that clause (viii) shall not prevent or otherwise affect a determination that any change or effect underlying such failure to meet projections or forecasts has resulted in, or contributed to, or would reasonably be expected to result in, a Material Adverse Effect, to the extent that such change or effect is not otherwise excluded from this definition of Material Adverse Effect) and (ix) COVID-19 or any other epidemic, pandemic or disease outbreak, or any Applicable Law, directive, pronouncement or guideline issued by a Governmental Authority, the Centers for Disease Control and Prevention (or similar national or international organization), the World Health Organization or industry group providing for business closures, changes to business operations, sheltering-in-place or other restrictions that relate to, or arise out of, an epidemic, pandemic or disease outbreak (including the COVID-19 pandemic) or any change in such Applicable Law, directive, pronouncement or guideline or interpretation thereof following the date of this Agreement or the Companys or any of its Subsidiaries compliance therewith; provided that, in the case of clauses (i)-(v) and (ix), such changes may be taken into account to the extent that such changes have had a disproportionate impact on the Company and its Subsidiaries, taken as a whole, as compared to other competitors or similarly situated companies operating in the industries or markets in which the Company and its Subsidiaries operate.
New Company Class A Common Stock means the Class A Common Stock, par value $0.0001 per share, as set forth in the Intermediate TopCo Certificate of Incorporation.
A-9
Table of Contents
New Company Class B Common Stock means the Class B Common Stock, par value $0.0001 per share, as set forth in the Intermediate TopCo Certificate of Incorporation.
New SPAC Class A Common Shares means the shares of Class A Common Stock of the SPAC, par value $0.0001 per share, as set forth in the New SPAC Certificate of Incorporation.
New SPAC Class B Common Shares means the shares of Class B Common Stock of the SPAC, par value $0.0001 per share, as set forth in the New SPAC Certificate of Incorporation.
New SPAC Common Stock means, collectively, the New SPAC Class A Common Shares and the New SPAC Class B Common Shares.
New SPAC Preferred Shares means the shares of preferred stock of the SPAC, par value $0.0001 per share, as set forth in the New SPAC Certificate of Incorporation.
NYSE means the New York Stock Exchange.
Open Source Software means any Software that is subject to or licensed, provided or distributed under, any license meeting the Open Source Definition (as promulgated by the Open Source Initiative as of the date of this Agreement) or the Free Software Definition (as promulgated by the Free Software Foundation as of the date of this Agreement) or any similar license for free, publicly available or open source Software, including the GNU General Public License, the Lesser GNU General Public License, the Apache License, the BSD License, Mozilla Public License (MPL), the MIT License or any other license that includes similar terms.
Owned Intellectual Property Rights means all Intellectual Property Rights (including Intellectual Property Rights in Company Software) owned or purported to be owned by the Company or any of its Subsidiaries.
Owned Real Property means all real property owned by the Company or its Subsidiaries.
Payroll Tax Executive Order means any U.S. presidential memorandum, executive order or similar pronouncement permitting or requiring the deferral of any payroll Taxes (including those imposed by Sections 3101(a) and 3201 of the Code).
Per Ordinary Share Cash Consideration means (x) Total Cash Consideration for Ordinary Shares, divided by (y) the number of Cash Electing Shares.
Per Ordinary Share SPAC Stock Consideration means a number of New SPAC Class A Common Shares equal to (w) (A) the Total Equity Consideration for Ordinary Shares, multiplied by (B) the SPAC Stock Election Proportion, divided by (z) $10.00 per share.
Per Ordinary Share Unit Consideration means a number of Units equal to (x) (A) the Total Equity Consideration for Ordinary Shares, multiplied by (B) the Unit Election Proportion, divided by (z) $10.00 per Unit.
Permitted Liens means (i) statutory or common law Liens of mechanics, materialmen, warehousemen, landlords, carriers, repairmen, construction contractors and other similar Liens that arise in the ordinary course of business, that relate to amounts not yet delinquent or that are being contested in good faith through appropriate Actions, in each case only to the extent appropriate reserves have been established in accordance with GAAP, (ii) Liens arising under original purchase price conditional sales contracts and equipment leases with third parties entered into in the ordinary course of business, (iii) Liens for Taxes not yet due and payable or which are being contested in good faith through appropriate Actions and, in each case, with respect to which appropriate reserves
A-10
Table of Contents
have been established in accordance with GAAP, (iv) covenants conditions, easements, restrictions, encroachments and other similar matters of record as reflected in title or other public records affecting the real property which do not detract from the value or interfere with the present or contemplated use, occupancy or operation of any property, (v) Liens securing any Indebtedness of the Company and its Subsidiaries and (vi) non-exclusive licenses of Intellectual Property Rights granted to customers, distributors and vendors in the ordinary course of business.
Person means an individual, firm, corporation, partnership, limited liability company, incorporated or unincorporated association, joint venture, joint stock company, trust or other entity or organization, including a Governmental Authority.
Personal Information means, in addition to any definition for personal information or any similar term (e.g., personal data or personally identifiable information or PII) provided by applicable Law, or by the Company or any of its Subsidiaries in any of its privacy policies or Contracts, all information that identifies, could be used to identify or is otherwise related to an individual person or device, including, but not limited to, (i) name, physical address, telephone number, email address, biometric identifiers, financial information, financial account number or government-issued identifier, (ii) any data regarding an individuals activities online or on a mobile device or application, and (iii) Internet Protocol addresses, device identifiers or other persistent identifiers. Personal Information may relate to any individual, including a current, prospective, or former customer, end user or employee of any Person.
PIPE Investors means those Persons who are participating in the PIPE Financing pursuant to a PIPE Subscription Agreement entered into with the SPAC as of the date hereof.
PPP Loans means any loans applied for or received by the Company or any of its Subsidiaries pursuant to the Paycheck Protection Program.
Pre-Closing Tax Period means any taxable period ending on or before the Closing Date, and in the case of a Straddle Period, the portion of such taxable period ending on the Closing Date.
Primary Capital means $50,000,000.
Privacy Laws shall mean any and all Applicable Law and self-regulatory guidelines that are binding on the Company (including of any applicable foreign jurisdiction) relating to the receipt, collection, compilation, use, storage, processing, sharing, safeguarding, security (technical, physical or administrative), disposal, destruction, disclosure or transfer (including cross-border) of Personal Information, including the Federal Trade Commission Act, General Data Protection Regulation (EU) 2016/679, the UK Data Protection Act 2018, the Federal Trade Commission Act, California Consumer Privacy Act (CCPA), Payment Card Industry Data Security Standard (PCI-DSS), any and all Applicable Law relating to breach notification or marketing in connection with Personal Information, and any Applicable Law relating to the use of biometric identifiers.
Prospectus means the prospectus, dated June 29, 2020, filed by the SPAC with the SEC.
Proxy Statement means the proxy statement that will be used for the SPAC Special Meeting to adopt and approve the Transaction Proposals and other matters reasonably related to the Transaction Proposals, as may be amended or supplemented, all in accordance with and as required by the Existing SPAC Certificate of Incorporation and Existing SPAC Bylaws, Applicable Law and any applicable rules and regulations of the SEC and NYSE.
Registration Statement means the Registration Statement on Form S-4, or other appropriate form determined by the Parties, including any pre-effective or post-effective amendments or supplements thereto, to be filed with the SEC by the SPAC under the Securities Act with respect to the New SPAC Class A Common Shares to be issued pursuant to this Agreement.
A-11
Table of Contents
Regulatory Consent Authority means the Antitrust Division of the United States Department of Justice, the United States Federal Trade Commission, the Committee on Foreign Investment in the United States, the Directorate of Defense Trade Controls, the French Ministry of the Economy and Finance and any other Governmental Authority charged with oversight of antitrust and/or foreign direct investment matters.
Release means any spilling, leaking, pumping, pouring, emitting, emptying, discharging, injecting, escaping, leaching, dumping, or disposing into the environment.
R&W Insurance Policy shall mean any buyer-side representations and warranties insurance policy with respect to the representations and warranties of the Company and the Sellers, in the name of and for the benefit of the SPAC.
Requisite Existing SPAC Stockholders means a majority of the Existing SPAC Stockholders who attend and vote at the SPAC Special Meeting.
Sanctioned Country shall mean, at any time, a country or territory which is itself the subject or target of territorial Sanctions (at the time of this Agreement, Crimea, Cuba, Iran, North Korea and Syria).
Sanctioned Person shall mean any Person that is the subject or target of Sanctions, including (i) any Person listed in any Sanctions-related list maintained by OFAC or the U.S. Department of State, the United Nations Security Council, Canada, the European Union, Her Majestys Treasury of the United Kingdom, Switzerland or any European Union member state; (ii) any Person located, organized, resident in or national of a Sanctioned Country; or (iii) any Person fifty percent (50%) or more owned, directly or indirectly, or otherwise controlled by or acting on behalf of any such Person or Persons described in the foregoing clauses (i) and (ii).
Sanctions shall mean economic or financial sanctions or trade embargoes imposed, administered or enforced from time to time by the U.S. government through OFAC or the U.S. Department of State, the United Nations Security Council, Canada, the European Union or any European Union member state, Her Majestys Treasury of the United Kingdom or Switzerland.
SEC means the U.S. Securities and Exchange Commission.
Securities Act means the Securities Act of 1933, as amended.
Seller-Borne Transaction Expenses means the following enumerated fees, costs and expenses of the SPAC and the Company and its Subsidiaries incurred prior to and through the Closing Date, whether paid or unpaid prior to the Closing, in connection with the IPO, the negotiation, preparation and execution of this Agreement and the other Transaction Agreements, the performance of and compliance with the terms of all Transaction Agreements to be performed or complied with at or before Closing, and the consummation of the Transactions, whether paid or unpaid prior to the Closing: (i) the fees, costs, expenses and disbursements of Freshfields Bruckhaus Deringer LLP, in its capacity as counsel to the Company and the Charterhouse Parties, and Lazard Ltd., in its capacity as a financial advisor to the Company, (ii) the aggregate amount of the Exit Bonuses, any other transaction or similar bonuses or other compensatory amounts payable in connection with the execution and delivery of this Agreement and/or the consummation of the Transactions, and the employer portion of payroll taxes on any such amounts, and (iii) solely to the extent the aggregate fees, costs and expenses described in clauses (i), (ii)(A)-(C) and (iii)(A)-(B) of the definition of Transaction Expenses exceed $60,000,000, fifty percent (50%) of the amount of such excess, up to a maximum of $5,000,000.
Seller Material Adverse Effect means, with respect to the Sellers, any state of facts, development, effect, change, circumstance, event or occurrence that, individually or in the aggregate, has prevented, materially delayed or materially impaired, or would reasonably be expected to prevent, materially delay or materially impair, the ability of the Sellers, considered as a group, to consummate the Transactions.
A-12
Table of Contents
Service Provider means any officer, director, employee (whether temporary, full- or part-time) or individual independent contractor of the Company or any of its Subsidiaries.
Shareholder Loan Notes means, collectively, (i) those certain loan notes due 2026 issued to certain of the Sellers by Mirion Technologies (HoldingSub1), Ltd. pursuant to that certain Second Amended and Restated Loan Note Instrument dated September 10, 2018; and (ii) those certain loan notes due 2026 issued to certain of the Sellers by Mirion Technologies (HoldingSub1), Ltd. pursuant to that certain Loan Note Instrument dated December 16, 2020.
Software means all: (i) computer programs, including all software implementations of algorithms, models and methodologies, whether in source code or object code; (ii) databases and compilations, including all data and collections of data, whether machine readable or otherwise; (iii) descriptions, flow-charts and other work product used to design, plan, organize and develop any of the foregoing, screens, user interfaces, report formats, firmware, development tools, templates, menus, buttons, images, videos, models and icons; and (iv) documentation, including user manuals and other training documentation, related to any of the foregoing.
SPAC Material Adverse Effect means, with respect to the SPAC, any state of facts, development, effect, change, circumstance, event or occurrence that, individually or in the aggregate, has prevented, materially delayed or materially impaired, or would reasonably be expected to prevent, materially delay or materially impair, the ability of the SPAC to consummate the Transactions.
SPAC Special Meeting means the meeting of the Existing SPAC Stockholders, for the purpose of obtaining the approval of the Transaction Proposals and other matters reasonably related to the Transaction Proposals.
SPAC Stock Election Proportion means the number of SPAC Stock Electing Shares, divided by the aggregate number of SPAC Stock Electing Shares and Unit Electing Shares.
Sponsor means GS Sponsor II LLC, a Delaware limited liability company.
Straddle Period means any taxable period that includes (but does not end on) the Closing Date.
Subsidiary means, with respect to any Person, any partnership, limited liability company, corporation or other business entity of which: (i) if a corporation, a majority of the total voting power of shares of capital stock entitled (without regard to the occurrence of any contingency) to vote in the election of directors, managers, or trustees thereof is at the time owned or controlled, directly or indirectly, by that Person or one or more of the other Subsidiaries of that Person or a combination thereof; (ii) if a partnership, limited liability company or other business entity, a majority of the partnership or other similar ownership interests thereof is at the time owned or controlled, directly or indirectly, by that Person or one or more Subsidiaries of that Person or a combination thereof; or (iii) in any case, such Person controls the management thereof.
Tax means (i) any federal, state, provincial, territorial, local, non-U.S. and other net income, alternative or add-on minimum, franchise, gross income, adjusted gross income or gross receipts, employment-related (including employee withholding or employer payroll, ad valorem, transfer, franchise, license, excise, severance, stamp, occupation, premium, personal property, real property, capital stock, profits, disability, occupation, windfall profit, registration, value added, social security, estimated, customs, duties, sales or use tax, or any other tax or like assessment or charge, in each case, in the nature of a tax, together with any interest, penalty, addition to tax or additional amount imposed with respect thereto by a Governmental Authority and (ii) any liability in respect of any items described in clause (i) payable as a result of being a member of a combined, consolidated, unitary or affiliated group (including pursuant to Treasury Regulations Section 1.1502-6 or any comparable provision of Applicable Law), or as a transferee or successor.
A-13
Table of Contents
Tax Return means any return, report, real property transfer tax return, statement, refund, claim, declaration, information return, statement, estimate or other document filed or required to be filed with a Governmental Authority in respect of Taxes, including any schedule or attachment thereto and including any amendments thereof.
Technology means, collectively, all Software, information, formulae, algorithms, procedures, methods, techniques, research and development, technical data, programs, subroutines, tools, materials, processes, apparatus, creations, and other similar materials, and all recordings, graphs, reports, analyses, and other writings, and other tangible embodiments of the foregoing, in any form whether or not specifically listed herein, and all related technology, that are used in, incorporated in, embodied in, displayed by or related to, or are used in connection with the foregoing.
Total Cash Consideration for Ordinary Shares means, if any, (x) Cash Consideration less (y) the Total Loan Note Cash Consideration (provided that such amount shall in no case be less than zero).
Total Consideration means $1,700,000,000.
Total Consideration for Ordinary Shares means (x) Total Consideration less (y) the Total Loan Note Consideration.
Total Equity Consideration for Ordinary Shares means (x) Total Consideration for Ordinary Shares less (y) the Total Cash Consideration for Ordinary Shares (if any).
Total Loan Note Consideration means an amount equal to, as of the Closing Date, the total principal and accrued interest with respect to all of the Loan Notes (the Closing Date Loan Note Balance), which amount as of June 30, 2021 and December 31, 2021 shall be as set forth on Schedule 2.06.
Transaction Agreements means this Agreement, the Joinder Agreements, the Sponsor Support Agreement, the PIPE Subscription Agreements, the Backstop Agreement, the Debt Commitment Letter, the Registration Rights Agreement, the Election Agreements, the New SPAC Certificate of Incorporation, the New SPAC Bylaws, the Intermediate TopCo Certificate of Incorporation, the Confidentiality Agreement and all agreements, documents, instruments and certificates entered into in connection herewith or therewith and any and all exhibits and schedules thereto.
Transaction Expenses means all fees, costs and expenses of the SPAC and the Company and its Subsidiaries incurred prior to and through the Closing Date, whether paid or unpaid prior to the Closing, in connection with the IPO, the negotiation, preparation and execution of this Agreement and the other Transaction Agreements, the performance of and compliance with the terms of all Transaction Agreements to be performed or complied with at or before Closing, and the consummation of the Transactions, including (i) any deferred underwriter fees, discounts and commissions in connection with the SPACs IPO, (ii) the unreimbursed fees, costs, expenses and disbursements of counsel, accountants, advisors and consultants of the SPAC and the Company and its Subsidiaries, specifically including those of (A) Davis Polk & Wardwell LLP, (B) PricewaterhouseCoopers LLP, and (C) Boston Consulting Group, Inc., (iii) the fees, costs and expenses incurred in connection with the PIPE Financing, the Debt Financing and the Payoff Letter, specifically including (A) any cash financing fees in connection therewith, (B) any third-party advisory expenses in connection therewith and (C) any consent fees and similar costs associated therewith, in the case of each of clauses (iii)(A) and (iii)(B), that were approved in writing prior to April 14, 2021 by the Charterhouse Entities, (iv) the costs and expenses associated with any filings with or notifications to any Governmental Authority in connection with the transactions contemplated by the Transaction Agreements, including pursuant to the HSR Act, (v) the fees, costs and expenses associated with the preparation and filing of the Registration Statement and the Proxy Statement and the SPAC Special Meeting and (vi) the fees, costs and expenses incurred in connection with the R&W Insurance Policy.
A-14
Table of Contents
Transaction Proposals means (a) the adoption and approval of this Agreement, (b) the amendment and restatement of the New SPAC Certificate of Incorporation, (c) the issuance of New SPAC Class A Common Shares in connection with (x) the Transactions (including as may be required under the NYSE) and (y) the PIPE Financing; (d) the issuance of New SPAC Class B Common Shares in connection with the Transactions; (e) the approval of the Equity Incentive Plan; (f) the election of the directors constituting the board of directors of the SPAC; (g) the adoption and approval of any other proposals that the SEC (or staff members thereof) may indicate are necessary in its comments to the Registration Statement or the Proxy Statement or correspondence related thereto; (h) any other proposals the Parties agree are necessary or desirable to consummate the Transactions; and (i) adjournment of the SPAC Special Meeting, if necessary, to permit further solicitation of proxies because there are not sufficient votes to approve and adopt any of the foregoing.
Transactions means the transactions contemplated by this Agreement and the other Transaction Agreements.
Transfer Tax means any direct or indirect transfer (including real estate transfer), sales, use, stamp, documentary, registration, conveyance, recording or other similar Taxes or governmental fees, including any interest, penalty or addition to Tax with respect thereto.
Treasury Regulations means the temporary and final regulations promulgated under the Code.
Trust Agreement means the Investment Management Trust Agreement, dated as of June 29, 2020, by and between the SPAC and Continental Stock Transfer & Trust Company, a New York corporation.
Trustee means Continental Stock Transfer & Trust Company.
Unit means, together, one New SPAC Class B Common Share and one share of New Company Class B Common Stock.
Unit Election Proportion means the number of Unit Electing Shares divided by the aggregate number of SPAC Stock Electing Shares and Unit Electing Shares.
(b) Each of the following terms is defined in the Section set forth opposite such term:
| Term | Section | |||
| Approvals |
4.13 |
|||
| Agreement |
Preamble |
|||
| Alternative Financing |
8.09 |
|||
| Alternative Financing Commitment Letter |
8.09 |
|||
| Anti-Corruption Laws |
4.23 |
|||
| Acquisition Transaction |
8.08(b) |
|||
| Audited Financial Statements |
4.08(a) |
|||
| Backstop Agreement |
Recitals |
|||
| Backstop Party |
Recitals |
|||
| Balance Sheet Date |
4.08(a) |
|||
| Business Combination Proposal |
8.08(c) |
|||
| Call Option |
Recitals |
|||
| Cash Electing Share |
2.02(a)(i) |
|||
| Cash Election for Loan Notes |
2.02(a)(ii) |
|||
| Cash Election for Shares |
2.02(a)(i) |
|||
| Charterhouse Director Designation Agreement |
Recitals |
|||
| Charterhouse Parties |
Preamble |
|||
| Closing |
2.03(a) |
|||
| Closing Date |
2.03(a) |
|||
| Closing Step Plan |
Recitals |
A-15
Table of Contents
| Term | Section | |||
| Company |
Preamble |
|||
| Company Closing Statement |
2.07 |
|||
| Company Cure Period |
12.01(d) |
|||
| Company Disclosure Schedule |
ARTICLE 4 |
|||
| Company Securities |
4.05(b) |
|||
| Company Software |
4.15(g) |
|||
| Completion 8-K |
8.05 |
|||
| DCL Beneficiary |
Recitals |
|||
| Debt Commitment Letter |
4.30(a) |
|||
| Debt Financing |
4.30(a) |
|||
| Debt Payoff Amount |
2.04(e)(vii) |
|||
| Election Agreement |
2.02(a) |
|||
| Election Deadline |
2.02(e) |
|||
| Election Period |
2.02(d) |
|||
| e-mail |
13.03 |
|||
| End Date |
12.01(b) |
|||
| Environmental Permits |
4.20(a) |
|||
| Equity Incentive Plan |
10.01 |
|||
| Exchange Agent |
2.08(a) |
|||
| Exchange Fund |
2.08(d) |
|||
| Existing SPAC Preferred Stock |
5.05(a) |
|||
| Financial Statements |
4.08(a) |
|||
| HRP |
6.04(b) |
|||
| Interim Period |
6.01 |
|||
| Intermediate TopCo |
Recitals |
|||
| Intermediate TopCo Bylaws |
Recitals |
|||
| Intermediate TopCo Certificate of Incorporation |
Recitals |
|||
| Jersey Merger Sub |
Recitals |
|||
| Joining Seller |
Preamble |
|||
| Leases |
4.14(b) |
|||
| Licensed Intellectual Property Rights |
4.15(b) |
|||
| Loan Note Cash Consideration |
2.02(a)(ii) |
|||
| Loan Notes SPAC Stock Consideration |
2.02(a)(ii) |
|||
| Loan Notes Unit Consideration |
2.02(a)(ii) |
|||
| Material Contracts |
4.11(a) |
|||
| Material Customers |
4.11(a)(xiii) |
|||
| Material Suppliers |
4.11(a)(xiii) |
|||
| Minimum Cash Condition |
11.03(d) |
|||
| New SPAC Certification of Incorporation |
Recitals |
|||
| New SPAC Bylaws |
Recitals |
|||
| Option Agreement |
Recitals |
|||
| Parties |
Preamble |
|||
| Payoff Letter |
2.04(e)(vii) |
|||
| Personnel IP Contracts |
4.15(e) |
|||
| PIPE Financing |
Recitals |
|||
| PIPE Financing Amount |
Recitals |
|||
| PIPE Subscription Agreement |
Recitals |
|||
| Pre-Closing Step Plan |
Recitals |
|||
| Private Placement Warrants |
5.05(a) |
|||
| Public Warrants |
5.05(a) |
|||
| Registered Intellectual Property Rights |
4.15(a) |
A-16
Table of Contents
| Term | Section | |||
| Registration Rights Agreement |
Recitals |
|||
| Related Parties |
4.21 |
|||
| SEC Documents |
5.06(a) |
|||
| Seller |
Preamble |
|||
| Seller-Borne Expense Portion |
2.04(d) |
|||
| Seller Disclosure Schedule |
3 |
|||
| Seller Total Consideration |
2.04(d) |
|||
| Sellers |
Preamble |
|||
| SPAC |
Preamble |
|||
| SPAC Closing Statement |
2.06 |
|||
| SPAC Cure Period |
12.01(e) |
|||
| SPAC Sponsor Director Designation Agreement |
Recitals |
|||
| SPAC Material Contract |
5.16 |
|||
| SPAC Disclosure Schedule |
5 |
|||
| SPAC Stock Election for Loan Notes |
2.02(a)(ii) |
|||
| SPAC Stock Election for Shares |
2.02(a)(i) |
|||
| SPAC Stock Electing Share |
2.02(a)(i) |
|||
| SPAC Stockholder Redemption |
8.04(a) |
|||
| Sponsor Support Agreement |
Recitals |
|||
| Subsidiary Securities |
4.06(b) |
|||
| Supporting Company Holders |
Preamble |
|||
| Tax Contest |
9.03 |
|||
| Terminating Company Breach |
12.01(d) |
|||
| Terminating SPAC Breach |
12.01(e) |
|||
| Total Loan Note Cash Consideration |
2.02(a)(ii) |
|||
| Title IV Plan |
4.18(b) |
|||
| Trust Account |
5.20(a) |
|||
| Trust Amount |
5.20(a) |
|||
| Unit Election for Loan Notes |
2.02(a)(ii) |
|||
| Unit Election for Shares |
2.02(a)(i) |
|||
| Unit Electing Share |
2.02(a)(i) |
|||
| Up-C Merger |
Recitals |
|||
| USRPIs |
2.04(e)(ii) |
|||
| Waived 280G Benefits |
10.03 |
|||
| WARN |
4.17(f) |
Section 1.02. Other Definitional and Interpretative Provisions. The words hereof, herein and hereunder and words of like import used in this Agreement shall refer to this Agreement as a whole and not to any particular provision of this Agreement. The captions herein are included for convenience of reference only and shall be ignored in the construction or interpretation hereof. References to Articles, Sections, Exhibits and Schedules are to Articles, Sections, Exhibits and Schedules of this Agreement unless otherwise specified. All Exhibits and Schedules annexed hereto or referred to herein are hereby incorporated in and made a part of this Agreement as if set forth in full herein. Any capitalized terms used in any Exhibit or Schedule but not otherwise defined therein shall have the meaning as defined in this Agreement, and any accounting term not otherwise defined herein has the meaning assigned to it in accordance with GAAP. Any singular term in this Agreement shall be deemed to include the plural, and any plural term the singular. References to him, her, it, itself and other like references shall be deemed to include the masculine or feminine reference, as the case may be. Whenever the words include, includes or including are used in this Agreement, they shall be deemed to be followed by the words without limitation, whether or not they are in fact followed by those words or words of like import. The word will shall be construed to have the same meaning and effect as the word shall. The word or when used in this Agreement is not exclusive. Writing, written and comparable terms refer to
A-17
Table of Contents
printing, typing and other means of reproducing words (including electronic media) in a visible form. References to any statute, rule, regulation, law or Applicable Law shall be deemed to refer to all Applicable Law as amended or supplemented from time to time and to any rules, regulations and interpretations promulgated thereunder, unless otherwise explicitly specified. References to any agreement or contract are to that agreement or contract as amended, modified or supplemented from time to time in accordance with the terms hereof and thereof; provided that with respect to any agreement or contract listed on any schedules hereto, all such amendments, modifications or supplements must also be listed in the appropriate schedule. References to any Person include the successors and permitted assigns of that Person. References from or through any date mean, unless otherwise specified, from and including or through and including, respectively. References to days shall refer to calendar days unless Business Days are specified. If any action is to be taken or given on or by a particular calendar day, and such calendar day is not a Business Day, then such action may be deferred until the next Business Day. All monetary figures shall be in United States dollars unless otherwise specified. The Parties have participated jointly in the negotiation and drafting of this Agreement and each has been represented by counsel of its choosing; in the event an ambiguity or question of intent or interpretation arises, this Agreement shall be construed as jointly drafted by the Parties and no presumption or burden of proof shall arise favoring or disfavoring any Party by virtue of the authorship of any provision of this Agreement. The phrases provided to, furnished to, made available and phrases of similar import when used herein, unless the context otherwise requires, means that a copy of the information or material referred to has been provided no later than 9:00 a.m. on the day immediately prior to the date of this Agreement to the Party to which such information or material is to be provided or furnished in the virtual data room set up by the Company in connection with this Agreement.
Section 1.03. Equitable Adjustments. If, between the date of this Agreement and the Closing, the outstanding Existing Company Shares or shares of Existing SPAC Common Stock shall have been changed into a different number of shares or a different class, by reason of any stock or share dividend, subdivision, reclassification, reorganization, recapitalization, split, combination or exchange of shares, or any similar event shall have occurred, or if there shall have been any breach by the SPAC with respect to its covenant not to issue shares of Existing SPAC Common Stock or rights to acquire Existing SPAC Common Stock, then any number, value (including dollar value) or amount contained herein which is based upon the number of Existing Company Shares or shares of Existing SPAC Common Stock, as applicable, will be appropriately adjusted to provide to the holders of Existing Company Shares or the holders of Existing SPAC Common Stock, as applicable, the same economic effect as contemplated by this Agreement prior to such event; provided, however, that this Section 1.03 shall not be construed to permit the SPAC or the Company to take any action with respect to their respective securities that is prohibited by the terms and conditions of this Agreement.
ARTICLE 2
BUSINESS COMBINATION
Section 2.01. Company Articles Amendment. No later than ten (10) Business Days following the date of this Agreement, the Company and the Supporting Company Holders shall take, or cause to be taken, such actions as are required to amend and restate the Existing Company Articles in the form set forth as Exhibit P (the Company Articles Amendment).
Section 2.02. Election Procedures. Each Seller shall have the right, subject to the limitations set forth in this Article 2, to submit an election in accordance with the following procedures:
(a) Each Seller must specify in a request in the form attached hereto as Exhibit G (an Election Agreement):
(i) the respective number of Existing Company Shares owned by such Seller in respect of which such Seller desires to receive (in each case, if any):
(x) the Per Ordinary Share Cash Consideration (any such election, a Cash Election for Shares, and each Existing Company Share with respect to which such Cash Election was made, a Cash Electing Share),
A-18
Table of Contents
(y) the Per Ordinary Share Unit Consideration (any such election, a Unit Election for Shares, and each Existing Company Share with respect to which such Unit Election for Shares was made, a Unit Electing Share), or
(z) the Per Ordinary Share SPAC Stock Consideration (any such election, a SPAC Stock Election for Shares, and each Existing Company Share with respect to which such SPAC Stock Election for Shares was made, a SPAC Stock Electing Share)
; provided, that if, as determined immediately prior to Closing, the number of Existing Company Shares in respect of which Sellers have made a Cash Election for Shares would cause the aggregate Per Share Cash Consideration to exceed the Total Cash Consideration for Ordinary Shares, the number of Existing Company Shares subject to a Cash Election for Shares shall be automatically adjusted, pro rata based on each electing Sellers aggregate ownership, such that the aggregate Per Share Cash Consideration shall equal the Total Cash Consideration for Ordinary Shares and, with respect to any Existing Company Shares so deemed to have made an alternate election as a result of such adjustment, the Sellers shall be deemed to have made the alternate Unit Election for Shares or SPAC Stock Election for Shares as set forth in such Sellers Election Agreement; and
(ii) with respect to the Loan Notes owned by such Seller whether such Seller desires to receive with respect to such Sellers Loan Notes:
(x) an amount in cash equal to the total principal and accrued interest as of the Closing Date with respect to such Loan Notes (Loan Note Cash Consideration) (any such election, a Cash Election for Loan Notes),
(y) a number of Units equal to the total principal and accrued interest as of the Closing Date with respect to such Loan Notes divided by $10.00 per Unit (Loan Notes Unit Consideration) (any such election, a Unit Election for Loan Notes), or
(z) a number of shares of SPAC Stock equal to the total principal and accrued interest as of the Closing Date with respect to such Loan Notes divided by $10.00 per Unit (Loan Notes SPAC Stock Consideration) (any such election, a SPAC Stock Election for Loan Notes)
; provided, that if, as determined immediately prior to Closing, the number of Loan Notes in respect of which Sellers have made a Cash Election for Loan Notes would cause the aggregate Loan Note Cash Consideration to exceed the Cash Consideration, the number of Loan Notes subject to a Cash Election for Loan Notes shall be automatically adjusted, pro rata based on each electing Sellers aggregate holdings of Loan Notes, such that the aggregate Loan Note Cash Consideration shall equal the Cash Consideration (as such aggregate Loan Note Cash Consideration may be adjusted by this proviso, the Total Loan Note Cash Consideration) and, with respect to any Loan Notes so deemed to have made an alternate election as a result of such adjustment, the Sellers shall be deemed to have made the alternate Unit Election for Loan Notes or SPAC Stock Election for Loan Notes as set forth in such Sellers Election Agreement.
(b) Sellers who hold Existing Company Shares or Loan Notes as nominees, trustees or in other representative capacities may submit a separate Election Agreement on or before the Election Deadline (as defined below) with respect to each beneficial owner for whom such nominee, trustee or representative holds such Existing Company Shares or Loan Notes.
(c) Unless expressly permitted in writing by the Company, the Charterhouse Parties and the SPAC, any election made pursuant to an Election Agreement shall be irrevocable.
(d) The SPAC shall direct the Exchange Agent to mail or transmit in electronic form the Election Agreement within two (2) Business Days (or such later date agreed by the Company, the Charterhouse Parties and the SPAC) after the date the Registration Statement is declared effective by the SEC, to the record holders of Existing Company Shares as of the date hereof, and following such date, shall use reasonable best efforts to make
A-19
Table of Contents
available as promptly as practicable the Election Agreement to any holder of Existing Company Shares who requests such Election Agreement prior to the Election Deadline. The time period between such mailing date and the Election Deadline is referred to herein as the Election Period.
(e) With respect to (i) any Seller of Existing Company Shares in respect of which a properly completed and duly executed Election Agreement has not been received by the Exchange Agent prior to the Election Deadline, together with any additional documents specified in the procedures set forth in the Election Agreement, (ii) any holder of Existing Company Shares that does not deliver a properly completed and duly executed Joinder Agreement, and (iii) any Seller of Loan Notes in respect of which a properly completed and duly executed Election Agreement has not been received by the Exchange Agent prior to the Election Deadline, together with any additional documents specified in the procedures set forth in the Election Agreement, such Persons shall be deemed to have made a SPAC Stock Election for Shares with respect to such Existing Company Shares or Loan Notes, as applicable. As used herein, unless otherwise agreed in advance by the parties, Election Deadline means 5:00 p.m. New York City time on the earliest practicable date which the Company, the Charterhouse Parties and the SPAC shall agree, but in any event, no less than ten (10) Business Days after the date on which the Registration Statement is declared effective by the SEC.
Section 2.03. Closing. (a) The closing (the Closing) of the transactions contemplated hereby shall occur simultaneously with respect to all Sellers and shall take place electronically at 10:00 a.m. Eastern Time, no later than the third (3rd) Business Day after the satisfaction (or waiver in accordance with this Agreement) of the last to occur of the conditions set forth in Article 11 (other than any such conditions that by their nature cannot be satisfied until the Closing Date, which shall be required to be so satisfied or (to the extent permitted by Applicable Law and this Agreement) waived on the Closing Date), or on such other date or at such other time or place as each of the SPAC, the Company and the Charterhouse Parties may agree in writing (such date on which the Closing occurs, the Closing Date).
Section 2.04. Closing Deliverables. At or before the Closing:
(a) Each Seller shall deliver or cause to be delivered to the SPAC:
(i) a duly executed counterpart to this Agreement or, as applicable, a Joinder Agreement;
(ii) in the case of any Seller that is a United States person within the meaning of Section 7701(a)(30) of the Code, a properly completed IRS Form W-9;
(iii) in the case of any Seller that is not a United States person within the meaning of Section 7701(a)(30) of the Code, a properly completed IRS Form W-8 appropriate to such Sellers circumstances; and
(iv) certificates for the number of Existing Company Shares listed opposite such Sellers name on Annex I with respect to which such Existing Company Shares are to be converted into New Company Class A Common Stock transferred to the SPAC, or, with respect to a Joining Seller, on Exhibit A to such Joining Sellers Joinder Agreement, duly endorsed and accompanied by a duly executed share transfer form.
(b) Each Seller shall deliver or cause to be delivered to Mirion Technologies (HoldingSub1), Ltd. certificates for the number of Loan Notes listed opposite such Sellers name on Annex I or, with respect to a Joining Seller, as specified on such Joining Sellers Joinder Agreement.
(c) Each Seller shall deliver to the Company and the SPAC duly executed counterparts to the Registration Rights Agreement.
(d) Each Seller agrees that the SPAC and the Company shall be entitled to, and shall, prior to payment of any such amounts owed to such Seller, deduct from that portion of the Total Loan Note Consideration and Total Consideration for Ordinary Shares payable to such Seller in respect of such Sellers Loan Notes and Ordinary
A-20
Table of Contents
Shares (such Sellers Seller Total Consideration) an amount equal to such Sellers aggregate portion of the Seller-Borne Transaction Expenses, on a pro rata basis based on the proportion that such Sellers Seller Total Consideration bears to Total Consideration (such amount such Sellers Seller-Borne Expense Portion). Each Sellers Seller-Borne Expense Portion will be deducted from, or forfeited by, such Seller from such Sellers Seller Total Consideration in equal proportion from the Seller Total Consideration to be received by Such Seller in cash, New SPAC Class A Common Shares or Units; providing for such purpose that the New SPAC Class A Common Shares or Units to be received by such Seller shall be valued at $10.00.
(e) The Company shall deliver or cause to be delivered:
(i) to the SPAC and the Sellers party thereto, a duly executed counterpart to this Agreement and the Registration Rights Agreement;
(ii) to the SPAC and Intermediate TopCo, (x) a certificate duly executed by the Company in form and substance required under Treasury Regulations Section 1.1445-11T, stating that either (A) fifty percent (50%) or more of the value of the gross assets of the Company does not consist of U.S. real property interests within the meaning of Section 897 of the Code and the Treasury Regulations thereunder (USRPIs) or (B) ninety percent (90%) or more of the value of the gross assets of the Company does not consist of USRPIs plus cash or cash equivalents and (y) a properly completed certification from the Company pursuant to Section 1446(f) and Treasury Regulations Section 1.1446(f)-2(b)(4)(i)(B);
(iii) to each holder of Existing Company Shares making a Unit Election for Shares (including with respect to a deemed Unit Election for Shares), in respect of each such Existing Company Share, certificates or other evidence of ownership set forth on the shareholder register of the Company with respect to the New Company Class B Common Stock to be owned by such holder following the transactions contemplated by this Article 2;
(iv) to each holder of a Loan Note electing to receive Units with respect to a portion of such holders Loan Note Equity Consideration, certificates or other evidence of ownership set forth on the loan note register of the Company with respect to that number of New Company Class B Common Stock equal to such holders applicable portion of the Loan Note Equity Consideration, based upon such holders portion of the aggregate principal and accrued interest with respect to all Loan Notes outstanding as of the Closing Date (such payment to be made in accordance with the Closing Step Plan);
(v) to each recipient set forth in Section 2.04(e)(v) of the Company Disclosure Schedule, the amount of Seller-Borne Transaction Expenses due to such recipient;
(vi) evidence reasonably acceptable to the SPAC that the Company has settled and terminated the Existing Investment Agreements in compliance with Section 6.06; and
(vii) to the SPAC, a payoff letter in customary form (the Payoff Letter), which payoff letter shall (i) indicate the total amount required to be paid to fully satisfy all principal, interest, prepayment premiums, penalties or similar obligations related to the Credit Agreement as of the Closing Date (the Debt Payoff Amount), and (ii) state that all Liens in connection therewith relating to the assets of the Company or its Subsidiaries shall be, upon the payment of the Debt Payoff Amount on the Closing Date, released.
(f) The SPAC shall deliver or cause to be delivered:
(i) to each Seller and the Company, a duly executed counterpart to this Agreement;
(ii) to the Company and the Sellers party thereto, a duly executed counterpart to the Registration Rights Agreement;
(iii) to each holder of Existing Company Shares electing to receive cash, an amount in cash equal to such holders Per Ordinary Share Cash Consideration, if any (such payment to be made in accordance with the Closing Step Plan);
A-21
Table of Contents
(iv) to each holder of Existing Company Shares making a Unit Election for Shares, in respect of each such Unit Electing Share, the portion of the Per Ordinary Share Unit Consideration comprising New SPAC Class B Common Shares;
(v) to each holder of Existing Company Shares making a SPAC Stock Election for Shares, in respect of such SPAC Stock Electing Share, the portion of the Per Ordinary Share SPAC Stock Consideration comprising New SPAC Class A Common Shares;
(vi) to each holder of a Loan Note electing to receive cash, an amount in cash equal to such holders Loan Note Cash Consideration, if any (such payment to be made in accordance with the Closing Step Plan);
(vii) to each holder of a Loan Note electing to receive New SPAC Class A Common Shares with respect to such holders portion of Loan Note Equity Consideration, that number of New SPAC Class A Common Shares equal to such holders applicable portion of the Loan Note Equity Consideration (such payment to be made in accordance with the Closing Step Plan);
(viii) to each holder of a Loan Note electing to receive Units with respect to such holders portion of Loan Note Equity Consideration, that number of shares of New SPAC Class B Common Shares equal to such holders applicable portion of the Loan Note Equity Consideration (such payment to be made in accordance with the Closing Step Plan);
(ix) to each recipient set forth in Section 2.04(f)(ix) of the SPAC Disclosure Schedule, the amount of Transaction Expenses due to such recipient; and
(x) to the Company, any remaining cash from the PIPE Financing and the Trust Account.
Section 2.05. Closing Transactions. At or prior to the Closing, the Parties shall cause the consummation of the following transactions in the following order:
(a) in connection with the Closing, and in accordance with the Closing Step Plan, the Company and its applicable Subsidiaries shall take all action necessary to effect the extinguishment of the Loan Notes including cancelling all Loan Note certificates, updating all loan note registers which relate to the Loan Notes and notifying the Official List of The International Stock Exchange in writing to cancel the listing of all Loan Notes with immediate effect from the Closing Date;
(b) the Sponsor shall waive any adjustment to the conversion ratio set forth in the Existing SPAC Certificate of Incorporation resulting from the transaction contemplated by the PIPE Subscription Agreements pursuant to and in accordance with the terms of the Sponsor Support Agreement, which Sponsor Support Agreement shall remain in full force and effect as of the Closing;
(c) prior to the Closing, the New SPAC Certificate of Incorporation authorizing the New SPAC Common Stock shall become effective;
(d) the PIPE Investors shall purchase, and the SPAC shall issue and sell to the PIPE Investors, the number of shares of New SPAC Class A Common Shares set forth in the PIPE Subscription Agreements against payment of the subscription price set forth in the PIPE Subscription Agreements;
(e) the SPAC shall deposit or cause to be deposited with the Exchange Agent the Exchange Fund; and
(f) the Parties shall execute the transactions described in the Closing Step Plan in the order in which they appear, with such amendments and modifications as the Company, the SPAC and the Charterhouse Parties may agree to effectuate the intent of the Parties under this Agreement, and the Parties agree that they shall, and shall cause their Subsidiaries to, deliver any such consents and approvals, and take all such actions, as are necessary and desirable to execute such transactions described in the Closing Step Plan (as may be modified and amended by the Company, the SPAC and the Charterhouse Parties hereunder), which shall include exercising and
A-22
Table of Contents
procuring the exercise of any and all voting rights attaching to the Existing Company Shares to which such Party is or may become entitled to vote in favor of any resolution approving the Up-C Merger (including without limitation the resolutions required to approve the merger agreement relating to the Up-C Merger under Article 127F(1) of the Companies (Jersey) Law 1991 (as amended)), or such other transactions as may be required by the Closing Step Plan (as may be modified and amended by the Company, the SPAC and the Charterhouse Parties hereunder).
Section 2.06. SPAC Closing Statement. At least two (2) Business Days prior to the SPAC Special Meeting and in any event not earlier than the time that Existing SPAC Investors may no longer elect redemption in accordance with the SPAC Stockholder Redemption, the SPAC shall prepare and deliver to the Company and the Charterhouse Parties a statement (the SPAC Closing Statement) setting forth in good faith: (a) the aggregate amount of cash in the Trust Account (prior to giving effect to the SPAC Stockholder Redemption), the PIPE Investment proceeds received and to be received by the SPAC on the Closing Date and, as communicated by the Company to the SPAC in writing at least five (5) Business Days prior the SPAC Special Meeting, and the Debt Financing Proceeds to be received by the DCL Beneficiary (or its applicable affiliate assignee or designee) prior to the Closing; (b) the aggregate amount of all payments required to be made in connection with the SPAC Stockholder Redemption; (c) the aggregate amount of Transaction Expenses (other than Seller-Borne Transaction Expenses); (d) the Available Closing Cash (excluding the Closing Balance Sheet Cash) resulting therefrom; (e) the number of shares of New SPAC Common Stock to be outstanding as of the Closing after giving effect to the SPAC Stockholder Redemption and the issuance of shares of New SPAC Common Stock pursuant to the PIPE Subscription Agreements; and (f) the Backstop Amount; in each case, including reasonable supporting detail therefor. The SPAC Closing Statement and each component thereof shall be prepared and calculated in accordance with the definitions contained in this Agreement. From and after delivery of the SPAC Closing Statement until the Closing, the SPAC shall (x) cooperate with and provide the Company and its representatives all information reasonably requested by the Company or any of its representatives and within the SPACs or its representatives possession or control in connection with the Companys review of the SPAC Closing Statement and (y) consider in good faith any comments to the SPAC Closing Statement provided by the Company, which comments the Company shall deliver to the SPAC no less than two (2) Business Days prior to the Closing Date, and the SPAC shall revise such SPAC Closing Statement to incorporate any changes SPAC determines, in its sole discretion, are necessary or appropriate given such comments. Schedule 2.06 to this Agreement sets forth, for informational purposes, an illustrative sources and uses calculation, calculated on the basis of certain assumptions regarding Available Closing Cash (including the components thereof), Transaction Expenses, the Debt Payoff Amount, the consideration elections to be made by the Sellers under this Agreement and such other assumptions as set forth therein.
Section 2.07. Company Closing Statement. At least two (2) Business Days prior to the Closing Date, the Company shall prepare and deliver to the SPAC a statement (the Company Closing Statement) setting forth in good faith as of the Closing Date: (a) the aggregate number of A Ordinary Shares issued and outstanding; (b) the aggregate number of B Ordinary Shares issued and outstanding (in the case of (a) and (b), prior to giving effect to the Up-C Merger); (c) the aggregate number of New Company Class A Common Stock after giving effect to the Up-C Merger; (d) the aggregate number of New Company Class B Common Stock after giving effect to the Up-C Merger; (e) the total principal and accrued interest with respect to the Shareholder Loan Notes; (f) the total principal and accrued interest with respect to the Management Loan Notes; (g) the Closing Balance Sheet Cash, (h) the Companys calculation of the Total Loan Note Consideration; and (i) the Companys calculation of the Total Consideration for Ordinary Shares, in each case, including reasonable supporting detail therefor. From and after delivery of the Company Closing Statement until the Closing, the Company shall (x) cooperate with and provide the SPAC and its representatives all information reasonably requested by the SPAC or any of its representatives and within the Companys or its representatives possession or control in connection with the SPACs review of the Company Closing Statement and (y) consider in good faith any comments to the Company Closing Statement provided by the SPAC, which comments the SPAC shall deliver to the Company no less than one (1) Business Day prior to the Closing Date, and the Company shall revise such
A-23
Table of Contents
Company Closing Statement to incorporate any changes the Company determines are necessary or appropriate given such comments.
Section 2.08. Exchange Procedures. Upon the terms and subject to the conditions of this Agreement:
(a) As soon as reasonably practicable after the date of this Agreement, the SPAC and the Company shall jointly appoint and engage an exchange agent (the Exchange Agent) for the purpose of, among other things, receiving elections and the exchanges and payments described in this Article 2; provided that the Company may determine in its sole election to act as the Exchange Agent for all purposes hereunder.
(b) Within ten (10) Business Days following the initial filing of the Registration Statement or the Proxy Statement, the Company or the Exchange Agent shall mail or otherwise deliver to each Seller a Letter of Transmittal, which shall specify, among other things, that delivery shall be effected, and risk of loss and title to the Existing Company Shares shall pass, only upon delivery of a completed and duly executed Letter of Transmittal to the Exchange Agent but in no event prior to the Closing. The Exchange Agent shall not issue to any Seller the portion of the Total Consideration to which such Seller is entitled unless such Seller has delivered a completed and duly executed Letter of Transmittal to the Exchange Agent. With respect to any Seller that has not delivered a completed and duly executed Letter of Transmittal to the Exchange Agent at or prior to the Closing, upon delivery of a completed and duly executed Letter of Transmittal to the Exchange Agent after the Closing, the Exchange Agent shall issue such portion of the Total Consideration to which such Seller is entitled pursuant to Section 2.02. With respect to any Seller of Existing Company Shares that delivers a completed and duly executed Letter of Transmittal to the Exchange Agent at or prior to the Closing, the SPAC shall instruct the Exchange Agent to issue to such Seller the portion of the Total Consideration to which such Seller is entitled pursuant to Section 2.02 at or promptly after the Closing.
(c) The SPAC shall enter into an exchange agent agreement with the Exchange Agent, which agreement shall set forth the duties, responsibilities and obligations of the Exchange Agent consistent with the terms of this Agreement, including with regard to the exchanges described in this Article 2.
(d) In furtherance of Section 2.05(d), the SPAC shall deposit or cause to be deposited with the Exchange Agent the cash portion of the Total Consideration and the aggregate amount of the equity portion of the Total Consideration (together, the Exchange Fund).
(e) Any portion of the Exchange Fund (including the proceeds of any investments of the Exchange Fund) that remains unclaimed by the Sellers for one year after the Closing shall be delivered to the SPAC. Any Seller who has not theretofore complied with this Article 2 shall thereafter look only to the SPAC for delivery of such holders Total Consideration, as applicable, upon due surrender of their certificates (or affidavits of loss in lieu as set forth below), in each case, without any interest thereon. Notwithstanding the foregoing, neither the Exchange Agent nor any Party shall be liable to any Person in respect of the consideration payable pursuant to this Agreement delivered to a public official pursuant to any applicable abandoned property, escheat or similar Applicable Law.
ARTICLE 3
REPRESENTATIONS AND WARRANTIES OF SELLERS
With respect to a Supporting Company Holder, except as set forth in such Supporting Company Holders Seller Disclosure Schedule (with respect to each Seller, its Seller Disclosure Schedule), such Supporting Company Holder represents and warrants to the SPAC as of the date hereof and as of the Closing Date, but only insofar as such representations and warranties relate to such Supporting Company Holder, and, with respect to a Joining Seller, except as set forth in such Joining Sellers Seller Disclosure Schedule, such Joining Seller represents and warrants to the SPAC as of the date of execution by such Joining Seller of such Joinder
A-24
Table of Contents
Agreement and as of the Closing Date, but only (i) with respect to Section 3.05 hereof and (ii) insofar as such representations and warranties relate to such Joining Seller, that:
Section 3.01. Corporate Existence and Power.
(a) If such Seller is a natural person, such Seller is of sound mind, has the legal capacity to enter into this Agreement and the other Transaction Agreements to which he or she is a party, has entered into or will enter into this Agreement and the other Transaction Agreements to which he or she is a party on his or her own will, and understands the nature of the obligations to be assumed by him or her under this Agreement and the other Transaction Agreements to which he or she is a party.
(b) If such Seller is not a natural person, such Seller is a corporation, exempted company or limited liability company, as applicable, duly organized, validly existing and in good standing under the Applicable Law of its jurisdiction of organization and has all organizational powers, authority and all governmental licenses, authorizations, permits, consents and approvals required to carry on its business as now conducted, except for those licenses, authorizations, permits, consents and approvals the absence of which would not have, and would not reasonably be expected to have, a Seller Material Adverse Effect.
Section 3.02. Seller Authorization. The execution, delivery and performance by such Seller of this Agreement and each other Transaction Agreement to which such Seller is or will be a party and the consummation of the transactions contemplated hereby and thereby are within such Sellers powers and authority and have been duly authorized by all necessary action on the part of such Seller, and no other proceedings on the part of such Seller are necessary to authorize this Agreement or to consummate the Transactions. This Agreement and the other Transaction Agreements to which it is, or will be a party constitute, or will constitute when entered into, a valid and binding agreement of such Seller, assuming this Agreement and the other Transaction Agreements to which such Seller is or will be a party constitute the valid and binding obligation of the other parties hereto and thereto, enforceable against such Seller in accordance with their terms, except insofar as enforceability may be limited by Enforceability Exceptions.
Section 3.03. Governmental Authorization. The execution, delivery and performance by such Seller of this Agreement and the other Transaction Agreements to which it is a party and the consummation by such Seller of the transactions contemplated hereby requires no consent, approval, authorization, permit, action by or in respect of, or filing with or notification to, any Governmental Authority, other than (i) compliance with any applicable requirements of the HSR Act; (ii) compliance with any applicable requirements of the Securities Act and the Exchange Act; (iii) such actions and filings as may be required by any applicable state securities or blue sky law; (iv) such actions and filings as may be required by any Applicable Law; and (v) any such action or filing the failure of which to make or obtain would not have, and would not reasonably be expected to have, a Seller Material Adverse Effect.
Section 3.04. Noncontravention. The execution, delivery and performance by such Seller of this Agreement and the consummation of the Transactions will not (i) violate the organizational documents of such Seller (if such Seller is not a natural person) and (ii) assuming compliance with the matters referred to in Section 3.03, violate any Applicable Law.
Section 3.05. Ownership and Transfer of Existing Company Shares. Such Seller has good and valid title to, and is the record and beneficial owner of, the number of Existing Company Shares and Loan Notes set forth opposite such Sellers name on Annex I hereto or, with respect to a Joining Seller, on such Joining Sellers Seller Disclosure Schedule, free and clear of any Lien, and, such Seller has the power to sell, transfer, assign and deliver its Existing Company Shares and Loan Notes to the SPAC at the Closing as provided in this Agreement, and such delivery will convey to the SPAC good, valid and marketable title to such Existing Company Shares and Loan Notes, free and clear of all Liens.
A-25
Table of Contents
Section 3.06. Information Supplied. None of the information related to such Seller supplied (or to be supplied prior to the Closing Date) by or on behalf of such Seller expressly for inclusion or incorporation by reference in the Registration Statement or the Proxy Statement will, at the time the Registration Statement is declared effective by the SEC or at the Closing, or at the date the Proxy Statement is mailed to Existing SPAC Stockholders or at the time of the SPAC Special Meeting, contain any untrue statement of a material fact or omit to state any material fact required to be stated therein or necessary in order to make the statements therein, in light of the circumstances under which they are made, not misleading. The Proxy Statement and the Registration Statement, insofar as it relates to information related to such Seller supplied by or on behalf of such Seller expressly for inclusion or incorporation by reference therein, will comply as to form in all material respects with the provisions of the Securities Act, the Exchange Act and the rules and regulations thereunder. Notwithstanding the foregoing, such Seller makes no representation, warranty or covenant with respect to statements made or incorporated by reference therein based on information supplied by the Company or the SPAC for inclusion or incorporation by reference in the Registration Statement, the Proxy Statement or any SEC Documents.
Section 3.07. Finders Fees. Except as set forth on such Sellers Seller Disclosure Schedule, there is no investment banker, broker, finder or other intermediary that has been retained by or is authorized to act on behalf of such Seller who might be entitled to any fee or commission from the Company or any of its Subsidiaries in connection with the Transactions.
Section 3.08. No Other Representations. Such Seller acknowledges that such Seller and its advisors, have made their own investigation of the SPAC and, except as provided in this Agreement, are not relying on any representation or warranty whatsoever as to the condition, merchantability, suitability or fitness for a particular purpose or trade as to any of the assets of the SPAC, the prospects (financial or otherwise) or the viability or likelihood of success of the business of the SPAC as conducted after the Closing, as contained in any materials provided by the SPAC or any of its Affiliates or any of their respective directors, officers, employees, shareholders, partners, members or representatives or otherwise.
ARTICLE 4
REPRESENTATIONS AND WARRANTIES OF THE COMPANY
Except as set forth in the Company Disclosure Schedule (the Company Disclosure Schedule), the Company represents and warrants to the SPAC as of the date hereof and as of the Closing Date that:
Section 4.01. Existence and Power. The Company is a private company limited by shares duly incorporated, validly existing and in good standing under the laws of its jurisdiction of formation and has all corporate powers and all governmental licenses, authorizations, permits, consents and approvals required to carry on its business as now conducted and to own, operate and lease its properties, rights and assets, except for those licenses, authorizations, permits, consents and approvals the absence of which would not, and would not reasonably be expected to, individually or in the aggregate, have a Material Adverse Effect. The Company is duly licensed or qualified to do business as a foreign entity and is in good standing in each jurisdiction in which the ownership of its property or the character of its activities is such that such license or qualification is necessary, except for those jurisdictions where failure to be so licensed or qualified would not, and would not reasonably be expected to, individually or in the aggregate, have a Material Adverse Effect. The Company is not in violation in any material respect of any of the provisions of the Existing Company Articles.
Section 4.02. Authorization. The execution, delivery and performance by the Company of this Agreement and each other Transaction Agreement to which the Company is a party and the consummation of the transactions contemplated hereby and thereby are within the Companys powers and authority and have been duly authorized by all necessary action on the part of the Company, and no other proceedings on the part of the Company are necessary to authorize this Agreement or to consummate the Transactions, subject to (i) due approval by Company shareholders of the Company Articles Amendment and (ii) action by Company
A-26
Table of Contents
shareholders required to approve the Up-C Merger. This Agreement and the other Transaction Agreements to which the Company is a party constitute a valid and binding agreement of the Company, assuming this Agreement and the other Transaction Agreements to which the Company is a party constitute the valid and binding obligation of the other parties hereto and thereto, enforceable against the Company in accordance with their respective terms, except insofar as enforceability may be limited by Enforceability Exceptions.
Section 4.03. Governmental Authorization. The execution, delivery and performance by the Company of this Agreement and the other Transaction Agreements to which it is a party and the consummation of the Transactions require no consent, approval, authorization, permit, action by or in respect of, or filing with or notification to, any Governmental Authority, other than (i) compliance with any applicable requirements of the HSR Act; (ii) compliance with any applicable requirements of the Securities Act and the Exchange Act; (iii) such actions and filings set forth in Section 4.03 of the Company Disclosure Schedule; and (iv) any such action or filing the failure of which to make or obtain would not, and would not reasonably be expected to, individually or in the aggregate, be material to the Company and its Subsidiaries, taken as a whole.
Section 4.04. Noncontravention. The execution, delivery and performance by the Company of this Agreement and the other Transaction Agreements to which it is a party do not and will not, and the consummation of the Transactions will not, (i) conflict with or violate the organizational documents of the Company or any Subsidiary, (ii) assuming compliance with the matters referenced to in Section 4.03 and receipt of the approval of the Companys directors, violate any Applicable Law, (iii) require any consent, waiver or other action by any Person under, violate, result in a breach of or constitute a default (with or without notice or lapse of time, or both) under, or give rise to any right of termination, amendment, cancellation, modification or acceleration of any right or obligation of the Company, any Subsidiary or third party or to a loss or impairment of any benefit or right to which the Company or any Subsidiary is entitled under any provision of any Contract or other instrument binding upon the Company or any Subsidiary or (iv) result in the creation or imposition of any Lien on any asset or properties of the Company or any Subsidiary, except for any Permitted Liens, with such exceptions as would not, and would not reasonably be expected to, in the case of each of clauses (ii) through (iv), individually or in the aggregate, be material to the Company and its Subsidiaries, taken as a whole.
Section 4.05. Capitalization.
(a) Prior to any amendments taken in accordance with the Pre-Closing Step Plan or Closing Step Plan, the share capital of the Company is $100,000 divided into 3,000,000 A Ordinary Shares and 7,000,000 B Ordinary Shares, each with a par value of $0.01. As of the date hereof, there are outstanding 1,483,795 A Ordinary Shares and 5,353,970 B Ordinary Shares.
(b) All outstanding Existing Company Shares are duly authorized, validly issued, fully paid and nonassessable. Except as set forth in this Section 4.05, there are no outstanding (i) shares or voting securities of the Company, (ii) subscriptions, calls, options, warrants, rights (including preemptive rights), puts or other securities of the Company convertible into or exchangeable or exercisable for shares of capital stock or voting or other equity securities of the Company, or any other Contracts to which the Company is a party or by which the Company is bound obligating the Company to issue or sell any shares of, other equity interests in or debt securities of, the Company or (iii) equity equivalents, phantom stock, options, appreciation rights, stock units, profits interests or other rights to acquire from the Company, or other obligation of the Company to issue, any shares, voting or other equity securities or securities convertible into or exchangeable for shares or voting or equity securities of the Company (the items in clauses 4.05(b)(i), 4.05(b)(ii) and 4.05(b)(iii) being referred to collectively as the Company Securities). There are no outstanding obligations of the Company or any Subsidiary to repurchase, redeem or otherwise acquire any Company Securities. There are no outstanding bonds, debentures, notes or other Indebtedness of the Company having the right to vote (or convertible into, or exchangeable for, securities having the right to vote) on any matter for which the Companys shareholders may
A-27
Table of Contents
vote. The Company is not party to any shareholders agreement, voting agreement, proxies, registration rights agreement or other similar agreements relating to its equity interests.
Section 4.06. Subsidiaries.
(a) The Companys direct and indirect Subsidiaries, together with their jurisdiction of incorporation or organization, as applicable, are listed on Section 4.06(b) of the Company Disclosure Schedule. Each Subsidiary is a business entity that is duly organized, validly existing and in good standing under the laws of its jurisdiction of organization and has all organizational powers and all governmental licenses, authorizations, permits, consents and approvals required to carry on its business as now conducted, and to own, operate and lease its properties, rights and assets, and is duly licensed or qualified to do business as a foreign entity and is in good standing in each jurisdiction in which the ownership of its property or the character of its activities is such that where such license or qualification is necessary, except for those licenses, authorizations, consents and approvals the absence of which would not, or would not reasonably be expected to, individually or in the aggregate, be material to the Company and its Subsidiaries, taken as a whole. Complete and correct copies of the organizational documents of each such Subsidiary have been made available to the SPAC. No such Subsidiary is in violation in any material respect of any of the provisions of its organizational documents.
(b) All of the issued share capital, stock or other voting or equity securities of each Subsidiary have been duly authorized and validly issued and are fully paid and nonassessable. All of the ownership interests in each Subsidiary are owned by the Company, directly or indirectly, free and clear of any Lien and free of any other limitation or restriction (including any restriction on the right to vote, sell or otherwise dispose of such ownership interests) and have not been issued in violation of preemptive or similar rights. There are no outstanding (i) subscriptions, calls, options, warrants, rights (including preemptive rights), puts or other securities of any Subsidiary convertible into or exchangeable or exercisable for shares or voting or equity securities of any Subsidiary, or any other Contracts to which the Company or any Subsidiary is a party or by which the Company or any Subsidiary is bound obligating the Company or any Subsidiary to issue or sell any shares of, other equity interests in or debt securities of, any Subsidiary or (ii) equity equivalents, phantom stock, options, appreciation rights, stock units, profits interests or other rights to acquire from the Company or any Subsidiary, or other obligation of the Company or any Subsidiary to issue, any shares, voting or equity securities or securities convertible into or exchangeable for shares or voting or equity securities of any Subsidiary (the items in clauses 4.06(b)(i) and 4.06(b)(ii) being referred to collectively as the Subsidiary Securities). There are no outstanding obligations of the Company or any Subsidiary to repurchase, redeem or otherwise acquire any outstanding Subsidiary Securities. Except for the Subsidiary Securities, neither the Company nor any of its Subsidiaries owns any equity, ownership, profit, voting or similar interest in or any interest convertible, exchangeable or exercisable for, any equity, profit, voting or similar interest in, any Person. There are no outstanding bonds, debentures, notes or other Indebtedness of any Subsidiary having the right to vote (or convertible into, or exchangeable for, securities having the right to vote) on any matter for which such Subsidiarys shareholders may vote. No Subsidiary is party to any shareholders agreement, voting agreement, proxies, registration rights agreement or other similar agreements relating to its equity interests.
Section 4.07. Information Supplied. None of the information supplied or to be supplied by or on behalf of the Company prior to the Closing Date related to the Company expressly for inclusion or incorporation by reference in the Registration Statement or the Proxy Statement will, at the time the Registration Statement is declared effective by the SEC or at the Closing, or the Proxy Statement at the date it is mailed to Existing SPAC Stockholders or at the time of the SPAC Special Meeting, contain any untrue statement of a material fact or omit to state any material fact required to be stated therein or necessary in order to make the statements therein, in light of the circumstances under which they are made, not misleading. The Registration Statement and the Proxy Statement, insofar as it relates to information related to the Company supplied by or on behalf of the Company expressly for inclusion or incorporation by reference therein, will comply as to form in all material respects with the provisions of the Securities Act, the Exchange Act and the rules and regulations thereunder. Notwithstanding the foregoing, the Company makes no representation, warranty or covenant with respect to statements made or
A-28
Table of Contents
incorporated by reference therein based on information supplied by any Seller or the SPAC for inclusion or incorporation by reference in the Registration Statement, Proxy Statement or any SEC Documents.
Section 4.08. Financial Statements.
(a) Attached to Section 4.08(a) of the Company Disclosure Schedule are true, correct, accurate and complete copies of (i) the audited consolidated balance sheets of the Company and its Subsidiaries as of June 30, 2020 and 2019 and the related audited consolidated statements of operations, comprehensive loss, stockholders deficit and cash flows for the years ended June 30, 2020, 2019 and 2018 (collectively, the Audited Financial Statements), and (ii) the unaudited interim consolidated balance sheet as of the Company and its Subsidiaries as of March 31, 2021 (the Balance Sheet Date) and the related unaudited interim consolidated statements of operations, comprehensive loss, stockholders deficit and cash flows for the nine (9) months ended March 31, 2021 of the Company and its Subsidiaries (the Financial Statements). The Financial Statements (x) fairly present, in conformity with GAAP applied on a consistent basis (except as may be indicated in the notes thereto), the consolidated financial position of the Company and its Subsidiaries as of the dates thereof and their consolidated results of operations and cash flows for the periods then ended; provided that such unaudited interim consolidated financial statements do not include all of the notes or the information contained in such notes as required by GAAP for complete financial statements and are subject to normal year-end adjustments and do not present information with respect to general and administrative expenses or depreciation, in each case as required by GAAP. The Audited Financial Statements were audited, in all material respects, in accordance with the standards of the American Institute of Certified Public Accountants (except as may be indicated in the notes thereto).
(b) The Company has established and maintained internal controls that are sufficient to provide reasonable assurance (i) that transactions, receipts and expenditures of the Company and its Subsidiaries are being executed and made only in accordance with appropriate authorizations of management of the Company, (ii) that transactions are recorded as necessary to permit preparation of financial statements in conformity with GAAP and to maintain accountability for assets, (iii) regarding prevention or timely detection of unauthorized acquisition, use or disposition of the assets of the Company and its Subsidiaries and (iv) that accounts, notes and other receivables and inventory are recorded accurately. The Company has not identified or been made aware of, and has not received from its independent auditors any notification of, any (x) significant deficiency in the internal controls over financial reporting of the Company or any of its Subsidiaries, (y) material weakness in the internal controls over financial reporting of the Company or any its Subsidiaries or (z) fraud, whether or not material, that involves management or other employees of the Company or any of its Subsidiaries who have a role in the internal controls over financial reporting of the Company or any of its Subsidiaries.
(c) There are no outstanding loans or other extensions of credit made by the Company or any of its Subsidiaries to any executive officer (as defined in Rule 3b-7 under the Exchange Act) or director of the Company.
Section 4.09. Absence of Certain Changes.
(a) Since the Balance Sheet Date, except as contemplated by this Agreement or any of the other Transaction Agreements, the business of the Company and its Subsidiaries has been conducted in the ordinary course consistent with past practice, and neither the Company nor any of its Subsidiaries has taken or permitted to occur any action that, were it to be taken from and after the date hereof, would require the prior written consent of the SPAC pursuant to clauses (a), (b), (c), (d), (i), (j), (k), (o) or (s) of Section 6.01.
(b) Since the Balance Sheet Date, no Material Adverse Effect has occurred.
A-29
Table of Contents
Section 4.10. No Undisclosed Material Liabilities. There are no liabilities of the Company or any Subsidiary of any kind (whether direct or indirect, absolute, accrued, contingent or otherwise) required by GAAP to be disclosed on a balance sheet, other than:
(a) liabilities provided for on the audited consolidated balance sheet as of June 30, 2020 or disclosed in the notes thereto, or on the unaudited interim consolidated balance sheet as of March 31, 2021;
(b) liabilities disclosed in, related to or arising under any agreements, instruments or other matters disclosed in this Agreement or any Schedule hereto, or incurred in connection with the Transactions;
(c) liabilities incurred in the ordinary course of business consistent with past practice since June 30, 2020; or
(d) liabilities which would not, individually or in the aggregate, be material to the Company and its Subsidiaries, taken as a whole.
Section 4.11. Material Contracts. (a) Section 4.11 of the Company Disclosure Schedule sets forth a true, correct and complete list of the following (collectively, the Material Contracts):
(i) any lease or sublease (whether of real or personal property) providing for annual rentals of $500,000 or more that cannot be terminated on sixty (60) days notice or less without payment by the Company or any Subsidiary of any material penalty;
(ii) any Contract for the purchase of materials, supplies, goods, services, equipment or other assets providing for either (A) annual payments by the Company and its Subsidiaries of $1,000,000 or more or (B) aggregate payments by the Company and its Subsidiaries of $5,000,000 or more, in each case that cannot be terminated on sixty (60) days notice or less without payment by the Company or any Subsidiary of any material penalty;
(iii) any sales, distribution or other similar Contracts providing for the sale by the Company or any Subsidiary of materials, supplies, goods, services, equipment or other assets that provides for annual payments to the Company and the Subsidiaries of $5,000,000 or more;
(iv) any partnership, joint venture or other similar Contract or arrangement;
(v) any Contracts for the sale of any of the business, properties or assets of the Company or any of its Subsidiaries or the acquisition by the Company or any of its Subsidiaries of any operating business, properties or assets, whether by merger, purchase or sale of stock or assets or otherwise, in each case involving consideration therefor in an amount in excess of $5,000,000 (other than Contracts for the purchase of inventory or supplies entered into in the ordinary course of business consistent with past practice);
(vi) any obligation to make payments, contingent or otherwise, arising out of the prior acquisition of the business, assets or stock of other Persons;
(vii) any agreement relating to Indebtedness for borrowed money or the deferred purchase price of property (in either case, whether incurred, assumed or guaranteed by the Company or any of its Subsidiaries or secured by any asset), except any such agreement with an aggregate outstanding principal amount not exceeding $1,500,000;
(viii) any Contract that that purports to limit (A) the localities in which the Company and its Subsidiaries businesses are conducted, (B) the Company or any of its Subsidiaries from engaging in any line of business or (C) the Company or any of its Subsidiaries from developing, marketing or selling products or services, in each case, in any manner that is material to the Company and its Subsidiaries, taken as a whole, including any non-compete agreements or agreements limiting the ability of any of Company or any of its Subsidiaries from soliciting customers or employees, in a manner that is material to the Company and its Subsidiaries, taken as a whole;
(ix) any collective bargaining agreement or other material Contract with any labor organization, union, association, works council or similar entity in respect of employees of the Company or any of its Subsidiaries;
A-30
Table of Contents
(x) any agreement with any Related Party;
(xi) any (A) Outbound IP License (other than non-exclusive licenses granted to customers or vendors in the ordinary course of business) or (B) Inbound IP License (other than (x) contracts for any Software that is commercially available on standard and non-negotiated terms for an annual or aggregate license fee of no more than $100,000 or (y) licenses to Open Source Software);
(xii) any agreement with any Governmental Authority-funded academic institution or research center or Governmental Authority that provides for the provision of funding to the Company or any Subsidiary for research and development or similar activities involving the creation of any material Intellectual Property Rights or other assets and that has resulted in such Governmental Authority, research center or academic institution having any ownership of or step-in ownership right to any such Intellectual Property Rights;
(xiii) any Contract with the top twenty (20) customers of the Company and its Subsidiaries, taken as a whole (the Material Customers) and top twenty (20) suppliers and distributors of the Company and its Subsidiaries, taken as a whole (the Material Suppliers) as determined by revenue and dollar volume of payments, respectively, in each case during the twelve (12)-month period prior to the date of this Agreement;
(xiv) any Contract (other than those made in the ordinary course of business) providing for the grant of any preferential rights to purchase or lease any asset of the Company;
(xv) any Contract that imposes obligations on the Company or any of its Subsidiaries to provide most favored nation pricing to any of its customers, or that contains any take or pay or minimum requirements with any of its suppliers, right of first refusal or other similar provisions with respect to any transaction engaged in by the Company or any of its Subsidiaries;
(xvi) any material Contract (other than those made in the ordinary course of business consistent with past practice) with any Governmental Authority; or
(xvii) any commitment to enter into any Contract of the type described in this Section 4.11(a).
(b) Except as would not, or would not reasonably be expected to, individually or in the aggregate, be material to the Company and its Subsidiaries, taken as a whole, each Material Contract is in full force and effect and represents a legal, valid and binding obligation of the Company or applicable Subsidiary party thereto and, to the knowledge of the Company, represents a legal, valid and binding obligation of the counterparties thereto, except insofar as enforceability may be limited by Enforceability Exceptions. True, correct and complete copies of all Material Contracts have been made available to the SPAC.
(c) Neither the Company nor, to the knowledge of the Company, any other party thereto, is in breach of or in default under, and no event has occurred which with notice or lapse of time or both would become a breach of or default under, any Material Contract, and no party to any Material Contract has given any written notice of any claim of any such breach, default or event, which, individually or in the aggregate, are reasonably likely to be material to the Company and its Subsidiaries, taken as a whole.
Section 4.12. Litigation. There is no, and for the past three (3) years has been no, (a) Action, suit, investigation or proceeding pending, or, to the knowledge of the Company, threatened, against or affecting the Company or any Subsidiary or any of their respective properties or assets or any of the directors or officers of the Company or any of its Subsidiaries with regard to their actions as such, and, to the knowledge of the Company, no facts exist that would reasonably be expected to form the basis for any such Action; (b) pending or, to the knowledge of the Company, threatened in writing, audit, examination or investigation by any Governmental Authority against the Company or any of its Subsidiaries or any of their respective properties or assets, or any of the directors, managers or officers of the Company or any of its Subsidiaries with regard to their actions as such, and, to the knowledge of the Company, no facts exist that would reasonably be expected to form the basis for any such audit, examination or investigation; (c) pending or threatened Action by the Company or any of its
A-31
Table of Contents
Subsidiaries against any third party; (d) settlement or similar agreement that imposes any material ongoing obligation or restriction on the Company or any of its Subsidiaries; and (e) award, injunction, judgment, regulatory or supervisory mandate, order, writ, decree or ruling entered, issued, made, or rendered by any Governmental Authority imposed or, to the knowledge of the Company, threatened to be imposed upon the Company or any of its Subsidiaries or any of its respective properties or assets, or any of the directors, managers or officers of the Company or any of its Subsidiaries with regard to their actions as such except as would not, or would not reasonably be expected to, individually or in the aggregate, be material to the Company and its Subsidiaries, taken as a whole.
Section 4.13. Compliance with Laws and Court Orders. Neither the Company nor any Subsidiary is, nor for the past five (5) years has been, in violation of any Applicable Law, except for violations that have not been, or would not reasonably be expected to be, individually or in the aggregate, material to the Company and its Subsidiaries, taken as a whole. No written or, to the knowledge of the Company, oral notice of non-compliance with any Applicable Law has been received by the Company or any of its Subsidiaries in the past five (5) years. The Company and each of its Subsidiaries is in possession of all franchises, grants, authorizations, licenses, permits, consents, certificates, approvals and orders from Governmental Entities (Approvals) necessary to own, lease and operate the properties it purports to own, operate or lease and to carry on its business as it is now being conducted, except where the failure to have such Approvals would not, or would not reasonably be expected to, individually or in the aggregate, be material to the Company and its Subsidiaries, taken as a whole. Each Approval held by the Company or any of its Subsidiaries, as applicable, is valid, binding and in full force and effect in all material respects. None of the Company or any of its Subsidiaries (i) are in default or violation (and no event has occurred that, with notice or the lapse of time or both, would constitute a default or violation) of any material term, condition or provision of any such Approval, or (ii) have received any notice from a Governmental Authority that has issued any such Approval that it intends to cancel, terminate, modify or not renew any such Approval, except in the case of clauses (i) and (ii) as would not, or would not reasonably be expected to, individually or in the aggregate, be material to the Company and its Subsidiaries, taken as a whole.
Section 4.14. Real Property.
(a) Section 4.14(a) of the Company Disclosure Schedule sets forth a complete and accurate list of each Owned Real Property. The Company or one of its Subsidiaries owns and has good, valid, and marketable fee simple title to the Owned Real Property, free and clear of all Liens other than Permitted Liens. There are no outstanding options, rights of first offer or rights of first refusal by any third party to lease or purchase the Owned Real Property or any portion thereof or interest therein. Except as would not be material to the Company or its Subsidiaries, all buildings, structures, improvements, fixtures, building systems and equipment, and all components thereof, located on the Owned Real Property are in good operating condition and repair (normal wear and tear expected) and are sufficient for the use and occupancy of the Owned Real Property as currently conducted.
(b) Section 4.14(b) of the Company Disclosure Schedule sets forth a complete and accurate list of each Leased Real Property. The Company has made available to the SPAC true, correct and complete copies of the leases (including all modifications, amendments, guarantees, supplements, waivers and side letters thereto) pursuant to which the Company or any of its Subsidiaries occupies (or has been granted an option to occupy) the Leased Real Property or is otherwise a party with respect to the Leased Real Property (the Leases). The Company and its Subsidiaries have valid and subsisting leasehold estates in, and enjoy peaceful and undisturbed possession of, all Leased Real Property, free and clear of all Liens, subject only to Permitted Liens. With respect to each Lease, (i) such Lease is valid, binding and enforceable and in full force and effect against the Company or a Subsidiary and, to the Companys knowledge, the other party thereto in accordance with its terms, subject to the Enforceability Exceptions, (ii) each Lease has not been materially amended or modified except as reflected in the modifications, amendments, supplements, waivers and side letters made available to the SPAC, (iii) neither the Company nor one of its Subsidiaries has received or given any written notice of default or breach under any of the Leases and, to the knowledge of the Company, neither the Company nor any of its Subsidiaries has
A-32
Table of Contents
received oral notice of any default that has not been cured within the applicable cure period and (iv) there does not exist under any Lease any event or condition which, with notice or lapse of time or both, would become a material default by the Company or any of its Subsidiaries or, to the Companys knowledge, the other party thereto. Each Leased Real Property is in good condition and repair (normal wear and tear excepted) and is sufficient for the operation of the business as currently conducted.
(c) Neither the Company nor any of its Subsidiaries has a written sublease granting any Person (other than another Subsidiary of the Company) the right to use or occupy Leased Real Property which is still in effect.
(d) Neither the Company nor its Subsidiaries has collaterally assigned or granted any other security interest in the Leased Real Property or any interest therein which is still in effect.
(e) Neither the Company nor any of its Subsidiaries is in default or violation of, or not in material compliance with, any legal requirements applicable to its occupancy of the Owned Real Property and the Leased Real Property.
(f) No construction or expansion is currently being performed or is planned for 2021 at any of the Owned Real Property or Leased Real Property that is expected to result in liability to the Company or any of its Subsidiaries in excess of $1,000,000 in such calendar year.
(g) Neither the whole nor any part of the Owned Real Property is subject to any pending suit for condemnation or other taking by any government authority, and, to the knowledge of the Company, no such condemnation or other taking is threatened or contemplated.
Section 4.15. Intellectual Property. (a) Section 4.15(a) of the Company Disclosure Schedule contains a complete and accurate list of all issuances, registrations and applications therefor in respect of any Patents, Trademarks, Copyrights and Internet domain names included in the Owned Intellectual Property Rights (collectively, Registered Intellectual Property Rights), including, for each item of such Registered Intellectual Property Rights, the record owner of such item, the jurisdiction in which such item has been issued, registered, or filed, and the issuance, registration or application number and date for such item. With respect to all material Registered Intellectual Property Rights, all necessary registration, maintenance, renewal, and other relevant filing fees due through the date hereof have been timely paid and all necessary documents and certificates in connection therewith have been timely filed (except for any failure that has been, or can be, cured by subsequent payment of such fees or the filing of such documents or certificates) with the relevant Patent, Trademark, Copyright, Internet domain name, or other authorities in the United States or foreign jurisdictions, as the case may be, for the purposes of maintaining the Registered Intellectual Property Rights in full force and effect. All Registered Intellectual Property Rights are subsisting and, to the knowledge of the Company, valid and enforceable.
(b) (i) (A) Except as set forth on Section 4.15(b)(i)(A) of the Company Disclosure Schedule, the Company or one of its Subsidiaries solely and exclusively owns all rights, title and interest in and to all material Owned Intellectual Property Rights, free and clear of all Liens (except Permitted Liens) and (B) the Company and its Subsidiaries have a valid right to use pursuant to a valid written IP License all other Intellectual Property Rights used in and necessary for the conduct and operation of their respective businesses (the Licensed Intellectual Property Rights) and (ii) the Owned Intellectual Property Rights and Licensed Intellectual Property Rights constitute (when used within the scope of the applicable IP License) all of the Intellectual Property Rights used or practiced in, held for use or practice in or necessary for the operation of the respective business of the Company and each of its Subsidiaries as currently conducted.
(c) None of the Company or any of its Subsidiaries, nor the conduct of their business, has infringed, misappropriated, diluted or otherwise violated, or does infringe, misappropriate, dilute or otherwise violate any Intellectual Property Rights of any third party in any material respect. No Actions are pending, threatened in
A-33
Table of Contents
writing or, to the knowledge of the Company, otherwise threatened against the Company or any of its Subsidiaries by any third party (i) claiming infringement, misappropriation or other violation of Intellectual Property Rights owned by such third party by the Company or any of its Subsidiaries or (ii) challenging the ownership, validity or enforceability of any Owned Intellectual Property Rights. To the knowledge of the Company, no third party is currently infringing any Owned Intellectual Property Rights in any material respect and, to the knowledge of the Company, there are no facts or circumstances that would form the basis for any claim or challenge alleging any such material infringement.
(d) The Company and each of its Subsidiaries have taken, and take, commercially reasonable steps to preserve, protect and maintain the secrecy, confidentiality and value of all material Trade Secrets or confidential and proprietary information (i) included in the Owned Intellectual Property Rights or (ii) owned by any third party to whom the Company or any of its Subsidiaries has a confidentiality obligation. No material Trade Secret or confidential and proprietary information has been authorized to be disclosed or, to the knowledge of the Company, has been actually disclosed to any third party other than pursuant to a written confidentiality contract restricting the disclosure and use thereof.
(e) The Company and each of its Subsidiaries have entered into valid and enforceable written contracts with all past and current employees, contractors and consultants who have been employed, engaged or otherwise retained at by the Company or any of its Subsidiaries and who have contributed to the discovery, conception, development, creation, or reduction to practice of any material Intellectual Property Rights for or on behalf of the Company or any of its Subsidiaries or otherwise within the scope of such employment, engagement or retention, pursuant to which each such Person, to the Companys knowledge, effectively and validly, assigns to the Company or its applicable Subsidiary all of such Persons right, title, and interest in and to all such Intellectual Property Rights (collectively, the Personnel IP Contracts), except to the extent such rights automatically vest in the Company by operation of law. To the knowledge of the Company, all Personnel IP Contracts are in full force and effect and have not suffered a material default, violation or breach.
(f) The Company and each of its Subsidiaries have taken and take commercially reasonable steps to protect and maintain the performance, confidentiality, and security of all IT Systems (and all Software, information and data stored or contained therein or transmitted thereby). The IT Systems are adequate and sufficient for the operation of the respective businesses of the Company and each of its Subsidiaries in all material respects. To the knowledge of the Company, there have been no security breaches or unauthorized use, access or intrusions of any IT Systems.
(g) Except as set forth in Section 4.15(g) of the Company Disclosure Schedule, all Software owned or purported to be owned by the Company or developed by the Company (Company Software) was created solely by employees, contractors or consultants of the Company or its Subsidiaries within the scope of their employment, engagement or retention with or by the Company or any of its Subsidiaries.
(h) To the knowledge of the Company, the Company and each of its Subsidiaries have complied and complies with all license terms applicable to any Open Source Software that is included, incorporated or embedded in, linked to, combined or distributed with, or used in the delivery or provision of any material Company Software. To the knowledge of the Company, no Open Source Software is included, incorporated or embedded in, linked to, combined or distributed with or used in the delivery or provision of any material Company Software, in each case, in a manner that requires that any material Company Software be (i) made available or distributed in source code form, (ii) licensed for the purpose of making derivative works, (iii) licensed under terms that allow reverse engineering, reverse assembly or disassembly of any kind or (iv) redistributable at no charge.
(i) None of the source code or related source materials for any Company Software has been provided to, or to the Companys knowledge, used or accessed by, any Person other than employees, consultants or contractors of the Company or any of its Subsidiaries who have entered into written confidentiality Contracts with respect to
A-34
Table of Contents
such source code or related source materials. None of the Company or any of its Subsidiaries is a party to any source code escrow contract or any other contract (or a party to any contract obligating the Company to enter into a source code escrow contract or any other contract) requiring the deposit of any source code or related source materials for any Company Software, and no source code or related source materials for any Company Software has been placed into escrow for the benefit of any third party.
(j) All Company Software is sufficient in all material respects for the purposes for which it is used in the respective businesses of the Company and each of its Subsidiaries. The Company and each of its Subsidiaries have implemented industry standard procedures to mitigate against the likelihood that any Company Software contains any (i) contaminant or other Software routines or hardware components designed to permit unauthorized access to or disable, erase or otherwise harm Software, hardware or data or (ii) spyware or trackware (as such terms are commonly understood in the software industry).
(k) No funding, facilities or resources of any Governmental Authority or any research or academic institution was used in the creation or development of any Owned Intellectual Property Rights in a manner that has resulted in such Governmental Authority or research or academic institution having any ownership of or step-in ownership right to any such Intellectual Property Rights.
Section 4.16. Insurance Coverage. Section 4.16 of the Company Disclosure Schedule contains a true, correct and complete list of each material insurance policy maintained by the Company and its Subsidiaries as of the date of this Agreement, copies of which have been made available to the SPAC. All such policies are in full force and effect, and there are no material claims by the Company or any of its Subsidiary pending under any of such policies as to which coverage has been questioned, denied or disputed by the underwriters of such policies or bonds or in respect of which such underwriters have reserved their rights. To the knowledge of the Company, the coverages provided by such insurance policies or fidelity or surety bonds are usual and customary in amount and scope for the Company and its Subsidiaries business and operations as concurrently conducted, and sufficient to comply with any insurance required to be maintained by Material Contracts.
Section 4.17. Employees.
(a) Section 4.17(a) of the Company Disclosure Schedule sets forth a true and complete list, as of the date hereof, of the names (or anonymized with employee numbers if required by Applicable Law), titles, annual salaries and incentive compensation opportunity of all officers of the Company and its Subsidiaries, including, for the avoidance of doubt, any individuals appointed to a management board, and all other Key Employees.
(b) Except as disclosed on Section 4.17(b) of the Company Disclosure Schedule, neither the Company nor its Subsidiaries is a party to or bound by any labor agreement, collective bargaining agreement or other labor Contract applicable to persons employed by the Company and its Subsidiaries. Except as disclosed on Schedule 4.17(b) of the Company Disclosure Schedule, no employees of the Company or its Subsidiaries are represented by any labor union, labor organization, works council or other employee representative body with respect to their employment with the Company or its Subsidiaries. There are no representation proceedings or petitions seeking a representation proceeding presently pending or, to the knowledge of the Company, threatened to be brought or filed, with the National Labor Relations Board or other labor relations tribunal or other similar body, nor has any such representation proceeding, petition, or demand been brought, filed, made, or, to the knowledge of the Company, threatened in the last three (3) years. In the past three (3) years, there have been no labor organizing activities (including, without limitation, any requests or applications for the recognition or establishment of any trade unions, works councils or other employee representative bodies) involving the Company or its Subsidiaries with respect to any employees of the Company and its Subsidiaries or, to the knowledge of the Company, threatened by any labor organization, works council or group of employees.
(c) In the past three (3) years, there have been no strikes, work stoppages, slowdowns, lockouts or other material labor disputes, arbitrations, grievances or unfair labor practice charges pending or, to the knowledge of
A-35
Table of Contents
the Company, threatened in writing against or affecting the Company or its Subsidiaries involving any current or former Service Provider. There are no material charges, grievances or complaints, in each case related to alleged unfair labor practices of the Company or any of its Subsidiaries, pending or, to the knowledge of the Company, threatened in writing by or on behalf of any current or former Service Provider or any labor organization. There are no continuing obligations of the Company or its Subsidiaries pursuant to the resolution of any such proceeding that is no longer pending.
(d) As of the date of this Agreement, none of the Companys officers or other Key Employees has given written notice of any intent to terminate his or her employment with the Company or its Subsidiaries as a result of the consummation of the Transactions or otherwise within one (1) year of the Closing Date. The Company and its Subsidiaries are in material compliance and, to the knowledge of the Company, each Key Employee is in material compliance with the terms of any employment, nondisclosure, and restrictive covenant agreements between the Company and its Subsidiaries and such individuals.
(e) To the knowledge of the Company, no written notice has been received by the Company or its Subsidiaries in the past three (3) years asserting or alleging discrimination, harassment or other similar misconduct against any officer, director, executive or other Key Employee of the Company or its Subsidiaries in his or her capacity as an officer, director, executive or Key Employee or in any other capacity.
(f) Except as disclosed on Section 4.17(f) of the Company Disclosure Schedule, there are no material complaints, charges, proceedings, investigations, or claims against the Company or its Subsidiaries pending or, to the knowledge of the Company, threatened that could be brought or filed, with any Governmental Authority or any relevant court or tribunal based on, arising out of, in connection with or otherwise relating to the employment, engagement, or termination of employment or engagement or failure to employ or engage by the Company and its Subsidiaries, of any Service Provider. The Company and its Subsidiaries are in material compliance with all Applicable Law respecting employment, employment practices and social security rules, including, without limitation, all laws respecting terms and conditions of employment, the classification and payment of employees, workers, and/or individual contractors and consultants, wages, profit-sharing, bonus plans, working-time, the Worker Adjustment and Retraining Notification Act and any similar foreign, state or local mass layoff or plant closing laws (collectively, WARN) or other mass layoff, plant closing, collective redundancies or similar with respect to the Company or its Subsidiaries, collective bargaining, company-wide agreements, social contributions and social declarations, immigration, benefits, labor relations, discrimination, harassment, civil rights, safety and health and workers compensation. There are no material pending, or to the knowledge of the Company, threatened, Actions against the Company or any of its Subsidiaries in respect of any current or former Service Provider.
(g) Except as disclosed on Section 4.17(g) of the Company Disclosure Schedule, there has been no mass layoff or plant closing or similar actions (as defined by WARN) within the six (6) months prior to the Closing.
(h) The Company and its Subsidiaries maintain, and in the past three (3) years have maintained, an affirmative action plan where required by Applicable Law.
(i) Neither the Company nor its Subsidiaries is liable for any arrears of wages, fees, benefits or other compensation or penalties with respect thereto, except in each case as would not be material to the Company and its Subsidiaries taken as a whole.
(j) To the knowledge of the Company, neither the execution or delivery of this Agreement nor the consummation of the Transactions contemplated by this Agreement will result in any material breach or other violation of any collective bargaining agreement, employment agreement, consulting agreement, or any other labor-related agreement to which the Company or its Subsidiaries are a party or bound. The Company and its Subsidiaries have satisfied in all material respects any pre-signing legal or contractual requirement to provide
A-36
Table of Contents
notice to, seek the opinion of, provide a right of advice to or enter into any consultation procedure with, any labor union, labor organization, works council or other employee representative body, which is representing any employee of the Company or its Subsidiaries, in connection with the execution of this Agreement or the Transactions contemplated by this Agreement.
(k) The mass layoff (so-called plan de sauvegarde de lemploi) currently being implemented by Mirion Technologies (Canberra) SAS in France is in compliance with Applicable Law.
(l) Except as disclosed on Section 4.17(l) of the Company Disclosure Schedule, the Company and its Subsidiaries do not have any material outstanding obligations as a result of settlement agreements entered into with their respective employees.
(m) The Company has provided to the SPAC copies of all executive officer, director and material Key Employees employment contracts and consultancy agreements in existence between the Company or its Subsidiaries.
Section 4.18. Employee Benefit Plans. (a) Section 4.18(a) of the Company Disclosure Schedule sets forth a true, correct and complete list of each material Employee Plan. The Company has provided to the SPAC complete copies of each material Employee Plan and all amendments thereto, and to the extent applicable: (i) all plan documents, including any related trust documents, insurance contracts or other funding arrangements, and all amendments thereto; (ii) for the most recent plan year: (A) the IRS Form 5500 and all schedules thereto or other applicable annual report where applicable; (B) audited financial statements; and (C) actuarial or other valuation reports; (iii) the most recent IRS determination letter or opinion letter, as applicable, or applicable letter of good standing or other confirmation from the relevant tax authority in relation to an Employee Plan subject to the Applicable Law of a jurisdiction outside the United States; (iv) any material, non-routine correspondence with any Governmental Authority regarding any Employee Plan during the past three (3) years; (v) the most recent summary plan descriptions or employee booklet where applicable and (vi) the past ten (10) years of contribution history to the IUE-CWA Pension Fund or any other multiemployer plan to which the Company or an of its Subsidiaries contributes or is obligated to contribute.
(b) None of the Company, any ERISA Affiliate or any predecessor thereof sponsors, maintains or contributes to, or has in the past sponsored, maintained or contributed to, any plan or arrangement subject to Title IV of ERISA. Except as set forth in Section 4.18(b) of the Company Disclosure Schedule, as of the date hereof, the fair market value of the assets of each Employee Plan subject to Title IV of ERISA, Section 412 of the Code, Section 302 of ERISA or a defined benefit plan not subject to ERISA (other than a multiemployer plan, as defined in Section 3(37) of ERISA) (a Title IV Plan) (excluding for these purposes any accrued but unpaid contributions) exceeded the present value of all benefits accrued under such Title IV Plan determined using the assumptions used for plan funding purposes. Neither the Company nor any ERISA Affiliate thereof has incurred, or reasonably expects to incur prior to the Closing Date (i) any liability under Title IV of ERISA arising in connection with the termination of, or a complete or partial withdrawal from, any plan covered or previously covered by Title IV of ERISA or (ii) any liability under Section 4971 of the Code that in either case could become a liability of the SPAC or any of its ERISA Affiliates after the Closing Date. To the knowledge of the Company, in the event the Company or any ERISA Affiliate were to withdraw from any multiemployer plan to which it currently contributes, the withdrawal liability would not reasonably be expected to be in excess of $1,000,000. No multiemployer plan to which the Company or any of its ERISA Affiliates contributes or has liability has been determined by an applicable Governmental Authority to be in endangered, critical, or critical and declining status or subject to a rehabilitation plan, and neither the Company nor any of its ERISA Affiliates has withdrawn from such a plan or has liability other than the obligation to make ordinary course contributions. No Employee Plan is a registered pension plan as that term is defined in the section 248(1) of the Income Tax Act (Canada).
(c) None of the Company, any ERISA Affiliate or any predecessor thereof contributes to, or has in the past six (6) years contributed to, any (i) multiemployer plan, as defined in Section 3(37) of ERISA; (ii) a multiple
A-37
Table of Contents
employer plan as defined in Section 413(c) of the Code; or (iii) a multiple employer welfare arrangement within the meaning of Section 3(40) of ERISA.
(d) Each Employee Plan which is intended to be qualified under Section 401(a) of the Code has received a favorable determination letter or is entitled to rely on an advisory or opinion letter from the Internal Revenue Service, or has pending or has time remaining in which to file an application for such determination from the Internal Revenue Service, and any trust intended to be exempt from federal income taxation under the provisions of Section 501(a) of the Code is so exempt, and, to the knowledge of the Company, no circumstances exist with respect to any such Employee Plan which would reasonably be expected to cause the loss of such qualification or exemption. Each Employee Plan has been maintained in all material respects in compliance with its terms and with the requirements prescribed by any and all statutes, orders, rules and regulations, including ERISA and the Code, which are applicable to such Employee Plan. To the knowledge of the Company, no events have occurred within the past six (6) years with respect to any Employee Plan that could result in payment or assessment by or against the Company of any material excise taxes under Sections 4972, 4975, 4976, 4977, 4979, 4980B, 4980D, 4980E or 5000 of the Code, except as would not be material to the Company taken as a whole. In the past six (6) years, no non-exempt prohibited transaction (within the meaning of Section 406 of ERISA and Section 4975 of the Code) has occurred or is reasonably expected to occur with respect to any Employee Plan.
(e) Neither the Company nor any Subsidiary has any current or projected liability in respect of post-employment or post-retirement health or medical or life insurance benefits for retired, former or current Service Providers, except as may be required under the Consolidated Omnibus Budget Reconciliation Act of 1985, as amended, or similar state or other Applicable Law and at the sole expense of such participant or the participants beneficiary.
(f) No material actions, suits, claims (other than routine claims for benefits in the ordinary course), audits, inquiries, proceedings or lawsuits are pending, or, to the knowledge of the Company, threatened against any Employee Plan, the assets of any of the trusts under such plans or the plan sponsor or administrator, or against any fiduciary of any Employee Plan with respect to the operation thereof. No event has occurred, and to the knowledge of the Company, no condition exists that would, by reason of the Companys affiliation with any of its ERISA Affiliates, subject the Company to any material tax, fine, lien, penalty or other liability imposed by ERISA, the Code or other Applicable Law.
(g) All contributions, reserves or premium payments required to be made or accrued as of the date hereof to the Employee Plans and Government Plans have been timely made or accrued in all material respects.
(h) Neither the execution and delivery of this Agreement nor the consummation of the Closing will, either alone or in connection with any other event(s): (i) result in any payment or benefit becoming due to any current or former Service Provider or under an Employee Plan; (ii) increase any amount of compensation or benefits otherwise payable to any current or former Service Provider or under any Employee Plan; (iii) result in the acceleration of the time of payment, funding or vesting of any benefits to any current or former Service Provider or under any Employee Plan; or (iv) limit the right to merge, amend or terminate any Employee Plan.
(i) Neither the execution and delivery of this Agreement nor the consummation of the Closing shall, either alone or in connection with any other event(s), give rise to any excess parachute payment as defined in Section 280G(b)(1) of the Code, any excise tax owing under Section 4999 of the Code or any other amount that would not be deductible under Section 280G of the Code.
(j) Section 4.18(j) of the Company Disclosure Schedule sets forth all Company obligations to gross-up or reimburse any individual for any tax or related interest or penalties incurred by such individual, including under Sections 409A or 4999 of the Code or otherwise.
(k) Each Foreign Employee Plan required to be registered has been registered and has been maintained in good standing with applicable regulatory authorities and has received applicable favorable tax treatment and, to
A-38
Table of Contents
the knowledge of the Company, no event has occurred since the date of the most recent approval or application therefor relating to any such Foreign Employee Plan that would reasonably be expected to adversely affect any such approval or good standing; and each Foreign Employee Plan required to be fully funded or fully insured, is fully funded or fully insured, including any back-service obligations, on an ongoing and termination or solvency basis (determined using reasonable actuarial assumptions) in compliance with all Applicable Law, in each of the foregoing cases except as would not be material to the Company taken as a whole and any material liability that is not fully funded has been fully and fairly disclosed in the relevant audited accounts and details of such liability are set out in Section 4.18(k) of the Company Disclosure Schedule.
(l) The Company and its Subsidiaries have in all material respects paid the contributions payable to social and healthcare schemes and social security bodies (organismes sociaux, de prévoyance, de protection sociale).
(m) The Company and its Subsidiaries are not under any obligation to any of their executive officers, directors or Key Employees or their assigns (ayants droits) in connection with pension rights, top-up pensions (retraite complémentaire), stock option plans or any other material benefit in kind.
Section 4.19. Taxes.
(a) All material Tax Returns required to be filed by or on behalf of the Company or any of its Subsidiaries (taking into account applicable extensions) have been timely filed with the appropriate Governmental Authority, and all such Tax Returns are true, correct and complete in all material respects.
(b) The Company and its Subsidiaries have timely paid all material amounts of Taxes (whether or not shown on any Tax Return) that are due and payable by the Company and its Subsidiaries, except with respect to matters contested in good faith by appropriate proceedings and with respect to which adequate reserves have been made in accordance with GAAP.
(c) Except for Permitted Liens, there are no Liens for Taxes upon the property or assets of the Company or any of its Subsidiaries.
(d) The Company and its Subsidiaries have deducted, withheld and timely paid to the appropriate Governmental Authority all amounts of Taxes required to be deducted, withheld or paid in connection with amounts paid or owing to any employee, former employee, independent contractor, creditor, stockholder or other Person (including any other third party), and the Company and its Subsidiaries have complied with all related reporting and record keeping requirements.
(e) None of the Company or any of its Subsidiaries has received from any Governmental Authority any written notice of any threatened, proposed, or assessed deficiency for material amounts of Taxes of the Company or any of its Subsidiaries, except for such deficiencies that have been satisfied by payment, settled or withdrawn. No audit or other proceeding by any Governmental Authority is in progress with respect to any material amounts of Taxes due from the Company or any of its Subsidiaries, and neither the Company nor any of its Subsidiaries has received written notice from any Governmental Authority that any such audit or proceeding is pending.
(f) Neither the Company nor any of its Subsidiaries has a pending request for a private letter ruling, administrative relief, technical advice or a change of any method of accounting with any Governmental Authority. No written rulings, clearances or similar agreements have been entered into with or issued by any Governmental Authority with respect to the Company or any of its Subsidiaries which agreement, clearance or ruling would be effective after the Closing Date and could reasonably be expected to have a material effect on the Tax treatment of the Company or any of its Subsidiaries after the Closing Date.
(g) Neither the Company nor any of its Subsidiaries has waived any statute of limitations in respect of Taxes or agreed to any extension of the statute of limitations for assessment, collection or other imposition of any Tax (other than any extension obtained in the ordinary course of business), which extension is currently in effect.
A-39
Table of Contents
(h) Neither the Company nor any of its Subsidiaries is a party to or bound by any Tax sharing, indemnification or allocation agreement or any other similar agreement, other than (i) as set forth in the Existing Company Articles, (ii) any customary commercial contracts entered into in the ordinary course of business which do not primarily relate to Taxes or (iii) any such agreement solely among the Company and its Subsidiaries.
(i) Since January 1, 2016, neither the Company nor any of its Subsidiaries (i) has ever been a member of an Affiliated Group (other than an Affiliated Group the common parent of which is the Company or any of its Subsidiaries and which consists only of the Company and its Subsidiaries), (ii) has liability for the Taxes of any other Person (other than the Company and its Subsidiaries) under Treasury Regulations Section 1.1502-6 (or any similar provision of Applicable Law), as transferor or successor, or by contract (other than pursuant to any customary commercial contract entered into in the ordinary course of business that does not principally relate to Taxes) or (iii) is liable to pay any material Tax in consequence of the failure by any other Person (other than the Company or any of its Subsidiaries) to discharge such Tax in circumstances where such other Person is primarily liable for such Tax.
(j) (i) Neither the Company nor any of its Subsidiaries has been a party to any listed transaction within the meaning of Treasury Regulations Section 1.6011-4(b)(2) or any similar provision of Applicable Law, including the EU Mandatory Disclosure Directive or UK disclosure of tax avoidance schedules legislation.
(k) The Company is and has been since formation properly treated as a partnership (and not as a publicly traded partnership within the meaning of Section 7704(b) of the Code) for U.S. federal, state, and local income Tax purposes. Section 4.19(k) of the Company Disclosure Schedule accurately sets forth the U.S. federal Tax classification of each Subsidiary of the Company as of the date of this Agreement.
(l) Neither the Company nor any of its Subsidiaries will be required to include any material item of income in, or exclude any material item of deduction from, taxable income for any taxable period (or portion thereof) ending after the Closing Date as a result of any: (i) change in method of accounting for a taxable period ending on or prior to the Closing Date or change in method of accounting required as a result of the Transactions, (ii) closing agreement as described in Section 7121 of the Code (or any similar provision of Applicable Law) executed on or prior to the Closing Date, (iii) installment sale or open transaction disposition made on or prior to the Closing Date, (iv) cash method of accounting or long-term contract method of accounting utilized prior to the Closing Date, or (v) prepaid amount received on or prior to the Closing Date.
(m) No Subsidiary has (i) deferred any amount of the employers share of any applicable employment taxes under Section 2302 of the CARES Act, (ii) received any credits under Sections 7001 through 7005 of the FFCRA or section 2301 of the CARES Act, or (iii) deferred any payroll tax obligations pursuant to the Coronavirus Response and Relief Supplemental Appropriation Act of 2021, Internal Revenue Service Notice 2021-11, Internal Revenue Service Notice 2020-65 or other guidance issued pursuant to any Payroll Tax Executive Order.
(n) Neither the Company nor any of its Subsidiaries has constituted either a distributing corporation or a controlled corporation in a distribution of stock qualifying for tax-free treatment under Section 355 of the Code in the three (3) years prior to the date of this Agreement.
(o) Neither the Company nor any Company Subsidiary has been involved in a transaction or arrangement which had a main purpose, or the main purpose, of avoiding Tax.
(p) No claim has been made by any Governmental Authority in a jurisdiction in which the Company or any Company Subsidiary does not file a particular type of Tax Return (or pay a particular type of Tax) that such Company or Company Subsidiary is or may be required to pay such type of Tax to (or file such type of Tax Return with) that Governmental Authority.
A-40
Table of Contents
(q) All payments by, to or among the Company, the Subsidiaries and their Affiliates comply with all applicable transfer pricing requirements imposed by any Governmental Authority, including Section 482 of the Code and the Treasury Regulations promulgated thereunder.
(r) No restricted securities have been acquired by (or by any affiliate of) any of the Companys or any Subsidiarys current, prospective or former employees, directors or officers resident in the United Kingdom for Tax purposes without a valid joint election under Section 431(1) of the Income Tax (Earnings and Pensions) Act 2003 having been signed by the current, prospective or former employee, director or officer and its employer in respect thereof within fourteen (14) days of such acquisition.
(s) To the knowledge of the Company, the sale of the Company pursuant to this Agreement will not give rise to a deemed disposal or realization by the Company or any Subsidiary of any asset or liability for any Taxation purpose in circumstances where such deemed disposal or realization would give rise to a charge to material Tax for the Company or any Subsidiary.
(t) All documents which are required to evidence title of the Company or any Subsidiary to any material asset held by them and which are liable to Transfer Tax or are required to be stamped either with a particular stamp denoting that no Transfer Tax is chargeable or that the document has been produced to the appropriate authority, have been properly and duly stamped and the appropriate Transfer Tax has been paid (together with any related interest and penalties).
(u) If the Company were treated as a corporation for U.S. federal tax purposes, it would not be an investment company within the meaning of Section 351(e)(1) of the Code and Treasury Regulations Section 1.351-1(c)(1)(ii), taking into account Section 351(e)(1)(B) of the Code.
Section 4.20. Environmental Matters.
(a) Except as to matters that, individually or in the aggregate, would not reasonably be expected to be material to the Company and its Subsidiaries, taken as a whole, (i) the Company and each Subsidiary is and, for the last three (3) years, has been in compliance with all Environmental Laws, which compliance include obtaining, maintaining, and complying with all Approvals required under or pursuant to Environmental Laws for their operation (hereinafter, Environmental Permits) and neither the Company nor any Subsidiary has received any notice from the body that has issued any such Environmental Permit that it intends to cancel, terminate, revoke, suspend, modify or not renew any such Environmental Permit; (ii) (x) no written notice, order, request for information, complaint or penalty has been received by the Company or any Subsidiary, and (y) there are no judicial, administrative or other actions, suits or proceedings pending or threatened, in the case of each of (x) and (y), which allege a violation of or liability under or pursuant to any Environmental Law and relate to the Company or any Subsidiary or any real property currently or formerly owned, operated or leased by or for the Company or any Subsidiary; (iii) neither the Company nor any Subsidiary (or any of their respective predecessors) is subject to any Order arising under or relating to any Environmental Law, Environmental Permit or Hazardous Material; (iv) there has been no Release of any Hazardous Materials by the Company or any Subsidiary (or any of their respective predecessors) at, in, on or, under, to or from (x) any Leased Real Property or Owned Real Property or, to the knowledge of the Company, at, in, on or under any property or facility previously owned, leased or operated by or to which any Hazardous Material has been transported for disposal, recycling or treatment by or on behalf of, in each case the Company or any of its Subsidiaries (or any of their respective predecessors) which would reasonably be expected to give rise to liabilities or obligations of the Company or any Subsidiary under Environmental Laws; and (v) to the knowledge of the Company, no conditions currently exist with respect to any property currently or formerly owned, used, leased, or operated by the Company or any Subsidiary that would reasonably be expected to result in the Company or any Subsidiary incurring liabilities or obligations under Environmental Laws.
A-41
Table of Contents
(b) The consummation of the transactions contemplated hereby requires no filings or notifications to be made or actions to be taken pursuant to the New Jersey Industrial Site Recovery Act (N.J.A.C. 7:26B), the Connecticut Transfer Act (CGS §§ 22a-134 to 134e) or any similar and applicable environmental transfer laws.
(c) The Company has provided to the SPAC as of the date hereof copies of (i) all material environmental assessments (including any phase I or II environmental assessments), studies, audits, analyses or reports relating to the Company or any Subsidiary or any real property currently or formerly owned, operated or leased by the Company or any Subsidiary, (ii) all Environmental Permits and (iii) all material, non-privileged documents relating to any material and outstanding liabilities of the Company or any Subsidiary under Environmental Law, to the extent such are in the possession, custody, or reasonable control of the Company or the Subsidiaries.
Section 4.21. Affiliate Transactions. Except as set forth on Schedule 4.21 of the Company Disclosure Schedule, no (i) officer, director, employee, shareholder, equityholder, member, manager or partner of the Company or any of its Subsidiaries, as applicable, (ii) any immediate family member of any of the foregoing, or (iii) any of their respective Affiliates (collectively, the Related Parties) is a party to or has any material interest in any Contract or has any material interest in any property used by the Company or any of its Subsidiaries. None of the Related Parties, on the one hand, and none of the Company or any of its Subsidiaries, on the other hand, owe any material amount to the other, other than pursuant to agreements or promissory notes entered into on an arms-length basis.
Section 4.22. Finders Fees. Except for Lazard Ltd., there is no investment banker, broker, finder or other intermediary that has been retained by or is authorized to act on behalf of the Company who might be entitled to any fee or commission from the Company or any of its Subsidiaries in connection with the Transactions.
Section 4.23. Anti-Bribery; Anti-Corruption. For the last five (5) years, none of the Company, any of its Subsidiaries, or, to the knowledge of the Company, any of the their respective directors, officers, employees, Affiliates or any other Persons acting on their behalf, at their direction or for their benefit has, in connection with the operation of the business of the Company or any of its Subsidiaries, directly or indirectly: (a) made, offered or promised to make or offer any payment, loan or transfer of anything of value, including any reward, advantage or benefit of any kind, to or for the benefit of any government official, candidate for public office, political party or political campaign, or any official of such party or campaign, for the purpose of: (i) influencing any act or decision of such government official, candidate, party or campaign or any official of such party or campaign; (ii) inducing such government official, candidate, party or campaign or any official of such party or campaign to do or omit to do any act in violation of a lawful duty; (iii) obtaining or retaining business for or with any Person; (iv) expediting or securing the performance of official acts of a routine nature; or (v) otherwise securing any improper advantage; (b) paid, offered or agreed or promised to make or offer any bribe, payoff, influence payment, kickback, unlawful rebate or other similar unlawful payment of any nature; (c) made, offered or agreed or promised to make or offer any unlawful contributions, gifts, entertainment or other unlawful expenditures; (d) established or maintained any unlawful fund of corporate monies or other properties; (e) created or caused the creation of any false or inaccurate books and records related to any of the foregoing; or (f) otherwise violated any provision of the Foreign Corrupt Practices Act of 1977, as amended, 15 U.S.C. §§78dd-1, et seq., the United Kingdom Bribery Act 2010 or any other Applicable Law related to anti-corruption or anti-bribery (the Anti-Corruption Laws). None of the Company, any of its Subsidiaries, or any of their respective directors, officers, employees, Affiliates or, to the knowledge of the Company, any other Persons acting on their behalf, at their direction or for their benefit, (i) is or has been the subject of an unresolved claim or allegation relating to (A) any potential violation of the Anti-Corruption Laws or (B) any potentially unlawful payment, contribution, gift, bribe, rebate, payoff, influence payment, kickback or other payment or the provision of anything of value, directly or indirectly, to an official, to any political party or official thereof or to any candidate for political office, or (ii) has received any notice or other communication from, or made a voluntary disclosure to, any Governmental Authority regarding any actual, alleged or potential violation of, or failure to comply with, any Anti-Corruption Law. For the last five (5) years, the Company and its Subsidiaries have had and maintained a system or systems
A-42
Table of Contents
of internal controls reasonably designed to (x) ensure compliance with the Anti-Corruption Laws and (y) prevent and detect violations of the Anti-Corruption Laws.
Section 4.24. International Trade; Sanctions.
(a) For the last five (5) years, the Company, its Subsidiaries, and their respective directors, officers, employees, Affiliates, and, to the knowledge of the Company, any other Persons acting on their behalf, in connection with the operation of the business of the Company and its Subsidiaries, and in each case in all material respects, (i) have been in compliance with all applicable Customs & International Trade Laws, (ii) have obtained all import and export licenses and all other consents, notices, waivers, approvals, orders, authorizations, registrations, declarations, classifications and filings required for the export, deemed export, import, re-export, deemed re-export or transfer of goods, services, software and technology required for the operation of the respective businesses of the Company and its Subsidiaries, including the Customs & International Trade Authorizations; (iii) have not been the subject of any civil or criminal fine, penalty, seizure, forfeiture, revocation of a Customs & International Trade Authorization, debarment or denial of future Customs & International Trade Authorizations in connection with any actual or alleged violation of any applicable Customs & International Trade Laws; and (iv) have not received any actual or, to the knowledge of the Company, threatened claims, investigations or requests for information by a Governmental Authority with respect to material Customs & International Trade Authorizations or compliance with applicable Customs & International Trade Laws and have not made any disclosures to any Governmental Authority with respect to any actual or potential noncompliance with any applicable Customs & International Trade Laws. The Company and its Subsidiaries have in place adequate controls and systems reasonably designed to ensure compliance with applicable Customs & International Trade Laws in each of the jurisdictions in which the Company, any of its Subsidiaries, or any of their respective Affiliates is incorporated or does business.
(b) None of the Company, any of its Subsidiaries, or any of their respective directors, officers, employees, Affiliates or, to the knowledge of the Company, any other Persons acting on their behalf is or has been within the past five (5) years, a Sanctioned Person. For the past five (5) years, the Company, its Subsidiaries, and their respective directors, officers, employees, Affiliates and, to the knowledge of the Company, any other Persons acting on their behalf have, in connection with the operation of the business of the Company and its Subsidiaries, been in compliance with any applicable Sanctions. For the past five (5) years, (i) no Governmental Authority has initiated any action or imposed any civil or criminal fine, penalty, seizure, forfeiture, revocation of an authorization, debarment or denial of future authorizations against the Company, any of its Subsidiaries, or any of their respective directors, officers, employees, Affiliates, or, to the knowledge of the Company, any other Persons acting on their behalf in connection with any actual or alleged violation of any applicable Sanctions, (ii) there have been no actual or threatened claims, investigations or requests for information by a Governmental Authority received by the Company, its Subsidiaries, or any of their respective Affiliates compliance with applicable Sanctions and (iii) and no disclosures have been made to any Governmental Authority with respect to any actual or potential noncompliance with applicable Sanctions. The Company and its Subsidiaries have in place adequate controls and systems reasonably designed to ensure compliance with applicable Sanctions in each of the jurisdictions in which the Company, any of its Subsidiaries, or any of their respective Affiliates is incorporated or does business.
Section 4.25. Data Privacy.
(a) The Company and its Subsidiaries are, and for the past three (3) years have been, and to the knowledge of the Company, any Person acting for or on the Companys or any Subsidiarys behalf is and has, for the past three (3) years, been in material compliance with (i) all applicable Privacy Laws, (ii) all of the Companys and its Subsidiaries external-facing policies and notices regarding Personal Information, and (iii) all of the Companys contractual obligations with respect to the receipt, collection, compilation, use, storage, processing, sharing, safeguarding, security (technical, physical and administrative), disposal, destruction, disclosure, or transfer (including cross-border) of Personal Information. Neither the Company nor any of its Subsidiaries have received
A-43
Table of Contents
any written notice of any claims (including written notice from third parties acting on behalf of the Company or any of its Subsidiaries) of or investigations or regulatory inquiries related to, or been charged in an administrative or judicial proceeding with, the violation of any Privacy Laws or contractual commitments with respect to any Personal Information. Except as would not reasonably be expected to have a material effect, to the knowledge of the Company, there are no facts or circumstances that could reasonably form the basis of any such notice or claim.
(b) The Company and its Subsidiaries have (i) for the past three (3) years, implemented and maintained commercially reasonable technical and organizational safeguards, which safeguards are at least consistent with practices in the industry in which the Company operates, to protect all Personal Information and other confidential data in the Companys or its Subsidiaries possession or under its control against loss, theft, misuse or unauthorized access, use, modification, alteration, destruction or disclosure and (ii) taken commercially reasonable steps to ensure that all third-party service providers, outsourcers, processors or other third parties who process, store or otherwise handle any Personal Information for or on behalf of the Company have agreed to comply with applicable Privacy Laws, maintain the privacy and confidentiality of Personal Information, and protect and secure Personal Information from loss, theft, misuse or unauthorized access, use, modification, alteration, destruction or disclosure.
(c) To the knowledge of the Company, there have been no breaches, security incidents, misuse of or unauthorized access to or disclosure of any Personal Information in the possession or control of the Company or collected, used or processed by or on behalf of the Company, and the Company has not provided or been required to provide any notices to any Person in connection with any disclosure of any Personal Information. The Company and its Subsidiaries have implemented commercially reasonable disaster recovery and business continuity plans, and taken commercially reasonable actions consistent with such plans, to the extent required, to safeguard all confidential data and Personal Information in its possession or control. The Company and its Subsidiaries have conducted commercially reasonable privacy and data security testing or audits at reasonable and appropriate intervals and have resolved or remediated any material privacy or data security issues or vulnerabilities identified. Neither the Company or any of its Subsidiaries nor any third party acting at the discretion or authorization of the Company or any of its Subsidiaries has paid any perpetrator of any data breach incident or cyber-attack.
(d) Except as would not reasonably be expected to have a material effect, the Company is not subject to any contractual requirements or other legal obligations that, following the Closing, would prohibit the Company or any of its Subsidiaries from receiving, accessing, storing or using any Personal Information in the manner in which the Company and its Subsidiaries received, accessed, stored and used such Personal Information prior to the Closing.
Section 4.26. Customers and Suppliers. Since January 1, 2019, none of the Company nor any of its Subsidiaries has received any written or, to the knowledge of the Company, oral notice that the Company or any of its Subsidiaries is in material breach of or material default under any Contract with any Material Customer or Material Supplier or that any such Material Customer or Material Supplier intends to cease doing business with the Company or any of its Subsidiaries or materially decrease the volume of business that it is presently conducting with the Company or any of its Subsidiaries.
Section 4.27. Product Liabilities and Recalls. Since the date that is three (3) years prior to the date hereof, (a) each product and service offering manufactured or sold by the Company or any of its Subsidiaries has been manufactured or sold in material conformity with all contractual commitments and all standard warranties, in each case, to the extent applicable; (b) the Company and its Subsidiaries have not incurred any material obligations for replacement or repair of any of their products or service offerings or other damages in connection therewith; (c) there are no existing or, to the knowledge of the Company, threatened, product warranty, product liability or product recall or similar claims involving any of the products of the Company or any of its Subsidiaries; (d) there have been no product recalls of any of the products of the Company or any of its
A-44
Table of Contents
Subsidiaries; and (e) the Company and its Subsidiaries have not been denied product liability insurance coverage by a third-party insurance provider.
Section 4.28. No Other Representations. The Company acknowledges that the Company and its advisors, have made their own investigation of the SPAC and, except as provided in this Agreement, are not relying on any representation or warranty whatsoever as to the condition, merchantability, suitability or fitness for a particular purpose or trade as to any of the assets of the SPAC, the prospects (financial or otherwise) or the viability or likelihood of success of the business of the SPAC as conducted after the Closing, as contained in any materials provided by the SPAC or any of its Affiliates or any of their respective directors, officers, employees, shareholders, partners, members or representatives or otherwise.
Section 4.29. PPP Loans. The applicable Subsidiaries of the Company were in compliance with all applicable eligibility and certification requirements to apply for and obtain the PPP Loans at the time of their initial application for the PPP Loans. All claims and certifications made by the applicable Subsidiaries of Company in connection with applying for, obtaining and seeking forgiveness of the PPP Loans were true, correct and made in good faith at the time such claims and certifications were made. The applicable Subsidiaries of the Company are, and have been since their application for the PPP Loans, in compliance with all applicable requirements and Applicable Law in connection with the PPP Loans, including any restrictions on the use of any borrowed funds. In connection with applying for, obtaining and seeking forgiveness of the PPP Loans, no directors, officers or, to the knowledge of the Company, other employees of the Company or its Subsidiaries have been debarred or otherwise prohibited from engaging in any contracting activities with any Governmental Authority.
Section 4.30. Debt Financing.
(a) The Company has delivered to the SPAC a true, complete and fully executed copy of a commitment letter (including all related exhibits, schedules, annexes, supplements and term sheets thereto and the fee letter executed in connection therewith, as each of the foregoing may be amended, supplemented, replaced, substituted, terminated or otherwise modified or waived in each case, as permitted or contemplated by Sections 6.10(a), 6.10(c) and/or 8.09, from time to time after the date hereof, the Debt Commitment Letter) from the Debt Financing Sources identified therein confirming their respective commitments to provide the DCL Beneficiary (or its applicable affiliate assignee or designee) the debt facilities referred to therein (the Debt Financing).
(b) As of the date hereof, the Debt Commitment Letter is in full force and effect and is the legal, valid and binding obligation of the DCL Beneficiary and, to the knowledge of the DCL Beneficiary, the other parties thereto, enforceable against the DCL Beneficiary and, to the knowledge of the DCL Beneficiary, the other parties thereto in accordance with its terms (subject to the Enforceability Exceptions). As of the date hereof, the Debt Commitment Letter has not been amended, restated, or otherwise modified or waived, and the respective commitments contained in the Debt Commitment Letter has not been withdrawn, rescinded or otherwise modified. All fees (if any) required to be paid under the Debt Commitment Letter on or prior to the date hereof have been paid in full.
(c) As of the date hereof, neither the DCL Beneficiary nor, to the knowledge of the DCL Beneficiary and/or the Company, the other parties thereto have breached any of the covenants or other obligations set forth in, or is in default under, the Debt Commitment Letter. As of the date hereof, to the knowledge of the DCL Beneficiary and/or the Company no event has occurred that, with or without notice, lapse of time or both, would or would reasonably be expected to constitute or result in a breach or default on the part of the DCL Beneficiary or any other party to the Debt Commitment Letter.
(d) There are no conditions precedent directly or indirectly related to the funding of the full amount of the Debt Financing other than as expressly set forth in the Debt Commitment Letter (including the fee letter). Other than the Debt Commitment Letter (including the fee letter), there are no other contracts, arrangements or other
A-45
Table of Contents
agreements, to which the Company or any Affiliate thereof is a party related to the Debt Financing (except for customary non-disclosure agreements, non-reliance letters and similar written agreements, in each case which do not impact the conditionality or amount of the Debt Financing). As of the date hereof, assuming the satisfaction of the conditions in Sections 11.01 and 11.03, to the knowledge of the Company, the Company has no reason to believe that any of the conditions to the Debt Financing will not be satisfied or that the Debt Financing will not be available to the DCL Beneficiary (or its applicable affiliate assignee or designee) on the Closing Date.
Section 4.31. Exclusivity of Representations and Warranties.
(a) Except as otherwise expressly set forth in this Article 4 (as modified by the Company Disclosure Schedule), any certificate delivered pursuant to this Agreement or in any other Transaction Agreement, the Sellers and the Company expressly disclaim any representations or warranties of any kind or nature in respect of any such Seller or the Company, express or implied, including any representations or warranties as to the Companys Subsidiaries, their respective businesses and affairs or the Transactions.
(b) Without limiting the generality of the foregoing, except for the representations and warranties in this Article 4, any certificate delivered pursuant to this Agreement or in any other Transaction Agreement, neither the Sellers, the Company nor any other Person has made, and shall not be deemed to have made, any representations or warranties in the materials relating to the business and affairs of the Company or its Subsidiaries that have been made available to the SPAC, including due diligence materials, or in any presentation of the business and affairs of the Company or its Subsidiaries by the management of the Company or others in connection with the Transactions, and no statement contained in any of such materials or made in any such presentation shall be deemed a representation or warranty hereunder or otherwise or deemed to be relied upon by the SPAC in executing, delivering and performing this Agreement and the Transactions. It is understood that any cost estimates, projections or other predictions, any data, any financial information or any memorandum or offering materials or presentations, including any offering memorandum or similar materials made available by the Sellers or the Sellers or the Companys representatives on behalf of the Company, or by the Company directly, are not and shall not be deemed to be or to include representations or warranties made by the Sellers or the Company, and are not and shall not be deemed to be relied upon by the SPAC in executing, delivering and performing the Transaction Agreements and the Transactions, except, in each such case, to the extent of any representation or warranty provided in this Article 4, any certificate delivered pursuant to this Agreement or in any other Transaction Agreement with respect to any such matters.
ARTICLE 5
REPRESENTATIONS AND WARRANTIES OF THE SPAC
Except (a) as set forth in the SPAC Disclosure Schedule (the SPAC Disclosure Schedule); and (b) as disclosed in the SEC Documents filed or furnished with the SEC prior to the date of this Agreement (to the extent the qualifying nature of such disclosure is readily apparent from the content of such SEC Documents, excluding disclosures referred to in Forward-Looking Statements, Risk Factors and any other disclosures therein to the extent they are of a predictive or cautionary nature or related to forward-looking statements), the SPAC represents and warrants to the Sellers and the Company as of the date hereof and as of the Closing Date that:
Section 5.01. Corporate Existence and Power. The SPAC is a corporation duly incorporated, validly existing and in good standing under the laws of its jurisdiction of incorporation and has all corporate powers and all material governmental licenses, authorizations, permits, consents and approvals required to carry on its business as now conducted, except for those licenses, authorizations, permits, consents and approvals the absence of which would not, and would not reasonably be expected to, individually or in the aggregate, have a SPAC Material Adverse Effect.
Section 5.02. Authorization. The execution, delivery and performance by the SPAC of this Agreement and the other Transaction Agreements and the consummation of the Transactions are within the corporate powers and
A-46
Table of Contents
authority of the SPAC and have been duly authorized by all necessary corporate action on the part of the SPAC, other than, as of the date hereof, approval of the Transaction Proposals by the Requisite Existing SPAC Stockholders at the SPAC Special Meeting. The approval of the Transaction Proposals by the Requisite Existing SPAC Stockholders at the SPAC Special Meeting is the only vote of the holders of any class or series of capital stock of the SPAC necessary to approve the Transactions. This Agreement and the other Transaction Agreements to which the SPAC is a party constitutes a valid and binding agreement of the SPAC, assuming this Agreement constitutes the valid and binding obligation of the other parties hereto and thereto, enforceable against the SPAC in accordance with their terms, except insofar as enforceability may be limited by Enforceability Exceptions.
Section 5.03. Governmental Authorization. The execution, delivery and performance by the SPAC of this Agreement and the other Transaction Agreements to which it is a party and the consummation of the Transactions requires no consent, approval, authorization, permit, action by or in respect of, or filing with or notification to, any Governmental Authority, other than (i) compliance with any applicable requirements of the HSR Act; (ii) compliance with any applicable requirements of the Securities Act and the Exchange Act; (iii) such actions and filings as may be required by any Applicable Law set forth in Section 5.03 of the SPAC Disclosure Schedule; and (iv) any such action or filing the failure of which to make or obtain would not, individually or in the aggregate, prevent, materially delay or materially impair the ability of the SPAC to consummate the Transactions or the ability of the SPAC to perform its obligations under this Agreement or the other Transaction Agreements to which it is a party.
Section 5.04. Noncontravention. The execution, delivery and performance by the SPAC of this Agreement and the other Transaction Agreements to which it is a party do not, the performance of this Agreement and the other Transaction Agreements to which it is a party by the SPAC shall not, and the consummation of the Transactions will not (i) conflict with or violate the organizational documents of the SPAC, (ii) assuming compliance with the matters referred to in Section 5.03, violate any Applicable Law, (iii) require any consent, waiver or other action by any Person under, violate, result in a breach of or constitute a default (with or without notice or lapse of time, or both) under or give rise to any right of termination, amendment, cancellation, modification or acceleration of any right or obligation of the SPAC or any third party or to a loss or impairment of any benefit or right to which the SPAC is entitled under any provision of any Contract or other instrument binding upon the SPAC or (iv) result in the creation or imposition of any material Lien on any asset or properties of the SPAC, except for any Permitted Liens, with such exceptions as would not, and would not reasonably be expected to, in the case of each of clauses (iii) and (iv), individually or in the aggregate, have a SPAC Material Adverse Effect.
Section 5.05. Capital Structure.
(a) The authorized capital stock of the SPAC consists of (i) 500,000,000 authorized and 75,000,000 issued and outstanding shares of Class A common stock of the SPAC, par value $0.0001 per share; (ii) 50,000,000 authorized and 18,750,000 issued and outstanding shares of Class B common stock of the SPAC, par value $0.0001 per share; and (iii) 5,000,000 authorized and no issued and outstanding shares of preferred stock of the SPAC, par value $0.0001 per share (Existing SPAC Preferred Stock), and there are issued and outstanding warrants to purchase 8,500,000 shares of Class A common stock of the SPAC (the Private Placement Warrants) at an exercise price of $11.50 per share, and issued and outstanding warrants to purchase 18,750,000 shares of Class A common stock of the SPAC (the Public Warrants) at an exercise price of $11.50 per share. All outstanding shares of Existing SPAC Common Stock and Existing SPAC Preferred Stock and all Private Placement Warrants have been duly authorized, validly issued and fully paid, are non-assessable and are not subject to preemptive rights.
(b) Immediately prior to the closing of the transactions contemplated by the Closing Step Plan, the authorized capital stock of the SPAC will consist of (i) 2,000,000,000 New SPAC Class A Common Shares, (ii) 100,000,000 New SPAC Class B Common Shares, and (iii) 100,000,000 New SPAC Preferred Shares, of which the SPAC has committed to issue 90,000,000 New SPAC Class A Common Shares to the PIPE Investors, up to
A-47
Table of Contents
12,500,000 New SPAC Class A Common Shares to the Backstop Party, a number of New SPAC Class A Common Shares to holders of SPAC Stock Electing Shares and holders of the Loan Notes (subject to Article 2 and the elections made pursuant to the Election Agreements), and a number of New SPAC Class B Common Shares to holders of Unit Electing Shares (subject to Article 2 and the elections made pursuant to the Election Agreements). Immediately prior to the closing of the transactions contemplated by the Closing Step Plan, the SPAC will have up to 8,500,000 Private Placement Warrants issued and outstanding that will entitle the holder thereof to purchase New SPAC Class A Common Shares at an exercise price of $11.50 per share on the terms and conditions set forth in the applicable warrant agreement, of which up to 8,500,000 Private Placement Warrants will be issued to the Sponsor.
(c) Subject to approval of the Transaction Proposals by the Requisite Existing SPAC Stockholders, the equity portion of the Total Consideration, when delivered, as applicable, shall be duly authorized and validly issued, fully paid and non-assessable, issued in compliance with all Applicable Law and not subject to, and not issued in violation of, any options, warrants, calls, rights (including preemptive rights), organizational documents, commitments or agreements to which the SPAC or any Subsidiary of the SPAC is a party or by which it is bound, and shall be issued to the holders of Unit Electing Shares and the holders of SPAC Stock Electing Shares with good and valid title, free and clear of any Liens other than Liens arising out of, under or in connection with applicable federal, state and local securities laws.
(d) Except for the Public Warrants, the Private Placement Warrants and the PIPE Subscription Agreements, there are no subscriptions, calls, options, warrants, rights (including preemptive rights), puts or other securities convertible into or exchangeable or exercisable for the share of Existing SPAC Common Stock, New SPAC Common Stock or other equity interests of the SPAC, or any other contracts to which the SPAC is a party or by which the SPAC is bound obligating the SPAC to issue, transfer, register or sell, or cause to be issued, transferred, registered or sold, any shares of capital stock of, other equity interests in or debt securities of, the SPAC to grant, extend or enter into options, warrants, calls, rights, subscriptions or other securities. Other than in connection with the SPAC Stockholder Redemptions, there are no outstanding contractual obligations of the SPAC to repurchase, redeem or otherwise acquire any securities or equity interests of the SPAC. There are no outstanding bonds, debentures, notes or other Indebtedness of the SPAC having the right to vote (or convertible into, or exchangeable for, securities having the right to vote) on any matter for which the SPACs stockholders may vote. The SPAC is not a party to any shareholders agreement, voting agreement or registration rights agreement relating to Existing SPAC Common Stock, New SPAC Common Stock or any other equity interests of the SPAC. The SPAC does not own any capital stock or any other equity interests in any other Person other than investments made in accordance with the Trust Account. The SPAC does not have any right, option, warrant, conversion right, stock appreciation right, redemption right, repurchase right, agreement, arrangement or commitment of any character under which a Person is or may become obligated to issue or sell, or give any right to subscribe for or acquire, or in any way dispose of, any shares of the capital stock or other equity interests, or any securities or obligations exercisable or exchangeable for or convertible into any shares of the capital stock or other equity interests, of such Person.
Section 5.06. SEC Documents; Controls.
(a) Since June 29, 2020, the SPAC has timely filed or furnished with the SEC all forms, reports, schedules and statements required to be filed or furnished under the Securities Act or Exchange Act (such forms, reports, schedules, and statements other than the Proxy Statement and the Registration Statement, the SEC Documents). As of their respective filing (or furnishing) dates, each of the SEC Documents, as amended (including all exhibits and schedules thereto and documents incorporated by reference therein), complied in all material respects with the applicable requirements of the Securities Act or the Exchange Act, as the case may be, and the rules and regulations of the SEC thereunder applicable to such SEC Documents, and none of the SEC Documents contained, when filed or, if amended prior to the date hereof, as of the date of such amendment with respect to those disclosures that are amended, any untrue statement of material fact or omitted to state a material fact required to be stated therein or necessary to make the statements therein, in light of the circumstances under
A-48
Table of Contents
which they were made, not misleading. None of the SEC Documents are the subject of ongoing SEC review or outstanding SEC comment and, to the SPACs knowledge, neither the SEC nor any other Governmental Authority is conducting any investigation or review of any SEC Document. No notice of any SEC review or investigation of the SPAC or the SEC Documents has been received by the SPAC.
(b) The financial statements of the SPAC included in the SEC Documents, including all notes and schedules thereto, complied in all material respects when filed, or if amended prior to the date hereof, as of the date of such amendment, with the rules and regulations of the SEC with respect thereto, were prepared in accordance with GAAP (except as may be indicated in the notes thereto, or in the case of the unaudited statements, as permitted by Rule 10-01 of Regulation S-X of the SEC) and fairly present in all material respects in accordance with the applicable requirements of GAAP (except as may be indicated in the notes thereto, subject, in the case of the unaudited statements, to normal year-end audit adjustments that are not material) the financial position of the SPAC, as of their respective dates, and the results of operations, changes in stockholders equity and cash flows of the SPAC, for the periods presented therein.
(c) The SPAC has established and maintains disclosure controls and procedures and internal control over financial reporting (as such terms are defined in paragraphs (e) and (f), respectively, of Rule 13a-15 under the Exchange Act and the listing standards of the NYSE). To the SPACs knowledge and except as disclosed in the SEC Documents, the SPACs internal controls provide reasonable assurance regarding the reliability of the SPACs financial reporting and the preparation of financial statements for external purposes in material conformity with GAAP and reasonably designed to ensure that material information relating to the SPAC is accumulated and communicated to the SPACs management as appropriate. To the SPACs knowledge and except as disclosed in the SEC Documents, since the SPACs formation, there have been no significant deficiencies or material weakness in the SPACs internal control over financial reporting (whether or not remediated) and no change in the SPACs control over financial reporting that has materially affected, or is reasonably likely to materially affect, the SPACs internal control over financial reporting.
Section 5.07. Listing. The issued and outstanding shares of Class A common stock of the SPAC, par value $0.0001 per share, are registered pursuant to Section 12(b) of the Exchange Act and are listed for trading on the NYSE. As of the date hereof, there is no Action pending, or to the knowledge of the SPAC, threatened, against the SPAC by the NYSE or the SEC with respect to any intention by such entity to deregister any such SPAC shares or prohibit or terminate the listing of any such SPAC shares on the NYSE.
Section 5.08. The Registration Statement and the Proxy Statement. At the time it is declared effective by the SEC and at the Closing, the Registration Statement, and when first filed in accordance with Rule 424(b) or filed pursuant to Section 14A, the Proxy Statement (or any amendment or supplement thereto), will comply in all material respects with the applicable requirements of the Securities Act and the Exchange Act. At the time it is declared effective by the SEC and at the Closing, the Registration Statement, and on the date of any filing pursuant to Rule 424(b), the date the Proxy Statement is first mailed to the Existing SPAC Stockholders and at the time of the SPAC Special Meeting, the Proxy Statement (together with any amendments or supplements thereto) will not include any untrue statement of material fact or omit to state a material fact necessary in order to make the statements therein, in the light of the circumstances under which they were made, not misleading; provided, however, that the SPAC makes no representations or warranties as to the information contained in or omitted from the Registration Statement or the Proxy Statement in reliance upon and in conformity with information furnished in writing to the SPAC by or on behalf of the Company or any Seller specifically for inclusion in the Registration Statement or the Proxy Statement.
Section 5.09. Absence of Certain Changes. Since its respective formation through the date of this Agreement, the SPAC has not (a) conducted business other than its formation, the public offering of its securities (and the related private offerings), public reporting and its search for an initial Business Combination as described in the Prospectus (including the investigation of the Company and its Subsidiaries and the negotiation and execution of this Agreement) and related activities or (b) been subject to (i) a material adverse change or a
A-49
Table of Contents
material adverse effect, individually or in the aggregate, upon on its assets, financial condition, business or operations, taken as a whole, or (ii) any effect, change, event or occurrence that would, individually or in the aggregate, prevent, materially delay or materially impair the ability of the SPAC to consummate the Transactions.
Section 5.10. Litigation. There is no Action, suit, investigation or proceeding pending against, or to the knowledge of the SPAC, threatened against or affecting, the SPAC before any court or arbitrator or any Governmental Authority which in any manner challenges or seeks to prevent, enjoin, alter or delay the Transactions.
Section 5.11. Compliance with Applicable Law. Since the date of its incorporation, the SPAC has not been in violation of any Applicable Law, except for violations which would not be reasonably likely, individually or in the aggregate, to result in material liability to the SPAC.
Section 5.12. Taxes.
(a) All material Tax Returns required to be filed by or on behalf of the SPAC (taking into account applicable extensions) have been timely filed with the appropriate Governmental Authority, and all such Tax Returns are true, correct and complete in all material respects.
(b) The SPAC timely has paid all material amounts of Taxes (whether or not shown on any Tax Return) that are due and payable by the SPAC, except with respect to matters contested in good faith by appropriate proceedings and with respect to which adequate reserves have been made in accordance with GAAP.
(c) Except for Permitted Liens, there are no Liens for Taxes upon the property or assets of the SPAC.
(d) The SPAC has deducted, withheld and timely paid to the appropriate Governmental Authority all amounts of Taxes required to be deducted, withheld or paid in connection with amounts paid or owing to any employee, former employee, independent contractor, creditor, stockholder or other Person (including any other third party), and the SPAC has complied with all related reporting and record keeping requirements.
(e) The SPAC has not received from any Governmental Authority any written notice of any threatened, proposed, or assessed deficiency for material amounts of Taxes of the SPAC, except for such deficiencies that have been satisfied by payment, settled or withdrawn. No audit or other proceeding by any Governmental Authority is in progress with respect to any material amounts of Taxes due from the SPAC, and the SPAC has not received written notice from any Governmental Authority that any such audit or proceeding is contemplated or pending.
(f) The SPAC does not have a pending request for a private letter ruling, administrative relief, technical advice or a change of any method of accounting with any Governmental Authority. No written rulings, clearances or similar agreements have been entered into with or issued by any Governmental Authority with respect to the SPAC which agreement, clearance or ruling would be effective after the Closing Date and could reasonably be expected to have a material effect on the Tax treatment of the SPAC after the Closing Date.
(g) The SPAC has not waived any statute of limitations in respect of Taxes or agreed to any extension of the statute of limitations for assessment, collection or other imposition of any Tax (other than any extension obtained in the ordinary course of business), which extension is currently in effect.
(h) The SPAC is not a party to or bound by any Tax sharing, indemnification or allocation agreement or any other similar agreement, other than (i) as set forth in the Exiting SPAC Certificate of Incorporation or Existing SPAC Bylaws or (ii) any customary commercial contracts entered into in the ordinary course of business which do not primarily relate to Taxes.
A-50
Table of Contents
(i) Other than prior to the IPO, the SPAC (i) has never been a member of an Affiliated Group and (ii) has no liability for the Taxes of any other Person under Treasury Regulations Section 1.1502-6 (or any similar provision of Applicable Law), as transferor or successor, or by contract (other than pursuant to any customary commercial contract entered into in the ordinary course of business that does not principally relate to Taxes).
(j) The SPAC has not been a party to any listed transaction within the meaning of Treasury Regulations Section 1.6011-4(b)(2).
(k) The SPAC is, and has been since formation, properly treated as a corporation for U.S. federal, state and local income Tax purposes.
(l) The SPAC will not be required to include any material item of income in, or exclude any material item of deduction from, taxable income for any taxable period (or portion thereof) ending after the Closing Date as a result of any (i) change in method of accounting for a taxable period ending on or prior to the Closing Date or change in method of accounting required as a result of the Transactions, (ii) closing agreement as described in Section 7121 of the Code (or any similar provision of Applicable Law) executed on or prior to the Closing Date, (iii) installment sale or open transaction disposition made on or prior to the Closing Date, (iv) cash method of accounting or long-term contract method of accounting utilized prior to the Closing Date or (v) prepaid amount received on or prior to the Closing Date.
(m) The SPAC has not (i) deferred any amount of the employers share of any applicable employment taxes under Section 2303 of the CARES Act, (ii) received any credits under Sections 7001 through 7005 of the FFCRA or Section 2301 of the CARES Act or (iii) deferred any payroll tax obligations pursuant to the Coronavirus Response and Relief Supplemental Appropriation Act of 2021, Internal Revenue Service Notice 2021-11, Internal Revenue Service Notice 2020-65 or other guidance issued pursuant to any Payroll Tax Executive Order.
(n) The SPAC has not constituted either a distributing corporation or a controlled corporation in a distribution of stock qualifying for tax-free treatment under Section 355 of the Code in the two (2) years prior to the date of this Agreement.
(o) The SPAC has not been involved in a transaction or arrangement which had a main purpose, or the main purpose, of avoiding Tax.
(p) No claim has been made by any Governmental Authority in a jurisdiction in which the SPAC does not file a particular type of Tax Return (or pay a particular type of Tax) that the SPAC is or may be required to pay such type of Tax to (or file such type of Tax Return with) that Governmental Authority.
Section 5.13. Employees and Employee Benefits Plans. The SPAC does not (a) have any paid employees or (b) maintain, sponsor, contribute to or otherwise have any liability under any employee benefit plans. Neither the execution and delivery of this Agreement or the other Transaction Agreements nor the Transactions will: (a) result in any payment from the SPAC (including severance, unemployment compensation, golden parachute, bonus or otherwise) becoming due to any director, officer or employee of the SPAC; or (b) result in the acceleration of the time of payment or vesting of any such benefits from the SPAC. Other than reimbursement of any out-of-pocket expenses incurred by the SPACs officers and directors in connection with activities on the SPACs behalf in an aggregate amount not in excess of the amount of cash held by the SPAC outside of the Trust Account, the SPAC has no unsatisfied material liability with respect to any officer or director.
Section 5.14. Affiliate Transactions. Except for equity ownership or employment relationships (including any employment or similar Contract) expressly contemplated by this Agreement, any non-disclosure or confidentiality Contract entered into in connection with the wall-crossing of the Existing SPAC Stockholders, any other Transaction Agreement or any contract that is an exhibit to the SEC Documents or described therein,
A-51
Table of Contents
(a) there are no transactions or contracts, or series of related transactions or contracts, between the SPAC, on the one hand, and any related party of the SPAC, the Sponsor, any beneficial owner (as defined in Rule 13d-3 under the Exchange Act) of five percent (5%) or more of the Existing SPAC Common Stock or, to the knowledge of the SPAC, any of their respective associates or immediate family members (as such terms are defined in Rule 12b-2 and Rule 16a-1 of the Exchange Act), on the other hand, nor is any Indebtedness owed by or to the SPAC, on the one hand, to or by the Sponsor or any such related party, beneficial owner, associate or immediate family member, on the other hand, and (b) none of the officers or directors (or members of a similar governing body) of the SPAC, the Sponsor, any beneficial owner of five percent (5%) or more of the Existing SPAC Common Stock or, to the knowledge of the SPAC, their respective associates or immediate family members, has any material interest in, (i) any material tangible or real property that the SPAC uses, owns or leases or (ii) any customer, vendor or other material business relation of the SPAC or the Sponsor that, in each case of (a) or (b), would prevent, materially delay or materially impact the ability of the SPAC to perform its obligations under this Agreement or any other Transaction Agreement to which it is a party.
Section 5.15. Properties. The SPAC does not own, license or otherwise have any right, title or interest in any material Intellectual Property Rights. The SPAC does not own, or otherwise have an interest in, any real property, including under any real property lease, sublease, space sharing, license or other occupancy agreement.
Section 5.16. Contracts. Other than this Agreement, the other Transaction Agreements, Transaction Expenses incurred by the SPAC or any contracts that are exhibits to the SEC Documents, there are no Contracts to which the SPAC is a party or by which any of its properties or assets may be bound, subject or affected, which (a) creates or imposes a liability greater than $250,000, (b) may not be cancelled by the SPAC on less than sixty (60) calendar days prior notice without payment of a material penalty or termination fee or (c) prohibits, prevents, restricts or impairs in any material respect any business practice of the SPAC as its business is currently conducted or any acquisition of material property by the SPAC, or restricts in any material respect the ability of the SPAC to compete with any other Person (each such contract, a SPAC Material Contract). All SPAC Material Contracts have been made available to the Company.
Section 5.17. Finders Fees. Except as set forth on Section 5.17 of the SPAC Disclosure Schedule, there is no investment banker, broker, finder or other intermediary that has been retained by or is authorized to act on behalf of the SPAC who might be entitled to any fee or commission in connection with the Transactions.
Section 5.18. Financial Ability. Assuming the Debt Financing is fully funded on the Closing Date in accordance with the Debt Commitment Letter, the aggregate net proceeds of the Debt Financing and the PIPE Financing and the funds to be contributed to the SPAC from the Trust Account are in an amount sufficient to fund the Cash Consideration and the Primary Capital, to pay the Debt Payoff Amount, to pay the Transaction Expenses and to satisfy all of the SPACs other obligations under this Agreement required to be paid on the Closing Date.
Section 5.19. PIPE Financing; Backstop Agreement
(a) The SPAC has delivered to the Company true, correct and complete copies of each of the PIPE Subscription Agreements entered into by the SPAC with the applicable PIPE Investors named therein, pursuant to which the PIPE Investors have committed to provide the PIPE Financing. To the knowledge of the SPAC, with respect to each PIPE Investor, the PIPE Subscription Agreement with such PIPE Investor is in full force and effect and has not been withdrawn or terminated, or otherwise amended or modified, in any respect, and no withdrawal, termination, amendment or modification is contemplated by the SPAC. Each PIPE Subscription Agreement is a legal, valid and binding obligation of the SPAC and, to the knowledge of the SPAC, each PIPE Investor, and none of the execution, delivery or performance of obligations under such PIPE Subscription Agreement by the SPAC or, to the knowledge of the SPAC, each PIPE Investor, violates any Applicable Law. There are no other agreements, side letters, or arrangements between the SPAC and any PIPE Investor relating to any PIPE Subscription Agreement that could affect the obligation of such PIPE Investors to contribute to the
A-52
Table of Contents
SPAC the applicable portion of the PIPE Financing Amount set forth in the PIPE Subscription Agreement of such PIPE Investors, and, as of the date hereof, the SPAC does not know of any facts or circumstances that may reasonably be expected to result in any of the conditions set forth in any PIPE Subscription Agreement not being satisfied, or the PIPE Financing Amount not being available to the SPAC, on the Closing Date. No event has occurred that, with or without notice, lapse of time or both, would constitute a default or breach on the part of the SPAC under any material term or condition of any PIPE Subscription Agreement and, as of the date hereof, the SPAC has no reason to believe that it will be unable to satisfy in all material respects on a timely basis any term or condition of closing to be satisfied by it contained in any PIPE Subscription Agreement. The PIPE Subscription Agreements contain all of the conditions precedent (other than the conditions contained in the other agreements related to the transactions contemplated herein) to the obligations of the PIPE Investors to contribute to the SPAC the applicable portion of the PIPE Financing Amount set forth in the PIPE Subscription Agreements on the terms therein.
(b) The SPAC has delivered to the Company a true, correct and complete copy of the Backstop Agreement entered into by the SPAC with the Backstop Party, pursuant to which the Backstop Party has committed to provide the Backstop Amount on the terms and conditions set forth in the Backstop Agreement. To the knowledge of the SPAC, the Backstop Agreement with the Backstop Party is in full force and effect and has not been withdrawn or terminated, or otherwise amended or modified, in any respect, and no withdrawal, termination, amendment or modification is contemplated by the SPAC. The Backstop Agreement is a legal, valid and binding obligation of the SPAC and, to the knowledge of the SPAC, the Backstop Party, and none of the execution, delivery or performance of obligations under such Backstop Agreement by the SPAC or, to the knowledge of the SPAC, the Backstop Party, violates any Applicable Law. There are no other agreements, side letters, or arrangements between the SPAC and the Backstop Party relating to the Backstop Agreement that could affect the obligation of the Backstop Party to contribute to the SPAC the applicable portion of the Backstop Amount, if any is required to be contributed thereunder, set forth in the Backstop Agreement, and, as of the date hereof, the SPAC does not know of any facts or circumstances that may reasonably be expected to result in any of the conditions set forth in the Backstop Agreement not being satisfied, or the Backstop Amount not being available to the SPAC, if any, on the Closing Date. No event has occurred that, with or without notice, lapse of time or both, would constitute a default or breach on the part of the SPAC under any material term or condition of the Backstop Agreement and, as of the date hereof, the SPAC has no reason to believe that it will be unable to satisfy in all material respects on a timely basis any term or condition of closing to be satisfied by it contained in the Backstop Agreement. The Backstop Agreement contains all of the conditions precedent (other than the conditions contained in the other agreements related to the transactions contemplated herein) to the obligations of the Backstop Party to contribute to the SPAC the Backstop Amount (if required) set forth in the Backstop Agreement on the terms therein.
(c) No fees, consideration or other discounts are payable or have been agreed by the SPAC or any of its Subsidiaries to any PIPE Investor in respect of its portion of the PIPE Financing Amount, or to the Backstop Party with respect to the Backstop Amount, except as set forth in the PIPE Subscription Agreements or the Backstop Agreement, respectively.
Section 5.20. Trust Account.
(a) As of the date hereof, the SPAC has at least $750,000,000 (the Trust Amount) in the account established by the SPAC for the benefit of its public shareholders (the Trust Account), with such funds invested in United States Government securities or money market funds meeting certain conditions under Rule 2a-7 promulgated under the Investment Company Act and held in trust by the Trustee pursuant to the Trust Agreement. The SPAC has delivered to the Company a true, complete and fully executed copy of the Trust Agreement.
(b) The Trust Agreement is in full force and effect and is a valid and binding obligation of the SPAC and, to the knowledge of the SPAC, the other parties thereto, enforceable against the SPAC and the other parties thereto
A-53
Table of Contents
in accordance with its terms (subject to the Enforceability Exceptions). The Trust Agreement has not been amended, restated or modified, and no amendment, restatement or modification of the Trust Agreement is contemplated, and the respective rights and obligations contained in the Trust Agreement have not been withdrawn, rescinded or otherwise modified.
(c) Neither the SPAC nor, to the knowledge of the SPAC, the other parties thereto have breached any of the covenants or other obligations set forth in, or are in default under, the Trust Agreement, and to the knowledge of the SPAC, no event has occurred or circumstance exists that, with or without notice, lapse of time or both, would or would reasonably be likely to (i) constitute or result in a breach or default on the part of any Person under the Trust Agreement or (ii) constitute or result in a failure by the SPAC or the other parties thereto to satisfy a condition precedent to or other contingency to be satisfied by the SPAC or the other parties thereto set forth in the Trust Agreement.
(d) Other than the Trust Agreement, there are no other contracts, side letters, arrangements or understandings (whether written or unwritten, express or implied), to the knowledge of the SPAC, that would entitle any Person (other than Existing SPAC Investors who have previously validly elected to redeem their shares of Existing SPAC Common Stock pursuant to a SPAC Stockholder Redemption) to any portion of proceeds in the Trust Account. As of the date hereof, assuming the satisfaction of the conditions to the SPACs obligation to consummate the transactions contemplated hereby, the SPAC has no reason to believe that, subject to the SPAC Stockholder Redemptions, the full Trust Amount will not be available to the SPAC or an Affiliate thereof on the Closing Date.
(e) Prior to the Closing, none of the funds held in the Trust Account may be released except (i) to pay income and franchise taxes on any interest income earned in the Trust Account, (ii) to pay working capital related costs, and (iii) to satisfy obligations in respect of the SPAC Stockholder Redemptions.
(f) There are no Actions pending or, to the knowledge of the SPAC, threatened in writing with respect to the Trust Account.
Section 5.21. No Other Representations. The SPAC acknowledges that the SPAC and its advisors, have made their own investigation of the Company and its Subsidiaries and, except as provided in this Agreement, are not relying on any representation or warranty whatsoever as to the condition, merchantability, suitability or fitness for a particular purpose or trade as to any of the assets of the Company or any of its Subsidiaries, the prospects (financial or otherwise) or the viability or likelihood of success of the business of the Company and its Subsidiaries as conducted after the Closing, as contained in any materials provided by the Company or any of its Affiliates or any of their respective directors, officers, employees, shareholders, partners, members or representatives or otherwise.
Section 5.22. Exclusivity of Representations and Warranties.
(a) Except as otherwise expressly set forth in this Article 5 (as modified by the SPAC Disclosure Schedule), any certificate delivered pursuant to this Agreement or in any other Transaction Agreements, the SPAC expressly disclaim any representations or warranties of any kind or nature in respect of the SPAC, express or implied, including the Transactions.
(b) Without limiting the generality of the foregoing, except for the representations and warranties in this Article 5, any certificate delivered pursuant to this Agreement or in any other Transaction Agreement, neither the SPAC nor any other Person has made, and shall not be deemed to have made, any representations or warranties in the materials relating to the business and affairs of the SPAC that have been made available to the Company and the Sellers, including due diligence materials, or in any presentation of the business and affairs of the SPAC by the management of the SPAC or others in connection with the Transactions, and no statement contained in any of such materials or made in any such presentation shall be deemed a representation or warranty hereunder or
A-54
Table of Contents
otherwise or deemed to be relied upon by the Company or any Seller in executing, delivering and performing this Agreement and the Transactions. It is understood that any cost estimates, projections or other predictions, any data, any financial information or any memorandum or offering materials or presentations, including any offering memorandum or similar materials made available by the SPAC the SPACs representatives on behalf of the SPAC, or by the SPAC directly, are not and shall not be deemed to be or to include representations or warranties made by the SPAC, and are not and shall not be deemed to be relied upon by the Company or any Seller in executing, delivering and performing the Transaction Agreements and the Transactions, except, in each such case, to the extent of any representation or warranty provided in this Article 5, any certificate delivered pursuant to this Agreement or in any other Transaction Agreement with respect to any such matters.
ARTICLE 6
COVENANTS OF THE COMPANY
The Company agrees that:
Section 6.01. Conduct of Business. From the date of this Agreement until the earlier of the Closing or the termination of this Agreement in accordance with its terms (the Interim Period), the Company shall, and shall cause its Subsidiaries to, except as expressly contemplated by this Agreement, set forth on Schedule 6.01 or consented to by the SPAC in writing (which consent shall not be unreasonably conditioned, withheld or delayed), operate its business in the ordinary course of business consistent with past practice (provided that any action taken, or omitted to be taken, that relates to, or arises out of, COVID-19 shall be deemed to be in the ordinary course of business consistent with past practice). Notwithstanding anything to the contrary contained herein, nothing herein shall prevent the Company or any of its Subsidiaries from taking or failing to take any action, including the establishment of any policy, procedure or protocol, in specific response to COVID-19 or any COVID-19 Measures, and (x) all such actions or failure to take such actions shall be deemed to constitute an action taken in the ordinary course of business consistent with past practice and shall not require prior written consent of the SPAC pursuant to the subsequent sentence of this Section 6.01 and (y) no such actions or failure to take such actions in specific response to COVID-19 or any COVID-19 Measures shall serve as a basis for the SPAC to assert that any of the conditions to the Closing contained herein have not been satisfied. Without limiting the generality of the foregoing, except as contemplated by this Agreement, any other Transaction Agreement, the Pre-Closing Step Plan or the Closing Step Plan, as set forth on Schedule 6.01, as consented to by the SPAC in writing (which consent shall not be unreasonably conditioned, withheld or delayed), or as required by Applicable Law, the Company shall not, and the Company shall cause its Subsidiaries not to, during the Interim Period:
(a) change or amend the Existing Company Articles or other organizational documents of the Company or any of its Subsidiaries;
(b) make, declare, set aside, establish a record date for or pay any dividend or distribution, other than (i) any dividends or distributions from any wholly-owned Subsidiary of the Company to the Company or any other wholly owned Subsidiaries of the Company and (ii) in the case of a Subsidiary that is a non-wholly-owned Subsidiary, dividends or distributions to each owner of the equity interests thereof, so long as any Subsidiary of the Company that holds equity interests in such class of equity interests shares at least ratably in such dividend or distribution with respect to such class of equity interests;
(c) enter into, assume, assign, partially or completely amend any material term of, modify any material term of or terminate (excluding any expiration in accordance with its terms) any collective bargaining or similar agreement (including agreements with works councils and trade unions and side letters) to which the Company or its Subsidiaries is a party or by which it is bound, other than in the ordinary course of business consistent with past practice;
(d) (i) issue, deliver, sell, transfer, pledge, dispose of or place any Lien (other than a Permitted Lien) on any shares or any other equity or voting securities of the Company or any of its Subsidiaries or (ii) issue any
A-55
Table of Contents
securities (including any shares, voting securities or loan capital) or grant any options, appreciation rights, share units, profits interests, warrants or other rights to purchase or obtain any shares or any other equity or voting securities or loan capital of the Company and/or any of its Subsidiaries, other than issuances of Existing Company Shares upon the exercise of any warrants to purchase such Existing Company Shares;
(e) sell, assign, transfer, convey, abandon, subject to a Lien, or otherwise dispose of any Owned Real Property;
(f) sell, assign, transfer, convey, lease, license, abandon, allow to lapse or expire, subject to or grant any Lien (other than Permitted Liens) on, or otherwise dispose of, any material assets, rights or properties (including Leased Real Property) of the Company and its Subsidiaries, taken as a whole, other than in the ordinary course of business;
(g) waive, release, compromise, settle or satisfy any pending or threatened claim (which shall include, but not be limited to, any pending or threatened Action) or compromise or settle any liability, (i) if such settlement would require payment by the Company in an amount greater than $1,000,000 individually or in the aggregate, (ii) to the extent such settlement includes an agreement to accept or concede material injunctive relief or (iii) to the extent such settlement involves a Governmental Authority or alleged criminal wrongdoing;
(h) agree to modify in any respect materially adverse to the Company and its Subsidiaries any confidentiality or similar Contract to which the Company or any of its Subsidiaries are a party;
(i) directly or indirectly acquire by merging or consolidating with, or by purchasing a substantial portion of the assets of, or by purchasing all of or a substantial equity interest in, or by any other manner, any business or any corporation, partnership, limited liability company, joint venture, association or other entity or Person or division thereof except for any transaction permitted pursuant to Section 6.01(i) of the Company Disclosure Schedule (provided, that, no such transaction listed on Section 6.01(i) of the Company Disclosure Schedule shall be permitted without the consent of the SPAC if the merger, consolidation, acquisition or similar transaction involves a target business with (x) PPP Loans or (y) operations or sales in any jurisdiction listed in Schedule 11.02(g));
(j) make any loans or advance any money or other property to any Person, except for (i) advances in the ordinary course of business to employees or officers of the Company or any of its Subsidiaries for expenses not to exceed $50,000 individually or $500,000 in the aggregate, (ii) prepayments and deposits paid to suppliers of the Company or any of its Subsidiaries in the ordinary course of business or (iii) trade credit extended to customers of the Company or any of its Subsidiaries in the ordinary course of business consistent with past practice;
(k) redeem, purchase or otherwise acquire any shares (or other equity interests) of the Company or any of its Subsidiaries or any securities or obligations convertible (whether currently convertible or convertible only after the passage of time or the occurrence of certain events) into or exchangeable for any shares of capital stock (or other equity interests) of the Company or any of its Subsidiaries;
(l) adjust, split, combine, subdivide, recapitalize, reclassify or otherwise effect any change in respect of any shares or other equity interests or securities of the Company, other than in connection with the Closing Step Plan;
(m) make any change in its customary accounting principles or methods of accounting materially affecting the reported consolidated assets, liabilities or results of operations of the Company and its Subsidiaries, other than (i) as may be required by Applicable Law, GAAP or regulatory guidelines, or (ii) in connection with any voluntary early adoption of Applicable Law, GAAP or regulatory guidelines;
(n) adopt or enter into a plan of complete or partial liquidation, dissolution, merger, consolidation or recapitalization of the Company or its Subsidiaries;
A-56
Table of Contents
(o) make, change or revoke any material Tax election, adopt or change any material accounting method with respect to Taxes, file any amended material Tax Return, settle or compromise any material Tax liability, surrender any right to claim a material refund of Taxes, consent to any extension or waiver of the limitations period applicable to any material tax claim or assessment (other than any extension pursuant to an extension to file any Tax Return), change its Tax residence or start to trade to a material extent through a permanent establishment or other taxable presence, or enter into any material closing agreement with respect to any Tax;
(p) except as otherwise required by any existing Employee Plan or Applicable Law (i) increase or grant any increase in the compensation, bonus, or other benefits (other than de minimis fringe or other benefits) of, or pay, grant or promise any bonus to, any current or former Key Employee, except for in connection with any promotion or material increase in responsibility of any such Key Employee in the ordinary course of business consistent with past practice (measured by applicable jurisdiction); (ii) grant or pay any severance (other than in the ordinary course of business consistent with past practice under a severance program, policy or practice previously provided or described to the SPAC and set forth on Section 4.18(a) of the Company Disclosure Schedule) or change in control pay or benefits to, or otherwise increase the severance or change in control pay or benefits of, any current or former Service Provider; (iii) enter into, amend (other than immaterial amendments) or terminate any Employee Plan or any employee benefit plan, policy, program, agreement, trust or arrangement that would have constituted an Employee Plan if it had been in effect on the date of this Agreement; (iv) take any action to accelerate the vesting or payment of, or otherwise fund or secure the payment of, any compensation or non-de minimis benefits under any Employee Plan; (v) grant any equity or equity based compensation awards; or (vi) hire or terminate (other than for cause) any Key Employee;
(q) voluntarily fail to maintain in full force and effect material insurance policies covering the Company and its Subsidiaries and their respective properties, assets and businesses in a form and amount consistent with past practices;
(r) enter into any transaction or amend in any material respect any existing agreement with any Related Party (excluding, to the extent permitted under Section 6.01(p), (i) ordinary course payments of annual compensation, provision of benefits or reimbursement of expenses in respect of members or shareholders who are officers or directors of the Company or its Subsidiaries and (ii) repayment or amendment of the terms of any of the Executive Loans);
(s) enter into any agreement that materially restricts the ability of the Company or its Subsidiaries to engage or compete in any line of business or enter into a new line of business;
(t) make any capital expenditure that in the aggregate exceeds $2,500,000, other than any capital expenditure (or series of related capital expenditures) consistent in all material respects with the Companys annual capital expenditures budget for periods following the date hereof;
(u) receive, collect, compile, use, store, process, share, safeguard, secure (technically, physically or administratively), dispose of, destroy, disclose, or transfer (including cross-border) any Personal Information (or fail to do any of the foregoing, as applicable) in violation of any (i) applicable Privacy Laws, (ii) external-facing privacy policies or notices of the Company or any of its Subsidiaries, or (iii) contractual obligations of the Company with respect to any Personal Information;
(v) incur, guarantee or otherwise become liable for (whether directly, contingently or otherwise) any Indebtedness, including any loans under the Payment Protection Program;
(w) enter into, assume, assign, partially or completely amend any material term of, modify any material term of or terminate (excluding any expiration in accordance with its terms) any Contract of a type required to be listed on Section 4.11(a) pursuant to clauses (ii) and (iii) and for which payments to or from the Company or any
A-57
Table of Contents
of its Subsidiaries would be expected to exceed $5,000,000 annually or $20,000,000 in the aggregate, other than entry into such agreements in the ordinary course of business consistent with past practice; or
(x) enter into any Contract, or otherwise become obligated, to do any action prohibited under this Section 6.01.
Section 6.02. Inspection. Subject to confidentiality obligations and similar restrictions that may be applicable to information furnished to the Company or any of its Subsidiaries by third parties that may be in the Companys or any of its Subsidiaries possession from time to time, and except for any information which (a) relates to the interactions with prospective buyers of the Company prior to the date hereof or their negotiation of this Agreement or the other Transaction Agreements or the Transactions, (b) is prohibited from being disclosed by Applicable Law or (c) on the advice of legal counsel of the Company would result in the loss of attorney-client privilege or other privilege from disclosure, the Company shall, and shall cause its Subsidiaries to, afford to the SPAC reasonable access during the Interim Period, during normal business hours and with reasonable advance notice, in such manner as to not interfere with the normal operation of the Company and its Subsidiaries, and so long as reasonably feasible or permissible under Applicable Law, to all of their respective properties, books, contracts, commitments, records and appropriate officers and employees of the Company and its Subsidiaries, and shall use its commercially reasonable efforts to furnish the SPAC with all financial and operating data and other information concerning the affairs of the Company and its Subsidiaries that are in the possession of the Company or its Subsidiaries, in each case, as the SPAC may reasonably request solely for purposes of consummating the Transactions; provided, however, that the SPAC shall not be permitted to perform any environmental sampling at any Leased Real Property, including sampling of soil, groundwater, surface water, building materials, or air or wastewater emissions. The Parties shall use commercially reasonable efforts to make alternative arrangements for such disclosure where the restrictions in the preceding sentence apply. Any request pursuant to this Section 6.02 shall be made in a time and manner so as not to delay the Closing. All information obtained by the SPAC under this Agreement shall be subject to the Confidentiality Agreement prior to the Closing.
Section 6.03. No Claim Against the Trust Account. The Company and each of the Sellers acknowledge that it has read the Prospectus and other SEC Documents, the Existing SPAC Certificate of Incorporation, the Existing SPAC Bylaws and the Trust Agreement and understands that the SPAC has established the Trust Account described therein for the benefit of the SPACs public stockholders and that disbursements from the Trust Account are available only in the limited circumstances set forth in the Trust Agreement. The Company and each Seller further acknowledge that, if the transactions contemplated by this Agreement, or, in the event of a termination of this Agreement, another Business Combination, are not consummated by July 2, 2022 or such later date as approved by the stockholders of the SPAC to complete a Business Combination, the SPAC will be obligated to return to its stockholders the amounts being held in the Trust Account. Accordingly, the Company and each Seller (on behalf of itself and its respective controlled Affiliates) hereby waives any past, present or future claim of any kind against, and any right to access, the Trust Account, the Trustee and the SPAC, or to collect from the Trust Account any monies that may be owed to them by the SPAC or any of its Affiliates for any reason whatsoever, and will not seek recourse against the Trust Account at any time for any reason whatsoever. This Section 6.03 shall survive the termination of this Agreement for any reason; provided that nothing herein shall serve to limit or prohibit the Companys or each Sellers right to pursue a claim against the SPAC or any of its Affiliates for legal relief against assets held outside the Trust Account (including from and after the consummation of a Business Combination other than as contemplated by this Agreement) or pursuant to Section 13.14 for specific performance or other injunctive relief.
Section 6.04. Pre-Closing Actions by the Company.
(a) As promptly as practicable following the date of this Agreement, the Company, the Charterhouse Parties and the SPAC will use their reasonable best efforts to agree on a final Pre-Closing Steps Plan, an illustrative version of which is attached as Exhibit I (which version, if the parties cannot agree, shall be deemed to be the
A-58
Table of Contents
agreed version). Prior to the Closing, the Company shall, and shall cause its Subsidiaries to, to take, or cause to be taken, all actions and to do, or cause to be done, all things necessary to effectuate the agreed-upon Pre-Closing Step Plan, with such amendments, modifications, re-orderings and the like as the Company may determine to be reasonably necessary and desirable to effect the Transactions; provided that the Company shall not make any such amendment, modification, re-ordering or the like without the consent of the SPAC and the Charterhouse Parties (not to be unreasonably withheld, conditioned or delayed), other than any such amendment, modification, re-ordering or the like having an immaterial impact on the SPAC, the stockholders of the SPAC or the holders of the Public Warrants or Private Placement Warrants. The Company shall keep the SPAC and the Charterhouse Parties reasonably informed with respect to the Pre-Closing Steps Plan and make available to the SPAC copies of any transfer, assignment or other relevant document pursuant to which the Pre-Closing Step Plan shall be effected, which shall be in form and substance reasonably acceptable to each of the Company, the Charterhouse Parties and the SPAC.
(b) The Company shall take the actions set forth in Section 6.04(b) of the Company Disclosure Schedule to supplement existing policies, procedures and practices concerning sales channel partners and shall, together with the SPAC, jointly instruct and work with Weil, Gotshal & Manges LLP (or such other counsel as the parties agree) at the cost and expense of the SPAC to direct and oversee the due diligence process in relation to the sales channels partners located or making sales in the jurisdictions set out in Schedule 11.02(g) (each such partner an HRP). The Company, the SPAC and its representatives shall use commercially reasonable efforts and cooperate with each other in connection with the Company taking each of the actions set forth on Section 6.04(b) of the Company Disclosure Schedule as soon as practicable after the date of this Agreement.
Section 6.05. Drag Along.
(a) The Charterhouse Parties shall, promptly following the effectiveness of the Registration Statement (and in any event no later than five (5) Business Days after such effectiveness): (i) cause the holders of a majority of the Existing Company Shares, which holders, together with the Supporting Company Holders, shall be sufficient to constitute the Vendor Shareholders (as defined in the Existing Company Articles), to deliver to (A) the SPAC and the Company a duly executed Joinder Agreement and a duly executed Election Agreement and (B) the Exchange Agent, such Election Form and, if applicable, properly completed stock transfer form(s), in each case in respect of the Existing Company Shares and Loan Notes held by such holder; and (ii) cause the applicable Vendor Shareholders to provide a Drag Along Notice to the Called Shareholders, with such notice to contain such information as is required by and to be served in accordance with the Existing Company Articles. To the extent (i) any Existing Company Shares and/or Loan Notes are transferred to any third party other than pursuant to the terms of this Agreement or (ii) any securities (including any shares, voting securities or loan capital) are issued in the capital of the Company or any of its Subsidiaries during the Interim Period, the Charterhouse Parties shall cause the Vendor Shareholders to provide a Drag Along Notice, with such notice to contain such information as is required by and to be served in accordance with the Existing Company Articles, to any and all owners of share or loan capital of the Company or any of its Subsidiaries from time to time as soon as reasonably practicable (and in any event no later than five (5) Business Days prior to the Closing Date).
(b) Promptly following the provision of the Drag Along Notice by the Vendor Shareholders to the Called Shareholders in accordance with Section 6.05(a), the Charterhouse Parties shall cause all Called Shareholders to deliver, at least five (5) Business Days prior to the Closing Date, (i) to the SPAC and the Company a duly executed Joinder Agreement and a duly executed Election Agreement and (ii) to the Exchange Agent, such Election Agreement and properly completed stock and/or note transfer form(s), in each case in respect of the Existing Company Shares and the Loan Notes held by such Called Shareholder. Subject to the satisfaction of the obligations in Section 6.05(a), in the event that any Called Shareholder has not delivered to the SPAC and the Company a duly executed Joinder Agreement and a duly executed Election Agreement (and to the Exchange Agent such Election Agreement and properly completed stock and/or note transfer form(s)) prior to the date that is five (5) Business Days prior to the Closing Date, then the Company shall cause any director of the Company or any Vendor Shareholder to, pursuant to Article 48.10 of the Existing Company Articles, execute and deliver a
A-59
Table of Contents
Joinder Agreement, an Election Agreement and such stock and/or note transfer form(s) (and any such other agreements or documents necessary) in the name of and as an agent for any such Called Shareholder, effective as of the Closing.
(c) From and after the date hereof until the earlier of the Closing or the termination of this Agreement in accordance with Section 12.01, none of the Charterhouse Parties shall sell, transfer or assign any of such Charterhouse Partys Existing Company Shares, other than to an Affiliate of such Charterhouse Party who agrees to be bound by the terms and conditions of the Existing Company Articles and of this Agreement.
(d) Prior to the Closing, the Charterhouse Parties shall not consent to any transfer of any Existing Company Shares without the prior written consent of the SPAC other than to an Affiliate of such transferee who agrees to be bound by the terms and conditions of the Existing Company Articles and of this Agreement.
Section 6.06. Termination of Affiliate Transactions. Except for the Contracts set forth on Section 6.06 of the Company Disclosure Schedule, the Company, the Sellers and the other parties thereto, as applicable, shall cause the Contracts set forth on Section 4.21 of the Company Disclosure Schedule and any Contract that would have been listed on Section 4.21 of the Company Disclosure Schedule if such Contracts were identified prior to the date hereof or entered into as of the date hereof, respectively, to be settled and terminated prior to the Closing without any further or continuing liability on the part of the Company or any of its Subsidiaries or the SPAC or any of its Affiliates.
Section 6.07. Title Insurance Cooperation. From the date hereof until the Closing, the Company shall reasonably cooperate with the SPAC and the SPACs lender to obtain, owners and lenders title insurance policies with respect to the Owned Real Property, dated as of the Closing Date, issued from a title insurance company and in amounts reasonably satisfactory to the SPAC, including to deliver such customary affidavits from officers of the Company and its Subsidiaries as reasonably requested by the title insurance company , including any affidavit required by the title company in order to issue a non-imputation endorsement.
Section 6.08. No SPAC Securities Transactions. Neither the Company, any Seller nor any of their respective controlled Affiliates, directly or indirectly, shall engage in any transactions involving the securities of the SPAC prior to the time of the making of a public announcement regarding all of the material terms of the Transactions. The Company and each Seller shall use its respective reasonable best efforts to require each of its respective officers, directors, employees, agents, advisors, contractors, associates, clients, customers and representatives to comply with the foregoing requirement.
Section 6.09. Repayment of Employee Loans. No later than ten (10) Business Days prior to the Closing, the Company shall take such action as may be necessary such that, as of the Closing, (i) all Executive Loans shall be terminated (including by way of automatic termination in accordance with its terms) and of no further continued force or effect without any obligations or liabilities surviving the Closing, and (ii) all accounts payable to either party to such agreements shall be settled and fully discharged with no further obligation or liability to either party.
Section 6.10. Debt Financing.
(a) The Company shall cause the DCL Beneficiary to use its reasonable best efforts to, and shall cause each of its Subsidiaries to use its reasonable best efforts to, arrange and obtain the Debt Financing on terms and conditions not less favorable than (taken as a whole) those set forth in the Debt Commitment Letter (or such other terms as are reasonably acceptable to the DCL Beneficiary and the SPAC, such consent of the SPAC not to be unreasonably withheld, delayed or conditioned) and prior to the End Date (as may be extended pursuant to Section 12.01(b)), including using reasonable best efforts to take all actions within its control to (i) maintain in effect the Debt Commitment Letter (subject to any amendment, supplement, replacement, substitution, termination or other modification or waiver that is not prohibited by clause (c) below), (ii) promptly negotiate
A-60
Table of Contents
and enter into definitive agreements with respect thereto on the terms and conditions contained in the Debt Commitment Letter (including the flex provisions) or on other terms no less favorable (taken as a whole) to the DCL Beneficiary and/or its applicable Subsidiaries (or such other terms as are reasonably acceptable to the DCL Beneficiary (or its applicable Subsidiaries) and the SPAC (such consent of the SPAC not to be unreasonably withheld, delayed or conditioned), (iii) satisfy or obtain a waiver thereof on a timely basis all conditions applicable to the DCL Beneficiary and/or its Subsidiaries in the Debt Commitment Letter and such definitive agreements thereto that are within its (or their) control, (iv) assuming that all conditions contained in the Debt Commitment Letter have been satisfied, consummate the Debt Financing at or prior to the Closing and (v) enforce its rights under the Debt Commitment Letter.
(b) At the reasonable request of the SPAC from time to time, the Company shall keep the SPAC reasonably informed as to the status of the Debt Financing process. The Company shall give the SPAC prompt notice (i) of the termination, written repudiation, rescission, cancellation or expiration of the Debt Commitment Letter or the definitive agreements related to the Debt Financing, (ii) of any material breach or default (or any event or circumstance that, with or without notice, lapse of time or both, would reasonably be expected to give rise to a material breach or material default) by any party to the Debt Commitment Letter or any definitive agreements related to the Debt Financing, in each case of which the Company becomes aware, (iii) of the receipt of any written notice or other written communication, in each case received from any Debt Financing Source with respect to any (A) material breach of the Company or any of its Subsidiaries obligations under the Debt Commitment Letter or definitive agreements related to the Debt Financing, or material default, termination or repudiation by any party of the Debt Commitment Letter or definitive agreements related to the Debt Financing or (B) material dispute between or among any parties to the Debt Commitment Letter or definitive agreements related to the Debt Financing or any provisions of the Debt Commitment Letter, with respect to the obligation to fund the Debt Financing or the amount of the Debt Financing to be funded at the Closing or (iv) if, at any time, the Company believes in good faith that it will not be able to obtain all or any portion of the Debt Financing on terms and conditions, in the manner, or from the sources contemplated by the Debt Commitment Letter or definitive agreements related to the Debt Financing; provided that in no event shall the Company be under any obligation to disclose any information pursuant to the foregoing clauses (A) or (B) that would waive the protection of attorney-client or similar privilege if the Company shall have used reasonable best efforts to disclose such information in a way that would not waive such privilege and provided notice to the SPAC that such information is being withheld on such basis. As soon as reasonably practicable, the Company shall provide any information reasonably requested in writing by the SPAC relating to any circumstance referred to in clause (i), (ii), (iii) or (iv) of the immediately preceding sentence (subject to the exclusions and qualifications set forth in the proviso of the immediately preceding sentence).
(c) The Company and the DCL Beneficiary shall have the right from time to time to amend, supplement or otherwise modify or waive its rights under the Debt Commitment Letter with the consent of the SPAC (such consent not to be unreasonably withheld, delayed or conditioned). The Company shall furnish to the SPAC a copy of any amendment, modification, waiver or consent of or relating to the Debt Commitment Letter promptly upon execution thereof.
(d) The Company and the SPAC each acknowledge and agree that the obtaining of any financing is not a condition to the Closing.
(e) For purposes of this Agreement (other than with respect to representations made by the Company and/or the DCL Beneficiary that speak as of the date hereof), references to (i) Debt Financing shall include the financing contemplated by the Debt Commitment Letter as permitted or contemplated to be amended, modified, supplemented, restated, replaced or substituted by Sections 6.10(a), 6.10(c) and/or 8.09, (ii) Debt Commitment Letter shall also include any amendment, modification, restatement, supplement and replacement or substitution permitted or contemplated by Sections 6.10(a), 6.10(c) and/or 8.09, along with any flex provisions or other similar terms set forth therein and (iii) Debt Financing Sources shall include lenders and other financing sources (including underwriters, placement agents and initial purchasers) providing the Debt Financing pursuant
A-61
Table of Contents
to the Debt Commitment Letter, as permitted to be amended, modified, supplemented, restated, replaced or substituted by Sections 6.10(a), 6.10(c) and/or 8.09.
Section 6.11. PPP Loans. Prior to the Closing, the Company shall, or shall cause its applicable Subsidiaries to, repay in full any PPP Loans outstanding as of the date of this Agreement (including any PPP Loans that as of the date hereof are subject to (i) an application for forgiveness, (ii) an audit by the U.S. Small Business Administration or the lender of such PPP Loan, or (iii) an escrow agreement).
ARTICLE 7
COVENANTS OF THE SPAC
The SPAC agrees that:
Section 7.01. Conduct of Business During the Interim Period.
(a) During the Interim Period, except as set forth on Section 7.01 of the SPAC Disclosure Schedule or as contemplated by this Agreement, any other Transaction Agreement, the Closing Step Plan, the PIPE Investment, as required by Applicable Law or as consented to by the Company and the Charterhouse Parties in writing (which consent shall not be unreasonably conditioned, withheld, delayed or denied, except, in the case of clauses (i), (ii), (iv), (vi) and (vii) below, as to which the Companys and the Charterhouse Parties consent may be granted or withheld in their sole discretion), the SPAC shall not:
(i) change, modify or amend the Trust Agreement, the Existing SPAC Certificate of Incorporation or the Existing SPAC Bylaws;
(ii) declare, set aside or pay any dividends on, or make any other distribution in respect of any outstanding capital stock of, or other equity interests in, the SPAC; split, combine or reclassify any capital stock of, or other equity interests in, the SPAC; or other than in connection with the SPAC Stockholder Redemptions or as otherwise required by the Existing SPAC Certificate of Incorporation or the Existing SPAC Bylaws in order to consummate the transactions contemplated hereby, repurchase, redeem or otherwise acquire, or offer to repurchase, redeem or otherwise acquire, any capital stock of, or other equity interests in, the SPAC;
(iii) make, change or revoke any material Tax election, adopt or change any material accounting method with respect to Taxes, file any amended material Tax Return, settle or compromise any material Tax liability, surrender any right to claim a material refund of Taxes, consent to any extension or waiver of the limitations period applicable to any material tax claim or assessment (other than any extension pursuant to an extension to file any Tax Return), change its Tax residence or start to trade to a material extent through a permanent establishment other taxable presence, or enter into any material closing agreement with respect to any Tax;
(iv) enter into, renew or amend in any material respect any transaction or Contract with a Related Party of the SPAC (including, for the avoidance of doubt, (x) the Sponsors or anyone related by blood, marriage or adoption to any director or officer of Sponsor and (y) any Person in which any Sponsor has a direct or indirect legal, contractual or beneficial ownership interest of five percent (5%) or greater), in each case, other than on arms length terms in the ordinary course of business;
(v) waive, release, compromise, settle or satisfy any pending or threatened material claim (which shall include, but not be limited to, any pending or threatened Action) or compromise or settle any liability;
(vi) incur, guarantee or otherwise become liable for (whether directly, contingently or otherwise) any Indebtedness; or
(vii) offer, issue, deliver, grant or sell, or authorize or propose to offer, issue, deliver, grant or sell, any capital stock of, other equity interests, equity equivalents, stock appreciation rights, phantom stock
A-62
Table of Contents
ownership interests or similar rights in, the SPAC or any securities convertible into, or any rights, warrants or options to acquire, any such capital stock or equity interests, other than the issuance of New SPAC Class A Common Shares at not less than $10 per share on the terms set forth in the Subscription Agreements;
(viii) amend, modify or waive any of the terms or rights set forth in any Private Placement Warrant;
(ix) merge or consolidate itself with any Person, restructure, reorganize or completely or partially liquidate or dissolve, or adopt or enter into a plan of complete or partial liquidation, dissolution, restructuring, recapitalization or other reorganization of the SPAC (other than the Transactions); or
(x) enter into any agreement, or otherwise become obligated, to do any action prohibited under this Section 7.01.
(b) During the Interim Period, the SPAC shall comply with, and continue performing under, as applicable, the Existing SPAC Certificate of Incorporation and the Existing SPAC Bylaws, the Trust Agreement, the Transaction Agreements and all other agreements or contracts to which SPAC or its Subsidiaries may be a party.
Section 7.02. SPAC Board of Directors. The SPAC shall take all necessary action to cause the Board of Directors of the SPAC as of immediately following the Closing to consist of nine (9) directors, of whom one (1) shall be the Chief Executive Officer of the SPAC upon the Closing (i.e., the Chief Executive Officer of the Company immediately prior to the Closing), two (2) shall be named by the Sponsor, one (1) shall be named by the Charterhouse Parties and the remainder shall be mutually agreed by the Charterhouse Parties, the Company and the SPAC prior to the Closing.
Section 7.03. Governing Documents. Prior to the Closing and immediately prior to the closing of the PIPE Financing, the SPAC shall file the New SPAC Certificate of Incorporation with the Secretary of State of the State of Delaware, which shall provide, among other things, that (i) the SPAC will have two classes of common stock, New SPAC Class A Common Shares and New SPAC Class B Common Shares, and (ii) the Board of Directors of the SPAC will be composed of up to nine (9) directors.
Section 7.04. Registration Rights Agreement. Concurrently with the Closing, the SPAC shall cause the existing Registration Rights Agreement, dated June 29, 2020, by and among the SPAC, the Sponsor, the Charterhouse Parties and the other parties listed on the signature pages thereto, to be amended and restated in the form of the Amended and Restated Registration Rights Agreement attached as Exhibit E hereto.
Section 7.05. NYSE Listing. From the date hereof through the Closing, the SPAC shall use reasonable best efforts to ensure that the SPAC remains listed as a public company, and that shares of Class A common stock of the SPAC remain listed, on the NYSE. The SPAC shall use reasonable best efforts to ensure that the New SPAC Class A Common Shares are listed on the NYSE, as of the Closing.
Section 7.06. SPAC Public Filings. From the date hereof through the Closing, the SPAC shall use reasonable best efforts to keep current and timely file all reports required to be filed or furnished with the SEC and otherwise comply in all material respects with its reporting obligations under Applicable Law.
Section 7.07. PIPE Subscription Agreements. Unless otherwise approved in writing by the Company and the Charterhouse Parties, the SPAC shall not permit any material amendment or modification to be made to, or any waiver of any provision or remedy under, or any replacements or terminations of, the PIPE Subscription Agreements in any manner adverse to the Company or the Charterhouse Parties. The SPAC shall use its reasonable best efforts to take, or cause to be taken, all actions and do, or cause to be done, all things necessary, proper or advisable to consummate the transactions contemplated by the PIPE Subscription Agreements on the terms and conditions described therein, including using its reasonable best efforts to enforce its rights under the PIPE Subscription Agreements to cause the PIPE Investors to pay to (or as directed by) the SPAC the applicable
A-63
Table of Contents
purchase price under each PIPE Investors applicable Subscription Agreement in accordance with its terms. The SPAC shall not permit any PIPE Investor to transfer any New SPAC Common Stock that would trigger any filing or notification with a Regulatory Consent Authority, other than those filings and notifications listed on Section 11.01(c) of the Company Disclosure Schedule, without the prior written consent of the Company and the Charterhouse Parties.
Section 7.08. Backstop Agreement. Unless otherwise approved in writing by the Company and the Charterhouse Parties, the SPAC shall not permit any material amendment or modification to be made to, or any waiver of any provision or remedy under, or any replacement or termination of, the Backstop Agreement in any manner adverse to the Company or the Charterhouse Parties. The SPAC shall use its reasonable best efforts to take, or cause to be taken, all actions and do, or cause to be done, all things necessary, proper or advisable to consummate the transactions contemplated by the Backstop Agreement on the terms and conditions described therein, including using its reasonable best efforts to enforce its rights under the Backstop Agreement to cause the Backstop Party to pay to (or as directed by) the SPAC the applicable purchase price under the Backstop Agreement in accordance with its terms. The SPAC shall not permit the Backstop Party to transfer any New SPAC Common Stock that would trigger any filing or notification with a Regulatory Consent Authority, other than those filings and notifications listed on Section 11.01(c) of the Company Disclosure Schedule, without the prior written consent of the Company and the Charterhouse Parties.
Section 7.09. Trust Account. Upon the satisfaction (or waiver by the SPAC) of the conditions set forth in Article 11, and in accordance with and pursuant to the Trust Agreement (i) at the Closing, (A) the SPAC shall cause the documents, opinions and notices required to be delivered to the Trustee pursuant to the Trust Agreement to be so delivered and (B) the SPAC shall make arrangements to cause the Trustee to (1) pay as and when due all amounts payable to Existing SPAC Investors who shall have previously validly elected to redeem their shares of Existing SPAC Common Stock pursuant to a SPAC Stockholder Redemption and (2) promptly thereafter, pay all remaining amounts then available in the Trust Account in accordance with this Agreement and the Trust Agreement and (ii) thereafter, the Trust Account shall terminate, except as otherwise provided in the Trust Agreement. The SPAC shall not amend, supplement or otherwise modify or waive its rights under the Trust Agreement without the prior written consent of each of the Charterhouse Parties and the Company.
Section 7.10. Financing Cooperation; Alternative Financing. The SPAC shall use its reasonable best efforts to, and shall cause its respective representatives to use their reasonable best efforts to, provide all cooperation in connection with the arrangement of the Debt Financing as may be reasonably requested by Company that is necessary or customary for financings of the type contemplated by the Debt Commitment Letter.
ARTICLE 8
JOINT COVENANTS
The SPAC, the Company and the Sellers agree that:
Section 8.01. Best Efforts; Further Assurances.
(a) Subject to the terms and conditions of this Agreement and Section 4.03 of the Company Disclosure Schedule and Section 5.03 of the SPAC Disclosure Schedule, the SPAC, the Sellers and the Company will use their respective reasonable best efforts to take, or cause to be taken, all actions and to do, or cause to be done, all things necessary or advisable under Applicable Law to consummate the Transactions. Without limiting the foregoing, the SPAC agrees to take all steps necessary or advisable to eliminate impediments under any antitrust, competition, or other Applicable Law (including mitigation measures imposed by CFIUS, ITAR or MINEFI) that are asserted by any Governmental Authority or any other party having jurisdiction over the Transactions so as to enable the Parties to close the Transactions prior to the End Date, as may be extended, including but not limited to (i) negotiating, committing to and effecting by consent decree, hold separate orders, or otherwise, the sale,
A-64
Table of Contents
divestiture or disposition of such assets, categories of assets or businesses of the SPAC or the Company; (ii) terminating existing relationships, contractual rights or obligations; (iii) terminating any venture or other arrangement; (iv) licensing any portion of the business of the SPAC or of the assets being purchased; and (v) defending through litigation on the merits any claim asserted in court by any party in order to avoid entry of, or to have vacated or terminated, any decree, order or judgment that would prevent the Closing from occurring prior to the End Date; provided that, notwithstanding anything to the contrary in this Agreement, other than as relates to removing any impediment under any antitrust or competition Applicable Law, unless mutually agreed by the SPAC, the Charterhouse Parties and the Company, the SPAC will not be required to take (and the Sellers, the Company and their Subsidiaries shall not take, without the prior written consent of the SPAC) any actions to satisfy the conditions in Section 11.01(a) or Section 11.01(c) herein that, individually or in the aggregate, would reasonably be expected to result in the consequences set forth on Section 8.01 of the Company Disclosure Schedule or (2) result in any other person designated by a Governmental Authority becoming an equity investor or being granted the right to a seat on the board of directors, board of managers, or similar managing body of the SPAC, the Company or any of the Companys material Subsidiaries. The SPAC, the Company and the Sellers agree to execute and deliver such other documents, certificates, agreements and other writings and to take such other actions as may be necessary or advisable in order to consummate or implement expeditiously the Transactions.
(b) In furtherance and not in limitation of the foregoing, each of the SPAC, the Company and the Sellers shall, as applicable, make an appropriate filing of a Notification and Report Form pursuant to the HSR Act and the other filings and notices set forth on Sections 4.03 of the Company Disclosure Schedule and 5.03 of the SPAC Disclosure Schedule with respect to the Transactions as promptly as practicable after the date hereof and to supply as promptly as practicable any additional information and documentary material that may be requested pursuant to such filings and notifications and to take all other actions necessary to obtain all required consents or approvals or cause the expiration or termination of any applicable waiting period as soon as practicable and advisable. The SPAC, the Sellers and the Company shall: (a) promptly inform the other of any communication to or from any Governmental Authority regarding the Transactions; (b) permit each other to review in advance any proposed written communication to any such Governmental Authority and consider reasonable comments thereto (c) not agree to participate in any substantive meeting or discussion with any such Governmental Authority in respect of any filing, investigation or inquiry concerning this Agreement or the Transactions unless, to the extent reasonably practicable, it consults with the other Party in advance and, to the extent permitted by such Governmental Authority, gives the other Party the opportunity to attend; and (e) promptly furnish each other with copies of all correspondence, filings (not to include filings made pursuant to the HSR Act) and written communications (to the extent allowed under Applicable Law and redacted or limited to outside counsel only as appropriate to protect confidential or commercially sensitive information) between such Party and their Affiliates and their respective agents, representatives and advisors, on one hand, and any such Governmental Authority, on the other hand, in each case, with respect to this Agreement and the Transactions.
Section 8.02. Certain Filings. Not in limitation of Section 8.01, the Sellers, the SPAC and the Company shall cooperate with one another (i) in determining whether any action by or in respect of, or filing with, any Governmental Authority is required, or any actions, consents, approvals or waivers are required to be obtained from parties to any Material Contracts, in connection with the consummation of the Transactions and (ii) in using their respective reasonable best efforts to take such actions or make any such filings, furnishing information required in connection therewith and seeking timely to obtain any such actions, consents, approvals or waivers (including with respect to the matters set forth on Schedule 8.02).
Section 8.03. Indemnification and Insurance.
(a) The SPAC agrees that all rights held by each present and former director and officer of the Company and any of its Subsidiaries to indemnification and exculpation from liabilities for acts or omissions occurring at or prior to the Closing, whether asserted or claimed prior to, at or after the Closing, provided in the respective
A-65
Table of Contents
organizational documents of the Company or such Subsidiary in effect on the date of this Agreement shall survive the transactions contemplated hereby and shall continue in full force and effect. Without limiting the foregoing, the SPAC shall cause the Company and each of its Subsidiaries (i) to maintain for a period of not less than six (6) years from the Closing provisions in its applicable organizational documents concerning the indemnification and exculpation (including provisions relating to expense advancement) of the Companys and its Subsidiaries former and current officers, directors, employees, and agents that are no less favorable to those Persons than the provisions of the organizational documents of the Company or such Subsidiary, as applicable, in each case, as of the date of this Agreement and (ii) not to amend, repeal or otherwise modify such provisions in any manner that would adversely affect the rights of those Persons thereunder, in each case, except as required by Applicable Law.
(b) The Parties shall negotiate in good faith to establish a directors and officers liability insurance policy, to be in place as of the Closing, that includes full prior acts coverage and continuity. If the Parties are unable to establish such a policy within fourteen (14) days prior to the anticipated Closing Date, after using their respective commercially reasonable efforts in good faith to do so, the Company shall cause coverage to be extended under the Companys current directors and officers liability insurance policies by obtaining a six (6) year tail policy containing terms not materially less favorable than the terms of such current insurance coverage with respect to claims existing or occurring at or prior to the Closing. If any claim is asserted or made within such six (6) year period, the provisions of this Section 8.03 shall be continued in respect of such claim until the final disposition thereof.
(c) Notwithstanding anything contained in this Agreement to the contrary, this Section 8.03 shall survive the consummation of the transactions contemplated hereby indefinitely and shall be binding, jointly and severally, on all successors and assigns of the SPAC and the Company. In the event that the SPAC or the Company or any of their respective successors or assigns consolidates with or merges into any other Person and shall not be the continuing or surviving corporation or entity of such consolidation or merger or transfers or conveys all or substantially all of its properties and assets to any Person, then, and in each such case, proper provision shall be made so that the successors and assigns of the SPAC or the Company, as the case may be, shall succeed to the obligations set forth in this Section 8.03.
(d) The SPAC shall maintain customary D&O insurance on behalf of any Person who is or was a director or officer of the SPAC (at any time, including prior to the date hereof) against any liability asserted against such Person and incurred by such Person in any such capacity, or arising out of such Persons status as such, whether or not the SPAC would have the power to indemnify such Person against such liability under the provisions of the New SPAC Certificate of Incorporation, the New SPAC Bylaws, Section 145 of the DGCL or any other provision of Applicable Law.
Section 8.04. Registration Statement; Proxy Statement; SPAC Special Meeting.
(a) As promptly as practicable after the date of this Agreement, the SPAC and the Company shall, in accordance with this Section 8.04(a), prepare, and the SPAC shall file with the SEC, (i) in preliminary form, the Proxy Statement, to be filed as part of the Registration Statement and sent to the Existing SPAC Stockholders in advance of the SPAC Special Meeting, for the purpose of, among other things: (a) providing the Existing SPAC Stockholders with the opportunity to redeem Existing SPAC Class A Common Shares by tendering such shares for redemption not later than 5:00 p.m. Eastern Time on the date that is two (2) Business Days prior to the date of the SPAC Special Meeting (the SPAC Stockholder Redemption), and (b) soliciting proxies from holders of Existing SPAC Class A Common Shares to vote at the SPAC Special Meeting, as adjourned or postponed, in favor of the Transaction Proposals and (ii) the Registration Statement, in which the Proxy Statement will be included as a prospectus. Without the prior written consent of the Company and the Charterhouse Parties, the Transaction Proposals shall be the only matters (other than procedural matters) which the SPAC shall propose to be acted on by the Existing SPAC Stockholders at the SPAC Special Meeting, as adjourned or postponed. The SPAC and the Company shall use commercially reasonable efforts to cooperate with each other and their
A-66
Table of Contents
respective representatives in the preparation of the Registration Statement and the Proxy Statement (including by executing and delivering to counsel of the Company and/or the SPAC, as the case may be, letters of representation customary for transactions of this type and reasonably satisfactory to counsel of the Company and/or the SPAC). The Registration Statement and the Proxy Statement will comply as to form and substance with the applicable requirements of the Securities Act and Exchange Act, as applicable, and the rules and regulations thereunder. The SPAC shall (A) have the Registration Statement declared effective under the Securities Act as promptly as practicable after the filing thereof and keep the Registration Statement effective as long as is necessary to consummate the Transactions, (B) file the definitive Proxy Statement with the SEC, (C) cause the Proxy Statement to be mailed to its Existing SPAC Stockholders of record, as of the record date to be established by the board of directors of the SPAC in accordance with Section 8.04(f), as promptly as practicable following the effective date of the Registration Statement and (C) promptly (and in no event later than the fifth (5th) Business Day following the date of this Agreement) commence a broker search in accordance with Rule 14a-12 of the Exchange Act.
(b) Prior to filing with the SEC, the SPAC will make available to the Company and the Charterhouse Parties drafts of the Registration Statement, Proxy Statement and any other documents to be filed with the SEC, both preliminary and final, and any amendment or supplement to the Registration Statement, Proxy Statement or such other document and will provide the Company and the Charterhouse Parties with a reasonable opportunity to comment on such drafts and shall consider such comments in good faith. Other than routine filings required by the Exchange Act, the SPAC shall not file any such documents with the SEC without the prior written consent of the Company and the Charterhouse Parties (such consent not to be unreasonably withheld, conditioned or delayed). The SPAC will advise the Company promptly after it receives notice thereof, of: (i) the time when the Registration Statement and the Proxy Statement has been filed; (ii) the time when the Registration Statement has been declared effective under the Securities Act; (iii) the filing of any supplement or amendment to the Registration Statement or the Proxy Statement; (iv) any request by the SEC for amendment of the Registration Statement or the Proxy Statement; (v) any comments from the SEC relating to the Registration Statement or the Proxy Statement and responses thereto; and (vi) requests by the SEC for additional information. The SPAC shall respond to any SEC comments on the Registration Statement and the Proxy Statement as promptly as practicable; provided, that prior to responding to any requests or comments from the SEC, the SPAC will make available to the Company and the Charterhouse Parties drafts of any such response and provide the Company and the Charterhouse Parties with a reasonable opportunity to comment on such drafts.
(c) Notwithstanding anything in this Agreement to the contrary, if, at any time prior to the SPAC Special Meeting, there shall be discovered any information that should be set forth in an amendment or supplement to the Registration Statement or the Proxy Statement so that the Registration Statement or the Proxy Statement would not include any misstatement of a material fact or omit to state any material fact necessary to make the statements therein, in light of the circumstances under which they were made, not misleading, the SPAC shall promptly file an amendment or supplement to the Registration Statement or the Proxy Statement, as applicable, containing such information. If, at any time prior to the Closing, the Company or any Seller discovers any information, event or circumstance relating to the Company such Seller, its or their respective business or any of its or their respective Affiliates, officers, directors or employees that should be set forth in an amendment or a supplement to the Registration Statement or the Proxy Statement so that the Registration Statement and the Proxy Statement would not include any misstatement of a material fact or omit to state any material fact necessary to make the statements therein, in light of the circumstances under which they were made, not misleading, then the Company or such Seller, as applicable, shall promptly inform the SPAC of such information, event or circumstance.
(d) The SPAC shall make all necessary filings with respect to the Transactions under the Securities Act, the Exchange Act and applicable blue sky laws, and any rules and regulations thereunder. The Company agrees to promptly provide the SPAC with all information concerning the business, management, operations and financial condition of the Company and its Subsidiaries, in each case, reasonably requested by the SPAC for inclusion in the Registration Statement and the Proxy Statement.
A-67
Table of Contents
(e) Without limiting the generality of Section 8.04(d), the Company shall promptly furnish to the SPAC for inclusion in the Registration Statement and the Proxy Statement, (i) audited consolidated financial statements of the Company and its Subsidiaries as of and for the years ended June 30, 2018, 2019 and 2020, prepared in accordance with GAAP and Regulation S-X and audited by the Companys independent auditor in accordance with PCAOB auditing standards; (ii) other financial statements, reports and information with respect to the Company and its Subsidiaries that may be required to be included in the Registration Statement and the Proxy Statement under the rules of the SEC and (iii) auditors reports and consents to use such financial statements and reports therein.
(f) The SPAC shall, prior to or as promptly as practicable following the effective date of the Registration Statement, establish a record date (which date shall be mutually agreed with the Company) for, duly call and give notice of, the SPAC Special Meeting. The SPAC shall convene and hold the SPAC Special Meeting, for the purpose of obtaining the approval of the Transaction Proposals, which meeting shall be held as promptly as practicable following the declaration of effectiveness of the Registration Statement and not more than forty-five (45) days after the date on which the SPAC commences the mailing of the Proxy Statement to its stockholders. The SPAC shall use its reasonable best efforts to take all actions necessary (in its discretion or at the request of the Company) to obtain the approval of the Transaction Proposals at the SPAC Special Meeting, including as such SPAC Special Meeting may be adjourned or postponed in accordance with this Agreement, including by soliciting proxies as promptly as practicable in accordance with Applicable Law for the purpose of seeking such approval. The SPAC shall include its board recommendation in the Proxy Statement. The board of directors of the SPAC shall not (and no committee or subgroup thereof shall) change, withdraw, withhold, qualify or modify, or publicly propose to change, withdraw, withhold, qualify or modify, such board recommendation for any reason. Notwithstanding anything to the contrary contained in this Agreement, the SPAC shall be entitled to (and, in the case of the following clauses (ii) and (iii), at the request of the Company, shall) postpone or adjourn the SPAC Special Meeting for a period of no longer than fifteen (15) days: (i) to ensure that any supplement or amendment to the Proxy Statement that the board of directors of the SPAC has determined in good faith is required by Applicable Law is disclosed to the Existing SPAC Stockholders and for such supplement or amendment to be promptly disseminated to such Existing SPAC Stockholders prior to the SPAC Special Meeting; (ii) if, as of the time for which the SPAC Special Meeting is originally scheduled (as set forth in the Proxy Statement), there are insufficient Existing SPAC Class A Common Shares represented (either in person or by proxy) to constitute a quorum necessary to conduct the business to be conducted at the SPAC Special Meeting; (iii) in order to solicit additional proxies from Existing SPAC Stockholders for purposes of obtaining approval of the Transaction Proposals; or (iv) only with the prior written consent of the Company to seek withdrawal of redemption requests, for purposes of satisfying the Minimum Cash Condition; provided, that, notwithstanding any longer adjournment or postponement period specified at the beginning of this sentence, in the event of any such postponement or adjournment, the SPAC Special Meeting shall be reconvened as promptly as practicable following such time as the matters described in such clauses have been resolved.
Section 8.05. Form 8-K Filings. The SPAC, the Company and the Charterhouse Parties shall mutually agree upon and issue a press release announcing the effectiveness of this Agreement. The SPAC, the Company and the Charterhouse Parties shall cooperate in good faith with respect to the prompt preparation of, and, as promptly as practicable after the date hereof (but in any event within four (4) Business Days thereafter), the SPAC shall file with the SEC a Current Report on Form 8-K pursuant to the Exchange Act to report the execution of this Agreement. Prior to the Closing, the SPAC, the Company and the Charterhouse Parties shall mutually agree upon and prepare the press release announcing the consummation of the transactions contemplated by this Agreement, which shall be issued concurrently with or promptly after the Closing. The SPAC, the Company and the Charterhouse Parties shall cooperate in good faith with respect to the preparation of, and, at least five (5) days prior to the Closing, the SPAC shall prepare a draft Form 8-K announcing the Closing, together with, or incorporating by reference, the required pro forma financial statements and the historical financial statements prepared by the Company and its accountant (the Completion 8-K). Concurrently with the Closing, or as soon as practicable (but in any event within four (4) Business Days) thereafter, the SPAC shall file the Completion 8-K with the SEC.
A-68
Table of Contents
Section 8.06. Public Announcements. The Parties agree to consult with each other before issuing any press release or making any public statement with respect to this Agreement or the other Transaction Agreements or the Transactions and, except for any press releases and public announcements the making of which may be required by Applicable Law or any listing agreement with any national securities exchange, will not issue any such press release or make any such public statement prior to such consultation except: (i) if such announcement or other communication is required by Applicable Law, in which case the disclosing Party shall, to the extent permitted by Applicable Law, first allow such other Parties to review such announcement or communication and have the opportunity to comment thereon and the disclosing Party shall consider such comments in good faith; (ii) to the extent such announcements or other communications contain only information previously disclosed in a public statement, press release or other communication previously approved in accordance with Section 8.05 or this Section 8.06; and (iii) announcements and communications to Governmental Authorities in connection with registrations, declarations and filings relating to the Transactions required to be made under this Agreement.
Section 8.07. Notification of Certain Matters. Each Party shall give prompt notice to the other Party of (a) any Action or investigation that would have been required to be disclosed under Section 4.12 if the Company had knowledge of it as of the date hereof or Section 5.10 if the SPAC had knowledge of it as of the date hereof; (b) the occurrence or non-occurrence of any event whose occurrence or non-occurrence, as the case may be, could reasonably be expected to cause any condition set forth in Section 11.02 or Section 11.03 not to be satisfied at any time from the date of this Agreement to the Closing; (c) any notice or other communication from any third Person alleging that the consent of such third Person is or may be required in connection with the Transactions; and (d) any regulatory notice, report or results of inspection from a Governmental Authority in respect of the Transactions.
Section 8.08. Exclusivity.
(a) During the Interim Period, no Seller shall sell, assign, transfer, redeem or repay (or enter into any Contract or otherwise become obligated to sell, assign, transfer, redeem or repay) any share or loan capital of the Company or any of its Subsidiaries, other than transfers to an Affiliate of such Seller who agrees to be bound by the terms and conditions of the Existing Company Articles and of this Agreement; provided, that, the execution, delivery and performance of this Agreement and the other Transaction Agreements and the consummation of the Transactions shall not be deemed a violation of this Section 8.08(a).
(b) During the Interim Period, neither the Company nor any Seller shall take, nor shall the Company or any Seller permit any of their respective Affiliates or representatives to take, whether directly or indirectly, any action to solicit, initiate or engage in discussions or negotiations with, or enter into any agreement with, or encourage, or provide information to, any Person (other than the SPAC and/or any of its Affiliates or representatives) concerning any purchase of any of the Companys equity securities or the issuance and sale of any securities of, or membership interests in, the Company or its Subsidiaries (other than any purchases of equity securities by the Company from employees of the Company or its Subsidiaries) or any merger or sale of substantial assets involving the Company or its Subsidiaries, other than immaterial assets or assets sold in the ordinary course of business (each such acquisition transaction, but excluding the Transactions, an Acquisition Transaction); provided, that, the execution, delivery and performance of this Agreement and the other Transaction Agreements and the consummation of the Transactions shall not be deemed a violation of this Section 8.08(b). The Company and each Seller shall, and shall cause their respective Affiliates and representatives to, immediately cease any and all existing discussions or negotiations with any Person conducted prior to the date hereof with respect to, or which is reasonably likely to give rise to or result in, an Acquisition Transaction.
(c) During the Interim Period, the SPAC shall not take, nor shall it permit any of its Affiliates or representatives to take, whether directly or indirectly, any action to solicit, initiate, continue or engage in discussions or negotiations with, or enter into any agreement with, or encourage, respond, provide information to or commence due diligence with respect to, any Person (other than the Company, its shareholders and/or any of their Affiliates or representatives), concerning, relating to or which is intended or is reasonably likely to give rise
A-69
Table of Contents
to or result in, any offer, inquiry, proposal or indication of interest, written or oral relating to any Business Combination (a Business Combination Proposal) other than with the Company, its shareholders and their respective Affiliates and representatives; provided, that, the execution, delivery and performance of this Agreement and the other Transaction Agreements and the consummation of the Transactions shall not be deemed a violation of this Section 8.08(c). The SPAC shall, and shall cause its Affiliates and representatives to, immediately cease any and all existing discussions or negotiations with any Person conducted prior to the date hereof with respect to, or which is reasonably likely to give rise to or result in, a Business Combination Proposal.
(d) Each Party shall promptly (and in no event later than twenty-four (24) hours after receipt of such inquiry, proposal, offer or submission) notify the other Parties if it or, to its knowledge, any of its or its representatives receives any inquiry, proposal, offer or submission with respect to an Acquisition Transaction or a Business Combination Proposal, as applicable (including the identity of the Person making such inquiry or submitting such proposal, offer or submission), after the execution and delivery of this Agreement. If either Party or its representatives receives an inquiry, proposal, offer or submission with respect to an Acquisition Transaction or a Business Combination Proposal, as applicable, such Party shall provide the other Parties with a copy of such inquiry, proposal, offer or submission as soon as reasonably practicable after receipt of such inquiry, proposal, offer or submission.
Section 8.09. Alternative Financing. In the event that any portion of the Debt Financing necessary for the consummation of the Business Combination becomes unavailable on the terms and conditions contemplated by the Debt Commitment Letter (or otherwise on terms and conditions acceptable to the Company and the SPAC (as contemplated by Section 6.10(a)), including the flex provisions) (other than (x) in the case of the obligations of the Company under this Section 8.09, as a result of (A) the SPACs failure to satisfy the conditions set forth in Section 11.03(a) or (B) the failure of the condition in paragraph 1 of the Conditions Exhibit to the Debt Commitment Letter (as in effect on the date hereof) which failure arises primarily as a result of a unilateral consent under, or a unilateral waiver of, this Agreement by the SPAC and (y) in the case of the obligations of the SPAC under this Section 8.09, as a result of the Company and the Sellers failure to satisfy the conditions set forth in Section 11.02(a)) (i) the Company shall promptly notify the SPAC thereof and (ii) each of the Company and the SPAC shall jointly use reasonable best efforts to (A) arrange and obtain any such portion from alternative sources (which may include existing Debt Financing Sources) (an Alternative Financing), on terms, taken as whole, that are not materially more adverse to the Company and its Subsidiaries (including after giving effect to the market flex provisions) or that are otherwise acceptable to the Company (in its sole discretion) as promptly as practicable following the occurrence of such event; provided that the terms of such Alternative Financing shall not (A) impose new or additional conditions precedent or expand upon the conditions precedent to the Debt Financing as set forth in the existing Debt Commitment Letter that could reasonably be expected to prevent or materially delay the Closing, (B) reduce the aggregate amount of available Debt Financing to less than the amount required to consummate the transactions contemplated by this Agreement (together with the other sources of financing contemplated hereby) or (C) otherwise reasonably be expected to materially delay or prevent the Closing. The Company and/or the SPAC shall deliver to the SPAC or the Company (as applicable) true, correct and complete copies of all agreements entered into in connection with any such Alternative Financing promptly following the execution thereof (the Alternative Financing Commitment Letter). Unless otherwise agreed by the Company in its sole discretion (at such time), notwithstanding anything to the contrary contained in this Agreement, in no event shall Company or its Affiliates be required to pay any fees or interest rates applicable to the Debt Financing that, taken as a whole, are materially in excess of those contemplated by the Debt Commitment Letters as in effect on the date hereof (including the market flex provisions).
ARTICLE 9
TAX MATTERS
Section 9.01. Transfer Taxes. All Transfer Taxes incurred in connection with the Transactions, including any real property transfer Tax and any similar Tax, shall be borne and paid by the SPAC, and the SPAC will, at
A-70
Table of Contents
its own expense, file all necessary Tax Returns and other documentation with respect to all such Taxes and fees, and, if required by Applicable Law, the Sellers will, and will cause their respective Affiliates to, join in the execution of any such Returns and other documentation.
Section 9.02. Tax Returns. The Company shall prepare, or cause to be prepared, and file, or cause to be filed, all Tax Returns for the Company and its Subsidiaries (including any Tax Returns of the Company and its Subsidiaries that relate to a Pre-Closing Tax Period and are required to be filed after the Closing); provided, that the Company shall use commercially reasonable efforts to engage a certified public accounting firm satisfactory to the SPAC to assist in the preparation and filing of any Tax Returns of the Company and/or its Subsidiaries in the United Kingdom for taxable periods that include the transactions contemplated by this Agreement. With respect to any Flow-Through Tax Return of the Company that relates to a Pre-Closing Tax Period (including, for the avoidance of doubt, a Straddle Period), (i) such Tax Return shall be prepared on a basis consistent with past practices, except as otherwise required by Applicable Law, (ii) the Company shall submit such Tax Return to the Sellers no later than thirty (30) days prior to the due date for the filing of any such Tax Return for review and (iii) the Company shall reflect any reasonable comments requested by the Sellers; provided such comments are provided no later than fifteen (15) days prior to the due date for the filing of any such Tax Return.
Section 9.03. Tax Contest. After the Closing, each Party shall promptly notify the other Parties in writing upon receipt by the applicable Party or its Affiliates of notice of any audit, examination, claim, investigation, litigation or other proceeding or Action (i) with respect to any Flow-Through Tax Return of the Company that relates to a Pre-Closing Tax Period (including, for the avoidance of doubt, a Straddle Period) or (ii) that could reasonably be expected to affect a Flow-Through Tax Return of the Company that relates to a Pre-Closing Tax Period (any such proceeding under (i) or (ii), a Tax Contest). The SPAC shall control any Tax Contest, provided that (a) the SPAC shall keep the Sellers reasonably informed of any material developments with respect to such Tax Contest, (b) the Sellers shall have the right to participate in any such Tax Contest at their own cost and expense and (iii) the SPAC shall not settle, resolve or abandon any such Tax Contest without the prior written consent of the Sellers (not to be unreasonably withheld, conditioned or delayed).
Section 9.04. Cooperation. Subject to the other provisions of this Section 9.04, the SPAC, the Company and the Sellers shall cooperate fully, as and to the extent reasonably requested, in connection with (i) the filing of Tax Returns and (ii) any audit, examination, claim, investigation, litigation or other proceeding or Action with respect to Taxes and Tax Returns. Such cooperation shall include the retention, and (upon the other Partys request) the provision, of records and information which are reasonably relevant to any such audit, litigation or other proceeding or Action and making employees available on a mutually convenient basis to provide additional information and explanation of any material provided hereunder; provided, that the Party requesting assistance shall pay the reasonable out-of-pocket expenses incurred by the Party providing such assistance.
Section 9.05. Straddle Periods. For purposes of this Agreement, in the case of any Taxes that are payable for a Straddle Period, (i) real property and personal property Taxes shall be apportioned on a per diem basis and (ii) all other Taxes shall be apportioned based on an interim closing of the books.
Section 9.06. Post-Closing Actions. After the Closing, without the prior written consent of the Sellers (which consent shall not be unreasonably withheld, conditioned or delayed), the SPAC shall not (and shall neither cause nor permit the Company and its Subsidiaries to) take any of the following actions except as required by Applicable Law: (i) file (except in accordance with Section 9.02 or Section 9.09), amend, re-file or otherwise modify any Flow-Through Tax Return of the Company or any of its Subsidiaries that relates to any Pre-Closing Tax Period, (ii) make, change or revoke any Tax election affecting a Flow-Through Tax Return of the Company or any of its Subsidiaries that relates to any Pre-Closing Tax Period (except in accordance with Section 9.09) or (iii) initiate any discussion, voluntary disclosure or examination with any Governmental Authority regarding any Flow-Through Tax Returns of the Company or any of its Subsidiaries that relates to any Pre-Closing Tax Period.
A-71
Table of Contents
Section 9.07. Election. The Parties agree and shall cause the Company and each Subsidiary of the Company that is classified as a partnership for U.S. federal income tax purposes to have in effect for the tax period that includes the Closing Date a valid election pursuant to Section 754 of the Code.
Section 9.08. Withholding. Notwithstanding anything to the contrary in this Agreement or any Transaction Agreement, each Party shall be entitled to deduct and withhold from the consideration otherwise payable to any Person pursuant to this Agreement or any Transaction Agreement such amounts as it is required to deduct and withhold with respect to the making of such payment under any provision of Applicable Law. If any Party so withholds amounts, such amounts shall be treated for all purposes of this Agreement as having been paid to the Person in respect of which such Party made such deduction and withholding to the extent such amounts are properly paid over to the appropriate Governmental Authority, and such Party shall furnish to such Person within ten (10) Business Days of such payment the original or certificated copy of a receipt issued by such Governmental Authority evidencing such payment. In the event that one Party determines that any portion of a payment under this Agreement or any Transaction Agreement would be subject to withholding under Applicable Law, such Party shall promptly notify the other Parties of such determination but in no event later than ten (10) days prior to the date on which such payment is due. The Parties shall reasonably cooperate to eliminate or minimize any such withholding.
Section 9.09. Partnership Audits. Notwithstanding anything herein to the contrary, the Company shall make a push out election under Section 6226 of the Code (and any corresponding provisions of Applicable Law) with respect to any imputed underpayment (within the meaning of Section 6225 of the Code) or any interest or penalty related thereto relating to any Pre-Closing Tax Period and the Parties hereby agree to reasonably cooperate (or cause their Affiliates to cooperate) with any and all actions necessary to facilitate any such election.
ARTICLE 10
EMPLOYEE BENEFITS
Section 10.01. Equity Incentive Plan. Prior to the SPAC Special Meeting, the SPAC shall adopt an equity incentive plan substantially in the form attached hereto as Exhibit F (the Equity Incentive Plan), that provides for grant of awards to employees and other service providers of the SPAC, with the maximum number of New SPAC Class A Common Shares available for issuance under the Equity Incentive Plan not exceeding in the aggregate ten percent (10%) ) of the New SPAC Class A Common Shares outstanding immediately following the Closing. The total number of New SPAC Class A Common Shares available for issuance under the Equity Incentive Plan shall be increased on the first day of each fiscal year following the date on which the Equity Incentive Plan is adopted in an amount equal to the least of (i) three percent (3%) of the outstanding New SPAC Class A Common Shares on the last day of the immediately preceding fiscal year, (ii) five percent (5%) of the New SPAC Class A Common Shares immediately following the Closing and (iii) such number of New SPAC Class A Common Shares as determined by the Committee (as defined and designated under the terms of the Equity Incentive Plan) in its discretion. The Equity Incentive Plan shall be subject to approval of the Existing SPAC Shareholders and shall become effective upon adoption and such approval.
Section 10.02. [Reserved.]
Section 10.03. 280G Approval. To the extent that any disqualified individual (within the meaning of Section 280G(c) of the Code and the regulations thereunder) has the right to receive any payments or benefits that could be deemed to constitute parachute payments (within the meaning of Section 280G(b)(2)(A) of the Code and the regulations thereunder), then, the Company will: (a) at least four (4) Business Days prior to the Closing Date, solicit and use its reasonable best efforts to obtain from each such disqualified individual a waiver of such disqualified individuals rights to some or all of such payments or benefits (the Waived 280G Benefits) so that any remaining payments and/or benefits shall not be deemed to be excess parachute payments (within the meaning of Section 280G of the Code and the regulations thereunder); and (b) at least one
A-72
Table of Contents
(1) Business Day prior to the Closing Date, with respect to each individual who agrees to the waiver described in clause (a), submit to a vote of holders of the equity interests of the Company entitled to vote on such matters, in the manner required under Section 280G(b)(5) of the Code and the regulations promulgated thereunder, along with adequate disclosure intended to satisfy such requirements (including Q&A 7 of Section 1.280G-1 of such regulations), the right of any such disqualified individual to receive the Waived 280G Benefits. At least five (5) days prior to soliciting such waivers and approval, the Company shall provide drafts of such waivers and approval materials and the calculations and related back-up documentation to the SPAC for its review and comment, and the Company shall incorporate changes reasonably requested by the SPAC. To the extent applicable, prior to the Closing Date, the Company shall deliver to the SPAC evidence that a vote of the stockholders of the Company was solicited in accordance with the foregoing and whether the requisite number of votes of the stockholders of the Company was obtained with respect to the Waived 280G Benefits or that the vote did not pass and the Waived 280G Benefits will not be paid or retained.
ARTICLE 11
CONDITIONS TO CLOSING
Section 11.01. Conditions to Obligations of All Parties. The obligations of the Parties to consummate, or cause to be consummated, the Closing are subject to the satisfaction of the following conditions (one or more of which may be waived, in accordance with Section 13.04, if legally permitted, in writing by each of the SPAC, the Company and the Charterhouse Parties):
(a) No provision of Applicable Law, and no judgment, injunction, order or decree of any applicable Governmental Authority, shall prohibit the consummation of the Closing.
(b) Any applicable waiting period under the HSR Act relating to the Transactions (and any extensions thereof or any timing agreements, understandings or commitments obtained by request or other action of the United States Federal Trade Commission or the Antitrust Division of the United States Department of Justice, as applicable) shall have expired or been terminated.
(c) The filings and notifications listed on Section 11.01(c) of the Company Disclosure Schedule shall have been made with, and all approvals required in connection therewith shall have been received from, the applicable Governmental Authorities.
(d) The Registration Statement shall have become effective in accordance with the Securities Act, no stop order shall have been issued by the SEC with respect to the Registration Statement and no Action seeking such stop order shall have been threatened or initiated.
(e) The Requisite Existing SPAC Stockholders shall have duly approved the Transaction Proposals in accordance with Applicable Law, the Existing SPAC Certificate of Incorporation, the Existing SPAC Bylaws and the rules and regulations of the NYSE.
(f) The SPAC shall have at least $5,000,001 of net tangible assets (as determined in accordance with Rule 3a51-1(g)(1) of the Exchange Act) remaining after the SPAC Stockholder Redemptions.
Section 11.02. Additional Conditions to Obligation of the SPAC. The obligation of the SPAC to consummate, or cause to be consummated, the Closing is subject to the satisfaction of the following further conditions (one or more of which may be waived in writing by the SPAC):
(a) The Company and the Sellers shall have performed in all material respects all of their respective obligations hereunder required to be performed by such Person on or prior to the Closing Date.
A-73
Table of Contents
(b) (i) The representations and warranties of the Company contained in Section 4.09(b) (Absence of Certain Changes) of this Agreement shall be true and correct in all respects as of the date of this Agreement and as of the Closing Date, as if made at and as of the Closing Date; (ii) the representations and warranties of the Company contained in Section 4.01 (Existence and Power), Section 4.02 (Authorization), Section 4.05 (Capitalization), and Section 4.22 (Finders Fees) shall be true and correct in all but de minimis respects (without giving effect to any limitations as to materiality or Material Adverse Effect or any similar limitation set forth therein) as of the date of this Agreement and as of the Closing Date, as if made at and as of the Closing Date (except to the extent such representations and warranties expressly relate to an earlier date, and in such case, shall be so true and correct in all but de minimis respects at and as of such earlier date); and (iii) each other representation and warranty of the Company contained in Article 4 shall be true and correct (without giving effect to any limitations as to materiality or Material Adverse Effect or any similar limitation set forth therein) as of the date of this Agreement and as of the Closing Date, as if made at and as of the Closing Date (except to the extent such representations and warranties expressly relate to an earlier date, and in such case, shall be true and correct at and as of such earlier date), except, in the case of this clause (iii), where the failure to be so true and correct, individually or in the aggregate, has not had, and would not reasonably be expected to have, a Material Adverse Effect.
(c) (i) The representations and warranties of the Sellers contained in Article 3 shall be true and correct (without giving effect to any limitations as to materiality or Material Adverse Effect or any similar limitation set forth therein) as of the date of this Agreement and as of the Closing Date, as if made at and as of such date (except to the extent such representations and warranties expressly relate to an earlier date, and in such case, shall be true and correct at and as of such earlier date), except where the failure to be so true and correct, individually or in the aggregate, has not had, and would not reasonably be expected to have, a Seller Material Adverse Effect.
(d) The SPAC shall have received a certificate signed by an officer of the Company, dated the Closing Date, certifying that the conditions specified in Section 11.02(a), Section 11.02(b) and Section 11.02(c) have been fulfilled.
(e) A majority of the holders of Existing Company Shares shall have delivered to the Called Shareholders the Drag Along Notice.
(f) No Material Adverse Effect shall have occurred since the date of this Agreement.
(g) The Company and its Subsidiaries shall have issued notice of suspension or termination of any contracts to the relevant HRPs and otherwise ceased doing business with the relevant HRP as requested in writing by the SPAC; provided, that with respect to each HRP for which the SPAC has made such a request, the SPAC shall have provided the Company with (i) written notice at least five (5) Business Days prior to the Closing Date and (ii) the reason(s) for which the SPAC has made such a request (which may include an inability to obtain sufficient information about the relevant HRP).
Section 11.03. Additional Conditions to Obligation of the Company and the Sellers. The obligation of the Company and the Sellers to consummate, or cause to be consummated, the Closing is subject to the satisfaction of the following further conditions (one or more of which may be waived, in accordance with Section 13.04, in writing by the Company and the Charterhouse Parties):
(a) The SPAC shall have performed in all material respects all of its obligations hereunder required to be performed by it on or prior to the Closing Date.
(b) (i) The representations and warranties of the SPAC contained in Section 5.09(b) of this Agreement shall be true and correct in all respects as of the Closing Date, as if made at and as of such date; (ii) the representations and warranties of the SPAC contained in Section 5.01, Section 5.02, Section 5.05, and Section 5.17 shall be true and correct (without giving effect to any limitations as to materiality or material adverse effect or any similar
A-74
Table of Contents
limitation set forth therein) in all but de minimis respects as of the Closing Date, as if made at and as of such date (except to the extent such representations and warranties expressly relate to an earlier date, and in such case, shall be so true and correct in all but de minimis respects at and as of such earlier date); and (iii) each other representation and warranty of the SPAC contained in Article 5 shall be true and correct (without giving effect to any limitations as to materiality or material adverse effect or any similar limitation set forth therein) as of the Closing Date, as if made at and as of such date (except to the extent such representations and warranties expressly relate to an earlier date, and in such case, shall be true and correct at and as of such earlier date), except, in the case of this clause (iii), where the failure to be so true and correct has not had, and would not reasonably be expected to have, a SPAC Material Adverse Effect.
(c) The Company shall have received a certificate signed by an officer of the SPAC, dated the Closing Date, certifying that the conditions specified in Section 11.03(a) and Section 11.03(b) have been fulfilled.
(d) The Available Closing Cash shall not be less than $1,310,000,000 (the Minimum Cash Condition).
(e) Since the date of this Agreement, there has been no development, effect, change, circumstance, event or occurrence, that, individually or in the aggregate, has had, or would reasonably be expected to have a SPAC Material Adverse Effect.
ARTICLE 12
TERMINATION
Section 12.01. Grounds for Termination. This Agreement may be terminated at any time prior to the Closing:
(a) by mutual written agreement of the SPAC, the Company and the Charterhouse Parties;
(b) by the SPAC, on the one hand, and the Company and the Charterhouse Parties, on the other hand, if the Closing shall not have been consummated on or before November 30, 2021 (as may be extended hereunder, or by mutual agreement of the parties, the End Date); provided, however, that if the conditions specified in Sections 11.01(b) and 11.01(c) have not been satisfied by November 30, 2021, either the SPAC, on the one hand, or the Company and the Charterhouse Parties, on the other hand, may by written notice to the other party, extend the End Date to January 31, 2022; provided, further however, that if, following such extension of the End Date, the conditions specified in Sections 11.01(b) and 11.01(c) have not been satisfied by January 31, 2022 because of a failure to receive the specified approval or approvals noted on Section 11.01(c) of the Company Disclosure Schedule, either the SPAC, on the one hand, or the Company and the Charterhouse Parties, on the other hand, may by written notice to the other party, extend the End Date to March 31, 2022;
(c) by written notice from either the Company and the Charterhouse Parties, on the one hand, or the SPAC, on the other hand, to the other(s) if consummation of the transactions contemplated hereby would violate any nonappealable final order, decree or judgment of any court or Governmental Authority having competent jurisdiction;
(d) by written notice to the Company and the Charterhouse Parties from the SPAC if there is any breach of any representation, warranty or covenant on the part of the Company or the Sellers set forth in this Agreement, such that the conditions specified in Sections 11.02(a), 11.02(b) and 11.02(c) would not be satisfied at the Closing (a Terminating Company Breach), except that, if such Terminating Company Breach is curable by the Company through the exercise of its commercially reasonable efforts, then, for a period of up to thirty (30) days (or any shorter period of the time that remains between the date the SPAC provides written notice of such breach and the End Date (as may be extended pursuant to Section 12.01(b))) after receipt by the Company of notice from the SPAC of such breach, but only as long as the Company continues to use its commercially
A-75
Table of Contents
reasonable efforts to cure such Terminating Company Breach (the Company Cure Period), such termination shall not be effective, and such termination shall become effective only if the Terminating Company Breach is not cured within the Company Cure Period; provided that the SPACs right to terminate this Agreement under clause (b) of this Section 12.01 or this clause (d) shall not be available if the SPACs failure to fulfill any obligation under this Agreement has been the primary cause of, or primarily resulted in, the failure of the Closing to occur on or before such date;
(e) by written notice to the SPAC from the Company and the Charterhouse Parties if there is any breach of any representation, warranty or covenant on the part of the SPAC set forth in this Agreement, such that the conditions specified in Sections 11.03(a) and 11.03(b) would not be satisfied at the Closing (a Terminating SPAC Breach), except that, if such Terminating SPAC Breach is curable by the SPAC through the exercise of its commercially reasonable efforts, then, for a period of up to thirty (30) days (or any shorter period of the time that remains between the date the Company provides written notice of such breach and the End Date (as may be extended pursuant to Section 12.01(b)), as applicable) after receipt by the SPAC of notice from the Company of such breach, but only as long as the SPAC continues to use its commercially reasonable efforts to cure such Terminating SPAC Breach (the SPAC Cure Period), such termination shall not be effective, and such termination shall become effective only if the Terminating SPAC Breach is not cured within the SPAC Cure Period; provided that the right of the Company and the Charterhouse Parties to terminate this Agreement under clause (b) of this Section 12.01 or this clause (e) shall not be available if the Companys failure to fulfill any obligation under this Agreement has been the primary cause of, or primarily resulted in, the failure of the Closing to occur on or before such date;
(f) by written notice from either the Company and the Charterhouse Parties, on the one hand, or the SPAC, on the other hand, to the other(s) if the approval of the Transaction Proposals by the Requisite Existing SPAC Stockholders is not obtained at the SPAC Special Meeting (subject to any adjournment, postponement or recess of the meeting); provided that the right to terminate this Agreement under this clause (f) shall not be available to the SPAC if, at the time of such termination, the SPAC is in breach of Section 8.04; or
(g) by written notice from either the Company and the Charterhouse Parties, on the one hand, or the SPAC, on the other hand, to the other(s) if the Minimum Cash Condition is incapable of being satisfied and the Company and the Charterhouse Parties have not waived the Minimum Cash Condition within fifteen (15) Business Days of receipt by the Company and the Charterhouse Parties of a written notice from the SPAC that the Minimum Cash Condition has not been satisfied and is incapable of being satisfied.
Section 12.02. Effect of Termination. If this Agreement is terminated as permitted by Section 12.01, this Agreement shall forthwith become void and have no effect, without any liability on the part of any Party or its respective Affiliates, shareholders, stockholders, directors, officers, employees, agents, consultants or representatives to the other Parties to this Agreement; provided that if such termination shall result from (i) Fraud, (ii) the knowing and willful failure of a Party to fulfill a condition to the performance of the obligations of the other Party, (iii) the knowing and willful failure of a Party to perform a covenant of this Agreement or (iv) the knowing and willful breach by a Party hereto of any representation or warranty or agreement contained herein, such Party shall, subject to the terms of this Agreement, be fully liable for any and all losses, damages, claims, costs or expenses incurred or suffered by the other Parties as a result of such failure or breach. The provisions of Sections 6.03, this Section 12.02 and Article 13 shall survive any termination hereof pursuant to Section 12.01.
ARTICLE 13
MISCELLANEOUS
Section 13.01. Non-Survival of Representations, Warranties and Covenants. None of the representations, warranties, covenants and agreements in this Agreement or in any instrument, document or certificate delivered
A-76
Table of Contents
pursuant to this Agreement shall survive the Closing, except for (i) those covenants and agreements contained herein and therein which by their terms expressly apply in whole or in part after the Closing and then only to such extent until such covenants and agreements have been fully performed, and (ii) the provisions of this Article 13. Nothing in this Section 13.01 shall limit or prohibit the rights of the SPAC to pursue recoveries under the R&W Insurance Policy or any other representation and warranty insurance policy.
Section 13.02. Non-Recourse. This Agreement may only be enforced against, and any claim or cause of action based upon, arising out of, or related to this Agreement or the transactions contemplated hereby may only be brought against, the entities that are expressly named as Parties and then only with respect to the specific obligations set forth herein with respect to such Party. Except to the extent a Party (and then only to the extent of the specific obligations undertaken by such Party in this Agreement), (i) no past, present or future director, officer, employee, incorporator, member, partner, stockholder, Affiliate, agent, attorney, advisor or representative of any Party and (ii) no past, present or future director, officer, employee, incorporator, member, partner, stockholder, Affiliate, agent, attorney, advisor or representative of any of the foregoing shall have any liability (whether in contract, tort, equity or otherwise) for any one or more of the representations, warranties, covenants, agreements or other obligations or liabilities of any one or more of the Company, the SPAC or the Sellers under this Agreement of or for any claim based on, arising out of, or related to this Agreement or the transactions contemplated hereby.
Section 13.03. Notices. All notices, requests and other communications to any party hereunder shall be in writing (including electronic mail (e-mail) transmission, so long as a receipt of such e-mail is requested and received) and shall be given,
if to the SPAC, to:
GS Acquisition Holdings Corp II
200 West Street
New York, New York 10282
Attention: Thomas R. Knott, David S. Plutzer
E-mail: tom.knott@gs.com; david.plutzer@gs.com
with a copy (which copy shall not constitute notice) to:
Weil, Gotshal & Manges LLP
767 Fifth Avenue
New York, New York 10153
Attention: Michael J. Aiello, Brian Parness
E-mail: michael.aiello@weil.com; brian.parness@weil.com
if to the Company or any Seller other than Charterhouse Capital Partners LLP, to:
Mirion Technologies (TopCo), Ltd.
22 Grenville Street, St. Helier
Jersey JE4 8PX, Channel Islands
Attention: Thomas D. Logan, Emmanuelle Lee, Mirion Legal
E-mail: tlogan@mirion.com; elee@mirion.com; legal@mirion.com
with a copy (which copy shall not constitute notice) to:
Davis Polk & Wardwell LLP
1600 El Camino Real Ste. 100
Menlo Park, California 94025
Attention: Alan F. Denenberg, Stephen Salmon
E-mail: alan.denenberg@davispolk.com; stephen.salmon@davispolk.com
A-77
Table of Contents
if to the Charterhouse Parties, to:
The Charterhouse Parties
6th Floor, Belgrave House, 76 Buckingham Palace Road
London, SW1W 9TQ, United Kingdom
Attention: Christopher Warren, Thomas Patrick
E-mail: chris.warren@charterhouse.co.uk;
tom.patrick@charterhouse.co.uk
with a copy (which copy shall not constitute notice) to:
Freshfields Bruckhaus Deringer LLP
601 Lexington Avenue, 31st Floor
New York, New York 10019
Attention: Valerie Ford Jacob
E-mail: valerie.jacob@freshfields.com
Freshfields Bruckhaus Deringer LLP
9 avenue de Messine
75008 Paris, France
Attention: Yann Gozal
E-mail: yann.gozal@freshfields.com
Freshfields Bruckhaus Deringer LLP
100 Bishopsgate
London EC2P 2SR, United Kingdom
Attention: Charles Hayes
E-mail: charles.hayes@freshfields.com
or such other address or e-mail address as such party may hereafter specify for the purpose by notice to the other Parties hereto. All such notices, requests and other communications shall be deemed received on the date of receipt by the recipient thereof if received prior to 5:00 p.m. in the place of receipt and such day is a Business Day. Otherwise, any such notice, request or communication shall be deemed not to have been received until the next succeeding Business Day.
Section 13.04. Amendments and Waivers. (a) Any provision of this Agreement may be amended or waived if, but only if, such amendment or waiver is in writing and is signed, in the case of an amendment, by each of the Company, the SPAC and the Charterhouse Parties, on behalf of the Sellers, or in the case of a waiver, by (i) the SPAC, if the SPAC is the Party against whom the waiver is to be effective, (ii) the Company and the Charterhouse Parties, if the Company is the Party against whom the waiver is to be effective, or (iii) the Charterhouse Parties, if any or all of the Sellers are the Parties against whom the waiver is to be effective.
(a) No failure or delay by any Party in exercising any right, power or privilege hereunder shall operate as a waiver thereof nor shall any single or partial exercise thereof preclude any other or further exercise thereof or the exercise of any other right, power or privilege. The rights and remedies herein provided shall be cumulative and not exclusive of any rights or remedies provided by law.
Section 13.05. Expenses. Except as otherwise provided herein, all costs and expenses incurred in connection with this Agreement shall be paid by the Party incurring such cost or expense. For the avoidance of doubt, if this Agreement is terminated in accordance with Article 12, all costs and expenses incurred in connection with this Agreement shall be paid by the Party incurring such cost or expense.
Section 13.06. Successors and Assigns. The provisions of this Agreement shall be binding upon and inure to the benefit of the Parties hereto and their respective successors and assigns; provided that no Party may assign,
A-78
Table of Contents
delegate or otherwise transfer any of its rights or obligations under this Agreement without the consent of each other Party hereto, except that the SPAC may assign, delegate or otherwise transfer any of its rights or obligations under this Agreement, without the consent of any other Party hereto, to any insurer that underwrites an R&W Insurance Policy, or the agent of any such insurer, in the event of Fraud.
Section 13.07. Governing Law. This Agreement shall be governed by and construed in accordance with the law of the State of Delaware, without regard to the conflicts of law rules of such state.
Section 13.08. Jurisdiction. The Parties hereto agree that any suit, Action or proceeding seeking to enforce any provision of, or based on any matter arising out of or in connection with, this Agreement or the transactions contemplated hereby shall be brought in the Court of Chancery of the State of Delaware or, if such court does not have jurisdiction, to the Superior Court of the State of Delaware or, if jurisdiction is vested exclusively in federal courts of the United States, the federal courts of the United States sitting in the State of Delaware, so long as one of such courts shall have subject matter jurisdiction over such suit, Action or proceeding, and that any cause of action arising out of this Agreement shall be deemed to have arisen from a transaction of business in the State of Delaware, and each of the Parties hereby irrevocably consents to the jurisdiction of such courts (and of the appropriate appellate courts therefrom) in any such suit, Action or proceeding and irrevocably waives, to the fullest extent permitted by law, any objection that it may now or hereafter have to the laying of the venue of any such suit, Action or proceeding in any such court or that any such suit, Action or proceeding brought in any such court has been brought in an inconvenient forum. Process in any such suit, Action or proceeding may be served on any Party anywhere in the world, whether within or without the jurisdiction of any such court. Without limiting the foregoing, each Party agrees that service of process on such Party as provided in Section 13.01 shall be deemed effective service of process on such Party.
Section 13.09. WAIVER OF JURY TRIAL. EACH OF THE PARTIES HERETO HEREBY IRREVOCABLY WAIVES ANY AND ALL RIGHT TO TRIAL BY JURY IN ANY ACTION OR LEGAL PROCEEDING ARISING OUT OF OR RELATED TO THIS AGREEMENT OR THE TRANSACTIONS CONTEMPLATED HEREBY.
Section 13.10. Counterparts; Effectiveness; Third Party Beneficiaries. This Agreement may be signed in any number of counterparts, each of which shall be an original, with the same effect as if the signatures thereto and hereto were upon the same instrument. This Agreement shall become effective when each Party hereto shall have received a counterpart hereof signed by the other Party hereto. Until and unless each party has received a counterpart hereof signed by the other Parties hereto, this Agreement shall have no effect and no Party shall have any right or obligation hereunder (whether by virtue of any other oral or written agreement or other communication). Counterparts may be delivered via facsimile, electronic mail (including .pdf or any electronic signature complying with the U.S. federal ESIGN Act of 2000 (e.g., www.docusign.com)) or other transmission method and any counterpart so delivered shall be deemed to have been duly and validly delivered and be valid and effective as delivery of a manually executed counterpart of this Agreement. Each of the Parties hereto represents that it has undertaken commercially reasonable steps to verify the identity of each individual person executing any such counterparts via electronic signature on behalf of such Party and has and will maintain sufficient records of the same. No provision of this Agreement is intended to confer any rights, benefits, remedies, obligations, or liabilities hereunder upon any Person other than the Parties and their respective successors and assigns, except that (i) each director and officer of the Company and its Subsidiaries as of the date hereof shall be a third-party beneficiary of Section 8.03 and shall have the right to enforce such provision directly to the extent he or she may deem such enforcement necessary or advisable to protect his or her rights thereunder and (ii) the Debt Financing Sources are express third party beneficiaries of, and may enforce, the survival provisions set forth in Section 12.02, and any provision of this Section 13.10 and Section 13.15.
Section 13.11. Entire Agreement. This Agreement and the other Transaction Agreements constitute the entire agreement among the Parties with respect to the subject matter hereof and supersede all prior agreements and understandings, both oral and written, among the Parties with respect to the subject matter hereof.
A-79
Table of Contents
Section 13.12. Severability. If any term, provision, covenant or restriction of this Agreement is held by a court of competent jurisdiction or other authority to be invalid, void or unenforceable, the remainder of the terms, provisions, covenants and restrictions of this Agreement shall remain in full force and effect and shall in no way be affected, impaired or invalidated so long as the economic or legal substance of the transactions contemplated hereby is not affected in any manner materially adverse to any Party. Upon such a determination, the Parties shall negotiate in good faith to modify this Agreement so as to effect the original intent of the Parties as closely as possible in an acceptable manner in order that the transactions contemplated hereby be consummated as originally contemplated to the fullest extent possible.
Section 13.13. Disclosure Schedules. The Sellers, the Company and the SPAC have set forth information on Schedules in a section thereof that corresponds to the section of this Agreement to which it relates. A matter set forth in one section of a Schedule need not be set forth in any other section so long as its relevance to such other section of the Schedule or section of this Agreement is reasonably apparent on the face of the information disclosed therein to the Person to which such disclosure is being made. The Parties acknowledge and agree that (a) the Schedules to this Agreement may include certain items and information not required to be set forth therein, provided solely for informational purposes for the convenience of the Company, the Sellers and the SPAC, as applicable; (b) the disclosure by the Sellers, the Company or the SPAC of any matter in the Schedules shall not be deemed to constitute an acknowledgment by the Sellers, the Company or the SPAC, as applicable, that the matter is required to be disclosed by the terms of this Agreement or that the matter is material; and (c) no disclosure in the Schedules relating to any possible breach or violation of any agreement, law or regulation shall be construed as an admission or indication that any such breach or violation exists or has actually occurred, and no disclosure in the Schedules shall be construed as an admission against interest by the Company, any Seller or the SPAC, as applicable, to any third party regarding any matter whatsoever, including that any agreement or document is enforceable or currently in effect or that there are any obligations remaining to be performed or any rights that may be exercised under such agreement or document.
Section 13.14. Specific Performance. The Parties hereto agree that irreparable damage would occur if any provision of this Agreement were not performed in accordance with the terms hereof and that the Parties shall be entitled to an injunction or injunctions to prevent breaches of this Agreement or to enforce specifically the performance of the terms and provisions hereof, in addition to any other remedy to which they are entitled at law or in equity. The Parties hereby further acknowledge and agree that prior to Closing, the Sellers and the Company shall be entitled to seek specific performance to enforce specifically the terms and provisions of, and to prevent or cure breaches of, Section 7.09 (Trust Account), including by compelling the SPAC to enforce its rights under the Trust Agreement through the commencement of litigation and other legal actions against the counterparties of the Trustee.
Section 13.15. Debt Financing Sources. Notwithstanding anything in this Agreement to the contrary, each of the Parties hereby:
(a) agrees that any proceeding, whether in law or in equity, whether in contract or in tort or otherwise, involving the Debt Financing Sources, arising out of or relating to, this Agreement or any of the transactions contemplated hereby, shall be subject to the exclusive jurisdiction of any federal or state court in the Borough of Manhattan, New York, New York, so long as such forum is and remains available, and any appellate court thereof and each party hereto irrevocably submits itself and its property with respect to any such proceeding to the exclusive jurisdiction of such court,
(b) agrees that any such proceeding shall be governed by the laws of the State of New York (without giving effect to any conflicts of law principles that would result in the application of the laws of another state), except as otherwise provided in the applicable documentation relating to the Debt Financing,
(c) agrees not to bring or support any proceeding of any kind or description, whether in law or in equity, whether in contract or in tort or otherwise, against any Debt Financing Source in any way arising out of or
A-80
Table of Contents
relating to, this Agreement or any of the transactions contemplated hereby in any forum other than any federal or state court in the Borough of Manhattan, New York, New York,
(d) agrees that service of process in any such legal proceeding or proceeding shall be effective if notice is given in accordance with Section 13.03,
(e) irrevocably waives, to the fullest extent that it may effectively do so, the defense of an inconvenient forum to the maintenance of such proceeding in any such court,
(f) agrees that a final judgment in any such proceeding shall be conclusive and may be enforced in other jurisdictions by suit on the judgment or in any other manner provided by law,
(g) knowingly, intentionally and voluntarily waives to the fullest extent permitted by applicable law trial by jury in any proceeding brought against any Debt Financing Source in any way arising out of or relating to this Agreement or any of the transactions contemplated hereby,
(h) agrees that none of the Debt Financing Sources shall have any liability to any of the Parties relating to or arising out of this Agreement or any of the transactions contemplated hereby (excluding the Debt Commitment Letter), whether in law or in equity, whether in contract or in tort or otherwise (and each of the Parties and each of their respective Affiliates and each of their and their respective Affiliates respective directors, officers, employees, agents, managers, consultants, advisors and other representatives, including legal counsel, accountants and financial advisors, hereby acknowledges that they have no recourse against, and hereby waive any rights or claims against, the Debt Financing Sources in connection therewith); provided that nothing in this Agreement shall limit the liability of the Debt Financing Sources (or the rights and remedies of the DCL Beneficiary and/or its applicable Affiliates) pursuant to the documentation related to the Debt Financing, including the Debt Commitment Letter, and
(i) agrees that the Debt Financing Sources are express third party beneficiaries of, and may enforce, the survival provisions set forth in Section 12.02, and any provision of Section 13.10 and this Section 13.15 and that such provisions and the definition of Debt Financing Sources shall not be amended in any way adverse to any Debt Financing Source without the prior written consent of the Debt Financing Sources party to the Debt Commitment Letter.
A-81
Table of Contents
IN WITNESS WHEREOF, the parties hereto have caused this Agreement to be duly executed by their respective authorized officers as of the day and year first above written.
| GS ACQUISITION HOLDINGS CORP II | ||
| By: | /s/ Thomas R. Knott | |
| Name: Thomas R. Knott | ||
| Title: Authorized Signatory | ||
| MIRION TECHNOLOGIES (TOPCO), LTD. | ||
| By: | /s/ Christopher Warren | |
| Name: Christopher Warren | ||
| Title: Authorized Signatory | ||
| CCP IX LP NO. 1, acting by its General Partner, CHARTERHOUSE GENERAL PARTNERS (IX) LIMITED (for the limited purpose set forth herein) |
||
| By: | /s/ Thomas Patrick | |
| Name: Thomas Patrick | ||
| Title: Authorized Signatory | ||
| CCP IX LP NO. 2, acting by its General Partner, CHARTERHOUSE GENERAL PARTNERS (IX) LIMITED (for the limited purpose set forth herein) |
||
| By: | /s/ Thomas Patrick | |
| Name: Thomas Patrick | ||
| Title: Authorized Signatory | ||
| CCP IX CO-INVESTMENT LP, acting by its General Partner, CHARTERHOUSE GENERAL PARTNERS (IX) LIMITED (for the limited purpose set forth herein) |
||
| By: | /s/ Thomas Patrick | |
| Name: Thomas Patrick | ||
| Title: Authorized Signatory | ||
| CCP IX CO-INVESTMENT NO. 2 LP, acting by its General Partner, CHARTERHOUSE GENERAL PARTNERS (IX) LIMITED (for the limited purpose set forth herein) |
||
| By: | /s/ Thomas Patrick | |
| Name: Thomas Patrick | ||
| Title: Authorized Signatory | ||
Table of Contents
| THOMAS D. LOGAN (for the limited purpose set forth herein) |
||
| /s/ THOMAS D. LOGAN | ||
| EMMANUELLE LEE (for the limited purpose set forth herein) |
||
| /s/ EMMANUELLE LEE | ||
| BRIAN SCHOPFER (for the limited purpose set forth herein) |
||
| /s/ BRIAN SCHOPFER | ||
| MICHAEL FREED (for the limited purpose set forth herein) |
||
| /s/ MICHAEL FREED | ||
| MICHAEL BRUMBAUGH (for the limited purpose set forth herein) |
||
| /s/ MICHAEL BRUMBAUGH | ||
| IAIN WILSON (for the limited purpose set forth herein) |
||
| /s/ IAIN WILSON | ||
| SHELIA RICHARDSON (for the limited purpose set forth herein) |
||
| /s/ SHELIA RICHARDSON | ||
| BRUNO MOREL (for the limited purpose set forth herein) |
||
| /s/ BRUNO MOREL | ||
| LOUIS BIACCHI (for the limited purpose set forth herein) |
||
| /s/ LOUIS BIACCHI | ||
| JAMES COCKS (for the limited purpose set forth herein) |
||
| /s/ JAMES COCKS | ||
Table of Contents
| BERTRAND DUBAN (for the limited purpose set forth herein) |
||
| /s/ BERTRAND DUBAN | ||
| LOIC ELOY (for the limited purpose set forth herein) |
||
| /s/ LOIC ELOY | ||
| J.P. MORGAN TRUST COMPANY OF DELAWARE in its capacity as Trustee of the ALISON PAIGE LOGAN GST EXEMPT TRUST (for the limited purpose set forth herein) |
||
| /s/ Mark A. Kleinman | ||
| J.P. MORGAN TRUST COMPANY OF DELAWARE in its capacity as Trustee of the THOMAS DARRELL LOGAN, JR. GST EXEMPT TRUST (for the limited purpose set forth herein) |
||
| /s/ Mark A. Kleinman | ||
| J.P. MORGAN TRUST COMPANY OF DELAWARE in its capacity as Trustee of the MARY HANCOCK LOGAN GST EXEMPT TRUST (for the limited purpose set forth herein) |
||
| /s/ Mark A. Kleinman | ||
| /s/ Emmanuelle Lee |
| Emmanuelle Lee |
| /s/ Gregory C. Lee |
| Gregory C. Lee |
| both in their capacity as Trustee of the LEE REVOCABLE LIVING TRUST |
| Date: ______________________ 2021 |
| Location: San Ramon, California |
Table of Contents
Final Form
FORM OF CERTIFICATE OF INCORPORATION
OF
MIRION TECHNOLOGIES, INC.
ARTICLE 1
NAME
The name of the corporation is Mirion Technologies, Inc. (the Corporation). Capitalized terms used in this Certificate of Incorporation without definition shall have the meanings assigned thereto in Section 11.04.
ARTICLE 2
REGISTERED OFFICE AND AGENT
The address of the registered office of the Corporation in the State of Delaware is 850 New Burton Road, Suite 201, Dover, Delaware 19904, Kent County. The name of the registered agent of the Corporation in the State of Delaware at such address is Cogency Global Inc.
ARTICLE 3
PURPOSE AND POWERS
The purpose of the Corporation is to engage in any lawful act or activity for which corporations may be organized under the General Corporation Law of the State of Delaware as the same exists or may hereafter be amended (the DGCL).
ARTICLE 4
CAPITAL STOCK
Section 4.01. Authorized Shares.
(a) The total number of shares of all classes of stock that the Corporation shall have authority to issue is 2,200,000,000 shares, consisting of:
(i) 2,000,000,000 shares of Class A Common Stock, par value $0.0001 per share (the Class A Common Stock);
(ii) 100,000,000 shares of Class B Common Stock, par value $0.0001 per share (the Class B Common Stock and, together with the Class A Common Stock, the Common Stock); and
(iii) 100,000,000 shares of preferred stock, par value $0.0001 per share (the Preferred Stock).
(b) Subject to the rights of the holders of any series of Preferred Stock then outstanding, the number of authorized shares of any class or series of the Common Stock or the Preferred Stock may be increased or decreased by the affirmative vote of the holders of a majority of the total voting power of the outstanding shares of capital stock of the Corporation entitled to vote thereon, voting together as a single class, irrespective of the provisions of Section 242(b)(2) of the General Corporation Law, and no vote of the holders of any class of the
B-1
Table of Contents
Common Stock or the Preferred Stock voting separately as a class or series will be required therefor. Notwithstanding the immediately preceding sentence, the number of authorized shares of any particular class or series may not be decreased below the number of shares of such class or series then outstanding, plus, in the case of Class A Common Stock, the number of shares of Class A Common Stock issuable in connection with the redemption or exchange of all outstanding shares of Mirion IntermediateCo Class B Common Stock for Class A Common Stock pursuant to Section 4.04 of the Mirion IntermediateCo Charter (assuming for this purpose that such redemption or exchange is settled in shares of Class A Common Stock).
Section 4.02. Preferred Stock. The board of directors of the Corporation (the Board of Directors) is hereby empowered, without any action or vote by the Corporations stockholders (except as may otherwise be provided by the terms of any class or series of Preferred Stock then outstanding), to authorize by resolution or resolutions from time to time the issuance of one or more classes or series of Preferred Stock and to fix the designations, powers, preferences and relative, participating, optional or other rights, if any, and the qualifications, limitations or restrictions thereof, if any, with respect to each such class or series of Preferred Stock and the number of shares constituting each such class or series, and to increase or decrease the number of shares of any such class or series to the extent permitted by the DGCL.
Section 4.03. Common Stock. The rights, powers, preferences, privileges, restrictions and other matters relating to the Common Stock are as follows:
(a) Voting Rights.
(i) Except as otherwise required in this Certificate of Incorporation or by applicable law, the holders of Common Stock will vote together as a single class on all matters on which stockholders generally are entitled to vote (or, if any holders of Preferred Stock are entitled to vote together with the holders of Common Stock, as a single class with the holders of Preferred Stock).
(ii) Each holder of Class A Common Stock will be entitled to one vote for each share of Class A Common Stock held of record by such holder on all matters on which stockholders generally are entitled to vote, and each holder of Class B Common Stock will be entitled to one vote for each share of Class B Common Stock held of record by such holder on all matters on which stockholders generally are entitled to vote, except that, in each case, to the fullest extent permitted by law and subject to Section 4.03(a)(iii), holders of shares of each class of Common Stock, as such, will have no voting power with respect to, and will not be entitled to vote on, any amendment to this Certificate of Incorporation (including any certificate of designations relating to any series of Preferred Stock) that relates solely to the terms of any outstanding Preferred Stock if the holders of such Preferred Stock are entitled to vote as a separate class thereon under this Certificate of Incorporation (including any certificate of designations relating to any series of Preferred Stock) or under the DGCL.
(iii)
(A) The holders of the outstanding shares of Class A Common Stock shall be entitled to vote separately upon any amendment to this Certificate of Incorporation (including by merger, consolidation, reorganization or similar event) that would alter or change the powers, preferences or special rights of such class of Common Stock in a manner that is materially and disproportionately adverse as compared to any alteration or change to the Class B Common Stock; and
(B) The holders of the outstanding shares of Class B Common Stock shall be entitled to vote separately upon any amendment to this Certificate of Incorporation (including by merger, consolidation, reorganization or similar event) that would alter or change the powers, preferences or special rights of such class of Common Stock in a manner that is materially and disproportionately adverse as compared to any alteration or change to the Class A Common Stock, it being understood that this Section 4.03(a)(iii)(B) shall not apply to any amendment in connection with a merger, consolidation or other business combination if such merger, consolidation or other business combination constitutes a Disposition Event in which holders of Paired Interests are required to cause
B-2
Table of Contents
their Mirion IntermediateCo Class B Common Stock to be redeemed and their associated shares of Class B Common Stock to be retired, or exchange such Paired Interests, pursuant to Section 4.04(d) of the Mirion IntermediateCo Charter in such Disposition Event and receive consideration in such Disposition Event in accordance with the terms of the Mirion IntermediateCo Charter as in effect prior to such Disposition Event.
(iv) If at any time the ratio at which shares of Mirion IntermediateCo Class B Common Stock are redeemable or exchangeable for shares of Class A Common Stock pursuant to the Mirion IntermediateCo Charter is amended, the number of votes per share of Class B Common Stock to which holders of shares of Class B Common Stock are entitled pursuant to Section 4.03(a)(i) shall be adjusted accordingly.
(b) Dividends; Stock Splits or Combinations.
(i) Subject to applicable law and the rights, if any, of the holders of any outstanding series of Preferred Stock or any class or series of stock having a preference senior to or the right to participate with the Class A Common Stock with respect to the payment of dividends, dividends of cash or property may be declared and paid on the Class A Common Stock out of the assets of the Corporation that are by law available therefor, at the times and in the amounts as the Board of Directors in its discretion may determine.
(ii) Except as provided in Section 4.03(b)(iii) with respect to stock dividends, dividends of cash or property may not be declared or paid on shares of Class B Common Stock.
(iii) In no event will any stock dividend, stock split, reverse stock split, combination of stock, reclassification or recapitalization be declared or made on any class of Common Stock (each, a Stock Adjustment) unless (A) a corresponding Stock Adjustment for all other classes of Common Stock not so adjusted at the time outstanding is made in the same proportion and the same manner and (B) the Stock Adjustment has been reflected in the same economically equivalent manner on all shares of common stock of Mirion IntermediateCo. Stock dividends with respect to each class of Common Stock may only be paid with shares of stock of the same class of Common Stock.
(c) Liquidation Rights. In the event of any voluntary or involuntary liquidation, dissolution or winding-up of the affairs of the Corporation, after payment or provision for payment of the debts and other liabilities of the Corporation and of the preferential and other amounts, if any, to which the holders of Preferred Stock are entitled, if any, the holders of all outstanding shares of Class A Common Stock will be entitled to receive, pari passu, an amount per share equal to the par value thereof, and thereafter the holders of all outstanding shares of Class A Common Stock will be entitled to receive the remaining assets of the Corporation available for distribution ratably in proportion to the number of shares of Class A Common Stock. The holders of shares of Class B Common Stock, as such, will not be entitled to receive, with respect to such shares, any assets of the Corporation in excess of the par value thereof, in the event of any voluntary or involuntary liquidation, dissolution or winding up of the affairs of the Corporation; provided this Section 4.03(c) shall not limit the rights of the holders of shares of Class B Common Stock to cause their shares of Mirion IntermediateCo Class B Common Stock to be redeemed or exchanged by Mirion IntermediateCo for shares of Class A Common Stock or cash in accordance with Section 4.04 of the Mirion IntermediateCo Charter (or for the consideration payable in respect of shares of Class A Common Stock in such voluntary or involuntary liquidation, dissolution or winding-up).
(d) Transfer Restrictions; Retirement of Class B Common Stock.
(i) No holder of shares of Class B Common Stock may Transfer such shares to any person unless such holder Transfers a corresponding number of shares of Mirion IntermediateCo Class B Common Stock (as part of Paired Interests) to the same Person in accordance with the Mirion IntermediateCo Charter. If any outstanding share of Class B Common Stock ceases to be held by a holder of a share of Mirion IntermediateCo Class B Common Stock, such share shall automatically and without further action on the part of the Corporation or such holder be transferred to the Corporation for no consideration and retired.
B-3
Table of Contents
(ii) To the extent that any holder of shares of Mirion IntermediateCo Class B Common Stock exercises its right pursuant to Section 4.04 of the Mirion IntermediateCo Charter to have its Mirion IntermediateCo Class B Common Stock redeemed by Mirion IntermediateCo in accordance with the Mirion IntermediateCo Charter, then simultaneously with the payment of cash or Class A Common Stock consideration in accordance with the Mirion IntermediateCo Charter, the Corporation shall cancel for no consideration a number of shares of Class B Common Stock registered in the name of the redeeming or exchanging holder equal to the number of Mirion IntermediateCo Class B Common Stock held by such holder that are redeemed or exchanged in such redemption or exchange transaction.
(iii) The shares of Class B Common Stock may be notated with one or more of the following legends:
(A) THE SHARES REPRESENTED HEREBY ARE SUBJECT TO TRANSFER RESTRICTIONS SET FORTH IN THE CORPORATIONS CERTIFICATE OF INCORPORATION. NO TRANSFER MAY BE EFFECTED EXCEPT IN COMPLIANCE WITH THE TERMS OF THE CERTIFICATE OF INCORPORATION;
(B) THE SHARES REPRESENTED HEREBY HAVE NOT BEEN REGISTERED UNDER THE SECURITIES ACT OF 1933, AS AMENDED, AND HAVE BEEN ACQUIRED FOR INVESTMENT AND NOT WITH A VIEW TO, OR IN CONNECTION WITH, THE SALE OR DISTRIBUTION THEREOF. NO SUCH TRANSFER MAY BE EFFECTED WITHOUT AN EFFECTIVE REGISTRATION STATEMENT RELATED THERETO OR AN OPINION OF COUNSEL IN A FORM SATISFACTORY TO THE COMPANY THAT SUCH REGISTRATION IS NOT REQUIRED UNDER THE SECURITIES ACT OF 1933; and
(C) Any other legend required by applicable securities laws of any jurisdiction to the extent such laws are applicable to such shares of Class B Common Stock.
(e) Reservation of Shares of Class A Common Stock. The Corporation will at all times reserve and keep available out of its authorized and unissued shares of Class A Common Stock, solely for the purpose of the issuance upon redemption or exchange of shares of Mirion IntermediateCo Class B Common Stock, the number of shares of Class A Common Stock that are issuable upon conversion of all outstanding shares of Mirion IntermediateCo Class B Common Stock, pursuant to Section 4.04(e) of the Mirion IntermediateCo Charter (assuming for this purpose that such redemption or exchange is settled in shares of Class A Common Stock). The Corporation covenants that all the shares of Class A Common Stock that are issued upon the redemption or exchange of such shares of Mirion IntermediateCo Class B Common Stock will, upon issuance, be validly issued, fully paid and non-assessable.
(f) Taxes. The issuance of shares of Class A Common Stock upon the exercise by holders of shares of Class B Common Stock of their right under Section 4.04 of the Mirion IntermediateCo Charter to cause Mirion IntermediateCo to redeem shares of Mirion IntermediateCo Class B Common Stock will be made without charge to the holders of the shares of Class B Common Stock for any transfer taxes, stamp taxes or duties or other similar tax in respect of the issuance; provided, however, that if any such shares of Class A Common Stock are to be issued in a name other than that of the then record holder of the shares of Mirion IntermediateCo Class B Common Stock being redeemed or exchanged (or The Depository Trust Company or its nominee for the account of a participant of The Depository Trust Company that will hold the shares for the account of such holder), then such holder and/or the Person in whose name such shares are to be delivered, shall pay to the Corporation the amount of any tax that may be payable in respect of any transfer involved in the issuance or shall establish to the reasonable satisfaction of the Corporation that the tax has been paid or is not payable.
Section 4.04. Incorporation of Mirion IntermediateCo Charter. The Corporation will take the actions required, and observe the restrictions, applicable to it set forth under Sections 4.02(b) and 4.04 of the Mirion IntermediateCo Charter.
B-4
Table of Contents
ARTICLE 5
BYLAWS
The Board of Directors shall have the power to adopt, amend or repeal the bylaws of the Corporation (the Bylaws).
The stockholders may adopt, amend or repeal the Bylaws only with the affirmative vote of the holders of not less than 66 2/3% of the voting power of all outstanding securities of the Corporation generally entitled to vote in the election of directors, voting together as a single class.
ARTICLE 6
BOARD OF DIRECTORS
Section 6.01. Power of the Board of Directors. The business and affairs of the Corporation shall be managed by or under the direction of the Board of Directors.
Section 6.02. Number of Directors. The number of directors which shall constitute the Board of Directors shall be fixed exclusively by one or more resolutions adopted from time to time solely by the affirmative vote of a majority of the Board of Directors.
Section 6.03. Election of Directors.
(a) Each director shall be elected annually by the stockholders and shall serve for a term ending on the date of the annual meeting of stockholders next following the annual meeting at which such director was elected. Notwithstanding the foregoing, each director shall hold office until such directors successor shall have been duly elected and qualified or until such directors earlier death, resignation or removal. In no event will a decrease in the number of directors shorten the term of any incumbent director.
(b) There shall be no cumulative voting in the election of directors. Election of directors need not be by written ballot unless the Bylaws so provide.
Section 6.04. Vacancies. Vacancies on the Board of Directors resulting from death, resignation, removal or otherwise and newly created directorships resulting from any increase in the number of directors shall, except as otherwise required by law, be filled solely by a majority of the directors then in office (although less than a quorum) or by the sole remaining director.
Section 6.05. Removal. Any director may be removed from office by the stockholders, with or without cause, by the affirmative vote of the holders of not less than a majority of the total voting power of all outstanding securities of the Corporation generally entitled to vote in the election of directors, voting together as a single class.
Section 6.06. Preferred Stock Directors. Notwithstanding anything else contained herein, whenever the holders of one or more classes or series of Preferred Stock shall have the right, voting separately as a class or series, to elect directors, the election, term of office, filling of vacancies, removal and other features of such directorships shall be governed by the terms of such class or series of Preferred Stock adopted by resolution or resolutions adopted by the Board of Directors pursuant to Section 4.02 hereto, and such directors so elected shall not be subject to the provisions of this Article 6 unless otherwise provided therein.
ARTICLE 7
MEETINGS OF STOCKHOLDERS
Section 7.01. Annual Meetings. An annual meeting of stockholders for the election of directors to succeed those whose terms expire and for the transaction of such other business as may properly come before the meeting shall be held at such place, on such date, and at such time as the Board of Directors shall determine.
B-5
Table of Contents
Section 7.02. Special Meetings. Special meetings of the stockholders may be called only by (a) the Board of Directors acting pursuant to a resolution adopted by a majority of the Board of Directors, (b) the chairman of the Board of Directors or (c) the Chief Executive Officer of the Corporation. Notwithstanding the foregoing, whenever holders of one or more classes or series of Preferred Stock shall have the right, voting separately as a class or series, to elect directors, such holders may call, pursuant to the terms of such class or series of Preferred Stock adopted by resolution or resolutions of the Board of Directors pursuant to Section 4.02 hereto, special meetings of holders of such Preferred Stock.
Section 7.03. No Action by Written Consent. Subject to the rights of the holders of any class or series of Preferred Stock then outstanding, as may be set forth in the resolution or resolutions adopted by the Board of Directors pursuant to Section 4.02 hereto for such class or series of Preferred Stock, any action required or permitted to be taken at any annual or special meeting of stockholders may be taken only upon the vote of stockholders at an annual or special meeting duly noticed and called in accordance with the DGCL, as amended from time to time, and this Article 7, and may not be taken by written consent of stockholders without a meeting.
ARTICLE 8
INDEMNIFICATION
Section 8.01. Limited Liability. A director of the Corporation shall not be liable to the Corporation or its stockholders for monetary damages for breach of fiduciary duty as a director to the fullest extent permitted by the DGCL. Without limiting the effect of the preceding sentence, if the DGCL is hereafter amended to authorize the further elimination or limitation of the liability of a director, then the liability of a director of the Corporation shall be eliminated or limited to the fullest extent permitted by the DGCL, as so amended.
Section 8.02. Right to Indemnification.
(a) Each Person (and the heirs, executors or administrators of such Person) who was or is a party or is threatened to be made a party to, or is involved in any threatened, pending or completed action, suit or proceeding, whether civil, criminal, administrative or investigative, by reason of the fact that such Person is or was a director or officer of the Corporation or is or was serving at the request of the Corporation as a director or officer of another corporation, partnership, joint venture, trust or other enterprise (each, an Indemnified Person), shall be indemnified and held harmless by the Corporation to the fullest extent permitted by the DGCL. The right to indemnification conferred in this Article 8 shall also include the right to be paid by the Corporation the expenses incurred in connection with any such proceeding in advance of its final disposition to the fullest extent authorized by the DGCL provided, that, such Indemnified Person agrees to repay such amount if it shall ultimately be determined that such Indemnified Person is not entitled to be indemnified by the Corporation as authorized in this Article 8. The right to indemnification conferred in this Article 8 shall be a contract right.
(b) The Corporation may, by action of its Board of Directors, provide indemnification to such of the employees and agents of the Corporation to such extent and to such effect as the Board of Directors shall determine to be appropriate and authorized by the DGCL.
Section 8.03. Insurance. The Corporation shall have power to purchase and maintain insurance on behalf of any Person who is or was a director, officer, employee or agent of the Corporation, or is or was serving at the request of the Corporation as a director, officer, employee or agent of another corporation, partnership, joint venture, trust or other enterprise against any expense, liability or loss incurred by such Person in any such capacity or arising out of such Persons status as such, whether or not the Corporation would have the power to indemnify such Person against such liability under the DGCL.
Section 8.04. Priority of Indemnification. With respect to any Indemnified Person who has rights to indemnification, advancement of expenses or insurance provided by any stockholder of the Corporation, the
B-6
Table of Contents
Corporation and its respective direct and indirect Subsidiaries (collectively, the Company Group), then the Company Group shall be primarily liable for all indemnification, reimbursements, advancements or similar payments (the Indemnity Obligations) afforded to such Indemnified Person acting in such capacity or capacities on behalf or at the request of the Board of Directors, the Corporation or any other member of the Company Group, in such capacity, whether the Indemnity Obligations are created by law, organizational or constituent documents, contract or otherwise. Notwithstanding the fact that any stockholder of the Corporation and its Affiliates other than the Company Group (such Persons (excluding any member of the Company Group), together with their respective heirs, successors and assigns, the Other Indemnitors) may have concurrent liability to an Indemnified Person with respect to the Indemnity Obligations, the Corporation hereby agrees that in no event shall the Corporation or any other member of the Company Group have any right or claim against any of the Other Indemnitors for contribution or have rights of subrogation against any Other Indemnitors through an Indemnified Person for any payment made by the Corporation or any other member of the Company Group with respect to any Indemnity Obligation. In addition, the Corporation hereby agrees that no advancement or payment by the Other Indemnitors on behalf of an Indemnified Person with respect to any claim for which an Indemnified Person has sought indemnification from the Corporation or any other member of the Company Group shall affect the foregoing and the Other Indemnitors shall have a right of contribution and/or be subrogated to the extent of such advancement or payment to all of the rights of recovery of such Indemnified Person against the Corporation or any other member of the Company Group.
Section 8.05. Nonexclusivity of Rights. The rights and authority conferred in this Article 8 shall not be exclusive of any other right that any Person may otherwise have or hereafter acquire.
Section 8.06. Preservation of Rights. Neither the amendment nor repeal of this Article 8, nor the adoption of any provision of this Certificate of Incorporation or the Bylaws, nor, to the fullest extent permitted by the DGCL, any modification of law, shall adversely affect any right or protection of any Person granted pursuant hereto existing at, or arising out of or related to any event, act or omission that occurred prior to, the time of such amendment, repeal, adoption or modification (regardless of when any proceeding (or part thereof) relating to such event, act or omission arises or is first threatened, commenced or completed).
ARTICLE 9
CORPORATE OPPORTUNITIES
In the event that a member of the Board of Directors who is not an employee of the Corporation or its Subsidiaries, or any employee or agent of such member, other than someone who is an employee of the Corporation or its Subsidiaries (collectively, the Covered Persons), acquires knowledge of any business opportunity matter, potential transaction, interest or other matter, even if such business opportunity matter, potential transaction, interest or other matter is one that the Corporation or its Subsidiaries might reasonably be deemed to have pursued or had the ability or desire to pursue if granted the opportunity to do so, and each such Covered Persons shall have no duty to communicate or offer such business opportunity matter, potential transaction, interest or other matter to the Corporation (and there shall be no restriction on the Covered Persons using the general knowledge and understanding of the industry in which the Corporation operates which it has gained as a Covered Person in considering and pursuing such business opportunity matter, potential transaction, interest or other matter or in making investment, voting, monitoring, governance or other decisions relating to other entities or securities), unless such matter, transaction or interest is presented to, or acquired, created or developed by, or otherwise comes into the possession of, a Covered Person expressly and solely in connection with such individuals service as a member of the Board of Directors of the Corporation (a Corporate Opportunity), then the Corporation to the maximum extent permitted from time to time under the DGCL (including Section 122(17) thereof):
(a) renounces any expectancy that such Covered Person offer an opportunity to participate in such Corporate Opportunity to the Corporation; and
B-7
Table of Contents
(b) waives any claim that such opportunity constituted a Corporate Opportunity that should have been presented by such Covered Person to the Corporation or any of its Affiliates.
In addition to and notwithstanding the foregoing, a Corporate Opportunity shall not be deemed to belong to the Corporation if it is a business opportunity that the Corporation is not financially able or contractually permitted or legally able to undertake, or that is, from its nature, not in the line of the Corporations business or is of no practical advantage to it or that is one in which the Corporation has no interest or reasonable expectancy. No amendment or repeal of this paragraph shall apply to or have any effect on the liability or alleged liability of any officer, director or stockholder of the Corporation for or with respect to any opportunities of which such officer, director or stockholder becomes aware prior to such amendment or repeal.
ARTICLE 10
EXCLUSIVE FORUM
Section 10.01. Corporate Claim Exclusive Forum. Unless the Corporation consents in writing to the selection of an alternative forum, the Court of Chancery of the State of Delaware (or, if and only if the Court of Chancery of the State of Delaware lacks subject matter jurisdiction, any state court located within the State of Delaware or, if and only if all such state courts lack subject matter jurisdiction, the federal district court for the District of Delaware) and any appellate court therefrom shall be the sole and exclusive forum for the following claims or causes of action under the Delaware statutory or common law:
(a) any derivative claim or cause of action brought on behalf of the Corporation;
(b) any claim or cause of action for breach of a fiduciary duty owed by any current or former director, officer or other employee of the Corporation, to the Corporation or the Corporations stockholders;
(c) any claim or cause of action against the Corporation or any current or former director, officer or other employee of the Corporation, arising out of or pursuant to any provision of the DGCL, this Certificate of Incorporation or the Bylaws (as each may be amended from time to time);
(d) any claim or cause of action seeking to interpret, apply, enforce or determine the validity of this Certificate of Incorporation or the Bylaws (as each may be amended from time to time, including any right, obligation, or remedy thereunder);
(e) any claim or cause of action as to which the DGCL confers jurisdiction on the Court of Chancery of the State of Delaware; and
(f) any claim or cause of action against the Corporation or any current or former director, officer or other employee of the Corporation, governed by the internal-affairs doctrine, in all cases to the fullest extent permitted by law and subject to the court having personal jurisdiction over the indispensable parties named as defendants.
This Section 10.01 shall not apply to claims or causes of action brought to enforce a duty or liability created by the Securities Act of 1933, as amended (the Securities Act), or the Securities Exchange Act of 1934, as amended (the Exchange Act), or any other claim for which the federal courts have exclusive jurisdiction.
Section 10.02. Securities Act Exclusive Forum. Unless the Corporation consents in writing to the selection of an alternative forum, to the fullest extent permitted by law, the federal district courts of the United States of America shall be the exclusive forum for the resolution of any complaint asserting a cause of action arising under the Securities Act. This Section 10.02 shall not apply to claims or causes of action brought to enforce a duty or liability created by the Exchange Act, or any other claim for which the federal courts have exclusive jurisdiction.
B-8
Table of Contents
ARTICLE 11
AMENDMENTS; MISCELLANEOUS; CERTAIN DEFINITIONS
Section 11.01. Amendments. The Corporation reserves the right to amend this Certificate of Incorporation in any manner permitted by the DGCL and all rights and powers conferred upon stockholders, directors and officers herein are granted subject to this reservation. Notwithstanding the foregoing, the provisions set forth in Section 4.03 and Articles 5, 6, 7, 8, 9 and this 11 (and any defined terms referenced therein) may not be repealed or amended in any respect, and no other provision may be adopted, amended or repealed which would have the effect of modifying or permitting the circumvention of the provisions set forth therein, unless such action is approved by the affirmative vote of the holders of not less than 66 2/3% of the total voting power of all outstanding securities of the Corporation generally entitled to vote in the election of directors, voting together as a single class.
Section 11.02. Severability. If any provision of this Certificate of Incorporation becomes or is declared on any ground by a court of competent jurisdiction to be illegal, unenforceable or void, portions of such provision, or such provision in its entirety, to the extent necessary, shall be severed from this Certificate of Incorporation, and the court will replace such illegal, void or unenforceable provision of this Certificate of Incorporation with a valid and enforceable provision that most accurately reflects the Corporations intent, in order to achieve, to the maximum extent possible, the same economic, business and other purposes of the illegal, void or unenforceable provision. The balance of this Certificate of Incorporation shall be enforceable in accordance with its terms.
Section 11.03. Deemed Notice. Any Person holding, owning or otherwise acquiring any interest in any security of the Corporation shall be deemed to have notice of and consented to all of the provisions of this Certificate of Incorporation.
Section 11.04. Certain Definitions. As used in this Certificate of Incorporation, unless the context otherwise requires or as set forth in another Article or Section of this Certificate of Incorporation, the term:
(a) Affiliate means, with respect to any Person, any other person or entity who, as of the relevant time for which the determination of affiliation is being made, directly or indirectly controls, is controlled by or is under common control with such Person.
(b) Disposition Event means any merger, consolidation or other business combination of the Corporation, whether effectuated through one transaction or series of related transactions (including a tender offer followed by a merger in which holders of Class A Common Stock receive the same consideration per share paid in the tender offer), unless, following such transaction, all or substantially all of the holders of the voting power of all outstanding classes of Common Stock and series of Preferred Stock that are generally entitled to vote in the election of Directors prior to such transaction or series of transactions, continue to hold a majority of the voting power of the surviving entity (or its parent) resulting from such transaction or series of transactions in substantially the same proportions as immediately prior to such transaction or series of transactions.
(c) Mirion IntermediateCo means Mirion IntermediateCo, Inc., a Delaware corporation.
(d) Mirion IntermediateCo Charter means the certificate of incorporation of Mirion IntermediateCo, as it may be amended, restated or otherwise modified from time to time.
(e) Mirion IntermediateCo Class B Common Stock means the shares of Class B common stock, par value $0.0001 per share, of Mirion IntermediateCo.
(f) Paired Interest means one share of Class B Common Stock and one share of Mirion IntermediateCo Class B Common Stock, subject to adjustment pursuant to Section 4.03(a)(iv).
(g) Person means any individual, partnership, firm, corporation, limited liability company, association, trust, unincorporated organization or other entity.
B-9
Table of Contents
(h) Subsidiary means, with respect to any Person, any corporation, partnership, limited liability company, association, joint venture or other business entity of which more than 50% of the total voting power of equity securities or other ownership interests entitled (without regard to the occurrence of any contingency) to vote in the election of the Person or Persons (whether directors, managers, trustees or other Persons performing similar functions) having the power to direct or cause the direction of the management and policies thereof is at the time owned or controlled, directly or indirectly, by that Person or one or more of the other Subsidiaries of that Person or a combination thereof.
(i) Transfer of a share of Class B Common Stock means, directly or indirectly, any sale, assignment, exchange, gift, bequest, pledge, hypothecation or other transfer, disposition or encumbrance of such share or any legal or beneficial interest in such share, in whole or in part, whether or not for value and whether voluntary or involuntary or by operation of law; provided, however, that the following shall not be considered a Transfer:
(i) the granting of a revocable proxy to officers or directors of the Corporation at the request of the Board of Directors in connection with actions to be taken at an annual or special meeting of stockholders or in connection with any action by written consent of the stockholders solicited by the Board of Directors;
(ii) entering into a customary voting or support agreement (with or without granting a proxy) in connection with any merger, consolidation or other business combination of the Corporation, whether effectuated through one transaction or series of related transactions (including a tender offer followed by a merger in which holders of Class A Common Stock receive the same consideration per share paid in the tender offer);
(iii) the pledge of shares of capital stock of the Corporation by a stockholder that creates a mere security interest in such shares pursuant to a bona fide loan or indebtedness transaction so long as such stockholder continues to exercise sole voting control over such pledged shares; provided, however, that a foreclosure on such shares or other similar action by the pledgee shall constitute a Transfer hereunder unless subject to an exception in the definition thereof;
(iv) entering into a trading plan pursuant to Rule 10b5-1 under the Securities Exchange Act of 1934, as amended, with a broker or other nominee; provided, however, that a sale of shares of Class A Common Stock upon redemption or exchange of Mirion IntermediateCo Class B Common Stock pursuant to such plan shall constitute a Transfer at the time of such sale unless subject to an exception in the definition thereof; or
(v) the fact that the spouse of any holder of Class B Common Stock possesses or obtains an interest in such holders shares of Class B Common Stock arising solely by reason of the application of the community property laws of any jurisdiction, so long as no other event or circumstance shall exist or have occurred that constitutes a Transfer of such shares of Class B Common Stock.
[Remainder of this page intentionally left blank]
B-10
Table of Contents
IN WITNESS WHEREOF, the undersigned has executed this Certificate of Incorporation this __ day of ___ 2021.
|
|
| Name: Title: |
[Signature Page to Certificate of Incorporation]
Table of Contents
Final Form
FORM OF AMENDED AND RESTATED BYLAWS
OF
MIRION TECHNOLOGIES, INC.
* * * * *
ARTICLE 1
OFFICES
Section 1.01. Registered Office. The address of the registered office of Mirion Technologies, Inc. (the Corporation) in the State of Delaware is 850 New Burton Road, Suite 201, Dover, Delaware 19904, Kent County. The name of the registered agent of the Corporation at such address is Cogency Global Inc.
Section 1.02. Other Offices. The Corporation may also have offices at such other places both within and without the State of Delaware as the board of the directors of the Corporation (the Board of Directors) may from time to time determine or the business of the Corporation may require.
Section 1.03. Books. The books of the Corporation may be kept within or without the State of Delaware as the Board of Directors may from time to time determine or the business of the Corporation may require.
ARTICLE 2
MEETINGS OF STOCKHOLDERS
Section 2.01. Time and Place of Meetings. All meetings of stockholders shall be held at such place, either within or without the State of Delaware, on such date and at such time as may be determined from time to time by the Board of Directors (or the chairperson of the Board of Directors in the absence of a designation by the Board of Directors). The Board of Directors may, in its sole discretion, determine that a meeting of the stockholders shall not be held at any place, but instead be held solely by means of remote communication authorized by and in accordance with Section 211(a)(2) of the General Corporation Law of the State of Delaware as the same exists or may hereafter be amended (DGCL).
Section 2.02. Annual Meetings. An annual meeting of stockholders shall be held for the election of directors and to transact such other business as may properly be brought before the meeting.
Section 2.03. Special Meetings.
(a) Except as otherwise provided in the Corporations certificate of incorporation (as the same may be modified or further amended, restated, amended and restated or otherwise modified from time to time, the Certificate of Incorporation), special meetings of the stockholders may be called only by the (i) the Board of Directors acting pursuant to a resolution adopted by a majority of the Board of Directors, (ii) the chairperson of the Board of Directors, (iii) the Chief Executive Officer of the Corporation or (iv) whenever holders of none or more classes of series of preferred stock of the Corporation (the Preferred Stock) shall have the right, voting separately as a class or series, to elect directors, such holders may call, pursuant to the terms of such class or series of Preferred Stock, special meetings of holders of such Preferred Stock.
(b) A special meeting shall be held at such date, time and place as may be fixed by the Board of Directors in accordance with these Amended and Restated Bylaws (these Bylaws).
C-1
Table of Contents
(c) Business conducted at a special meeting shall be limited to the matters described in the applicable request for such special meeting and any other matters as the Board of Directors shall determine.
Section 2.04. Notice of Meetings and Adjourned Meetings; Waivers of Notice. (a) Whenever stockholders are required or permitted to take any action at a meeting, a written notice of the meeting shall be given which shall state the place, if any, date and hour of the meeting, the means of remote communications, if any, by which stockholders and proxy holders may be deemed to be present in person and vote at such meeting, and, in the case of a special meeting, the purpose or purposes for which the meeting is called. Unless otherwise provided by the DGCL, such notice shall be given not less than 10 nor more than 60 days before the date of the meeting to each stockholder of record entitled to vote at such meeting. The Board of Directors or the chairperson of the meeting may adjourn the meeting to another time or place (whether or not a quorum is present), and notice need not be given of the adjourned meeting if the time, place, if any, and the means of remote communications, if any, by which stockholders and proxy holders may be deemed to be present in person and vote at such meeting, are announced at the meeting at which such adjournment is made. At the adjourned meeting, the Corporation may transact any business which might have been transacted at the original meeting. If the adjournment is for more than 30 days, or after the adjournment a new record date is fixed for the adjourned meeting, a notice of the adjourned meeting shall be given to each stockholder of record entitled to vote at the meeting.
(b) A written waiver of any such notice signed by the person entitled thereto, or a waiver by electronic transmission by the person entitled to notice, whether before or after the time stated therein, shall be deemed equivalent to notice. Attendance of a person at a meeting shall constitute a waiver of notice of such meeting, except when the person attends the meeting for the express purpose of objecting, at the beginning of the meeting, to the transaction of any business because the meeting is not lawfully called or convened. Business transacted at any special meeting of stockholders shall be limited to the purposes stated in the notice.
Section 2.05. Quorum. Unless otherwise provided under the Certificate of Incorporation or these Bylaws and subject to the DGCL, the presence, in person or by proxy, of the holders of a majority of the total voting power of all outstanding securities of the Corporation generally entitled to vote at a meeting of stockholders shall constitute a quorum for the transaction of business, except that when specified business is to be voted on by a class or series of securities voting as a separate class or series, the holders of a majority in voting power of the outstanding securities of such class or series shall constitute a quorum of such class or series. If, however, such quorum shall not be present or represented at any meeting of the stockholders, the chairperson of the meeting or a majority in voting interest of the stockholders present in person or represented by proxy may adjourn the meeting without notice other than announcement at the meeting, until a quorum shall be present or represented. At such adjourned meeting at which a quorum shall be present or represented any business may be transacted that might have been transacted at the meeting as originally notified.
Section 2.06. Voting. (a) Unless otherwise provided in the Certificate of Incorporation and subject to the DGCL, each stockholder shall be entitled to one vote for each outstanding share of capital stock of the Corporation held by such stockholder. Any share of capital stock of the Corporation held by the Corporation shall have no voting rights. Except as otherwise required by law, the Certificate of Incorporation or these Bylaws, in all matters other than the election of directors, the affirmative vote of the holders of a majority of the votes cast at the meeting on the subject matter shall be the act of the stockholders. Abstentions and broker non-votes shall not be counted as votes cast. Subject to the rights of the holders of any class or series of preferred stock to elect additional directors under specific circumstances, as may be set forth in the certificate of designations for such class or series of preferred stock, directors shall be elected by a plurality of the votes of the shares of capital stock of the Corporation present in person or represented by proxy at the meeting and entitled to vote on the election of directors.
(b) Each stockholder entitled to vote at a meeting of stockholders or to express consent or dissent to a corporate action in writing without a meeting may authorize another person or persons to act for such stockholder by proxy, appointed by an instrument in writing, subscribed by such stockholder or by his attorney thereunto
C-2
Table of Contents
authorized, or by proxy sent by cable, telegram or by any means of electronic communication permitted by law, which results in a writing from such stockholder or by his attorney, and delivered to the secretary of the meeting. No proxy shall be voted after three (3) years from its date, unless said proxy provides for a longer period.
Section 2.07. No Action by Written Consent. (a) Subject to the rights of the holders of any class or series of preferred stock then outstanding as may be set forth in the certificate of designations for such class or series of preferred stock or except as provided for in the Certificate of Incorporation, any action required or permitted to be taken at any annual or special meeting of stockholders may be taken only upon the vote of stockholders at an annual or special meeting duly noticed and called in accordance with the DGCL and may not be taken by written consent of stockholders without a meeting.
Section 2.08. Organization. At each meeting of stockholders, the chairperson of the Board of Directors, if one shall have been elected, or in the chairpersons absence or if one shall not have been elected, the director designated by the vote of the majority of the directors present at such meeting, shall act as chairperson of the meeting. The Secretary (or in the Secretarys absence or inability to act, the person whom the chairperson of the meeting shall appoint secretary of the meeting) shall act as secretary of the meeting and keep the minutes thereof.
Section 2.09. Order of Business. The order of business at all meetings of stockholders shall be as determined by the chairperson of the meeting.
Section 2.10. Nomination of Directors and Proposal of Other Business.
(a) Annual Meetings of Stockholders. (i) Nominations of persons for election to the Board of Directors or the proposal of other business to be transacted by the stockholders at an annual meeting of stockholders may be made only (A) pursuant to the Corporations notice of meeting (or any supplement thereto), (B) by or at the direction of the Board of Directors or any committee thereof (C) as may be provided in the certificate of designations for any class or series of preferred stock or (D) by any stockholder of the Corporation who is a stockholder of record at the time of giving of notice provided for in paragraph (ii) of this Section 2.10(a) and at the time of the annual meeting, who shall be entitled to vote at the meeting and who complies with the procedures set forth in this Section 2.10(a), and, except as otherwise required by law, any failure to comply with these procedures shall result in the nullification of such nomination or proposal.
(ii) For nominations or other business to be properly brought before an annual meeting of stockholders by a stockholder pursuant to Section 2.10(a)(i)(D), the stockholder must have given timely notice thereof in writing to the Secretary of the Corporation and any such proposed business (other than the nominations of persons for election to the Board of Directors) must constitute a proper matter for stockholder action. To be timely, a stockholders notice shall be delivered to, or mailed and received by, the Secretary of the Corporation at the principal executive offices of the Corporation not less than 120 days nor more than 150 days prior to the first anniversary of the preceding years annual meeting of stockholders; provided, however, that in the event that the date of the annual meeting is advanced more than 30 days prior to such anniversary date or delayed more than 70 days after such anniversary date then to be timely such notice must be received by the Corporation no earlier than 120 days prior to such annual meeting and no later than the later of 70 days prior to the date of the meeting or the 10th day following the day on which public announcement of the date of the meeting was first made by the Corporation. In no event shall the adjournment or postponement of any meeting, or any announcement thereof, commence a new time period (or extend any time period) for the giving of a stockholders notice as described above.
(iii) A stockholders notice to the Secretary shall set forth (A) as to each person whom the stockholder proposes to nominate for election or reelection as a director: (1) all information relating to such person that is required to be disclosed in solicitations of proxies for election of directors, or is otherwise required, in each case pursuant to Regulation 14A under the Securities Exchange Act of 1934 (as amended (together with the rules and regulations promulgated thereunder), the Exchange Act) including such persons written consent to being named in the proxy statement as a nominee and to serving as a director if elected;
C-3
Table of Contents
and (2) a reasonably detailed description of any compensatory, payment or other financial agreement, arrangement or understanding that such person has with any other person or entity other than the Corporation including the amount of any payment or payments received or receivable thereunder, in each case in connection with candidacy or service as a director of the Corporation (a Third-Party Compensation Arrangement), (B) as to any other business that the stockholder proposes to bring before the meeting, a brief description of the business desired to be brought before the meeting, the text of the proposal or business (including the text of any resolutions proposed for consideration and in the event that such business includes a proposal to amend these Bylaws, the text of the proposed amendment), the reasons for conducting such business and any material interest in such business of such stockholder and the beneficial owner, if any, on whose behalf the proposal is made and (C) as to the stockholder giving the notice and the beneficial owner, if any, on whose behalf the proposal is made:
(1) the name and address of such stockholder (as they appear on the Corporations books) and any such beneficial owner;
(2) for each class or series, the number of shares of capital stock of the Corporation that are held of record or are beneficially owned by such stockholder and by any such beneficial owner;
(3) a description of any agreement, arrangement or understanding between or among such stockholder and any such beneficial owner, any of their respective affiliates or associates, and any other person or persons (including their names) in connection with the proposal of such nomination or other business;
(4) a description of any agreement, arrangement or understanding (including, regardless of the form of settlement, any derivative, long or short positions, profit interests, forwards, futures, swaps, options, warrants, convertible securities, stock appreciation or similar rights, hedging transactions and borrowed or loaned shares) that has been entered into by or on behalf of, or any other agreement, arrangement or understanding that has been made, the effect or intent of which is to create or mitigate loss to, manage risk or benefit of share price changes for, or increase or decrease the voting power of, such stockholder or any such beneficial owner with respect to the Corporations securities;
(5) a representation that the stockholder is a holder of record of stock of the Corporation entitled to vote at such meeting and intends to appear in person or by proxy at the meeting to bring such nomination or other business before the meeting;
(6) a representation as to whether such stockholder or any such beneficial owner intends or is part of a group that intends to (i) deliver a proxy statement and/or form of proxy to holders of at least the percentage of the voting power of the Corporations outstanding capital stock required to approve or adopt the proposal or to elect each such nominee and/or (ii) otherwise to solicit proxies from stockholders in support of such proposal or nomination;
(7) any other information relating to such stockholder, beneficial owner, if any, or director nominee or proposed business that would be required to be disclosed in a proxy statement or other filing required to be made in connection with the solicitation of proxies in support of such nominee or proposal pursuant to Section 14 of the Exchange Act; and
(8) such other information relating to any proposed item of business as the Corporation may reasonably require to determine whether such proposed item of business is a proper matter for stockholder action.
If requested by the Corporation, the information required under clauses 2.10(a)(iii)(C)(2), (3) and (4) of the preceding sentence of this Section 2.10 shall be supplemented by such stockholder and any such beneficial owner not later than 10 days after the record date for the meeting to disclose such information as of the record date.
(b) Special Meetings of Stockholders. Except to the extent required by the DGCL, and subject to Section 2.03 of these Bylaws, special meetings of stockholders may be called only in accordance with the
C-4
Table of Contents
Certificate of Incorporation. Only such business shall be conducted at a special meeting of stockholders as shall have been brought before the meeting pursuant to the Corporations notice of meeting. If the election of directors is included as business to be brought before a special meeting in the Corporations notice of meeting, then nominations of persons for election to the Board of Directors at a special meeting of stockholders may be made by any stockholder who is a stockholder of record at the time of giving of notice provided for in this Section 2.10(b) and at the time of the special meeting, who shall be entitled to vote at the meeting and who complies with the procedures set forth in this Section 2.10(b). For nominations to be properly brought by a stockholder before a special meeting of stockholders pursuant to this Section 2.10(b), the stockholder must have given timely notice thereof in writing to the Secretary of the Corporation. To be timely, a stockholders notice shall be delivered to or mailed and received at the principal executive offices of the Corporation (A) not earlier than 150 days prior to the date of the special meeting nor (B) later than the later of 120 days prior to the date of the special meeting or the 10th day following the day on which public announcement of the date of the special meeting was first made. A stockholders notice to the Secretary shall comply with the notice requirements of Section 2.10(a)(iii).
(c) General. (i) To be eligible to be a nominee for election as a director, the proposed nominee must provide to the Secretary of the Corporation in accordance with the applicable time periods prescribed for delivery of notice under Section 2.10(a)(ii) or Section 2.10(b): (1) a completed D&O questionnaire (in the form provided by the secretary of the Corporation at the request of the nominating stockholder) containing information regarding the nominees background and qualifications and such other information as may reasonably be required by the Corporation to determine the eligibility of such proposed nominee to serve as a director of the Corporation or to serve as an independent director of the Corporation, (2) a written representation that, unless previously disclosed to the Corporation, the nominee is not and will not become a party to any voting agreement, arrangement or understanding with any person or entity as to how such nominee, if elected as a director, will vote on any issue or that could interfere with such persons ability to comply, if elected as a director, with his/her fiduciary duties under applicable law, (3) a written representation and agreement that, unless previously disclosed to the Corporation pursuant to Section 2.10(a)(iii)(A)(2), the nominee is not and will not become a party to any Third-Party Compensation Arrangement and (4) a written representation that, if elected as a director, such nominee would be in compliance and will continue to comply with the Corporations corporate governance guidelines as disclosed on the Corporations website, as amended from time to time. At the request of the Board of Directors, any person nominated by the Board of Directors for election as a director shall furnish to the Secretary of the Corporation the information that is required to be set forth in a stockholders notice of nomination that pertains to the nominee.
(ii) No person shall be eligible to be nominated by a stockholder to serve as a director of the Corporation unless nominated in accordance with the procedures set forth in this Section 2.10. No business proposed by a stockholder shall be conducted at a stockholder meeting except in accordance with this Section 2.10.
(iii) The chairperson of the meeting shall, if the facts warrant, determine and declare to the meeting that a nomination was not made in accordance with the procedures prescribed by these Bylaws or that business was not properly brought before the meeting, and if he/she should so determine, he/she shall so declare to the meeting and the defective nomination shall be disregarded or such business shall not be transacted, as the case may be. Notwithstanding the foregoing provisions of this Section 2.10, unless otherwise required by law, if the stockholder (or a qualified representative of the stockholder) does not appear at the annual or special meeting of stockholders of the Corporation to present a nomination or other proposed business, such nomination shall be disregarded or such proposed business shall not be transacted, as the case may be, notwithstanding that proxies in respect of such vote may have been received by the Corporation and counted for purposes of determining a quorum. For purposes of this Section 2.10, to be considered a qualified representative of the stockholder, a person must be a duly authorized officer, manager or partner of such stockholder or must be authorized by a writing executed by such stockholder or an electronic transmission delivered by such stockholder to act for such stockholder as proxy at the meeting of stockholders and such person must produce such writing or electronic transmission, or a reliable reproduction of the writing or electronic transmission, at the meeting of stockholders.
C-5
Table of Contents
(iv) Without limiting the foregoing provisions of this Section 2.10, a stockholder shall also comply with all applicable requirements of the Exchange Act with respect to the matters set forth in this Section 2.10; provided, however, that any references in these Bylaws to the Exchange Act are not intended to and shall not limit any requirements applicable to nominations or proposals as to any other business to be considered pursuant to this Section 2.10, and compliance with 2.10(a)(i)(C) and 2.10(b) shall be the exclusive means for a stockholder to make nominations or submit other business (other than as provided in Section 2.10(c)(v)).
(v) Notwithstanding anything to the contrary, the notice requirements set forth herein with respect to the proposal of any business pursuant to this Section 2.10 shall be deemed satisfied by a stockholder if such stockholder has submitted a proposal to the Corporation in compliance with Rule 14a-8 under the Exchange Act, and such stockholders proposal has been included in a proxy statement that has been prepared by the Corporation to solicit proxies for the meeting of stockholders.
ARTICLE 3
DIRECTORS
Section 3.01. General Powers. Except as otherwise provided in the DGCL or the Certificate of Incorporation, the business and affairs of the Corporation shall be managed by or under the direction of the Board of Directors.
Section 3.02. Number, Election and Term Of Office. Subject to the Certificate of Incorporation, the number of directors which shall constitute the whole Board of Directors shall be fixed from time to time solely by resolution adopted by a majority of the Board of Directors. As set forth in Article 6 of the Certificate of Incorporation, each director shall be elected annually by the stockholders. Except as otherwise provided in the Certificate of Incorporation, each director shall serve for a term ending on the date of the annual meeting of stockholders next following the annual meeting at which such director was elected. Notwithstanding the foregoing, each director shall hold office until such directors successor shall have been duly elected and qualified or until such directors earlier death, resignation or removal. Directors need not be stockholders.
Section 3.03. Quorum and Manner of Acting. Unless the Certificate of Incorporation or these Bylaws require a greater number, a majority of the Board of Directors shall constitute a quorum for the transaction of business at any meeting of the Board of Directors and, except as otherwise expressly required by law or by the Certificate of Incorporation, the act of a majority of the directors present at a meeting at which a quorum is present shall be the act of the Board of Directors. When a meeting is adjourned to another time or place (whether or not a quorum is present), notice need not be given of the adjourned meeting if the time and place thereof are announced at the meeting at which the adjournment is taken. At the adjourned meeting, the Board of Directors may transact any business which might have been transacted at the original meeting. If a quorum shall not be present at any meeting of the Board of Directors, the directors present thereat shall adjourn the meeting, from time to time, without notice other than announcement at the meeting, until a quorum shall be present.
Section 3.04. Time and Place of Meetings. The Board of Directors shall hold its meetings at such place, either within or without the State of Delaware, and at such time as may be determined from time to time by the Board of Directors (or the chairperson of the Board of Directors in the absence of a determination by the Board of Directors).
Section 3.05. Annual Meeting. The Board of Directors shall meet for the purpose of organization, the election of officers and the transaction of other business, as soon as practicable after each annual meeting of stockholders, on the same day and at the same place where such annual meeting shall be held. Notice of such meeting need not be given. In the event such annual meeting is not so held, the annual meeting of the Board of Directors may be held at such place either within or without the State of Delaware, on such date and at such time as shall be specified in a notice thereof given as hereinafter provided in Section 3.07 herein or in a waiver of notice thereof signed by any director who chooses to waive the requirement of notice.
C-6
Table of Contents
Section 3.06. Regular Meetings. After the place and time of regular meetings of the Board of Directors shall have been determined and notice thereof shall have been once given to each member of the Board of Directors, regular meetings may be held without further notice being given.
Section 3.07. Special Meetings. Special meetings of the Board of Directors may be called by the chairperson of the Board of Directors, the Chief Executive Officer or on the written request of a majority of the Board of Directors. Notice of special meetings of the Board of Directors shall be given to each director at least 48 hours before the date of the meeting in such manner as is determined by the Board of Directors.
Section 3.08. Committees. The Board of Directors may designate one or more committees, each committee to consist of one or more of the directors of the Corporation. The Board of Directors may designate one or more directors as alternate members of any committee, who may replace any absent or disqualified member at any meeting of the committee. In the absence or disqualification of a member of a committee, the member or members present at any meeting and not disqualified from voting, whether or not such member or members constitute a quorum, may unanimously appoint another member of the Board of Directors to act at the meeting in the place of any such absent or disqualified member. Any such committee, to the extent provided in the resolution of the Board of Directors, shall have and may exercise all the powers and authority of the Board of Directors in the management of the business and affairs of the Corporation, and may authorize the seal of the Corporation to be affixed to all papers which may require it; but no such committee shall have the power or authority in reference to the following matters: (a) approving or adopting, or recommending to the stockholders, any action or matter expressly required by the DGCL to be submitted to the stockholders for approval or (b) adopting, amending or repealing any Bylaw of the Corporation. Each committee shall keep regular minutes of its meetings and report the same to the Board of Directors when required.
Section 3.09. Action by Consent. Unless otherwise restricted by the Certificate of Incorporation or these Bylaws, any action required or permitted to be taken at any meeting of the Board of Directors or of any committee thereof may be taken without a meeting, if all members of the Board of Directors or committee, as the case may be, consent thereto in writing or by electronic transmission, and the writing or writings or electronic transmission or transmissions, are filed with the minutes of proceedings of the Board of Directors or committee. Such filing shall be in paper form if the minutes are maintained in paper form and shall be in electronic form if the minutes are maintained in electronic form.
Section 3.10. Telephonic Meetings. Unless otherwise restricted by the Certificate of Incorporation or these Bylaws, members of the Board of Directors, or any committee designated by the Board of Directors, may participate in a meeting of the Board of Directors, or such committee, as the case may be, by means of conference telephone or other communications equipment by means of which all persons participating in the meeting can hear each other, and such participation in a meeting shall constitute presence in person at the meeting.
Section 3.11. Resignation. Any director may resign from the Board of Directors at any time by giving notice to the Board of Directors or to the Secretary of the Corporation in writing or by electronic transmission. The resignation of any director shall take effect upon receipt of notice thereof or at such later time as shall be specified in such notice; and unless otherwise specified therein, the acceptance of such resignation shall not be necessary to make it effective.
Section 3.12. Vacancies. Unless otherwise provided in the Certificate of Incorporation, vacancies on the Board of Directors resulting from death, resignation, removal or otherwise and newly created directorships resulting from any increase in the number of directors shall, except as otherwise required by law, be filled solely by a majority of the directors then in office (although less than a quorum) or by the sole remaining director, and each director so elected shall hold office until his or her successor is elected and qualified, or until his or her earlier death, resignation or removal. If there are no directors in office, then an election of directors may be held in accordance with the DGCL. Unless otherwise provided in the Certificate of Incorporation, when one or more
C-7
Table of Contents
directors shall resign from the Board of Directors, effective at a future date, a majority of the directors then in office, including those who have so resigned, shall have the power to fill such vacancy or vacancies, the vote thereon to take effect when such resignation or resignations shall become effective, and each director so chosen shall hold office as provided in the filling of the other vacancies.
Section 3.13. Removal. Any director or the entire Board of Directors may be removed, with or without cause, at any time by the affirmative vote of the holders of a majority of the outstanding capital stock of the Corporation then entitled to vote at any election of directors and the vacancies thus created may be filled in accordance with Section 3.12 herein.
Section 3.14. Compensation. Unless otherwise restricted by the Certificate of Incorporation or these Bylaws, the Board of Directors shall have authority to fix the compensation of directors, including fees and reimbursement of expenses.
Section 3.15. Preferred Stock Directors. Notwithstanding anything else contained herein, whenever the holders of one or more classes or series of preferred stock shall have the right, voting separately as a class or series, to elect directors, the election, term of office, filling of vacancies, removal and other features of such directorships shall be governed by the terms of the resolutions applicable thereto adopted by the Board of Directors pursuant to the Certificate of Incorporation, and such directors so elected shall not be subject to the provisions of Sections 3.02, 3.12 and 3.13 of this Article 3 unless otherwise provided therein.
ARTICLE 4
OFFICERS
Section 4.01. Officers. The officers of the Corporation shall be a Chief Executive Officer, a Chief Financial Officer, one or more Vice Presidents, a Treasurer and a Secretary, who shall have the duty, among other things, to record the proceedings of the meetings of stockholders and directors in a book kept for that purpose. The Corporation may also have such other officers, including one or more Controllers, Assistant Controllers, Assistant Treasurers or Assistant Secretaries. One person may hold the offices and perform the duties of any two or more of said offices, except that no one person shall hold the offices and perform the duties of Chief Executive Officer and Secretary.
Section 4.02. Appointment, Term of Office and Remuneration. The officers of the Corporation shall be appointed by, and the remuneration of the officers of the Corporation shall be fixed by, the Board of Directors or any other officer authorized by the Board of Directors; provided, however, the ability of any officer to appoint officers of the Corporation, specify the duties thereof or fix the compensation thereof may be limited or restricted by a resolution of the Board of Directors. Each such officer shall hold office until such officers successor is appointed, or until such officers earlier death, resignation or removal. Any vacancy in any office shall be filled in such manner as the Board of Directors or any officer authorized by the Board of Directors shall determine.
Section 4.03. Removal. Any officer may be removed, with or without cause, at any time, by resolution adopted by the Board of Directors or by any officer authorized by the Board of Directors; provided, however, the ability of any officer to remove officers of the Corporation may be limited or restricted by a resolution of the Board of Directors.
Section 4.04. Resignations. Any officer may resign at any time by giving notice to the Board of Directors or Chief Executive Officer (or to an officer if the Board of Directors has delegated to such officer the power to appoint and to remove such officer) in writing or by electronic transmission. The resignation of any officer shall take effect upon receipt of notice thereof or at such later time as shall be specified in such notice; and unless otherwise specified therein, the acceptance of such resignation shall not be necessary to make it effective.
C-8
Table of Contents
Section 4.05. Powers and Duties. The officers of the Corporation shall have such powers and perform such duties incident to each of their respective offices and such other duties as may from time to time be conferred upon or assigned to them by the Board of Directors.
ARTICLE 5
CAPITAL STOCK
Section 5.01. Uncertificated Shares. The shares of the Corporation shall be uncertificated, provided that the Board of Directors may provide by resolution or resolutions that some or all of any or all classes or series of its stock shall be represented by certificates or a combination of certificated and uncertificated shares. Any such resolution that shares of a class or series will only be uncertificated shall not apply to shares represented by a certificate until such certificate is surrendered to the Corporation. Except as otherwise required by law, the rights and obligations of the holders of uncertificated shares and the rights and obligations of the holders of shares represented by certificates of the same class and series shall be identical. Every holder of stock represented by certificates shall be entitled to have a certificate signed by any two officers of the Corporation, such officers to be designated by the Corporation in its sole discretion. All officers of the Corporation are authorized by these Bylaws to provide such signature. Any or all of the signatures on the certificate may be a facsimile. In case any officer, transfer agent or registrar who has signed or whose facsimile signature has been placed upon a certificate shall have ceased to be such officer, transfer agent or registrar before such certificate is issued, it may be issued by the Corporation with the same effect as if such person were such officer, transfer agent or registrar at the date of issue. A Corporation shall not have power to issue a certificate in bearer form.
Section 5.02. Transfer of Shares. Shares of the stock of the Corporation may be transferred on the record of stockholders of the Corporation by the holder thereof or by such holders duly authorized attorney upon surrender of a certificate therefor properly endorsed or upon receipt of proper transfer instructions from the registered holder of uncertificated shares or by such holders duly authorized attorney and upon compliance with appropriate procedures for transferring shares in uncertificated form, unless waived by the Corporation.
Section 5.03. Authority for Additional Rules Regarding Transfer. The Board of Directors shall have the power and authority to make all such rules and regulations as they may deem expedient concerning the issue, transfer and registration of certificated or uncertificated shares of the stock of the Corporation, as well as for the issuance of new certificates in lieu of those which may be lost or destroyed, and may require of any stockholder requesting replacement of lost or destroyed certificates, bond in such amount and in such form as they may deem expedient to indemnify the Corporation, and/or the transfer agents, and/or the registrars of its stock against any claims arising in connection therewith.
ARTICLE 6
GENERAL PROVISIONS
Section 6.01. Fixing the Record Date. (a) In order that the Corporation may determine the stockholders entitled to notice of any meeting of stockholders or any adjournment thereof, the Board of Directors may fix a record date, which record date shall not precede the date upon which the resolution fixing such record date is adopted by the Board of Directors, and which record date shall not be more than 60 nor less than 10 days before the date of such meeting. If the Board of Directors so fixes a date, such date shall also be the record date for determining the stockholders entitled to vote at such meeting unless the Board of Directors determines, at the time it fixes such record date, that a later date on or before the date of the meeting shall be the date for making such determination. If no record date is fixed by the Board of Directors, the record date for determining stockholders entitled to notice of or to vote at a meeting of stockholders shall be at the close of business on the day next preceding the day on which notice is given, or, if notice is waived, at the close of business on the day next preceding the day on which the meeting is held. A determination of stockholders of record entitled to notice
C-9
Table of Contents
of or to vote at a meeting of stockholders shall apply to any adjournment of the meeting; provided that the Board of Directors may in its discretion or as required by law fix a new record date for determination of stockholders entitled to vote at the adjourned meeting, and in such case shall fix the same date or an earlier date as the record date for stockholders entitled to notice of such adjourned meeting.
(b) In order that the Corporation may determine the stockholders entitled to receive payment of any dividend or other distribution or allotment of any rights or the stockholders entitled to exercise any rights in respect of any change, conversion or exchange of stock, or for the purpose of any other lawful action, the Board of Directors may fix a record date, which record date shall not precede the date upon which the resolution fixing the record date is adopted, and which record date shall be not more than 60 days prior to such action. If no record date is fixed, the record date for determining stockholders for any such purpose shall be at the close of business on the day on which the Board of Directors adopts the resolution relating thereto.
Section 6.02. Dividends. Subject to limitations contained in the DGCL and the Certificate of Incorporation, the Board of Directors may declare and pay dividends upon the shares of capital stock of the Corporation, which dividends may be paid either in cash, in property or in shares of the capital stock of the Corporation.
Section 6.03. Year. The fiscal year of the Corporation shall commence on July 1 and end on June 30 of each year.
Section 6.04. Corporate Seal. The corporate seal shall have inscribed thereon the name of the Corporation, the year of its organization and the words Corporate Seal, Delaware. The seal may be used by causing it or a facsimile thereof to be impressed, affixed or otherwise reproduced.
Section 6.05. Voting of Stock Owned by the Corporation. The Board of Directors may authorize any person, on behalf of the Corporation, to attend, vote at and grant proxies to be used at any meeting of stockholders of any corporation (except this Corporation) in which the Corporation may hold stock.
Section 6.06. Amendments. These Bylaws or any of them may be altered, amended or repealed, or new Bylaws may be made, by the stockholders entitled to vote thereon at any annual or special meeting thereof or by the Board of Directors. Unless a higher percentage is required by the Certificate of Incorporation as to any matter that is the subject of these Bylaws, all such amendments must be approved by the affirmative vote of the holders of not less than 66 2/3% of the total voting power of all outstanding securities of the Corporation generally entitled to vote in the election of directors, voting together as a single class, or by a majority of the Board of Directors.
C-10
Table of Contents
EXECUTION VERSION
June 17, 2021
GS Acquisition Holdings Corp II
200 West Street
New York, New York 10282
Re: Sponsor Agreement
Ladies and Gentlemen:
This letter (this Letter Agreement) is being delivered to you in connection with the Business Combination Agreement (together with the exhibits and schedules thereto, as amended, supplemented, otherwise modified, the Business Combination Agreement), dated as of the date hereof, by and among GS Acquisition Holdings Corp II, a Delaware corporation (the SPAC), Mirion Technologies (TopCo), Ltd., a Jersey private company (the Company), CCP IX LP No. 1, CCP IX LP No. 2, CCP IX Co-Investment LP and CCP IX Co-Investment No. 2 LP (collectively, the Charterhouse Parties), each of the other persons set forth on Annex I thereto and the other holders of Existing Company Shares from time to time becoming a party thereto by executing a Joinder Agreement in the form of Exhibit H thereto (collectively, together with each Charterhouse Party, the Sellers), and hereby amends and restates in its entirety that certain letter, dated June 29, 2020, from GS Sponsor II LLC, a Delaware limited liability company (the Sponsor), and the other undersigned persons (each such other undersigned person, an Insider and collectively, the Insiders) to the SPAC (the Prior Letter Agreement). Capitalized terms used but not otherwise defined herein shall have the respective meanings ascribed to such terms in the Business Combination Agreement. Certain capitalized terms used herein are defined in paragraph 9 hereof.
In order to induce the Company, the Sellers and the SPAC to enter into the Business Combination Agreement and for other good and valuable consideration, the receipt and sufficiency of which are hereby acknowledged, Sponsor and each of the Insiders hereby severally (and not jointly and severally) agrees with the SPAC, and at all times prior to any valid termination of the Business Combination Agreement, the Company and the Charterhouse Parties, as follows:
1. The Sponsor and each Insider hereby unconditionally and irrevocably agrees: (i) that at any duly called meeting of the stockholders of the SPAC (or any adjournment or postponement thereof), and in any action by written consent of the stockholders of the SPAC requested by the SPACs board of directors or to be undertaken as contemplated by the Transactions, the Sponsor and each such Insider shall, if a meeting is held, appear at the meeting, in person or by proxy, or otherwise cause all of its Shares to be counted as present thereat for purposes of establishing a quorum, and it shall vote or consent (or cause to be voted or consented), in person, in writing or by proxy, all of its Shares (a) in favor of the adoption of the Business Combination Agreement and approval of the Transactions and all other Transaction Proposals (and any actions required in furtherance thereof), (b) against any action, proposal, transaction or agreement that would result in a breach of any representation, warranty, covenant, obligation or agreement of the SPAC contained in the Business Combination Agreement, (c) in favor of any other proposals set forth in the SPACs proxy statement to be filed by the SPAC with the U.S. Securities and Exchange Commission (the Commission) relating to the Transactions (including any proxy supplements thereto, the Proxy Statement), (d) for any proposal to adjourn or postpone the applicable stockholder meeting to a later date if (and only if) (1) there are not sufficient votes for approval of the Business Combination Agreement and any other proposals related thereto, as set forth in the Proxy Statement, on the dates on which such meetings are held or (2) the Closing condition in Section 11.03(d) of the Business Combination Agreement has not been satisfied, and (e) against the following actions or proposals: (1) any Business Combination Proposal or any other proposal in opposition to approval of the Business Combination Agreement or in competition with
D-1
Table of Contents
the Business Combination Agreement; and (2) (A) any change in the present capitalization of the SPAC or any amendment of the SPACs Charter (as defined below), except to the extent expressly contemplated by the Business Combination Agreement, (B) any liquidation, dissolution or other change in the SPACs corporate structure or business, (C) any action, proposal, transaction or agreement that would result in a breach in any material respect of any covenant, representation or warranty or other obligation or agreement of the Sponsor or such Insider under this Letter Agreement, and (D) any other action or proposal involving the SPAC or any of its subsidiaries that is intended or would reasonably be expected to prevent, delay or impede the timely consummation of the Transactions and (ii) not to redeem, elect to redeem or tender or submit for redemption, or knowingly cause any other Person to do any such thing on its behalf, any Shares owned by it in connection with such stockholder approval or proposed Business Combination, or in connection with any vote to amend the SPACs Charter or otherwise in connection with the Transactions. Prior to any valid termination of the Business Combination Agreement, the Sponsor and each Insider shall be bound by and comply with Sections 8.08 (Exclusivity) and 8.06 (Public Announcements) of the Business Combination Agreement (and any relevant definitions contained in any such Sections), which Sections apply mutatis mutandis, as if such Person were a signatory to the Business Combination Agreement with respect to such provisions. The obligations of the Sponsor and the Insiders specified in this paragraph 1 shall apply whether or not the Transactions or any action described above is recommended by the SPACs board of directors or any such recommendation changes while this Letter Agreement remains in force.
The Sponsor and each Insider hereby agrees that in the event that the SPAC fails to consummate a Business Combination by June 29, 2022, or such later period approved by the SPACs stockholders in accordance with the SPACs amended and restated certificate of incorporation (the Charter), the Sponsor and each Insider shall take all reasonable steps to cause the SPAC to (i) cease all operations except for the purpose of winding up, (ii) as promptly as reasonably possible but not more than ten (10) Business Days thereafter, subject to lawfully available funds therefor, redeem 100% of the Class A Common Shares sold as part of the Units in the IPO (the Offering Shares), at a per share price, payable in cash, equal to the aggregate amount then on deposit in the Trust Account, including interest (which interest shall be net of taxes payable) and less up to $100,000 of interest to pay dissolution expenses, divided by the number of then outstanding Offering Shares, which redemption will completely extinguish all Public Stockholders rights as stockholders (including the right to receive further liquidation distributions, if any), subject to applicable law, and (iii) as promptly as reasonably possible following such redemption, subject to the approval of the SPACs remaining stockholders and the SPACs board of directors, dissolve and liquidate, subject in each case to the SPACs obligations under Delaware law to provide for claims of creditors and the other requirements of applicable law. The Sponsor and each Insider agrees to not propose any amendment to the SPACs Charter that would modify the substance or timing of the SPACs obligation to redeem 100% of the Offering Shares if the SPAC does not complete a Business Combination within the required time periods set forth in the Charter, or with respect to any other material provisions relating to stockholders rights or pre- initial Business Combination activity, unless the SPAC provides its Public Stockholders with the opportunity to redeem their Offering Shares upon approval of any such amendment at a per share price, payable in cash, equal to the aggregate amount then on deposit in the Trust Account, including interest (which interest shall be net of taxes payable), divided by the number of then outstanding Offering Shares.
The Sponsor and each Insider agrees and acknowledges that, with respect to the Founder Shares held by it, it has no right, title, interest or claim of any kind in or to any monies held in the Trust Account or any other asset of the SPAC as a result of any liquidation of the SPAC. The Sponsor and each Insider hereby further waives, with respect to any Shares held by it, any redemption rights, if any, it may have in connection with (x) the consummation of a Business Combination, including, without limitation, any such rights available in the context of a stockholder vote to approve such Business Combination or in the context of a tender offer made by the SPAC to purchase Class A Common Shares and (y) a stockholder vote to approve an amendment to the Charter (A) to modify the substance or timing of the SPACs obligation to allow redemptions in connection with the SPACs initial Business Combination or to redeem 100% of the Offering Shares if the SPAC has not consummated its initial Business Combination within 24 months from the closing of the IPO or (B) with respect to any other provision relating to stockholders rights or pre-initial Business Combination activity (although the
D-2
Table of Contents
Sponsor and the Insiders and their respective Affiliates shall be entitled to redemption and liquidation rights with respect to any Offering Shares it or they hold if the SPAC fails to consummate a Business Combination within the time period set forth in the Charter).
2. Without limiting their obligations under paragraph 5 below, during the period commencing on the date hereof and ending on the earlier of (a) the valid termination of the Business Combination Agreement or (b) the Closing, the Sponsor and each Insider shall not, without the prior written consent of the SPAC, Transfer any Units, Shares, warrants to purchase Class A Common Shares (Warrants) or any securities convertible into, or exercisable, or exchangeable for, Class A Common Shares owned by it. In the event that (i) any Class A Common Shares, Warrants or other equity securities of the SPAC are issued to the Sponsor or any Insider after the date hereof pursuant to any stock dividend, stock split, recapitalization, reclassification, combination or exchange of Shares or Warrants, on or affecting the Shares or Warrants owned by the Sponsor or any Insider or otherwise, (ii) the Sponsor or any Insider purchases or otherwise acquires beneficial ownership of any Shares, Warrants or other equity securities of the SPAC after the date hereof or (iii) the Sponsor or any Insider acquires the right to vote, direct the voting of or share in the voting of any Shares, Warrants or other equity securities of the SPAC after the date hereof (such Shares, Warrants or other equity securities of the SPAC described in clauses (i), (ii) and (iii), the New Shares), then such New Shares acquired or purchased by the Sponsor or any Insider shall be subject to the terms of this paragraph 2 and paragraph 1 above to the same extent as if they constituted the Shares or Warrants owned by the Sponsor or any Insider as of the date hereof.
3. In the event of the liquidation of the Trust Account, the Sponsor (which for purposes of clarification shall not extend to any other stockholders, members or managers of the Sponsor or any other Insider) agrees to indemnify and hold harmless the SPAC against any and all loss, liability, claim, damage and expense whatsoever (including, but not limited to, any and all legal or other expenses reasonably incurred in investigating, preparing or defending against any litigation, whether pending or threatened, or any claim whatsoever) to which the SPAC may become subject as a result of any claim by (i) any third party (other than the SPACs independent registered public accounting firm) for services rendered or products sold to the SPAC or (ii) a prospective target business with which the SPAC has entered into a letter of intent, confidentiality or similar agreement for a Business Combination (a Target); provided, however, that such indemnification of the SPAC by the Sponsor shall apply only to the extent necessary to ensure that such claims by a third party for services rendered (other than the SPACs independent registered public accounting firm) or a Target do not reduce the amount of funds in the Trust Account to below (i) $10.00 per Offering Share or (ii) such lesser amount per Offering Share held in the Trust Account as of the date of the liquidation of the Trust Account due to reductions in the value of the trust assets as of the date of the liquidation of the Trust Account, in each case, net of the amounts of interest which may be withdrawn to pay taxes. For the avoidance of doubt, the Sponsor shall not be required to indemnify the SPAC pursuant to this paragraph 3 in the event of (i) any claims by a third party who executed a waiver of any and all rights to seek access to the Trust Account, and (ii) any claims under the SPACs indemnity of Goldman Sachs & Co. LLC and Citigroup Global Markets Inc., as the representatives of the several underwriters (each an Underwriter and collectively, the Underwriters) named in the underwriting agreement between the SPAC and the Underwriters dated as of June 29, 2020, against certain liabilities, including liabilities under the Securities Act of 1933, as amended (the Securities Act). In the event that any such executed waiver is deemed to be unenforceable against such third party, the Sponsor shall not be responsible to the extent of any liability for such third party claims. The Sponsor shall have the right to defend against any such claim with counsel of its choice reasonably satisfactory to the SPAC if, within 15 days following written receipt of notice of the claim to the Sponsor, the Sponsor notifies the SPAC in writing that it shall undertake such defense. For the avoidance of doubt, none of the SPACs officers or directors will indemnify the SPAC for claims by third parties, including, without limitation, claims by third party vendors and Targets.
4. The Sponsor and each Insider hereby agrees and acknowledges that: (i) the Underwriters, the SPAC, and, prior to any valid termination of the Business Combination Agreement, the Company and the Charterhouse Parties, would be irreparably injured in the event of a breach by such Sponsor or Insider of its obligations under this Letter Agreement (with respect to the Underwriters, only such provisions as were contained in the Prior
D-3
Table of Contents
Letter Agreement) (ii) monetary damages may not be an adequate remedy for such breach and (iii) the non-breaching party shall be entitled to seek injunctive relief, including to prevent breaches or threatened breaches of this Letter Agreement and to specifically enforce its terms, in addition to any other remedy that such party may have in law or in equity in the event of such breach.
5. (a) In the event that the Closing does not occur for any reason (including, without limitation, as a result of the valid termination of the Business Combination Agreement), the Sponsor and each Insider agrees that it shall not Transfer any Founder Shares (or Class A Common Shares issuable upon conversion thereof) until the earlier of (A) one year after the completion of the SPACs initial Business Combination and (B) subsequent to the completion of the SPACs initial Business Combination, (x) the date on which the SPAC completes a liquidation, merger, stock exchange, reorganization or other similar transaction that results in all of the Public Stockholders having the right to exchange their Class A Common Shares for cash, securities or other property or (y) if the last reported sale price of the Class A Common Shares equals or exceeds $12.00 per share (as adjusted for stock splits, stock dividends, reorganizations, recapitalizations and the like) for any 20 trading days within any 30-day trading period commencing at least 150 days after the SPACs initial Business Combination (the Standalone Founder Shares Lock-up Period).
(b) In the event that the Closing does occur, the Sponsor and each Insider agrees that it shall not Transfer, or knowingly cause any other Person to Transfer on its behalf (A) any Founder Shares it owns or otherwise has a beneficial interest in or controls as at the time immediately prior to Closing until the earlier of (i) the one year anniversary of the Closing Date and (ii) the day following the trading date when the last reported sale price of the Class A Common Shares first equals or exceeds $12.00 per share (as adjusted for stock splits, stock dividends, reorganizations, recapitalizations and the like) for any 20 trading days within any 30-day trading period commencing at least 150 days after the Closing Date, subject to the clear market provisions in the Registration Rights Agreement and (B) any shares issued pursuant to the Sponsor PIPE Commitment if retained by the Sponsor or its Affiliates (but, for the avoidance of doubt, not if distributed to its employees and investment partners) for a period of 180 days after the Closing Date, subject to the clear market provisions in the Registration Rights Agreement, (such periods, the Business Combination Agreement Lock-Up Periods and, together with the Standalone Founder Shares Lock-Up Period, the Lock-Up Periods).
(c) Notwithstanding the provisions set forth in paragraphs 2 and 5(a) and (b), but subject to the provisions set forth in paragraph 5(d), upon the valid termination of the Business Combination Agreement, the following Transfers of the Founder Shares that are held by the Sponsor, any Insider or any of their permitted transferees (that have complied with this paragraph 5(c)), are permitted (a) to the SPACs officers or directors, any affiliates or family members of any of the SPACs officers or directors, any members of the Sponsor, or any affiliates of the Sponsor or any employee or partner of any such affiliate, (b) in the case of an individual, transfers by gift to a member of the individuals immediate family or to a trust, the beneficiary of which is a member of the individuals immediate family or an affiliate of such person, or to a charitable organization; (c) in the case of an individual, transfers by virtue of laws of descent and distribution upon death of the individual; (d) in the case of an individual, transfers pursuant to a qualified domestic relations order; (e) transfers by private sales or transfers made in connection with the consummation of the SPACs Business Combination at prices no greater than the price at which the securities were originally purchased; (f) transfers in the event of the SPACs liquidation prior to the SPACs completion of an initial Business Combination; (g) transfers by virtue of the laws of the State of Delaware or the Sponsors limited liability company agreement, as amended, upon dissolution of the Sponsor; (h) in the event of the SPACs completion of a liquidation, merger, stock exchange, reorganization or other similar transaction which results in all of the Public Stockholders having the right to exchange their Class A Common Shares for cash, securities or other property subsequent to the SPACs completion of an initial Business Combination; or (i) to a nominee or custodian of a person or entity to whom a disposition or transfer would be permissible under clauses (a) through (h) above; provided, however, that in the case of clauses (a) through (e), these permitted transferees must enter into a written agreement with the SPAC agreeing to be bound by the transfer restrictions and other applicable restrictions in this Letter Agreement.
D-4
Table of Contents
(d) Vesting Provisions. The Sponsor and GS Acquisition Holdings II Employee Participation LLC agree that all of the Founder Shares as of the Closing (the Vesting Shares) shall be subject to the vesting and forfeiture provisions set forth in this paragraph 5(d). The Sponsor and GS Acquisition Holdings II Employee Participation LLC each agrees that it shall not Transfer, or knowingly cause any other Person to Transfer on its behalf, any unvested Founder Shares prior to the later of (x) the expiration of the Business Combination Agreement Lock-up Period and (y) the date such Founder Shares become vested pursuant to this paragraph 5(d).
(i) Vesting of Founder Shares.
(1) 33-1/3% of the Founder Shares shall vest at such time as a Stock Price Level equal to $12.00 (the First Vesting Price) is achieved on or before the date that is five years after the Closing Date.
(2) 33-1/3% of the Founder Shares shall vest at such time as a Stock Price Level equal to $14.00 (the Second Vesting Price) is achieved on or before the date that is five years after the Closing Date.
(3) 33-1/3% of the Founder Shares shall vest at such time as a Stock Price Level equal to $16.00 (the Third Vesting Price) is achieved on or before the date that is five years after the Closing Date.
(4) Founder Shares that do not vest in accordance with this paragraph 5(d)(i) on or before the date that is five years after the Closing Date will be forfeited immediately following the five-year anniversary of the Closing Date.
(ii) Acceleration of Vesting upon a Sale. In the event of a Sale prior to the fifth anniversary of the Closing Date or the vesting of unvested Founder Shares, vesting shall be accelerated or the unvested Founder Shares will be forfeited, as follows:
(1) With respect to the unvested Founder Shares that were eligible to vest pursuant to paragraph 5(d)(i)(1), as the case may be, if such Sale occurs on or before the date that is five years after the Closing Date, then (i) such Founder Shares will fully vest as of immediately prior to the closing of such Sale only if the per share price of the Class A Common Shares paid or implied in such Sale equals or exceeds the First Vesting Price and (ii) no portion of such Founder Shares or Class A Common Shares will vest in connection with such Sale if the per share price of the Class A Common Shares paid or implied in such Sale is less than the First Vesting Price.
(2) With respect to the unvested Founder Shares that were eligible to vest pursuant to paragraph 5(d)(i)(2), as the case may be, if such Sale occurs on or before the date that is five years after the Closing Date, then (i) such Founder Shares will fully vest as of immediately prior to the closing of such Sale only if the per share price of the Class A Common Shares paid or implied in such Sale equals or exceeds the Second Vesting Price and (ii) no portion of such Founder Shares or Class A Common Shares will vest in connection with such Sale if the per share price of the Class A Common Shares paid or implied in such Sale is less than the Second Vesting Price.
(3) With respect to the unvested Founder Shares that were eligible to vest pursuant to paragraph 5(d)(i)(3), as the case may be, if such Sale occurs on or before the date that is five years after the Closing Date, then (i) such Founder Shares will fully vest as of immediately prior to the closing of such Sale only if the per share price of the Class A Common Shares paid or implied in such Sale equals or exceeds the Third Vesting Price and (ii) no portion of such Founder Shares or Class A Common Shares will vest in connection with such Sale if the per share price of the Class A Common Shares paid or implied in such Sale is less than the Third Vesting Price.
(4) Unvested Founder Shares that do not vest in accordance with this paragraph 5(d)(ii) upon the occurrence of a Sale will be forfeited immediately prior to the closing of such Sale and in accordance with paragraph 5(d)(iii).
D-5
Table of Contents
(5) For purposes of this paragraph 5(d)(ii), Sale means (A) a purchase, sale, exchange, business combination or other transaction (including a merger or consolidation of SPAC with or into any other corporation or other entity) in which the equity securities of SPAC, its successor or the surviving entity of such business combination or other transaction are not registered under the Securities Exchange Act of 1934, as amended (the Exchange Act) or listed or quoted for trading on a national securities exchange or (B) a sale, lease, exchange or other transfer in one transaction or a series of related transactions of all or substantially all of SPACs assets. For avoidance of doubt, following a transaction or business combination that is not a Sale hereunder, including a transaction or business combination in which the equity securities of the surviving entity of such business combination or other transaction are registered under the Exchange Act and listed or quoted for trading on a national securities exchange, the equitable adjustment provisions of paragraph 20 shall apply, including, without limitation, to performance vesting criteria.
(iii) Voting. Holders of Founder Shares subject to the vesting provisions of this paragraph 5(d) shall be entitled to vote such Founder Shares and receive dividends and other distributions with respect to such Founder Shares prior to vesting; provided, that dividends and other distributions with respect to Founder Shares that are subject to vesting pursuant to paragraph 5(d)(i) shall be set aside by the SPAC and shall only be paid to such holders upon the vesting of such Founder Shares; for the avoidance of doubt, (i) such dividends and other distributions shall be paid only on the portion of the unvested Founder Shares that vest and (ii) if any dividends or other distributions with respect to Founder Shares that are subject to vesting pursuant to paragraph 5(d)(i) are set aside and such Founder Shares are subsequently forfeited, such set aside dividends or distributions shall become the property of the SPAC; provided further, that (i) the amount of any dividends and other distributions with respect to the unvested Founder Shares and set aside by SPAC pursuant to this paragraph 5(d)(iii) shall not be reported as taxable income (on IRS Form 1099 or otherwise) to the holders of Founder Shares unless and until such dividends are paid in cash or in kind (which, for the avoidance of doubt, for purposes of this Letter Agreement, shall not include any transaction subject to paragraph 20 hereof), as the case may be and (ii) the parties to this Letter Agreement shall not take any position inconsistent with such reporting except to the extent otherwise required by a determination as defined in Section 1313 of the Code. References in this paragraph 5(d)(iii) to the Code shall include references to any similar or analogous provisions of state or local law.
(iv) Forfeiture. Founder Shares or Class A Common Shares that are forfeited pursuant to paragraph 5(d)(i) or 5(d)(ii) shall be promptly transferred by Sponsor to the SPAC, without any consideration for such Transfer, and cancelled.
(v) Stock Price Level. For purposes of this paragraph 5(d), the applicable Stock Price Level will be considered achieved only when the volume weighted average price per share of Class A Common Shares on the New York Stock Exchange, or such other securities exchange where the Class A Common Shares are listed or quoted, equals or exceeds the applicable threshold for any 20 trading days during a 30 consecutive trading day period. The Stock Price Levels (and the share price levels in a Sale in paragraph 5(d)(ii)) will be equitably adjusted on account of any stock split, reverse stock split or similar equity restructuring transaction.
(e) Waiver of Conversion Ratio Adjustment.
(1) (A) Section 4.3(b)(i) of the Charter provides that each share of Class B Common Stock (as defined therein) shall automatically convert into one Class A Common Share (the Initial Conversion Ratio) at the time of closing of the SPACs initial Business Combination, and (B) Section 4.3(b)(ii) of the Charter provides that the Initial Conversion Ratio shall be adjusted (the Adjustment) in the event that additional Class A Common Shares or equity-linked securities (as defined therein) are issued or deemed issued in excess of the amounts offered in the IPO and in relation to the closing of the initial Business Combination, such that the Sponsor and the Insiders shall continue to own 20% of the issued and outstanding Shares after giving effect to such issuance.
(2) As of, and conditioned upon the Closing, the Sponsor and each Insider hereby irrevocably relinquishes and waives any and all rights the Sponsor and each Insider has or will have under Section 4.3(b)(ii)
D-6
Table of Contents
of the Charter to receive Class A Common Shares in excess of the number issuable at the Initial Conversion Ratio upon conversion of the existing Founder Shares held by it in connection with the Closing as a result of any Adjustment, and, as a result, the Founder Shares shall convert into Class A Common Shares (or such equivalent security) at Closing on a one-for-one basis.
6. The Sponsor and each Insider represents and warrants that it has never been suspended or expelled from membership in any securities or commodities exchange or association or had a securities or commodities license or registration denied, suspended or revoked. Each Insider represents that such Insiders biographical information furnished to the SPAC, if any (including any such information included in the Prospectus), is true and accurate in all respects and does not omit any material information with respect to such Insiders background. The Sponsor and each Insiders questionnaire furnished to the SPAC, if any, is true and accurate in all respects. The Sponsor and each Insider represents and warrants that: it is not subject to or a respondent in any legal action for, any injunction, cease-and-desist order or order or stipulation to desist or refrain from any act or practice relating to the offering of securities in any jurisdiction; it has never been convicted of, or pleaded guilty to, any crime (i) involving fraud, (ii) relating to any financial transaction or handling of funds of another person, or (iii) pertaining to any dealings in any securities and it is not currently a defendant in any such criminal proceeding.
7. Except as disclosed on Schedule 5.17 (Brokers Fees) of the Business Combination Agreement, neither the Sponsor nor any Insider nor any Affiliate of the Sponsor or any Insider, nor any director or officer of the SPAC, shall receive from the SPAC any finders fee, reimbursement, consulting fee, monies in respect of any repayment of a loan or other compensation prior to, or in connection with any services rendered in order to effectuate the consummation of the SPACs initial Business Combination (regardless of the type of transaction that it is).
8. The Sponsor and each Insider has full legal capacity, right, power and organizational authority, without violating any agreement to which it is bound (including, without limitation, any non-competition or non-solicitation agreement) or any documents pursuant to which it is organized, to enter into this Letter Agreement.
9. As used herein, the following terms shall have the respective meanings set forth below:
Affiliate shall mean, with respect to any Person, any other Person directly or indirectly controlling, controlled by or under common control with such Person; provided that (i) neither the Company nor any Subsidiary shall be considered an Affiliate of any Seller (as defined in the Business Combination Agreement) and (ii) in no event shall the SPAC be considered an Affiliate of The Goldman Sachs Group, Inc. or of any investment fund affiliated with The Goldman Sachs Group, Inc., nor shall any portfolio company of any investment fund affiliated with The Goldman Sachs Group, Inc. be considered to be an Affiliate of the SPAC. For purposes of this definition, control (including with correlative meanings, the terms controlling, controlled by and under common control with), as applied to any Person, means the possession, directly or indirectly, of the power to direct or cause the direction of the management and policies of such Person, whether through the ownership of voting securities, by contract or otherwise.
Business Combination shall mean a merger, capital stock exchange, asset acquisition, stock purchase, reorganization or similar business combination, involving the SPAC and one or more businesses.
Class A Common Shares shall mean shares of Class A common stock, par value $0.0001 per share, issued by the SPAC.
Founder Shares shall mean the 18,750,000 shares of Class B common stock, par value $0.0001 per share, issued by the SPAC and owned by the Sponsor and GS Acquisition Holdings II Employee Participation LLC.
Governmental Authority shall mean any domestic or foreign national, state, multi-state, municipal or other local government, any subdivision, agency, commission or authority thereof, or any quasi-
D-7
Table of Contents
governmental or private body exercising any regulatory or taxing authority (to the extent that the rules, regulations or orders of such organization or authority have the force of Law), or any court, tribunal or arbitrator of competent jurisdiction.
Law shall mean any applicable foreign, federal, state, local law, statute, code, ordinance, rule, regulation, order or other legal requirement of any Governmental Authority.
Lien shall mean any lien, encumbrance, pledge, mortgage, deed of trust, security interest, lease, charge, option, right of first refusal or first offer, easement, servitude or other transfer restriction.
Person shall mean any individual, corporation, partnership, limited liability company, firm, joint venture, association, joint-stock company, trust, unincorporated organization, Governmental Authority or other entity.
Private Placement Warrants shall mean the Warrants to purchase up to 8,500,000 Class A Common Shares owned by the Sponsor and GS Acquisition Holdings II Employee Participation LLC.
Public Stockholders shall mean the holders of securities issued in the IPO.
Shares shall mean, collectively, the Class A Common Shares and the Founder Shares.
Subsidiary shall mean, with respect to any Person, any partnership, limited liability company, corporation or other business entity of which: (a) if a corporation, a majority of the total voting power of shares of capital stock entitled (without regard to the occurrence of any contingency) to vote in the election of directors, managers, or trustees thereof is at the time owned or controlled, directly or indirectly, by that Person or one or more of the other Subsidiaries of that Person or a combination thereof; (b) if a partnership, limited liability company or other business entity, a majority of the partnership or other similar ownership interests thereof is at the time owned or controlled, directly or indirectly, by that Person or one or more Subsidiaries of that Person or a combination thereof; or (c) in any case, such Person controls the management thereof.
Transfer shall mean the (a) sale or assignment of, offer to sell, contract or agreement to sell, hypothecate, pledge, grant of any option to purchase or otherwise dispose of or agreement to dispose of, directly or indirectly, or establishment or increase of a put equivalent position or liquidation with respect to or decrease of a call equivalent position within the meaning of Section 16 of the Exchange Act, and the rules and regulations of the Commission promulgated thereunder with respect to, any security, (b) entry into any swap or other arrangement that transfers to another, in whole or in part, any of the economic consequences of ownership of any security, whether any such transaction is to be settled by delivery of such securities, in cash or otherwise, or (c) public announcement of any intention to effect any transaction specified in clause (a) or (b) herein.
Trust Account shall mean the trust fund into which a portion of the net proceeds of the IPO were deposited.
Units has the meaning given in the Prior Letter Agreement.
10. This Letter Agreement and the other agreements referenced herein constitutes the entire agreement and understanding of the parties hereto in respect of the subject matter hereof and supersede all prior understandings, agreements, or representations by or among the parties hereto, written or oral, to the extent they relate in any way to the subject matter hereof or the transactions contemplated hereby, including, without limitation, the Prior Letter Agreement. This Letter Agreement may not be changed, amended, modified or waived (other than to correct a typographical error) as to any particular provision, except by a written instrument executed by all parties hereto, the Company and the Charterhouse Parties, it being acknowledged and agreed that the Companys and the Charterhouse Parties execution of such an instrument will not be required after any valid termination of the Business Combination Agreement.
11. Except as otherwise provided herein, no party hereto may assign either this Letter Agreement or any of its rights, interests, or obligations hereunder without the prior written consent of the other parties, the Company
D-8
Table of Contents
and the Charterhouse Parties (except that, following any valid termination of the Business Combination Agreement, no consent from the Company and the Charterhouse Parties shall be required). Any purported assignment in violation of this paragraph shall be void and ineffectual and shall not operate to transfer or assign any interest or title to the purported assignee. This Letter Agreement shall be binding on the SPAC, the Sponsor and each Insider and their respective successors, heirs and assigns and permitted transferees.
12. Nothing in this Letter Agreement shall be construed to confer upon, or give to, any person or entity other than the parties hereto, the Company and the Charterhouse Parties any right, remedy or claim under or by reason of this Letter Agreement or of any covenant, condition, stipulation, promise or agreement hereof. All covenants, conditions, stipulations, promises and agreements contained in this Letter Agreement shall be for the sole and exclusive benefit of the parties hereto (and, prior to any valid termination of the Business Combination Agreement, the Company and the Charterhouse Parties) and their successors, heirs, personal representatives and assigns and permitted transferees. Notwithstanding anything herein to the contrary, each of the SPAC, the Sponsor and each Insider acknowledges and agrees that, until the valid termination of the Business Combination Agreement, the Company and the Charterhouse Parties is each an express third party beneficiary of this Letter Agreement and may directly enforce (including by an action for specific performance, injunctive relief or other equitable relief) each of the provisions set forth in this Letter Agreement as though directly party hereto. The Sponsor and each Insider understands and acknowledges that the Company and the Charterhouse Parties are entering into the Business Combination Agreement in reliance upon such Sponsors and Insiders execution, delivery and performance of this Letter Agreement.
13. This Letter Agreement may be executed in any number of original or facsimile counterparts and each of such counterparts shall for all purposes be deemed to be an original, and all such counterparts shall together constitute but one and the same instrument.
14. This Letter Agreement shall be deemed severable, and the invalidity or unenforceability of any term or provision hereof shall not affect the validity or enforceability of this Letter Agreement or of any other term or provision hereof. Furthermore, in lieu of any such invalid or unenforceable term or provision, the parties hereto intend that there shall be added as a part of this Letter Agreement a provision as similar in terms to such invalid or unenforceable provision as may be possible and be valid and enforceable.
15. This Letter Agreement shall be governed by and construed in accordance with the law of the State of Delaware, without regard to the conflicts of law rules of such state. Any and all claims or causes of action (each, an Action) based upon, arising out of, or related to this Letter Agreement or the transactions contemplated hereby, shall be brought in the Court of Chancery of the State of Delaware or, if such court does not have jurisdiction, to the Superior Court of the State of Delaware or, if jurisdiction is vested exclusively in federal courts of the United States, the federal courts of the United States sitting in the State of Delaware, so long as one of such courts shall have subject matter jurisdiction over such suit, Action or proceeding, and that any cause of action arising out of this Letter Agreement shall be deemed to have arisen from a transaction of business in the State of Delaware, and each of the parties hereby irrevocably consents to the jurisdiction of such courts (and of the appropriate appellate courts therefrom) in any such suit, Action or proceeding and irrevocably waives, to the fullest extent permitted by law, any objection that it may now or hereafter have to the laying of the venue of any such suit, Action or proceeding in any such court or that any such suit, Action or proceeding brought in any such court has been brought in an inconvenient forum. Process in any such suit, Action or proceeding may be served on any party anywhere in the world, whether within or without the jurisdiction of any such court. EACH OF THE PARTIES HERETO HEREBY IRREVOCABLY WAIVES ANY AND ALL RIGHT TO TRIAL BY JURY IN ANY ACTION OR LEGAL PROCEEDING ARISING OUT OF OR RELATED TO THIS LETTER AGREEMENT OR THE TRANSACTIONS CONTEMPLATED HEREBY.
16. Any notice, consent or request to be given in connection with any of the terms or provisions of this Letter Agreement shall be in writing and shall be sent by express mail or similar private courier service, by certified mail (return receipt requested), by hand delivery or email transmission to the receiving partys address
D-9
Table of Contents
or email address set forth above or on the receiving partys signature page hereto; provided, that any such notice, consent or request to be given to the SPAC, the Charterhouse Parties or the Company at any time prior to the valid termination of the Business Combination Agreement shall be given in accordance with the terms of Section 13.03 (Notices) of the Business Combination Agreement.
17. This Letter Agreement shall terminate and be void and of no further force or effect on the earlier of (i) the latest of (x) the expiration of the applicable Lock-up Period and (y) the vesting in full and delivery of all, or forfeiture and cancellation of all, Vesting Shares, or (ii) the liquidation of the SPAC; provided, however, that paragraph 3 of this Letter Agreement shall survive such liquidation for a period of six years; provided, further, that no such termination shall relieve the Sponsor, any Insider or the SPAC from any liability resulting from a breach of this Letter Agreement occurring prior to such termination.
18. Each party hereto that is also a party to that certain Registration Rights Agreement, dated as of June 29, 2020, by and among SPAC, the Sponsor and the other parties signatory thereto (the Existing Registration Rights Agreement) hereby agrees to terminate the Existing Registration Rights Agreement effective as of the Closing. On or about the date hereof, the Sponsor and each Insider contemplated to become a party to the Amended and Restated Registration Rights Agreement shall deliver to the SPAC such agreement, duly executed by such Person, in the form attached to the Business Combination Agreement.
19. Each of the Sponsor and the Insiders hereby represents and warrants (severally and not jointly as to itself) to the SPAC, the Charterhouse Parties and the Company as follows: (i) it is duly organized, validly existing and, where applicable, in good standing under the laws of the jurisdiction in which it is incorporated, formed, organized or constituted, and the execution, delivery and performance of this Letter Agreement and the consummation of the transactions contemplated hereby are within such Persons corporate, limited liability company or organizational powers and have been duly authorized by all necessary corporate, limited liability company or organizational actions on the part of such Person; (ii) if this Letter Agreement is being executed in a representative or fiduciary capacity, the Person signing this Letter Agreement has full power and authority to enter into this Letter Agreement on behalf of the Sponsor or Insider; (iii) this Letter Agreement has been duly executed and delivered by such Person and, assuming due authorization, execution and delivery by the other parties to this Letter Agreement, this Letter Agreement constitutes a legally valid and binding obligation of such Person, enforceable against such Person in accordance with the terms hereof (except as enforceability may be limited by bankruptcy Laws, other similar Laws affecting creditors rights and general principles of equity affecting the availability of specific performance and other equitable remedies); (iv) the execution and delivery of this Letter Agreement by such Person does not, and the performance by such Person of its obligations hereunder will not, (A) conflict with or result in a violation of the organizational documents of such Person, (B) require any consent or approval that has not been given or other action that has not been taken by any third party (including under any Contract binding upon such Person or such Persons Founder Shares or Private Placement Warrants, as applicable, or in respect of any trust arrangements), in each case, to the extent such consent, approval or other action would prevent, enjoin or materially delay the performance by such Person of its obligations under this Letter Agreement, or (C) result in any violation of any Law having application to the Person or any of its properties, including its Founder Shares or Private Placement Warrants, that would reasonably be expected to have any adverse effect on the legal authority of the Person to enter into and timely perform its obligations under this Letter Agreement; (v) there are no Actions pending against such Person or, to the knowledge of such Person, threatened against such Person, before (or, in the case of threatened Actions, that would be before) any arbitrator or any Governmental Authority, which in any manner challenges or seeks to prevent, enjoin or materially delay the performance by such Person of its obligations under this Letter Agreement; (vi) except for fees described on Schedule 5.17 (Brokers Fees) of the Business Combination Agreement, no financial advisor, investment banker, broker, finder or other similar intermediary is entitled to any fee or commission from such Person, the SPAC, any of its subsidiaries or any of their respective Affiliates in connection with the Business Combination Agreement or this Letter Agreement or any of the respective transactions contemplated thereby and hereby, in each case, based upon any arrangement or agreement made by or, to the knowledge of such Person, on behalf of such Person, for which the SPAC, the Company, the
D-10
Table of Contents
Charterhouse Parties or any of their respective Affiliates would have any obligations or liabilities of any kind or nature; (vii) such Person has had the opportunity to read the Business Combination Agreement and this Letter Agreement and has had the opportunity to consult with its tax and legal advisors; (viii) such Person has not entered into, and shall not enter into, any agreement, arrangement or understanding that would in any way restrict, limit or interfere with the timely performance of such Persons obligations hereunder; (ix) the Founder Shares and Private Placement Warrants are the only equity securities in the SPAC (including, without limitation, any equity securities convertible into, or which can be exercised or exchanged for, equity securities of the SPAC) owned of record or beneficially by such Person as of the date hereof and such Person has the sole power to dispose of (or sole power to cause the disposition of) and the sole power to vote (or sole power to direct the voting of) such Founder Shares and Private Placement Warrants and none of such Founder Shares or Private Placement Warrants is subject to any proxy, voting trust or other agreement or arrangement with respect to the voting of such Founder Shares or Private Placement Warrants, except as provided in this Letter Agreement; (x) such Person is not currently (and at all times through Closing will refrain from being or becoming) a member of a group (within the meaning of Section 13(d)(3) or Section 14(d)(2) of the Exchange Act, or any successor provision), including any group acting for the purpose of acquiring, holding or disposing of equity securities of the SPAC (within the meaning of Rule 13d-5(b)(1) under the Exchange Act) and (xi) except as otherwise described in this Letter Agreement, such Person has the direct or indirect interest in all of its Class A Common Shares, Founder Shares and Private Placement Warrants, which are held through the Sponsor, the Sponsor has good title to all such Founder Shares, Private Placement Warrants and Class A Common Shares held by the Sponsor, and there exist no Liens or any other limitation or restriction (including, without limitation, any restriction on the right to vote, sell or otherwise dispose of such securities (other than transfer restrictions under the Securities Act) affecting any such securities, other than pursuant to (A) this Letter Agreement, (B) the Charter, (C) the Business Combination Agreement, (D) the Existing Registration Rights Agreement, or (E) any applicable securities laws.
20. If, and as often as, there are any changes in the SPAC (or any successor or surviving entity), the Class A Common Shares, the Founder Shares or the Private Placement Warrants by way of stock split, stock dividend, combination or reclassification, or through merger, consolidation, reorganization, recapitalization or business combination, or by any other means, equitable adjustment shall be made to the provisions of this Letter Agreement as may be required so that the rights, privileges, duties and obligations hereunder shall continue with respect to the SPAC, the SPACs successor or the surviving entity of such transaction, the Class A Common Shares, the Founder Shares or the Private Placement Warrants, each as so changed. For the avoidance of doubt, such equitable adjustment shall be made to the performance criteria set forth in paragraph 5(d).
21. Each of the parties hereto agrees to promptly execute and deliver hereafter any further document, agreement or instrument of assignment, transfer or conveyance as may be necessary or desirable to effectuate the purposes hereof and as may be reasonably requested in writing by another party hereto, the Company or the Charterhouse Parties.
22. No failure or delay by a party hereto in exercising any right, power or remedy under this Letter Agreement, and no course of dealing between the parties hereto, shall operate as a waiver of any such right, power or remedy of such party. No single or partial exercise of any right, power or remedy under this Letter Agreement by a party hereto, nor any abandonment or discontinuance of steps to enforce any such right, power or remedy, shall preclude such party from any other or further exercise thereof or the exercise of any other right, power or remedy hereunder. The election of any remedy by a party hereto shall not constitute a waiver of the right of such party to pursue other available remedies. No notice to or demand on a party not expressly required under this Letter Agreement shall entitle the party receiving such notice or demand to any other or further notice or demand in similar or other circumstances or constitute a waiver of the rights of the party giving such notice or demand to any other or further action in any circumstances without such notice or demand.
[Signature Page Follows]
D-11
Table of Contents
| Sincerely, | ||
| GS SPONSOR II LLC | ||
| By: GSAM Holdings LLC, as sole Manager | ||
| By: | /s/ Tom Knott | |
| Name: Tom Knott | ||
| Title: Authorized Signatory | ||
| GSAM HOLDINGS LLC | ||
| By: | /s/ Tom Knott | |
| Name: Tom Knott | ||
| Title: Authorized Signatory | ||
| GS ACQUISITION HOLDINGS II EMPLOYEE PARTICIPATION LLC |
||
| By: GSAM Gen-Par, L.L.C., its manager | ||
| By: | /s/ Raanan A. Agus | |
| Name: Raanan A. Agus | ||
| Title: Vice President | ||
Table of Contents
| Acknowledged and Agreed: | ||
| GS ACQUISITION HOLDINGS CORP II | ||
| By: | /s/ Tom Knott | |
| Name: Tom Knott | ||
| Title: Chief Executive Officer, Chief Financial Officer and Secretary | ||
Table of Contents
Execution Version
SUBSCRIPTION AGREEMENT
This SUBSCRIPTION AGREEMENT (this Subscription Agreement) is entered into this 17th day of June, 2021, by and among GS Acquisition Holdings Corp II, a Delaware corporation (the Issuer), and the entity named on the signature page hereto (Subscriber). Defined terms used but not otherwise defined herein shall have the respective meanings ascribed thereto in the Business Combination Agreement (as defined below).
RECITALS
WHEREAS, the Issuer shall, substantially concurrently with the execution of this Subscription Agreement, enter into a Business Combination Agreement (as it may be amended or supplemented from time to time, the Business Combination Agreement), between the Issuer, Mirion Technologies (TopCo), Ltd., a Jersey private company limited by shares (the Company), CCP IX LP No. 1, CCP IX LP No. 2, CCP IX Co-Investment LP and CCP IX Co-Investment No. 2 LP (collectively, the Charterhouse Parties), each acting by their general partner, Charterhouse General Partners (IX) Limited, and the other Sellers named therein;
WHEREAS, in connection with the transactions contemplated by the Business Combination Agreement (collectively, and together with the Subscription (as defined below) the Transactions), Subscriber desires to subscribe for and purchase from the Issuer that number of shares of Class A common stock, par value $0.0001 per share (the Class A Shares), of the Issuer set forth on the signature page hereto (the Acquired Shares) for a purchase price of $10.00 per share for the aggregate purchase price set forth on the signature page hereto (the Purchase Price), and the Issuer desires to issue and sell to Subscriber the Acquired Shares in consideration of the payment of the Purchase Price therefor by or on behalf of Subscriber to the Issuer, all on the terms and conditions set forth herein; and
WHEREAS, in connection with the Transactions, certain other accredited investors (as defined in Rule 501(a)(1), (2), (3) or (7) under the Securities Act of 1933, as amended (the Securities Act)) (each, an Other Subscriber) have, severally and not jointly, entered into separate subscription agreements with the Issuer (the Other Subscription Agreements), pursuant to which such Other Subscribers have agreed to purchase Class A Shares at the same per share purchase price as the Subscriber, and the aggregate amount of securities to be sold by the Issuer pursuant to this Subscription Agreement and the Other Subscription Agreements equals, as of the date hereof, 90,000,000 Class A Shares (the Offering).
NOW, THEREFORE, in consideration of the foregoing and the mutual representations, warranties and covenants, and subject to the conditions, herein contained, and intending to be legally bound hereby, the parties hereto hereby agree as follows:
For ease of administration, this single Subscription Agreement is being executed so as to enable each Subscriber identified on the signature page to enter into a Subscription Agreement, severally, but not jointly. The parties agree that (i) the Subscription Agreement shall be treated as if it were a separate agreement with respect to each Subscriber listed on the signature page, as if each Subscriber entity had executed a separate Subscription Agreement naming only itself as Subscriber, and (ii) no Subscriber listed on the signature page shall have any liability under the Subscription Agreement for the obligations of any other Subscriber so listed.
1. Subscription. Pursuant to the terms and subject to the conditions set forth herein, at the Closing, Subscriber hereby agrees to subscribe for and purchase from the Issuer, and the Issuer hereby agrees to issue and sell to Subscriber, upon the payment of the Purchase Price, the Acquired Shares (such subscription and issuance, the Subscription).
E-1
Table of Contents
2. Subscription Closing.
(a) The closing of the Subscription contemplated hereby (the Subscription Closing) shall occur on the Closing Date substantially concurrent with the Closing. Not less than five (5) business days prior to the anticipated Closing Date, the Issuer shall provide written notice to Subscriber (such notice, as updated from time to time, the Closing Notice) of such anticipated Closing Date; provided, that the Issuer may, upon at least one (1) business days written notice, delay from time to time the anticipated Closing Date up to ten (10) business days following the anticipated Closing Date identified in the Closing Notice. Subscriber shall deliver to the Issuer at least three (3) business days prior to the then anticipated Closing Date identified in the Closing Notice (unless a later time is otherwise agreed by the Issuer), to be held in escrow until the Subscription Closing, the Purchase Price for the Acquired Shares by wire transfer of U.S. dollars in immediately available funds to the account specified by the Issuer in the Closing Notice. Such funds shall be held on behalf of Subscriber until the Subscription Closing in an escrow account by an escrow agent selected by the Issuer, subject to such escrow agent meeting any requirements specified by Subscriber to the Issuer prior to the date hereof. On the Closing Date, the Issuer shall deliver to Subscriber (i) the Acquired Shares in book-entry form, free and clear of any liens or other restrictions whatsoever (other than those arising under state or federal securities laws or as set forth herein), in the name of Subscriber (or its nominee in accordance with its delivery instructions) or to a custodian designated by Subscriber, as applicable, and (ii) a copy of the records of the Issuers transfer agent (the Transfer Agent) showing Subscriber (or such nominee or custodian) as the owner of the Acquired Shares on and as of the Closing Date. If the Closing does not occur on the same day as the Subscription Closing, the Issuer shall promptly (but not later than two (2) business days thereafter) return the Purchase Price to Subscriber by wire transfer of U.S. dollars in immediately available funds to the account specified by Subscriber, and any book-entries with respect to the Acquired Shares shall be deemed cancelled; provided, that, unless this Subscription Agreement has been validly terminated pursuant to Section 6 hereof, the return of the funds shall not terminate this Subscription Agreement or otherwise relieve any party of any of its obligations hereunder (including Subscribers obligation to purchase the Acquired Shares at the Subscription Closing).
(b) The Subscription Closing shall be subject to satisfaction or written waiver of the conditions that, on the Closing Date:
(i) all conditions precedent to the consummation of the Transactions set forth in the Business Combination Agreement shall have been satisfied or waived by the party entitled to the benefit thereof under the Business Combination Agreement (other than those conditions that may only be satisfied at the consummation of the Transactions, but subject to satisfaction or waiver by such party of such conditions as of the consummation of the Transactions);
(ii) solely with respect to Subscribers obligation to close, (A) no amendment or waiver of the Business Combination Agreement or the Issuers organizational documents (other than as contemplated by the Business Combination Agreement) shall have occurred that materially and adversely affects the economic benefits of the Acquired Shares that Subscriber is acquiring pursuant to this Subscription Agreement and (B) there shall have been no amendment or waiver to any Other Subscription Agreement (including via side letter or other agreement, except as contemplated by Section 3(p)) that materially benefits the Other Subscriber thereunder unless Subscriber has been offered the same benefit;
(iii) solely with respect to Subscribers obligation to close, all representations and warranties made by the Issuer in this Subscription Agreement shall be true and correct in all material respects as of the Closing Date (other than (i) those representations and warranties expressly made as of an earlier date, which shall be true and correct in all material respects as of such date and (ii) those representations and warranties that are already qualified by materiality or the absence of a Material Adverse Effect (as defined below), which shall be true and correct as of the Closing Date), in each case without giving effect to the consummation of the Transactions; provided, that in the event this condition would otherwise fail to be satisfied as a result of a breach of one or more of the representations and warranties of the Issuer contained in this Subscription Agreement and the facts underlying such breach would also cause a condition to the Companys or the Charterhouse Parties obligations
E-2
Table of Contents
under the Business Combination Agreement to fail to be satisfied, this condition shall nevertheless be deemed satisfied in the event both the Company and the Charterhouse Parties waives such condition with respect to such breach under the Business Combination Agreement;
(iv) solely with respect to the Issuers obligation to close, all representations and warranties made by Subscriber in this Subscription Agreement shall be true and correct in all material respects as of the Closing Date (other than (i) those representations and warranties expressly made as of an earlier date, which shall be true and correct in all material respects as of such date and (ii) those representations and warranties that are already qualified by materiality or the absence of a Subscriber Material Adverse Effect (as defined below), which shall be true and correct as of the Closing Date), in each case without giving effect to the consummation of the Transactions;
(v) solely with respect to Subscribers obligation to close, the Issuer shall have performed, satisfied and complied in all material respects with all covenants, agreements and conditions required by this Subscription Agreement to be performed, satisfied or complied with by it at or prior to the Subscription Closing, except where the failure of such performance or compliance would not or would not reasonably be expected to prevent, materially delay, or materially impair the ability of the Issuer to consummate the Closing;
(vi) solely with respect to the Issuers obligation to close, Subscriber shall have performed, satisfied and complied in all material respects with all covenants, agreements and conditions required by this Subscription Agreement to be performed, satisfied or complied with by it at or prior to the Subscription Closing, except where the failure of such performance or compliance would not or would not reasonably be expected to prevent, materially delay, or materially impair the ability of the Subscriber to consummate the Subscription Closing;
(vii) there shall not be in force any order, judgment, injunction, decree, writ, stipulation, determination or award, in each case, entered by or with any Governmental Authority, statute, rule or regulation enjoining or prohibiting the consummation of the Subscription; and
(viii) no suspension by the New York Stock Exchange (the NYSE) of the qualification of the Acquired Shares for offering or sale or trading in the United States, or initiation or threatening of any proceedings by the NYSE for any of such purposes, shall have occurred and the Acquired Shares shall have been approved for listing on the NYSE, subject to official notice of issuance.
(c) At the Subscription Closing, the parties hereto shall take, or cause to be taken, all actions and do, or cause to be done, all things necessary, proper or advisable to consummate the transactions contemplated by this Subscription Agreement on the terms and conditions described herein no later than immediately prior to the consummation of the Transactions.
(d) For purposes of this Subscription Agreement, business day shall mean any day other than (i) any Saturday or Sunday or (ii) any other day on which banks located in New York, New York or London, United Kingdom, are required or authorized by applicable law to be closed for business.
3. Issuers Representations, Warranties and Agreements. The Issuer hereby represents and warrants that:
(a) The Issuer has been duly incorporated and is validly existing as a corporation in good standing under the Delaware General Corporate Law (DGCL). The Issuer has all corporate power and authority to own, lease and operate its properties and, subject to obtaining all required approvals necessary in connection with the performance of the Business Combination Agreement and the consummation of the Transactions (collectively, the Required Approvals), conduct its business as presently conducted and to enter into, deliver and perform its obligations under this Subscription Agreement.
(b) As of the Closing Date, the Acquired Shares will be duly authorized by the Issuer and, when issued and delivered to Subscriber against full payment for the Acquired Shares in accordance with the terms of this
E-3
Table of Contents
Subscription Agreement and registered with the Transfer Agent, the Acquired Shares will be validly issued, fully paid and non-assessable, free and clear of any liens and other restrictions (other than those arising under this Subscription Agreement or applicable law) and will not have been issued in violation of or subject to any preemptive or similar rights created under the Issuers certificate of incorporation and bylaws or under the DGCL or otherwise.
(c) This Subscription Agreement has been duly authorized, validly executed and delivered by the Issuer and, assuming that this Subscription Agreement constitutes the valid and binding agreement of Subscriber, is the valid and binding obligation of the Issuer, and is enforceable against the Issuer in accordance with its terms, except as may be limited or otherwise affected by (i) applicable bankruptcy, insolvency, fraudulent conveyance, reorganization, moratorium or other laws relating to or affecting the rights of creditors generally, and (ii) principles of equity, whether considered at law or equity.
(d) Subject to obtaining the Required Approvals, the execution, delivery and performance by the Issuer of this Subscription Agreement (including compliance by the Issuer with all of the provisions hereof), and the issuance and sale by the Issuer of the Acquired Shares, will not conflict with or result in a breach or violation of any of the terms or provisions of, or constitute a default under, or result in the creation or imposition of any lien, charge or encumbrance upon any of the property or assets of the Issuer pursuant to the terms of (i) any indenture, mortgage, deed of trust, loan agreement, lease, license or other agreement or instrument to which the Issuer is a party or by which the Issuer is bound or to which any of the property or assets of the Issuer is subject, which would be reasonably likely to have, individually or in the aggregate, a material adverse effect on the business, properties, financial condition, stockholders equity or results of operations of the Issuer (a Material Adverse Effect) or materially affect the validity of the Acquired Shares or the legal authority of the Issuer to comply in all material respects with the Issuers obligations under this Subscription Agreement; (ii) the organizational documents of the Issuer; or (iii) any statute or any judgment, order, rule or regulation of any court or governmental agency or body, domestic or foreign, having jurisdiction over the Issuer or any of its properties that would be reasonably likely to have, individually or in the aggregate, a Material Adverse Effect or materially affect the validity of the Acquired Shares or the legal authority of the Issuer to comply in all material respects with the Issuers obligations under this Subscription Agreement.
(e) Other than the Issuers Class B common stock, par value $0.0001 per share (the Class B Shares), there are no securities or instruments issued by or to which the Issuer is a party containing anti-dilution provisions that will be triggered by the issuance of (i) the Acquired Shares or (ii) the shares to be issued pursuant to any Other Subscription Agreement that have not been or will not be validly waived on or prior to the Closing Date; provided, that the holders of the Class B Shares will waive any such anti-dilution provisions in connection with the Transactions.
(f) The Issuer is not in default or violation (and no event has occurred which, with notice or the lapse of time or both, would constitute a default or violation) of any term, condition or provision of (i) the organizational documents of the Issuer, (ii) any loan or credit agreement, note, bond, mortgage, indenture, lease or other agreement, permit, franchise or license to which the Issuer is now a party or by which the Issuers properties or assets are bound or (iii) any statute or any judgment, order, rule or regulation of any court or governmental agency or body, domestic or foreign, having jurisdiction over the Issuer or any of its properties, except, in the case of clauses (ii) and (iii), for defaults or violations that have not had and would not be reasonably likely to have, individually or in the aggregate, a Material Adverse Effect.
(g) The Issuer is not required to obtain any consent, waiver, authorization or order of, give any notice to, or make any filing or registration with, any court or other federal, state, local or other governmental authority, self-regulatory organization or other person in connection with the execution, delivery and performance by the Issuer of this Subscription Agreement (including, without limitation, the issuance of the Acquired Shares), other than (i) filings with the Securities and Exchange Commission (the Commission), (ii) filings required by applicable state securities laws, (iii) filings required in accordance with Section 10(q) of this Subscription Agreement;
E-4
Table of Contents
(iv) filings required by the NYSE, including with respect to obtaining stockholder approval of the Transactions; (v) filings set forth in Section 4.03 of the Business Combination Agreement, and (vi) any consent, waiver, authorization, order, notice or filing the failure of which to obtain or make would not be reasonably likely to have, individually or in the aggregate, a Material Adverse Effect.
(h) As of the date of this Subscription Agreement, the authorized capital stock of the Issuer consists of (i) 5,000,000 shares of preferred stock, par value $0.0001 per share (Preferred Shares), (ii) 500,000,000 Class A Shares, and (iii) 50,000,000 Class B Shares. As of the date of this Subscription Agreement: (A) no Preferred Shares are issued and outstanding, (B) 75,000,000 Class A Shares are issued and outstanding, (C) 18,750,000 Class B Shares are issued and outstanding, (D) 8,500,000 warrants to purchase 8,500,000 Existing Class A Shares (the Private Placement Warrants) are outstanding and (E) 18,750,000 warrants to purchase 18,750,000 Existing Class A Shares (the Public Warrants) are outstanding. All issued and outstanding Class A Shares and Class B Shares have been duly authorized and validly issued, and are fully paid and non-assessable. Except as set forth above and pursuant to the Other Subscription Agreements, the Business Combination Agreement and the other agreements and arrangements referred to therein, as of the date hereof and immediately prior to Closing, there are no outstanding options, warrants or other rights to subscribe for, purchase or acquire from the Issuer any Class A Shares, Class B Shares or other equity interests in the Issuer, or securities convertible into or exchangeable or exercisable for such equity interests. There are no stockholder agreements, voting trusts or other agreements or understandings to which the Issuer is a party or by which it is bound relating to the voting of any securities of the Issuer, other than (1) as set forth in the SEC Documents (as defined below) and (2) as contemplated by the Business Combination Agreement and the other agreements and arrangements referred to therein.
(i) The Issuer has not received any written communication from a governmental entity that alleges that the Issuer is not in compliance with or is in default or violation of any applicable law, except where such non-compliance, default or violation would not be reasonably likely to have, individually or in the aggregate, a Material Adverse Effect.
(j) The issued and outstanding Class A Shares are registered pursuant to Section 12(b) of the Securities Exchange Act of 1934, as amended (the Exchange Act), and are listed for trading on the NYSE under the symbol GSAH. There is no suit, action, proceeding or investigation pending or, to the knowledge of the Issuer, threatened against the Issuer by the NYSE or the Commission, respectively, to prohibit or terminate the listing of the Class A Shares on the NYSE or to deregister the Class A Shares under the Exchange Act. The Issuer has taken no action that is designed to terminate the registration of the Class A Shares under the Exchange Act.
(k) Assuming the accuracy of Subscribers representations and warranties set forth in Section 4 of this Subscription Agreement, no registration under the Securities Act is required for the offer and sale of the Acquired Shares by the Issuer to Subscriber.
(l) Neither the Issuer nor any person acting on its behalf has offered or sold the Acquired Shares by any form of general solicitation or general advertising, including, but not limited to, the following: (1) any advertisement, article, notice or other communication published in any newspaper, magazine, or similar media or broadcast over television or radio; (2) any website posting or widely distributed email; or (3) any seminar or meeting whose attendees have been invited by any general solicitation or general advertising.
(m) A copy of each form, report, statement, schedule, prospectus, proxy, registration statement and other document, if any, filed by the Issuer with the Commission since its initial registration of the Class A Shares under the Exchange Act (the SEC Documents) is available to Subscriber via the Commissions EDGAR system. None of the SEC Documents filed under the Exchange Act contained, when filed or, if amended prior to the date of this Subscription Agreement, as of the date of such amendment with respect to those disclosures that are amended, any untrue statement of a material fact or omitted to state a material fact required to be stated therein or necessary to make the statements therein, in the light of the circumstances under which they were
E-5
Table of Contents
made, not misleading; provided, that with respect to the information about the Company, the Charterhouse Parties and their respective affiliates contained in the Schedule 14A and related proxy materials (or other SEC document) to be filed by the Issuer the representation and warranty in this sentence is made to the Issuers knowledge. The Issuer has timely filed each report, statement, schedule, prospectus, and registration statement that the Issuer was required to file with the Commission since its initial registration of the Class A Shares under the Exchange Act. There are no material outstanding or unresolved comments in comment letters from the staff of the Division of Corporation Finance (the Staff) of the Commission with respect to any of the SEC Documents.
(n) Except for such matters as have not had and would not be reasonably likely to have, individually or in the aggregate, a Material Adverse Effect, there is no (i) action, suit, claim or other proceeding, in each case by or before any governmental authority pending, or, to the knowledge of the Issuer, threatened against the Issuer or (ii) judgment, decree, injunction, ruling or order of any governmental entity or arbitrator outstanding against the Issuer.
(o) Other than the Agent (as defined below), the Issuer has not dealt with any broker, finder, commission agent, placement agent or arranger in connection with the sale of the Acquired Shares, and the Issuer is not under any obligation to pay any brokers fee or commission in connection with the sale of the Acquired Shares other than to the Agent. Neither the Issuer nor any of its affiliates nor any other person acting on its behalf (other than its officers acting in such capacity) has solicited offers for, or offered or sold, the Acquired Shares other than through the Agent. No broker, finder or other financial consultant has acted on behalf of Issuer in connection with this Subscription Agreement or the transactions contemplated hereby in such a way as to create any liability on Subscriber.
(p) There are no Other Subscription Agreements, side letter agreements or other agreements or understandings (including written summaries of any oral understandings) with any Other Subscriber (other than Subscribers in connection with the Other Subscription Agreements) (collectively, the PIPE Agreements) which include terms and conditions that are materially more advantageous to any such Other Subscriber (as compared to Subscriber), other than such PIPE Agreements containing any of the following: (i) any rights or benefits granted to an Other Subscriber in connection with such Other Subscribers compliance with any law, regulation or policy specifically applicable to such Other Subscriber or in connection with the taxable status of an Other Subscriber, (ii) any rights or benefits which are personal to an Other Subscriber based solely on its place of organization or headquarters, organizational form, legal status, or other particular restrictions applicable to, such Other Subscriber, (iii) any rights with respect to the confidentiality or disclosure of an Other Subscribers identity, or (iv) any rights or benefits granted to Goldman Sachs & Co. LLC or any of their respective Representatives (as defined below) providing for the ability to transfer or assign all or a portion of its rights under such Other Subscription Agreement to which it is a party. Notwithstanding the foregoing clauses (i)-(iv), the price per Class A Share payable by each Other Subscriber pursuant to any PIPE Agreements shall be the same as the price per Acquired Share payable by Subscriber.
(q) Notwithstanding anything to the contrary contained in this Section 3 of this Subscription Agreement, no representation or warranty is made by the Issuer as to the historical accounting treatment of its issued and outstanding Private Placement Warrants or Public Warrants or other changes in accounting arising in connection with any required restatement of the Issuers historical financial statements, or as to any deficiencies in disclosure (including with respect to financial statement presentation or accounting and disclosure controls) arising from the treatment of such warrants as equity rather than liabilities or changes in the Issuers historical financial statements and SEC Documents.
(r) Issuer acknowledges and agrees that, notwithstanding anything herein to the contrary, the Acquired Shares may be pledged by Subscriber in connection with a bona fide margin agreement, provided such pledge shall be (i) pursuant to an available exemption from the registration requirements of the Securities Act or (ii) pursuant to, and in accordance with, a registration statement that is effective under the Securities Act at the
E-6
Table of Contents
time of such pledge, and Subscriber effecting a pledge of Acquired Shares shall not be required to provide Issuer with any notice thereof; provided, however, that neither Issuer nor its counsel shall be required to take any action (or refrain from taking any action) in connection with any such pledge, other than providing any such lender of such margin agreement with an acknowledgment that the Acquired Shares are not subject to any contractual prohibition on pledging or a lock-up agreement, in each case with the Issuer, the form of such acknowledgment to be subject to review and comment by Issuer in all respects.
(s) The Issuer has not offered Class A Shares or any similar securities during the six months prior to the date hereof to anyone other than in connection with the Transactions and to Subscriber and other investors in connection with the Other Subscription Agreements.
4. Subscribers Representations, Warranties and Agreements. Subscriber hereby represents and warrants that:
(a) If Subscriber is not a natural person, (i) Subscriber has been duly organized, formed or incorporated, as the case may be, and is validly existing in good standing under the laws of its jurisdiction of organization, formation or incorporation, as the case may be, with all requisite power and authority to enter into, deliver and perform its obligations under this Subscription Agreement, and (ii) this Subscription Agreement has been duly authorized, validly executed and delivered by Subscriber.
(b) If Subscriber is a natural person, (i) Subscriber has all requisite power and authority to enter into, deliver and perform its obligations under this Subscription Agreement, (ii) Subscribers signature on this Subscription Agreement is genuine and Subscriber has duly executed and delivered this Subscription Agreement, and (iii) Subscriber has all requisite legal competence and capacity to acquire and hold the Acquired Shares and to execute, deliver and comply with the terms of this Subscription Agreement.
(c) Assuming that this Subscription Agreement constitutes the valid and binding agreement of the Issuer, this Subscription Agreement is the valid and binding obligation of Subscriber, and is enforceable against Subscriber in accordance with its terms, except as may be limited or otherwise affected by (i) applicable bankruptcy, insolvency, fraudulent conveyance, reorganization, moratorium or other laws relating to or affecting the rights of creditors generally, and (ii) principles of equity, whether considered at law or equity.
(d) The execution, delivery and performance by Subscriber of this Subscription Agreement and the consummation of the transactions contemplated herein do not and will not conflict with or result in a breach or violation of any of the terms or provisions of, or constitute a default under, or result in the creation or imposition of any lien, charge or encumbrance upon any of the property or assets of Subscriber or any of its subsidiaries, if applicable, pursuant to the terms of (i) any indenture, mortgage, deed of trust, loan agreement, lease, license or other agreement or instrument to which Subscriber or, if applicable, any of its subsidiaries is a party or by which Subscriber or, if applicable, any of its subsidiaries is bound or to which any of the property or assets of Subscriber or, if applicable, any of its subsidiaries is subject, which would be reasonably likely to have, individually or in the aggregate, a material adverse effect on the business, properties or financial condition of Subscriber, or, if applicable, the stockholders equity or results of operations of Subscriber or, if applicable, any of its subsidiaries, taken as a whole (a Subscriber Material Adverse Effect), or materially affect the legal authority of Subscriber to comply in all material respects with Subscribers obligations under this Subscription Agreement, (ii) the organizational documents of Subscriber if Subscriber is not a natural person, or (iii) any statute or any judgment, order, rule or regulation of any court or governmental agency or body, domestic or foreign, having jurisdiction over Subscriber or, if applicable, any of its subsidiaries or any of their respective properties that would be reasonably likely to have, individually or in the aggregate, a Subscriber Material Adverse Effect or materially affect the legal authority of Subscriber to comply in all material respects with Subscribers obligations under this Subscription Agreement.
(e) Subscriber is (i) an accredited investor, satisfying the applicable requirements set forth on Schedule A, (ii) an Institutional Account as defined in FINRA Rule 4512(c) and (iii) experienced in investing in private
E-7
Table of Contents
equity transactions and capable of evaluating investment risks independently, both in general and in connection with its participation in the Offering. Subscriber represents that it is purchasing its entire beneficial ownership interest in the Acquired Shares for its own account (and not for the account of others) for investment purposes and not with a view to the distribution thereof in violation of the securities laws of the United States or any other jurisdiction, provided, that (subject to the securities laws of the United States or any other jurisdiction) disposition of Subscribers property shall at all times be within Subscribers control. Subscriber understands that (1) the Acquired Shares (A) have not been registered under the securities laws of the United States or any other jurisdiction and may be resold or transferred in the United States or otherwise only if registered pursuant to the provisions of the Securities Act or if an exemption from registration is available, and (B) may only be resold or transferred in compliance with applicable law and the restrictions on transfer set forth in this Subscription Agreement, and that (2) the Issuer is not required to register the Acquired Shares other than as provided in Section 5 of this Agreement. Subscriber further represents and warrants that it will not sell, transfer or otherwise dispose of the Acquired Shares or any interest therein except in a registered transaction or in a transaction exempt from or not subject to the registration requirements of the Securities Act and except in accordance with the terms and conditions of this Subscription Agreement. Subscriber acknowledges that the Acquired Shares will be subject to transfer restrictions as set forth on Exhibit A to this Subscription Agreement.
(f) Subscriber represents and warrants that Subscriber is not (i) a person or entity named on the List of Specially Designated Nationals and Blocked Persons administered by the U.S. Treasury Departments Office of Foreign Assets Control (OFAC) or in any Executive Order issued by the President of the United States and administered by OFAC (OFAC List), or a person or entity prohibited by any OFAC sanctions program, (ii) a Designated National as defined in the Cuban Assets Control Regulations, 31 C.F.R. Part 515 or (iii) a non-U.S. shell bank or providing banking services indirectly to a non-U.S. shell bank. Subscriber agrees to provide law enforcement agencies, if requested thereby, such records as required by applicable law, provided, that Subscriber is permitted to do so under applicable law. Subscriber represents that if it is a financial institution subject to the Bank Secrecy Act (31 U.S.C. Section 5311 et seq.) (the BSA), as amended by the USA PATRIOT Act of 2001 (the PATRIOT Act), and its implementing regulations (collectively, the BSA/PATRIOT Act), that Subscriber, directly or indirectly through a third-party administrator, maintains policies and procedures reasonably designed to comply with applicable obligations under the BSA/PATRIOT Act. Subscriber also represents that, to the extent required, it, directly or indirectly through a third-party administrator, maintains policies and procedures reasonably designed for the screening of its investors against the OFAC sanctions programs, including the OFAC List. Subscriber further represents and warrants that, to the extent required, it, directly or indirectly through a third-party administrator, maintains policies and procedures reasonably designed to ensure that the funds held by Subscriber and used to purchase the Acquired Shares were legally derived.
(g) The purchase of Acquired Shares by Subscriber has not been solicited by or through anyone other than the Issuer or the Agent.
(h) Subscriber acknowledges that the Acquired Shares will not be eligible for resale pursuant to Rule 144A promulgated under the Securities Act. Subscriber understands and agrees that the Acquired Shares will be subject to transfer restrictions as set forth on Exhibit A to this Subscription Agreement, unless and until such transfer restrictions have been removed in accordance with Section 9(d) of this Subscription Agreement and, as a result of these transfer restrictions, Subscriber may not be able to readily resell the Acquired Shares and may be required to bear the financial risk of an investment in the Acquired Shares for an indefinite period of time. Subscriber also acknowledges that the Acquired Shares will not immediately be eligible for offer, resale, transfer, pledge or disposition pursuant to Rule 144 promulgated under the Securities Act (Rule 144), and that the provisions of Rule 144(i) will apply to the Acquired Shares. Subscriber understands that it has been advised to consult legal counsel prior to making any offer, resale, pledge or transfer of any of the Acquired Shares.
(i) Subscriber understands and agrees that Subscriber is purchasing the Acquired Shares directly from the Issuer. Subscriber further acknowledges that (i) there have been no, and will be no, representations, warranties, covenants or agreements (express or implied, of any kind or character) made to Subscriber in connection with
E-8
Table of Contents
Subscribers purchase of the Acquired Shares by the Issuer, the Agent, the Company, the Charterhouse Parties, any other party to the Business Combination Agreement or participant in the Transactions or any of their respective Representatives, expressly or by implication, other than those representations, warranties, covenants and agreements of the Issuer expressly set forth in this Subscription Agreement, (ii) the Agent has not provided any advice or recommendation to Subscriber in connection with Subscribers purchase of the Acquired Shares, and (iii) the Agent will not have any responsibility to Subscriber with respect to (x) any representations, warranties or agreements made by any person or entity under or in connection with the Subscription or any of the documents furnished pursuant thereto or in connection therewith, or the execution, legality, validity or enforceability (with respect to any person) thereof, or (y) the business, affairs, financial condition, operations, properties or prospects of, or any other matter concerning the Issuer, the Company, the Charterhouse Parties or the Transactions.
(j) In making its decision to purchase the Acquired Shares, Subscriber represents that it has relied solely upon the independent investigation made by Subscriber and has independently made its own analysis and decision to enter into this Subscription Agreement and purchase the Acquired Shares, in each case, based on such information as such Subscriber has deemed appropriate and without reliance upon the Agent or any of Agents affiliates. Subscriber acknowledges and agrees that Subscriber has received such information as Subscriber deems necessary in order to make an investment decision with respect to the Acquired Shares, including with respect to the Issuer, the Transactions, the Company, the Charterhouse Parties and their respective affiliates and Representatives. Subscriber represents and warrants that Subscriber and Subscribers professional advisor(s), if any, (i) were given the opportunity to ask questions and receive answers concerning the terms and conditions of the Subscription, the Issuer, the Company, the Charterhouse Parties and to obtain any additional information which the Issuer possessed or could acquire without unreasonable effort or expense and (ii) received, reviewed and understood the offering materials made available to it in connection with the Subscription and (iii) conducted and completed its own independent due diligence with respect to the Transactions. Except for the representations, warranties and agreements of the Issuer set forth in this Subscription Agreement, Subscriber is relying exclusively on its own sources of information, investment analysis and due diligence (including professional advice it may deem appropriate) with respect to the Subscription, the Acquired Shares and the business, condition (financial and otherwise), management, operations, properties and prospects of the Issuer, Company and the Charterhouse Parties, including but not limited to all business, legal, regulatory, accounting, credit and tax matters.
(k) Subscriber became aware of this offering of the Acquired Shares solely by means of direct contact between Subscriber and the Issuer or by means of contact from Goldman Sachs & Co. LLC, acting as a placement agent for the Issuer (together with its affiliates and any of its or their control persons, officers, directors and employees, the Agent), and the Acquired Shares were offered to Subscriber solely by direct contact between Subscriber and the Issuer or by contact between Subscriber and the Agent. Subscriber did not become aware of this offering of the Acquired Shares, nor were the Acquired Shares offered to Subscriber, by any other means. Subscriber acknowledges that the Issuer represents and warrants that the Acquired Shares (i) were not offered by any form of general solicitation or general advertising, including methods described in Section 502(c) of Regulation D of the Securities Act and (ii) are not being offered in a manner involving a public offering under, or in a distribution in violation of, the Securities Act, or any state securities laws offering under, or in a distribution in violation of, the Securities Act, or any state securities laws.
(l) Subscriber acknowledges that it is aware that there are substantial risks incident to the purchase and ownership of the Acquired Shares, including those set forth in the SEC Documents and the investor presentation provided by the Issuer. Subscriber is able to fend for itself in the transactions contemplated herein, has such knowledge and experience in financial and business matters as to be capable of evaluating the merits and risks of an investment in the Acquired Shares, and Subscriber has sought such accounting, legal and tax advice as Subscriber has considered necessary to make an informed investment decision.
(m) Without limiting the representations, warranties and covenants set forth in this Subscription Agreement, Subscriber represents and acknowledges that Subscriber has, alone, or together with any professional advisor(s),
E-9
Table of Contents
adequately analyzed and fully considered the risks of an investment in the Acquired Shares and determined that the Acquired Shares are a suitable investment for Subscriber and that Subscriber is able at this time and in the foreseeable future to bear the economic risk of a total loss of Subscribers investment in the Issuer. Subscriber has determined based on its own independent review and such professional advice as it deems appropriate that its purchase of the Acquired Shares (i) is fully consistent with its financial needs, objectives and condition, (ii) comply and is fully consistent with all investment policies, guidelines and other restrictions applicable to it, and (iii) is a fit, proper and suitable investment for it, notwithstanding the substantial risks inherent in investing in or holding the Acquired Shares. Subscriber acknowledges that it is able to bear the substantial risk associated with the purchase of the Acquired Shares, and specifically that a possibility of total loss exists.
(n) Subscriber understands and agrees that no federal or state agency has passed upon or endorsed the merits of the offering of the Acquired Shares or made any findings or determination as to the fairness of an investment in the Acquired Shares.
(o) If Subscriber is or is acting on behalf of (i) an employee benefit plan that is subject to Title I of the Employee Retirement Income Security Act of 1974, as amended (ERISA), (ii) a plan, an individual retirement account or other arrangement that is subject to Section 4975 of the Internal Revenue Code of 1986, as amended (the Code), (iii) an entity whose underlying assets are considered to include plan assets of any such plan, account or arrangement described in clauses (i) and (ii) (each, an ERISA Plan), or (iv) an employee benefit plan that is a governmental plan (as defined in Section 3(32) of ERISA), a church plan (as defined in Section 3(33) of ERISA), a non-U.S. plan (as described in Section 4(b)(4) of ERISA) or other plan that is not subject to the foregoing clauses (i), (ii) or (iii) but may be subject to provisions under any other federal, state, local, non-U.S. or other laws or regulations that are similar to such provisions of ERISA or the Code (collectively, Similar Laws, and together with ERISA Plans, Plans), Subscriber represents and warrants that (A) it has not relied on the Issuer or any of its affiliates (the Transaction Parties) for investment advice or as the Plans fiduciary, with respect to its decision to acquire and hold the Acquired Shares, and none of the Transaction Parties shall at any time be relied on as the Plans fiduciary with respect to any decision in connection with Subscribers investment in the Acquired Shares; (B) the decision to invest in the Acquired Shares has been made at the recommendation or direction of a fiduciary (for purposes of ERISA and/or Section 4975 of the Code, or any applicable Similar Law) with respect to Subscribers investment in the Acquired Shares who is independent of the Transaction Parties; and (C) its purchase of the Acquired Shares will not constitute a non-exempt prohibited transaction under Section 406 of ERISA or Section 4975 of the Code, or any applicable Similar Law.
(p) No foreign person (as defined in 31 C.F.R. §800.224) in which the national or subnational governments of a single foreign state have a substantial interest (as defined in 31 C.F.R. §800.244) will acquire a substantial interest (as defined in 31 C.F.R. §800.244) in the Issuer as a result of the purchase and sale of the Acquired Shares hereunder such that a declaration to the Committee on Foreign Investment in the United States would be mandatory under 31 C.F.R. §800.401, and Subscriber will not (i) have control (as defined in 31 C.F.R. §800.208) over the Issuer from and after the Closing as a result of the purchase and sale of the Acquired Shares hereunder or (ii) have any of the rights with respect to the Issuer that are set forth at 31 C.F.R. §800.211(b) as rights of a foreign person with respect to a TID U.S. business.
(q) No broker, finder or other financial consultant has acted on behalf of Subscriber in connection with this Subscription Agreement or the transactions contemplated hereby in such a way as to create any liability on the Issuer.
(r) The Subscriber acknowledges that (i) the Issuer and the Agent currently may have, and later may come into possession of, information regarding the Issuer that is not known to the Subscriber and that may be material to a decision to enter into this transaction to purchase the Acquired Shares (Excluded Information), (ii) the Subscriber has determined to enter into this transaction to purchase the Acquired Shares notwithstanding its lack of knowledge of the Excluded Information, (iii) neither the Issuer nor the Agent shall have liability to the
E-10
Table of Contents
Subscriber with respect to the non-disclosure of the Excluded Information and (iv) other Persons (including Other Subscribers) may have received additional information, including participating in meetings with Representatives of the Issuer, the Agent and the Company and their respective Affiliates in connection with the transactions contemplated hereby (including, but not limited to financial, legal and other due diligence information and reports). Affiliate means, with respect to any person or entity, any other person or entity who, directly or indirectly, controls, is controlled by, or is under direct or indirect common control with, such person or entity, and control, when used with respect to any specified person or entity, shall mean the power to direct or cause the direction of the management and policies of such person or entity, directly or indirectly, whether through ownership of voting securities or partnership or other ownership interests, by contract or otherwise; and the terms controlling and controlled shall have correlative meanings.
(s) The Subscriber acknowledges that certain information provided to it was based on projections, and such projections were prepared based on assumptions and estimates that are inherently uncertain and are subject to a wide variety of significant business, economic and competitive risks and uncertainties that could cause actual results to differ materially from those contained in the projections. The Subscriber acknowledges that such information and projections were prepared without the participation of the Agent and the Agent assumes no responsibility for independent verification of, or the accuracy or completeness of, such information or projections.
(t) The Subscriber acknowledges that the Agent and any of its respective affiliates or any of the Agents or its affiliates control persons, officers, directors, employees or other representatives, legal counsel, financial advisors, accountants or agents (collectively, Representatives) have made no independent investigation with respect to the Issuer or the Acquired Shares or the accuracy, completeness or adequacy of any information supplied to the Subscriber by the Issuer. Subscriber acknowledges and agrees that neither the Agent nor any Representative of the Agent has provided Subscriber with any information or advice with respect to the Acquired Shares nor is such information or advice necessary or desired. In connection with the issue and purchase of the Acquired Shares, Subscriber acknowledges that the Agent is acting solely as the Issuers placement agent in connection with the sale of the Acquired Shares and is not acting as an underwriter or in any other capacity and is not and shall not be construed as a fiduciary or financial advisor for Subscriber, the Company or any other person or entity.
(u) Subscriber acknowledges that it is not relying upon, and has not relied upon, any statement, representation or warranty made by the Agent in making its investment or decision to invest in Issuer.
(v) Subscriber agrees that the Agent shall not be liable to Subscriber for any action heretofore or hereafter taken or omitted to be taken by Agent or have any liability or obligation (including without limitation, for or with respect to any losses, claims, damages, obligations, penalties, judgments, awards, liabilities, costs, expenses or disbursements incurred by you, the Company or any other person or entity), whether in contract, tort or otherwise, to any Subscriber, or to any person claiming through such Subscriber, in respect of the Transactions.
(w) Subscriber is an entity having total liquid assets and net assets in excess of the Purchase Price as of the date hereof and as of each date the Purchase Price would be required to be funded to the Issuer pursuant and was not formed for the purpose of acquiring the Acquired Shares. At the Subscription Closing, Subscriber will have sufficient immediately available funds to pay the Purchase Price pursuant to Section 2(a) of this Subscription Agreement.
5. Registration Rights.
5.1 The Issuer agrees that, as soon as reasonably practicable, but in any event within 30 calendar days after the Closing Date (the Filing Date), the Issuer will file with the Commission (at the Issuers sole cost and expense) a registration statement for a shelf registration on Form S-1 (the Registration Statement) registering the resale of the Acquired Shares that are eligible for registration (determined as of two business days prior to
E-11
Table of Contents
such filing) (the Registrable Securities), and the Issuer shall use its commercially reasonable efforts to have the Registration Statement declared effective as soon as practicable after the filing thereof, but no later than the earlier of (i) the 60th calendar day (or 90th calendar day if the Commission notifies the Issuer that it will review the Registration Statement) following the Closing and (ii) the 10th business day after the date the Issuer is notified (orally or in writing, whichever is earlier) by the Commission that the Registration Statement will not be reviewed or will not be subject to further review (such earlier date, the Effectiveness Date); provided, however, that the Issuers obligations to include the Registrable Securities in the Registration Statement are contingent upon Subscriber furnishing a completed and executed selling shareholders questionnaire in customary form to the Issuer that contains the information required by Commission rules for a Registration Statement regarding Subscriber, the securities of the Issuer held by Subscriber and the intended method of disposition of the Registrable Securities (which shall be limited to non-underwritten public offerings) to effect the registration of the Registrable Securities, and Subscriber shall execute such documents in connection with such registration as the Issuer may reasonably request that are customary of a selling stockholder in similar situations, including providing that the Issuer shall be entitled to postpone and suspend the effectiveness or use of the Registration Statement during any customary blackout or similar period or as permitted hereunder; provided that Subscriber shall not in connection with the foregoing be required to execute any lock-up or similar agreement or otherwise be subject to any contractual restrictions on the ability to transfer the Acquired Shares. For purposes of clarification, any failure by the Issuer to file the Registration Statement by the Filing Date or to effect such Registration Statement by the Effectiveness Date shall not otherwise relieve the Issuer of its obligations to file or effect the Registration Statement as set forth above in this Section 5. Notwithstanding the foregoing, if the Commission prevents the Issuer from including any or all of the Acquired Shares proposed to be registered under the Registration Statement due to limitations on the use of Rule 415 of the Securities Act for the resale of the Acquired Shares by the applicable stockholders or otherwise, such Registration Statement shall register for resale such number of the Acquired Shares which is equal to the maximum number of the Acquired Shares as is permitted by the Commission. In such event, the number of the Acquired Shares to be registered for each selling stockholder named in the Registration Statement shall be reduced pro rata among all such selling stockholders and as promptly as practicable after being permitted to register additional Subscribed Shares under Rule 415 under the Securities Act, the Issuer shall amend the Registration Statement or file a new Registration Statement to register such additional Subscribed Shares and cause such amendment or Registration Statement to become effective as promptly as practicable. Unless required under applicable laws and Commission rules, in no event shall the Subscriber be identified as a statutory underwriter in the Registration Statement; provided, that if the Subscriber is required to be so identified as a statutory underwriter in the Registration Statement, the Subscriber will have an opportunity to withdraw its Registrable Securities from the Registration Statement. Following the Effectiveness Date, if the transfer restrictions as set forth on Exhibit A to this Subscription Agreement are no longer required by the Securities Act or any applicable state securities laws, upon written request of Subscriber, the Issuer shall use its commercially reasonable efforts to cooperate with Subscriber to have such transfer restrictions removed, including providing authorization (and, if required, a legal opinion at the Issuers sole expense to the effect that the removal of such restrictive legends in such circumstances may be effected under the Securities Act) to the Issuers transfer agent; provided that Subscriber will provide customary written representations and broker letters as may be reasonably requested by Issuers transfer agent or Issuers legal counsel; provided further that Subscriber understands that the transfer restrictions will not be removable from Subscribers Acquired Shares except in connection with a sale pursuant to an effective registration statement, Rule 144 (including all conditions of Rule 144(i)(2)) or another exemption from the Securities Act.
5.2 In the case of the registration effected by the Issuer pursuant to this Subscription Agreement, the Issuer shall, upon reasonable request, inform Subscriber as to the status of such registration. At its expense the Issuer shall:
5.2.1 except for such times as the Issuer is permitted hereunder to suspend the use of the prospectus forming part of a Registration Statement, use its commercially reasonable efforts to keep such registration, and any qualification, exemption or compliance under state securities laws which the Issuer determines to obtain, continuously effective with respect to Subscriber, and to keep the
E-12
Table of Contents
applicable Registration Statement or any subsequent shelf registration statement free of any material misstatements or omissions, until the earlier of the following: (i) Subscriber ceases to hold any Registrable Securities, (ii) the date all Registrable Securities held by Subscriber may be sold without restriction under Rule 144, including without limitation, any volume and manner of sale restrictions which may be applicable to affiliates under Rule 144 and without the requirement for the Issuer to be in compliance with the current public information required under Rule 144(c)(1) (or Rule 144(i)(2), if applicable) and (iii) three years from the date of effectiveness of the Registration Statement;
5.2.2 advise Subscriber as promptly as reasonably possible and in any case within two (2) business days:
(a) when the Registration Statement any amendment thereto has been filed with the Commission and when such Registration Statement or any post-effective amendment thereto has become effective;
(b) after it shall have received notice or obtained knowledge thereof, of the issuance by the Commission of any stop order suspending the effectiveness the Registration Statement or the initiation of any proceedings for such purpose;
(c) of the receipt by the Issuer of any notification with respect to the suspension of the qualification of the Registrable Securities included therein for sale in any jurisdiction or the initiation or threatening of any proceeding for such purpose; and
(d) subject to the provisions in this Subscription Agreement, of the occurrence of any event that requires the making of any changes in the Registration Statement or any prospectus so that, as of such date, the statements therein are not misleading and do not omit to state a material fact required to be stated therein or necessary to make the statements therein (in the case of a prospectus, in the light of the circumstances under which they were made) not misleading.
Notwithstanding anything to the contrary set forth herein, the Issuer shall not, when so advising Subscriber of such events, provide Subscriber with any material, non-public information regarding the Issuer other than to the extent that providing notice to Subscriber of the occurrence of the events listed in (a) through (d) above constitutes material, non-public information regarding the Issuer;
5.2.3 use its commercially reasonable efforts to obtain the withdrawal of any order suspending the effectiveness of the Registration Statement as soon as reasonably practicable;
5.2.4 upon the occurrence of any event contemplated in Section 5.2.2(d), except for such times as the Issuer is permitted hereunder to suspend, and has suspended, the use of a prospectus forming part of the Registration Statement, use its commercially reasonable efforts to as soon as reasonably practicable prepare a post-effective amendment to the Registration Statement or a supplement to the related prospectus, or file any other required document so that, as thereafter delivered to purchasers of the Registrable Securities included therein, such prospectus will not include any untrue statement of a material fact or omit to state any material fact necessary to make the statements therein, in the light of the circumstances under which they were made, not misleading; and
5.2.5 use its commercially reasonable efforts to cause all the Acquired Shares to be listed on each securities exchange or market, if any, on which the Issuers Acquired Shares are then listed.
5.3 Suspension Event.
5.3.1 Notwithstanding anything to the contrary in this Subscription Agreement, the Issuer shall be entitled to delay or postpone the effectiveness of the Registration Statement, and from time to time to require Subscriber not to sell under the Registration Statement or to suspend the effectiveness thereof, if the filing, effectiveness or continued use of any Registration Statement would require the
E-13
Table of Contents
Issuer to make any public disclosure of material non-public information, which disclosure, in the good faith determination of the board of directors of the Issuer, after consultation with counsel to the Issuer, (a) would be required to be made in any Registration Statement in order for the applicable Registration Statement not to contain any untrue statement of a material fact or omit to state a material fact necessary to make the statements contained therein not misleading, (b) would not be required to be made at such time if the Registration Statement were not being filed, and (c) the Issuer has a bona fide business purpose for not making such information public, including, without limitation, that such disclosure could materially affect a bona fide business, financing, merger, acquisition or other strategic transaction of or by the Issuer or would require premature disclosure of information that could otherwise materially adversely affect the Issuer (each such circumstance, a Suspension Event); provided, however, that the Issuer may not delay or suspend the Registration Statement on more than two occasions or for more than sixty (60) consecutive calendar days, or more than ninety (90) total calendar days, in each case during any twelve-month period. Upon receipt of any written notice from the Issuer of the happening of any Suspension Event during the period that the Registration Statement is effective or if as a result of a Suspension Event the Registration Statement or related prospectus contains any untrue statement of a material fact or omits to state any material fact required to be stated therein or necessary to make the statements therein, in light of the circumstances under which they were made (in the case of the prospectus) not misleading, Subscriber agrees that (i) it will immediately discontinue offers and sales of the Acquired Shares under the Registration Statement (excluding, for the avoidance of doubt, sales conducted pursuant to Rule 144) until Subscriber receives copies of a supplemental or amended prospectus (which the Issuer agrees to promptly prepare) that corrects the misstatement(s) or omission(s) referred to above and receives notice that any post-effective amendment has become effective or unless otherwise notified by the Issuer that it may resume such offers and sales, and (ii) it will maintain the confidentiality of any information included in such written notice delivered by the Issuer except (A) for disclosure to the Subscribers employees, agents and professional advisers who need to know such information and are obligated to keep it confidential, (B) for disclosures to the extent required in order to comply with reporting obligations to its limited partners who have agreed to keep such information confidential and (C) as required by law. Notwithstanding anything to the contrary set forth herein, the Issuer shall not, when so advising Subscriber of such events, provide Subscriber with any material, non-public information regarding the Issuer other than to the extent that providing notice to Subscriber of the occurrence of such events would constitute material, non-public information regarding the Issuer. If so directed by the Issuer, Subscriber will deliver to the Issuer or, in Subscribers sole discretion destroy, all copies of the prospectus covering the Acquired Shares in Subscribers possession; provided, however, that this obligation to deliver or destroy all copies of the prospectus covering the Acquired Shares shall not apply (i) to the extent Subscriber is required to retain a copy of such prospectus (a) in order to comply with applicable legal, regulatory, self-regulatory or professional requirements or (b) in accordance with a bona fide pre-existing document retention policy or (ii) to copies stored electronically on archival servers as a result of automatic data back-up.
5.3.2 Opt-Out Notice. Subscriber may deliver written notice (including via email) (an Opt-Out Notice) to the Issuer requesting that Subscriber not receive notices from the Issuer otherwise required by this Section 5; provided, however, that Subscriber may later revoke any such Opt-Out Notice in writing. Following receipt of an Opt-Out Notice from Subscriber (unless subsequently revoked), (i) the Issuer shall not deliver any such notices to Subscriber and Subscriber shall no longer be entitled to the rights associated with any such notice and (ii) each time prior to Subscribers intended use of an effective registration statement, Subscriber will notify the Issuer in writing at least two (2) business days in advance of such intended use, and if a notice of a Suspension Event was previously delivered (or would have been delivered but for the provisions of this Section 5.3.2) and the related suspension period remains in effect, the Issuer will so notify Subscriber, within one (1) business day of Subscribers notification to the Issuer, by delivering to Subscriber a copy of such notice of Suspension Event that would have been provided, and thereafter will provide
E-14
Table of Contents
Subscriber with the related notice of the conclusion of such Suspension Event immediately upon its availability, and Subscriber shall comply with any restrictions on using such Registration Statement during such Suspension Event.
5.4 Subscriber Indemnification. The Issuer agrees to indemnify and hold Subscriber, each person, if any, who controls Subscriber within the meaning of either Section 15 of the Securities Act or Section 20 of the Exchange Act, and each affiliate of Subscriber within the meaning of Rule 405 under the Securities Act, and each underwriter pursuant to the applicable underwriting agreement with such underwriter, and each broker, placement agent or sales agent to or through which Subscriber effects or executes the resale of any Acquired Shares, harmless against any and all losses, claims, damages and liabilities (including, without limitation, any legal or other expenses reasonably incurred in connection with defending or investigating any such action or claim) incurred by Subscriber directly that are caused by any untrue statement or alleged untrue statement of a material fact contained in the Registration Statement or any other registration statement which covers Registrable Securities of Subscriber (including, in each case, the prospectus contained therein) or any amendment thereof (including the prospectus contained therein) or caused by any omission or alleged omission to state therein a material fact necessary in order to make the statements therein (in the case of a prospectus, in the light of the circumstances under which they were made), not misleading, except insofar as the same are caused by or contained in any information or affidavit so furnished in writing to the Issuer by Subscriber expressly for use therein.
5.5 Issuer Indemnification. Subscriber agrees to, severally and not jointly with any Other Subscriber or other person named as a selling stockholder in the Registration Statement, indemnify and hold harmless the Issuer, each person, if any, who controls the Issuer within the meaning of either Section 15 of the Securities Act or Section 20 of the Exchange Act, and each affiliate of the Issuer within the meaning of Rule 405 under the Securities Act, harmless against any and all losses, claims, damages and liabilities (including, without limitation, any legal or other expenses reasonably incurred in connection with defending or investigating any such action or claim) incurred by the Issuer directly that are caused by any untrue statement or alleged untrue statement of a material fact contained in the Registration Statement or any other registration statement which covers Registrable Securities of Subscriber (including, in each case, the prospectus contained therein) or any amendment thereof (including the prospectus contained therein) or caused by any omission or alleged omission to state therein of a material fact necessary in order to make the statements therein (in the case of a prospectus, in light of the circumstances under which they were made), not misleading, insofar as the same are caused by or contained in any information or affidavit so furnished in writing to the Issuer by Subscriber expressly for use therein. Notwithstanding the foregoing, in no event will the Subscribers indemnification obligations under this Section 5, in the aggregate, be greater in amount than the dollar amount of the net proceeds received by such Subscriber upon the sale of the Acquired Shares giving rise to such indemnification obligation.
5.6 Any person or entity entitled to indemnification herein shall (A) give prompt written notice to the indemnifying party of any claim with respect to which it seeks indemnification (provided that the failure to give prompt notice shall not impair any persons or entitys right to indemnification hereunder to the extent such failure has not prejudiced the indemnifying party) and (B) unless in such indemnified partys reasonable judgment a conflict of interest between such indemnified and indemnifying parties may exist with respect to such claim, permit such indemnifying party to assume the defense of such claim with counsel reasonably satisfactory to the indemnified party. If such defense is assumed, the indemnifying party shall not be subject to any liability for any settlement made by the indemnified party without its consent (but such consent shall not be unreasonably withheld, conditioned or delayed). An indemnifying party who is not entitled to, or elects not to, assume the defense of a claim shall not be obligated to pay the fees and expenses of more than one counsel for all parties indemnified by such indemnifying party with respect to such claim, unless in the reasonable judgment of any indemnified party a conflict of interest may exist between such indemnified party and any other of such indemnified parties with respect to such claim. No indemnifying party shall, without the consent of the indemnified party, consent to the entry of any judgment or enter into any settlement which cannot be settled in all respects by the payment of money (and such money is so paid by the indemnifying party pursuant to the terms of
E-15
Table of Contents
such settlement) or which settlement includes a statement or admission of fault and culpability on the part of such indemnified party or which does not include as an unconditional term thereof the giving by the claimant or plaintiff to such indemnified party of a release from all liability in respect to such claim or litigation.
5.7 If the indemnification provided under Sections 5.4 through 5.6 from the indemnifying party is unavailable or insufficient to hold harmless an indemnified party in respect of any losses, claims, damages, liabilities and expenses referred to herein, then the indemnifying party, in lieu of indemnifying the indemnified party, shall contribute to the amount paid or payable by the indemnified party as a result of such losses, claims, damages, liabilities and expenses in such proportion as is appropriate to reflect the relative fault of the indemnifying party and the indemnified party, as well as any other relevant equitable considerations. The relative fault of the indemnifying party and indemnified party shall be determined by reference to, among other things, whether any action in question, including any untrue or alleged untrue statement of a material fact or omission or alleged omission to state a material fact, was made by, or relates to information supplied by, such indemnifying party or indemnified party, and the indemnifying partys and indemnified partys relative intent, knowledge, access to information and opportunity to correct or prevent such action. The amount paid or payable by a party as a result of the losses or other liabilities referred to above shall be deemed to include, subject to the limitations set forth in this Section 5, any legal or other fees, charges or expenses reasonably incurred by such party in connection with any investigation or proceeding. No person guilty of fraudulent misrepresentation (within the meaning of Section 11(f) of the Securities Act) shall be entitled to contribution pursuant to this Section 5.7 from any person who was not guilty of such fraudulent misrepresentation. Each indemnifying partys obligation to make a contribution pursuant to this Section 5.7 shall be individual, not joint and several, and in no event shall the liability of any Subscriber under this Section 5, in the aggregate, be greater in amount than the dollar amount of the net proceeds received by such Subscriber upon the sale of the Acquired Shares giving rise to such indemnification obligation.
6. Termination. This Subscription Agreement shall terminate and be void and of no further force and effect, and all rights and obligations of the parties hereunder shall terminate without any further liability on the part of any party in respect thereof, upon the earliest to occur of (i) such date and time as the Business Combination Agreement is validly terminated in accordance with its terms; (ii) upon the mutual written agreement of each of the parties hereto to terminate this Subscription Agreement; (iii) the event that any conditions contained in Section 2 herein are not satisfied or waived on or prior to the Closing and, as a result thereof, the Subscription and the other transactions contemplated by this Subscription Agreement are not or will not be consummated at the Subscription Closing; and (iv) the first anniversary of the date of this Subscription Agreement if the Subscription Closing has not occurred on or before such first anniversary; provided, that nothing herein will relieve any party from liability for any willful breach hereof prior to the time of termination, and each party will be entitled to any remedies at law or in equity to recover losses, liabilities or damages arising from such breach. The Issuer shall promptly notify Subscriber of the termination of the Business Combination Agreement promptly after the termination of such agreement.
7. No Short Sales. Subscriber agrees that, from the date of this Subscription Agreement until the Closing or the earlier termination of this Subscription Agreement, none of Subscriber or any person or entity acting on behalf of Subscriber pursuant to any understanding with Subscriber will engage in any Short Sales with respect to securities of the Issuer. For the purposes hereof, Short Sales shall include, without limitation, all short sales as defined in Rule 200 promulgated under Regulation SHO under the Exchange Act, and all types of direct and indirect stock pledges (other than pledges in the ordinary course of business as part of prime brokerage arrangements), forward sale contracts, options, puts, calls, swaps and similar arrangements (including on a total return basis), including through non-U.S. broker dealers or foreign regulated brokers. This Section 7 shall not apply to any sale (including the exercise of any redemption right) of securities of the Issuer (i) held by the Subscriber, its controlled affiliates or any person or entity acting on behalf of Subscriber or any of its controlled affiliates prior to the execution of this Subscription Agreement or (ii) purchased by Subscriber, its controlled affiliates or any person or entity acting on behalf of Subscriber or any of its controlled affiliates in open market transactions after the execution of this Subscription Agreement. Further, for the avoidance of doubt, this
E-16
Table of Contents
Section 7 shall not apply to ordinary course, non-speculative hedging transactions. Notwithstanding the foregoing, (a) nothing herein shall prohibit other entities under common management with Subscriber that have no knowledge of this Subscription Agreement or of Subscribers participation in the Offering (including Subscribers affiliates) from entering into any Short Sales and (b) in the case of a Subscriber that is a multi-managed investment vehicle whereby separate portfolio managers manage separate portions of such Subscribers assets and the portfolio managers have no knowledge of the investment decisions made by the portfolio managers managing other portions of such Subscribers assets, then, in each case, the representation set forth above shall only apply with respect to the portion of assets managed by the portfolio manager that made the investment decision to purchase the Acquired Shares covered by this Subscription Agreement.
8. Trust Account Waiver. Subscriber acknowledges that the Issuer is a blank check company with the powers and privileges to effect a merger, share exchange, asset acquisition, share purchase, reorganization or similar business combination involving the Issuer and one or more businesses. Subscriber further acknowledges that, as described in the Issuers prospectus relating to its initial public offering dated June 29, 2020, available at www.sec.gov, substantially all of the Issuers assets consist of the cash proceeds of the Issuers initial public offering and private placements of its securities, and substantially all of those proceeds have been deposited in a trust account (the Trust Account) for the benefit of the Issuer, its public stockholders and the underwriters of the Issuers initial public offering. For and in consideration of the Issuer entering into this Subscription Agreement, the receipt and sufficiency of which are hereby acknowledged, Subscriber, on behalf of itself, and its affiliates and Representatives (acting on behalf of Subscriber), hereby irrevocably waives any and all right, title and interest, or any claim of any kind they have or may have in the future as a result of, or arising out of, this Subscription Agreement, in or to any monies held in the Trust Account, and agrees not to seek recourse or make or bring any action, suit, claim or other proceeding against the Trust Account as a result of, or arising out of, this Subscription Agreement, the transactions contemplated hereby or the Acquired Shares, regardless of whether such claim arises based on contract, tort, equity or any other theory of legal liability. Subscriber acknowledges and agrees that it shall not have any redemption rights with respect to the Acquired Shares pursuant to the Issuers organizational documents in connection with the Transactions or any other business combination, any subsequent liquidation of the Trust Account or the Issuer or otherwise. In the event Subscriber has any claim against the Issuer as a result of, or arising out of, this Subscription Agreement, the transactions contemplated hereby or the Acquired Shares, it shall pursue such claim solely against the Issuer and its assets outside the Trust Account and not against the Trust Account or any monies or other assets in the Trust Account; provided, however, that nothing in this Section 8 shall (x) serve to limit or prohibit the Subscribers right to pursue a claim against Issuer for legal relief against assets held outside the Trust Account, for specific performance or other equitable relief, (y) serve to limit or prohibit any claims that the Subscriber may have in the future against Issuers assets or funds that are not held in the Trust Account (including any funds that have been released from the Trust Account and any assets that have been purchased or acquired with any such funds), or (z) be deemed to limit Subscribers right, title, interest or claim to the Trust Account by virtue of Subscribers record or beneficial ownership of Class A Shares of the Issuer acquired by any means other than pursuant to this Subscription Agreement.
9. Issuers Covenants. With a view to making available to Subscriber the benefits of Rule 144 promulgated under the Securities Act or any other similar rule or regulation of the Commission that may at any time permit Subscriber to sell securities of the Issuer to the public without registration, the Issuer agrees, until the Acquired Shares are sold by Subscriber, to:
(a) make and keep public information available, as those terms are understood and defined in Rule 144;
(b) file with the Commission in a timely manner all reports and other documents required of the Issuer under the Securities Act and the Exchange Act so long as the Issuer remains subject to such requirements and the filing of such reports and other documents is required for the applicable provisions of Rule 144;
(c) furnish to Subscriber so long as it owns the Acquired Shares, as promptly as reasonably practicable upon request, (x) a written statement by the Issuer, if true, that it has complied with the reporting requirements of Rule
E-17
Table of Contents
144, the Securities Act and the Exchange Act, (y) a copy of the most recent annual or quarterly report of the Issuer and such other reports and documents so filed by the Issuer with the Commission and (z) such other information as may be reasonably requested to permit Subscriber to sell such securities pursuant to Rule 144 without registration; and
(d) in connection with a sale by Subscriber pursuant to Rule 144, if the transfer restrictions as set forth on Exhibit A to this Subscription Agreement are no longer required by the Securities Act or any applicable state securities laws, upon request of Subscriber, the Issuer shall use its commercially reasonable efforts to cooperate with Subscriber to have such transfer restrictions removed, including providing authorization to the Issuers transfer agent.
10. Miscellaneous.
(a) Subscriber acknowledges that (i) the Issuer will rely on the acknowledgments, understandings, agreements, representations and warranties made by Subscriber contained in this Subscription Agreement and (ii) the Agent will rely on the acknowledgments, understandings, agreements, representations and warranties made by Subscriber contained in Section 4 and Section 10 of this Subscription Agreement. Issuer acknowledges that the Subscriber and the Agent will rely on the acknowledgements, understandings, agreements, representations and warranties of Issuer contained in this Subscription Agreement. Prior to the Subscription Closing, Subscriber and the Issuer agree to promptly notify the other party and the Agent if any of its acknowledgments, understandings, agreements, representations and warranties set forth herein are no longer accurate in all material respects.
(b) Each of the Issuer, Subscriber and the Agent is entitled to rely upon this Subscription Agreement and is irrevocably authorized to produce this Subscription Agreement or a copy hereof to any interested party in any administrative or legal proceeding or official inquiry with respect to the matters covered hereby; provided, however, that the foregoing shall not give the Agent any rights other than those expressly set forth herein. Disclosure of Subscribers name shall be subject to the provisions set forth in Section 10(q) of this Subscription Agreement.
(c) Neither this Subscription Agreement nor any rights that may accrue to Subscriber hereunder may be transferred or assigned other than the transfer and assignment by Subscriber of (i) the Acquired Shares acquired hereunder, if any, subsequent to Subscribers purchase of such Acquired Shares at the Subscription Closing and in accordance with Subscribers representations and warranties herein; (ii) any or all of Subscribers rights and obligations under this Subscription Agreement to its affiliates or to one or more funds or accounts managed by the same investment manager or investment adviser that manages or advises the Subscriber, subject to, if such transfer or assignment is prior to the Subscription Closing, such assignees executing a subscription agreement in substantially the same form as this Subscription Agreement, including with respect to the Purchase Price and other terms and conditions; and (iii) after the Subscription Closing, the Subscribers rights pursuant to Section 5, Section 9 and Section 10 of this Subscription Agreement to any purchaser of the Acquired Shares that receives the Acquired Shares without the removal of the transfer restrictions set forth on Exhibit A of this Subscription Agreement.
(d) All the agreements, representations and warranties made by each party hereto in this Subscription Agreement shall survive the Subscription Closing, in each case, until the expiration of any statute of limitations under applicable law.
(e) The Issuer may request from Subscriber such additional information as the Issuer may deem to be reasonably necessary to evaluate the eligibility of Subscriber to acquire the Acquired Shares and to comply with the Issuers registration obligations under Section 5 of this Subscription Agreement, and Subscriber shall promptly provide such information as may be reasonably requested, to the extent within Subscribers possession and control or otherwise readily available to Subscriber; provided that the Issuer agrees to keep any such information confidential except to the extent required to be disclosed by applicable law.
E-18
Table of Contents
(f) This Subscription Agreement may not be modified, waived or terminated (other than pursuant to Section 6 above) except by an instrument in writing, signed by the party against whom enforcement of such modification, waiver, or termination is sought.
(g) This Subscription Agreement constitutes the entire agreement between the parties with respect to the subject matter hereof, and supersedes all prior agreements and understandings, both written and oral, between the parties with respect to the subject matter hereof.
(h) Except as otherwise provided herein, this Subscription Agreement shall be binding upon, and inure to the benefit of the parties hereto and their heirs, executors, administrators, successors, legal representatives, and permitted assigns, and the agreements, representations, warranties, covenants and acknowledgments contained herein shall be deemed to be made by, and be binding upon, such heirs, executors, administrators, successors, legal representatives and permitted assigns. Except as set forth in Sections 5.4 through 5.7, this Subscription Agreement shall not confer rights or remedies upon any person other than the parties hereto and their respective successors and assigns; provided, however, each of the parties hereby agrees that Agent is an intended third party beneficiary of (i) Section 4 of this Subscription Agreement with respect to the representations, warranties and agreements of the Subscriber contained therein, (ii) the representations, warranties and agreements of the Issuer contained in this Subscription Agreement and (iii) this Section 10.
(i) Subject to Section 10(c), and except as otherwise provided herein, this Subscription Agreement shall be binding upon, and inure to the benefit of the parties hereto and their heirs, executors, administrators, successors, legal representatives, and permitted assigns, and the agreements, representations, warranties, covenants and acknowledgments contained herein shall be deemed to be made by, and be binding upon, such heirs, executors, administrators, successors, legal representatives and permitted assigns.
(j) If any provision of this Subscription Agreement shall be invalid, illegal or unenforceable, the validity, legality or enforceability of the remaining provisions of this Subscription Agreement shall not in any way be affected or impaired thereby and shall continue in full force and effect.
(k) This Subscription Agreement may be executed in one (1) or more counterparts via facsimile or email (including pdf or any by electronic signature complying with the U.S. federal ESIGN Act of 2000, e.g. www.docusign.com or www.echosign.com) or other transmission method and by different parties in separate counterparts, with the same effect as if all parties hereto had signed the same document. All counterparts so executed and delivered shall be construed together and constitute one and the same agreement. Delivery by facsimile or electronic transmission to counsel for the other parties of a counterpart executed by a party shall be deemed to meet the requirements of the previous sentence.
(l) Each party shall pay all of its own expenses in connection with this Subscription Agreement and the transactions contemplated herein.
(m) [Reserved].
(n) Notices. All notices and other communications required or permitted hereunder shall be in writing and either delivered personally, emailed or sent by overnight mail via a reputable overnight carrier, or sent by certified or registered mail, postage prepaid, and shall be deemed to be given and received (i) when so delivered personally, (ii) when sent, with no mail undeliverable or other rejection notice, if sent by email, or (iii) three (3) business days after the date of mailing to the address below or to such other address or addresses as such person
E-19
Table of Contents
may hereafter designate by notice given hereunder. Such communications, to be valid, must be addressed as follows:
(1) if to Subscriber, to such address or addresses set forth on the signature page hereto;
(2) if to the Issuer, to:
GS Acquisition Holdings Corp II
200 West Street
New York, New York 10282
Attention: Thomas R. Knott
David S. Plutzer
Email: tom.knott@gs.com
david.plutzer@gs.com
with a copy to (which copy shall not constitute notice):
Weil, Gotshal & Manges LLP
767 Fifth Avenue
New York, New York 10153
Attention: Michael J. Aiello
Brian Parness
Email: michael.aiello@weil.com
brian.parness@weil.com
(o) This Subscription Agreement, and any action, suit, dispute, controversy or claim arising out of this Subscription Agreement or the validity, interpretation, breach or termination of this Subscription Agreement, shall be governed by and construed in accordance with the internal laws of the State of Delaware regardless of the law that might otherwise govern under applicable principles of conflicts of law thereof.
(p) Each of the parties irrevocably consents to the exclusive jurisdiction and venue of the courts of the State of Delaware or the federal courts located in the State of Delaware in connection with any matter based upon or arising out of this Subscription Agreement, agrees that process may be served upon them in any manner authorized by the laws of the State of Delaware for such person and waives and covenants not to assert or plead any objection which they might otherwise have to such manner of service of process. Each party and any person asserting rights as a third-party beneficiary may do so only if he, she or it hereby waives, and shall not assert as a defense in any legal dispute, that: (a) such person is not personally subject to the jurisdiction of the above named courts for any reason; (b) such Action may not be brought or is not maintainable in such court; (c) such persons property is exempt or immune from execution; (d) such Action is brought in an inconvenient forum; or (e) the venue of such Action is improper. Each party and any person asserting rights as a third-party beneficiary hereby agrees not to commence or prosecute any such action, claim, cause of action or suit other than before one of the above-named courts, nor to make any motion or take any other action seeking or intending to cause the transfer or removal of any such action, claim, cause of action or suit to any court other than one of the above-named courts, whether on the grounds of inconvenient forum or otherwise. Each party hereby consents to service of process in any such proceeding in any manner permitted by Delaware law, and further consents to service of process by nationally recognized overnight courier service guaranteeing overnight delivery, or by registered or certified mail, return receipt requested, at its address specified pursuant to Section 10(n) of this Subscription Agreement. Process in any such suit, Action or proceeding may be served on any party anywhere in the world, whether within or without the jurisdiction of any such court. Notwithstanding the foregoing in this Section 10(p), any party may commence any action, claim, cause of action or suit in a court other than the above-named courts solely for the purpose of enforcing an order or judgment issued by one of the above-named courts.
TO THE EXTENT NOT PROHIBITED BY APPLICABLE LAW WHICH CANNOT BE WAIVED, EACH OF THE PARTIES AND ANY PERSON ASSERTING RIGHTS AS A THIRD-PARTY
E-20
Table of Contents
BENEFICIARY MAY DO SO ONLY IF HE, SHE OR IT IRREVOCABLY AND UNCONDITIONALLY WAIVES ANY AND ALL RIGHT TO TRIAL BY JURY ON ANY CLAIMS OR COUNTERCLAIMS ASSERTED IN ANY LEGAL DISPUTE RELATING TO THIS SUBSCRIPTION AGREEMENT OR THE TRANSACTIONS CONTEMPLATED HEREBY AND FOR ANY COUNTERCLAIM RELATING THERETO, IN EACH CASE WHETHER NOW EXISTING OR HEREAFTER ARISING. IF THE SUBJECT MATTER OF ANY SUCH LEGAL DISPUTE IS ONE IN WHICH THE WAIVER OF JURY TRIAL IS PROHIBITED, NO PARTY NOR ANY PERSON ASSERTING RIGHTS AS A THIRD-PARTY BENEFICIARY SHALL ASSERT IN SUCH LEGAL DISPUTE A NONCOMPULSORY COUNTERCLAIM ARISING OUT OF OR RELATING TO THIS SUBSCRIPTION AGREEMENT. FURTHERMORE, NO PARTY NOR ANY PERSON ASSERTING RIGHTS AS A THIRD-PARTY BENEFICIARY SHALL SEEK TO CONSOLIDATE ANY SUCH LEGAL DISPUTE WITH A SEPARATE ACTION OR OTHER LEGAL PROCEEDING IN WHICH A JURY TRIAL CANNOT BE WAIVED.
(q) The Issuer shall, no later than 9:00 a.m., New York City time, on the first (1st) business day immediately following the date of this Subscription Agreement, issue one or more press releases or file with the Commission a Current Report on Form 8-K (collectively, the Disclosure Document) disclosing all material terms of the transactions contemplated hereby, the Transactions and any other material, nonpublic information that the Issuer has provided to Subscriber at any time prior to the filing of the Disclosure Document unless the Issuer determines, in compliance with applicable laws and regulations, that any of such information is no longer material. From and after the issuance of the Disclosure Document, to the Issuers knowledge, Subscriber shall not be in possession of any material, non-public information received from or on behalf of the Issuer or any of its officers, directors or employees. Notwithstanding anything in this Subscription Agreement to the contrary, each party hereto acknowledges and agrees that without the prior written consent of the other party hereto it will not publicly make reference to such other party or any of its affiliates (i) in connection with the Transactions or this Subscription Agreement or (ii) in any promotional materials, media, or similar circumstances, except, in each case, as required by law or regulation or at the request of the Staff of the Commission or regulatory agency or under the regulations of the NYSE, including: (a) as required by the federal securities laws, (b) in connection with the filing by the Issuer of this Subscription Agreement (or a form of this Subscription Agreement) with the Commission or (c) in connection with the filing by the Issuer of a registration statement under the Securities Act or a proxy statement under Schedule 14A and related proxy materials with the Commission with respect to the Transactions.
(r) Except as expressly set forth in this Subscription Agreement, no former, current or future equity holders, controlling persons, directors, officers, employees, agents, affiliates, members, managers, general or limited partners, Representatives or assignees of Subscriber or any former, current or future equity holder, controlling person, director, officer, employee, agent, affiliate, member, manager, general or limited partner, Representative or assignee of any of the foregoing, shall have any obligation to the Issuer or to any other person hereunder in connection with the transactions contemplated hereby.
(s) The obligations of Subscriber and each Other Subscriber in connection with the Offering are several and not joint, and Subscriber shall not be responsible in any way for the performance of the obligations of any Other Subscriber in connection with the Offering. Nothing contained herein or in any Other Subscription Agreement, and no action taken by Subscriber or any Other Subscriber pursuant hereto or thereto, shall be deemed to constitute the Subscriber and Other Subscribers as a partnership, an association, a joint venture or any other kind of entity, or create a presumption that the Subscribers and Other Subscribers are in any way acting in concert or as a group with respect to such obligations or the transactions contemplated hereby. Subscriber and each Other Subscriber shall be entitled to independently protect and enforce its rights, and it shall not be necessary for any Other Subscriber to be joined as an additional party in any proceeding for such purpose.
(t) No failure or delay by a party hereto in exercising any right, power or remedy under this Subscription Agreement, and no course of dealing between the parties hereto, shall operate as a waiver of any such right,
E-21
Table of Contents
power or remedy of such party. No single or partial exercise of any right, power or remedy under this Subscription Agreement by a party hereto, nor any abandonment or discontinuance of steps to enforce any such right, power or remedy, shall preclude such party from any other or further exercise thereof or the exercise of any other right, power or remedy hereunder. The election of any remedy by a party hereto shall not constitute a waiver of the right of such party to pursue other available remedies. No notice to or demand on a party not expressly required under this Subscription Agreement shall entitle the party receiving such notice or demand to any other or further notice or demand in similar or other circumstances or constitute a waiver of the rights of the party giving such notice or demand to any other or further action in any circumstances without such notice or demand.
(u) Remedies.
(1) The parties agree that irreparable damage would occur if this Subscription Agreement was not performed or the Subscription Closing is not consummated in accordance with its specific terms or was otherwise breached and that money damages or other legal remedies would not be an adequate remedy for any such damage. It is accordingly agreed that the parties hereto shall be entitled to equitable relief, including in the form of an injunction or injunctions, to prevent breaches or threatened breaches of this Subscription Agreement and to enforce specifically the terms and provisions of this Subscription Agreement in an appropriate court of competent jurisdiction as set forth in Section 10(p), this being in addition to any other remedy to which any party is entitled at law or in equity, including money damages (but subject to Section 8). The right to specific enforcement shall include the right of the parties hereto to cause the other parties hereto to cause the transactions contemplated hereby to be consummated on the terms and subject to the conditions and limitations set forth in this Subscription Agreement. The parties hereto further agree (i) to waive any requirement for the security or posting of any bond in connection with any such equitable remedy, (ii) not to assert that a remedy of specific enforcement pursuant to this Section 10(u) is unenforceable, invalid, contrary to applicable law or inequitable for any reason and (iii) to waive any defenses in any action for specific performance, including the defense that a remedy at law would be adequate.
(2) The parties acknowledge and agree that this Section 10(u) is an integral part of the transactions contemplated hereby and without that right, the parties hereto would not have entered into this Subscription Agreement.
(v) Massachusetts Business Trust. If Subscriber is a Massachusetts Business Trust, a copy of the Declaration of Trust of Subscriber or any affiliate thereof is on file with the Secretary of State of the Commonwealth of Massachusetts and notice is hereby given that the Subscription Agreement is executed on behalf of the trustees of Subscriber or any affiliate thereof as trustees and not individually and that the obligations of the Subscription Agreement are not binding on any of the trustees, officers or stockholders of Subscriber or any affiliate thereof individually but are binding only upon Subscriber or any affiliate thereof and its assets and property.
[Signature Pages Follow]
E-22
Table of Contents
IN WITNESS WHEREOF, each of the Issuer and Subscriber has executed or caused this Subscription Agreement to be executed by its duly authorized representative as of the date set forth below.
| GS ACQUISITION HOLDINGS CORP II | ||||
| By: | ||||
| Name: | Thomas R. Knott | |||
| Title: | Authorized Signatory | |||
[Signature Page to Subscription Agreement]
Table of Contents
Accepted and agreed this ______ day of ______, 2021.
SUBSCRIBER
| Signature of Subscriber: | Signature of Joint Subscriber, if applicable: | |||||||
| By: | By: | |||||||
| Name: | Name: | |||||||
| Title: | Title: | |||||||
| Name of Subscriber: | Name of Joint Subscriber, if applicable: | |||
|
|
|
|||
| (Please print. Please indicate name and capacity of person signing above) |
(Please Print. Please indicate name and capacity of person signing above) |
|||
|
|
| Name in which securities are to be registered (if different from the name of Subscriber listed directly above): |
| Email Address: |
||
| If there are joint investors, please check one: |
||
| ☐ Joint Tenants with Rights of Survivorship |
||
| ☐ Tenants-in-Common |
||
| ☐ Community Property |
||
| Subscribers EIN: __________________________ |
Joint Subscribers EIN: ________________ |
|
[Signature Page to Subscription Agreement]
Table of Contents
| Business Address-Street: | Mailing Address-Street (if different): | |||
|
|
|
|||
|
|
|
|||
| City, State, Zip: | City, State, Zip: | |||
| Attn: | Attn: | |||
| Telephone No.: __________________________ | Telephone No.: __________________________ | |||
| Facsimile No.: __________________________ | Facsimile No.: __________________________ | |||
Aggregate Number of Shares subscribed for:
_____________________
Aggregate Purchase Price: $ ______________________ .
You must pay the Purchase Price by wire transfer of United States dollars in immediately available funds, to be held in escrow until the Subscription Closing, to the account specified by the Issuer in the Closing Notice.
[Signature Page to Subscription Agreement]
Table of Contents
SCHEDULE A
ELIGIBILITY REPRESENTATIONS OF SUBSCRIBER
This page should be completed by Subscriber
and constitutes a part of the Subscription Agreement.
| A. | INSTITUTIONAL ACCREDITED INVESTOR STATUS (Please check the applicable subparagraphs): |
☐ We are an institutional accredited investor (as described in Rule 501(a)(1), (2), (3) or (7) under the Securities Act) and have marked and initialed the appropriate box on the following page indicating the provision under which we qualify as an accredited investor.
*** AND ***
| B. | AFFILIATE STATUS |
(Please check the applicable box) SUBSCRIBER:
| ☐ | is: |
| ☐ | is not: |
an affiliate (as defined in Rule 144 under the Securities Act) of the Issuer or acting on behalf of an affiliate of the Issuer.
*** AND ***
| C. | INSTITUTIONAL ACCOUNT STATUS |
(Please check the applicable box) SUBSCRIBER:
| ☐ | is: |
| ☐ | is not: |
an Institutional Account (as defined in FINRA Rule 4512(c)).
This page should be completed by Subscriber
and constitutes a part of the Subscription Agreement.
Table of Contents
Rule 501(a) under the Securities Act, in relevant part, states that an accredited investor shall mean any person who comes within any of the below listed categories, or who the Issuer reasonably believes comes within any of the below listed categories, at the time of the sale of the securities to that person. Subscriber has indicated, by marking and initialing the appropriate box below, the provision(s) below which apply to Subscriber and under which Subscriber accordingly qualifies as an accredited investor.
☐ Any bank as defined in section 3(a)(2) of the Securities Act, or any savings and loan association or other institution as defined in section 3(a)(5)(A) of the Securities Act whether acting in its individual or fiduciary capacity;
☐ Any broker or dealer registered pursuant to section 15 of the Securities Exchange Act of 1934, as amended;
☐ Any insurance company as defined in section 2(a)(13) of the Securities Act;
☐ Any investment company registered under the Investment Company Act of 1940, as amended (the Investment Company Act) or a business development company as defined in section 2(a)(48) of the Investment Company Act;
☐ Any Small Business Investment Company licensed by the U.S. Small Business Administration under section 301(c) or (d) of the Small Business Investment Act of 1958, as amended;
☐ Any plan established and maintained by a state, its political subdivisions, or any agency or instrumentality of a state or its political subdivisions, for the benefit of its employees, if such plan has total assets in excess of $5,000,000;
☐ Any employee benefit plan within the meaning of the Employee Retirement Income Security Act of 1974, as amended (ERISA), if (i) the investment decision is made by a plan fiduciary, as defined in section 3(21) of ERISA, which is either a bank, a savings and loan association, an insurance company, or a registered investment adviser, (ii) the employee benefit plan has total assets in excess of $5,000,000 or, (iii) such plan is a self-directed plan, with investment decisions made solely by persons that are accredited investors;
☐ Any private business development company as defined in section 202(a)(22) of the Investment Advisers Act of 1940, as amended;
☐ Any (i) corporation, limited liability company or partnership, (ii) Massachusetts or similar business trust, or (iii) organization described in section 501(c)(3) of the Internal Revenue Code of 1986, as amended, not formed for the specific purpose of acquiring the securities offered, and with total assets in excess of $5,000,000; or
☐ Any trust, with total assets in excess of $5,000,000, not formed for the specific purpose of acquiring the securities offered, whose purchase is directed by a sophisticated person as described in Section 230.506(b)(2)(ii) of Regulation D.
Table of Contents
Exhibit A
NO TRANSFER, SALE, ASSIGNMENT, PLEDGE, HYPOTHECATION OR OTHER DISPOSITION OF THE ACQUIRED SHARES OR ANY INTEREST OR PARTICIPATION THEREIN MAY BE MADE EXCEPT (A) PURSUANT TO AN EFFECTIVE REGISTRATION STATEMENT UNDER THE SECURITIES ACT OF 1933 (THE ACT) OR (B) PURSUANT TO AN EXEMPTION FROM REGISTRATION UNDER THE ACT AND APPLICABLE STATE SECURITIES LAWS AND, IN THE CASE OF CLAUSE (B), UNLESS, IF THE ISSUER REQUESTS, THE ISSUER RECEIVES AN OPINION OF COUNSEL IN FORM AND SUBSTANCE REASONABLY SATISFACTORY TO THE ISSUER TO THE EFFECT THAT REGISTRATION IS NOT REQUIRED UNDER THE ACT.
Any transferee of the Acquired Shares or any interest therein, by its acceptance thereof, shall be deemed to have made the representations set forth in Section 4 of the Subscription Agreement (other than the representations set forth in Section 4(g), the first two sentences of Section 4(k) and Section 4(u) (collectively, the Excluded Representations)). The Issuer shall not be required to register the transfer of any Acquired Shares to any transferee unless the Issuer receives from the proposed transferee a written instrument in form and substance reasonably satisfactory to the Issuer in which such transferee makes the representations and warranties set forth in Section 4 of the Subscription Agreement (other than the Excluded Representations) and, if the Issuer so requests, an opinion of counsel in form and substance reasonably satisfactory to the Issuer to the effect that registration under the Securities Act is not required in connection with such transfer; provided, that no opinion of counsel will be required for a pledge of the Acquired Shares if the Issuer receives a representation from the pledgor and pledgee that the pledge is a bona fide pledge and, in the event that the pledgee acquires the shares that are the subject of the pledge, the pledgee agrees to the representations and warranties set forth in Section 4 of the Subscription Agreement. The foregoing shall not apply to any sale of the Acquired Shares made in accordance with Rule 144 or pursuant to an effective registration statement; provided, that the transferor of the Acquired Shares provides to the Issuer such representations with respect to compliance as is reasonably requested by the Issuer.
Table of Contents
Final Form
FORM OF AMENDED AND RESTATED
REGISTRATION RIGHTS AGREEMENT
THIS AMENDED AND RESTATED REGISTRATION RIGHTS AGREEMENT (this Agreement), dated as of [●], 2021, is made and entered into by and among Mirion Technologies, Inc., a Delaware corporation (the Company), GS Sponsor II LLC, a Delaware limited liability company (the GS Sponsor Member), GS Acquisition Holdings II Employee Participation LLC, a Delaware limited liability company (the GS Employee Vehicle, and together with the GS Sponsor Member, the GS Founder Share Members), GSAM Holdings LLC, a Delaware limited liability company (the GS Equity Investor), the Charterhouse Holders (as defined below) and the Target Shareholders (as defined below) listed on the signature pages hereto. Such Target Shareholders, together with the GS Founder Share Members, the GS Equity Investor and the Charterhouse Holders and any person or entity who hereafter becomes a party to this Agreement pursuant to Sections 6.2 or 6.10 of this Agreement are each referred to herein as a Holder and collectively the Holders.
RECITALS
WHEREAS, on June 29, 2020, the Company and the GS Sponsor Member entered into that certain Warrant Purchase Agreement (the Warrant Purchase Agreement), pursuant to which the GS Sponsor Member purchased 8,500,000 warrants (the Sponsor Warrants) in a private placement transaction occurring simultaneously with the closing of the Companys initial public offering on June 29, 2020;
WHEREAS, on June 29, 2020, the Company and the GS Founder Share Members entered into that certain Registration Rights Agreement (the Existing Registration Rights Agreement), pursuant to which the Company granted the GS Founder Share Members certain registration rights with respect to certain securities of the Company;
WHEREAS, the GS Founder Share Members own an aggregate of 18,750,000 shares of the Companys Class A Common Stock (as defined below) received upon the conversion of a like amount of shares of the Companys former Class B common stock (the Founder Shares);
WHEREAS, upon the closing of the transactions (the Transactions) contemplated by that certain Business Combination Agreement, dated as of June 17, 2021 (the Business Combination Agreement), by and among the Company, Mirion Technologies (TopCo), Ltd., a Jersey private company limited by shares (Mirion), CCP IX LP No. 1, CCP IX LP No. 2, CCP IX Co-Investment LP and CCP IX Co-Investment No. 2 LP (collectively, the Charterhouse Holders), each of the other persons and entities set forth on Annex I thereto (together with the Charterhouse Holders, the Supporting Company Holders) and the other holders of ordinary shares of Mirion from time to time becoming a party thereto (each, a Joining Seller and collectively, the Joining Sellers and, together with each Supporting Company Holder, each, a Target Shareholder, and collectively, the Target Shareholders), the Founder Shares were converted into shares of the Companys Class A common stock, par value $0.0001 per share (the Class A Common Stock), on a one-for-one basis;
WHEREAS, in connection with the Transactions, certain Target Shareholders received shares of Class A Common Stock and certain Target Shareholders received shares of the Companys Class B common stock, par value $0.0001 per share (the Class B Common Stock and, together with the Class A Common Stock, the Common Stock), as part of Paired Interests (as defined in the Companys Amended and Restated Certificate of Incorporation as in effect upon the closing of the Transactions (as it may be amended, restated or otherwise modified from time to time, the Company Charter));
F-1
Table of Contents
WHEREAS, on the date hereof, the GS Equity Investor and certain other investors (such other investors, collectively, the Other PIPE Investors) purchased 90,000,000 shares of the Companys Class A Common Stock in a transaction exempt from registration under the Securities Act (the PIPE Shares);
WHEREAS, on the date hereof, the GS Equity Investor purchased [●] shares of the Companys Class A Common Stock in a transaction exempt from registration under the Securities Act (the Backstop Shares);
WHEREAS, on the date hereof, the GS Equity Investor purchased [●] shares of the Companys Class A Common Stock from certain of the Charterhouse Holders and Target Shareholders (the Option Shares);
WHEREAS, pursuant to Section 5.6 of the Existing Registration Rights Agreement, the provisions, covenants and conditions set forth therein may be amended or modified upon the written consent of the Company and the Holders (as defined in the Existing Registration Rights Agreement) of at least a majority-in-interest of the Registrable Securities (as defined in the Existing Registration Rights Agreement) at the time in question; and
WHEREAS, the GS Founder Share Members constituted a majority-in-interest of the Registrable Securities under the Existing Registration Rights Agreement, and the Company and the GS Founder Share Members each desire to amend and restate the Existing Registration Rights Agreement, in order to provide the Holders with registration rights with respect to the Registrable Securities on the terms set forth herein.
NOW, THEREFORE, in consideration of the representations, covenants and agreements contained herein, and certain other good and valuable consideration, the receipt and sufficiency of which are hereby acknowledged, the parties hereto, intending to be legally bound, hereby agree as follows:
ARTICLE I
DEFINITIONS
1.1 Definitions. The terms defined in this Article I shall, for all purposes of this Agreement, have the respective meanings set forth below:
Additional Holder shall have the meaning given in Section 6.10.
Additional Holder Common Stock shall have the meaning given in Section 6.10.
Adverse Disclosure shall mean any public disclosure of material non-public information, which disclosure, in the good faith judgment of the Board (i) would be required to be made in any Registration Statement or Prospectus in order for the applicable Registration Statement or Prospectus not to contain any untrue statement of a material fact or omit to state a material fact necessary to make the statements contained therein (in the case of any prospectus and any preliminary prospectus, in the light of the circumstances under which they were made) not misleading, (ii) would not be required to be made at such time if the Registration Statement were not being filed, declared effective or used, as the case may be, and (iii) the Company has a bona fide business purpose for not making such information public.
Affiliate shall mean with respect to a specified person, each other person that directly, or indirectly through one or more intermediaries, controls or is controlled by, or is under common control with, the person specified; provided that no Holder shall be deemed an Affiliate of any other Holder by reason of an investment in the Company or holding of Common Stock (or securities convertible, exercisable or exchangeable for shares of Common Stock). As used in this definition, control (including with correlative meanings, controlled by and under common control with) means possession, directly or indirectly, of power to direct or cause the direction of management or policies (whether through ownership of voting securities or by contract or other agreement).
F-2
Table of Contents
Agreement shall have the meaning given in the Preamble.
Backstop Shares shall have the meaning given in the Recitals hereto.
Block Trade shall have the meaning given in Section 2.4.
Board shall mean the Board of Directors of the Company.
Business Combination Agreement shall have the meaning given in the Recitals hereto.
Change of Control means the transfer to or acquisition by (whether by tender offer, merger, consolidation, division or other similar transaction), in one transaction or a series of related transactions, a person or entity or group of affiliated persons or entities (other than an underwriter pursuant to an offering), of the Companys voting securities if, after such transfer or acquisition, such person, entity or group of affiliated persons or entities would beneficially own (as defined in Rule 13d-3 promulgated under the Exchange Act) more than 50% of the outstanding voting securities of the Company.
Charterhouse Demand Lock-up Period shall have the meaning given in Section 2.1.1.
Charterhouse Demand Period shall have the meaning given in Section 2.1.1.
Charterhouse Demand Right shall have the meaning given in Section 2.1.1.
Charterhouse Director means the Charterhouse Director (as defined in the Charterhouse Director Nomination Agreement).
Charterhouse Director Nomination Agreement shall mean that certain Director Nomination Agreement, dated as of the date hereof and as may be amended, restated or otherwise modified from time to time, by and among the Company and the Charterhouse Holders.
Charterhouse Holders shall have the meaning given in the Recitals hereto.
Charterhouse Lock-up Period shall mean, with respect to any shares of Common Stock received by the Charterhouse Holders pursuant to the Business Combination Agreement (the Charterhouse Lock-up Securities), the period ending on the 181st day after the Closing Date; provided, that any Charterhouse Lock-up Securities Transferred to the GS Equity Investor pursuant to the Option Agreement (as defined in the Business Combination Agreement) shall not be subject to the Charterhouse Lock-up Period.
Charterhouse Lock-up Securities shall have the meaning set forth in the definition of Charterhouse Lock-up Period.
Class A Common Stock shall have the meaning given in the Recitals hereto.
Class B Common Stock shall have the meaning given in the Recitals hereto.
Closing shall have the meaning given in the Business Combination Agreement.
Closing Date shall have the meaning given in the Business Combination Agreement.
Commission shall mean the U.S. Securities and Exchange Commission.
Common Stock shall have the meaning given in the Recitals hereto.
F-3
Table of Contents
Company shall have the meaning given in the Preamble and includes the Companys successors by recapitalization, merger, consolidation, spin-off and reorganization or similar transaction.
Company Charter shall have the meaning given in the Recitals hereto.
Demand Registration shall have the meaning given in Section 2.1.2.
Demanding Holder shall have the meaning given in Section 2.1.2.
Exchange Act shall mean the Securities Exchange Act of 1934, as it may be amended from time to time.
Excluded Registration shall mean a Registration Statement (or any registered offering with respect thereto) (i) filed in connection with any employee stock option or other benefit plan (including any Form S-8), (ii) on Form S-4 (or similar form that relates to a transaction subject to Rule 145 under the Securities Act or any successor rule thereto), (iii) for an offering of debt, warrants, units or other securities that are convertible into, or exchangeable or exercisable for, equity securities of the Company (or for equity securities issued upon conversion, exchange or exercise of such debt, warrants, units or other securities), (iv) for a dividend reinvestment plan, (v) for the resale of securities issued in connection with a reorganization, merger, acquisition or similar transaction, (vi) in connection with a Charterhouse Demand Right or (vii) in connection with any Demand Registration or Block Trade.
Filing Date shall have the meaning given in Section 2.3.1.
Form S-1 Shelf shall have the meaning given in Section 2.3.1.
Form S-3 Shelf shall have the meaning given in Section 2.3.1.
Founder Shares shall have the meaning given in the Recitals hereto and shall be deemed to include the shares of Class A Common Stock issued upon conversion thereof.
Founder Shares Lock-up Period shall mean, with respect to the Founder Shares, the period ending on the earlier of (A) one year after the Closing Date and (B) following the Closing Date, (x) if the last reported sale price of the Class A Common Stock equals or exceeds $12.00 per share (as adjusted for stock splits, stock dividends, reorganizations, recapitalizations and the like) for any 20 trading days within any 30-trading day period commencing at least 150 days after the Closing Date or (y) the date on which the Company completes a liquidation, merger, stock exchange, reorganization or other similar transaction that results in all of the Companys public stockholders having the right to exchange their shares of Class A Common Stock for cash, securities or other property.
GS Director means one of the SPAC Sponsor Directors (as defined in the GS Director Nomination Agreement).
GS Director Nomination Agreement shall mean that certain Director Nomination Agreement, dated as of the date hereof and as may be amended, restated or otherwise modified from time to time, by and among the Company and the GS Sponsor Member.
GS Employee Vehicle shall have the meaning given in the Preamble.
GS Equity Investor shall have the meaning given in the Preamble.
F-4
Table of Contents
GS Founder Share Members shall have the meaning given in the Preamble.
GS Holders means the GS Founder Share Members and the GS Equity Investor.
GS Sponsor Member shall have the meaning given in the Preamble.
Holder Information shall have the meaning given in Section 4.1.2.
Holders shall have the meaning given in the Preamble, for so long as such person or entity holds any Registrable Securities.
Insider Letter shall mean that certain amended and restated letter agreement, dated as of the date hereof, by and among the Company, the GS Founder Share Members and each of the other parties thereto.
Joinder shall have the meaning given in Section 6.2.6.
Joining Seller shall have the meaning given in the Recitals hereto.
Lock-up Period shall mean, as applicable, (i) the Founder Shares Lock-up Period, (ii) the Sponsor Warrant Lock-up Period, (iii) the PIPE Shares Lock-up Period, (iv) the Target Shareholder Lock-up Period, (v) the Charterhouse Demand Lock-up Period, and (vi) the Charterhouse Lock-up Period.
Maximum Number of Securities shall have the meaning given in Section 2.6.
Mirion shall have the meaning given in the Recitals hereto.
Misstatement shall mean an untrue statement of a material fact or an omission to state a material fact required to be stated in a Registration Statement or Prospectus or necessary to make the statements in a Registration Statement or Prospectus (in the case of a Prospectus, in the light of the circumstances under which they were made) not misleading.
Option Shares shall have the meaning given in the Recitals hereto.
Other PIPE Investors shall have the meaning given in the Recitals hereto.
Other PIPE Investors Subscription Agreements shall mean the respective Subscription Agreements, each dated as of June 17, 2021, by and between the Company and the Other PIPE Investors.
Paired Interest shall have the meaning given in the Recitals hereto.
Permitted Transferees shall mean:
(A) with respect to the GS Founder Share Members and their respective Permitted Transferees, any person or entity to whom the GS Founder Share Members are permitted to transfer such Registrable Securities prior to the expiration of the Founder Shares Lock-up Period or Sponsor Warrants Lock-up Period, as the case may be, pursuant to and in accordance with the Insider Letter and any other applicable agreement between such GS Founder Share Member and/or their respective Permitted Transferees and the Company and to any transferee thereafter;
(B) with respect to the GS Equity Investor and its Permitted Transferees, any employees, investor partners or clients of the GS Equity Investor or its Affiliates, who, for the avoidance of doubt, shall not be subject to any Lock-up Period upon a valid transfer of Registrable Securities; and
(C) with respect to the Target Shareholders and their respective Permitted Transferees, any transferee permitted under clauses (i) through (viii) of Section 5.1(b).
F-5
Table of Contents
Piggyback-Eligible Holder shall mean, as of the applicable time, a Holder owning, collectively with its Affiliates, at least 100,000 shares of Common Stock (as such number may be adjusted for stock splits, combinations, recapitalizations, stock dividends or similar transactions that effect a change in the number of outstanding shares of the Company).
Piggyback Registration shall have the meaning given in Section 2.2.
PIPE Shares shall have the meaning given in the Recitals hereto.
PIPE Shares Lock-up Period shall mean, with respect to the PIPE Shares held by the GS Equity Investor and its Affiliates (excluding Permitted Transferees), the period ending 180 days after the Closing Date. For the avoidance of doubt, any PIPE Shares distributed by the GS Equity Investor or its Affiliates to their respective Permitted Transferees shall not be subject to the PIPE Shares Lock-up Period.
Prospectus shall mean the prospectus included in any Registration Statement, as supplemented by any and all prospectus supplements and as amended by any and all post-effective amendments and including all material incorporated by reference in such prospectus.
Registrable Security shall mean (a) any shares of Class A Common Stock either outstanding or underlying warrants to purchase shares of Class A Common Stock held by a Holder immediately following the Closing (including any securities distributable pursuant to the Business Combination Agreement and any PIPE Shares), (b) shares of Class A Common Stock issued or issuable upon the exchange of any Paired Interests held by a Holder immediately following the Closing (including any Paired Interests distributable pursuant to the Business Combination Agreement), (c) any outstanding shares of Common Stock or any other equity security (including warrants to purchase shares of Common Stock and shares of Common Stock issued or issuable upon the exercise of any equity security) of the Company acquired by any Charterhouse Holder following the date hereof to the extent that such securities are restricted securities (as defined in Rule 144) or are considered to be held by such Charterhouse Holder as an affiliate (as defined in Rule 144) of the Company, (d) any Additional Holder Common Stock and (e) any other equity security of the Company or any of its subsidiaries issued or issuable with respect to any securities referenced in clause (a), (b), (c) or (d) above by way of a stock dividend or stock split or in connection with a recapitalization, merger, consolidation, spin-off, reorganization or similar transaction; provided, however, that, as to any particular Registrable Security, such securities shall cease to be Registrable Securities upon the earliest to occur of:
(i) a Registration Statement with respect to the sale of such securities shall have become effective under the Securities Act and such securities shall have been sold, transferred, disposed of or exchanged in accordance with such Registration Statement by the applicable Holder;
(ii) (A) such securities shall have been otherwise transferred, (B) new certificates for such securities not bearing (or book entry positions not subject to) a legend restricting further transfer shall have been delivered by the Company and (C) subsequent public distribution of such securities shall not require registration under the Securities Act; provided, that this clause (ii) shall not apply to securities held by Permitted Transferees to the extent subsequent distribution of such securities by such Permitted Transferees requires registration under the Securities Act;
(iii) such securities shall have ceased to be outstanding;
(iv) such securities have been sold without registration pursuant to Section 4(a)(1) of the Securities Act or Rule 144 or Rule 145 promulgated under the Securities Act (or any successor rule promulgated thereafter by the Commission);
(v) such securities have been sold to, or through, a broker, dealer or underwriter in a public distribution or other public securities transaction; or
(vi) in the case of Registrable Securities included under clause (c) of the definition thereof, such securities may be sold pursuant to Section 4(a)(1) of the Securities Act or Rule 144 or Rule 145 promulgated under the
F-6
Table of Contents
Securities Act (or any successor rule promulgated thereafter by the Commission) without any volume, current public information or manner of sale restrictions.
Registration shall mean a registration effected by preparing and filing a registration statement or similar document in compliance with the requirements of the Securities Act, and the applicable rules and regulations promulgated thereunder, and such registration statement becoming effective.
Registration Expenses shall mean the out-of-pocket expenses of a Registration or Underwritten Offering, excluding Selling Expenses, and including, without limitation, the following:
(A) all registration and filing fees (including fees with respect to filings required to be made with the Financial Industry Regulatory Authority, Inc.) and any national securities exchange on which the Class A Common Stock is then listed;
(B) fees and expenses of compliance with securities or blue sky laws (including reasonable fees and disbursements of outside counsel for the Underwriters in connection with blue sky qualifications of Registrable Securities);
(C) printing, messenger, telephone and delivery expenses;
(D) reasonable fees and disbursements of counsel for the Company;
(E) reasonable fees and disbursements of all independent registered public accountants of the Company incurred specifically in connection with such Registration or Underwritten Offering; and
(F) reasonable and documented fees and expenses of one (1) legal counsel selected by a majority-of-interest of Holders participating in such Registration or Underwritten Offering (this clause (F), Holder Counsel Expenses).
Registration Statement shall mean any registration statement that covers the Registrable Securities pursuant to the provisions of this Agreement, including the Prospectus included in such registration statement, amendments (including post-effective amendments) and supplements to such registration statement, and all exhibits to and all material incorporated by reference in such registration statement.
Requesting Holders shall have the meaning given in Section 2.1.2.
Requisite Percentage shall mean (a) with respect to each of the GS Founder Share Members, at least 100,000 of the shares of Common Stock directly held by such GS Founder Share Member immediately following the Closing Date and (b) with respect to the Charterhouse Holders, at least 100,000 of the shares of Common Stock received by the Charterhouse Holders in the aggregate in connection with the Transactions so long as such shares of Common Stock remain Registrable Securities.
Rule 144 shall mean Rule 144 promulgated under the Securities Act.
Securities Act shall mean the Securities Act of 1933, as amended from time to time.
Selling Expenses means all underwriting discounts, selling commissions and stock transfer taxes applicable to the sale of Registrable Securities, and fees and disbursements of counsel for any Holder, except for Holder Counsel Expenses.
Shelf Registration shall mean a registration of securities pursuant to a registration statement filed with the Commission in accordance with and pursuant to Rule 415 promulgated under the Securities Act (or any successor rule then in effect).
F-7
Table of Contents
Sponsor Underwriter Lock-up Period shall mean a period of time no less than 45 days and no longer than 90 days beginning on the date of the pricing of an underwritten offering pursuant to the Charterhouse Holders exercise of its first demand right pursuant to Section 2.1.1, which period shall be equal to the length of the lock-up period of the Charterhouse Holders with the underwriters of such underwritten offering.
Sponsor Warrant Lock-up Period shall mean, with respect to Sponsor Warrants and any of the Class A Common Stock issued or issuable upon the exercise or conversion of the Sponsor Warrants that are held by the initial purchasers of such Sponsor Warrants or their Permitted Transferees, the period ending 30 days after the Closing Date.
Sponsor Warrants shall have the meaning given in the Recitals hereto.
Subsequent Shelf Registration shall have the meaning given in Section 2.3.2.
Supporting Company Holders shall have the meaning given in the Recitals hereto.
Target Shareholder shall have the meaning given in the Recitals hereto.
Target Shareholder Lockup shall have the meaning given in Section 5.1.
Target Shareholder Lock-up Period shall have the meaning given in Section 5.1.
Target Shareholder Lock-up Securities shall have the meaning given in Section 5.1.
Transactions shall have the meaning given in the Recitals hereto.
Transfer shall mean the (a) sale or assignment of, offer to sell, contract or agreement to sell, hypothecate, pledge, grant of any option to purchase or otherwise dispose of or agreement to dispose of, directly or indirectly, or establishment or increase of a put equivalent position or liquidation with respect to or decrease of a call equivalent position within the meaning of Section 16 of the Exchange Act with respect to, any security, (b) entry into any hedging, swap or other arrangement that transfers to another, in whole or in part, any of the economic consequences of ownership of any security, whether any such transaction is to be settled by delivery of such securities, in cash or otherwise, or (c) public announcement of any intention to effect any transaction specified in clause (a) or (b).
Underwriter shall mean a securities dealer who purchases any Registrable Securities as principal in an Underwritten Offering and not as part of such dealers market-making activities.
Underwritten Offering shall mean a Registration in which securities of the Company are sold to an Underwriter in a firm commitment underwriting for distribution to the public, including for the avoidance of doubt an Underwritten Shelf Takedown.
Underwritten Shelf Takedown shall have the meaning given in Section 2.3.4.
ARTICLE II
REGISTRATIONS
2.1 Demand Registration.
2.1.1 Charterhouse Demand Registration. The Charterhouse Holders will have the exclusive right (the Charterhouse Demand Right) for a 90-day period following expiration of the Charterhouse Lock-up Period
F-8
Table of Contents
(the Charterhouse Demand Period) to exercise a single demand right and for which Piggyback Registration rights shall not be applicable; provided, for the avoidance of doubt, the Charterhouse Holders can determine, in their sole discretion, to include other stockholders in such Registration. During the Charterhouse Lock-up Period and the Charterhouse Demand Period (or such earlier period ending the date of the expiration of the Sponsor Underwriter Lock-up Period if an Underwritten Offering pursuant to the Charterhouse Demand Right closes) (the Charterhouse Demand Lock-up Period), (a) the GS Founder Share Members shall not Transfer any shares of Common Stock (other than to their Permitted Transferees) or request a Demand Registration and (b) the GS Equity Investor and its Affiliates shall not Transfer any PIPE Shares, Backstop Shares or Option Shares (other than any such shares distributed to the GS Equity Investors Permitted Transferees) or request a Demand Registration (in each case of clauses (a) and (b), whether as part of a Shelf Registration, an unregistered transaction or otherwise); provided that such period shall be extended for any day during which the Registration Statement is not effective or sales pursuant to the Registration Statement are suspended. The GS Founder Share Members, the GS Founder Share Members Permitted Transferees, the GS Equity Investor and each Holder that is an executive officer or director of the Company or the beneficial owner of more than five percent (5%) of the outstanding shares of Class A Common Stock, shall, if requested by the Underwriters, execute a customary lock-up agreement (in each case on substantially the same terms and conditions as all such Holders, including customary mfn release provisions) in favor of the managing Underwriters not to, Transfer any shares of Common Stock or any securities convertible into or exercisable or exchangeable for Common Stock or exercise any demand or piggyback rights hereunder, during the Sponsor Underwriter Lock-up Period, except as expressly permitted by such lock-up agreement or in the event the managing Underwriters otherwise agree.
2.1.2 Request for Registration. Subject to the provisions of Sections 2.1.1, 2.8 and 2.9 hereof, at any time and from time to time on or after the date the Charterhouse Demand Period ends, (i) the Charterhouse Holders, (ii) the GS Holders or (iii) the Holders of at least thirty percent (30%) in interest of the then outstanding number of Registrable Securities (any of (i), (ii) or (iii), the Demanding Holders) may make a written demand for Registration under the Securities Act of all or part of their Registrable Securities, which written demand shall describe the amount and type of securities to be included in such Registration and the intended methods of distribution thereof (such written demand, a Demand Registration); provided that no shares that are subject to a Lock-Up Period at the time that the Registration Statement that is subject to such Demand Registration is required to be filed may be included in such Demand Registration or counted towards such 30% in interest of the Demanding Holders. The Company shall, within 10 days of the Companys receipt of the Demand Registration, notify, in writing, all other Holders of Registrable Securities of such demand, and each Holder of Registrable Securities who thereafter wishes to include all or a portion of such Holders Registrable Securities in a Registration pursuant to a Demand Registration (each such Holder that includes all or a portion of such Holders Registrable Securities in such Registration, a Requesting Holder) shall so notify the Company, in writing, within 5 days after the receipt by the Holder of the notice from the Company. Upon receipt by the Company of any such written notification from a Requesting Holder to the Company, such Requesting Holder shall be entitled to have their Registrable Securities included in a Registration pursuant to a Demand Registration and the Company shall effect, as soon thereafter as reasonably practicable, the Registration of all Registrable Securities requested by the Demanding Holders and Requesting Holders pursuant to such Demand Registration, including by using commercially reasonable efforts to file a Registration Statement relating thereto as soon as reasonably practicable, but not more than 45 days immediately after the Companys receipt of the Demand Registration; provided the Company shall not be required to file such Registration Statement (or an amendment thereto) during any period for which it has not yet filed financial statements with the Commission that would be required to be included in such Registration Statement and the due date for filing of such financial statements under the rules and regulations of the Commission has not yet elapsed.
2.1.3 Effective Registration. Notwithstanding the provisions of Section 2.1.2 above or any other part of this Agreement, a Registration pursuant to a Demand Registration shall not count as a Registration hereunder unless and until (i) the Registration Statement filed with the Commission with respect to a Registration pursuant to a Demand Registration has been declared effective by the Commission and (ii) the Company has materially complied with all of its obligations under this Agreement with respect thereto; provided, further, that if, after such Registration Statement has been declared effective, an offering of Registrable Securities in a Registration
F-9
Table of Contents
pursuant to a Demand Registration is subsequently interfered with by any stop order or injunction of the Commission, federal or state court or any other governmental agency, the Registration Statement with respect to such Registration shall be deemed not to have been declared effective, unless and until, (i) such stop order or injunction is removed, rescinded or otherwise terminated and (ii) a majority-in-interest of the Demanding Holders initiating such Demand Registration thereafter affirmatively elect to continue with such Registration and accordingly notify the Company in writing; provided, further, that the Company shall not be obligated or required to file another Registration Statement until the Registration Statement that has been previously filed with respect to a Registration pursuant to a Demand Registration becomes effective or is subsequently terminated.
2.2 Piggyback Registration. If the Company or any Holder proposes to conduct a registered offering of equity securities (whether for its account or for the account of one more of its stockholders) other than an Excluded Registration, then the Company shall give written notice of such proposed filing to all of the Piggyback-Eligible Holders at such time as soon as reasonably practicable but not less than ten business days before the anticipated filing date of such Registration Statement or, in the case of an Underwritten Offering pursuant to a Shelf Registration, the applicable red herring prospectus or prospectus supplement used for marketing such offering, which notice shall (A) describe the amount and type of securities to be included in such offering, the intended methods of distribution and the name of the proposed managing Underwriters, if any, in such offering, and (B) offer to all of the Piggyback-Eligible Holders of Registrable Securities the opportunity to include in such registered offering such number of Registrable Securities as such Piggyback-Eligible Holders may request in writing within five business days after receipt of such written notice (such Registration, a Piggyback Registration). Subject to Section 2.7, the Company shall, in good faith, cause such Registrable Securities to be included in such Piggyback Registration and, if applicable, shall use its commercially reasonable efforts to cause the managing Underwriter or Underwriters of such Piggyback Registration to permit the Registrable Securities requested by the Piggyback-Eligible Holders pursuant to this Section 2.2 to be included therein on the same terms and conditions as any similar securities of the Company included in such registered offering and to permit the sale or other disposition of such Registrable Securities in accordance with the intended methods of distribution thereof. The inclusion of any Holders Registrable Securities in a Piggyback Registration shall be subject to such Holders agreement to enter into an underwriting agreement in customary form with the Underwriters selected for such Underwritten Offering. For purposes of clarity, any Piggyback Registration effected pursuant to Section 2.2 hereof shall not be counted as a demand for an Underwritten Shelf Takedown.
2.3 Shelf Registrations.
2.3.1 Filing. The Company shall use commercially reasonable efforts to file a Registration Statement with the Commission for a Shelf Registration on Form S-1 (the Form S-1 Shelf), since it will be ineligible to use a Form S-3 (the Form S-3 Shelf and, together with a Form S-1 Shelf, a Shelf) as soon as reasonably practicable but no later than 30 calendar days following the Closing Date (the Filing Date), covering the resale of all Registrable Securities (determined as of two business days prior to such filing) on a delayed or continuous basis and shall use its commercially reasonable efforts to have such Shelf declared effective as soon as reasonably practicable after the filing thereof and no later than the earlier of (x) the 90th calendar day following the Filing Date if the Commission notifies the Company that it will review the Shelf and (y) the 10th business day after the date the Company is notified in writing by the Commission that such Shelf will not be reviewed or will not be subject to further review. Such Shelf shall provide for the resale of the Registrable Securities included therein pursuant to any method or combination of methods legally available to, and requested by, any Holder named therein. The Company shall maintain a Shelf in accordance with the terms hereof, and shall prepare and file with the Commission such amendments, including post-effective amendments, and supplements as may be necessary to keep a Shelf continuously effective, available for use to permit all Holders named therein to sell their Registrable Securities included therein and in compliance with the provisions of the Securities Act until such time as there are no longer any Registrable Securities. The Company shall use its commercially reasonable efforts to convert the Form S-1 Shelf (and any Subsequent Shelf Registration) to a Form S-3 Shelf as soon as reasonably practicable after the Company is eligible to use Form S-3.
F-10
Table of Contents
2.3.2 Subsequent Shelf Registration. If any Shelf ceases to be effective under the Securities Act for any reason at any time while Registrable Securities included thereon are still outstanding, the Company shall, subject to Section 3.4, use its commercially reasonable efforts to as promptly as is reasonably practicable cause such Shelf to again become effective under the Securities Act (including using its commercially reasonable efforts to obtaining the prompt withdrawal of any order suspending the effectiveness of such Shelf), and shall use its commercially reasonable efforts to as promptly as is reasonably practicable amend such Shelf in a manner reasonably expected to result in the withdrawal of any order suspending the effectiveness of such Shelf or file an additional registration statement as a Shelf Registration (a Subsequent Shelf Registration) registering the resale of all Registrable Securities (determined as of two business days prior to such filing), and pursuant to any method or combination of methods legally available to, and requested by, any Holder named therein. If a Subsequent Shelf Registration is filed, the Company shall use its commercially reasonable efforts to (i) cause such Subsequent Shelf Registration to become effective under the Securities Act as promptly as is reasonably practicable after the filing thereof (it being agreed that the Subsequent Shelf Registration shall be an automatic shelf registration statement (as defined in Rule 405 promulgated under the Securities Act) if the Company is a well-known seasoned issuer (as defined in Rule 405 promulgated under the Securities Act) at the most recent applicable eligibility determination date) and (ii) keep such Subsequent Shelf Registration continuously effective, available for use to permit all Holders named therein to sell their Registrable Securities included therein and in compliance with the provisions of the Securities Act until such time as there are no longer any Registrable Securities. Any such Subsequent Shelf Registration shall be on Form S-3 to the extent that the Company is eligible to use such form. Otherwise, such Subsequent Shelf Registration shall be on another appropriate form.
2.3.3 Additional Registrable Securities. In the event that any Holder holds Registrable Securities that are not registered for resale on a delayed or continuous basis, the Company, upon request of the Charterhouse Holders or the GS Founder Share Members, shall promptly use its commercially reasonable efforts to cause the resale of such Registrable Securities to be covered by either, at the Companys option, any then available Shelf (including by means of a post-effective amendment) or by filing a Subsequent Shelf Registration and cause the same to become effective as soon as reasonably practicable after such filing and such Shelf or Subsequent Shelf Registration shall be subject to the terms hereof; provided, however, the Company shall only be required to cause such Registrable Securities to be so covered twice per calendar year for each of the Charterhouse Holders and the GS Founder Share Members.
2.3.4 Requests for Underwritten Shelf Takedowns. Subject to Section 2.4, at any time and from time to time when an effective Form S-3 Shelf is on file with the Commission, the Demanding Holders may request to sell all or any portion of such Demanding Holders Registrable Securities in an Underwritten Offering that is registered pursuant to the Shelf (each, an Underwritten Shelf Takedown). All requests for Underwritten Shelf Takedowns (other than Block Trades) shall be made by giving written notice to the Company at least fifteen business days prior to the public announcement of such Underwritten Shelf Takedown, which shall specify the approximate number of Registrable Securities proposed to be sold in the Underwritten Shelf Takedown and the expected price range (net of underwriting discounts and commissions) of such Underwritten Shelf Takedown. The Company shall include in any Underwritten Shelf Takedown the securities requested to be included by any Holder within 5 business days of receipt of a notice of such Underwritten Shelf Takedown pursuant to written contractual Piggyback Registration rights of such holder (including those set forth herein).
2.4 Block Trades. If the Charterhouse Holders, the GS Founder Share Members or the GS Equity Investor wish to consummate an overnight block trade (on either an SEC-registered or non-registered basis, a Block Trade), then notwithstanding the time periods and piggyback rights otherwise provided herein, such Holder shall, if it would like the assistance of the Company, endeavor to give the Company sufficient advance notice in order to prepare the appropriate documentation for such transaction; provided the Company shall have no obligations under this Section 2.4 if the proposed aggregate offering amount of the Block Trade is less than $40 million. If requesting an SEC-registered Block Trade requiring the Company to file a prospectus or prospectus supplement, the applicable Holders (1) shall give the Company written notice of the transaction and the anticipated launch date of the transaction at least three (3) business days prior to the anticipated launch date of the transaction, (2) the Company shall be required to only notify the Charterhouse Holders, the GS Founder
F-11
Table of Contents
Share Members and the GS Equity Investor of the transaction and none of the other Holders; provided, however, that the Charterhouse Holders, the GS Founder Share Members and the GS Equity Investor may each determine, in its discretion, to notify other Holders, (3) the Charterhouse Holders, the GS Founder Share Members and the GS Equity Investor, and any other Holders that have been notified of the Block Trade pursuant to the foregoing clause (2), shall have one (1) business day prior to the launch of the transaction to determine if they wish to participate in the Block Trade and (4) the Company shall include in the Block Trade only shares held by the Charterhouse Holders, the GS Founder Share Members, the GS Equity Investor or any other Holders participating pursuant to the foregoing clauses (2) and (3), subject to Section 2.6. This Section 2.4 shall not restrict a Holder from undertaking a Block Trade independently that is either non-registered or does not otherwise require Company assistance in filing a prospectus or prospectus supplement.
2.5 Underwritten Offering. The Demanding Holders holding a majority of the Registrable Securities included in a Demand Registration or Underwritten Shelf Takedown, or in the case of a Registration pursuant to a Charterhouse Demand Right, the Charterhouse Holders solely, may advise the Company that such Demand Registration or Underwritten Shelf Takedown shall be in the form of an Underwritten Offering and shall have the right to select the Underwriters for such offering (which shall consist of one or more reputable nationally recognized investment banks) subject to the Companys prior approval, which shall not be unreasonably withheld, conditioned or delayed. All such Holders proposing to distribute their Registrable Securities through an Underwritten Offering under this Section 2.4 shall sell such Registrable Securities pursuant to the terms of an underwriting agreement in customary form with the Underwriters selected for such Underwritten Offering and execute such documents and certificates and cooperate with requirements of such underwriting agreement as may be reasonably requested in connection therewith, including providing any legal opinions customarily provided by selling stockholders in an Underwritten Offering.
2.6 Reduction of Underwritten Offering or Block Trade. If the managing Underwriter or Underwriters (or other sales agent) in an Underwritten Offering or Block Trade, in good faith, advise the Company and the participating Holders that the dollar amount or number of Registrable Securities exceeds the maximum dollar amount or maximum number of equity securities that can be sold in the Underwritten Offering or Block Trade without adversely affecting the proposed offering price, the timing, the distribution method or the probability of success of such offering (such maximum dollar amount or maximum number of such securities, as applicable, the Maximum Number of Securities), then the Company shall include in such Underwritten Offering or Block Trade, as follows:
(a) if the Underwritten Offering is pursuant to a Demand Registration, an Underwritten Shelf Takedown or Block Trade, the Registrable Securities of the Holders that can be sold without exceeding the Maximum Number of Securities, determined pro rata based on the respective number of Registrable Securities that each Holder has requested be included in such Underwritten Offering; and
(b) if the Underwritten Offering is undertaken for the Companys account, the Company shall include in any such Registration or registered offering (i) first, the shares of Common Stock or other equity securities that the Company desires to sell, which can be sold without exceeding the Maximum Number of Securities, and (ii) second, to the extent that the Maximum Number of Securities has not been reached under the foregoing clause (i), the Registrable Securities of Holders exercising their rights to register their Registrable Securities pursuant to Section 2.2 hereof determined pro rata based on the respective number of Registrable Securities that each Holder has requested be included in such Underwritten Offering; provided that the Registrable Securities of the Charterhouse Holders shall not be reduced below the amount that is included pursuant to clause (i) less the Maximum Number of Securities unless they are the only Holders participating in such Underwritten Offering.
2.7 Registration Withdrawal. Any Holder participating in a Registration has the right to withdraw from a Registration for any or no reason whatsoever upon written notification to the Company and the Underwriter or Underwriters (if any) of their intention to withdraw from such Registration prior to: (i) the effectiveness of the Registration Statement filed with the Commission with respect to the Registration or (ii) in the case of an Underwritten Shelf Takedown, the public announcement thereof.
F-12
Table of Contents
2.8 Restrictions on Registration Rights. Notwithstanding anything to the contrary in this Agreement, the Company shall not be obligated to effect, or to take any action to effect, any Registration or Underwritten Offering pursuant to Article II if:
(a) during the period starting with the date 60 days prior to the Companys good faith estimate of the date of the filing of, and ending on a date 120 days after the effective date of, a Company initiated Registration and provided that the Company has delivered written notice to the Holders prior to receipt of a Demand Registration pursuant to Section 2.1.2, it continues to actively employ, in good faith, its commercially reasonable efforts to cause the applicable Registration Statement to become effective;
(b) the Holders have requested an Underwritten Offering and the Company and the Holders are unable to obtain the commitment of underwriters to firmly underwrite the offer;
(c) in the case of a Demand Registration, the Registrable Securities to be included therein have a gross aggregate offering price of less than $100 million;
(d) in the case of an Underwritten Shelf Takedown, the Registrable Securities to be included therein have a gross aggregate offering price of less than $50 million;
(e) the Company has effected at least four (4) Demand Registrations, Underwritten Shelf Takedowns or Underwritten Offerings (excluding Piggyback Registrations), each resulting in a consummated offering, in the twelve (12) month period immediately preceding the date of a request for such Registration; provided this clause (e) shall not apply to non-registered Block Trades or Block Trades in which the Company does not need to file a prospectus or prospectus supplement;
(f) any Registration Statement is suspended pursuant to Section 3.4; or
(g) in the good faith judgment of the Board, such Registration would be seriously detrimental to the Company and the Board concludes as a result that it is essential to defer the filing of such Registration Statement at such time, then in each case the Company shall furnish to such Holders a certificate signed by the Chairman of the Board, the Chief Executive Officer (or a Co-Chief Executive Officer, if applicable), the President or the Secretary of the Company stating that in the good faith judgment of the Board it would be seriously detrimental to the Company for such Registration Statement to be filed in the near future and that it is therefore essential to defer the filing of such Registration Statement; provided in the case of this clause (g), the Company shall only have the right to defer such filing for a period of not more than 90 days and not more than once in any 12-month period.
Notwithstanding anything to the contrary contained in this Agreement, the Company shall not be required to effect or permit any Registration or cause any Registration Statement to become effective, with respect to any Registrable Securities held by any Holder, until after the expiration of any Lock-up Period applicable to such Registrable Securities.
2.9 Market Stand-off. In connection with any Underwritten Offering of Common Stock pursuant to this Agreement, if requested by the Underwriters managing the offering, each Holder that is an executive officer or director of the Company or the beneficial owner of more than five percent (5%) of the outstanding shares of Class A Common Stock and any other Holder reasonably requested by the managing Underwriter (including any Holder selling securities in such Underwritten Offering), agrees not to, and to execute a customary lock-up agreement (in each case on substantially the same terms and conditions as all such Holders, including customary mfn release provisions) in favor of the managing Underwriters not to, Transfer any shares of Common Stock or any securities convertible into or exercisable or exchangeable for Common Stock (other than those included in such offering) or exercise any demand or piggyback rights hereunder, during the ninety (90) day period (or such shorter time agreed to by the managing Underwriters (but in no event less than 45 days)) beginning on the date of pricing such offering, except as expressly permitted by such lock-up agreement or in the event the managing Underwriters otherwise agree.
F-13
Table of Contents
ARTICLE III
COMPANY PROCEDURES
3.1 General Procedures. In connection with any Registration, whether pursuant to the filing of a new Registration Statement, effecting an Underwritten Shelf Takedown or effecting an underwritten Block Trade, the Company shall use its commercially reasonable efforts to effect such Registration to permit the sale of such Registrable Securities in accordance with the intended plan of distribution thereof, and pursuant thereto the Company shall, as expeditiously as possible (without limiting the generality of the Companys obligations pursuant to Section 2.3):
3.1.1 prepare and file with the Commission as soon as reasonably practicable a Registration Statement with respect to such Registrable Securities and use its commercially reasonable efforts to cause such Registration Statement to become effective and remain effective until all Registrable Securities have ceased to be Registrable Securities;
3.1.2 prepare and file with the Commission such amendments and post-effective amendments to the Registration Statement, and such supplements to the Prospectus, as may be reasonably requested by a majority-in-interest of the Holders with Registrable Securities registered on such Registration Statement, the GS Founder Share Members or the Charterhouse Holders (provided that such GS Founder Share Members and/or the Charterhouse Holders, as applicable, hold at least some of the Registrable Securities registered on such Registration Statement or are named in the Registration Statement) or any Underwriter of Registrable Securities or as may be required by the rules, regulations or instructions applicable to the registration form used by the Company or by the Securities Act or rules and regulations thereunder to keep the Registration Statement effective until all Registrable Securities covered by such Registration Statement are sold in accordance with the intended plan of distribution set forth in such Registration Statement or supplement to the Prospectus;
3.1.3 prior to filing a Registration Statement or Prospectus, or any amendment or supplement thereto, furnish without charge to the Underwriters, if any, and the Holders of Registrable Securities included in such Registration, and such Holders legal counsel, copies of such Registration Statement as proposed to be filed, each amendment and supplement to such Registration Statement (in each case including all exhibits thereto and documents incorporated by reference therein), the Prospectus included in such Registration Statement (including each preliminary Prospectus), and such other documents as the Underwriters and the Holders of Registrable Securities included in such Registration or the legal counsel for any such Holders may request in order to facilitate the disposition of the Registrable Securities owned by such Holders;
3.1.4 prior to any public offering of Registrable Securities, use its commercially reasonable efforts to (i) register or qualify the Registrable Securities covered by the Registration Statement under such securities or blue sky laws of such jurisdictions in the United States as the Holders of Registrable Securities included in such Registration Statement (in light of their intended plan of distribution) may reasonably request (or provide evidence satisfactory to such Holders that the Registrable Securities are exempt from such registration or qualification) and (ii) take such action necessary to cause such Registrable Securities covered by the Registration Statement to be registered with or approved by such other governmental authorities as may be necessary by virtue of the business and operations of the Company and do any and all other acts and things that may be necessary or advisable to enable the Holders of Registrable Securities included in such Registration Statement to consummate the disposition of such Registrable Securities in such jurisdictions; provided, however, that the Company shall not be required to qualify generally to do business in any jurisdiction where it would not otherwise be required to qualify or take any action to which it would be subject to general service of process or taxation in any such jurisdiction where it is not then otherwise so subject;
3.1.5 cause all such Registrable Securities to be listed on each national securities exchange on which such class of Registrable Security issued by the Company is then listed;
3.1.6 provide a transfer agent and registrar for all such Registrable Securities no later than the effective date of such Registration Statement;
F-14
Table of Contents
3.1.7 advise each seller of such Registrable Securities, promptly after it shall receive notice or obtain knowledge thereof, of the issuance of any stop order by the Commission suspending the effectiveness of such Registration Statement or the initiation or threatening of any proceeding for such purpose and promptly use its commercially reasonable efforts to prevent the issuance of any stop order or to obtain its withdrawal if such stop order should be issued;
3.1.8 at least two business days prior to the filing of any Registration Statement or Prospectus or any amendment or supplement to such Registration Statement or Prospectus (or such shorter period of time as may be necessary in order to comply with the Securities Act, the Exchange Act, and the rules and regulations promulgated under the Securities Act or Exchange Act, as applicable), furnish a copy thereof to each seller of such Registrable Securities or its counsel (excluding any exhibits thereto and any filing made under the Exchange Act that is to be incorporated by reference therein);
3.1.9 notify the Holders at any time when a Prospectus relating to such Registration Statement is required to be delivered under the Securities Act, of the happening of any event as a result of which the Prospectus included in such Registration Statement, as then in effect, includes a Misstatement, and then to correct such Misstatement as set forth in Section 3.4;
3.1.10 permit a representative of the Holders, the Underwriters, if any, and any attorney or accountant retained by such Holders or Underwriter to participate, at each such persons own expense, in the preparation of the Registration Statement, and cause the Companys officers, directors and employees to supply all information reasonably requested by any such representative, Underwriter, attorney or accountant in connection with the Registration; provided, however, that such representatives and Underwriters agree to confidentiality arrangements, in form and substance reasonably satisfactory to the Company, prior to the release or disclosure of any such information;
3.1.11 in connection with a Registration that is an Underwritten Offering, request the Companys independent registered public accountants to provide an accountants comfort letter, in customary form and covering such matters of the type customarily covered by accountants comfort letters;
3.1.12 in connection with a Registration that is an Underwritten Offering, obtain an opinion and a negative assurance letter of counsel representing the Company for the purposes of such Registration covering customary legal matters with respect to the Registration in respect of which such opinion is being given;
3.1.13 enter into and perform its obligations under an underwriting agreement or distribution agreement, in usual and customary form, with the managing Underwriter or the broker, placement agent or sales agent of such offering or sale;
3.1.14 with respect to an Underwritten Offering, use its commercially reasonable efforts to make available senior executives of the Company to participate in customary road show presentations that may be reasonably requested by the Underwriter in any Underwritten Offering;
3.1.15 otherwise, in good faith, cooperate reasonably with, and take such customary actions as may reasonably be requested by the participating Holders and the broker, placement agent or sales agent, if any, and the Underwriter, if any, as applicable; and
3.1.16 upon the reasonable request of a Holder, the Company shall (i) authorize the Companys transfer agent to remove any legend on share certificates of such Holders Class A Common Stock restricting further transfer (or any similar restriction in book entry positions of such Holder) if such restrictions are no longer required by the Securities Act or any applicable state securities laws or any agreement with the Company to which such Holder is a party, including if such shares subject to such a restriction have been or will imminently be sold on a Registration Statement, (ii) request the Companys transfer agent to issue in lieu thereof shares of Class A Common Stock without such restrictions to the Holder upon, as applicable, surrender of any stock certificates evidencing such shares of Class A Common Stock, or to update the applicable book entry position of such Holder so that it no longer is subject to such a restriction, and (iii) use commercially reasonable efforts to cooperate with such Holder to have such Holders shares of Class A Common Stock transferred into a book-entry
F-15
Table of Contents
position at The Depository Trust Company, in each case, subject to delivery of customary documentation, including any documentation required by such restrictive legend or book-entry notation.
3.2 Registration Expenses. The Registration Expenses of all Registrations shall be borne by the Company. It is acknowledged by each Holder that, with respect to such Holders Registrable Securities being sold, all Selling Expenses of such Holder will be borne by such Holder.
3.3 Requirements for Participation in Registration Statement in Underwritten Offerings. Notwithstanding anything in this Agreement to the contrary, if any Holder does not provide the Company with its requested Holder Information, the Company may exclude such Holders Registrable Securities from the applicable Registration Statement or Prospectus if the Company determines that such information is necessary to effect the registration and such Holder continues thereafter to withhold such information. No person may participate in any Underwritten Offering for equity securities of the Company pursuant to a Registration initiated by the Company hereunder unless such person (i) agrees to sell such persons securities on the basis provided in any underwriting arrangements approved by the Company and (ii) completes and executes all customary questionnaires, powers of attorney, indemnities, lock-up agreements, underwriting agreements and other customary documents as may be reasonably required under the terms of such underwriting arrangements. The exclusion of a Holders Registrable Securities as a result of this Section 3.3 shall not affect the registration of the other Registrable Securities to be included in such Registration.
3.4 Suspension of Sales; Adverse Disclosure; Restrictions on Registration Rights. Upon receipt of written notice from the Company that a Registration Statement or Prospectus contains a Misstatement, each of the Holders shall forthwith discontinue disposition of Registrable Securities until he, she or it has received copies of a supplemented or amended Prospectus correcting the Misstatement (it being understood that the Company hereby covenants to prepare and file such supplement or amendment as soon as reasonably practicable after the time of such notice), or until he, she or it is advised in writing by the Company that the use of the Prospectus may be resumed. If the filing, initial effectiveness or continued use of a Registration Statement in respect of any Registration at any time would require the Company to make an Adverse Disclosure or would require the inclusion in such Registration Statement of financial statements that are unavailable to the Company for reasons beyond the Companys control, the Company may, upon giving prompt written notice of such action to the Holders, delay the filing or initial effectiveness of, or suspend use of, such Registration Statement for the shortest reasonable period of time; provided, however, the Company may not delay or suspend the Registration Statement on more than two occasions or for more than sixty (60) consecutive calendar days, or more than ninety (90) total calendar days, in each case during any twelve-month period. In the event the Company exercises its rights under the preceding sentence, the Holders agree to suspend, immediately upon their receipt of the notice referred to above, their use of the Prospectus relating to any Registration in connection with any sale or offer to sell Registrable Securities. The Company shall immediately notify the Holders of the expiration of any period during which it exercised its rights under this Section 3.4.
3.5 Reporting Obligations. As long as any Holder shall own Registrable Securities, the Company, at all times while it shall be a reporting company under the Exchange Act, covenants to file timely (or obtain extensions in respect thereof and file within the applicable grace period) all reports required to be filed by the Company after the date hereof pursuant to Sections 13(a) or 15(d) of the Exchange Act. The Company further covenants that it shall furnish to any Holder, so long as the Holder owns any Registrable Securities: (i) to the extent accurate, a written statement by the Company that it has complied with the reporting requirements of Rule 144 (at any time after the time period contemplated by Rule 144(i)(2)), the Securities Act and the Exchange Act; and (ii) such other information as may be reasonably requested in availing any Holder of any rule or regulation of the SEC that permits the selling of any such securities without registration (at any time after the Company has become subject to the reporting requirements under the Exchange Act) or pursuant to Form S-3 (at any time after the Company so qualifies to use such form).
F-16
Table of Contents
ARTICLE IV
INDEMNIFICATION AND CONTRIBUTION
4.1 Indemnification.
4.1.1 The Company agrees to indemnify, to the extent permitted by law, each Holder of Registrable Securities, its officers, directors and agents and each person who controls such Holder (within the meaning of the Securities Act) against all losses, claims, damages, liabilities and out-of-pocket expenses (including, without limitation, reasonable outside attorneys fees) resulting from any untrue or alleged untrue statement of material fact contained or incorporated by reference in any Registration Statement, Prospectus or preliminary Prospectus or any amendment thereof or supplement thereto or any omission or alleged omission of a material fact required to be stated therein or necessary to make the statements therein not misleading, except insofar as the same are caused by or contained in any information or affidavit so furnished in writing to the Company by such Holder expressly for use therein.
4.1.2 In connection with any Registration Statement in which a Holder of Registrable Securities is participating, such Holder shall furnish to the Company in writing such information and affidavits as the Company reasonably requests for use in connection with any such Registration Statement or Prospectus (the Holder Information) and, to the extent permitted by law, shall indemnify the Company, its directors, officers and agents and each person who controls the Company (within the meaning of the Securities Act) against all losses, claims, damages, liabilities and out-of-pocket expenses (including, without limitation, reasonable outside attorneys fees) resulting from any untrue or alleged untrue statement of material fact contained in any Registration Statement, Prospectus or preliminary Prospectus or any amendment thereof or supplement thereto or any omission or alleged omission of a material fact required to be stated therein or necessary to make the statements therein not misleading, but only to the extent that such untrue statement or omission is contained in the Holder Information so furnished in writing by such Holder expressly for use therein; provided, however, that the obligation to indemnify shall be several, not joint and several, among such Holders of Registrable Securities, and the liability of each such Holder of Registrable Securities shall be in proportion to and limited to the net proceeds received by such Holder from the sale of Registrable Securities pursuant to such Registration Statement.
4.1.3 Any person entitled to indemnification herein shall (i) give prompt written notice to the indemnifying party of any claim with respect to which he, she or it seeks indemnification (provided that the failure to give prompt notice shall not impair any persons right to indemnification hereunder to the extent such failure has not materially prejudiced the indemnifying party) and (ii) unless in such indemnified partys reasonable judgment a conflict of interest between such indemnified and indemnifying parties may exist with respect to such claim, permit such indemnifying party to assume the defense of such claim with counsel reasonably satisfactory to the indemnified party. If such defense is assumed, the indemnifying party shall not be subject to any liability for any settlement made by the indemnified party without its consent (but such consent shall not be unreasonably withheld). An indemnifying party who is not entitled to, or elects not to, assume the defense of a claim shall not be obligated to pay the fees and expenses of more than one counsel for all parties indemnified by such indemnifying party with respect to such claim, unless in the reasonable judgment of any indemnified party a conflict of interest may exist between such indemnified party and any other of such indemnified parties with respect to such claim. No indemnifying party shall, without the consent of the indemnified party, consent to the entry of any judgment or enter into any settlement that cannot be settled in all respects by the payment of money (and such money is so paid by the indemnifying party pursuant to the terms of such settlement) or which settlement includes a statement or admission of fault and culpability on the part of such indemnified party or which settlement does not include as an unconditional term thereof the giving by the claimant or plaintiff to such indemnified party of a release from all liability in respect to such claim or litigation.
4.1.4 The indemnification provided for under this Agreement shall remain in full force and effect regardless of any investigation made by or on behalf of the indemnified party or any officer, director or controlling person of such indemnified party and shall survive the transfer of securities. The Company and each Holder of
F-17
Table of Contents
Registrable Securities participating in an offering also agrees to make such provisions as are reasonably requested by any indemnified party for contribution to such party in the event the Companys or such Holders indemnification is unavailable for any reason.
4.1.5 If the indemnification provided under Section 4.1 from the indemnifying party is unavailable or insufficient to hold harmless an indemnified party in respect of any losses, claims, damages, liabilities or out-of-pocket expenses referred to herein, then the indemnifying party, in lieu of indemnifying the indemnified party, shall contribute to the amount paid or payable by the indemnified party as a result of such losses, claims, damages, liabilities and out-of-pocket expenses in such proportion as is appropriate to reflect the relative fault of the indemnifying party and the indemnified party, as well as any other relevant equitable considerations. The relative fault of the indemnifying party and indemnified party shall be determined by reference to, among other things, whether any action in question, including any untrue or alleged untrue statement of a material fact or omission or alleged omission to state a material fact, was made by, or relates to information supplied by, such indemnifying party or indemnified party, and the indemnifying partys and indemnified partys relative intent, knowledge, access to information and opportunity to correct or prevent such action; provided, however, that the liability of any Holder under this Section 4.1.5 shall be limited to the amount of the net proceeds received by such Holder in such offering giving rise to such liability. The amount paid or payable by a party as a result of the losses or other liabilities referred to above shall be deemed to include, subject to the limitations set forth in Sections 4.1.1, 4.1.2 and 4.1.3 above, any legal or other fees, charges or out-of-pocket expenses reasonably incurred by such party in connection with any investigation or proceeding. The parties hereto agree that it would not be just and equitable if contribution pursuant to this Section 4.1.5 were determined by pro rata allocation or by any other method of allocation, which does not take account of the equitable considerations referred to in this Section 4.1.5. No person guilty of fraudulent misrepresentation (within the meaning of Section 11(f) of the Securities Act) shall be entitled to contribution pursuant to this Section 4.1.5 from any person who was not guilty of such fraudulent misrepresentation.
ARTICLE V
LOCK-UP AGREEMENT
5.1 Target Shareholder Lock-Up Agreement.
(a) Each Target Shareholder agrees not to Transfer any shares of Common Stock or Paired Interests received by him, her or it pursuant to the Business Combination Agreement (the Target Shareholder Lock-up Securities) from the date hereof until, and including, the 180th day after the Closing Date (the Target Shareholder Lock-up Period), subject to Section 5.1(b) (the Target Shareholder Lockup).
(b) Notwithstanding Section 5.1(a), a Target Shareholder may Transfer Target Shareholder Lock-up Securities:
(i) by will, other testamentary document or intestacy;
(ii) as a bona fide gift or gifts, including to charitable organizations or for bona fide estate planning purposes;
(iii) to any trust for the direct or indirect benefit of the Target Shareholder or the immediate family of the Target Shareholder, or if the Target Shareholder is a trust, to a trustor or beneficiary of the trust or to the estate of a beneficiary of such trust;
(iv) to a partnership, limited liability company or other entity of which such Target Shareholder and the immediate family of such Target Shareholder are the legal and beneficial owner of all of the outstanding equity securities or similar interests;
(v) if the Target Shareholder is a corporation, partnership, limited liability company, trust or other business entity, (A) to another corporation, partnership, limited liability company, trust or other business entity that is an affiliate (as defined in Rule 405 promulgated under the Securities Act) of such Target Shareholder, or
F-18
Table of Contents
to any investment fund or other entity controlling, controlled by, managing or managed by or under common control with such Target Shareholder or affiliates of such Target Shareholder (including, for the avoidance of doubt, where such Target Shareholder is a partnership, to its general partner or a successor partnership or fund, or any other funds managed by such partnership), or (B) as part of a distribution to members or shareholders of such Target Shareholder;
(vi) to a nominee or custodian of any person or entity to whom a Transfer would be permissible under clauses (i) through (v) above;
(vii) in the case of an individual, by operation of law, such as pursuant to a qualified domestic order, divorce settlement, divorce decree, separation agreement or related court order;
(viii) with the prior written consent of the Board (subject to the determination of the Board in its sole discretion at any time); provided such consent must be approved by each of the Charterhouse Director (unless waived by the Charterhouse Holders) and the GS Directors (unless waived by the GS Sponsor Member);
(ix) from an employee or a director of, or a service provider to, the Company or any of its subsidiaries upon the death, disability or termination of employment or services, in each case, of such person;
(x) pursuant to a bona fide third-party tender offer, merger, consolidation or other similar transaction that is approved by the Board and made to all holders of shares of the Companys capital stock involving a Change of Control (as defined below) (including negotiating and entering into an agreement providing for any such transaction), provided that in the event that such tender offer, merger, consolidation or other similar transaction is not completed, the Target Shareholder Lock-up Securities shall remain subject to the Target Shareholder Lockup; and
(xi) to the GS Equity Investor pursuant to the Option Agreement (as defined in the Business Combination Agreement);
provided that:
(x) in the case of any Transfer of Target Shareholder Lock-up Securities pursuant to clauses (i) through (vi), (1) such Transfer shall not involve a disposition for value; (2) the Target Shareholder Lock-up Securities shall remain subject to the Target Shareholder Lockup and the transferee shall sign a Joinder before such Transfer is effective; (3) any required public report or filing (including filings under Section 16(a) of the Exchange Act), shall disclose the nature of such Transfer and that the Target Shareholder Lock-up Securities remain subject to the Target Shareholder Lockup; and (4) there shall be no voluntary public disclosure or other announcement of such Transfer; and
(y) a Target Shareholder may enter into a trading plan established in accordance with Rule 10b5-1 under the Exchange Act during the Target Shareholder Lock-up Period so long as no Transfers are effected under such trading plan prior to the expiration of the Target Shareholder Lock-up Period and no voluntary public disclosure or announcement of such plan is made.
ARTICLE VI
MISCELLANEOUS
6.1 Notices. Any notice or communication under this Agreement must be in writing and given by mail, hand delivery, electronic mail or facsimile. Each notice or communication that is mailed, delivered or transmitted in the manner described above shall be deemed sufficiently given, served, sent and received, in the case of mailed notices, on the third business day following the date on which it is mailed and, in the case of notices delivered by hand delivery, electronic mail or facsimile, at such time as it is delivered to the addressee or at such time as delivery is refused by the addressee upon presentation.
F-19
Table of Contents
Any notice or communication under this Agreement must be addressed:
if to the Company, to:
Mirion Technologies, Inc.
1218 Menlo Drive
Atlanta, GA 30318
Attention: General Counsel
Email: elee@mirion.com; legal@mirion.com
with a copy (which copy shall not constitute notice) to:
Davis Polk & Wardwell LLP
1600 El Camino Real Ste. 100
Menlo Park, California 94025
Attention: Alan F. Denenberg, Stephen Salmon, Bryan M. Quinn
E-mail: alan.denenberg@davispolk.com; stephen.salmon@davispolk.com; bryan.quinn@davispolk.com
if to the Charterhouse Holders, to:
The Charterhouse Holders
6th Floor, Belgrave House, 76 Buckingham Palace Road
London, SW1W 9TQ, United Kingdom
Attention: Christopher Warren, Thomas Patrick
E-mail: chris.warren@charterhouse.co.uk; tom.patrick@charterhouse.co.uk
with copies (which copies shall not constitute notice) to:
Freshfields Bruckhaus Deringer US LLP
601 Lexington Avenue, 31st Floor
New York, New York 10019
Attention: Valerie Ford Jacob
E-mail: valerie.jacob@freshfields.com
Freshfields Bruckhaus Deringer LLP
9 avenue de Messin
75008 Paris, France
Attention: Yann Gozal
E-mail: yann.gozal@freshfields.com
Freshfields Bruckhaus Deringer LLP
100 Bishopsgate
London EC2P 2SR, United Kingdom
Attention: Charles Hayes
E-mail: charles.hayes@freshfields.com
if to the GS Founder Share Members or the GS Equity Investor, to:
GS Sponsor II LLC, GS Acquisition Holdings II Employee Participation LLC, and GSAM Holdings LLC
200 West Street
New York, New York 10282
Attention: Thomas R. Knott, David S. Plutzer
E-mail: tom.knott@gs.com; david.plutzer@gs.com
F-20
Table of Contents
with copies (which copies shall not constitute notice) to:
Weil, Gotshal & Manges LLP
767 Fifth Avenue
New York, New York 10153
Attention: Michael J. Aiello, Brian Parness
E-mail: michael.aiello@weil.com; brian.parness@weil.com
and, if to any other Holder, at such Holders address as set forth in the Companys books and records. Any party may change its address for notice at any time and from time to time by written notice to the other parties hereto, and such change of address shall become effective 30 days after delivery of such notice as provided in this Section 6.1.
6.2 Assignment; No Third Party Beneficiaries.
6.2.1 This Agreement and the rights, duties and obligations of the Company hereunder may not be assigned or delegated by the Company in whole or in part.
6.2.2 Subject to Section 6.2.4 and Section 6.2.6, this Agreement and the rights, duties and obligations of a Holder hereunder may be assigned in whole or in part to such Holders Permitted Transferees. This Agreement and the provisions hereof shall be binding upon and shall inure to the benefit of each of the parties and its successors and Permitted Transferees.
6.2.3 For the avoidance of doubt, each of the GS Equity Investor or the Charterhouse Holders may assign its rights, duties and obligations under this Agreement to its Permitted Transferees and such Permitted Transferees shall, following the execution of a Joinder (as defined below) and effective upon such distribution, become a Holder hereunder, and the Registrable Securities which such Permitted Transferee receives in such distribution shall remain Registrable Securities until they cease to be Registrable Securities in accordance with the definition thereof.
6.2.4 Prior to the expiration of the applicable Lock-up Period, no Holder who is subject to a Lock-up Period may assign or delegate such Holders rights, duties or obligations under this Agreement, in whole or in part, except in connection with a transfer of Registrable Securities by such Holder to a Permitted Transferee and in accordance with the provisions of the agreement providing for such Lock-up Period and this Section 6.2; provided, that, with respect to each GS Founder Share Member and Charterhouse Holder, the rights, duties and obligations hereunder that are personal to such GS Founder Share Member and such Charterhouse Holder, as applicable, and may not be assigned or delegated in whole or in part, except that (x) a GS Founder Share Member shall be permitted to assign or delegate its rights, duties and obligations hereunder to one or more Affiliates of such GS Founder Share Member (it being understood that no such assignment or delegation shall reduce any rights, duties or obligations of such GS Founder Share Member or such transferees) and (y) a Charterhouse Holder shall be permitted to assign or delegate its rights, duties and obligations hereunder to one or more Affiliates of such Charterhouse Holder (it being understood that no such assignment or delegation shall reduce any rights, duties or obligations of such Charterhouse Holder or such transferees).
6.2.5 This Agreement shall not confer any rights or benefits on any persons that are not parties hereto, other than as expressly set forth in this Agreement and Section 6.2.
6.2.6 No assignment by any party hereto of such partys rights, duties and obligations hereunder shall be binding upon or obligate the Company unless and until the Company shall have received (i) written notice of such assignment as provided in Section 6.1 and (ii) an executed joinder to this Agreement from such successor or permitted assignee in the form of Exhibit A attached hereto (a Joinder). Any transfer or assignment made other than as provided in this Section 6.2 shall be null and void.
6.3 Counterparts. This Agreement may be executed in multiple counterparts, each of which shall be deemed an original, and all of which together shall constitute the same instrument, but only one of which need be produced. Counterparts may be delivered via facsimile, electronic mail (including any electronic signature
F-21
Table of Contents
covered by the U.S. federal ESIGN Act of 2000, Uniform Electronic Transactions Act, the Electronic Signatures and Records Act or other applicable law, e.g., www.docusign.com) or other transmission method, and any counterpart so delivered shall be deemed to have been duly and validly delivered and be valid and effective for all purposes.
6.4 Governing Law; Venue. NOTWITHSTANDING THE PLACE WHERE THIS AGREEMENT MAY BE EXECUTED BY ANY OF THE PARTIES HERETO, THE PARTIES EXPRESSLY AGREE THAT (1) THIS AGREEMENT SHALL BE GOVERNED BY AND CONSTRUED UNDER THE LAWS OF THE STATE OF NEW YORK, INCLUDING, WITHOUT LIMITATION, SECTIONS 5-1401 AND 5-1402 OF THE NEW YORK GENERAL OBLIGATIONS LAW AND NEW YORK CIVIL PRACTICE LAWS AND RULES 327(B), AS APPLIED TO AGREEMENTS AMONG NEW YORK RESIDENTS ENTERED INTO AND TO BE PERFORMED ENTIRELY WITHIN NEW YORK AND (2) SUBJECT TO APPLICABLE LAW, THE VENUE FOR ANY ACTION TAKEN WITH RESPECT TO THIS AGREEMENT SHALL BE ANY STATE OR FEDERAL COURT IN NEW YORK COUNTY IN THE STATE OF NEW YORK.
6.5 TRIAL BY JURY. EACH PARTY HERETO ACKNOWLEDGES AND AGREES THAT ANY CONTROVERSY THAT MAY ARISE UNDER THIS AGREEMENT IS LIKELY TO INVOLVE COMPLICATED AND DIFFICULT ISSUES, AND, THEREFORE, EACH SUCH PARTY HEREBY IRREVOCABLY AND UNCONDITIONALLY WAIVES, TO THE FULLEST EXTENT PERMITTED BY APPLICABLE LAW, ANY RIGHT SUCH PARTY MAY HAVE TO A TRIAL BY JURY IN RESPECT TO ANY ACTION DIRECTLY OR INDIRECTLY ARISING OUT OF, UNDER OR IN CONNECTION WITH OR RELATING TO THIS AGREEMENT OR THE TRANSACTIONS CONTEMPLATED BY THIS AGREEMENT.
6.6 Amendments and Modifications. Upon the written consent of the Company and the Holders of at least a majority-in-interest of the Registrable Securities at the time in question, compliance with any of the provisions, covenants and conditions set forth in this Agreement may be waived, or any of such provisions, covenants or conditions may be amended or modified; provided, however, that notwithstanding the foregoing, any amendment hereto or waiver hereof shall also require the written consent of the GS Founder Share Members (provided that the GS Founder Share Members or their Permitted Transferees hold, in the aggregate, the applicable Requisite Percentage) and/or the Charterhouse Holders (provided that the Charterhouse Holders or their Permitted Transferees hold, in the aggregate, the applicable Requisite Percentage); provided, further, that any amendment hereto or waiver hereof that adversely affects one Holder, solely in his, her or its capacity as a holder of the shares of capital stock of the Company, in a manner that is materially different from the other Holders (in such capacity) shall require the consent of the Holder so affected. No course of dealing between any Holder or the Company and any other party hereto or any failure or delay on the part of a Holder or the Company in exercising any rights or remedies under this Agreement shall operate as a waiver of any rights or remedies of any Holder or the Company. No single or partial exercise of any rights or remedies under this Agreement by a party shall operate as a waiver or preclude the exercise of any other rights or remedies hereunder or thereunder by such party. For the avoidance of doubt, a waiver pursuant to the first sentence of this Section 6.6 may waive piggyback rights for all Holders pursuant to Section 2.2 so long as no Holders participate in an offering for which such piggyback rights would be applicable.
6.7 Other Registration Rights. Other than (i) the Other PIPE Investors who have registration rights with respect to their PIPE Shares pursuant to their Other PIPE Investors Subscription Agreements, (ii) as provided in that certain Warrant Agreement, dated June 29, 2020, between the Company and Continental Stock Transfer & Trust Company and (iii) as provided in the Warrant Purchase Agreement, the Company represents and warrants that no person, other than a Holder of Registrable Securities, has any right to require the Company to register any securities of the Company for sale or to include such securities of the Company in any Registration Statement filed by the Company for the sale of securities for its own account or for the account of any other person.
6.8 Term. This Agreement shall terminate with respect to any Holder on the date that such Holder no longer holds any Registrable Securities; provided, however, the provisions of Article IV, Article V and this Article VI shall survive any termination.
F-22
Table of Contents
6.9 Holder Information. Each Holder agrees, if requested in writing, to represent to the Company the total number of Registrable Securities held by such Holder in order for the Company to make determinations hereunder.
6.10 Additional Holder; Joinder. In addition to persons or entities who may become Holders pursuant to Section 6.2 hereof, subject to the prior written consent of the GS Founder Share Members and the Charterhouse Holders so long as, with respect to each of such parties, the GS Founder Share Members and their Permitted Transferees and the Charterhouse Holders and their Permitted Transferees hold, in the aggregate, the applicable Requisite Percentage, the Company may make any person or entity who acquires shares of Class A Common Stock or rights to acquire shares of Class A Common Stock after the date hereof a party to this Agreement (each such person, an Additional Holder) by obtaining an executed Joinder from such Additional Holder in the form of Exhibit A attached hereto. Such Joinder shall specify the rights and obligations of the applicable Additional Holder under this Agreement. Upon the execution and delivery and subject to the terms of a Joinder by such Additional Holder, the shares of Class A Common Stock of the Company then owned, or underlying any rights then owned, by such Additional Holder (the Additional Holder Common Stock) shall be Registrable Securities to the extent provided herein and therein, and such Additional Holder shall be a Holder under this Agreement with respect to such Additional Holder Common Stock.
[SIGNATURE PAGES FOLLOW]
F-23
Table of Contents
IN WITNESS WHEREOF, the undersigned have caused this Agreement to be executed as of the date first written above.
| COMPANY: | ||
| MIRION TECHNOLOGIES, INC. | ||
| a Delaware corporation | ||
| By: |
|
|
| Name: | ||
| Title: | ||
[Signature Page to A&R Registration Rights Agreement]
Table of Contents
IN WITNESS WHEREOF, the undersigned have caused this Agreement to be executed as of the date first written above.
| HOLDERS: | ||
| GS SPONSOR II LLC | ||
| By: | GSAM HOLDINGS LLC, as sole manager | |
| By: |
|
|
| Name: Tom Knott | ||
| Title: Authorized Signatory | ||
| GS ACQUISITION HOLDINGS II EMPLOYEE PARTICIPATION LLC | ||
| By: | GSAM Gen-Par, L.L.C., its manager | |
| By: |
|
|
| Name: Raanan A. Agus | ||
| Title: Vice President | ||
[Signature Page to A&R Registration Rights Agreement]
Table of Contents
IN WITNESS WHEREOF, the undersigned have caused this Agreement to be executed as of the date first written above.
SELLERS:
[●]
[Signature Page to A&R Registration Rights Agreement]
Table of Contents
EXHIBIT A
REGISTRATION RIGHTS AGREEMENT JOINDER
The undersigned is executing and delivering this joinder (this Joinder) pursuant to the Amended and Restated Registration Rights Agreement, dated as of [●], 2021 (as the same may hereafter be amended, the Registration Rights Agreement), by and among Mirion Technologies, Inc., a Delaware corporation (the Company), and the other persons or entities named as parties therein. Capitalized terms used but not otherwise defined herein shall have the meanings provided in the Registration Rights Agreement.
By executing and delivering this Joinder to the Company, and upon acceptance hereof by the Company upon the execution of a counterpart hereof, the undersigned hereby agrees to become a party to, to be bound by and to comply with the Registration Rights Agreement as a Holder of Registrable Securities in the same manner as if the undersigned were an original signatory to the Registration Rights Agreement, and the undersigneds shares of Class A Common Stock (including any shares of Class A Common Stock issuable from the exchange of Paired Interests) shall be included as Registrable Securities under the Registration Rights Agreement to the extent provided therein.
[For Permitted Transferees of the GS Equity Investor executing this Joinder pursuant to Section 6.2 of the Registration Rights Agreement include the following:
Notwithstanding anything herein or the Registration Rights Agreement to the contrary, this Joinder will become effective only upon the distribution by the GS Equity Investor of PIPE Shares to the undersigned.]
Accordingly, the undersigned has executed and delivered this Joinder as of the ___day of ______________, 20__.
|
Signature of Stockholder |
|
|
| Print Name of Stockholder |
| By: |
| Its: |
| Address: |
|
|
|
|
[Signature Page to A&R Registration Rights Agreement]
Table of Contents
EXECUTION VERSION
BACKSTOP AGREEMENT
This Backstop Agreement (this Agreement) is entered into as of June 17, 2021, by and among GS Acquisition Holdings Corp II, a Delaware corporation (the Company), and GSAM Holdings LLC, a Delaware limited liability company (the Purchaser). Capitalized terms used but not defined in this Agreement shall have the meaning ascribed to such terms in that certain Business Combination Agreement, dated as of the date hereof, by and among the Company, Mirion Technologies (TopCo), Ltd., a Jersey private company limited by shares (Mirion), CCP IX LP No. 1, CCP IX LP No. 2, CCP IX Co-Investment LP and CCP IX Co-Investment No. 2 LP (the Charterhouse Parties), each acting by their general partner, Charterhouse General Partners (IX) Limited, and the other parties named therein (as amended, modified, supplemented or waived from time to time in accordance with its terms, the Business Combination Agreement).
WHEREAS, in connection with the entry into the Business Combination Agreement, Purchaser has allocated and committed up to $125,000,000.00 to subscribe for a number of shares of New SPAC Class A Common Shares subject to the amount of Existing SPAC Common Stock that is redeemed (and for which redemptions are not subsequently withdrawn) by Existing SPAC Stockholders in connection with the SPAC Special Meeting, if any (the GS Stockholder Redemptions); and
WHEREAS, the Purchaser is now entering into this Agreement with the Company, whereby at the Closing under the Business Combination Agreement, the Purchaser will acquire, and the Company will issue and sell to the Purchaser, New SPAC Class A Common Shares, on a private placement basis, solely to the extent necessary to fund GS Stockholder Redemptions on a share for share basis and in the amount determined pursuant to Section 2(a)(i) hereof and subject to the limitations set forth herein (the Backstop Purchase Shares).
NOW, THEREFORE, in consideration of the promises, representations, warranties and the mutual covenants contained in this Agreement, and for other good and valuable consideration, the receipt, sufficiency and adequacy of which are hereby acknowledged, the parties hereto agree as follows:
1. Backstop Limit; Backstop Notice.
(a) Backstop Limit. Notwithstanding anything to the contrary in this Agreement, the Purchaser shall never be required to fund an amount in connection with the GS Stockholder Redemptions that is greater than $125,000,000.00 (the Backstop Limit).
(b) Backstop Notice. On the date by which GS Stockholder Redemptions are required to be made in accordance with the Companys certificate of incorporation, as it may be amended from time to time (the Certificate of Incorporation) (which date is two (2) Business Days prior to the date of the SPAC Special Meeting), if and only to the extent that the GS Stockholder Redemptions are of such an amount that they would cause the Cash Shortfall (as such term is defined in the Business Combination Agreement) to be a positive value (i.e. greater than $0 (zero dollars), calculated without including the Backstop Amount (as such term is defined in the Business Combination Agreement) in Available Closing Cash (as such term is defined in the Business Combination Agreement) (the Threshold GS Redemptions Amount), the Company shall deliver a written notice (the Backstop Notice) to the Purchaser setting forth:
(i) the total number of shares of Existing SPAC Common Stock subject to the GS Stockholder Redemptions,
(ii) the Subscription Amount (as defined below),
G-1
Table of Contents
(iii) the resulting BPS Purchase Price (as defined below) (as calculated in accordance with Section 2(a)(i)), which amount, for the avoidance of doubt, shall in no event be greater than the Backstop Limit; and
(iv) the Companys wire instructions.
Subscription Amount means a number of Backstop Purchase Shares equal to the lesser of (x) 12,500,000, and (y) that number of Backstop Purchase Shares, if any, that would be required to be sold hereunder, at a purchase price of $10.00 per share, such that the Cash Shortfall (as such term is defined in the Business Combination Agreement) would be equal to $0 (zero dollars). Notwithstanding the forgoing, for the avoidance of doubt, the Subscription Amount shall not include any shares of New SPAC Class A Common Shares in respect of GS Stockholder Redemptions that have been subsequently withdrawn in accordance with the Companys Certificate of Incorporation and Applicable Law. A Backstop Notice cannot be made and the Company shall not be permitted to deliver a Backstop Notice or cause the Purchaser to acquire any Backstop Purchase Shares to the extent the GS Stockholder Redemptions do not meet the Threshold GS Redemptions Amount. Only one (1) Backstop Notice may be delivered hereunder.
2. Sale and Purchase.
(a) Backstop Purchase Shares.
(i) Subject to the terms and conditions hereof, following delivery of the Backstop Notice by the Company to the Purchaser hereunder, the Company shall issue and sell to the Purchaser, and the Purchaser shall purchase from the Company a number of Backstop Purchase Shares equal to the Subscription Amount for an aggregate purchase price equal to the product of (x) $10.00 multiplied by (y) the number of Backstop Purchase Shares to be issued and sold hereunder (such aggregate purchase price, the BPS Purchase Price). The numbers of shares, per share amounts and purchase price of the Backstop Purchase Shares and the BPS Purchase Price, as applicable, shall be appropriately adjusted to reflect any stock split, stock dividend, stock combination, recapitalization or the like occurring after the date hereof.
(ii) The delivery of the Backstop Notice hereunder shall serve as notice to the Purchaser that the Purchaser will be required to pay the BPS Purchase Price, and acquire the Backstop Purchase Shares, at the BPS Closing (as defined below).
(iii) The closing of the sale of the Backstop Purchase Shares (the BPS Closing) shall be held on the Closing Date. At the BPS Closing, the Company will issue to the Purchaser the Backstop Purchase Shares, registered in the name of the Purchaser, against (and concurrently with) the payment of the BPS Purchase Price to the Company by wire transfer of immediately available funds to the account notified to the Purchaser by the Company in the Backstop Notice.
(b) Delivery of Backstop Purchase Shares.
(i) The Company shall register the Purchaser as the owner of the Backstop Purchase Shares purchased by the Purchaser hereunder (individually or collectively, the Securities) in the register of stockholders of the Company and with the Companys transfer agent by book entry on or promptly after (but in no event more than two (2) Business Days after) the date of the BPS Closing.
(ii) Each register and book entry for the Backstop Purchase Shares purchased by the Purchaser hereunder shall contain a notation, and each certificate (if any) evidencing the Backstop Purchase Shares shall be stamped or otherwise imprinted with a legend, in substantially the following form:
THE SECURITIES REPRESENTED HEREBY HAVE NOT BEEN REGISTERED UNDER THE U.S. SECURITIES ACT OF 1933, AS AMENDED, OR THE SECURITIES LAWS OF ANY STATE OR OTHER JURISDICTION, AND MAY NOT BE TRANSFERRED IN VIOLATION OF SUCH ACT AND LAWS.
(c) Registration Rights. The Purchaser shall enter into the Registration Rights Agreement at Closing, and the Purchaser shall have certain registration rights with respect to the Backstop Purchase Shares as referenced therein (the Registration Rights).
G-2
Table of Contents
3. Representations and Warranties of the Purchaser. The Purchaser represents and warrants to the Company as follows, as of the date hereof and as of the BPS Closing:
(a) Organization and Power. The Purchaser is duly organized, validly existing, and in good standing under the laws of the jurisdiction of its formation (if the concept of good standing is a recognized concept in such jurisdiction) and has all requisite power and authority to carry on its business as presently conducted and as proposed to be conducted.
(b) Authorization. The Purchaser has full power and authority, including any necessary corporate or other organizational authority, to enter into and perform its obligations under this Agreement and any other instrument to be entered into, executed and delivered by or on behalf of the Purchaser in connection with the purchase of the Backstop Purchase Shares. This Agreement, when executed and delivered by the Purchaser, will constitute the valid and legally binding obligation of the Purchaser, enforceable in accordance with its terms, except (a) as limited by applicable bankruptcy, insolvency, reorganization, moratorium, fraudulent conveyance and any other laws of general application affecting enforcement of creditors rights generally, (b) as limited by laws relating to the availability of specific performance, injunctive relief or other equitable remedies, or (c) to the extent the indemnification provisions contained in the Registration Rights may be limited by applicable federal or state securities laws. The signature of the person(s) signing on behalf of the Purchaser is binding on the Purchaser.
(c) Compliance with Other Instruments. The execution, delivery and performance by the Purchaser of this Agreement and the consummation by the Purchaser of the transactions contemplated by this Agreement will not result in any violation or default (i) of any provisions of its organizational documents, if applicable, (ii) of any instrument, judgment, order, writ or decree to which it is a party or by which it is bound, (iii) under any note, indenture or mortgage to which it is a party or by which it is bound, (iv) under any lease, agreement, contract or purchase order to which it is a party or by which it is bound or (v) of any provision of federal or state statute, rule or regulation applicable to the Purchaser, in each case (other than clause (i)), which would have a material adverse effect on the Purchaser or its ability to consummate the transactions contemplated by this Agreement.
(d) No Governmental or other Authorization Required; Consents. Except for any filings and approvals required pursuant to the terms of the Business Combination Agreement, filings with the SEC under the Exchange Act and such other reports under, and such other compliance with, the Exchange Act as may be required in connection with this Agreement, or as may have already been obtained, no authorization or approval or other action by, and no notice to or filing with, any Governmental Authority or any other person will be required to be obtained or made by the Purchaser in connection with the due execution, delivery and performance by the Purchaser of this Agreement.
(e) Restricted Securities. The Purchaser understands that the sale of the Securities to the Purchaser has not been, and will not be, registered under the Securities Act, by reason of a specific exemption from the registration provisions of the Securities Act which depends upon, among other things, the bona fide nature of the investment intent and the accuracy of the Purchasers representations as expressed herein. The Purchaser understands that the Securities are restricted securities under applicable U.S. federal and state securities laws and that, pursuant to these laws, the Purchaser must hold the Securities indefinitely unless they are registered with the SEC and qualified by state authorities, or an exemption from such registration and qualification requirements is available. The Purchaser acknowledges that the Company has no obligation to register or qualify the Securities for resale, except pursuant to the Registration Rights. The Purchaser further acknowledges that if an exemption from registration or qualification is available, it may be conditioned on various requirements including, but not limited to, the time and manner of sale, the holding period for the Securities, and requirements relating to the Company which are outside of the Purchasers control, and which the Company is under no obligation and may not be able to satisfy. The Purchaser acknowledges that the Company filed a registration statement on Form S-1 to consummate its initial public offering with the SEC (the IPO). The Purchaser understands that the sale of the Securities hereunder is not, and is not intended to be, part of the IPO, and that the Purchaser will not be able to rely on the protection of Section 11 of the Securities Act with respect to such sale of the Securities.
G-3
Table of Contents
(f) Review of Disclosed Material. The Purchaser is in receipt of and has carefully read and understands the following items (the Disclosed Material):
(i) the prospectus filed by the Company with the SEC to consummate its IPO (the Prospectus);
(ii) each filing made by the Company with the SEC following the filing of the Prospectus; and
(iii) the Business Combination Agreement (including any amendment thereto) and the New SPAC Certificate of Incorporation and New SPAC Bylaws, each of which is exhibited thereto.
(g) High Degree of Risk. The Purchaser understands that its agreement to purchase the Securities involves a high degree of risk which could cause the Purchaser to lose all or part of its investment, and the Purchaser has the ability to bear the economic risks of an investment in Securities, including a complete loss of its investment. Further, the Purchaser has carefully read, considered and understands (i) any risks identified in the Disclosed Material, and (ii) the risks related to the Transactions, the Company, Mirion and the Securities, and has had the opportunity to retain, at its own expense, and relied upon, appropriate professional advice regarding the financial, taxation and legal implications, risk and consequences of the foregoing
(h) Accredited Investor. The Purchaser is an accredited investor as defined in Rule 501(a) of Regulation D promulgated under the Securities Act.
(i) Adequacy of Financing. The Purchaser has, or will have at the BPS Closing, available to it sufficient clear funds to satisfy its obligations under this Agreement, without restriction or conditions on payment to the Company except as provided hereunder.
(j) Purchasers Knowledge and Skill. The Purchaser has knowledge, skill and experience in financial, business and investment matters relating to investments of this type and is capable of evaluating the merits and risks of such investment and protecting its interests in connection with the acquisition of Backstop Purchase Shares.
(k) Own Investigations. In making its investment decision to purchase Securities, the Purchaser is relying solely on investigations made by it and its representatives and its assessment, and the assessment of any of its professional advisers, of the merits of an acquisition of Securities.
(l) No SEC Approval. The Securities have not been approved or disapproved by the SEC or any state securities commission, nor has the SEC or any state securities commission passed upon the accuracy or adequacy of any representations by the Company.
(m) No Other Representations or Warranties. The Purchaser acknowledges that neither the Company nor any of its representatives has made or makes any representation or warranty to the Purchaser in respect of the Company, Mirion or the Transactions other than, in the case of the Company, the representations and warranties contained in this Agreement.
4. Representations and Warranties of the Company. The Company represents and warrants to the Purchaser as follows:
(a) Incorporation and Corporate Power. The Company is validly existing and in good standing under the laws of the State of Delaware, with all corporate power and authority to own, lease and operate its properties and conduct its business as presently conducted and to enter into, deliver and perform its obligations under this Agreement.
(b) Capitalization. The authorized share capital of the Company consists, as of the date hereof, of:
(i) 500,000,000 shares of Class A common stock, par value $0.0001 per share, 75,000,000 of which are issued and outstanding, and all of the outstanding shares of Class A common stock have been duly authorized, are fully paid and nonassessable and were issued in compliance with all Applicable Laws;
(ii) 50,000,000 shares of Class B common stock, par value $0.0001 per share, 18,750,000 of which are issued and outstanding, and all of the outstanding shares of Class B common stock have been duly authorized, are fully paid and nonassessable and were issued in compliance with all Applicable Laws; and
G-4
Table of Contents
(iii) 5,000,000 shares of Existing SPAC Preferred Stock, none of which are issued and outstanding.
(c) Authorization. All corporate action required to be taken by the Companys Board of Directors and shareholders in order to authorize the Company to enter into this Agreement, and to issue the Backstop Purchase Shares at the BPS Closing has been taken or will be taken prior to the BPS Closing, as applicable. All action on the part of the shareholders, directors and officers of the Company necessary for the execution and delivery of this Agreement, the performance of all obligations of the Company under this Agreement to be performed as of the BPS Closing, and the issuance and delivery of the Backstop Purchase Shares and the securities issuable upon conversion or exercise (in each case, if applicable) of the Backstop Purchase Shares has been taken or will be taken prior to the BPS Closing, as applicable. This Agreement, when executed and delivered by the Company, shall constitute the valid and legally binding obligation of the Company, enforceable against the Company in accordance with its terms except (i) as limited by applicable bankruptcy, insolvency, reorganization, moratorium, fraudulent conveyance, or other laws of general application relating to or affecting the enforcement of creditors rights generally, (ii) as limited by laws relating to the availability of specific performance, injunctive relief, or other equitable remedies, or (iii) to the extent the indemnification provisions contained in the Registration Rights may be limited by applicable federal or state securities laws.
(d) Valid Issuance of Backstop Purchase Shares. The Backstop Purchase Shares, when issued, sold and delivered in accordance with the terms and for the consideration set forth in this Agreement and registered in the register of members of the Company, will be validly issued, fully paid and nonassessable and free of all preemptive or similar rights, liens, encumbrances and charges with respect to the issue thereof and restrictions on transfer, other than restrictions on transfer specified under this Agreement, applicable state and federal securities laws and liens or encumbrances created by or imposed by the Purchaser. Assuming the accuracy of the representations of the Purchaser in this Agreement and subject to the filings described in Section 4(e) below, the Backstop Purchase Shares will be issued in compliance with all applicable federal and state securities laws.
(e) Governmental Consents and Filings. Assuming the accuracy of the representations and warranties made by the Purchaser in this Agreement, no consent, approval, order or authorization of, or registration, qualification, designation, declaration or filing with, any federal, state or local governmental authority is required on the part of the Company in connection with the consummation of the transactions contemplated by this Agreement, except for any filings required pursuant to Regulation D of the Securities Act, applicable state securities laws, and pursuant to the Registration Rights.
(f) Compliance with Other Instruments. The execution, delivery and performance of this Agreement and the consummation of the transactions contemplated by this Agreement by the Company will not result in any violation or default (i) of any provisions of the Companys Certificate of Incorporation or its other governing documents, (ii) of any instrument, judgment, order, writ or decree to which the Company is a party or by which the Company is bound, (iii) under any note, indenture or mortgage to which the Company is a party or by which the Company is bound, (iv) under any lease, agreement, contract or purchase order to which the Company is a party or by which the Company is bound or (v) of any provision of federal or state statute, rule or regulation applicable to the Company, in each case (other than clause (i)) which would have a material adverse effect on the Company or its ability to consummate the transactions contemplated by this Agreement.
(g) Limited Operations and Operating History. As of the date hereof, the Company has not conducted any operations other than organizational activities and activities in connection with its IPO, its search for a potential business combination and financing in connection therewith.
(h) Absence of Litigation. As of the date hereof, there is no action, suit, proceeding, inquiry or investigation before or by any court, public board, government agency, self-regulatory organization or body pending or, to the knowledge of the Company, threatened against or affecting the Company or any of the Companys officers or directors, whether of a civil or criminal nature or otherwise, in their capacities as such.
(i) No General Solicitation. Neither the Company, nor any of its officers, directors, employees, agents or shareholders has either directly or indirectly, including through a broker or finder, (i) engaged in any general solicitation, or (ii) published any advertisement in connection with the sale of the Backstop Purchase Shares.
G-5
Table of Contents
(j) No Other Representations and Warranties; Non-Reliance. Except for the specific representations and warranties contained in this Section 4 and in any certificate or agreement delivered pursuant hereto, the Company has not made, does not make and shall not be deemed to make any other express or implied representation or warranty with respect to the Company, the sale and purchase of the Backstop Purchase Shares, the IPO, the Transactions or a potential business combination, and the Company disclaims any such representation or warranty. Except for the specific representations and warranties expressly made by the Purchaser in Section 3 of this Agreement and in any certificate or agreement delivered pursuant hereto, the Company specifically disclaims that it is relying upon any other representations or warranties that may have been made by the Purchaser. Notwithstanding anything to the contrary in this Agreement, nothing in this Section 4(j) shall limit any claim or cause of action (or recovery in connection therewith) with respect to fraud.
5. Trust Account. Notwithstanding anything to the contrary set forth herein, the Purchaser acknowledges that the Company has established a trust account containing the proceeds of its IPO and from certain private placements (collectively, with interest accrued from time to time thereon, the Trust Account). The Purchaser agrees that (i) it has no right, title, interest or claim of any kind in or to any monies held in the Trust Account, and (ii) it shall have no right of set-off or any right, title, interest or claim of any kind (Claim) to, or to any monies in, the Trust Account, in each case in connection with this Agreement, and hereby irrevocably and unconditionally waives any Claim to, or to any monies in, the Trust Account that it may have in connection with this Agreement; provided, however, that nothing in this Section 5 shall be deemed to limit Purchasers right, title, interest or claim to the Trust Account by virtue of such Purchasers record or beneficial ownership of securities of the Company, including, but not limited to, any redemption right with respect to any such securities of the Company. In the event the Purchaser has any Claim against the Company under this Agreement, the Purchaser shall pursue such Claim solely against the Company and its assets outside the Trust Account and not against the property or any monies in the Trust Account. The Purchaser agrees and acknowledges that such waiver is material to this Agreement and has been specifically relied upon by the Company to induce the Company to enter into this Agreement and the Purchaser further intends and understands such waiver to be valid, binding and enforceable under Applicable Law. In the event the Purchaser, in connection with this Agreement, commences any action or proceeding which seeks, in whole or in part, relief against the funds held in the Trust Account or distributions therefrom or against any of the Companys stockholders, whether in the form of monetary damages or injunctive relief, the Purchaser shall be obligated to pay to the Company all of its legal fees and costs in connection with any such action in the event that the Company prevails in such action or proceeding.
6. BPS Closing Conditions.
(a) The obligation of the Purchaser to purchase the Backstop Purchase Shares at the BPS Closing under this Agreement shall be subject to the fulfillment, at or prior to the BPS Closing of each of the following conditions, any of which, to the extent permitted by Applicable Laws, may be waived by the Purchaser:
(i) The Transactions shall be consummated substantially concurrently with, and immediately following, the purchase of the Backstop Purchase Shares;
(ii) All conditions precedent to Closing set forth in the Business Combination Agreement shall have been satisfied or waived (other than those conditions which, by their nature, are to be satisfied upon Closing); and
(iii) No provision of Applicable Law, and no judgment, injunction, order or decree of any applicable Governmental Authority, shall prohibit the consummation of the transactions contemplated hereby.
(b) The obligation of the Company to sell the Backstop Purchase Shares at the BPS Closing under this Agreement shall be subject to the fulfillment, at or prior to the BPS Closing of each of the following conditions, any of which, to the extent permitted by applicable laws, may be waived by the Company:
(i) The Transactions shall be consummated substantially concurrently with, and immediately following, the purchase of the Backstop Purchase Shares;
G-6
Table of Contents
(ii) The representations and warranties of the Purchaser set forth in Section 3 of this Agreement shall have been true and correct as of the date hereof and shall be true and correct as of the BPS Closing, as applicable, with the same effect as though such representations and warranties had been made on and as of such date (other than any such representation or warranty that is made by its terms as of a specified date, which shall be true and correct as of such specified date), except where the failure to be so true and correct would not have a material adverse effect on the Purchaser or its ability to consummate the transactions contemplated by this Agreement;
(iii) The Purchaser shall have performed, satisfied and complied in all material respects with the covenants, agreements and conditions required by this Agreement to be performed, satisfied or complied with by the Purchaser at or prior to the BPS Closing; and
(iv) No provision of Applicable Law, and no judgment, injunction, order or decree of any applicable Governmental Authority, shall prohibit the consummation of the transactions contemplated hereby.
7. Termination. This Agreement may be terminated at any time prior to the BPS Closing:
(a) by written consent of each of the Company, the Purchaser, Mirion and the Charterhouse Parties; or
(b) automatically:
(i) upon the consummation of the Transactions without the sale to the Purchaser of any Backstop Purchase Shares (whether or not a Backstop Notice has been delivered); or
(ii) upon the termination of the Business Combination Agreement, as provided under the terms therein.
In the event of any termination of this Agreement pursuant to this Section 7, the BPS Purchase Price, if previously paid, and all the Purchasers funds paid in connection herewith shall be promptly returned to the Purchaser in accordance with written instructions provided by the Purchaser to the Company, and thereafter this Agreement shall forthwith become null and void and have no effect, without any liability on the part of the Purchaser or the Company and their respective directors, officers, employees, partners, managers, members, or shareholders and all rights and obligations of each party shall cease; provided, however, that nothing contained in this Section 7 shall relieve either party from liabilities or damages arising out of any fraud or willful breach by such party of any of its representations, warranties, covenants or agreements contained in this Agreement. Section 5 shall survive termination of this Agreement.
8. General Provisions.
(a) Notices. Any notice or communication required or permitted hereunder shall be in writing and either delivered personally, emailed or sent by overnight mail via a reputable overnight carrier, or sent by certified or registered mail, postage prepaid, and shall be deemed to be given and received (i) when so delivered personally, (ii) when sent, with no mail undeliverable or other rejection notice, if sent by email, or (iii) three (3) Business Days after the date of mailing to the address below or to such other address or addresses as such person may hereafter designate by notice given hereunder:
| (i) | If to the Purchaser, to: |
GSAM Holdings LLC
200 West Street
New York, New York 10282
Attention: Thomas R. Knott, David S. Plutzer
E-mail: tom.knott@gs.com; david.plutzer@gs.com
G-7
Table of Contents
with a copy (which shall not constitute notice) to:
Weil, Gotshal & Manges LLP
767 Fifth Avenue
New York, NY 10153
Attention: Michael J. Aiello, Brian Parness
E-mail: michael.aiello@weil.com; brian.parness@weil.com
| (ii) | If to the Company, to: |
GS Acquisition Holdings Corp II
200 West Street
New York, New York 10282
Attention: Thomas R. Knott, David S. Plutzer
E-mail: tom.knott@gs.com; david.plutzer@gs.com
with a copy (which shall not constitute notice), (1) if prior to Closing, to:
Weil, Gotshal & Manges LLP
767 Fifth Avenue
New York, NY 10153
Attention: Michael J. Aiello, Brian Parness
E-mail: michael.aiello@weil.com; brian.parness@weil.com
or (2) if following Closing to:
Davis Polk & Wardwell LLP
1600 El Camino Real Ste. 100
Menlo Park, California 94025
Attention: Alan F. Denenberg, Stephen Salmon
E-mail: alan.denenberg@davispolk.com; stephen.salmon@davispolk.com
with a copy (which copy shall not constitute notice) to:
Freshfields Bruckhaus Deringer LLP
601 Lexington Avenue, 31st Floor
New York, New York 10019
Attention: Valerie Ford Jacob
E-mail: valerie.jacob@freshfields.com
Freshfields Bruckhaus Deringer LLP
9 avenue de Messine
75008 Paris, France
Attention: Yann Gozal
E-mail: yann.gozal@freshfields.com
(b) Entire Agreement. This Agreement constitutes the entire agreement, and supersedes all other prior agreements, understandings, representations and warranties, both written and oral, among the parties, with respect to the subject matter hereof, including any commitment letter entered into relating to the subject matter hereof.
(c) No Third Party Beneficiaries; Exception. Except to the extent expressly set forth in Sections 7(a), 8(e), 8(j) and 8(q), this Agreement shall be binding on, and inure solely to the benefit of, the parties hereto and their respective successors and assigns, and nothing set forth in this Agreement shall be construed to confer upon or give any Person, other than the parties hereto and their respective successors and permitted assigns, any benefits, rights or remedies under or by reason of, or any rights to enforce or cause the Company to enforce, this Agreement; provided, however, that Mirion and the Charterhouse Parties are intended third party beneficiaries of Sections 2, 3, 7(a), 8(e), 8(j) and 8(q) of this Agreement to the extent expressly set forth therein.
G-8
Table of Contents
(d) Successors. All of the terms, agreements, covenants, representations, warranties, and conditions of this Agreement are binding upon, and inure to the benefit of and are enforceable by, the parties hereto and their respective successors. Nothing in this Agreement, express or implied, is intended to confer upon any party other than the parties hereto or their respective successors and assigns any rights, remedies, obligations or liabilities under or by reason of this Agreement, except as expressly provided in this Agreement.
(e) Assignments. Except as otherwise specifically provided herein, no party hereto may assign either this Agreement or any of its rights, interests, or obligations hereunder without the prior written consent of the other party, Mirion and the Charterhouse Parties. Notwithstanding the foregoing, the Purchaser may assign and delegate all or a portion of its rights and obligations to purchase the Backstop Purchase Shares to one or more other persons upon the consent of the Company, Mirion and the Charterhouse Parties (which consent shall not be unreasonably conditioned, withheld or delayed); provided, however, that no consent of the Company, Mirion or the Charterhouse Parties shall be required if such assignment or delegation is to an Affiliate, employee, partner or client of Purchaser or its Affiliates; provided, further, that no such assignment or delegation shall relieve the Purchaser of its obligations hereunder (including its obligation to purchase the Backstop Purchase Shares) and the Company shall be entitled to pursue all rights and remedies against the Purchaser in respect its obligations subject to the terms and conditions hereof. Any purported assignment or assumption of this Agreement or any right or obligation hereunder in contravention of this Section 8(e) shall be void ab initio.
(f) Counterparts. This Agreement may be executed in one or more counterparts, all of which when taken together shall be considered one and the same agreement and shall become effective when counterparts have been signed by each party and delivered to the other parties, it being understood that the parties need not sign the same counterpart.
(g) Headings and Captions. The headings and captions of the various subdivisions of this Agreement are for convenience of reference only and shall in no way modify or affect the meaning or construction of any of the terms or provisions hereof.
(h) Governing Law. This Agreement, and any claim or cause of action hereunder based upon, arising out of or related to this Agreement (whether based on law, in equity, in contract, in tort or any other theory) or the negotiation, execution, performance or enforcement of this Agreement, shall be governed by and construed in accordance with the laws of the State of Delaware, without giving effect to the principles of conflicts of law thereof.
(i) Consent to Jurisdiction; Waiver of Jury Trial. Each of the parties irrevocably consents to the exclusive jurisdiction and venue of the Court of Chancery of the State of Delaware, provided that if subject matter jurisdiction over the matter that is the subject of the legal proceeding is vested exclusively in the U.S. federal courts, such legal proceeding shall be heard in the U.S. District Court for the District of Delaware (together with the Court of Chancery of the State of Delaware, Chosen Courts), in connection with any matter based upon or arising out of this Agreement. Each party hereby waives, and shall not assert as a defense in any legal dispute, that (i) such person is not personally subject to the jurisdiction of the Chosen Courts for any reason, (ii) such legal proceeding may not be brought or is not maintainable in the Chosen Courts, (iii) such persons property is exempt or immune from execution, (iv) such legal proceeding is brought in an inconvenient forum or (v) the venue of such legal proceeding is improper. Each party hereby consents to service of process in any such proceeding in any manner permitted by Delaware law, further consents to service of process by nationally recognized overnight courier service guaranteeing overnight delivery, or by registered or certified mail, return receipt requested, at its address specified pursuant to Section 8(a) and waives and covenants not to assert or plead any objection which they might otherwise have to such manner of service of process. Notwithstanding the foregoing in this Section 8(i), a party may commence any action, claim, cause of action or suit in a court other than the Chosen Courts solely for the purpose of enforcing an order or judgment issued by the Chosen Courts. TO THE EXTENT NOT PROHIBITED BY APPLICABLE LAW WHICH CANNOT BE WAIVED, EACH OF THE PARTIES WAIVES ANY RIGHT TO TRIAL BY JURY ON ANY CLAIMS OR COUNTERCLAIMS ASSERTED IN ANY LEGAL DISPUTE RELATING TO THIS AGREEMENT WHETHER NOW EXISTING OR HEREAFTER ARISING. IF THE SUBJECT MATTER OF ANY SUCH LEGAL DISPUTE IS ONE IN
G-9
Table of Contents
WHICH THE WAIVER OF JURY TRIAL IS PROHIBITED, NO PARTY SHALL ASSERT IN SUCH LEGAL DISPUTE A NONCOMPULSORY COUNTERCLAIM ARISING OUT OF OR RELATING TO THIS AGREEMENT. FURTHERMORE, NO PARTY SHALL SEEK TO CONSOLIDATE ANY SUCH LEGAL DISPUTE WITH A SEPARATE ACTION OR OTHER LEGAL PROCEEDING IN WHICH A JURY TRIAL CANNOT BE WAIVED.
(j) Modifications and Amendments. This Agreement may not be amended, modified, supplemented or waived except by an instrument in writing, signed by the party against whom enforcement of such amendment, modification, supplement or waiver is sought; provided, that the prior written consent of Mirion and the Charterhouse Parties shall be required for any material amendments, modifications, waivers or supplements (which shall include amendments which (1) create additional conditionality to the Purchasers obligation to purchase the Backstop Purchase Shares, (2) change the Backstop Limit, (3) change Mirions and the Charterhouse Parties rights under this Agreement, or (4) change the economics or delay the timing of any Backstop Notice (including changing the threshold of GS Stockholder Redemptions before which a Backstop Notice can be given under Section 1(b)).
(k) Waiver of Damages. Notwithstanding anything to the contrary contained herein, in no event shall any party be liable for punitive damages in connection with this Agreement; provided, however, that in no event shall the Purchaser be liable for any form of damages, whether such damages are consequential, special or exemplary, in connection with this Agreement in excess of the sum of the Backstop Limit and any reasonable fees and expenses (including, without limitation, legal fees) associated with the collection of such damages.
(l) Severability. If any provision of this Agreement shall be invalid, illegal or unenforceable, the validity, legality or enforceability of the remaining provisions of this Agreement shall not in any way be affected or impaired thereby and shall continue in full force and effect.
(m) Expenses. The parties will each be responsible for their costs and expenses incurred in connection with the preparation, execution and performance of this Agreement and the consummation of the transactions contemplated hereby, including all fees and expenses of agents, representatives, financial advisors, legal counsel and accountants. The Company will be responsible for all fees and expenses incurred in connection with transfer agents, stamp taxes and all of The Depository Trust Companys fees associated with the issuance and resale of the Securities and any securities issuable upon conversion or exercise of the Securities (in each case, if applicable).
(n) Construction. The parties hereto have participated jointly in the negotiation and drafting of this Agreement. If an ambiguity or question of intent or interpretation arises, this Agreement will be construed as if drafted jointly by the parties hereto and no presumption or burden of proof will arise favoring or disfavoring any party hereto because of the authorship of any provision of this Agreement. Any reference to any federal, state, local, or foreign law will be deemed also to refer to law as amended and all rules and regulations promulgated thereunder, unless the context requires otherwise. The words include, includes, and including will be deemed to be followed by without limitation. Pronouns in masculine, feminine, and neuter genders will be construed to include any other gender, and words in the singular form will be construed to include the plural and vice versa, unless the context otherwise requires. The words this Agreement, herein, hereof, hereby, hereunder, and words of similar import refer to this Agreement as a whole and not to any particular subdivision unless expressly so limited. The parties hereto intend that each representation, warranty, and covenant contained herein will have independent significance. If any party hereto has breached any representation, warranty, or covenant contained herein in any respect, the fact that there exists another representation, warranty or covenant relating to the same subject matter (regardless of the relative levels of specificity) which such party hereto has not breached will not detract from or mitigate the fact that such party hereto is in breach of the first representation, warranty, or covenant.
(o) Waiver. No waiver by any party hereto of any default, misrepresentation, or breach of warranty or covenant hereunder, whether intentional or not, may be deemed to extend to any prior or subsequent default, misrepresentation, or breach of warranty or covenant hereunder or affect in any way any rights arising because of any prior or subsequent occurrence.
G-10
Table of Contents
(p) Confidentiality. Except as may be required by law, regulation or applicable stock exchange listing requirements, or upon the request of a Governmental Authority, unless and until the transactions contemplated hereby and the terms hereof are publicly announced or otherwise publicly disclosed by the Company, the parties hereto shall keep confidential and shall not publicly disclose the existence or terms of this Agreement.
(q) Specific Performance; Enforcement. The Purchaser agrees that irreparable damage may occur to the Company, Mirion and the Charterhouse Parties in the event any provision of this Agreement is not performed by the Purchaser in accordance with the terms hereof and that the Company, Mirion and the Charterhouse Parties shall be entitled to specific performance of the terms hereof, in addition to any other remedy at law or equity, without a requirement to post bond or any other security. Subject to the proviso in Section 8(c) and as provided in this Section 8(q), this Agreement may be enforced only by the Company and the Purchaser, and none of the Companys direct or indirect creditors nor any other person that is not a party to this Agreement shall have any right to enforce this Agreement or to cause the Company to enforce this Agreement.
(r) Further Assurances. Each party will, at the request of the other party, promptly take all actions, and execute and deliver all other agreements and documents, which may be reasonably required to give effect to the terms of and the transactions contemplated by this Agreement.
[Signature Page Follows]
G-11
Table of Contents
IN WITNESS WHEREOF, the undersigned have executed this Agreement to be effective as of the date first set forth above.
| GS ACQUISITION HOLDINGS CORP II | ||
| By: | /s/ Thomas R. Knott |
|
| Name: Thomas R. Knott | ||
| Title: Authorized Signatory | ||
| GSAM HOLDINGS LLC | ||
| By: | /s/ Thomas R. Knott |
|
| Name: Thomas R. Knott | ||
| Title: Authorized Signatory | ||
Table of Contents
Final Form
OPTION AGREEMENT
This Option Agreement (this Agreement) is entered into as of [●], 2021, by and between GSAM Holdings LLC, a Delaware limited liability company (the Purchaser), GS Acquisition Holdings Corp II, a Delaware corporation (the Company), and the persons named as Option Sellers on the signature pages hereto (the Option Sellers). Capitalized terms used but not defined in this Agreement shall have the meaning ascribed to such terms in that certain Business Combination Agreement, dated as of the date hereof, by and among the Company, Mirion Technologies (TopCo), Ltd., a Jersey private company limited by shares (Mirion), CCP IX LP No. 1, CCP IX LP No. 2, CCP IX Co-Investment LP and CCP IX Co-Investment No. 2 LP (the Charterhouse Parties), each acting by their general partner, Charterhouse General Partners (IX) Limited, and the other parties named therein (as amended, modified, supplemented or waived from time to time in accordance with its terms, the Business Combination Agreement).
WHEREAS, reference is made to that certain Backstop Agreement, dated as of June 16, 2021 by and between the Company and the Purchaser (as amended, modified, supplemented or waived from time to time in accordance with its terms, the Backstop Agreement) pursuant to which the Purchaser has allocated and committed up to $125,000,000.00 to subscribe for a number of shares of New SPAC Class A Common Shares subject to the amount of Existing SPAC Common Stock that is redeemed and not withdrawn by Existing SPAC Stockholders in connection with the SPAC Special Meeting, if any; and
WHEREAS, the Purchaser is now entering into this Agreement with the Option Sellers, which Option Sellers have made a Cash Election for Shares or a Cash Election for Loan Notes pursuant to the Business Combination Agreement, whereby, in connection with the Closing of the Transactions under the Business Combination Agreement (the BCA Closing), the Purchaser shall have the right (the Call Right), but not the obligation, to purchase from such Option Sellers, on a pro rata basis based on the proportion that such Option Sellers Seller Total Consideration bears to Total Consideration received by Option Sellers making a Cash Election for Shares or a Cash Election for Loan Notes, New SPAC Class A Common Shares issued to such Option Sellers pursuant to the Business Combination Agreement in the amount determined pursuant to Section 2(a) hereof and subject to the limitations set forth herein (the Call Option Shares).
NOW, THEREFORE, in consideration of the promises, representations, warranties and the mutual covenants contained in this Agreement, and for other good and valuable consideration, the receipt, sufficiency and adequacy of which are hereby acknowledged, the parties hereto agree as follows:
1. Purchaser Call Right. From the time that the Backstop Notice (as defined in the Backstop Agreement) is delivered by the Company pursuant to the Backstop Agreement until immediately prior to the Closing (the Call Option Period), the Purchaser shall be entitled to exercise its Call Right to purchase from each Option Seller, on a pro rata basis based on the proportion that such Option Sellers Seller Total Consideration bears to Total Consideration received by Option Sellers making a Cash Election for Shares or a Cash Election for Loan Notes, all or any portion of the Call Option Shares owned by such Option Seller pursuant to this Agreement. The Purchaser shall be entitled to exercise the Call Right in its sole discretion at any time during the Call Option Period by delivering written notice to the Company and the Option Sellers of its election to exercise such right in the form attached hereto as Exhibit A (a Call Notice).
2. Sale and Purchase.
(a) Subject to the terms and conditions hereof, at the Call Option Share Closing (as defined below), the Option Sellers shall sell to the Purchaser an aggregate number of Call Option Shares, free and clear of all liens,
H-1
Table of Contents
equal to (a) 12,500,000 minus (b) the number of shares acquired by the Purchaser pursuant to the Backstop Agreement (if any, the Backstop Shortfall Shares) for an aggregate purchase price equal to the product of (x) $10.00 multiplied by (y) the number of Backstop Shortfall Shares (such aggregate purchase price, the Call Option Purchase Price). The numbers of shares, per share amounts and purchase price of the Call Option Shares and the Call Option Purchase Price, as applicable, shall be appropriately adjusted to reflect any stock split, stock dividend, stock combination, recapitalization or the like occurring after the date hereof.
(b) The closing of the sale of the Call Option Shares (the Call Option Share Closing) shall be held on the Closing Date (as defined in the Business Combination Agreement) immediately following and in connection with the BCA Closing, unless such Call Option Share Closing would require the approval of a Governmental Authority, in which case the Call Option Share Closing shall occur immediately following such approval being obtained. At the Call Option Share Closing, the Purchaser shall pay (or cause to be paid) to the Option Sellers (to the account(s) specified in writing by the Option Sellers in the Election Agreements delivered pursuant to the Business Combination Agreement) the portion of the Call Option Purchase Price attributable to the Call Option Shares purchased by the Purchaser, and each Option Seller shall sell and transfer such Persons Call Option Shares to the Purchaser, free and clear of any lien or encumbrance pursuant to duly executed customary transfer instruments in a form acceptable to Purchaser.
(c) Delivery of Backstop Purchase Shares.
(i) The Company shall register the Purchaser as the owner of the Call Option Shares purchased by the Purchaser hereunder (individually or collectively, the Securities) in the register of stockholders of the Company and with the Companys transfer agent by book entry on or promptly after (but in no event more than two (2) Business Days after) the date of the Call Option Share Closing.
(ii) Each register and book entry for the Call Option Shares purchased by the Purchaser hereunder shall contain a notation, and each certificate (if any) evidencing the Call Option Shares shall be stamped or otherwise imprinted with a legend, in substantially the following form:
THE SECURITIES REPRESENTED HEREBY HAVE NOT BEEN REGISTERED UNDER THE U.S. SECURITIES ACT OF 1933, AS AMENDED, OR THE SECURITIES LAWS OF ANY STATE OR OTHER JURISDICTION, AND MAY NOT BE TRANSFERRED IN VIOLATION OF SUCH ACT AND LAWS.
(d) The Company acknowledges that Purchaser shall have registration rights with respect to the Call Option Shares as referenced in the Registration Rights Agreement that will be entered into by and among the Company, the Sponsor, the Charterhouse Parties, the Option Sellers, the Purchaser and certain other parties thereto in connection with the consummation of the Transactions (the Registration Rights).
(e) Notwithstanding any provision contained herein to the contrary, the Purchaser shall be entitled to deduct or withhold from any amounts otherwise payable to the Option Sellers pursuant to this Agreement such amounts as it is required to deduct or withhold with respect to the making of such payment under any provision of applicable tax law. To the extent that amounts are so withheld and properly paid over to the applicable Governmental Authority in accordance with Applicable Law, such withheld amounts shall be treated for purposes of this Agreement as having been paid to the Option Sellers, and the Purchaser shall furnish to the Option Sellers within ten (10) Business Days of such payment the original or certificated copy of a receipt issued by such Governmental Authority evidencing such payment. In the event that the Purchaser determines that any portion of a payment under this Agreement would be subject to withholding under Applicable Law, the Purchaser shall promptly notify the Option Sellers of such determination but in no event later than ten (10) days prior to the date on which such payment is due. The parties shall reasonably cooperate to eliminate or minimize any such withholding.
3. Representations and Warranties of the Purchaser. The Purchaser represents and warrants to the Company and each Option Seller as follows, as of the date hereof and as of the Call Option Share Closing:
(a) Organization and Power. The Purchaser is duly organized, validly existing, and in good standing under the laws of the jurisdiction of its formation (if the concept of good standing is a recognized concept in
H-2
Table of Contents
such jurisdiction) and has all requisite power and authority to carry on its business as presently conducted and as proposed to be conducted.
(b) Authorization. The Purchaser has full power and authority, including any necessary corporate or other organizational authority, to enter into and perform its obligations under this Agreement and any other instrument to be entered into, executed and delivered by or on behalf of the Purchaser in connection with the purchase of the Call Option Shares. This Agreement, when executed and delivered by the Purchaser, will constitute the valid and legally binding obligation of the Purchaser, enforceable in accordance with its terms, except (a) as limited by applicable bankruptcy, insolvency, reorganization, moratorium, fraudulent conveyance and any other laws of general application affecting enforcement of creditors rights generally, (b) as limited by laws relating to the availability of specific performance, injunctive relief or other equitable remedies, or (c) to the extent the indemnification provisions contained in the Registration Rights may be limited by applicable federal or state securities laws. The signature of the person(s) signing on behalf of the Purchaser is binding on the Purchaser.
(c) Compliance with Other Instruments. The execution, delivery and performance by the Purchaser of this Agreement and the consummation by the Purchaser of the transactions contemplated by this Agreement will not result in any violation or default (i) of any provisions of its organizational documents, if applicable, (ii) of any instrument, judgment, order, writ or decree to which it is a party or by which it is bound, (iii) under any note, indenture or mortgage to which it is a party or by which it is bound, (iv) under any lease, agreement, contract or purchase order to which it is a party or by which it is bound or (v) of any provision of federal or state statute, rule or regulation applicable to the Purchaser, in each case (other than clause (i)), which would have a material adverse effect on the Purchaser or its ability to consummate the transactions contemplated by this Agreement.
(d) No Governmental or other Authorization Required; Consents. Except for any filings and approvals required pursuant to the terms of the Business Combination Agreement, filings with the SEC under the Exchange Act and such other reports under, and such other compliance with, the Exchange Act as may be required in connection with this Agreement, or as may have already been obtained, no authorization or approval or other action by, and no notice to or filing with, any Governmental Authority or any other person will be required to be obtained or made by the Purchaser in connection with the due execution, delivery and performance by the Purchaser of this Agreement.
(e) Litigation. As of the date of this Agreement, there are no Actions pending or, to the knowledge of the Purchaser, threatened against the Purchaser, before any Governmental Authority that would prevent, materially impair or materially delay the Purchaser from performing its obligations hereunder.
(f) Restricted Securities. The Purchaser understands that the sale of the Securities to the Purchaser has not been, and will not be, registered under the Securities Act, by reason of a specific exemption from the registration provisions of the Securities Act which depends upon, among other things, the bona fide nature of the investment intent and the accuracy of the Purchasers representations as expressed herein. The Purchaser understands that the Securities are restricted securities under applicable U.S. federal and state securities laws and that, pursuant to these laws, the Purchaser must hold the Securities indefinitely unless they are registered with the SEC and qualified by state authorities, or an exemption from such registration and qualification requirements is available. The Purchaser acknowledges that the Company has no obligation to register or qualify the Securities for resale, except pursuant to the Registration Rights. The Purchaser further acknowledges that if an exemption from registration or qualification is available, it may be conditioned on various requirements including, but not limited to, the time and manner of sale, the holding period for the Securities, and requirements relating to the Company which are outside of the Purchasers control, and which the Company is under no obligation and may not be able to satisfy. The Purchaser acknowledges that the Company filed a registration statement on Form S-1 to consummate its initial public offering with the SEC (the IPO). The Purchaser understands that the sale of the Securities hereunder is not, and is not intended to be, part of the IPO, and that the Purchaser will not be able to rely on the protection of Section 11 of the Securities Act with respect to such sale of the Securities.
H-3
Table of Contents
(g) Review of Disclosed Material. The Purchaser is in receipt of and has carefully read and understands the following items (the Disclosed Material):
(i) the prospectus filed by the Company with the SEC to consummate its IPO (the Prospectus);
(ii) each filing made by the Company with the SEC following the filing of the Prospectus; and
(iii) the Business Combination Agreement (including any amendment thereto) and the New SPAC Certificate of Incorporation and New SPAC Bylaws, each of which is exhibited thereto.
(h) High Degree of Risk. The Purchaser understands that its agreement to purchase the Securities involves a high degree of risk which could cause the Purchaser to lose all or part of its investment, and the Purchaser has the ability to bear the economic risks of an investment in Securities, including a complete loss of its investment. Further, the Purchaser has carefully read, considered and understands (i) any risks identified in the Disclosed Material, and (ii) the risks related to the Transactions, the Company, Mirion and the Securities, and has had the opportunity to retain, at its own expense, and relied upon, appropriate professional advice regarding the financial, taxation and legal implications, risk and consequences of the foregoing.
(i) Accredited Investor. The Purchaser is an accredited investor as defined in Rule 501(a) of Regulation D promulgated under the Securities Act.
(j) Adequacy of Financing. The Purchaser has, or will have at the Call Option Share Closing, available to it sufficient clear funds to satisfy its obligations under this Agreement, without restriction or conditions on payment to the Option Sellers except as provided hereunder.
(k) Purchasers Knowledge and Skill. The Purchaser has knowledge, skill and experience in financial, business and investment matters relating to investments of this type and is capable of evaluating the merits and risks of such investment and protecting its interests in connection with the acquisition of the Securities.
(l) Own Investigations. In making its investment decision to purchase Securities, the Purchaser is relying solely on investigations made by it and its representatives and its assessment, and the assessment of any of its professional advisers, of the merits of an acquisition of Securities.
(m) No SEC Approval. The Securities have not been approved or disapproved by the SEC or any state securities commission, nor has the SEC or any state securities commission passed upon the accuracy or adequacy of any representations by the Company.
(n) No Other Representations or Warranties. The Purchaser acknowledges that neither the Option Sellers nor any of their representatives has made or makes any representation or warranty to the Purchaser in respect of such Option Seller or the Transactions other than, in the case of the Option Sellers, the representations and warranties contained in this Agreement.
4. Representations and Warranties of the Option Sellers. Each Option Seller hereby represents and warrants to the Purchaser as follows:
(a) Authority; Binding Obligation. If such Option Seller is an entity, such Option Seller has all requisite organizational power and authority to execute, deliver and perform this Agreement. The execution by such Option Seller of this Agreement and the performance of its obligations hereunder have been duly and validly authorized by all required limited partnership or similar corporate action on the part of such Option Seller, and no other proceedings on the part of such Option Seller are required to authorize this Agreement or to perform such Option Sellers obligations hereunder. If such Option Seller is an individual, such Option Seller has all requisite legal capacity, power and authority to execute, deliver and perform this Agreement. This Agreement has been duly executed and delivered by such Option Seller and assuming that this Agreement constitutes the legal, valid and binding obligation of the other parties thereto, constitutes the legal, valid and binding obligation of such Option Seller, enforceable against such Option Seller in accordance with its terms, except to the extent that the enforceability thereof may be limited by the bankruptcy, reorganization, insolvency, moratorium and similar laws of general application relating to or affecting the enforcement of rights of creditors and general principles of equity. The signature of the person(s) signing on behalf of such Option Seller is binding on such Option Seller.
H-4
Table of Contents
(b) Ownership of Shares. Immediately following the BCA Closing, such Option Seller shall be the beneficial or record owner of the New SPAC Class A Common Shares indicated on Schedule 1 hereto, free and clear of any and all liens, mortgages, pledges, security interests, charges, claims or restrictions, other than those created by this Agreement or as disclosed on Schedule 1.
(c) No Defaults or Conflicts. Neither the execution and delivery of this Agreement, or the performance by such Option Seller of its obligations hereunder (a) results in any violation of the applicable organizational documents of such Option Seller, (b) conflicts with, or results in a breach of any of the terms or provisions of, or constitutes a default under any material agreement or instrument to which such Shareholder is a party or by which it is bound or to which the New SPAC Class A Common Shares owned of record or beneficially by such Shareholder is subject; or (c) violates any existing Applicable Law, judgment, order or decree of any Governmental Authority having jurisdiction over such Option Seller or the New SPAC Class A Common Shares owned of record or beneficially by such Shareholder.
(d) No Governmental or other Authorization Required; Consents. Except for filings with the SEC under the Exchange Act and such other reports under, and such other compliance with, the Exchange Act as may be required in connection with this Agreement, no authorization or approval or other action by, and no notice to or filing with, any Governmental Authority or any other person will be required to be obtained or made by such Option Seller in connection with the due execution, delivery and performance by such Option Seller of this Agreement.
(e) Litigation. As of the date of this Agreement, there are no Actions pending or, to the knowledge of such Option Seller, threatened against such Option Seller, before any Governmental Authority that would prevent, materially impair or materially delay such Option Seller from performing their obligations hereunder.
(f) No Other Representations or Warranties. Such Option Seller acknowledges that neither the Purchaser nor any of its representatives has made or makes any representation or warranty to such Option Seller in respect of the Purchaser or the Transactions other than the representations and warranties contained in this Agreement.
5. Trust Account. Notwithstanding anything to the contrary set forth herein, the Purchaser and the Option Sellers acknowledge that the Company has established a trust account containing the proceeds of its IPO and from certain private placements (collectively, with interest accrued from time to time thereon, the Trust Account). The Purchaser and each Option Seller agrees that (i) it has no right, title, interest or claim of any kind in or to any monies held in the Trust Account, and (ii) it shall have no right of set-off or any right, title, interest or claim of any kind (Claim) to, or to any monies in, the Trust Account, in each case in connection with this Agreement, and hereby irrevocably and unconditionally waives any Claim to, or to any monies in, the Trust Account that the Purchaser or the Option Sellers may have in connection with this Agreement; provided, however, that nothing in this Section 5 shall be deemed to limit Purchasers or the Option Sellers right, title, interest or claim to the Trust Account by virtue of such Purchasers or Option Sellers record or beneficial ownership of securities of the Company, including, but not limited to, any redemption right with respect to any such securities of the Company. In the event the Purchaser or any Option Seller has any Claim against the Company under this Agreement, the Purchaser and each Option Seller shall pursue such Claim solely against the Company and its assets outside the Trust Account and not against the property or any monies in the Trust Account. The Purchaser and each Option Seller agrees and acknowledges that such waiver is material to this Agreement and has been specifically relied upon by the Company to induce the Company to enter into this Agreement and the Purchaser and each Option Seller further intends and understands such waiver to be valid, binding and enforceable under Applicable Law. In the event the Purchaser or any Option Seller, in connection with this Agreement, commences any action or proceeding which seeks, in whole or in part, relief against the funds held in the Trust Account or distributions therefrom or against any of the Companys stockholders, whether in the form of monetary damages or injunctive relief, the Purchaser and each Option Seller shall be obligated to pay to the Company all of its legal fees and costs in connection with any such action in the event that the Company prevails in such action or proceeding.
H-5
Table of Contents
6. Closing Conditions.
(a) The obligation of the Purchaser to purchase, and the obligation of the Option Sellers to sell, the Call Option Shares at the Call Option Share Closing under this Agreement shall be subject to the fulfillment, at or prior to the Call Option Share Closing of each of the following conditions, any of which, to the extent permitted by Applicable Laws, may be waived by the Purchaser and the Option Sellers:
(i) The Transactions shall be consummated substantially concurrently with, and immediately preceding, the purchase of the Call Option Shares, provided, that, if any approval by a Governmental Authority is required in connection with the transactions contemplated hereby, the transactions contemplated hereby shall be consummated following such approval being obtained; and
(ii) No provision of Applicable Law, and no judgment, injunction, order or decree of any applicable Governmental Authority, shall prohibit the consummation of the transactions contemplated hereby.
7. Termination. This Agreement may be terminated at any time prior to the Call Option Share Closing:
(a) by written consent of each of the Option Sellers, on the one hand, and the Purchaser, on the other;
(b) automatically upon the termination of the Business Combination Agreement, as provided under the terms therein; or
(c) automatically, if the Call Option Period has ended without valid delivery by the Purchaser of the Call Option Notice.
In the event of any termination of this Agreement pursuant to this Section 7, this Agreement shall forthwith become null and void and have no effect, without any liability on the part of the Purchaser or any Option Seller or any of their respective directors, officers, employees, partners, managers, members, or shareholders and all rights and obligations of each party shall cease; provided, however, that nothing contained in this Section 7 shall relieve either party from liabilities or damages arising out of any fraud or willful breach by such party of any of its representations, warranties, covenants or agreements contained in this Agreement. Section 7 shall survive termination of this Agreement. Section 5 shall survive termination of this Agreement.
8. General Provisions.
(a) Notices. Any notice or communication required or permitted hereunder shall be in writing and either delivered personally, emailed or sent by overnight mail via a reputable overnight carrier, or sent by certified or registered mail, postage prepaid, and shall be deemed to be given and received (i) when so delivered personally, (ii) when sent, with no mail undeliverable or other rejection notice, if sent by email, or (iii) three (3) Business Days after the date of mailing to the address below or to such other address or addresses as such person may hereafter designate by notice given hereunder:
(i) If to the Purchaser, to:
GSAM Holdings LLC
200 West Street
New York, New York 10282
Attention: Thomas R. Knott, David S. Plutzer
E-mail: tom.knott@gs.com; david.plutzer@gs.com
with a copy (which shall not constitute notice) to:
Weil, Gotshal & Manges LLP
767 Fifth Avenue
New York, NY 10153
Attention: Michael J. Aiello, Brian Parness
E-mail: michael.aiello@weil.com; brian.parness@weil.com
H-6
Table of Contents
(ii) If to the Company, to:
GS Acquisition Holdings Corp II
200 West Street
New York, New York 10282
Attention: Thomas R. Knott, David S. Plutzer
E-mail: tom.knott@gs.com; david.plutzer@gs.com
with a copy (which shall not constitute notice), (1) if prior to BCA Closing, to:
Weil, Gotshal & Manges LLP
767 Fifth Avenue
New York, NY 10153
Attention: Michael J. Aiello, Brian Parness
E-mail: michael.aiello@weil.com; brian.parness@weil.com
or (2) if following BCA Closing to:
Davis Polk & Wardwell LLP
1600 El Camino Real Ste. 100
Menlo Park, California 94025
Attention: Alan F. Denenberg, Stephen Salmon
E-mail:alan.denenberg@davispolk.com; stephen.salmon@davispolk.com
with a copy (which copy shall not constitute notice) to:
Freshfields Bruckhaus Deringer LLP
601 Lexington Avenue, 31st Floor
New York, New York 10019
Attention: Valerie Ford Jacob
E-mail: valerie.jacob@freshfields.com
Freshfields Bruckhaus Deringer LLP
9 avenue de Messine
75008 Paris, France
Attention: Yann Gozal
E-mail: yann.gozal@freshfields.com
(iii) If to an Option Seller, to:
the address set forth on the signature page hereto .
(b) Entire Agreement. This Agreement constitutes the entire agreement, and supersedes all other prior agreements, understandings, representations and warranties, both written and oral, among the parties, with respect to the subject matter hereof, including any commitment letter entered into relating to the subject matter hereof.
(c) No Third Party Beneficiaries. This Agreement shall be binding on, and inure solely to the benefit of, the parties hereto and their respective successors and assigns, and nothing set forth in this Agreement shall be construed to confer upon or give any Person, other than the parties hereto and their respective successors and permitted assigns, any benefits, rights or remedies under or by reason of, or any rights to enforce or cause the Purchaser or the Option Sellers to enforce, this Agreement.
(d) Successors. All of the terms, agreements, covenants, representations, warranties, and conditions of this Agreement are binding upon, and inure to the benefit of and are enforceable by, the parties hereto and their respective successors. Nothing in this Agreement, express or implied, is intended to confer upon any party other than the parties hereto or their respective successors and assigns any rights, remedies, obligations or liabilities under or by reason of this Agreement, except as expressly provided in this Agreement.
H-7
Table of Contents
(e) Assignments. Except as otherwise specifically provided herein, no party hereto may assign either this Agreement or any of its rights, interests, or obligations hereunder without the prior written consent of the other parties hereto. Notwithstanding the foregoing, the Purchaser may assign and delegate all or a portion of its rights to purchase the Call Option Shares to one or more other persons upon the consent of each of the Option Sellers (which consent shall not be unreasonably conditioned, withheld or delayed); provided, however, that no consent of the Option Sellers shall be required if such assignment or delegation is to an Affiliate, employee, partner or client of Purchaser or its Affiliates; provided, further, that no such assignment or delegation shall relieve the Purchaser of its obligations hereunder (including its obligation to purchase the Call Option Shares) and the Option Sellers shall be entitled to pursue all rights and remedies against the Purchaser in respect its obligations subject to the terms and conditions hereof. Any purported assignment or assumption of this Agreement or any right or obligation hereunder in contravention of this Section 5(e) shall be void ab initio.
(f) Counterparts. This Agreement may be executed in one or more counterparts, all of which when taken together shall be considered one and the same agreement and shall become effective when counterparts have been signed by each party and delivered to the other parties, it being understood that the parties need not sign the same counterpart.
(g) Headings and Captions. The headings and captions of the various subdivisions of this Agreement are for convenience of reference only and shall in no way modify or affect the meaning or construction of any of the terms or provisions hereof.
(h) Governing Law. This Agreement, and any claim or cause of action hereunder based upon, arising out of or related to this Agreement (whether based on law, in equity, in contract, in tort or any other theory) or the negotiation, execution, performance or enforcement of this Agreement, shall be governed by and construed in accordance with the laws of the State of Delaware, without giving effect to the principles of conflicts of law thereof.
(i) Consent to Jurisdiction; Waiver of Jury Trial. Each of the parties irrevocably consents to the exclusive jurisdiction and venue of the Court of Chancery of the State of Delaware, provided that if subject matter jurisdiction over the matter that is the subject of the legal proceeding is vested exclusively in the U.S. federal courts, such legal proceeding shall be heard in the U.S. District Court for the District of Delaware (together with the Court of Chancery of the State of Delaware, Chosen Courts), in connection with any matter based upon or arising out of this Agreement. Each party hereby waives, and shall not assert as a defense in any legal dispute, that (i) such person is not personally subject to the jurisdiction of the Chosen Courts for any reason, (ii) such legal proceeding may not be brought or is not maintainable in the Chosen Courts, (iii) such persons property is exempt or immune from execution, (iv) such legal proceeding is brought in an inconvenient forum or (v) the venue of such legal proceeding is improper. Each party hereby consents to service of process in any such proceeding in any manner permitted by Delaware law, further consents to service of process by nationally recognized overnight courier service guaranteeing overnight delivery, or by registered or certified mail, return receipt requested, at its address specified pursuant to Section 7(a) and waives and covenants not to assert or plead any objection which they might otherwise have to such manner of service of process. Notwithstanding the foregoing in this Section 7(i), a party may commence any action, claim, cause of action or suit in a court other than the Chosen Courts solely for the purpose of enforcing an order or judgment issued by the Chosen Courts. TO THE EXTENT NOT PROHIBITED BY APPLICABLE LAW WHICH CANNOT BE WAIVED, EACH OF THE PARTIES WAIVES ANY RIGHT TO TRIAL BY JURY ON ANY CLAIMS OR COUNTERCLAIMS ASSERTED IN ANY LEGAL DISPUTE RELATING TO THIS AGREEMENT WHETHER NOW EXISTING OR HEREAFTER ARISING. IF THE SUBJECT MATTER OF ANY SUCH LEGAL DISPUTE IS ONE IN WHICH THE WAIVER OF JURY TRIAL IS PROHIBITED, NO PARTY SHALL ASSERT IN SUCH LEGAL DISPUTE A NONCOMPULSORY COUNTERCLAIM ARISING OUT OF OR RELATING TO THIS AGREEMENT. FURTHERMORE, NO PARTY SHALL SEEK TO CONSOLIDATE ANY SUCH LEGAL DISPUTE WITH A SEPARATE ACTION OR OTHER LEGAL PROCEEDING IN WHICH A JURY TRIAL CANNOT BE WAIVED.
H-8
Table of Contents
(j) Modifications and Amendments. This Agreement may not be amended, modified, supplemented or waived except by an instrument in writing, signed by the party against whom enforcement of such amendment, modification, supplement or waiver is sought.
(k) Severability. If any provision of this Agreement shall be invalid, illegal or unenforceable, the validity, legality or enforceability of the remaining provisions of this Agreement shall not in any way be affected or impaired thereby and shall continue in full force and effect.
(l) Expenses. The parties will each be responsible for their costs and expenses incurred in connection with the preparation, execution and performance of this Agreement and the consummation of the transactions contemplated hereby, including all fees and expenses of agents, representatives, financial advisors, legal counsel and accountants. The Company will be responsible for all fees and expenses incurred in connection with transfer agents, stamp taxes and all of The Depository Trust Companys fees associated with the resale of the Call Option Shares.
(m) Construction. The parties hereto have participated jointly in the negotiation and drafting of this Agreement. If an ambiguity or question of intent or interpretation arises, this Agreement will be construed as if drafted jointly by the parties hereto and no presumption or burden of proof will arise favoring or disfavoring any party hereto because of the authorship of any provision of this Agreement. Any reference to any federal, state, local, or foreign law will be deemed also to refer to law as amended and all rules and regulations promulgated thereunder, unless the context requires otherwise. The words include, includes, and including will be deemed to be followed by without limitation. Pronouns in masculine, feminine, and neuter genders will be construed to include any other gender, and words in the singular form will be construed to include the plural and vice versa, unless the context otherwise requires. The words this Agreement, herein, hereof, hereby, hereunder, and words of similar import refer to this Agreement as a whole and not to any particular subdivision unless expressly so limited. The parties hereto intend that each representation, warranty, and covenant contained herein will have independent significance. If any party hereto has breached any representation, warranty, or covenant contained herein in any respect, the fact that there exists another representation, warranty or covenant relating to the same subject matter (regardless of the relative levels of specificity) which such party hereto has not breached will not detract from or mitigate the fact that such party hereto is in breach of the first representation, warranty, or covenant.
(n) Waiver. No waiver by any party hereto of any default, misrepresentation, or breach of warranty or covenant hereunder, whether intentional or not, may be deemed to extend to any prior or subsequent default, misrepresentation, or breach of warranty or covenant hereunder or affect in any way any rights arising because of any prior or subsequent occurrence.
(o) Specific Performance; Enforcement. Each party agrees that irreparable damage may occur to the other parties hereto in the event any provision of this Agreement is not performed by such party in accordance with the terms hereof and that the other parties shall be entitled to specific performance of the terms hereof, in addition to any other remedy at law or equity, without a requirement to post bond or any other security. This Agreement may be enforced only by the parties to this Agreement, and no person that is not a party to this Agreement shall have any right to enforce this Agreement.
(p) Further Assurances. Each party will, at the request of the other party, promptly take all actions, and execute and deliver all other agreements and documents, which may be reasonably required to give effect to the terms of and the transactions contemplated by this Agreement, including using reasonable best efforts to obtain any Governmental Approvals required to effect the Call Option Closing.
[Signature Page Follows]
H-9
Table of Contents
IN WITNESS WHEREOF, the undersigned have executed this Agreement to be effective as of the date first set forth above.
| GSAM HOLDINGS LLC | ||
| By: |
|
|
| Name: | ||
| Title: | ||
| GS ACQUISITION HOLDINGS CORP II | ||
| By: |
|
|
| Name: | ||
| Title: | ||
Table of Contents
| OPTION SELLERS: | ||
| [●] | ||
| By: |
|
|
| Name: | ||
| Title: | ||
| Address: | ||
| [●] | ||
| By: |
|
|
| Name: | ||
| Title: | ||
| Address: | ||
| [●] | ||
| By: |
|
|
| Name: | ||
| Title: | ||
| Address: | ||
Table of Contents
Schedule 1
| Option Seller |
New SPAC Class A Common Shares |
|
| [●] | [●] | |
| [●] | [●] | |
| [●] | [●] | |
| [●] | [●] | |
| [●] | [●] | |
| [●] | [●] |
Table of Contents
Exhibit A
Call Notice
(see attached)
Table of Contents
FORM OF CALL NOTICE
[PURCHASER HEADER]
[ADDRESS]
[DATE]
[OPTION SELLER ADDRESS]
Re: Call Notice
Ladies and Gentlemen:
Reference is made to that certain Option Agreement (the Agreement), dated as of [●], 2021. Each capitalized term used but not defined herein has the meaning given to it in the Agreement.
The Purchaser hereby exercises its Call Right under the Agreement to purchase [●] of your New SPAC Class A Common Shares.
If you have any questions, please contact [CONTACT] at [EMAIL].
| Sincerely, |
||
| GSAM HOLDINGS LLC | ||
| By: |
|
|
| Name: [●] | ||
| Title: [●] | ||
Table of Contents
Copy:
[NAME]
[ADDRESS]
Attention: [NAME]
Email: [EMAIL]
[NAME]
[ADDRESS]
Attention: [NAME]
Email: [EMAIL
[NAME]
[ADDRESS]
Attention: [NAME]
Email: [EMAIL
[NAME]
[ADDRESS]
Attention: [NAME]
Email: [EMAIL
Table of Contents
Final Form
MIRION TECHNOLOGIES, INC.
OMNIBUS INCENTIVE PLAN
Section 1. Purpose. The purpose of the Mirion Technologies, Inc. Omnibus Incentive Plan (as amended from time to time, the Plan) is to motivate and reward employees and other individuals to perform at the highest level and contribute significantly to the success of Mirion Technologies, Inc., a Delaware corporation (the Company), thereby furthering the best interests of the Company and its shareholders. The Plan shall serve as the primary plan under which equity-based incentives are awarded on a worldwide basis to Participants.
Section 2. Definitions. As used in the Plan, the following terms shall have the meanings set forth below:
(a) Affiliate means any entity that, directly or indirectly through one or more intermediaries controls, is controlled by or is under common control with, the Company.
(b) Award means any Option, SAR, Restricted Stock, RSU, Performance Award, Other Cash-Based Award or Other Stock-Based Award granted under the Plan.
(c) Award Agreement means any agreement, contract or other instrument or document (including in electronic form) evidencing any Award granted under the Plan, which may, but need not, be executed or acknowledged by a Participant.
(d) Beneficial Owner has the meaning ascribed to such term in Rule 13d-3 under the Exchange Act.
(e) Beneficiary means a Person entitled to receive payments or other benefits or exercise rights that are available under the Plan in the event of a Participants death. If no such Person can be named or is named by a Participant, or if no Beneficiary designated by a Participant is eligible to receive payments or other benefits or exercise rights that are available under the Plan at a Participants death, such Participants Beneficiary shall be such Participants estate.
(f) Board means the Board of Directors of the Company.
(g) Cause is as defined in Participants Service Agreement, if any, or Award Agreement or, if not so defined, means: (i) any theft, fraud, embezzlement, dishonesty, willful misconduct, breach of fiduciary duty for personal profit, falsification of any documents or records of the Company or any of its Affiliates, felony or similar act by Participant (whether or not related to Participants relationship with the Company); (ii) an act of moral turpitude by Participant, or any act that causes significant injury to, or is otherwise adversely affecting, the reputation, business, assets, operations or business relationship of the Company (or a Subsidiary or Affiliate, when applicable); (iii) any breach by Participant of any material agreement with or of any material duty of Participant to the Company or any Subsidiary or Affiliate thereof (including breach of confidentiality, non-disclosure, non-use non-competition or non-solicitation covenants towards the Company or any of its Affiliates) or failure to abide by code of conduct or other policies (including, without limitation, policies relating to confidentiality and reasonable workplace conduct); (iv) any act which constitutes a breach of a Participants fiduciary duty towards the Company or an Affiliate or Subsidiary, including disclosure of confidential or proprietary information thereof or acceptance or solicitation to receive unauthorized or undisclosed benefits, irrespective of their nature, or funds, or promises to receive either, from individuals, consultants or corporate entities that the Company or a Subsidiary does business with; (v) Participants unauthorized use, misappropriation, destruction, or diversion of any tangible or intangible asset or corporate opportunity of the Company or any of its Affiliates (including, without limitation, the improper use or disclosure of confidential or proprietary information); or (vi) any circumstances that constitute grounds for termination for cause under Participants Service Agreement with the Company or Affiliate, to the extent applicable. For the avoidance of doubt, the determination as to whether a termination is for Cause for purposes of this Plan, shall be made in good faith by the Committee and shall be final and binding on Participant.
I-1
Table of Contents
(h) Change in Control means the occurrence of any one or more of the following events:
(i) any Person, other than (A) any employee plan established by the Company or any Subsidiary, (B) the Company or any of its Affiliates, (C) an underwriter temporarily holding securities pursuant to an offering of such securities, or (D) an entity owned, directly or indirectly, by shareholders of the Company in substantially the same proportions as their ownership of the Company, is (or becomes, during any 12-month period) the Beneficial Owner, directly or indirectly, of securities of the Company (not including in the securities beneficially owned by such Person any securities acquired directly from the Company or its Affiliates other than in connection with the acquisition by the Company or its Affiliates of a business) representing 50% or more of the total voting power of the stock of the Company; provided that the provisions of this subsection (i) are not intended to apply to or include as a Change in Control any transaction that is specifically excepted from the definition of Change in Control under subsection (iii) below;
(ii) a change in the composition of the Board such that, during any 12-month period, the individuals who, as of the beginning of such period, constitute the Board (the Existing Board) cease for any reason to constitute at least 50% of the Board; provided, however, that any individual becoming a member of the Board subsequent to the beginning of such period whose election, or nomination for election by the Companys shareholders, was either (a) a result of the ordinary annual director elections or (b) approved by a vote of at least a majority of the Directors immediately prior to the date of such appointment or election, in each case, shall be considered as though such individual were a member of the Existing Board; provided further, that, notwithstanding the foregoing, no individual whose initial assumption of office occurs as a result of either an actual or threatened election contest (as such terms are used in Rule 14a-11 or Regulation 14A promulgated under the Exchange Act or successor statutes or rules containing analogous concepts) or other actual or threatened solicitation of proxies or consents by or on behalf of an individual, corporation, partnership, group, associate or other entity or Person other than the Board, shall in any event be considered to be a member of the Existing Board;
(iii) the consummation of a merger, amalgamation or consolidation of the Company with any other corporation or other entity, or the issuance of voting securities in connection with such a transaction pursuant to applicable stock exchange requirements; provided that immediately following such transaction the voting securities of the Company outstanding immediately prior thereto do not continue to represent (either by remaining outstanding or by being converted into voting securities of the surviving entity of such transaction or parent entity thereof) 50% or more of the total voting power of the Companys stock (or, if the Company is not the surviving entity of such merger or consolidation, 50% or more of the total voting power and total fair market value of the stock of such surviving entity or parent entity thereof); and provided, further, that such a transaction effected to implement a recapitalization of the Company (or similar transaction) in which no Person is or becomes the Beneficial Owner, directly or indirectly, of securities of the Company (not including in the securities beneficially owned by such Person any securities acquired directly from the Company or its Affiliates other than in connection with the acquisition by the Company or its Affiliates of a business) representing 50% or more of either the then-outstanding Shares or the combined voting power and total fair market value of the Companys then-outstanding voting securities shall not be considered a Change in Control; or
(iv) the sale or disposition by the Company of all or substantially all of the Companys assets in which any Person acquires (or has acquired during the 12-month period ending on the date of the most recent acquisition by such Person) assets from the Company that have a total gross fair market value equal to more than 50% of the total gross fair market value of all of the assets of the Company immediately prior to such acquisition or acquisitions.
Notwithstanding the foregoing, (A) no Change in Control shall be deemed to have occurred if there is consummated any transaction or series of integrated transactions immediately following which the record holders of the Shares immediately prior to such transaction or series of transactions continue to have
I-2
Table of Contents
substantially the same proportionate ownership in an entity which owns substantially all of the assets of the Company immediately prior to such transaction or series of transactions and (B) no Change in Control shall be deemed to have occurred upon the acquisition of additional control of the Company by any Person that is considered to effectively control the Company. In no event will a Change in Control be deemed to have occurred if any Participant is part of a group within the meaning of Section 13(d)(3) of the Exchange Act that effects a Change in Control. Notwithstanding the foregoing or any provision of any Award Agreement to the contrary, for any Award that provides for accelerated distribution on a Change in Control of amounts that constitute deferred compensation (as defined in Section 409A of the Code), if the event that constitutes such Change in Control does not also constitute a change in the ownership or effective control of the Company, or in the ownership of a substantial portion of the Companys assets (in either case, as defined in Section 409A of the Code), such amount shall not be distributed on such Change in Control but instead shall vest as of such Change in Control and shall be distributed on the scheduled payment date specified in the applicable Award Agreement, except to the extent that earlier distribution would not result in the Participant who holds such Award incurring interest or additional tax under Section 409A of the Code.
(i) Code means the Internal Revenue Code of 1986, as amended from time to time, and the rules, regulations and guidance thereunder. Any reference to a provision in the Code shall include any successor provision thereto.
(j) Committee means the compensation committee of the Board unless another committee is designated by the Board If there is no compensation committee of the Board and the Board does not designate another committee, references herein to the Committee shall refer to the Board.
(k) Consultant means any individual, including an advisor, who is providing bona fide services to the Company or any Subsidiary or who has accepted an offer of service or consultancy from the Company or any Subsidiary; provided that any such person may not receive any payment or exercise any right relating to an Award until such person has commenced service with the Company or its Subsidiaries or Affiliates. For purposes of the Plan, in the case of a Consultant, references to employment shall be deemed to refer to such Consultants service in such capacity, but in no event shall the Plan or any action taken hereunder be construed to create an employer-employee relationship between any such Consultant and the Company or of any of its Affiliates.
(l) Director means any member of the Board.
(m) Effective Date means the date on which the Plan is adopted by the Board and approved by the shareholders of the Company.
(n) Employee means any individual, including any officer, employed by the Company or any Subsidiary or Affiliate or any prospective employee or officer who has accepted an offer of employment from the Company or any Subsidiary or Affiliate, with the status of employment determined based upon such factors as are deemed appropriate by the Committee in its discretion, subject to any requirements of the Code or applicable laws; provided that any such person may not receive any payment or exercise any right relating to an Award until such person has commenced employment or service with the Company or its Subsidiaries or Affiliates. An employee on an approved leave of absence (including maternity leave) shall be considered as still in the employment of the Company or its Subsidiaries or Affiliates for purposes of eligibility for participation in the Plan.
(o) Exchange Act means the Securities Exchange Act of 1934, as amended from time to time, and the rules, regulations and guidance thereunder. Any reference to a provision in the Exchange Act shall include any successor provision thereto.
(p) Fair Market Value means (i) with respect to Shares, unless otherwise determined by the Committee, the closing price of a Share on the trading day immediately preceding the date of determination (or, if there is no reported sale on such date, on the last preceding date on which any reported sale occurred), on the principal stock market or exchange on which the Shares are quoted or traded, or if Shares are not so quoted or traded, the fair market value of a Share as determined by the Committee, and (ii) with respect to
I-3
Table of Contents
any property other than Shares, the fair market value of such property determined by such methods or procedures as shall be established from time to time by the Committee.
(q) Incentive Stock Option means an option representing the right to purchase Shares from the Company, granted pursuant to Section 6, that meets the requirements of Section 422 of the Code.
(r) Intrinsic Value with respect to an Option or SAR Award means (i) the excess, if any, of the price or implied price per Share in a Change in Control or other event over (ii) the exercise or hurdle price of such Award multiplied by (iii) the number of Shares covered by such Award.
(s) Non-Employee Director means a Director who either (i) is not a current employee or officer of the Company or an Affiliate, does not receive compensation, either directly or indirectly, from the Company or an Affiliate for services rendered as a consultant or in any capacity other than as a Director (except for an amount as to which disclosure would not be required under Item 404(a) of Regulation S-K promulgated pursuant to the Securities Act (Regulation S-K)), does not possess an interest in any other transaction for which disclosure would be required under Item 404(a) of Regulation S-K, and is not engaged in a business relationship for which disclosure would be required pursuant to Item 404(b) of Regulation S-K; or (ii) is otherwise considered a non-employee director for purposes of Rule 16b-3.
(t) Non-Qualified Stock Option means an option representing the right to purchase Shares from the Company, granted pursuant to Section 6, that is not an Incentive Stock Option.
(u) Option means an Incentive Stock Option or a Non-Qualified Stock Option.
(v) Other Cash-Based Award means an Award granted pursuant to Section 11, including cash awarded as a bonus or upon the attainment of specified performance criteria or otherwise as permitted under the Plan.
(w) Other Stock-Based Award means an Award granted pursuant to Section 11 that may be denominated or payable in, valued in whole or in part by reference to, or otherwise based on, or related to, Shares or factors that may influence the value of Shares, including convertible or exchangeable debt securities, other rights convertible or exchangeable into Shares, purchase rights for Shares, dividend rights or dividend equivalent rights or Awards with value and payment contingent upon performance of the Company or business units thereof or any other factors designated by the Committee.
(x) Participant means the recipient of an Award granted under the Plan.
(y) Performance Award means an Award granted pursuant to Section 10.
(z) Performance Period means the period established by the Committee with respect to any Performance Award during which the performance goals specified by the Committee with respect to such Award are to be measured.
(aa) Person has the meaning ascribed to such term in Section 3(a)(9) of the Exchange Act and used in Sections 13(d) and 14(d) thereof, including a group as defined in Section 13(d) thereof.
(bb) Restricted Stock means any Share subject to certain restrictions and forfeiture conditions, granted pursuant to Section 8.
(cc) Rule 16b-3 means Rule 16b-3 promulgated under the Exchange Act or any successor to Rule 16b-3, as in effect from time to time.
(dd) RSU means a contractual right granted pursuant to Section 9 that is denominated in Shares. Each RSU represents a right to receive the value of one Share (or a percentage of such value) in cash, Shares or a combination thereof. Awards of RSUs may include the right to receive dividend equivalents.
(ee) SAR means a right granted pursuant to Section 7 to receive upon exercise by the Participant or settlement, in cash, Shares or a combination thereof, the excess of (i) the Fair Market Value of one Share on the date of exercise or settlement over (ii) the exercise or hurdle price of the right on the date of grant.
I-4
Table of Contents
(ff) Service Agreement means any employment, severance, consulting or similar agreement between the Company or any of its Affiliates and a Participant.
(gg) Share means a share of the Companys Class A common stock, $0.0001 par value.
(hh) Subsidiary means an entity of which the Company directly or indirectly holds all or a majority of the value of the outstanding equity interests of such entity or a majority of the voting power with respect to the voting securities of such entity. Whether employment by or service with a Subsidiary is included within the scope of the Plan shall be determined by the Committee.
(ii) Substitute Award means an Award granted in assumption of, or in substitution for, an outstanding award previously granted by a company or other business acquired by the Company or with which the Company combines.
(jj) Termination of Service means, in the case of a Participant who is an Employee, cessation of the employment relationship such that the Participant is no longer an employee of the Company or any Subsidiary, or, in the case of a Participant who is a Consultant or Non-Employee Director, the date the performance of services for the Company or any Subsidiary has ended; provided, however, that in the case of a Participant who is an Employee, the transfer of employment from the Company to a Subsidiary, from a Subsidiary to the Company, from one Subsidiary to another Subsidiary or, unless the Committee determines otherwise, the cessation of employee status but the continuation of the performance of services for the Company or a Subsidiary as a Director or Consultant shall not be deemed a cessation of service that would constitute a Termination of Service; provided, further, that a Termination of Service shall be deemed to occur for a Participant employed by, or performing services for, a Subsidiary when such Subsidiary ceases to be a Subsidiary unless such Participants employment or service continues with the Company or another Subsidiary. Notwithstanding the foregoing, with respect to any Award subject to Section 409A of the Code (and not exempt therefrom), a Termination of Service occurs when a Participant experiences a separation of service (as such term is defined under Section 409A of the Code).
Section 3. Eligibility.
(a) Any Employee, Non-Employee Director or Consultant shall be eligible to be selected to receive an Award under the Plan, to the extent that an offer or receipt of an Award is permitted by applicable law, stock market or exchange rules and regulations or accounting or tax rules and regulations.
(b) Holders of equity compensation awards granted by a company that is acquired by the Company (or whose business is acquired by the Company) or with which the Company combines are eligible for grants of Substitute Awards under the Plan to the extent permitted under applicable regulations of any stock exchange on which the Company is listed.
Section 4. Administration.
(a) Administration of the Plan. The Plan shall be administered by the Committee. All decisions of the Committee shall be final, conclusive and binding upon all parties, including the Company, its shareholders, Participants and any Beneficiaries thereof. The Committee may issue rules and regulations for administration of the Plan.
(b) Delegation of Authority. To the extent permitted by applicable law, including under Section 157(c) of the Delaware General Corporation Law, the Committee may delegate to one or more officers of the Company some or all of its authority under the Plan, including the authority to grant Options and SARs or other Awards in the form of Share rights (except that such delegation shall not apply to any Award for a Person then covered by Section 16 of the Exchange Act), and the Committee may delegate to one or more committees of the Board (which may consist of solely one Director) some or all of its authority under the Plan, including the authority to grant all types of Awards, in accordance with applicable law.
(c) Authority of Committee. Subject to the terms of the Plan and applicable law, the Committee (or its delegate) shall have full discretion and authority to: (i) designate Participants; (ii) determine the type or
I-5
Table of Contents
types of Awards (including Substitute Awards) to be granted to each Participant under the Plan; (iii) determine the number of Shares to be covered by (or with respect to which payments, rights or other matters are to be calculated in connection with) Awards; (iv) determine the terms and conditions of any Award and prescribe the form of each Award Agreement, which need not be identical for each Participant; (v) determine whether, to what extent, under what circumstances and by which methods Awards may be settled or exercised in cash, Shares, other Awards, other property, net settlement (including broker-assisted cashless exercise), or any combination thereof, or canceled, forfeited or suspended; (vi) determine whether, to what extent and under what circumstances cash, Shares, other Awards, other property and other amounts payable with respect to an Award under the Plan shall be deferred either automatically or at the election of the holder thereof or of the Committee; (vii) amend terms or conditions of any outstanding Awards; (viii) correct any defect, supply any omission and reconcile any inconsistency in the Plan or any Award, in the manner and to the extent it shall deem desirable to carry the Plan into effect; (ix) interpret and administer the Plan and any instrument or agreement relating to, or Award made under, the Plan; (x) establish, amend, suspend or waive such rules and regulations and appoint such agents, trustees, brokers, depositories and advisors and determine such terms of their engagement as it shall deem appropriate for the proper administration of the Plan and due compliance with applicable law, stock market or exchange rules and regulations or accounting or tax rules and regulations; and (xi) make any other determination and take any other action that the Committee deems necessary or desirable for the administration of the Plan and due compliance with applicable law, stock market or exchange rules and regulations or accounting or tax rules and regulations. Notwithstanding anything to the contrary contained herein, the Board may, in its sole discretion, at any time and from time to time, grant Awards or administer the Plan. In any such case, the Board shall have all of the authority and responsibility granted to the Committee herein.
(d) Rule 16b-3 Compliance. To the extent an Award is intended to qualify for the exemption from Section 16(b) of the Exchange Act that is available under Rule 16b-3 of the Exchange Act, the Award will be granted by the Board or a Committee (or a subcommittee thereof) that consists solely of two or more Non-Employee Directors, as determined under Rule 16b-3(b)(3) of the Exchange Act and thereafter any action establishing or modifying the terms of the Award will be approved by the Board or a Committee (or a subcommittee) meeting such requirements to the extent necessary for such exemption to remain available.
Section 5. Shares Available for Awards.
(a) Subject to adjustment as provided in Section 5(c) and except for Substitute Awards, the maximum number of Shares available for issuance under the Plan shall not exceed in the aggregate [] Shares.1 The total number of Shares available for issuance under the Plan shall be increased on the first day of each Company fiscal year following the Effective Date in an amount equal to the lesser of (i) three percent (3%) of outstanding Shares on the last day of the immediately preceding fiscal year, (ii) []2 Shares and (iii) such number of Shares as determined by the Committee in its discretion. Shares underlying Substitute Awards and Shares remaining available for grant under a plan of an acquired company or of a company with which the Company combines (whether by way of amalgamation, merger, sale and purchase of shares or other securities or otherwise), appropriately adjusted to reflect the acquisition or combination transaction, shall not reduce the number of Shares remaining available for grant hereunder.
(b) If any Award is forfeited, cancelled, expires, terminates or otherwise lapses or is settled in cash, in whole or in part, without the delivery of Shares, then the Shares covered by such forfeited, expired, terminated or lapsed Award shall again be available for grant under the Plan. The following shall become available for issuance under the Plan: (i) any Shares withheld in respect of taxes relating to any Award and (ii) any Shares tendered or withheld to pay the exercise price of Options.
(c) In the event that the Committee determines that, as a result of any dividend or other distribution (other than an ordinary dividend or distribution), recapitalization, stock split, reverse stock split,
| 1 | To be 10% of Shares outstanding immediately following the closing of the business combination. |
| 2 | To be 5% of Shares outstanding immediately following the closing of the business combination. |
I-6
Table of Contents
reorganization, merger, amalgamation, consolidation, separation, rights offering, split-up, spin-off, combination, repurchase or exchange of Shares or other securities of the Company, issuance of warrants or other rights to acquire Shares or other securities of the Company, issuance of Shares pursuant to the anti-dilution provisions of securities of the Company, or other similar corporate transaction or event affecting the Shares, or of changes in applicable laws, regulations or accounting principles, an adjustment is necessary in order to prevent dilution or enlargement of the benefits or potential benefits intended to be made available under the Plan, then the Committee shall, subject to Section 19 and applicable law, adjust equitably so as to ensure no undue enrichment or harm (including by payment of cash), any or all of:
(i) the number and type of Shares (or other securities) which thereafter may be made the subject of Awards, including the aggregate limits specified in Section 5(a) and Section 5(f);
(ii) the number and type of Shares (or other securities) subject to outstanding Awards;
(iii) the grant, acquisition, exercise or hurdle price with respect to any Award or, if deemed appropriate, make provision for a cash payment to the holder of an outstanding Award; and
(iv) the terms and conditions of any outstanding Awards, including the performance criteria of any Performance Awards;
provided, however, that the number of Shares subject to any Award denominated in Shares shall always be a whole number.
(d) Any Shares delivered pursuant to an Award may consist, in whole or in part, of authorized and unissued Shares or Shares acquired by the Company.
(e) The aggregate value of all compensation granted or paid, as applicable, to any individual for service as a Non-Employee Director with respect to any calendar year, including Awards granted and cash fees paid by the Company to such Non-Employee Director, will not exceed (i) $500,000 in total value or (ii) in the event such Non-Employee Director is first appointed or elected to the Board, $750,000 in total value during the initial annual period, in each case calculating the value of any equity awards based on the grant date fair value of such equity awards for financial reporting purposes. The limitations in this Section 5(e) shall apply commencing with the first calendar year that begins following the Effective Date.
(f) Subject to adjustment as provided in Section 5(c)(i), the maximum number of Shares available for issuance with respect to Incentive Stock Options shall be []3. To the extent that the aggregate Fair Market Value (determined at the time of grant) of Shares with respect to which Incentive Stock Options are exercisable for the first time by any Optionholder during any calendar year (under all plans of the Company and any Affiliates) exceeds $100,000 (or such other limit established in the Code) or otherwise does not comply with the rules governing Incentive Stock Options, the Options or portions thereof that exceed such limit (according to the order in which they were granted) or otherwise do not comply with such rules will be treated as Nonqualified Stock Options, notwithstanding any contrary provision of the applicable Option Agreement(s).
Section 6. Options. The Committee is authorized to grant Options to Participants with the following terms and conditions and with such additional terms and conditions, in either case not inconsistent with the provisions of the Plan, as the Committee shall determine:
(a) The exercise price per Share under an Option shall be determined by the Committee at the time of grant; provided, however, that, except in the case of Substitute Awards, such exercise price shall not be less than the Fair Market Value of a Share on the date of grant of such Option.
(b) The term of each Option shall be fixed by the Committee but shall not exceed 10 years from the date of grant of such Option. Subject to Section 13, the Committee shall determine the time or times at which an Option becomes vested and exercisable in whole or in part.
| 3 | To be same number as initial share reserve. |
I-7
Table of Contents
(c) The Committee shall determine the methods by which, and the forms in which payment of the exercise price with respect thereto may be made or deemed to have been made, including cash, Shares, other Awards, other property, net settlement (including broker-assisted cashless exercise) or any combination thereof, having a Fair Market Value on the exercise date equal to the relevant exercise price.
(d) To the extent an Option is not previously exercised as to all of the Shares subject thereto, and, if the Fair Market Value of one Share is greater than the exercise price then in effect, then the Option shall be deemed automatically exercised immediately before its expiration.
(e) No grant of Options may be accompanied by a tandem award of dividend equivalents or provide for dividends, dividend equivalents or other distributions to be paid on such Options (except as provided under Section 5(c)).
(f) The terms of any Incentive Stock Option granted under the Plan shall comply in all respects with the provisions of Section 422 of the Code. Incentive Stock Options may be granted only to employees of the Company or of a parent or subsidiary corporation (as defined in Section 424 of the Code).
Section 7. Stock Appreciation Rights. The Committee is authorized to grant SARs to Participants with the following terms and conditions and with such additional terms and conditions, in either case not inconsistent with the provisions of the Plan, as the Committee shall determine:
(a) SARs may be granted under the Plan to Participants either alone (freestanding) or in addition to other Awards granted under the Plan (tandem) and may, but need not, relate to a specific Option granted under Section 6.
(b) The exercise or hurdle price per Share under a SAR shall be determined by the Committee; provided, however, that, except in the case of Substitute Awards, such exercise or hurdle price shall not be less than the Fair Market Value of a Share on the date of grant of such SAR.
(c) The term of each SAR shall be fixed by the Committee but shall not exceed 10 years from the date of grant of such SAR. Subject to Section 13, the Committee shall determine the time or times at which a SAR may be exercised or settled in whole or in part.
(d) Upon the exercise of a SAR, the Company shall pay to the Participant an amount equal to the number of Shares subject to the SAR multiplied by the excess, if any, of the Fair Market Value of one Share on the exercise date over the exercise or hurdle price of such SAR. The Company shall pay such excess in cash, in Shares valued at Fair Market Value, or any combination thereof, as determined by the Committee.
(e) To the extent a SAR is not previously exercised as to all of the Shares subject thereto, and, if the Fair Market Value of one Share is greater than the exercise price then in effect, then the SAR shall be deemed automatically exercised immediately before its expiration.
(f) No grant of SARs may be accompanied by a tandem award of dividend equivalents or provide for dividends, dividend equivalents or other distributions to be paid on such SARs (except as provided under Section 5(c)).
Section 8. Restricted Stock. The Committee is authorized to grant Awards of Restricted Stock to Participants with the following terms and conditions and with such additional terms and conditions, in either case not inconsistent with the provisions of the Plan, as the Committee shall determine:
(a) The Award Agreement shall specify the vesting schedule.
(b) Awards of Restricted Stock shall be subject to such restrictions as the Committee may impose, which restrictions may lapse separately or in combination at such time or times, in such installments or otherwise, as the Committee may deem appropriate.
(c) Subject to the restrictions set forth in the applicable Award Agreement, a Participant generally shall have the rights and privileges of a shareholder with respect to Awards of Restricted Stock, including the right to vote such Shares of Restricted Stock and the right to receive dividends.
I-8
Table of Contents
(d) The Committee may, in its discretion, specify in the applicable Award Agreement that any or all dividends or other distributions paid on Awards of Restricted Stock prior to vesting be paid either in cash or in additional Shares and either on a current or deferred basis and that such dividends or other distributions may be reinvested in additional Shares, which may be subject to the same restrictions as the underlying Awards.
(e) Any Award of Restricted Stock may be evidenced in such manner as the Committee may deem appropriate, including book-entry registration.
(f) The Committee may provide in an Award Agreement that an Award of Restricted Stock is conditioned upon the Participant making or refraining from making an election with respect to the Award under Section 83(b) of the Code. If a Participant makes an election pursuant to Section 83(b) of the Code with respect to an Award of Restricted Stock, such Participant shall be required to file promptly a copy of such election with the Company and the applicable Internal Revenue Service office.
Section 9. RSUs. The Committee is authorized to grant Awards of RSUs to Participants with the following terms and conditions and with such additional terms and conditions, in either case not inconsistent with the provisions of the Plan, as the Committee shall determine:
(a) The Award Agreement shall specify the vesting schedule and the delivery schedule (which may include deferred delivery later than the vesting date).
(b) Awards of RSUs shall be subject to such restrictions as the Committee may impose, which restrictions may lapse separately or in combination at such time or times, in such installments or otherwise, as the Committee may deem appropriate.
(c) An RSU shall not convey to a Participant the rights and privileges of a shareholder with respect to the Share subject to such RSU, such as the right to vote or the right to receive dividends, unless and until and to the extent a Share is issued to such Participant to settle such RSU.
(d) The Committee may, in its discretion, specify in the applicable Award Agreement that any or all dividend equivalents or other distributions paid on Awards of RSUs prior to vesting or settlement, as applicable, be paid either in cash or in additional Shares and either on a current or deferred basis and that such dividend equivalents or other distributions may be reinvested in additional Shares, which may be subject to the same restrictions as such Awards.
(e) Shares delivered upon the vesting and settlement of an RSU Award may be evidenced in such manner as the Committee may deem appropriate, including book-entry registration.
(f) The Committee may determine the form or forms (including cash, Shares, other Awards, other property or any combination thereof) in which payment of the amount owing upon settlement of any RSU Award may be made.
Section 10. Performance Awards. The Committee is authorized to grant Performance Awards to Participants with the following terms and conditions and with such additional terms and conditions, in either case not inconsistent with the provisions of the Plan, as the Committee shall determine:
(a) Performance Awards may be denominated as a cash amount, number of Shares or units or a combination thereof and are Awards that may be earned upon achievement or satisfaction of performance conditions specified by the Committee. In addition, the Committee may specify that any other Award shall constitute a Performance Award by conditioning the grant to a Participant or the right of a Participant to exercise the Award or have it settled, and the timing thereof, upon achievement or satisfaction of such performance conditions as may be specified by the Committee. The Committee may use such business criteria and other measures of performance as it may deem appropriate in establishing any performance conditions. Subject to the terms of the Plan, the performance goals to be achieved during any Performance Period, the length of any Performance Period, the amount of any Performance Award granted and the
I-9
Table of Contents
amount of any payment or transfer to be made pursuant to any Performance Award shall be determined by the Committee.
(b) Performance criteria may be measured on an absolute (e.g., plan or budget) or relative basis, and may be established on a corporate-wide basis, with respect to one or more business units, divisions, Subsidiaries or business segments, or on an individual basis. If the Committee determines that a change in the business, operations, corporate structure or capital structure of the Company, or the manner in which the Company conducts its business, or other events or circumstances render the performance objectives unsuitable, the Committee may modify the performance objectives or the related minimum acceptable level of achievement, in whole or in part, as the Committee deems appropriate and equitable such that it does not provide any undue enrichment or harm. Performance measures may vary from Performance Award to Performance Award and from Participant to Participant, and may be established on a stand-alone basis, in tandem or in the alternative. The Committee shall have the power to impose such other restrictions on Awards subject to this Section 10(b) as it may deem necessary or appropriate to ensure that such Awards satisfy all requirements of any applicable law, stock market or exchange rules and regulations or accounting or tax rules and regulations.
(c) Settlement of Performance Awards shall be in cash, Shares, other Awards, other property, net settlement, or any combination thereof, as determined in the discretion of the Committee.
(d) A Performance Award shall not convey to a Participant the rights and privileges of a shareholder with respect to the Shares subject to such Performance Award, such as the right to vote (except as relates to Restricted Stock) or the right to receive dividends, unless and until and to the extent a Share is issued to such Participant to settle such Performance Award. The Committee, in its sole discretion, may provide that a Performance Award shall convey the right to receive dividend equivalents on the Shares subject to such Performance Award with respect to any dividends declared during the period that such Performance Award is outstanding, in which case, such dividend equivalent rights shall accumulate and shall be paid in cash or Shares on the settlement date of the Performance Award, subject to the Participants earning of the Shares with respect to which such dividend equivalents are paid upon achievement or satisfaction of performance conditions specified by the Committee. Shares delivered upon the vesting and settlement of a Performance Award may be evidenced in such manner as the Committee may deem appropriate, including book-entry registration. For the avoidance of doubt, unless otherwise determined by the Committee, no dividend equivalent rights shall be provided with respect to any Shares subject to Performance Awards that are not earned or otherwise do not vest or settle pursuant to their terms.
(e) The Committee may, in its discretion, increase or reduce the amount of a settlement otherwise to be made in connection with a Performance Award.
Section 11. Other Cash-Based Awards and Other Stock-Based Awards. The Committee is authorized, subject to limitations under applicable law, to grant Other Cash-Based Awards (either independently or as an element of or supplement to any other Award under the Plan) and Other Stock-Based Awards. The Committee shall determine the terms and conditions of such Awards. Shares delivered pursuant to an Award in the nature of a purchase right granted under this Section 11 shall be purchased for such consideration, and paid for at such times, by such methods and in such forms, including cash, Shares, other Awards, other property, net settlement, broker-assisted cashless exercise or any combination thereof, as the Committee shall determine; provided that the purchase price therefor shall not be less than the Fair Market Value of such Shares on the date of grant of such right.
Section 12. Effect of Termination of Service or a Change in Control on Awards.
(a) The Committee may provide, by rule or regulation or in any applicable Award Agreement, or may determine in any individual case, the circumstances in which, and the extent to which, an Award may be exercised, settled, vested, paid or forfeited in the event of a Participants Termination of Service prior to the end of a Performance Period or vesting, exercise or settlement of such Award.
I-10
Table of Contents
(b) Subject to the last sentence of Section 2(jj), the Committee may determine, in its discretion, whether, and the extent to which, (i) an Award will vest during a leave of absence, (ii) a reduction in service level (for example, from full-time to part-time employment) will cause a reduction, or other change, to an Award and (iii) a leave of absence or reduction in service will be deemed a Termination of Service.
(c) In the event of a Change in Control, the Committee may, in its sole discretion, and on such terms and conditions as it deems appropriate, take any one or more of the following actions with respect to any outstanding Award, which need not be uniform with respect to all Participants and/or Awards:
(i) continuation or assumption of such Award by the Company (if it is the surviving corporation) or by the successor or surviving entity or its parent;
(ii) substitution or replacement of such Award by the successor or surviving entity or its parent with cash, securities, rights or other property to be paid or issued, as the case may be, by the successor or surviving entity (or a parent or subsidiary thereof), with substantially the same terms and value as such Award (including any applicable performance targets or criteria with respect thereto);
(iii) acceleration of the vesting of such Award and the lapse of any restrictions thereon and, in the case of an Option or SAR Award, acceleration of the right to exercise such Award during a specified period (and the termination of such Option or SAR Award without payment of any consideration therefor to the extent such Award is not timely exercised), in each case, either (A) immediately prior to or as of the date of the Change in Control, (B) upon a Participants involuntary Termination of Service (including upon a termination of the Participants employment by the Company (or a successor corporation or its parent) without Cause, by a Participant for good reason and/or due to a Participants death or disability, as such terms may be defined in the applicable Award Agreement and/or a Participants Service Agreement, as the case may be) on or within a specified period following the Change in Control or (C) upon the failure of the successor or surviving entity (or its parent) to continue or assume such Award;
(iv) in the case of a Performance Award, determination of the level of attainment of the applicable performance condition(s); and
(v) cancellation of such Award in consideration of a payment, with the form, amount and timing of such payment determined by the Committee in its sole discretion, subject to the following: (A) such payment shall be made in cash, securities, rights and/or other property; (B) the amount of such payment shall equal the value of such Award, as determined by the Committee in its sole discretion; provided that, in the case of an Option or SAR Award, if such value equals the Intrinsic Value of such Award, such value shall be deemed to be valid; provided further that, if the Intrinsic Value of an Option or SAR Award is equal to or less than zero, the Committee may, in its sole discretion, provide for the cancellation of such Award without payment of any consideration therefor (for the avoidance of doubt, in the event of a Change in Control, the Committee may, in its sole discretion, terminate any Option or SAR Awards for which the exercise or hurdle price is equal to or exceeds the per Share value of the consideration to be paid in the Change in Control transaction without payment of consideration therefor); and (C) such payment shall be made promptly following such Change in Control or on a specified date or dates following such Change in Control; provided that the timing of such payment shall comply with Section 409A of the Code.
Section 13. General Provisions Applicable to Awards.
(a) Awards shall be granted for such cash or other consideration, if any, as the Committee determines; provided that in no event shall Awards be issued for less than such minimal consideration as may be required by applicable law.
(b) Awards may, in the discretion of the Committee, be granted either alone or in addition to or in tandem with any other Award or any award granted under any other plan of the Company. Awards granted
I-11
Table of Contents
in addition to or in tandem with other Awards, or in addition to or in tandem with awards granted under any other plan of the Company, may be granted either at the same time as or at a different time from the grant of such other Awards or awards.
(c) Subject to the terms of the Plan, payments or transfers to be made by the Company upon the grant, exercise or settlement of an Award may be made in the form of cash, Shares, other Awards, other property, net settlement, or any combination thereof, as determined by the Committee in its discretion at the time of grant, and may be made in a single payment or transfer, in installments or on a deferred basis, in each case in accordance with rules and procedures established by the Committee. Such rules and procedures may include provisions for the payment or crediting of reasonable interest on installment or deferred payments or the grant or crediting of dividend equivalents in respect of installment or deferred payments.
(d) Except as may be permitted by the Committee or as specifically provided in an Award Agreement, (i) no Award and no right under any Award shall be assignable, alienable, saleable or transferable by a Participant other than by will or pursuant to Section 13(e) and (ii) during a Participants lifetime, each Award, and each right under any Award, shall be exercisable only by such Participant or, if permissible under applicable law, by such Participants guardian or legal representative. The provisions of this Section 13(d) shall not apply to any Award that has been fully exercised or settled, as the case may be, and shall not preclude forfeiture of an Award in accordance with the terms thereof.
(e) A Participant may designate a Beneficiary or change a previous Beneficiary designation only at such times as prescribed by the Committee, in its sole discretion, and only by using forms and following procedures approved or accepted by the Committee for that purpose.
(f) All certificates, if any, for Shares and/or other securities delivered under the Plan pursuant to any Award or the exercise or settlement thereof shall be subject to such stop transfer orders and other restrictions as the Committee may deem advisable under the Plan or the rules, regulations and other requirements of the Securities and Exchange Commission, any stock market or exchange upon which such Shares or other securities are then quoted, traded or listed, and any applicable securities laws, and the Committee may cause a legend or legends to be put on any such certificates to make appropriate reference to such restrictions.
(g) The Company will not be obligated to deliver any Shares under the Plan or remove restrictions from Shares previously delivered under the Plan until (i) all Award conditions have been met or removed to the Committees satisfaction, (ii) as determined by the Committee, all other legal matters regarding the issuance and delivery of such Shares have been satisfied, including any applicable securities laws, stock market or exchange rules and regulations or accounting or tax rules and regulations and (iii) the Participant has executed and delivered to the Company such representations or agreements as the Committee deems necessary or appropriate to satisfy any applicable laws. The Companys inability to obtain authority from any regulatory body having jurisdiction, which the Committee determines is necessary to the lawful issuance and sale of any Shares, will relieve the Company of any liability for failing to issue or sell such Shares as to which such requisite authority has not been obtained.
(h) The Committee may impose restrictions on any Award with respect to non-competition, non-solicitation, confidentiality and other restrictive covenants, or requirements to comply with minimum share ownership requirements, as it deems necessary or appropriate in its sole discretion, which such restrictions may be set forth in any applicable Award Agreement or otherwise.
Section 14. Amendments and Terminations.
(a) Amendment or Termination of the Plan. Except to the extent prohibited by applicable law and unless otherwise expressly provided in an Award Agreement or in the Plan, the Board may amend, alter, suspend, discontinue or terminate the Plan or any portion thereof at any time; provided, however, that no such amendment, alteration, suspension, discontinuation or termination shall be made without (i) shareholder approval if such approval is required by applicable law or the rules of the stock market or
I-12
Table of Contents
exchange, if any, on which the Shares are principally quoted or traded or (ii) subject to Section 5(c) and Section 12, the consent of the affected Participant, if such action would materially adversely affect the rights of such Participant under any outstanding Award, except (x) to the extent any such amendment, alteration, suspension, discontinuance or termination is made to cause the Plan to comply with applicable law, stock market or exchange rules and regulations or accounting or tax rules and regulations or (y) to impose any clawback or recoupment provisions on any Awards (including any amounts or benefits arising from such Awards) in accordance with Section 18. Notwithstanding anything to the contrary in the Plan, the Committee may amend the Plan, or create sub-plans, in such manner as may be necessary or desirable to enable the Plan to achieve its stated purposes in any jurisdiction in a tax-efficient manner and in compliance with local rules and regulations.
(b) Dissolution or Liquidation. In the event of the dissolution or liquidation of the Company, each Award shall terminate immediately prior to the consummation of such action, unless otherwise determined by the Committee.
(c) Terms of Awards. The Committee may waive any conditions or rights under, amend any terms of, or amend, alter, suspend, discontinue or terminate any Award theretofore granted (including by substituting another Award of the same or a different type), prospectively or retroactively, without the consent of any relevant Participant or holder or Beneficiary of an Award; provided, however, that, subject to Section 5(c) and Section 12, no such action shall materially adversely affect the rights of any affected Participant or holder or Beneficiary under any Award theretofore granted under the Plan, except (x) to the extent any such action is made to cause the Plan or Award to comply with applicable law, stock market or exchange rules and regulations or accounting or tax rules and regulations, or (y) to impose any clawback or recoupment provisions on any Awards (including any amounts or benefits arising from such Awards) in accordance with Section 18. The Committee shall be authorized to make adjustments in the terms and conditions of, and the criteria included in, Awards in recognition of events (including the events described in Section 5(c)) affecting the Company, or the financial statements of the Company, or of changes in applicable laws, regulations or accounting principles, whenever the Committee determines that such adjustments are appropriate in order to prevent dilution or enlargement of the benefits or potential benefits intended to be made available under the Plan.
(d) No Repricing. Except as provided in Section 5(c), the Committee may not, without shareholder approval, seek to effect any re-pricing of any previously granted underwater Option, SAR or similar Award by: (i) amending or modifying the terms of the Option, SAR or similar Award to lower the exercise price; (ii) cancelling the underwater Option, SAR or similar Award and granting either (A) replacement Options, SARs or similar Awards having a lower exercise price or (B) Restricted Shares, RSUs, Performance Awards or Other Share-Based Awards in exchange; or (iii) cancelling or repurchasing the underwater Options, SARs or similar Awards for cash or other securities. An Option, SAR or similar Award will be deemed to be underwater at any time when the Fair Market Value of the Shares covered by such Award is less than the exercise price of the Award.
Section 15. Miscellaneous.
(a) No Employee, Consultant, Non-Employee Director, Participant, or other Person shall have any claim to be granted any Award under the Plan, and there is no obligation for uniformity of treatment of employees, Participants or holders or Beneficiaries of Awards under the Plan. The terms and conditions of Awards need not be the same with respect to each recipient. Any Award granted under the Plan shall be a one-time Award that does not constitute a promise of future grants. The Company, in its sole discretion, maintains the right to make available future grants under the Plan.
(b) The grant of an Award shall not be construed as giving a Participant the right to be retained in the employ of, or to continue to provide services to, the Company or any Affiliate. Further, the Company or any applicable Affiliate may at any time dismiss a Participant, free from any liability, or any claim under the Plan, unless otherwise expressly provided in the Plan or in any Award Agreement or in any other agreement
I-13
Table of Contents
binding on the parties. The receipt of any Award under the Plan is not intended to confer any rights on the receiving Participant except as set forth in the applicable Award Agreement.
(c) In the event a Participants regular level of time commitment in the performance of his or her services for the Company and any Affiliates is reduced (for example, and without limitation, if the Participant is an employee of the Company and the Employee has a change in status from a full-time employee to a part-time employee (or serves as a Consultant or Director) or takes an extended leave of absence) after the date of grant of any Award to the Participant, the Board may determine, to the extent permitted by applicable law, to (i) make a corresponding reduction in the number of shares or cash amount subject to any portion of such Award that is scheduled to vest or become payable after the date of such change in time commitment, and (ii) in lieu of or in combination with such a reduction, extend the vesting or payment schedule applicable to such Award. In the event of any such reduction, the Participant will have no right with respect to any portion of the Award that is so reduced or extended.
(d) As a condition to accepting an Award under the Plan, the Participant agrees to execute any additional documents or instruments necessary or desirable, as determined in the Committees sole discretion, to carry out the purposes or intent of the Award, or facilitate compliance with securities and/or other regulatory requirements, in each case at the Committees request.
(e) No payment pursuant to the Plan shall be taken into account in determining any benefits under any severance, pension, retirement, savings, profit sharing, group insurance, welfare or other benefit plan of the Company or any Affiliate, except to the extent otherwise expressly provided in writing in such other plan or an agreement thereunder.
(f) Nothing contained in the Plan shall prevent the Company or any Affiliate from adopting or continuing in effect other or additional compensation arrangements, including the grant of options and other stock-based awards, and such arrangements may be either generally applicable or applicable only in specific cases.
(e) The Company shall be authorized to withhold from any Award granted or any payment due or transfer made under any Award or under the Plan or from any compensation or other amount owing to a Participant the amount (in cash, Shares, other Awards, other property, net settlement, or any combination thereof) of applicable withholding taxes due in respect of an Award, its grant, vesting, exercise or settlement or any payment or transfer under such Award or under the Plan and to take such other action (including providing for elective payment of such amounts in cash or Shares by such Participant) as may be necessary to satisfy all obligations for the payment of such taxes and, unless otherwise determined by the Committee in its discretion, to the extent such withholding would not result in liability classification of such Award (or any portion thereof) pursuant to FASB ASC Subtopic 718-10. As a condition to accepting an Award under the Plan, in the event that the amount of the Companys and/or its Affiliates withholding obligation in connection with such Award was greater than the amount actually withheld by the Company and/or its Affiliates, each Participant agrees to indemnify and hold the Company and/or its Affiliates harmless from any failure by the Company and/or its Affiliates to withhold the proper amount.
(g) If any provision of the Plan or any Award Agreement is or becomes or is deemed to be invalid, illegal or unenforceable in any jurisdiction, or as to any Person or Award, or would disqualify the Plan or any Award under any law deemed applicable by the Committee, such provision shall be construed or deemed amended to conform to applicable laws, or if it cannot be so construed or deemed amended without, in the determination of the Committee, materially altering the intent of the Plan or the Award Agreement, such provision shall be stricken as to such jurisdiction, Person or Award, and the remainder of the Plan and any such Award Agreement shall remain in full force and effect.
(h) Neither the Plan nor any Award shall create or be construed to create a trust or separate fund of any kind or a fiduciary relationship between the Company and a Participant or any other Person. To the extent that any Person acquires a right to receive payments from the Company pursuant to an Award, such right shall be no greater than the right of any unsecured general creditor of the Company.
I-14
Table of Contents
(i) Any reference herein or in an Award Agreement to a written agreement or document will include any agreement or document delivered electronically, filed publicly at www.sec.gov (or any successor website thereto) or posted on the Companys intranet (or other shared electronic medium controlled by the Company to which the Participant has access). By accepting any Award the Participant consents to receive documents by electronic delivery and to participate in the Plan through any on-line electronic system established and maintained by the Committees or another third party selected by the Committee. The form of delivery of any Shares (e.g., a stock certificate or electronic entry evidencing such shares) shall be determined by the Company.
(j) No fractional Shares shall be issued or delivered pursuant to the Plan or any Award, and the Committee shall determine whether cash or other securities shall be paid or transferred in lieu of any fractional Shares, or whether such fractional Shares or any rights thereto shall be canceled, terminated or otherwise eliminated.
(k) Awards may be granted to Participants who are non-United States nationals or employed or providing services outside the United States, or both, on such terms and conditions different from those applicable to Awards to Participants who are employed or providing services in the United States as may, in the judgment of the Committee, be necessary or desirable to recognize differences in local law, tax policy or custom. The Committee also may impose conditions on the exercise or vesting of Awards in order to minimize the Companys obligation with respect to tax equalization for Participants on assignments outside their home country.
Section 16. Effective Date of the Plan. The Plan shall be effective as of the Effective Date.
Section 17. Term of the Plan. No Award shall be granted under the Plan after the earliest to occur of (i) the 10-year anniversary of the Effective Date; (ii) the maximum number of Shares available for issuance under the Plan have been issued; or (iii) the Board terminates the Plan in accordance with Section 14(a). However, unless otherwise expressly provided in the Plan or in an applicable Award Agreement, any Award theretofore granted may extend beyond such date, and the authority of the Committee to amend, alter, adjust, suspend, discontinue or terminate any such Award, or to waive any conditions or rights under any such Award, and the authority of the Board to amend the Plan, shall extend beyond such date.
Section 18. Cancellation or Clawback of Awards.
(a) The Committee may specify in an Award Agreement that a Participants rights, payments and benefits with respect to an Award shall be subject to reduction, cancellation, forfeiture or recoupment upon the occurrence of certain specified events, in addition to any otherwise applicable vesting or performance conditions of an Award. Such events may include a Termination of Service with or without Cause (and, in the case of any Cause that is resulting from an indictment or other non-final determination, the Committee may provide for such Award to be held in escrow or abeyance until a final resolution of the matters related to such event occurs, at which time the Award shall either be reduced, cancelled or forfeited (as provided in such Award Agreement) or remain in effect, depending on the outcome), violation of material policies, breach of non-competition, non-solicitation, confidentiality or other restrictive covenants, or requirements to comply with minimum share ownership requirements, that may apply to the Participant, or other conduct by the Participant that is detrimental to the business or reputation of the Company and/or its Affiliates.
(b) The Committee shall have full authority to implement any policies and procedures necessary to comply with Section 10D of the Exchange Act and any rules promulgated thereunder and any other regulatory regimes. Notwithstanding anything to the contrary contained herein, any Awards granted under the Plan (including any amounts or benefits arising from such Awards) shall be subject to any clawback or recoupment arrangements or policies the Company has in place from time to time, and the Committee may, to the extent permitted by applicable law and stock exchange rules or by any applicable Company policy or arrangement, and shall, to the extent required, cancel or require reimbursement of any Awards granted to the
I-15
Table of Contents
Participant or any Shares issued or cash received upon vesting, exercise or settlement of any such Awards or sale of Shares underlying such Awards.
Section 19. Section 409A of the Code. With respect to Awards subject to Section 409A of the Code, the Plan is intended to comply with the requirements of Section 409A of the Code, and the provisions of the Plan and any Award Agreement shall be interpreted in a manner that satisfies the requirements of Section 409A of the Code, and the Plan shall be operated accordingly. If any provision of the Plan or any term or condition of any Award would otherwise frustrate or conflict with this intent, the provision, term or condition shall be interpreted and deemed amended so as to avoid this conflict. Notwithstanding anything in the Plan to the contrary, if the Board considers a Participant to be a specified employee under Section 409A of the Code at the time of such Participants separation from service (as defined in Section 409A of the Code), and any amount hereunder is deferred compensation subject to Section 409A of the Code, any distribution of such amount that otherwise would be made to such Participant with respect to an Award as a result of such separation from service shall not be made until the date that is six months after such separation from service, except to the extent that earlier distribution would not result in such Participants incurring interest or additional tax under Section 409A of the Code. If an Award includes a series of installment payments (within the meaning of Section 1.409A-2(b)(2)(iii) of the Treasury Regulations), a Participants right to such series of installment payments shall be treated as a right to a series of separate payments and not as a right to a single payment, and if an Award includes dividend equivalents (within the meaning of Section 1.409A-3(e) of the Treasury Regulations), a Participants right to such dividend equivalents shall be treated separately from the right to other amounts under the Award. Notwithstanding the foregoing, the tax treatment of the benefits provided under the Plan or any Award Agreement is not warranted or guaranteed, and in no event shall the Company be liable for all or any portion of any taxes, penalties, interest or other expenses that may be incurred by a Participant on account of non-compliance with Section 409A of the Code.
Section 20. Successors and Assigns. The terms of the Plan shall be binding upon and inure to the benefit of the Company and any successor entity, including any successor entity contemplated by Section 12(c).
Section 21. Data Protection. In connection with the Plan, the Company may need to process personal data provided by the Participant to the Company or its Affiliates, third party service providers or others acting on the Companys behalf. Examples of such personal data may include, without limitation, the Participants name, account information, social security number, tax number and contact information. The Company may process such personal data in its legitimate business interests for all purposes relating to the operation and performance of the Plan, including but not limited to:
(a) administering and maintaining Participant records;
(b) providing the services described in the Plan;
(c) providing information to future purchasers or merger partners of the Company or any Affiliate, or the business in which such Participant works; and
(d) responding to public authorities, court orders and legal investigations, as applicable.
The Company may share the Participants personal data with (i) Affiliates, (ii) trustees of any employee benefit trust, (iii) registrars, (iv) brokers, (v) third party administrators of the Plan, (vi) third party service providers acting on the Companys behalf to provide the services described above or (vii) regulators and others, as required by law.
If necessary, the Company may transfer the Participants personal data to any of the parties mentioned above in a country or territory that may not provide the same protection for the information as the Participants home country. Any transfer of the Participants personal data to recipients in a third country will be made subject to appropriate safeguards or applicable derogations provided for under applicable law. Further information on those safeguards or derogations can be obtained through the contact set forth in the Employee Privacy Notice (the Employee Privacy Notice) that previously has been provided by the
I-16
Table of Contents
Company or its applicable Affiliate to the Participant. The terms set forth in this Section 21 are supplementary to the terms set forth in the Employee Privacy Notice (which, among other things, further describes the rights of the Participant with respect to the Participants personal data); provided that, in the event of any conflict between the terms of this Section 21 and the terms of the Employee Privacy Notice, the terms of this Section 21 shall govern and control in relation to the Plan and any personal data of the Participant to the extent collected in connection therewith.
The Company will keep personal data collected in connection with the Plan for as long as necessary to operate the Plan or as necessary to comply with any legal or regulatory requirements.
A Participant has a right to (i) request access to and rectification or erasure of the personal data provided, (ii) request the restriction of the processing of his or her personal data, (iii) object to the processing of his or her personal data, (iv) receive the personal data provided to the Company and transmit such data to another party, and (v) to lodge a complaint with a supervisory authority.
Section 22. Governing Law. The Plan and each Award Agreement shall be governed by the laws of the State of Delaware, without application of the conflicts of law principles thereof.
I-17
Table of Contents
PART II INFORMATION NOT REQUIRED IN PROSPECTUS
Item 20. Indemnification of Officers and Directors
Section 145 of the DGCL provides that a corporation may indemnify directors and officers as well as other employees and individuals against expenses (including attorneys fees), judgments, fines and amounts paid in settlement actually and reasonably incurred by such person in connection with any threatened, pending or completed actions, suits or proceedings in which such person is made a party by reason of such person being or having been a director, officer, employee or agent of the Registrant. The DGCL provides that Section 145 is not exclusive of other rights to which those seeking indemnification may be entitled under any bylaws, agreement, vote of stockholders or disinterested directors or otherwise. The Registrants Certificate of Incorporation and Bylaws provide for indemnification by the Registrant of its directors and officers to the fullest extent permitted by the DGCL.
Section 102(b)(7) of the DGCL permits a corporation to provide in its Certificate of Incorporation that a director of the corporation shall not be personally liable to the corporation or its stockholders for monetary damages for breach of fiduciary duty as a director, except for liability (1) for any breach of the directors duty of loyalty to the corporation or its stockholders, (2) for acts or omissions not in good faith or which involve intentional misconduct or a knowing violation of law, (3) for unlawful payments of dividends or unlawful stock repurchases, redemptions or other distributions or (4) for any transaction from which the director derived an improper personal benefit. The Registrants Certificate of Incorporation provides for such limitation of liability to the fullest extent permitted by the DGCL.
The Registrant has entered into indemnification agreements with each of its directors and executive officers to provide contractual indemnification in addition to the indemnification provided in our Current Company Certificate. Each indemnification agreement provides for indemnification and advancements by the Registrant of certain expenses and costs relating to claims, suits or proceedings arising from his or her service to the Registrant or, at our request, service to other entities, as officers or directors to the maximum extent permitted by applicable law. We believe that these provisions and agreements are necessary to attract qualified directors.
The Registrant also maintains standard policies of insurance under which coverage is provided (1) to its directors and officers against loss arising from claims made by reason of breach of duty or other wrongful act, while acting in their capacity as directors and officers of the Registrant, and (2) to the Registrant with respect to payments which may be made by the Registrant to such officers and directors pursuant to any indemnification provision contained in the Registrants Certificate of Incorporation and Bylaws or otherwise as a matter of law.
Item 21. Exhibits and Financial Statement Schedules
| (a) | Exhibits. |
See the accompanying Exhibit Index for the list of Exhibits of this registration statement, which list of Exhibits is incorporated herein by reference.
Item 22. Undertakings
| (a) | The undersigned registrant hereby undertakes: |
| (1) | To file, during any period in which offers or sales are being made, a post-effective amendment to this registration statement: |
| (i) | To include any prospectus required by Section 10(a)(3) of the Securities Act; |
| (ii) | To reflect in the prospectus any facts or events arising after the effective date of the registration statement (or the most recent post-effective amendment thereof), which, individually or in the aggregate, represent a fundamental change in the information set forth in the registration |
II-1
Table of Contents
| statement. Notwithstanding the foregoing, any increase or decrease in volume of securities offered (if the total dollar value of securities offered would not exceed that which was registered) and any deviation from the low or high end of the estimated maximum offering range may be reflected in the form of prospectus filed with the SEC pursuant to Rule 424(b) if, in the aggregate, the changes in volume and price represent no more than a 20 percent change in the maximum aggregate offering price set forth in the Calculation of Registration Fee table in the effective registration statement; and |
| (iii) | To include any material information with respect to the plan of distribution not previously disclosed in the registration statement or any material change to such information in the registration statement. |
| (2) | That, for the purpose of determining any liability under the Securities Act, each such post-effective amendment shall be deemed to be a new registration statement relating to the securities offered therein, and the offering of such securities at that time shall be deemed to be the initial bona fide offering thereof. |
| (3) | To remove from registration by means of a post-effective amendment any of the securities being registered which remain unsold at the termination of the offering. |
| (4) | That, for the purpose of determining liability under the Securities Act to any purchasers, each prospectus filed pursuant to Rule 424(b) as part of a registration statement relating to an offering other than registration statements relying on Rule 430B or other than prospectuses filed in reliance on Rule 430A, shall be deemed to be part of and included in the registration statement as of the date it is first used after effectiveness. Provided, however, that no statement made in a registration statement or prospectus that is part of the registration statement or made in a document incorporated or deemed incorporated by reference into the registration statement or prospectus that is part of the registration statement will, as to a purchaser with a time of contract of sale prior to such first use, supersede or modify any statement that was made in the registration statement or prospectus that was part of the registration statement or made in any such document immediately prior to such date of first use. |
| (5) | That, for the purpose of determining liability of the registrant under the Securities Act to any purchaser in the initial distribution of the securities, the undersigned registrant undertakes that in a primary offering of securities of the undersigned registrant pursuant to this registration statement, regardless of the underwriting method used to sell the securities to the purchaser, if the securities are offered or sold to such purchaser by means of any of the following communications, the undersigned registrant will be a seller to the purchaser and will be considered to offer or sell such securities to such purchaser: |
| (i) | any preliminary prospectus or prospectus of the undersigned registrant relating to the offering required to be filed pursuant to Rule 424; |
| (ii) | any free writing prospectus relating to the offering prepared by or on behalf of the undersigned registrant or used or referred to by the undersigned registrant; |
| (iii) | the portion of any other free writing prospectus relating to the offering containing material information about the undersigned registrant or its securities provided by or on behalf of the undersigned registrant; and |
| (iv) | any other communication that is an offer in the offering made by the undersigned registrant to the purchaser. |
| (b) | The undersigned registrant hereby undertakes that, for purposes of determining any liability under the Securities Act, each filing of the registrants annual report pursuant to Section 13(a) or 15(d) of the Securities Exchange Act of 1934 (and, where applicable, each filing of an employee benefit plans annual report pursuant to Section 15(d) of the Securities Exchange Act of 1934) that is incorporated by reference in the registration statement shall be deemed to be a new registration statement relating to the securities offered therein, and the offering of such securities at that time shall be deemed to be the initial bona fide offering thereof. |
II-2
Table of Contents
| (c) | (1) | The undersigned registrant hereby undertakes as follows: that prior to any public reoffering of the securities registered hereunder through use of a prospectus which is a part of this registration statement, by any person or party who is deemed to be an underwriter within the meaning of Rule 145(c), the issuer undertakes that such reoffering prospectus will contain the information called for by the applicable registration form with respect to reofferings by persons who may be deemed underwriters, in addition to the information called for by the other items of this form. | ||
| (2) | The registrant undertakes that every prospectus (i) that is filed pursuant to paragraph (1) immediately preceding or (ii) that purports to meet the requirements of Section 10(a)(3) of the Securities Act and is used in connection with an offering of securities subject to Rule 415, will be filed as a part of an amendment to the registration statement and will not be used until such amendment is effective, and that, for purposes of determining any liability under the Securities Act, each such post-effective amendment shall be deemed to be a new registration statement relating to the securities offered therein, and the offering of such securities at that time shall be deemed to be the initial bona fide offering thereof. | |||
| (d) | Insofar as indemnification for liabilities arising under the Securities Act may be permitted to directors, officers and controlling persons of the registrant pursuant to the foregoing provisions, or otherwise, the registrant has been advised that in the opinion of the SEC such indemnification is against public policy as expressed in the Act and is, therefore, unenforceable. In the event that a claim for indemnification against such liabilities (other than the payment by the registrant of expenses incurred or paid by a director, officer or controlling person of the registrant in the successful defense of any action, suit or proceeding) is asserted by such director, officer or controlling person in connection with the securities being registered, the registrant will, unless in the opinion of its counsel the matter has been settled by controlling precedent, submit to a court of appropriate jurisdiction the question whether such indemnification by it is against public policy as expressed in the Act and will be governed by the final adjudication of such issue. |
| (e) | The undersigned registrant hereby undertakes to respond to requests for information that is incorporated by reference into the prospectus pursuant to Items 4, 10(b), 11, or 13 of Form S-4, within one business day of receipt of such request, and to send the incorporated documents by first class mail or other equally prompt means. This includes information contained in documents filed subsequent to the effective date of the registration statement through the date of responding to the request. |
| (f) | The undersigned registrant hereby undertakes to supply by means of a post-effective amendment all information concerning a transaction, and the company being acquired involved therein, that was not the subject of and included in the registration statement when it became effective. |
II-3
Table of Contents
EXHIBIT INDEX
II-4
Table of Contents
| * | Filed herewith |
| ** | Previously filed |
| | To be filed upon amendment |
II-5
Table of Contents
SIGNATURES AND POWER OF ATTORNEY
Pursuant to the requirements of the Securities Act of 1933, as amended, the registrant has duly caused this Registration Statement to be signed on its behalf by the undersigned, thereunto duly authorized, in the City of New York, State of New York, on August 11, 2021.
| GS ACQUISITION HOLDINGS CORP II |
||||
| By: | /s/ Tom Knott |
|||
| Name: | Tom Knott | |||
| Title: | Chief Executive Officer, Chief Financial | |||
| Officer and Secretary | ||||
Pursuant to the requirements of the Securities Act of 1933, as amended, this Registration Statement has been signed by the following person in the capacities and on the dates indicated:
| Signature |
Title |
Date |
||
| /s/ Tom Knott |
Chief Executive Officer, Chief Financial Officer and Secretary | August 11, 2021 | ||
| Tom Knott | (Principal Executive, Financial and Accounting Officer) | |||
| * |
Chairman of the Board of Directors | August 11, 2021 | ||
| Raanan A. Agus | ||||
| * |
Director | August 11, 2021 | ||
| William Frist | ||||
| * |
Director | August 11, 2021 | ||
| Steven S. Reinemund | ||||
| * |
Director | August 11, 2021 | ||
| David Robinson | ||||
| * |
Director | August 11, 2021 | ||
| Martha Sullivan | ||||
| *By: | /s/ Tom Knott |
|
| Tom Knott | ||
| Attorney-in-Fact |
II-6